Abstract
We compared the performance of four rapid diagnostic tests (RDTs) for imported malaria, and particularly Plasmodium falciparum infection, using thick and thin blood smears as the gold standard. All the tests are designed to detect at least one protein specific to P. falciparum ( Plasmodium histidine-rich protein 2 (PfHRP2) or Plasmodium LDH (PfLDH)) and one pan-Plasmodium protein (aldolase or Plasmodium LDH (pLDH)). 1,311 consecutive patients presenting to 9 French hospitals with suspected malaria were included in this prospective study between April 2006 and September 2008. Blood smears revealed malaria parasites in 374 cases (29%). For the diagnosis of P. falciparum infection, the three tests detecting PfHRP2 showed high and similar sensitivity (96%), positive predictive value (PPV) (90%) and negative predictive value (NPV) (98%). The PfLDH test showed lower sensitivity (83%) and NPV (80%), despite good PPV (98%). For the diagnosis of non-falciparum species, the PPV and NPV of tests targeting pLDH or aldolase were 94–99% and 52–64%, respectively. PfHRP2-based RDTs are thus an acceptable alternative to routine microscopy for diagnosing P. falciparum malaria. However, as malaria may be misdiagnosed with RDTs, all negative results must be confirmed by the reference diagnostic method when clinical, biological or other factors are highly suggestive of malaria.
Introduction
Malaria is endemic in 99 countries worldwide, and several million people travel from non-endemic countries to malaria-endemic regions each year [1]. Although more than 7000 cases of imported malaria are reported annually both in European countries and in the United States, malaria remains unusual in non-endemic countries and its clinical presentation is often nonspecific [2,3]. Misdiagnosis which remains too frequent, resulting in delays in antimalarial treatment [4]. A careful physical examination and interview, including any recent travel to a malaria-endemic region, are crucial for diagnostic evaluation. More than 90% of cases of imported malaria are due to Plasmodium falciparum, the species associated with severe morbidity and mortality. The remaining cases are due to P. ovale, P. vivax, P. malariae or, rarely, P . knolewsi , alone or in association with P. falciparum [4].
Malaria is a serious disease that must be diagnosed urgently. The reference routine test is microscopic examination of Giemsa-stained blood smears [5]. However, many centers cannot provide reliable round-the-clock smear-based diagnosis. Thus, a simple, sensitive test capable of reliably confirming or ruling out malaria would be a welcome addition to the diagnostic arsenal.
Rapid diagnostic tests (RDTs) that detect malaria parasite proteins by immunochromatography were first developed 20 years ago as a complement to microscopic diagnosis [6]. RDTs detect a variety of proteins, including P. falciparum histidine-rich protein 2 (PfHRP2) and P. falciparum lactate dehydrogenase (PfLDH), both specific to P. falciparum; and also Plasmodium LDH (pLDH) and aldolase, enzymes shared by the 5 human pathogenic Plasmodium species [6]. Such tests are now increasingly used outside of malaria-endemic areas [7–9]. Numerous studies including a Cochrane meta-analysis assessed the accuracy of RDTs for diagnosis of malaria in endemic settings [10–14]. Studies were conducted in Europe among travelers returning from endemic areas [15,16] but results are limited to P. ovale, P. malariae and P. vivax, and shown large differences in performance [9]. Tests available in non-endemic countries bear the CE (European Conformity) label but their performance is extremely variable; in addition, they have mainly been tested in endemic areas and have never been compared in the same study [17].
In 2008, WHO launched a comparative study of some RDTs on selected samples with P. falciparum and P. vivax [18] but studies using clinical samples are the most informative ones regarding test performance in routine use. Previous studies of patients presenting to emergency rooms in Europe with suspected imported malaria were all retrospective and compared only 1 or 2 RDTs with standard microscopic methods [15,16,19–21]. We report the results of a multicenter study of the diagnostic performance of four RDTs selected among those most widely used in Europe to diagnose imported malaria, performed in usual care settings in non-endemic areas. Thick and thin blood smears were considered as the gold standard and discordant results were explored in an attempt to obtain an analytical explanation.
Methods
Study design
This prospective study included patients with fever or history of fever and a history of travel to a malaria-endemic area and who underwent diagnostic tests in one of 9 French hospitals (from April 2006 to September 2008 in 6 centers and from September 2007 to September 2008 in 3 centers) (Table 1) (Protocol summary S1, Study protocol S1). The study was designed, conducted and reported in compliance with the Standards for the Reporting of Diagnostic Accuracy (STARD) guidelines [22] (STARD checklist S1).
Table 1. Characteristics of the participating centers.
| Participating center | Category of hospital | Patients | Number of suspected cases of malaria per year | Number of malaria diagnoses per year | Number of patients included in the study (%) (n=1311) |
|---|---|---|---|---|---|
| Bichat hospital | University Hospital | Adults | 752 | 126 (16.8%) | 571 (44.3%) |
| Jean-Verdier hospital | University hospital | Children + adults | 200 | 40 (20%) | 195 (15.1%) |
| Delafontaine hospital | General hospital | Children + adults | 719 | 95 (13.2%) | 145 (11.3%) |
| Institut Pasteur | Travel clinic | Adults | 650 | 25 (3.8%) | 97 (7.5%) |
| Pitié-Salpêtrière hospital | University Hospital | Adults | 1021 | 90 (8.8%) | 69 (5.4%)* |
| Robert-Ballanger hospital | General hospital | Children + adults | 345 | 57 (16.5%) | 68 (5.3%) |
| Robert-Debré hospital | University Hospital | Children | 670 | 65 (9.7%) | 63 (4.9%) |
| Pellegrin + Saint-André hospitals | University hospital | Children + adults | 600 | 80 (13.3%) | 80 (6.1%)* |
These sites participated in the study for 12 months only
Ethics Statement
This research was non interventional and, in accordance with French legislation (article R1121-2 of the French public health code), was authorized by two French authorities (CCTIRS and CNIL) and registered with the Ile-de-France XI ethics committee under identification number 06080 (Report of ethic committee S1, ANRS recommendation research study S1). No authorization was required from the latter body, in accordance with French legislation on non-interventional research. Patients (or the parents of minors) were individually informed, through a written document (approved by both CCTIRS and CNIL), and their non-objection to participation was systematically collected in their medical files. The study is registered with Clinical Trials.gov (identifier NCT00451269) (http://www.clinicaltrials.gov/).
