Abstract
Background
The consumption of dairy products may influence the risk of type 2 diabetes mellitus (T2DM), but inconsistent findings have been reported. Moreover, large variation in the types of dairy intake has not yet been fully explored.
Methods and Results
We conducted a systematic review and meta-analysis to clarify the dose–response association of dairy products intake and T2DM risk. We searched PubMed, EMBASE and Scopus for studies of dairy products intake and T2DM risk published up to the end of October 2012. Random-effects models were used to estimate summary relative risk (RR) statistics. Dose-response relations were evaluated using data from different dairy products in each study. We included 14 articles of cohort studies that reported RR estimates and 95% confidence intervals (95% CIs) of T2DM with dairy products intake. We found an inverse linear association of consumption of total dairy products (13 studies), low-fat dairy products (8 studies), cheese (7 studies) and yogurt (7 studies) and risk of T2DM. The pooled RRs were 0.94 (95% CI 0.91–0.97) and 0.88 (0.84–0.93) for 200 g/day total and low-fat dairy consumption, respectively. The pooled RRs were 0.80 (0.69–0.93) and 0.91 (0.82–1.00) for 30 g/d cheese and 50 g/d yogurt consumption, respectively. We also found a nonlinear association of total and low-fat dairy intake and T2DM risk, and the inverse association appeared to be strongest within 200 g/d intake.
Conclusion
A modest increase in daily intake of dairy products such as low fat dairy, cheese and yogurt may contribute to the prevention of T2DM, which needs confirmation in randomized controlled trials.
Introduction
The prevalence of type 2 diabetes mellitus (T2DM) is a growing public-health burden worldwide, particularly in developing countries. The prevalence of T2DM is estimated to reach 552 million worldwide by 2030 [1]. T2DM may cause substantial morbidity and mortality and is associated with enormous economic, health, and societal costs [2], [3]. Moreover, as compared with unaffected people, those with T2DM are at increased risk of other chronic illnesses, including cardiovascular disease; T2DM more than doubles the risk of a heart attack or stroke [4], [5]. Therefore, the identification of modifiable risk factors for primary prevention of T2DM is of considerable public health importance.
T2DM has genetic components but is also directly influenced by modifiable lifestyle factors, including dietary behaviors [6]. Dairy consumption might affect T2DM. Experimental studies indicated that dairy protein, such as whey protein, has insulinotropic and glucose-lowering properties [7].The Multi-Ethnic Study Atherosclerosis [8] and Cardiovascular Health study [9] suggested that fatty acids in dairy might be responsible for lower risk of T2DM. Epidemiological studies of dairy products and T2DM risk have given mixed results [10], [11], [12], [13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23]. Some cohort studies have reported inverse associations of intake of total and low-fat dairy products, milk and/or yogurt and T2DM risk, but other studies found no association [10], [11], [19], [20], [23]. One meta-analysis of 7 studies reported a significant inverse association of dairy intake and risk of T2DM [24]. However, the large variation in types of dairy consumed has not been fully explored. Furthermore, the dose–response relationship needs to be clarified as well as any gender or geographic differences in the T2DM risk. In addition, possible confounding by other lifestyle factors needs to be explored to firmly establish the potential preventive role of dairy products in T2DM.
We conducted a meta-analysis of population-based cohort studies to investigate dose–response associations of consumption of total, low-fat, and full-fat dairy products as well as different types of dairy products and risk of T2DM.
Methods
Data Sources and Search Strategy
We followed standard criteria for conducting and reporting meta-analyses of observational studies (MOOSE). Two authors (DG and NN) independently did a literature search MEDLINE via PubMed (published from 1966 to March 2013), EMBASE (published from 1980 to March 2013), and Scopus (www.scopus.com) with no restriction on language. To identify studies of milk or dairy product intake and T2DM risk, we used both the medical subject heading (MeSH) terms (“Diabetes Mellitus” AND (milk OR dairy)) and searched the text using the terms (‘diabetes’/exp OR diabetes') AND (‘dairy’/exp OR dairy OR ‘milk’/exp OR milk). We also searched the reference lists of all studies retrieved and published systematic reviews and meta-analysis.
Study Selection
All abstracts retrieved were examined independently by 2 investigators (DG and NN) who then retrieved the full text of potential articles. Disagreements were resolved by consensus, and if necessary, with a third author (CW). We included prospective cohort studies and case-cohort studies assessing the association of consumption of total dairy products or specific types of dairy products and T2DM. To be included in the analyses, articles needed to contain estimates of the relative risk (RR) (such as odds ratios [ORs], hazard ratios [HRs] or risk ratios) with 95% confidence intervals (95% CIs). We excluded animal studies, clinical trials, cross sectional studies, case-control studies, and studies that examined other associations. For the dose–response analysis, a quantitative measure of intake had to be provided. If the article lacked data, we attempted to contact the author.
Data Extraction and Quality Assessment
We extracted the following data from each study: country where the study was conducted, follow-up period, sample size, gender, age, number of cases, dietary assessment method (type, number of food items and whether the food intake had been validated), type of dairy product (e.g., total dairy, milk, cheese), quantity of intake, HRs, RR values, and ORs and 95% CIs for dairy product intake and, when available, the number of cases and participants or person-years for each category of dairy product consumption. Two authors (YL and ZM) independently performed the data extraction. Any disagreements were resolved by discussion.
Two independent reviewers (DG and NN) evaluated the quality of the selected studies by using a modified scoring system that was based on a recently used system (designed with reference to QUATSO [25], MOOSE [26], and STROBE [27]) that allowed for a total score of 0 to 6 points (6 indicating highest quality) [28]. The system allocates one point each for 1) any justification given for the cohort; 2) appropriate inclusion and exclusion criteria used; 3) outcome (diagnosis of T2DM not solely based on self-reporting); 4) intervention (participants' usual dairy consumption assessed with a validated tool); 5) statistical analysis (adjustments made for age, sex, body mass index, and family history of T2DM, total energy intake and physical activity, these being proven risk factors for type 2 diabetes); and 6) any other adjustments performed (such as glycemic load and dietary factors).
Statistical analysis
HRs and RRs were assumed to be approximately the same measure of relative risk. For articles reporting ORs, we estimated the RRs from the ORs using a previously published correction method [29]. To take into account heterogeneity between studies, we used a random-effects models to calculate summary RRs and 95% CIs for the highest versus lowest level of dairy product intake and for the dose–response analysis. The natural logarithm of the RR from each study was weighted by the inverse of its variance and pooled across studies. A two-tailed P<0.05 was considered statistically significant. Articles that reported findings for men and women separately were considered 2 studies when the observed items were combined.
For the dose–response analysis, we used GLST command in Stata software as the method proposed by Greenland and Longnecker [30] and Orsini et al. [31] to compute study-specific slopes (linear trends) and 95% CIs from the natural logs of the RRs and 95% CIs across categories of dairy product intake.
For each study, the median or mean level of dairy product intake for each category was assigned to each corresponding RR. When the median or mean intake per category was not provided, we assigned the midpoint of upper and lower boundaries in each category as the average intake. If the highest or the lowest category was open-ended, we assumed that the open-ended interval length had the same length as the adjacent interval. If the intake was reported in densities (i.e., per 1000 kcal), we recalculated the reported intake as absolute intake using the mean or median energy intake reported in the article [14]. When studies reported the intake in servings and times per day or week, we converted the intake to grams of intake per day using standard units of 244 g (or 244 ml) for milk, 43 g for cheese (2 slices) and 177 g for total dairy products from the serving sizes reported in the US Department of Agriculture Food and Nutrient Database for Dietary Studies [32]. Pooled estimates were expressed in rounded numbers that approximated a normal portion size and fitted within the range of dairy intake of all studies (i.e., 200 g for milk and total, low-fat, and full-fat dairy; 50 g for yogurt; and 30 g for cheese).
To examine a potential nonlinear association between dairy products intake and T2DM risk, we performed a 2-stage, random-effects, dose-response meta-analysis, as recently summarized [33]. In the first stage, we constructed study-specific restricted cubic spline models, with 4 knots at fixed percentiles (5%, 35%, 65%, 95%) of the exposure distribution by using generalized least-squares regression. In the second stage, we combined the 2 regression coefficients and the variance/covariance matrix that had been estimated within each study, using the restricted maximum likelihood method in a multivariate random-effects meta-analysis. The pooled relative risks for specific exposure values were then estimated. A P value for nonlinearity was calculated by testing the null hypothesis that the coefficient of the second spline was equal to zero.
Heterogeneity among studies was assessed by I2, the amount of total variation explained by the between-study variation, and the Q test. We conducted subgroup and random effects univariate and multivariate meta-regression to investigate potential sources of heterogeneity we performed for the primary outcomes. Publication bias was assessed with funnel plots, Begg's test and Egger's test. Stata v12.0 (Stata Corp, College Station, TX) was used for all the statistical analysis.