Patients
Patients attending a participating center with fever or history of fever and with a history of travel to malaria-endemic areas leading to prescription of laboratory tests for malaria were prospectively considered for enrollment. After reading a dedicated information sheet, patients were excluded if they (or the parents of minors) declined to participate. The non-objection to participation was systematically collected by investigators and recorded in the medical files of all participants. Patients were recruited in 8 hospitals (five university hospitals, three general hospitals) with emergency rooms or infectious and tropical diseases departments, and in one private hospital. These centers managed between 25 and 126 cases of malaria each year, in children and/or adults (Table 1). Patient participation in the study was limited to a single blood sample used both for the diagnosis of acute malaria and for other relevant tests. A physician collected the patient’s age, gender, birth country, country of residence, country of travel, date of return, malaria chemoprophylaxis, and any antimalarial treatment taken before consulting.
Preparation and reading of smears
The same venous blood sample taken in an anticoagulant (EDTA)-containing tube was used for blood smears and the four RDTs. Patient care decisions were based on the results of routine methods performed in each center, using the same venous blood sample. The 4 RDTs were performed simultaneously by technicians in each participating center. A sample of the same blood was immediately sent to the French National Malaria Reference Center (FNMRC), where reference thin and thick blood smears were prepared and stained upon receipt. Each slide was read by one of the three expert microscopists involved in this study, who were blinded to the patients’ characteristics and symptoms and to the results of the RDTs [23]. The results of the expert readings were not compared with those of the non-centralized readings. Thin smears prepared at the FNMRC were considered positive for malaria if one or more malaria parasites were visualized, and negative if no asexual form of Plasmodium was observed in 200 high-power fields (about 40 000 erythrocytes). Parasite density was expressed as the percentage of infected red cells. Thick blood smears prepared at FNMRC were considered positive if one or more malaria parasites were visualized and negative if no parasites were detected after examining 1000 white blood cells. The parasite species was determined. The isolated presence of P. falciparum gametocytes was noted but was not considered indicative of acute malaria.
To assess the reproducibility of the centralized microscopic examination, smears of 30 blood samples were read independently and blindly by a biologist who had no other role in the study.
Rapid diagnostic tests
All 4 tests were capable of detecting at least one P. falciparum-specific protein and one pan-Plasmodium protein. The Now ICT Malaria test (PfHRP2-test1, pan-aldolase test) (manufactured by Binax, distributed by Inverness, France) detects P. falciparum PfHPR2 and the aldolase of the 5 human Plasmodium species. The Core Malaria Pan/Pv/Pf test (PfHRP2-test2, pLDH-test2) (Ivagen, France) and the Palutop+4 test (PfHRP2-test3, pLDH-test3) (All Diag, France) detect PfHRP2, P. vivax PvLDH, and pLDH of the 5 human Plasmodium species. The Optimal-IT test (PfLDH-test, pLDH-test1) (Diamed, France) detects P. falciparum PfLDH and pLDH. The tests were based on lateral flow immunochromatography, in either cassette format (PfLDH-test, PfHRP2-test2, and PfHRP2-test3) or card format (PfHRP2-test1) (Table 2). PfHRP2-test1 and PfLDH-test are three-band tests, while PfHRP2-test2 and PfHRP2-test3 are four-band tests. All 4 tests include a control line that must be present for the test to be valid. The kits were stored in a dry environment between 18°C and 25°C.
Table 2. Names and targets of the rapid diagnostic tests for malaria.
| Test | Name | Distributor in France | Target | Species |
|---|---|---|---|---|
| PfHRP2-test1 | Now ICT Malaria | Inverness | PfHRP2 | Plasmodium falciparum |
| pan-aldolase test | Aldolase | All species | ||
| PfHRP2-test2 | Core Malaria | Ivagen | PfHRP2 | P. falciparum |
| pLDH-test2 | Pan/Pv/Pf | pLDH | All species | |
| PvLDH | Plasmodium vivax | |||
| PfHRP2-test3 | Palutop +4 | All Diag | PfHRP2 | P. falciparum |
| pLDH-test3 | pLDH | All species | ||
| PvLDH | P. vivax | |||
| PfLDH test | Optimal IT | Diamed | PfLDH | P. falciparum |
| pLDH-test1 | pLDH | All species |
The tests were performed according to the manufacturers’ instructions. Blood samples (5, 10 or 15 µl, depending on the test) were loaded into a transfer pipette. Samples and diluents were applied and reading was performed 10 to 20 minutes later, depending on the test. All the results were read by local trained technicians blinded to the results of the standard test. If the control line did not appear, the test was considered invalid and was repeated. Results were scored as negative (no test line visible) or positive (at least one test line visible). If the test line was barely visible, the result was scored as doubtful but was considered positive in subsequent analyses.
Inconsistencies
If the Plasmodium species could not be reliably identified on the FNMRC centralized thin blood smear, species PCR was performed by FNRMRC, as previously described, on DNA extracted from the EDTA sample, with primers specific for each of the five species of plasmodium [24,25]. In case of discrepancies between a positive RDT and negative blood smears, PCR was used to detect plasmodial DNA, as evidence of previous Plasmodium infection or submicroscopic parasitemia.
As parasite viability may influence the performance of pLDH-based tests [26], antimalarial drugs (chloroquine and its metabolite, amodiaquine and its metabolite, quinine, proguanil and its metabolite, mefloquine and doxycycline and its metabolite) were assayed by high-performance liquid chromatography on EDTA plasma to detect prior treatment when Pf-LDH- or pLDH-based RDTs were negative but blood smears were positive.
Sample size calculation
To achieve an estimated RDT sensitivity of 95% with an accuracy of 0.03 (half the 95% confidence interval [95% CI]), we needed a test sample population of 245 patients with confirmed malaria. As the rate of confirmed malaria among patients with clinical signs was approximately 20% during the year preceding the study in all the participating centers, we targeted an enrollment of approximately 1225 patients.
Statistical analysis
A patient was considered to have confirmed acute malaria in the presence of fever or a history of fever, and of asexual Plasmodium forms on a blood film. The isolated presence of P. falciparum gametocytes without asexual forms did not define a case of acute malaria [27]. The reproducibility of thin and thick blood smear reading was validated by using a Kappa concordance test. The performance of the four RDTs was compared to that of the routine “gold standard” test, i.e. centralized thin or thick blood smear, in terms of their sensitivity, specificity, positive and negative predictive values (PPV and NPV), and their 95% CIs for binomial proportions. Likelihood ratios were provided with 95% CIs calculated as risk ratios [28]. Cochran’s Q test was used to compare the sensitivity and specificity of the four RDTs. P values below 0.05 (two-tailed) were considered statistically significant. SAS v9-1 software (SAS Inst., Cary, NC, USA) was used for statistical analyses.