Results
Study characteristics
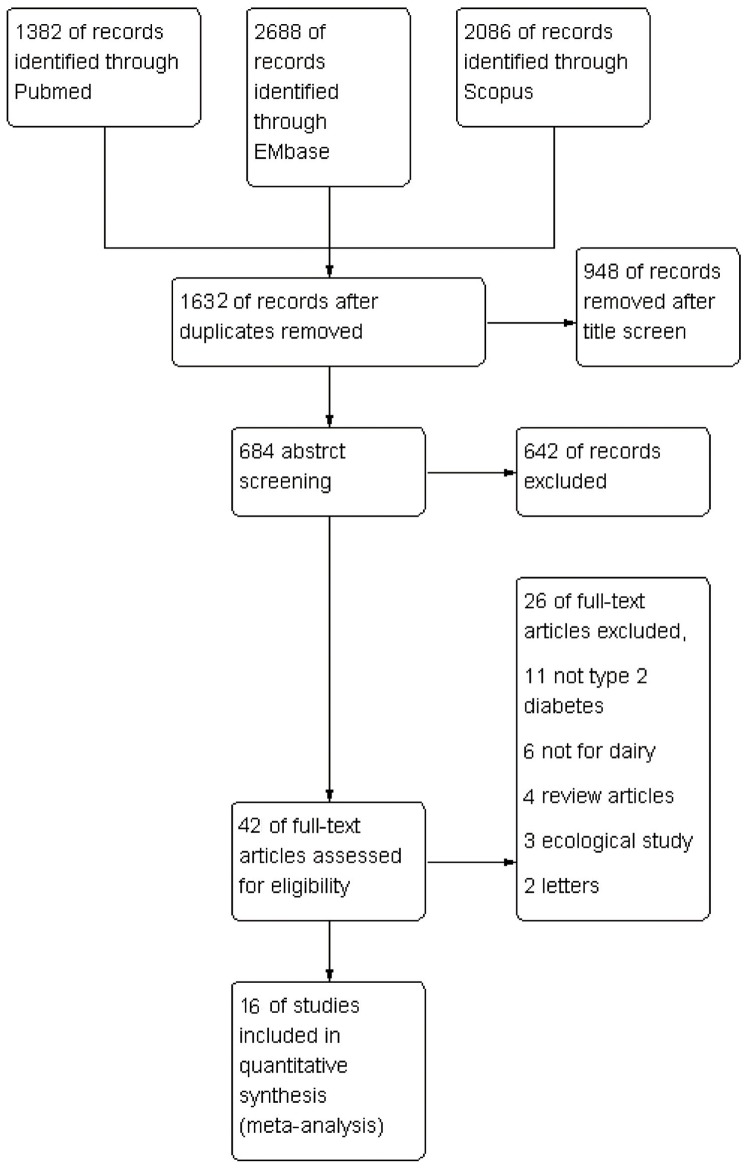
We included 15 prospective cohort studies and 1 case–cohort study in our analysis (Figure 1). 6 of the studies [12], [13], [14], [15], [16], [18] were performed in the United States, 6 in Europe [11], [19], [21], [23], [34], [35], 2 in Asia [17], [22] and 2 in Australia [10], [20]. The articles were published between 2005 and 2013 and included 526,998 subjects (including 29,789 T2DM cases). Characteristics of included studies are in Table 1. Figures 2 show assessments by risk of bias. The studies were generally of moderate quality. More than 75% of the studies met 4 of the quality items as reported and 8 studies met all requirements. Interrater reliability for assessing quality items was good (κ = 0.86, P<0.01).
Figure 1. Flow chart for the selection of studies for meta-analysis of the association of dairy products intake and type 2 diabetes (T2DM).
Table 1. Characteristics of the cohort studies of dairy products intake and type 2 diabetes mellitus (T2DM).
| Author,y | populati | Country | Men | Age, y | Follow | Subjects | Dietary | Dairy quantity | Relative risk | Assessment | Adjustment |
| on | (%) | -up,y | (cases) | Assessment | (high vs. low intake) | of T2DM | |||||
| Sluijs,2012 | (EPIC- | 8 countries | 50% | 52 | 16 | 24,475 | FFQ | Total dairy (628.9 g vs.79.7 g) | 0.97 (0.82,1.15) | Self reporting, primary | Center, age, sex, BMI, educational |
| InterAct) | in Europe | (10,694) | 24-h dietary | Milk (486.1 g vs. 0.3 g) | 1.08 (0.90,1.31) | care registers, | level, smoking, physical activity, | ||||
| recall data | Yogurt (190.4 g vs. 8.4 g) | 0.89 (0.77,1.03) | secondary care | alcohol intake, fruit plus vegetables, | |||||||
| Cheese (73.7 g vs. 3.2 g) | 0.83 (0.7,0.98) | registers, medication | red meat, processed meat, sugar | ||||||||
| use(drug registers), | |||||||||||
| hospital admissions, | |||||||||||
| Fermented dairy (220.7 g vs. 40.4 g) | 0.85 (0.73,0.99) | and mortality data | sweetened soft drinks, coffee, cereals, | ||||||||
| Grantham, | AusDiab | Australia | 45% | 52 | 5 | 5,582 | 121-item FFQ | Total dairy (408 g vs. 346 g) | 0.71 (0.48,1.05) | 75 g OGTT | Age, sex, energy intake, family |
| 2012 | (209) | Low-fat milk (375 g vs. 200 g) | 0.65 (0.44,0.94) | history of diabetes, education level, | |||||||
| Full-fat milk (375 g vs. 200 g) | 1.18 (0.78,1.79) | level of physical activity, smoking, | |||||||||
| Yogurt (73 g vs. 3 g) | 1.14 (0.78,1.67) | TAG, HDL cholesterol, systolic blood | |||||||||
| Cheese (20 g vs. 6 g) | 0.78 (0.48,1.15) | pressure, waist circumference and hip | |||||||||
| Louie, 2012 | BMES | Australia | 42% | 63.5 | 10 | 1,824 | 145-item FFQ | Total dairy (3.1 vs. 0.5) | 1.50 (0.47,4.77) | Self-reporting, taking | Age, sex, smoking, physical activity, |
| (145) | Low-fat dairy (2.1 vs. 0) | 1.09 (0.57,2.09) | medication for T2DM, | dietary glycemic load, fibre, total | |||||||
| Full-fat dairy (1.9 vs. 0.1) | 0.87(0.48,1.58) | fasting blood glucose | energy intake and family history of | ||||||||
| >7.0 mmol/L | type 2 diabetes, calcium. | ||||||||||
| Struijk, | Inter99 | Denmark | 47.5% | 30–60 | 5 | 5,232 | FFQ | Total dairy (578 g vs. 47 g) | 0.96 (0.58,1.58) | 75 g OGTT | Age, gender and intervention group, |
| 2012 | (214) | Low-fat dairy (536 g vs. 57 g) | 0.85 (0.52,1.40) | diabetes family history, education | |||||||
| Full-fat dairy (89 g vs. 4 g) | 0.94 (0.56,1.58) | level, physical activity smoking | |||||||||
| Milk (546 g vs. 16 g) | 0.95 (0.58,1.57) | status, alcohol intake, wholegrain | |||||||||
| Cheese (49 g vs. 4 g) | 0.78 (0.47,1.29) | cereal, meat, fish, coffee, tea, fruit, | |||||||||
| Fermented dairy (260 g vs. 13 g) | 0.86 (0.50,1.47) | vegetables, energy intake, change in | |||||||||
| diet waist circumference | |||||||||||
| Soedamah | Whitehall II | England | 72% | 56 | 9.8 | 4,186 | 114-item FFQ | Total dairy (575 g vs. 246 g) | 1.30 (0.95,1.77) | Self-reporting, and | Age, ethnicity and employment grade, |
| Muthu, 2012 | (273) | Low-fat dairy (458 g vs. 28 g) | 0.98 (0.73,1.31) | 75 g OGTT | smoking, alcohol intake, BMI, | ||||||
| Full-fat dairy (182 g vs. 27 g) | 1.23 (0.91,1.67) | physical activity and family history of | |||||||||
| Yogurt (117 g vs. 0 g) | 1.04 (0.87,1.58) | CHD/hypertension, fruit and | |||||||||
| Milk (441 g vs. 147 g) | 0.97 (0.71,1.31) | vegetables, bread, meat, fish, coffee, | |||||||||
| Fermented dairy (105 g vs. 17 g) | 1.17 (0.87,1.58) | tea and total energy intake. | |||||||||
| Cheese (31 g vs. 6 g) | 1.20 (0.88,1.64) | ||||||||||
| Margolis, | WHI-OS | USA | 0 | 50–79 | 8 | 82,076 | 122-item FFQ | Total dairy (3.4 vs.0.5) | 0.93 (0.83,1.04) | Self-reporting | Age, race/ethnicity, total energy |
| 2011 | (3946) | Low-fat dairy (2.8 vs.0.05) | 0.65 (0.44,0.96) | confirmed by review | intake, income, education, smoking, | ||||||
| Full-fat dairy (1.3 vs.0.06) | 0.80 (0.65,0.99) | of medical records | alcohol intake, family history of | ||||||||
| Yogurt (≥2/wk vs.<1/mo) | 0.46 (0.31,0.68) | diabetes, postmenopausal hormone | |||||||||
| therapy, blood pressure, BMI, | |||||||||||
| physical activity, dietary glycemic | |||||||||||
| load, dietary total fat, dietary total | |||||||||||
| fiber, magnesium | |||||||||||
| Malik, 2011 | NHS II cohort | USA | 0 | 34–53 | 8 | 37,038 | 133-item FFQ | Total dairy (2.14 vs. 0.62) | 0.75 (0.55,1.02) | self-reporting | Age, BMI, total energy intake, family |
| (550) | Low-fat dairy (1.44 vs.0.18) | 0.74 (0.54,1.01) | confirmed by | history of diabetes, smoking, physical | |||||||
| Full-fat dairy (1.14 vs. 0.19) | 0.72 (0.53,0.99) | review of medical | activity, alcohol, oral contraceptive | ||||||||
| records | use, hormone replacement therapy. | ||||||||||
| Polyunsaturated, saturated fat, glycemic | |||||||||||
| load, fiber, trans fat, processed meat, | |||||||||||
| carbonated soft drinks, fruit drinks,coffee | |||||||||||
| Kirii, 2009 | JPHC cohort | Japan | 57% | 40–69 | 5 | 59,796 | FFQ | Total dairy (≥300 g vs. <50 g) | Males 1.18 (0.90,1.56) | Self-reporting, | Age, area, BMI, family history of |