Results
A total of 1311 patients were included in the study (Table 1). Their mean age was 32.7 years (SD: 17.1) and 54% of patients were male. Although 90.2% of patients were living in France, 51.7% originated from malaria-endemic areas. Most patients had returned from Africa (85.0%, n=1060).
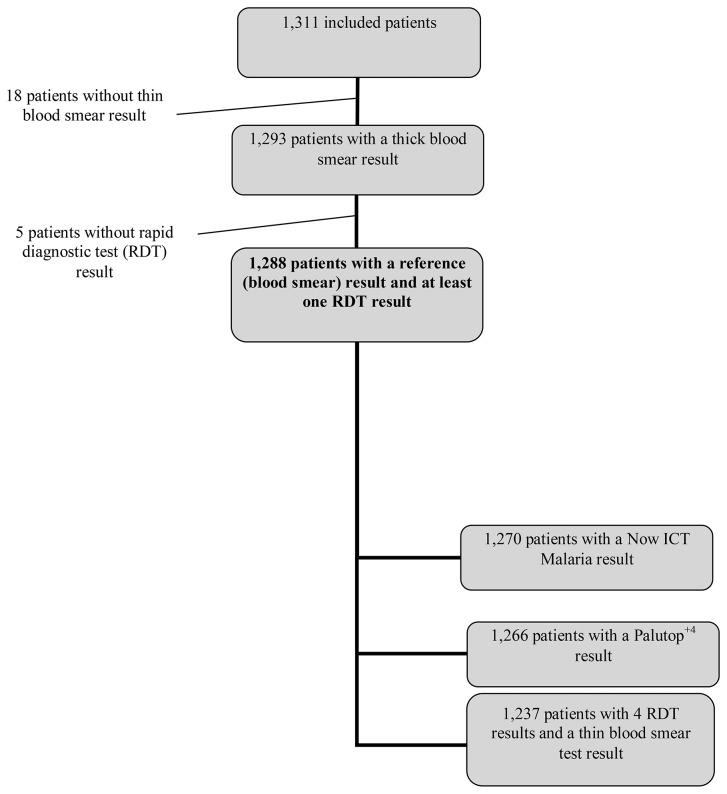
Results of the reference thin blood smear test were missing in 18 cases, and RDT results were missing in five cases (Figure 1); thus, 1288 patients were included in the analysis. Malaria was ruled out in 914 cases (71%) including 4 cases in which only P. falciparum gametocytes were detected. The reference tests confirmed malaria in 378 cases (28.8%). The species distribution was as follows: P. falciparum in 340 cases (89.9%), P. ovale in 17 cases (4.5%), P. vivax in 11 cases (2.9%), P. malariae in 8 cases (2.1%), P. falciparum associated with P. ovale in 1 case (0.3%), and P. falciparum associated with P. malariae in 1 case (0.3%). Median parasite density was 11 250/microliter (p/µl; range 8 to 9 000 000 p/µl). Parasite density was above 2000 p/µl in 59% of positive samples (n=221).
Figure 1. Flow diagram.
The kappa value for thin smear readings was 0.92 (0.76–1).
All the RDT results met the validation criteria. Figure 1 shows the number of results available for each of the four tests.
Considering P. falciparum, the sensitivity of the three RDTs targeting PfHRP2 was 96% (Table 3). P. falciparum malaria was missed in 8 of 337 cases with PfHRP2-test3, 9 of 336 cases with PfHRP2-test2, and 10 of 336 cases with PfHRP2-test1. However, the specificity of PfHRP2 detection was 97% whatever the RDT. The PPV and NPV of these 3 tests (PfHRP2-test1, PfHRP2-test2, and PfHRP2-test3) were 90% and 98% respectively. The sensitivity of PfHRP2 detection was related to parasite density and depended on the RDT used. Post-hoc analyses showed that sensitivity at parasite densities lower than 200 p/µl was 85% (28/33) with PfHRP2-test1, 88% (29/33) with PfHRP2-test2 and 94% (31/33) with PfHRP2-test3. Sensitivity at parasite densities between 200 and 2000 p/µl was 91% (48/53) with PfHRP2-test1 and PfHRP2-test2, and 92% (49/53) with PfHRP2-test3. Sensitivity was 100% with all 3 tests when parasite density was above 2000/µl. Because of the small test-group size, no statistical analysis was possible.
Table 3. Performance of the four rapid diagnostic tests according to their target antigens and Plasmodium species.
| Species | Target | PfHRP2-test1 | PfHRP2-test2 | PfHRP2-test3 | PfLDH | |
|---|---|---|---|---|---|---|
| N = 1237 | (Now ICT Malaria) | (Core Malaria) | (Palutop +4) | (Optimal-IT) | ||
| All positive | Sensitivity | 0.93 | 0.94 | 0.94 | 0.83 | |
| samples | [0.90-0.96] | [0.91-0.96] | [0.92-0.97] | [0.79-0.86] | ||
| Specificity | 0.97 | 0.96 | 0.97 | 0.99 | ||
| [0.96-0.98] | [0.95-0.97] | [0.96-0.98] | [0.99-1.00] | |||
| Species | Target | PfHRP2-test1 | PfHRP2-test2 | PfHRP2-test3 | PfLDH | |
| N = 1237 | (Now ICT Malaria) | (Core Malaria) | (Palutop +4) | (Optimal-IT) | ||
| P. falciparum | PfHRP2 | Sensitivity | 0.96 | 0.96 | 0.96 | 0.83 |
| (alone or | or | [0.94-0.98] | [0.94-0.98] | [0.94-0.98] | [0.79-0.87] | |
| mixed ) | PfLDH | Specificity | 0.97 | 0.97 | 0.97 | 1 |
| infection | [0.96-0.98] | [0.96-0.98] | [0.96-0.98] | [0.99-1] | ||
| PPV | 0.9 | 0.9 | 0.9 | 0.98 | ||
| [0.86-0.93] | [0.87-0.93] | [0.86-0.93] | [0.96-1] | |||
| NPV | 0.98 | 0.98 | 0.98 | 0.8 | ||
| [0.97-0.99] | [0.97-0.99] | [0.97-0.99] | [0.78-0.83] | |||
| LR+ | 34.8 | 36.2 | 34.5 | 188 | ||
| [23.6-51.2] | [24.4-53.7] | [23.5-50.9] | [70.6-499] | |||
| LR- | 0.04 | 0.04 | 0.04 | 0.17 | ||
| [0.02-0.06] | [0.02-0.06] | [0.03-0.07] | [0.13-0.21] | |||
| Species | Target | PvLDH | PvLDH | |||
| N=1256 | (CoreMalaria) | (Palutop +4) | ||||
| P. vivax | PvLDH | Sensitivity | 0.82 | 0.91 | ||
| [0.59-1] | [0.74-1] | |||||
| Specificity | 0.99 | 0.99 | ||||
| [0.99-1] | [0.99-1] | |||||
| PPV | 0.9 7 | 0.98 | ||||
| [0.89-1] | [0.9-1] | |||||
| NPV | 0.79 | 0.89 | ||||
| [0.76-0.81] | [0.88-0.91] | |||||
| LR+ | 145.5 | 162 | ||||
| [69.4-305.1] | [77.1-339] | |||||
| LR- | 0.18 | 0.09 | ||||
| [0.05-0.64] | [0.01-0.59] |
PPV: positive predictive value, NPV: negative predictive value, LR: likelihood ratio.
Sensitivity for PfLDH detection was 83%, missing 53 of the 337 cases of P. falciparum malaria. Specificity was 100%. The PPV and NPV of this test were 98% and 80% respectively (Table 3). Post-hoc analyses showed that sensitivity was low (41/85, 48.2%) when parasite density was below 2000 p/µl, but increased to 96.7% (238/246) when parasite density exceeded 2000 p/µl.
The RDTs targeting P. vivax LDH (PvLDH) had a specificity of 99% and a sensitivity of 82% or 91%, depending on the test (Table 3).
The sensitivity of P. falciparum pan-antigen detection was 73.6% for aldolase, and ranged from 69.5% to 81.3% for pLDH, depending on the test (p<0.001) (Table 4).
Table 4. Performance of the four rapid diagnostic tests for the detection of pan-antigen, pLDH and aldolase, according to the Plasmodium species and test.
| Species | Target |
|
Aldolase
|
pLDH-test1
|
pLDH-test2 | pLDH-test3 | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| N=1237 |
|
(Now ICT Malaria)
|
(Optimal-IT)
|
(CoreMalaria) | (Palutop +4) | |||||
| All species except | Pan-antigen: | Sensitivity | 0.57 [0.41-0.74] | 0.69 [0.53-0.84] | 0.6 [0.44-0.76] | 0.63 [0.47-0.79] | ||||
| P. falciparum | pLDH | Specificity |
1 [1–1] | 1 [0.99-1] | 0.99 [0.98-1] | 0.99 [0.99-1] | ||||
| (P. ovale, | or | PPV | 0.99 [0.94-1.0] | 0.98 [0.92-1.0] | 0.94 [0.86-1.0] | 0.96 [0.89-1.0] | ||||
| P. malariae, | Aldolase | NPV | 0.52 [0.49-0.55] | 0.64 [0.61-0.67] | 0.55 [0.52-0.58] | 0.58 [0.55-0.61] | ||||
| or P. vivax) | LR+ | 343.4 | 164.9 | 60.1 [34.2-106] | 94.4 [47.3-189] | |||||
| [86.0-1371] | [68.74-395.4] | |||||||||
| LR- | 0.43 [0.29-0.63] | 0.32 [0.19-0.51] | 0.4 [0.27-0.61] | 0.37 [0.24-0.58] | ||||||
| Plasmodium | Aldolase* | pLDH-test1* | pLDH-test2* | pLDH-test3* | Cochran Q test | |||||
| species | (Now ICT Malaria) | (Optimal-IT) | (CoreMalaria) | (Palutop +4) | P value | |||||
| P. falciparum | 246/334 (73.6%) | 271/334 (81.1%) | 232/334 (69.5%) | 264/334 (78.4%) | <0.0001 | |||||
| P. ovale | 7/18 (38.9%) | 10/18 (55.6%) | 8/18 (44.4%) | 8/18 (44.4%) | 0.10 | |||||
| P. vivax | 10/11 (90.9%) | 10/11 (90.9%) | 8/11 (72.8%) | 10/11 (90.9%) | 0.31 | |||||
| P. malariae | 5/9 (55.6%) | 6/9 (66.7%) | 5/9 (55.6%) | 6/9 (66.7%) | 0.39 | |||||
ratio of samples positive in the test to the number of samples positive in the reference method (sensitivity)
Regarding non-falciparum species, aldolase was detected as a pan-antigen in 57% of microscopically positive samples, whereas the sensitivity of pLDH detection ranged from 60% to 69%, depending on the test (Table 4). Specificity ranged from 99% to 100%, depending on the test (Table 4).
False-positive results are shown in Table 5. Most occurred with PfHRP2, and PCR was usually negative.
Table 5. False-positive results obtained with the four RDTs according to the detected antigen and the PCR results.
|
Name of the test
|
PfHRP2-test1 | PfHRP2-test2 | PfHRP2-test3 |
PfLDH test
|
|||
|---|---|---|---|---|---|---|---|
| pan-aldolase test | pLDH-test2 | pLDH-test3 |
pLDH-test1
|
||||
| (Now ICT Malaria) | (Core Malaria) | (Palutop +4) |
(Optimal-IT)
|
||||
| False positive RDT results | 26 | 34 | 29 | 6 | |||
| Detected antigen | PfHRP2 or PfLDH | 26 | 23 | 24 | 4 | ||
| pLDH | 6 | 2 | 2 | ||||
| PvLDH | 4 | 1 | |||||
| PvLDH + PfHRP2 | 1 | 1 | |||||
| Missing data | 1 | ||||||
| Gametocytes of P. falciparum | Present | 1 | 2 | 3 | 1 | ||
| Absent | 24 | 30 | 26 | 5 | |||
| Missing data | 1 | 2 | |||||
| PCR result | P.falciparum positive | 12 | 12 | 13 | 4 | ||
| Négative for all species | 13 | 21 | 15 | 2 | |||
| Missing data | 1 | 1 | 1 | ||||
False-negative RDT results (relative to microscopy and PCR) are shown in Table 6. Parasitaemia was between 8 p/µl and 117 000p/µl. Except for the PfLDH test/pLDH-test1 (Optimal-IT), most false-negative results involved P. ovale or P. malariae. Antimalarial drugs were detected in 21.7% to 33.9% of the false-negative samples, with no predominance of a particular drug.