| (1,114) | Females 0.71(0.51,0.98) | Validity verified by | diabetes mellitus, smoking, alcohol | ||||||||
| Milk (≥200 g vs. <50 g) | Males 1.02(0.85,1.24) | medical record data | intake hypertension, exercise, coffee, | ||||||||
| Females 0.87(0.70,1.09) | and plasma glucose | magnesium, total energy | |||||||||
| Cheese (≥5 g vs. 0 g) | Males 0.88 (0.64,1.21) | random samples. | |||||||||
| Females 1.12(0.80,1.57) | |||||||||||
| Yogurt (≥60 g vs. 0 g) | Males 1.01 (0.75,1.36) | ||||||||||
| Females 0.77(0.58,1.01) | |||||||||||
| Villegas, | SWHS cohort | China | 0 | 51 | 6.9 | 64,191 | FFQ | Milk (250 vs. 0) | 0.60 (0.41,0.88) | Self-reporting, | Age, energy intake, BMI, waist-hip |
| 2009 | (2,270) | fasting glucose and | ratio, smoking status, alcohol | ||||||||
| OGTT | consumption, physical activity, | ||||||||||
| income level, education level, | |||||||||||
| occupation, and hypertension. | |||||||||||
| Elwood | Caerphilly | UK | 100 | 45–59 | 20 | 640 | FFQ and 7- | Milk | 0.57 (0.20,1.63) | Self-reporting | Age, smoking, BMI and social class |
| 2007 | prospective | (41) | day weighed | ||||||||
| study | intake | ||||||||||
| Liu, 2006 | WHS cohort | USA | 0 | 55 | 10 | 37,183 | 131-item FFQ | Total dairy (>2.9 vs. <0.85) | 0.68 (0.52,0.89) | Diagnostic criteria | Age, total energy intake, |
| (1063) | Low-fat dairy (>2.0 vs. ≤0.27) | 0.69 (0.522,0.91) | of ADA, based on | randomized-treatment assignment, | |||||||
| Full-fat dairy (>1.33 vs. <0.2) | 0.99 (0.82,1.20) | self-reporting, 3 | family history of diabetes, smoking, | ||||||||
| Yogurt (≥2/wk vs.<1/mo) | 0.82 (0.70,0.97) | complementary | BMI, hypercholesterolemia, | ||||||||
| Whole milk (≥2/wk vs.<1/mo) | 1.04 (0.84,1.30) | approaches to | hypertension, physical activity | ||||||||
| Skim milk (≥2/wk vs.<1/mo) | 0.92 (0.78,1.09) | validate the cases | hormones, alcohol consumption, fiber, | ||||||||
| Cottage cheese (≥2/wk vs<1/mo) | 0.86 (0.71,1.05) | total fat, and dietary glycemic load, | |||||||||
| Ice cream (≥2/wk vs.<1/mo) | 0.88 (0.74,1.05) | calcium, vitamin D, and magnesium. | |||||||||
| Other cheese (≥2/wk vs.<1/mo) | 0.80 (0.64,1.01) | ||||||||||
| Van Dam, | Black Women’s | USA | 0 | 21–69 | 8 | 41,186 | FFQ | Total dairy (2.53 vs. 0.07) | 0.93 (0.75,1.15) | Self-reporting, | Age, total energy intake, BMI, |
| 2006 | Health Study | (1,964) | Low-fat dairy (1.22 vs. 0) | 0.87 (0.76,1.00) | validity verification | smoking physical activity, alcohol, | |||||
| Full-fat dairy (1.33 vs. 0.07) | 1.03 (0.88,1.20) | of a random sample | family history of diabetes, education | ||||||||
| level, coffee, sugar-sweetened soft | |||||||||||
| drink, processed meat, red meat, | |||||||||||
| . | calcium or magnesium intake | ||||||||||
| Pittas, 2006 | NHS cohort | US | 0 | 30–55 | 20 | 83,779 | FFQ | Total dairy (3.9 vs. 0.9) | 0.79 (0.70,0.90) | Criteria by | Age, BMI, hypertension, family |
| (4,843) | National Diabetes | history of diabetes, smoking, physical | |||||||||
| Data Group and | activity, caffeine, alcohol, and state of | ||||||||||
| ADA.self-reporting | residence, fat (saturated, | ||||||||||
| polyunsaturated, or trans), cereal fiber, | |||||||||||
| glycemic load, magnesium, and retinol | |||||||||||
| Choi, 2005 | HPFS cohort | USA | 100 | 43–75 | 12 | 41,254 | FFQ | Total dairy (≥2.9 vs. <0.9) | 0.75 (0.61,0.93) | Criteria by | Age, total energy intake, family history |
| (1243) | Low-fat dairy (>1.58 vs. <0.14) | 0.74 (0.60,0.91) | National Diabetes | of diabetes, smoking, BMI, | |||||||
| Full-fat dairy (>1.72 vs.<0.38) | 0.82 (0.66,1.02) | Data Group. Based | hypercholesterolemia, hypertension, | ||||||||
| Yogurt (≥2/wk vs. <1/mo) | 0.83 (0.66,1.06) | on self-reporting. | physical activity, alcohol, fiber, | ||||||||
| Whole milk (≥2/wk vs. <1/mo) | 1.19 (1.00,1.43) | Validity verified | trans-fat polyunsaturated to saturated | ||||||||
| Low-Fat milk (≥2/wk vs. <1/mo) | 0.78 (0.63,0.97) | with medical | fat, glycemic load | ||||||||
| Cottage cheese (≥2/wk vs. <1/mo) | 0.96 (0.80,1.17) | records in a sample | |||||||||
| Other cheese (≥2/wk vs. <1/mo) | 0.88 (0.67,1.16) | of 71 participants. | |||||||||
| Ice cream (≥2/wk vs. <1/mo) | 0.78 (0.64,0.95) | ||||||||||
| Montonen, | Finnish Mobile | Finland | 50 | 40–69 | 23 | 4304 | dietary history | Regular dairy (>305 vs. <39) | 0.81(0.62–1.08) | from the Social | Adjusted for age, sex, body mass |
| 2005 | Clinic Health | (383) | interview | Low fat dairy (>0 vs. 0) | 0.90(0.60,1.36) | Insurance | index, energy intake, smoking, family | ||||
| Examination | Whole milk (>878 vs. <326) | 1.06(0.75,1.50) | Institution’s | history of diabetes, and geographic | |||||||
| Survey | nationwide register | area | |||||||||
| of persons | |||||||||||
| receiving drug | |||||||||||
| reimbursement | |||||||||||
| Ericson, | Malmö Diet and | Sweden | 57 | 8 | 23 531 | 148-FFQ | Total dairy women (6.0 vs. 1.8) | 0·88 (0·70, 1·09) | Self report, and | sex, smoking status, alcohol | |
| 2013 | Cancer cohort | (837) | Total dairy men (6.3 vs. 1.8) | 1·20 (0·98, 1·47) | verified with an | consumption, leisure-time physical | |||||
| inquiry to the | activity, BMI, waist-to-hip ratio, | ||||||||||
| treating physician, | hypertension, history of high blood lipid | ||||||||||
| local cancer | levels at baseline, education, vitamin | ||||||||||
| registries | supplementation, non-consumption of the | ||||||||||
| respective food group, total energy intake | |||||||||||
| (kJ/day). |
FFQ, food-frequency questionnaire; OGGT, oral glucose tolerance test; BMI, body mass index TAG,triglyceride; HDL, High density lipoproteins; ADA, American Diabetes Association CHD, coronary heart disease; EPIC-InterAct European Prospective Investigation into Cancer and Nutrition cohort; AusDiab, Australian Diabetes Obesity and Lifestyle Study; BMES, Blue Mountains Eye Study; WHI-OS, Women’s Health Initiative observational study; NHS, The Nurses’ Health Study; JPHC, Japan Public Health Center-based Prospective Study; SWHS, Shanghai Women’s Health Study; WHS, Women’s Health Study; HPFS, Health Professionals Follow-up Study.
Figure 2. Methodological quality across included studies.
Total Dairy Products Intake and T2DM Risk
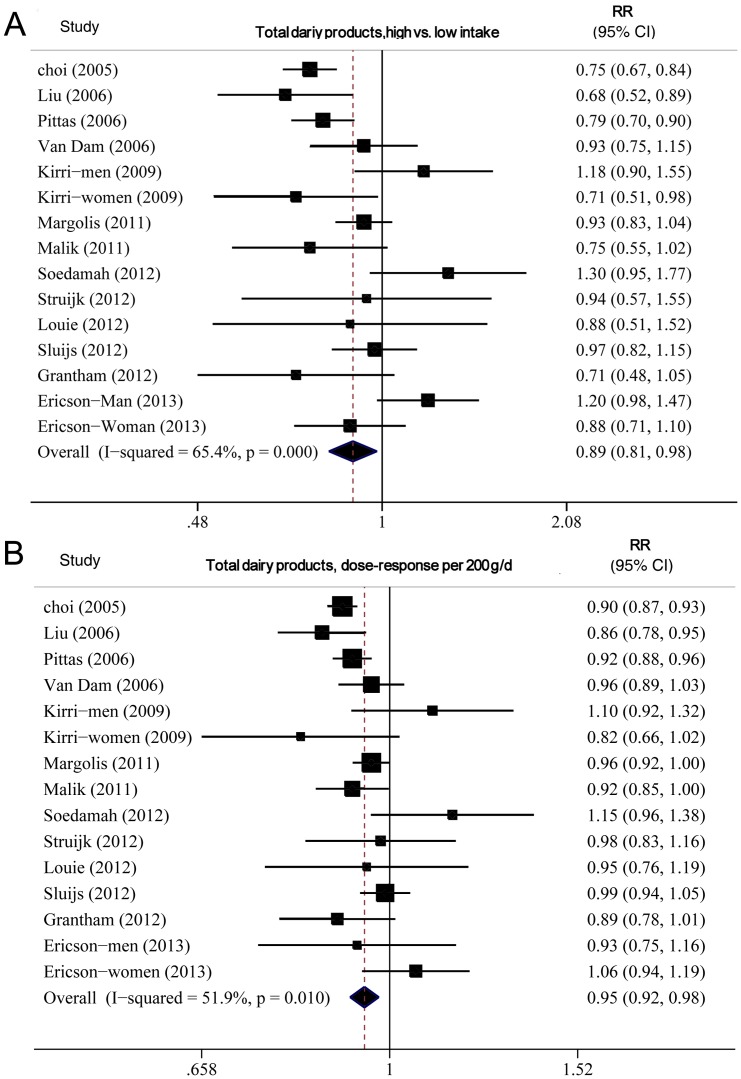
In all, 13 studies [10], [11], [12], [13], [14], [15], [16], [17], [18], [19], [20], [23], [34] including 457,893 subjects (27,095 cases) were analyzed.
In all, 13 studies(8–18; 21) including 457,893 subjects (27,095 cases) were analyzed.
High versus low intake
The summary RR for all studies was 0.89 (95% CI 0.81–0.98), with moderate heterogeneity, I2 = 65.4% and Pheterogeneity = 0.000 (Figure 3A).
Figure 3. Forest plot of relative risk (RR) for total dairy products intake and T2DM.
A, highest versus lowest intake. B, dose–response analysis (200 g/d). Weights are from random effects analysis.
Dose–response analysis
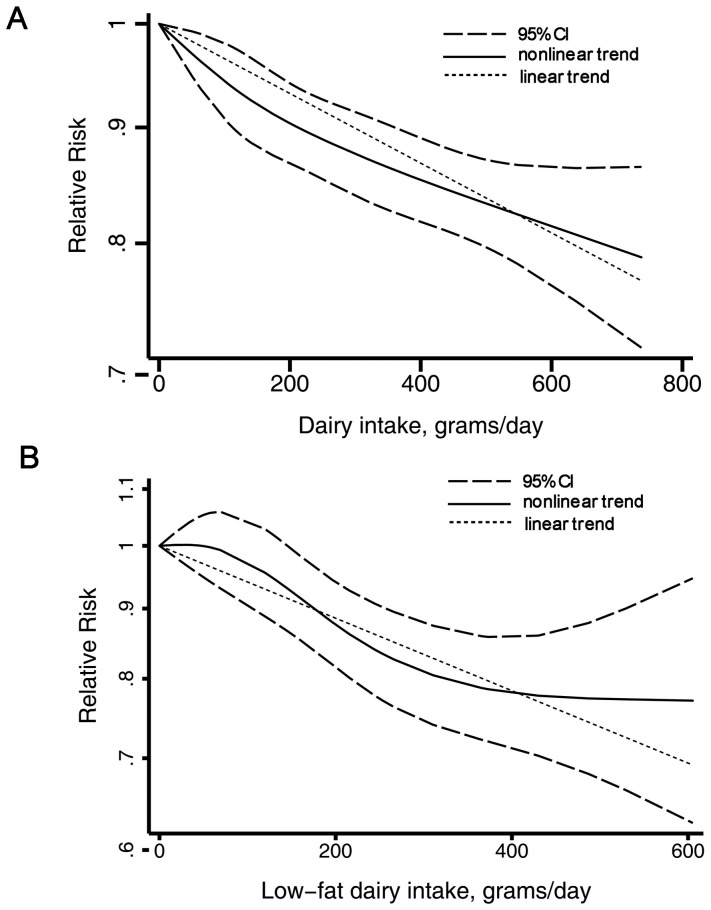
The summary RR for an increase of 200 g/day was 0.94 (0.91–0.97), with moderate heterogeneity, I2 = 51.6%, Pheterogeneity = 0.02 (Figure 3B). On subgroup analysis (Table 2), we found an inverse association of total dairy intake and T2DM risk in all strata except European studies and studies not adjusting for family history of T2DM, although in some analyses the associations were not statistically significant. None of the results differed significantly by sex (P = 0.21 for all comparisons). On univariate meta-regression analysis, geographic location, adjustment for family T2DM history, and glycemic load were significant predictors of the heterogeneity (p = 0.05, p = 0.04 and p = 0.04, respectively). But on multivariate meta-regression, we failed to identify the source of heterogeneity. We found no evidence of publication bias by Egger's test (P = 0.37), Begg's test (P = 0.58) or funnel plot(see Appendix Figure 1). We also found a nonlinear association of total dairy product intake and T2DM risk, Pfor nonlinearity <0.001, with most of the risk reduction occurring with intake up to about 200 g/d; higher intake were associated with a further but more modest decrease in risk (Figure 4A).
Table 2. Subgroup analyses of total and low-faty dairy products intake and T2DM, dose–response analysis.
| Total dairy | Low-fat dairy | |||||||||||||
| n | RR (95% CI) | I2 (%) | Pa | Pb | Pc | n | RR (95% CI) | I2 (%) | Pa | Pb | Pc | |||
| All studies | 12 | 0.94 (0.91,0.97) | 51.6 | 0.02 | 8 | 0.88 (0.84,0.93) | 16.3 | 0.30 | ||||||
| Duration | ||||||||||||||
| <10 | 6 | 0.95 (0.92,0.98) | 4.3 | 0.39 | 4 | 0.88 (0.82,0.95) | 32.5 | 0.21 | ||||||
| ≥10 | 6 | 0.94 (0.89,0.99) | 68.6 | 0.01 | 0.65 | 4 | 0.89 (0.82,0.96) | 23.5 | 0.27 | 0.93 | ||||
| Sex | ||||||||||||||
| Female | 6 | 0.93 (0.90,0.96) | 26.9 | 0.23 | 4 | 0.86 (0.80,0.92) | 29.6 | 0.23 | ||||||
| Male | 2 | 0.97 (0.80,1.18) | 78.2 | 0.03 | 1 | 0.85 (0.76,0.96) | ||||||||
| Both | 5 | 0.98 (0.92,1.05) | 24.5 | 0.26 | 0.21 | 3 | 0.94 (0.71,1.23) | 0 | 0.94 | 0.16 | ||||
| Geographic location | ||||||||||||||
| United States | 6 | 0.92 (0.90,0.95) | 42.7 | 0.12 | 5 | 0.86 (0.82,0.91) | 8 | 0.36 | ||||||
| Europe | 3 | 1.01 (0.94,1.08) | 18.6 | 0.29 | 2 | 0.97 (0.87,1.06) | 0 | 0.94 | 0.10 | 0.57 | ||||
| Asia | 1 | 0.96 (0.72,1.28) | 75.5 | 0.04 | 1 | 0.94 (0.71,1.24) | ||||||||
| Australia | 2 | 0.90 (0.81,1.01) | 0 | 0.62 | 0.04 | 0.08 | ||||||||
| No. of cases | ||||||||||||||
| <500 | 4 | 0.98 (0.88,1.10) | 41.5 | 0.16 | 3 | 0.97 (0.88,1.06) | 0 | 0.94 | ||||||
| 500–1500 | 4 | 0.91 (0.86,0.95) | 38.5 | 0.17 | 3 | 0.88 (0.82,0.93) | 0.9 | 0.37 | ||||||
| ≥1500 | 4 | 0.95 (0.92,0.98) | 32.7 | 0.22 | 0.43 | 3 | 0.83 (0.74,0.94) | 40 | 0.20 | 0.10 | ||||
| Study type | ||||||||||||||