Table 6. False-negative RDT results according to the Plasmodium species and antimalarial drug assay results.
|
Name of the test
|
PfHRP2-test1 | PfHRP2-test2 | PfHRP2-test3 | PfLDH test | |
|---|---|---|---|---|---|
|
|
pan-aldolase test | pLDH-test2 | pLDH-test | pLDH-test1 | |
|
|
(Now ICT Malaria) | (CoreMalaria) | (Palutop +4) | (Optimal-IT) | |
| False negative RDT results | 26 | 23 | 21 | 65 | |
| Parasitaemia | Minimum/Maximum | 16/8100 | 16/8100 | 16/8100 | 8/117000 |
| (p/µl) | Median [Q1 - Q3] | 232.0 [72.0-496.0] | 272.0 [72.0-1184.0] | 312.0 [120.0-1288.0] | 288.0 [68.0-756.0] |
| Species | P. falciparum | 10 (38.5%) | 9 (39.1%) | 8 (38.1%) | 53 (81.5%) |
| P. ovale | 11 (42.3%) | 8 (34.8%) | 9 (42.9%) | 8 (12.3%) | |
| P. vivax | 1 (3.8%) | 2 (8.7%) | 1 (4.8%) | 1 (1.5%) | |
| P. malariae | 4 (15.4%) | 4 (17.4%) | 3 (14.3%) | 3 (4.6%) | |
| Plasmatic | Négatif | 18 | 16 | 14 | 39 |
| antimalarial | Positif | 5 | 5 | 5 | 20 |
| detection | Not done | 3 | 2 | 2 | 6 |
Discussion
We prospectively evaluated the performance of four malaria RDTs on a large panel of samples (n=1288) obtained from travelers returning from malaria-endemic areas to France.
The overall sensitivity of the RDTs for Plasmodium infection (at least one target detected) was higher than 93%, possibly owing to the preponderance of P. falciparum in the samples studied, and to the good sensitivity of PfHRP2 detection for falciparum malaria, as reported by Abba et al. [10].
The PfHRP2-based tests emerged as a reliable alternative to routine microscopy for the diagnosis of P. falciparum malaria, and were more sensitive than the PfLDH-based tests. Taking microscopy as the standard, the sensitivity of the PfHRP2-based tests was 96% (95% CI, 94%-98%), which is higher than the threshold of 95% recommended by the World Health Organization (WHO) [29].
More than 60 RDT brands and 200 different products have already been developed. WHO and the Foundation for Innovative New Diagnostics (FIND) evaluated the sensitivity of 168 RDTs for the diagnosis of P. falciparum and P. vivax malaria [18]. This four-round evaluation showed that the tests submitted to Round 4 performed better, possibly reflecting improvements in test manufacture. The proportion of tests achieving a panel detection rate higher than 75% for a parasite density of >200/µl was higher in the latter study than previously reported. Performance varied widely among the tests at low parasite densities (below 200/µl), but the majority of tests gave high detection rates at densities of 2000 or 5000 parasites/μl [18], as also observed in our study. Only 3 among the 4 tests studied here were part of the WHO panel, namely Binax Now Malaria (PfHRP2-test1, pan-aldolase test) with performance approximately 87% at 200 parasites/µl and 98% at 2000 p/μl); Core Malaria Pan/Pv/Pf (PfHRP2-test2, pLDH-test2), respectively 89% and 98%; and Optimal iT (PfLDH-test, pLDH-test1), 50% at 200 p/µl and 98% at 2000 p/μl.
In areas where transmission rates are low, parasite density is likely to be lower in patients with symptomatic malaria [9,30]. Thus, test performance at a parasite density of 200/µl is an important criterion. Imported malaria is defined as the presence of Plasmodium sp. in blood, whatever the parasite density. This requires a highly sensitive diagnostic test, as false negativity may lead to a life-threatening delay in treatment. In non-endemic areas, RDTs are mainly useful for confirming infection suggested by low microscopic parasite density, and for determining the species. Nonetheless, despite an NPV of 98% and a sensitivity of 96% obtained here with the PfHRP2-based tests, a negative RDT result does not rule out imported P. falciparum malaria [31]. Of note, all but one of the false-negative PfHRP2 test results in our study involved samples with parasite densities below 2000/µl.
The specificity and NPV of the pLDH-based tests were both 100%. Specificity higher than 90% has been reported in endemic areas [14]. In contrast, the sensitivity of the pLDH-based test was only 83% (95% CI, 79%–87%) in our study, while previous reports have shown sensitivities ranging from 85.1% to 99% among travelers [17,32] and from 85.6% to 98% in endemic countries [12,13], values lower than those of the PfHPR2-based tests for P. falciparum malaria, especially when parasite density was low (<2000/µl) [18].
Thus, RDT performance relies on the choice of monoclonal antibodies [33]. Better sensitivity would allow malaria to be ruled out by a negative RDT result, thus avoiding inappropriate presumptive treatment in areas where this therapeutic strategy is applied [17,34-36].
Although severe malaria, including imported malaria, is almost always due to P. falciparum (and occasionally P. vivax or P . knowlesi , depending on the country visited) [37], species identification can guide the choice of treatment and avoid costly investigations. In our study, 11% of cases were due to non-falciparum species (P. ovale, P. vivax and P. malariae), and no cases of P . knowlesi infection were diagnosed.
The RDTs tested here were unable to identify non-falciparum species, especially P. ovale and P. malariae, as previously reported [38-40]. However, the sensitivity of the PvLDH-based tests was 91% (95% CI, 74%–100%) for P. vivax, especially with the PfHRP2 test3 – pLDH test3 (Palutop 4+) assay. Few PvLDH-based RDTs are available, and they have been evaluated in only a handful of studies. Meena et al. reported 76.6% sensitivity and 98.1% specificity for the FalciVax test (Orchid) [41]. Another study reported higher sensitivity (93.4%) for the SD Malaria Antigen P.v. test [39]. WHO reported a wide range of sensitivities for P. vivax detection with specific PvLDH RDTs, ranging from 5.9% to 100% (94.3% for the Core Malaria Pan/Pv/Pf) at higher parasite densities (2000/µl) [18].
False-positive results, representing 3% in our study, may have several causes. Nearly half (12/26) of the samples with positive RDT results and negative blood smears corresponded to PCR-confirmed P. falciparum malaria; these patients received no specific monitoring but were treated with antimalarial drugs in case of high suspicion of malaria. These samples may have been from patients with a suspected relapse of malaria, in which case their false positivity would indicate the persistence of PfHRP2 antigen in the bloodstream after treatment [42]; alternatively, they would confirm that RDTs can diagnose malaria attacks earlier than microscopy. We were unable to test these hypotheses. Cross-reactivity due to self-antibodies such as rheumatoid factor, especially in RDTs in which the conjugate is an IgM antibody [43], is also possible, as the manufacturers of the tests studied here do not specify the conjugate isotypes, and we were unable to screen the samples for rheumatoid factor or HAMA (human antimouse antibodies). A possible impact of gametocytes on RDT specificity [44,45] could not be excluded either, as too few samples contained gametocytes and no asexual forms to draw firm conclusion.