| Prospective | 11 | 0.93 (0.90,0.96) | 44.7 | 0.05 | 8 | 0.88 (0.84,0.93) | 16.3 | 0.30 | ||||||
| Case cohort | 1 | 0.99 (0.94,1.05) | 0.22 | |||||||||||
| Adjustment method | ||||||||||||||
| COX | 8 | 0.94 (0.91,0.97) | 63.9 | 0.01 | 0.88 (0.82,0.93) | 32.6 | 0.19 | |||||||
| Logistic | 4 | 0.94 (0.86,1.04) | 24.8 | 0.26 | 0.92 | 0.95 (0.82,1.10) | 0 | 0.95 | 0.39 | |||||
| Adjustment factors | ||||||||||||||
| BMI | Yes | 9 | 0.94 (0.91,0.97) | 53 | 0.02 | 6 | 0.87 (0.83,0.92) | 8 | 0.37 | |||||
| No | 3 | 0.98 (0.84,1.16) | 60.9 | 0.02 | 0.57 | 2 | 0.97 (0.86,1.10) | 2 | 0.79 | 0.14 | 0.45 | |||
| Diabetes history | Yes | 1 | 0.93 (0.90,0.95) | 30.2 | 0.16 | 1 | 0.87 (0.83,0.92) | 0 | 0.45 | |||||
| No | 2 | 1.04 (0.91,1.19) | 58.2 | 0.12 | 0.048 | 0.24 | 0 | 0.98 (0.86,1.12) | 0.16 | |||||
| Glycemic load | Yes | 6 | 0.92 (0.89,0.95) | 33.7 | 0.18 | 5 | 0.86 (0.81,0.92) | 13.3 | 0.33 | |||||
| No | 6 | 0.98 (0.92,1.03) | 37.7 | 0.14 | 0.067 | 0.41 | 3 | 0.93 (0.86,1.00) | 0 | 0.47 | 0.20 | |||
| Fat | Yes | 5 | 0.92 (0.89,0.95) | 46.4 | 0.11 | 4 | 0.85 (0.80,0.92) | 28.9 | 0.24 | |||||
| No | 7 | 0.98 (0.93,1.03) | 27.7 | 0.21 | 0.067 | 0.56 | 4 | 0.93 (0.86,1.00) | 0 | 0.68 | 0.17 | |||
| Fiber intake | Yes | 5 | 0.92 (0.89,0.95) | 46.4 | 0.11 | 4 | 0.85 (0.80,0.92) | 28.9 | 0.24 | |||||
| No | 7 | 0.94 (0.91,0.97) | 27.7 | 0.21 | 0.067 | 4 | 0.93 (0.86,1.00) | 0 | 0.68 | 0.17 | ||||
| Coffee | Yes | 6 | 0.98 (0.93,1.03) | 38 | 0.14 | 4 | 0.83 (0.77,0.89) | 0 | 0.64 | |||||
| No | 6 | 0.92 (0.89,0.95) | 38 | 0.17 | 0.069 | 0.47 | 4 | 0.92 (0.87,0.98) | 0 | 0.68 | 0.06 | 0.41 | ||
| Fruit, vegetables | Yes | 9 | 0.93 (0.90,0.96) | 37.1 | 0.11 | 2 | 0.94 (0.87,1.01) | 0 | 0.55 | |||||
| No | 3 | 0.99 (0.90,1.08) | 63.1 | 0.07 | 0.17 | 6 | 0.85 (0.81,0.91) | 0 | 0.44 | 0.1 | ||||
| Meat | Yes | 7 | 0.92 (0.89,0.95) | 44.5 | 0.08 | 4 | 0.83 (0.77,0.89) | 0 | 0.64 | |||||
| No | 5 | 0.97 (0.93,1.02) | 28.4 | 0.23 | 0.098 | 0.58 | 4 | 0.92 (0.87,0.98) | 0 | 0.68 | 0.06 | 0.34 | ||
| Cacium, magnesium | Yes | 7 | 0.95 (0.91,0.98) | 44.5 | 0.08 | 4 | 0.84 (0.78,0.90) | 0 | 0.50 | |||||
| No | 5 | 0.93 (0.88,0.99) | 48.4 | 0.10 | 0.50 | 4 | 0.92 (0.86,0.97) | 0 | 0.43 | 0.12 | ||||
| Energy intake | Yes | 9 | 0.93 (0.90,0.96) | 43.3 | 0.08 | 7 | 0.88 (0.83,0.94) | 28.2 | 0.21 | |||||
| No | 3 | 0.98 (0.92,1.04) | 33.6 | 0.21 | 0.13 | 1 | 0.88 (0.78,0.99) | 0.95 | ||||||
BMI, body mass index; n, the number of studies,Pa, for heterogeneity within each subgroup; Pb, for heterogeneity between subgroups with univariate meta-regression analysis; Pc, for heterogeneity with multivariate meta-regression analysis.
Figure 4. Dairy products and incidence of T2DM, nonlinear dose–response analysis.
A, total dairy. B, low-fat dairy.
Low- and Full-fat Dairy Intake and T2DM Risk
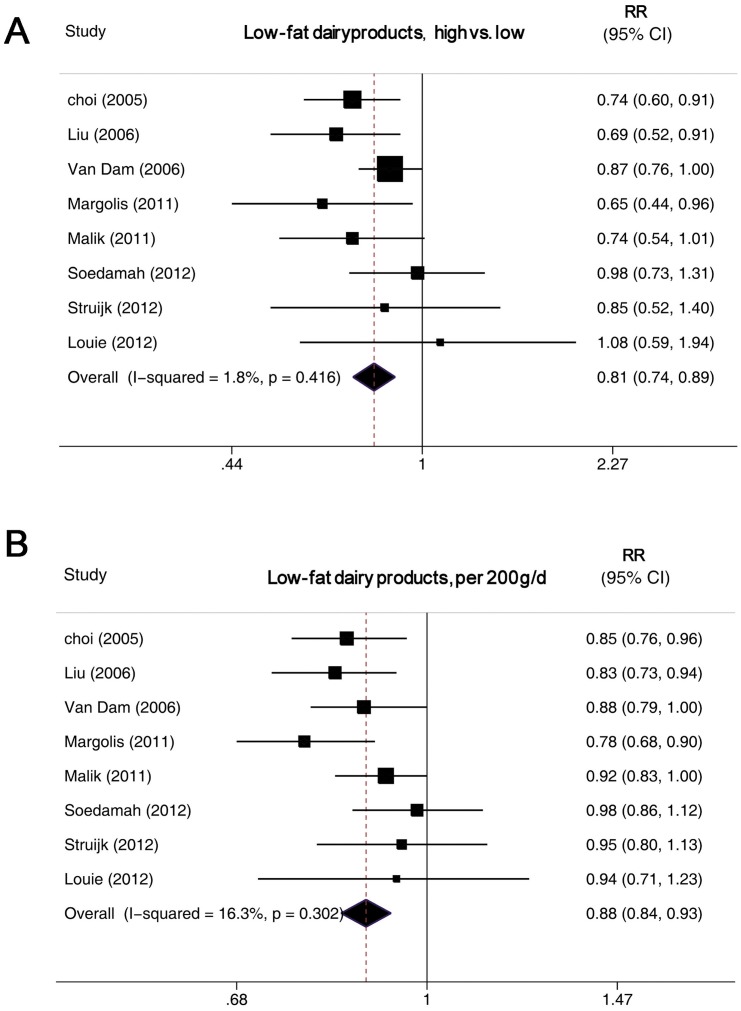
8 studies [10], [11], [12], [13], [14], [15], [16], [23] including 260,700 subjects (9,398 cases) were analyzed.
High versus low intake
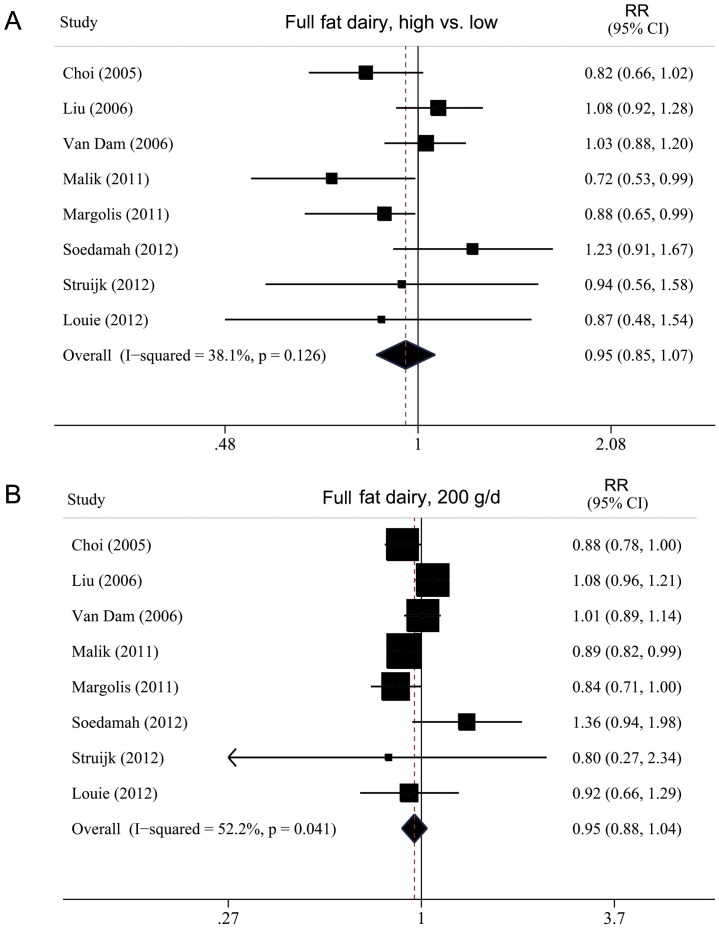
Low-fat dairy consumption was inversely associated with T2DM risk, with a pooled RR of 0.81 (0.74–0.89) (Figure 5A). Full-fat dairy consumption was not associated with T2DM risk, with a summary RR of 0.95 (0.85–1.07) (Figure 6A). We found no significant heterogeneity for the associations of low-fat (I2 = 1.8%; Pheterogeneity = 0.42) or full-fat dairy consumption (I2 = 38.1%; Pheterogeneity = 0.13).
Figure 5. Forest plot of RR for low-fat dairy products intake and T2DM.
A, highest versus lowest intake. B, dose–response analysis (200 g/d). Weights are from random-effects analysis.
Figure 6. Forest plot of RR for full-fat dairy products intake and T2DM.
A, highest versus lowest intake. B, dose–response analysis (200 g/d). Weights are from random-effects analysis.
Dose–response analysis
The summary RR for a 200-g/day increase in low-fat dairy intake was 0.88 (0.84–0.93), with no evidence of heterogeneity, I2 = 16.3% and Pheterogeneity = 0.32 (Figure 5B). The summary RR for a 200-g/day increase in full-fat dairy intake was 0.95 (0.88–1.04), with evidence of heterogeneity, I2 = 52.2% and Pheterogeneity = 0.04 (Figure 6B). We found an inverse association of low-fat dairy intake and T2DM risk for all strata, although in some analyses the associations were not statistically significant (Table 2). On univariate meta-regression analysis, the effect was weaker, although not significantly, for studies with than without adjustment for coffee and meat intake (P = 0.06 for both). But on multivariate meta-regression, we failed to identify the source of heterogeneity. On We found a nonlinear association of low-fat dairy intake and T2DM risk, Pfor nonlinearity = 0.02, with most of the risk reduction occurring with intake up to about 300 g/day; higher intake (>400 g/day) was not associated with a further decrease in risk (Figure 4B).
Milk Intake and T2DM Risk
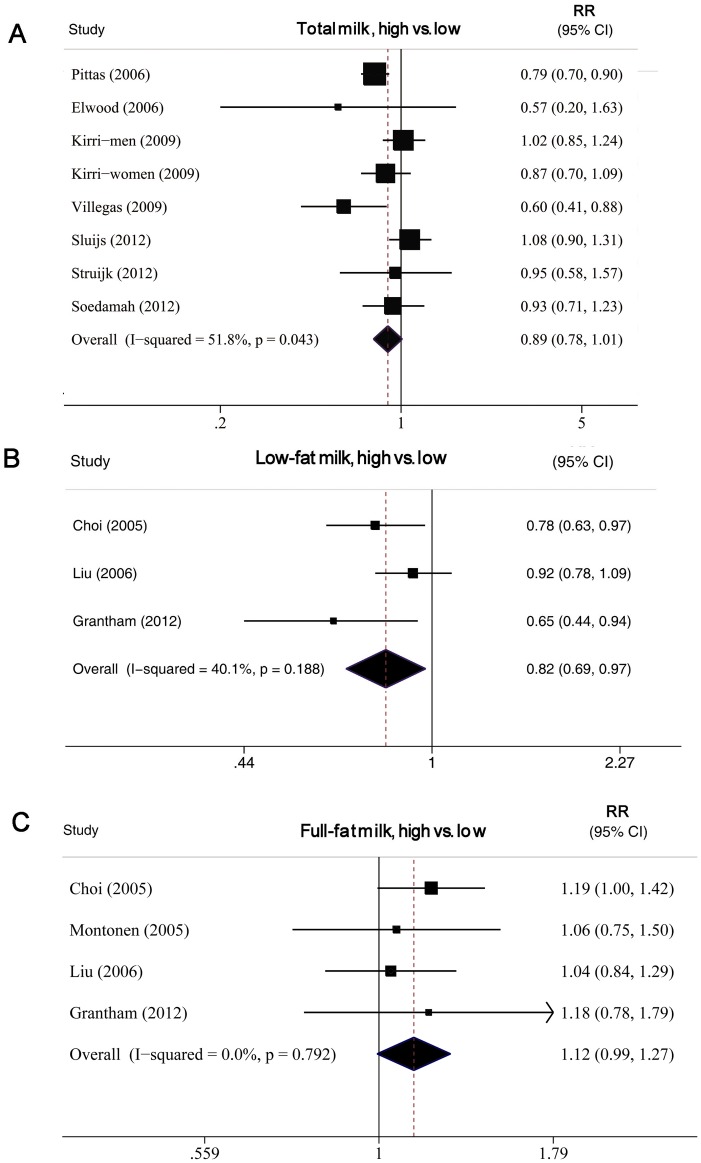
High versus low analysis
9 studies [11], [13], [15], [17], [18], [19], [20], [22], [23] including 327,039 subjects (21,755 cases) were analyzed. For 6 studies [11], [17], [18], [19], [22], [23], data were available on the association of total milk intake and T2DM risk, for 3 studies [13], [15], [20], milk consumption was analyzed as full-fat milk (or whole milk) and low-fat milk (or skim milk) and for 1 study, data was reported as full fat milk only [35]. The summary RR for total milk intake was 0.89 (0.78–1.01), with moderate heterogeneity, I2 = 51.8% and P heterogeneity = 0.043. The summary RR for low-fat and full-fat milk intake was 0.82 (0.69–0.97, I2 = 40% and Pheterogeneity = 0.19) and 1.12 (0.99–1.27, I2 = 0% and Pheterogeneity = 0.79), respectively (Figure 7).
Figure 7. Forest plot of RR for highest versus lowest milk intake and T2DM.
A, total milk. B, low fat milk. C, full fat milk. Weights are from random-effects analysis.
Dose–response analysis
8 studies [11], [13], [15], [17], [18], [19], [20], [22], [23] were analyzed. The summary RR for a 200-g/day increase in total milk intake was 0.89 (0.79–1.01), with evidence of moderate heterogeneity, I2 = 66.3% and Pheterogeneity = 0.005 (data not shown). The summary RR for a 200-g/day increase in full- and low-fat milk intake was 1.27 (0.97–1.67, I2 = 0% and Pheterogeneity = 0.58) and 0.83 (0.70–1.00, I2 = 14% and Pheterogeneity = 0.21), respectively.
Yogurt and Cheese Intake and T2DM Risk
Seven studies [11], [12], [13], [15], [17], [19], [20] including 254,552 subjects (18,532 cases) were analyzed for yogurt intake. Seven studies [11], [13], [15], [17], [19], [20], [23] including 178,429 subjects (14,810 cases) were analyzed for cheese intake.
High versus low intake
Yogurt and cheese intake were inversely associated with T2DM risk. The pooled RRs were 0.85 (0.75–0.97, I2 = 55% and Pheterogeneity = 0.02) and 0.82 (0.77–0.87, I2 = 0% and Pheterogeneity = 0.82), respectively.
Dose–response analysis
Yogurt and cheese intake were inversely associated with T2DM incidence. The pooled RRs were 0.91 (0.82–1.00, I2 = 74%, Pheterogeneity = 0.001) per 50 g/d and 0.80 (0.69–0.93, I2 = 59%, Pheterogeneity = 0.02) per 30 g/d, respectively.
Other dairy products
Intake of other types of dairy products except ice cream (n = 2 studies) were not significantly associated with T2DM risk (0.84 [0.73–0.95]). The pooled RR for total fermented dairy intake (n = 3 studies) was 0.94 (0.75–1.18) and cream (n = 2 studies) was 0.96 (0.84–1.12).
Discussion
This meta-analysis showed that total dairy intake was associated with a 6% lower risk of T2DM per 200 g/day consumption. When examining different types of dairy products in relation to T2DM risk, we found significant inverse associations of intake of low-fat dairy, low-fat milk, cheese and yogurt and T2DM. We found no association of intake of full-fat dairy as well as total and full-fat milk and T2DM. We also clarified a nonlinear association of both total and low-fat dairy intake and incidence of T2DM.
The hypothesis that dairy products intake protects against T2DM has received much interest among medical professionals and the general population. In intervention studies, the Dietary Approaches to Stop Hypertension (DASH) diet (a dietary pattern focusing on low-fat milk and other dairy products) increased high-density lipoprotein levels, reduced triglycerides levels, reduced blood pressure (both systolic and diastolic), contributed to weight loss, and reduced fasting blood glucose in both men and women as compared with the control diet [36]. In epidemiological studies, the association of dairy products intake and T2DM has been explored with inconsistent results [37].
Our findings for high versus low dairy intake are consistent with results from previous meta-analyses [24], which only included 7 studies. High versus low analyses are limited because true differences in the level and range of intake between studies are not considered and may contribute to heterogeneity in the results. With the accumulated evidence, we were able to enhance the precision of the risk estimates, perform dose–response analyses of different dairy products and explore the shape of the dose–response curve and sources of heterogeneity, thereby increasing the clinical relevance of our findings [38].
In addition, the presence of both linear and nonlinear dose–response relationships of specific dairy products strengthened the findings of an association of dairy products intake and risk of T2DM. In the linear dose-response analysis, we found a 6% and 12% lower risk of T2DM per 200 g/day intake of total and low-fat dairy products, respectively. Furthermore, we discovered a potential nonlinear association of total and low-fat dairy products intake and T2DM. A low threshold of 200 g/day total dairy and 300 g/day low fat dairy may reduce the risk by about 10% or 15% respectively. Intake above that level seems to have further but modest additional benefit for T2DM risk.
Dairy is a major source of dietary calcium and magnesium, 2 minerals that have a role in the development of T2DM, for potential in improving pancreatic B-cell function and insulin sensitivity [39]. Experimental [39], prospective cohort studies [40], [41] and a recent meta-analysis [42] have provided convincing evidence to support the direct effects of calcium and magnesium intake on insulin resistance and T2DM. In this study, we found that the association of dairy intake and T2DM risk remained unchanged after adjusting for diet calcium and/or magnesium (7 studies), so other major components in dairy products could account for the association. Recently, the beneficial physiological effects of dairy protein, such as whey protein, on the control of food intake and glucose metabolism have been reported. Studies have shown the insulinotropic and glucose-lowering properties of whey protein in healthy and T2DM subjects [43]. Furthermore, in addition to milk proteins, trans-palmitoleate, obtained primarily from dairy intake, is associated with reduced incidence of diabetes [9].