The choice of blood smears as the reference diagnostic test may represent a limitation of our study, as PCR has been proposed as the gold standard [46]. However, clearance of plasmodium DNA from the bloodstream following antimalarial treatment should be further studied to support and validate this option [47].
False-negative results for P. falciparum were observed mainly with the PfLDH-based test. Some of these samples were from patients treated with antimalarial drugs before their inclusion in the study, as shown by plasma drug assays; however, these treatments were not declared by the patients concerned, and their timing could not be determined. Dead parasites were observed on microscopy, but PfLDH production could have been halted by therapy [5,26], possibly contributing to the good specificity of the pLDH test [48].
The low sensitivity of RDT tests may also related to low parasite density, which could also explain the false-negative results obtained with the PfHRP2 detection tests, although some parasites isolated in South America and Africa have been shown not to produce PfHRP2 [49,50]. In such cases PfLDH detection would be of interest [51].
In conclusion, the three PfHRP2-based RDTs tested here showed high sensitivity and acceptable specificity for the diagnosis of imported and uncomplicated P. falciparum malaria, and thus appear to be a reliable, rapid and simple first-line diagnostic option for this potentially life-threatening disease, particularly in emergency settings. However, RDTs alone cannot rule out malaria, meaning that negative results must be confirmed by microscopy or PCR, and patients must be kept under medical supervision until the result is obtained [52,53].
Supporting Information
STARD checklist for reporting of studies of diagnostic accuracy
(DOC)
Recommendations about obligations for researchers to the patient (in French).
(PDF)
Acceptance of the project by the Ethic Committee (in French).
(PDF)
Detailed protocol of the research (in French).
(PDF)
Summary of the research protocol.
(DOC)
Acknowledgments
We thank the patients and guardians, and the personnel of the study centers. We also thank Delphine Prieur (Unité de Recherche Clinique Paris Nord) for the implementation and monitoring of the study. We thank David Young for his editorial assistance.
Funding Statement
The study was supported by a grant from the French Ministry of Health (PHRC AOR06066). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. World Health Organization, WHO Global Malaria Programme World malaria report 2011. Available: http://www.who.int/malaria/world_malaria_report_2011/en/. Accessed 21 June 2012.
- 2. European Centre for Disease Prevention and Control (2010) Annual Epidemiological Report on Communicable Diseases in Europe 2010 Stockholm: European Centre for Disease Prevention and Control; . Available: http://ecdc.europa.eu/en/publications/publications/1011_sur_annual_epidemiological_report_on_communicable_diseases_in_europe.pdf . Accessed 21 June 2012 [Google Scholar]
- 3. Hall-Baker PA, Nieves E, Jajosky RA Summary of Notifiable Diseases — United States, 2008. (2010) MMWR Surveill Summ 57: 1-94. Avalaible: http://www.cdc.gov/mmwr/mmwr_nd/index.html . Accessed 21 June 2012 [Google Scholar]
- 4. Casalino E, Le Bras J, Chaussin F, Fichelle A, Bouvet E (2002) Predictive factors of malaria in travelers to areas where malaria is endemic. Arch Intern Med 162: 1625-1630. doi:10.1001/archinte.162.14.1625. PubMed: 12123407. [DOI] [PubMed] [Google Scholar]
- 5. Rosenthal PJ (2012) How do we best diagnose malaria in Africa. Am J Trop Med Hyg 86: 192-193. doi:10.4269/ajtmh.2012.11-0619. PubMed: 22302846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Moody A (2002) Rapid diagnostic tests for malaria parasites. Clin Microbiol Rev 15: 66-78. doi:10.1128/CMR.15.1.66-78.2002. PubMed: 11781267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Chilton D, Malik ANJ, Armstrong M, Kettelhut M, Parker-Williams J et al. (2006) Use of rapid diagnostic tests for diagnosis of malaria in the UK. J Clin Pathol 59: 862-866. doi:10.1136/jcp.2005.032904. PubMed: 16603648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Stauffer WM, Cartwright CP, Olson DA, Juni BA, Taylor CM et al. (2009) Diagnostic performance of rapid diagnostic tests versus blood smears for malaria in US clinical practice. Clin Infect Dis 49: 908-913. doi:10.1086/605436. PubMed: 19686072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Maltha J, Gillet P, Jacobs J; Maltha J, Gillet P, Jacobs J (2013) Malaria rapid diagnostic tests in travel medicine. Clin Microbiol Infect 19: 408-415. Epub 2013. Feb 1. doi:10.1111/1469-0691.12152. PubMed: 23373854. [DOI] [PubMed] [Google Scholar]
- 10. Abba K, Deeks JJ, Olliaro P, Naing CM, Jackson SM et al. (2011) Rapid diagnostic tests for diagnosing uncomplicated P. falciparum malaria in endemic countries. Cochrane Database Syst Rev 7: CD008122: CD008122 PubMed: 21735422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Maltha J, Gillet P, Jacobs J; Maltha J, Gillet P, Jacobs J (2012) Malaria rapid diagnostic tests in endemic settings. Clin Microbiol Infect 19: 399-407. doi:10.1111/1469-0691.12151. PubMed: 23438048. Epub: 2013. Feb 25 PubMed; : 23438048 [DOI] [PubMed] [Google Scholar]
- 12. Ashton RA, Kefyalew T, Tesfaye G, Counihan H, Yadeta D et al. (2010) Performance of three multi-species rapid diagnostic tests for diagnosis of Plasmodium falciparum and Plasmodium vivax malaria in Oromia Regional State, Ethiopia. Malar J 9: 297 Available: http://www.malariajournal.com/content/9/1/297 Accessed 21 June 2012. doi:10.1186/1475-2875-9-297. PubMed: 20979601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Hendriksen IC, Mtove G, Pedro AJ, Gomes E, Silamut K et al. (2011) Evaluation of a PfHRP2 and a pLDH-based rapid diagnostic test for the diagnosis of severe malaria in 2 populations of African children. Clin Infect Dis 52: 1100-1107. doi:10.1093/cid/cir143. PubMed: 21467015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Ratsimbasoa A, Randriamanantena A, Raherinjafy R, Rasoarilalao N, Ménard D (2007) Which malaria rapid test for Madagascar? Field and laboratory evaluation of three tests and expert microscopy of samples from suspected malaria patients in Madagascar. Am J Trop Med Hyg 76: 481-485. PubMed: 17360871. [PubMed] [Google Scholar]
- 15. Rossi IA, D’Acremont V, Prod’hom G, Genton B (2012) Safety of falciparum malaria diagnostic strategy based on rapid diagnostic tests in returning travellers and migrants: a retrospective study. Malar J 11: 377. doi:10.1186/1475-2875-11-377. PubMed: 23158019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Bronner U, Karlsson L, Evengård B; Bronner U, Karlsson L, Evengård B (2011) Evaluation of rapid diagnostic tests for malaria in Swedish travellers. APMIS 119: 88-92. Epub 2010. Nov 17. doi:10.1111/j.1600-0463.2010.02696.x. PubMed: 21208275. [DOI] [PubMed] [Google Scholar]
- 17. Marx A, Pewsner D, Egger M, Nüesch R, Bucher HC et al. (2005) Meta-analysis: accuracy of rapid tests for malaria in travellers returning from endemic areas. Ann Intern Med 142: 836-846. doi:10.7326/0003-4819-142-10-200505170-00009. PubMed: 15897534. [DOI] [PubMed] [Google Scholar]
- 18. World Health Organization Malaria rapid diagnostic test performance Summary results of WHO product testing of malaria RDTs: round 1-4 (2008-2012). Available: http://www.who.int/malaria/publications/rdtmalaria_summary.pdf. Accessed 02 January 2013.