Our analysis of high- and low-fat dairy products revealed an inverse association of only low-fat dairy food intake and T2DM risk. This support the present recommendations by health authorities and governments to eat low-fat rather than full-fat dairy products [44]. We think the most prominent relationship was from residual confounding by factors related to a more unhealthy diet or lifestyle. On the other hand, we can not rule out the association between the intake of saturated fatty acid (SFA). Dairy products contributed to 15% of the total dietary SFA intake [45]. Although prospective cohorts demonstrate no significant association between SFA intake and risk of T2DM, some findings from experimental and observational studies have showed that SFA intake was inversely associated with insulin sensitivity [45], [46], [47]. Finally, the likelihood of publication bias effects may cause uncertain results. For analysis of the milk products, only 3 of 14 studies separately evaluated whole vs. low-fat milk, and thus it seems that publication bias could account for the observed difference between low vs. whole fat milk. Furthermore, because cheese, even low-fat cheese, has higher fat and saturated fat than whole milk yet was still associated with lower risk, it appears less likely that the observed difference between whole fat and low fat milk would be due to higher fat or saturated fat content in whole milk. Further confirmatory results of appropriately powered studies are still needed.
Cheese, which has far more fat than whole-fat milk, more than half of which is saturated Evidence suggests that saturated fat intake has an adverse effect on insulin sensitivity and increases the risk of T2DM. In our analysis, we found inverse association between both cheese and yogurt intake and incidence of T2DM. The exact mechanisms responsible for the significant inverse association between cheese and yogurt and T2DM are unknown. It could be partly explained by the fact that both dairy subgroups are a good source for vitamin K2. Vitamin K2 is exclusively synthesized by bacteria and is therefore only present in fermented dairy products such as cheese and yogurt due to the bacterial starter fermentation [48]. Vitamin K2 has recently been linked to a reduced risk of T2DM [49]. Additionally, these dairy subcategories are particularly high in the fat-soluble vitamin D, which has been found to be inversely associated with T2DM [50], [51].
We did not find a consistent pattern of difference or heterogeneity in results by sex or any other study characteristics examined, except for geographic location, which significantly modified the association between total dairy products intake and T2DM risk. We found a significant inverse association among US studies, with no evidence of a protective effect of total dairy food intake in European or Asia studies. This may be a chance finding, because only 3 European studies and 1 Asian study were included in this subgroup analysis or could be due to other factors. As well, differences in the ranges of intake or intake in the referent category could explain these results. Because of the nonlinear association between total dairy food intake and T2DM risk with the strongest reduction at low levels of intake, some studies may have missed an effect because the intake in the referent category may have been already sufficient to reduce risk. For example, in some European studies, intake of total dairy food in the referent category was >200 g/d but was <200 g/d for all US studies. As well, types of dairy food intake may vary between populations. In addition, differences in study size and follow-up time may contribute to the variations. Further cohort studies of specific dairy products and T2DM risk in different populations are needed.
Our meta-analysis contains some limitations. Publication bias is a major concern for analyses that depend on only a few studies. For example, in our analysis, only 4 of the 15 studies separately evaluated full or low-fat milk. So the efficiency of analysis on different milk production was limited. The inverse association we found between dairy products intake and T2DM risk could be due to unmeasured or residual confounding. Higher intake of dairy products, especially low-fat dairy products, is often associated with other lifestyle factors, including increased physical activity, low prevalence of smoking, and overweight/obesity, although different types of dairy products may be differentially associated with some of these confounders. In addition, the results were generally similar in the subgroup analyses when we stratified results by adjustment for confounding factors or other study characteristics, with no heterogeneity between subgroups for total and low-fat dairy product consumption. Only the analysis of total dairy products revealed some indication of heterogeneity, with studies that adjusted for family history of T2DM showing an inverse association with T2DM; studies that did not adjust for family history of T2DM showed a nonsignificant positive association, which suggests potential confounding.
Measurement errors in the assessment of dietary intake are known to bias effect estimates. Our results are based on data from cohort studies, in which dairy intake was mostly assessed by food-frequency questionnaires. In several studies, validation of the food-frequency questionnaires showed good correlations, of ≈0.6–0.7 for milk or (if not assessed) for protein and calcium, which are good indicators for milk intake. However, we cannot exclude that measurement errors might have resulted in attenuated associations. Dietary changes after baseline can also attenuate associations of dietary intake and T2DM risk; however, only 7 of the included studies [12], [13], [14], [17], [18], [22], [23] used repeated assessments of diet, and the results were similar when using only the baseline questionnaire for the analyses (data not shown). Furthermore, dietary intake data were collected between 1984 and 2003. In earlier studies, full-fat dairy was a major contributor to total dairy intake, whereas in later studies intake was more often low-fat dairy and publication year may have explained the study heterogeneity (p = 0.02). Finally, because all the studies were conducted primarily among middle-aged and older people, these results might not be generalizable to dairy intake in earlier life periods, which might have similar or different effects.
In conclusion, our results suggest a inverse association of intake of dairy products, such as low-fat dairy, cheese and yogurt and T2DM risk. Further cohort studies are warranted to investigate the specific types of dairy products in the association, the impact of measurement errors on estimates, any gender-specific recommendations, and biomarkers of dairy intake.
Supporting Information
Funnel plot of assessing evidence of publication bias. A. For total dairy; B. For low-fat dairy; C. For full-fat dairy.
(TIF)
Forest plot of RR for highest versus lowest yogurt and cheese intake and T2DM. A, yogurt. B, cheese. Weights are from random-effects analysis.
(TIF)
PRISMA 2009 Checklist
(DOC)
Acknowledgments
We thank Nita G. Forouhi, Dianna J. Magliano and Karen L. Margolis for providing additional data for the meta-analysis. We thank Nicola Orsini for additional help in statistical works. We thank Laura Smales from the http://www.biomedediting.com for manuscript editing.
Funding Statement
This study was supported by a grant from National Natural Science Foundation of China (no. 30900617 to D. Gao). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Guariguata L, Whiting D, Weil C, Unwin N (2011) The International Diabetes Federation diabetes atlas methodology for estimating global and national prevalence of diabetes in adults. Diabetes Res Clin Pract 94: 322–332. [DOI] [PubMed] [Google Scholar]
- 2. Polonsky KS (2012) The past 200 years in diabetes. N Engl J Med 367: 1332–1340. [DOI] [PubMed] [Google Scholar]
- 3. Straka RJ, Liu LZ, Girase PS, DeLorenzo A, Chapman RH (2009) Incremental cardiovascular costs and resource use associated with diabetes: an assessment of 29,863 patients in the US managed-care setting. Cardiovasc Diabetol 8: 53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Woodward M, Zhang X, Barzi F, Pan W, Ueshima H, et al. (2003) The effects of diabetes on the risks of major cardiovascular diseases and death in the Asia-Pacific region. Diabetes Care 26: 360–366. [DOI] [PubMed] [Google Scholar]
- 5. Huxley R, Barzi F, Woodward M (2006) Excess risk of fatal coronary heart disease associated with diabetes in men and women: meta-analysis of 37 prospective cohort studies. BMJ 332: 73–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Lakerveld J, Bot SD, Chinapaw MJ, van Tulder MW, van Oppen P, et al. (2008) Primary prevention of diabetes mellitus type 2 and cardiovascular diseases using a cognitive behavior program aimed at lifestyle changes in people at risk: Design of a randomized controlled trial. BMC Endocr Disord 8: 6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Jakubowicz D, Froy O (2013) Biochemical and metabolic mechanisms by which dietary whey protein may combat obesity and Type 2 diabetes. J Nutr Biochem 24: 1–5. [DOI] [PubMed] [Google Scholar]
- 8. Mozaffarian D, de Oliveira Otto MC, Lemaitre RN, Fretts AM, Hotamisligil G, et al. (2013) trans-Palmitoleic acid, other dairy fat biomarkers, and incident diabetes: the Multi-Ethnic Study of Atherosclerosis (MESA). Am J Clin Nutr 97: 854–861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Mozaffarian D, Cao H, King IB, Lemaitre RN, Song X, et al. (2010) Trans-palmitoleic acid, metabolic risk factors, and new-onset diabetes in U.S. adults: a cohort study. Ann Intern Med 153: 790–799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Louie JC, Flood VM, Rangan AM, Burlutsky G, Gill TP, et al.. (2012) Higher regular fat dairy consumption is associated with lower incidence of metabolic syndrome but not type 2 diabetes. Nutr Metab Cardiovasc Dis. [DOI] [PubMed]
- 11.Soedamah-Muthu SS, Masset G, Verberne L, Geleijnse JM, Brunner EJ (2012) Consumption of dairy products and associations with incident diabetes, CHD and mortality in the Whitehall II study. Br J Nutr: 1–9. [DOI] [PubMed]
- 12. Margolis KL, Wei F, de Boer IH, Howard BV, Liu S, et al. (2011) A diet high in low-fat dairy products lowers diabetes risk in postmenopausal women. J Nutr 141: 1969–1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Liu S, Choi HK, Ford E, Song Y, Klevak A, et al. (2006) A prospective study of dairy intake and the risk of type 2 diabetes in women. Diabetes Care 29: 1579–1584. [DOI] [PubMed] [Google Scholar]
- 14. Malik VS, Sun Q, van Dam RM, Rimm EB, Willett WC, et al. (2011) Adolescent dairy product consumption and risk of type 2 diabetes in middle-aged women. Am J Clin Nutr 94: 854–861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Choi HK, Willett WC, Stampfer MJ, Rimm E, Hu FB (2005) Dairy consumption and risk of type 2 diabetes mellitus in men: a prospective study. Arch Intern Med 165: 997–1003. [DOI] [PubMed] [Google Scholar]
- 16. van Dam RM, Hu FB, Rosenberg L, Krishnan S, Palmer JR (2006) Dietary calcium and magnesium, major food sources, and risk of type 2 diabetes in U.S. black women. Diabetes Care 29: 2238–2243. [DOI] [PubMed] [Google Scholar]
- 17. Kirii K, Mizoue T, Iso H, Takahashi Y, Kato M, et al. (2009) Calcium, vitamin D and dairy intake in relation to type 2 diabetes risk in a Japanese cohort. Diabetologia 52: 2542–2550. [DOI] [PubMed] [Google Scholar]
- 18. Pittas AG, Dawson-Hughes B, Li T, Van Dam RM, Willett WC, et al. (2006) Vitamin D and calcium intake in relation to type 2 diabetes in women. Diabetes Care 29: 650–656. [DOI] [PubMed] [Google Scholar]
- 19. Sluijs I, Forouhi NG, Beulens JW, van der Schouw YT, Agnoli C, et al. (2012) The amount and type of dairy product intake and incident type 2 diabetes: results from the EPIC-InterAct Study. Am J Clin Nutr 96: 382–390. [DOI] [PubMed] [Google Scholar]
- 20.Grantham NM, Magliano DJ, Hodge A, Jowett J, Meikle P, et al.. (2012) The association between dairy food intake and the incidence of diabetes in Australia: the Australian Diabetes Obesity and Lifestyle Study (AusDiab). Public Health Nutr: 1–7. [DOI] [PMC free article] [PubMed]
- 21. Elwood PC, Pickering JE, Fehily AM (2007) Milk and dairy consumption, diabetes and the metabolic syndrome: the Caerphilly prospective study. J Epidemiol Community Health 61: 695–698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Villegas R, Gao YT, Dai Q, Yang G, Cai H, et al. (2009) Dietary calcium and magnesium intakes and the risk of type 2 diabetes: the Shanghai Women's Health Study. Am J Clin Nutr 89: 1059–1067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Struijk EA, Heraclides A, Witte DR, Soedamah-Muthu SS, Geleijnse JM, et al.. (2012) Dairy product intake in relation to glucose regulation indices and risk of type 2 diabetes. Nutr Metab Cardiovasc Dis. [DOI] [PubMed]
- 24. Tong X, Dong JY, Wu ZW, Li W, Qin LQ (2011) Dairy consumption and risk of type 2 diabetes mellitus: a meta-analysis of cohort studies. Eur J Clin Nutr 65: 1027–1031. [DOI] [PubMed] [Google Scholar]
- 25. Wong WC, Cheung CS, Hart GJ (2008) Development of a quality assessment tool for systematic reviews of observational studies (QATSO) of HIV prevalence in men having sex with men and associated risk behaviours. Emerg Themes Epidemiol 5: 23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, et al. (2000) Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 283: 2008–2012. [DOI] [PubMed] [Google Scholar]
- 27. von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, et al. (2008) The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol 61: 344–349. [DOI] [PubMed] [Google Scholar]
- 28. Carter P, Gray LJ, Troughton J, Khunti K, Davies MJ (2010) Fruit and vegetable intake and incidence of type 2 diabetes mellitus: systematic review and meta-analysis. BMJ 341: c4229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Zhang J, Yu KF (1998) What's the relative risk? A method of correcting the odds ratio in cohort studies of common outcomes. JAMA 280: 1690–1691. [DOI] [PubMed] [Google Scholar]
- 30. Greenland S, Longnecker MP (1992) Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am J Epidemiol 135: 1301–1309. [DOI] [PubMed] [Google Scholar]
- 31.Orsini N BR, Greenland S (2006) Generalized least squares for trend estimation of summarized dose-response data. Stata Journal: 17.
- 32. Bodner-Montville J AJ, Ingwersen LA, Haggerty ES, Wilkinson Enns C, Perloff BP (2006) USDA Food and Nutrient Database for Dietary Studies: Released on the web. J Food Composition Analysis 19: 7. [Google Scholar]
- 33. Orsini N, Li R, Wolk A, Khudyakov P, Spiegelman D (2012) Meta-analysis for linear and nonlinear dose-response relations: examples, an evaluation of approximations, and software. Am J Epidemiol 175: 66–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Ericson U, Sonestedt E, Gullberg B, Hellstrand S, Hindy G, et al. (2013) High intakes of protein and processed meat associate with increased incidence of type 2 diabetes. Br J Nutr 109: 1143–1153. [DOI] [PubMed] [Google Scholar]
- 35. Montonen J, Jarvinen R, Heliovaara M, Reunanen A, Aromaa A, et al. (2005) Food consumption and the incidence of type II diabetes mellitus. Eur J Clin Nutr 59: 441–448. [DOI] [PubMed] [Google Scholar]
- 36. Azadbakht L, Mirmiran P, Esmaillzadeh A, Azizi T, Azizi F (2005) Beneficial effects of a Dietary Approaches to Stop Hypertension eating plan on features of the metabolic syndrome. Diabetes Care 28: 2823–2831. [DOI] [PubMed] [Google Scholar]
- 37. Elwood PC, Pickering JE, Givens DI, Gallacher JE (2010) The consumption of milk and dairy foods and the incidence of vascular disease and diabetes: an overview of the evidence. Lipids 45: 925–939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Thompson SG (1994) Why sources of heterogeneity in meta-analysis should be investigated. BMJ 309: 1351–1355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Belin RJ, He K (2007) Magnesium physiology and pathogenic mechanisms that contribute to the development of the metabolic syndrome. Magnes Res 20: 107–129. [PubMed] [Google Scholar]
- 40. Kirii K, Iso H, Date C, Fukui M, Tamakoshi A (2010) Magnesium intake and risk of self-reported type 2 diabetes among Japanese. J Am Coll Nutr 29: 99–106. [DOI] [PubMed] [Google Scholar]
- 41. Lopez-Ridaura R, Willett WC, Rimm EB, Liu S, Stampfer MJ, et al. (2004) Magnesium intake and risk of type 2 diabetes in men and women. Diabetes Care 27: 134–140. [DOI] [PubMed] [Google Scholar]
- 42. Dong JY, Xun P, He K, Qin LQ (2011) Magnesium intake and risk of type 2 diabetes: meta-analysis of prospective cohort studies. Diabetes Care 34: 2116–2122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Sousa GT, Lira FS, Rosa JC, de Oliveira EP, Oyama LM, et al. (2012) Dietary whey protein lessens several risk factors for metabolic diseases: a review. Lipids Health Dis 11: 67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. German JB, Gibson RA, Krauss RM, Nestel P, Lamarche B, et al. (2009) A reappraisal of the impact of dairy foods and milk fat on cardiovascular disease risk. Eur J Nutr 48: 191–203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kratz M, Baars T, Guyenet S (2012) The relationship between high-fat dairy consumption and obesity, cardiovascular, and metabolic disease. Eur J Nutr. [DOI] [PubMed]
- 46. Riserus U, Arner P, Brismar K, Vessby B (2002) Treatment with dietary trans10cis12 conjugated linoleic acid causes isomer-specific insulin resistance in obese men with the metabolic syndrome. Diabetes Care 25: 1516–1521. [DOI] [PubMed] [Google Scholar]
- 47.Niu K, Kobayashi Y, Guan L, Monma H, Guo H, et al.. (2012) Low-fat dairy, but not whole-/high-fat dairy, consumption is related with higher serum adiponectin levels in apparently healthy adults. Eur J Nutr. [DOI] [PubMed]
- 48. Schurgers LJ, Vermeer C (2000) Determination of phylloquinone and menaquinones in food. Effect of food matrix on circulating vitamin K concentrations. Haemostasis 30: 298–307. [DOI] [PubMed] [Google Scholar]
- 49. Beulens JW, van der AD, Grobbee DE, Sluijs I, Spijkerman AM, et al. (2010) Dietary phylloquinone and menaquinones intakes and risk of type 2 diabetes. Diabetes Care 33: 1699–1705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Khan H, Kunutsor S, Franco OH, Chowdhury R (2013) Vitamin D, type 2 diabetes and other metabolic outcomes: a systematic review and meta-analysis of prospective studies. Proc Nutr Soc 72: 89–97. [DOI] [PubMed] [Google Scholar]
- 51. Song Y, Wang L, Pittas AG, Del Gobbo LC, Zhang C, et al. (2013) Blood 25-hydroxy vitamin D levels and incident type 2 diabetes: a meta-analysis of prospective studies. Diabetes Care 36: 1422–1428. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Funnel plot of assessing evidence of publication bias. A. For total dairy; B. For low-fat dairy; C. For full-fat dairy.
(TIF)
Forest plot of RR for highest versus lowest yogurt and cheese intake and T2DM. A, yogurt. B, cheese. Weights are from random-effects analysis.
(TIF)
PRISMA 2009 Checklist
(DOC)