- 19. Alunni-Perret V, Vandenbos F, Kechkekian A, Marty P, Legros F et al. (2010) Fatal cerebral malaria diagnosed after death in a French patient. Am J Forensic Med Pathol 31: 269-272. doi:10.1097/PAF.0b013e3181e2efe4. PubMed: 20508488. [DOI] [PubMed] [Google Scholar]
- 20. Hermans V, Monzote L, Van den Sande B, Mukadi P, Sopheak T et al. (2011) Assessment of the knowledge of graphical symbols labelled on malaria rapid diagnostic tests in four international settings. Malar J 10: 331. doi:10.1186/1475-2875-10-331. PubMed: 22047089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Van Gool T, van Wolfswinkel ME, Koelewijn R, van Thiel PP, Jacobs J et al. (2011) A simple and fast method to exclude high Plasmodium falciparum parasitaemia in travellers with imported malaria. Malar J 10: 300. doi:10.1186/1475-2875-10-300. PubMed: 21999570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Bossuyt PM, Reitsma JB, Bruns DE, Gatsonis CA, Glasziou PP et al. (2003) Towards complete and accurate reporting of studies of diagnostic accuracy: the STARD initiative. Clin Chem 49: 1-6. doi:10.1373/49.1.1. PubMed: 12507953. [DOI] [PubMed] [Google Scholar]
- 23. World Health Organization (February 2009) Manual microscopy. Quality assurance manual, version 1. Available: http://www.who.int/malaria/publications/atoz/mmicroscopy_qam/en/index.html. Accessed 21 June 2012.
- 24. Snounou G (1996) Detection and identification of the four malaria parasite species infecting humans by PCR amplification. Methods Mol Biol 50: 263-291. PubMed: 8751365. [DOI] [PubMed] [Google Scholar]
- 25. Singh B, Kim Sung L, Matusop A, Radhakrishnan A, Shamsul SS et al. (2004) A large focus of naturally acquired Plasmodium knowlesi infections in human beings. Lancet. 363: 1017-1024. doi:10.1016/S0140-6736(04)15836-4. PubMed: 15051281. [DOI] [PubMed] [Google Scholar]
- 26. Piper R, Lebras J, Wentworth L, Hunt-Cooke A, Houzé S et al. (1999) Immunocapture diagnostic assays for malaria using Plasmodium lactate dehydrogenase (pLDH). Am J Trop Med Hyg 60: 109-118. PubMed: 9988333. [DOI] [PubMed] [Google Scholar]
- 27. World Health Organization (6-8 October 2009) Parasitological confirmation of malaria diagnosis: WHO technical consultation, Geneva. Available: http://www.who.int/malaria/publications/atoz/9789241599412/en/index.html. Accessed 21 June 2012.
- 28. Altman DG (2000) Diagnostic tests. In: Altman DG, Machin D, Bryant TN, Gardner MJ. Statistics with confidence. 2nd ed. London: BMJ Publishing Group Books; pp. 105-119. [Google Scholar]
- 29. World Health Organization WHO Global Malaria Programme. Good practices for selecting and procuring rapid diagnostic tests for malaria. Available: http://www.who.int/malaria/publications/atoz/9789241501125/en/index.html. Accessed 21 June 2012.
- 30. Murray CK, Gasser RA, Magill AJ, Miller RS (2008) Update on rapid diagnostic testing for malaria. Clin Microbiol Rev 21: 97-110. doi:10.1128/CMR.00035-07. PubMed: 18202438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Askling HH, Bruneel F, Burchard G, Castelli F, Chiodini PL et al. (2012) Management of imported malaria in Europe. Malar J 11: 328. doi:10.1186/1475-2875-11-328. PubMed: 22985344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Van Dijk DP, Gillet P, Vlieghe E, Cnops L, van Esbroeck M et al. (2009) Evaluation of the Palutop+4 malaria rapid diagnostic test in a non-endemic setting. Malar J 8: 293 Available: http://www.malariajournal.com/content/8/1/293 Accessed 21 June 2012. doi:10.1186/1475-2875-8-293. PubMed: 20003378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Piper RC, Buchanan I, Ho Choi Y, Makler T (2011) Opportunities for improving pLDH-based malaria diagnostic tests. Malar J 10: 213 Available: http://www.malariajournal.com/content/10/1/213. Accessed 21 June 2012. doi:10.1186/1475-2875-10-213. PubMed: 21806821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Jelinek T, Grobusch MP, Nothdurft HD (2000) Use of dipstick tests for the rapid diagnosis of malaria in nonimmune travellers. J Travel Med 7: 175-179. PubMed: 11003728. [DOI] [PubMed] [Google Scholar]
- 35. Playford EG, Walker J (2002) Evaluation of the ICT malaria Pf/Pv and the Optimal rapid diagnostic tests for malaria in febrile returned travellers. J Clin Microbiol 40: 4166-4171. doi:10.1128/JCM.40.11.4166-4171.2002. PubMed: 12409392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. De Monbrison F, Gérome P, Chaulet JF, Wallon M, Picot S et al. (2004) Comparative diagnostic performance of two commercial rapid tests for malaria in a non-endemic area. Eur J Clin Microbiol Infect Dis 23: 784-786. doi:10.1007/s10096-004-1202-9. PubMed: 15452770. [DOI] [PubMed] [Google Scholar]
- 37. Douglas NM, Anstey NM, Angus BJ, Nosten F, Price RN (2010) Artemisinin combination therapy for vivax malaria. Lancet Infect Dis 10: 405-416. doi:10.1016/S1473-3099(10)70079-7. PubMed: 20510281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. El-Moamly AM (2007) Antigen capture immuno-chromatographic strip format in detecting parasite-specific lactate deshydrogenase to diagnos malaria in nonimmune patients. J Egypt Soc Parasitol 37: 1017-1030. PubMed: 18383801. [PubMed] [Google Scholar]
- 39. Van der Palen M, Gillet P, Bottieau E, Cnops L, Van Esbroeck M et al. (2009) Test characteristics of two rapid antigen detection tests (SD FK50 and SD FK60) for the diagnosis of malaria in returned travellers. Malar J 8: 90 Available: http://www.malariajournal.com/content/8/1/90 Accessed 21 June 2012. doi:10.1186/1475-2875-8-90. PubMed: 19416497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Murray CK, Bennett JW (2009) Rapid Diagnosis of Malaria. Interdiscip Perspect. Infect Dis: 415953 Available: http://www.ncbi.nlm.nih.gov/pubmed/19547702. Accessed 21 June 2012. [DOI] [PMC free article] [PubMed]
- 41. Meena M, Joshi D, Joshi R, Sridhar S, Waghdhare S et al. (2009) Accuracy of a multispecies rapid diagnostic test kit for detection of malarial parasite at the point of care in a low endemicity region. Trans R Soc Trop Med Hyg 103: 1237-1244. doi:10.1016/j.trstmh.2009.04.011. PubMed: 19477476. [DOI] [PubMed] [Google Scholar]
- 42. Iqbal J, Siddique A, Jameel M, Hira PR (2004) Persistent histidine-rich protein 2, parasite lactate dehydrogenase, and panmalarial antigen reactivity after clearance of Plasmodium falciparum monoinfection. J Clin Microbiol 42: 4237-4241. doi:10.1128/JCM.42.9.4237-4241.2004. PubMed: 15365017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Laferi H, Kandel K, Pichler H (1997) False positive dipstick test for malaria. N Engl J Med 337: 1635-1636. doi:10.1056/NEJM199711273372219. PubMed: 9411233. [DOI] [PubMed] [Google Scholar]
- 44. Mueller I, Betuela I, Ginny M, Reeder JC, Genton B (2007) The sensitivity of the Optimal rapid diagnostic test to the presence of Plasmodium falciparum gametocytes compromises its ability to monitor treatment outcomes in an area of Papua New Guinea in which malaria is endemic. J Clin Microbiol 45: 627-630. doi:10.1128/JCM.00816-06. PubMed: 17135432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Hayward RE, Sullivan DJ, Day KP (2000) Plasmodium falciparum: histidine-rich protein II is expressed during gametocyte development. Exp Parasitol 96: 139-146. doi:10.1006/expr.2000.4557. PubMed: 11162364. [DOI] [PubMed] [Google Scholar]
- 46. Singh N, Shukla MM, Shukla MK, Mehra RK, Sharma S et al. (2010) Field and laboratory comparative evaluation of rapid malaria diagnostic tests versus traditional and molecular techniques in India. Malar J 9: 191 Available: http://www.malariajournal.com/content/9/1/191 Accessed 21 June 2012. doi:10.1186/1475-2875-9-191. PubMed: 20602766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Bell D, Peeling RW (2006) Evaluation of rapid diagnostic tests: malaria. Nat Rev Microbiol Suppl: 34-40. [DOI] [PubMed] [Google Scholar]
- 48. Houzé S, Boly MD, Le Bras J, Deloron P, Faucher JF (2009) PfHRP2 and PfLDH antigen detection for monitoring the efficacy of artemisinin-based combination therapy (ACT) in the treatment of uncomplicated falciparum malaria. Malar J 8: 211 Available: http://www.malariajournal.com/content/8/1/211. Accessed 21 June 2012. doi:10.1186/1475-2875-8-211. PubMed: 19735557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Gamboa D, Ho MF, Bendezu J, Torres K, Chiodini PL et al. (2010) A large proportion of P. falciparum isolates in the Amazon region of Peru lack pfhrp2 and pfhrp3: implications for malaria rapid diagnostic tests. PLOS ONE 5: e8091 Available: http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0008091 Accessed 21 June 2012. doi:10.1371/journal.pone.0008091. PubMed: 20111602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Koita OA, Doumbo OK, Ouattara A, Tall LK, Konaré A et al. (2012) False negative rapid diagnostic tests for malaria and deletion of the histidine-rich repeat region of the hrp2 gene. Am J Trop Med Hyg 86: 194-198. doi:10.4269/ajtmh.2012.10-0665. PubMed: 22302847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Bendezu J, Rosas A, Grande T, Rodriguez H, Llanos-Cuentas A et al. (2010) Field evaluation of a rapid diagnostic test (Parascreen) for malaria diagnosis in the Peruvian Amazon. Malar J 9: 154 Available: http://www.malariajournal.com/content/9/1/154 Accessed 21 June 2012. doi:10.1186/1475-2875-9-154. PubMed: 20529273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. D’Acremont V, Malila A, Swai N, Tillya R, Kahama-Maro J et al. (2010) Withholding antimalarials in febrile children who have a negative result for a rapid diagnostic test. Clin Infect Dis 51: 506-511. doi:10.1086/655688. PubMed: 20642354. [DOI] [PubMed] [Google Scholar]
- 53. Björkman A, Mårtensson A (2010) Risks and benefits of targeted malaria treatment based on rapid diagnostic test results. Clin Infect Dis 51: 512-514. doi:10.1086/655689. PubMed: 20642355. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
STARD checklist for reporting of studies of diagnostic accuracy
(DOC)
Recommendations about obligations for researchers to the patient (in French).
(PDF)
Acceptance of the project by the Ethic Committee (in French).
(PDF)
Detailed protocol of the research (in French).
(PDF)
Summary of the research protocol.
(DOC)