Abstract
There are potential health risks associated with the use of early weaning to prevent mother‐to‐child transmission of human immunodeficiency virus (HIV) in resource‐poor settings. Our objective was to examine growth and nutrient inadequacies among a cohort of children weaned early. Children participating in the Breastfeeding Antiretrovirals and Nutrition (BAN) Study in Lilongwe, Malawi, had HIV‐infected mothers, were weaned at 6 months and fed LNS until 12 months. 40 HIV‐negative, BAN‐exited children were compared with 40 HIV‐negative, community children matched on age, gender and local health clinic. Nutrient intake was calculated from 24‐h dietary recalls collected from BAN‐exited children. Anthropometric measurements were collected from BAN‐exited and matched community children at 15–16 months, and 2 months later. Longitudinal random effects sex‐stratified models were used to evaluate anthropometric differences between the two groups. BAN‐exited children consumed adequate energy, protein and carbohydrates but inadequate amounts of fat. The prevalence of inadequate micronutrient intakes were: 46% for vitamin A; 20% for vitamin B6; 69% for folate; 13% for vitamin C; 19% for iron; 23% for zinc. Regarding growth, BAN‐exited girls gained weight at a significantly lower rate {0.02 g kg−1 per day [95% confidence interval (CI): 0.01, 0.03]} than their matched comparison [0.05 g kg−1 per day (95% CI: 0.03, 0.07)]; BAN girls grew significantly slower [0.73 cm month−1 (95% CI: 0.40,1.06)] than their matched comparison (1.55 cm month−1[95% CI: 0.98, 2.12]). Among this sample of BAN‐exited children, early weaning was associated with dietary deficiencies and girls experienced reduced growth velocity. In resource‐poor settings, HIV prevention programmes must ensure that breastfeeding stop only once a nutritionally adequate and safe diet without breast milk can be provided.
Keywords: LNS, early breastfeeding cessation, HIV, Malawi, child growth
Introduction
Human immunodeficiency virus (HIV)‐infected mothers living in resource‐poor settings are advised to exclusively breastfeed (EBF) from 0 to 6 months, take antiretroviral drugs during pregnancy and breastfeeding, introduce complementary foods at 6 months, and stop breastfeeding once a nutritionally adequate, affordable, feasible, acceptable, sustainable and safe replacement diet can be provided (World Health Organization 2006, 2009). The most recent WHO guidelines state ‘mothers known to be HIV‐infected (and whose infants are HIV uninfected or of unknown HIV status) should EBF their infants for the first 6 months of life, introducing appropriate complementary foods thereafter, and continue breastfeeding for the first 12 months of life. Breastfeeding should then only stop once a nutritionally adequate and safe diet without breast milk can be provided’ (World Health Organization 2009). Because breastfeeding is the most cost‐effective infant feeding strategy for food insecure families and supplies optimal nutrition and protection against common childhood infections (e.g. diarrhoea, pneumonia), early cessation is potentially dangerous for infants living in resource‐poor settings (Habicht et al. 1986; Victora et al. 1987). The WHO's global strategy on infant and young child feeding recommends the infants of HIV‐negative mothers be EBF for the first 6 months of life and continue breastfeeding for up to 2 years of age or beyond (World Health Organization 2002).
Malawian infants are traditionally fed a complementary diet based on maize porridge in addition to breast milk (Hotz & Gibson 2001; National Statistics Office [Malawi] and ORC Macro 2005; Corneli et al. 2007). This combination often cannot meet infant nutrient needs thus the traditional diet alone increases the risk of malnutrition (Dewey & Brown 2003; Dewey et al. 2004; Adu‐Afarwuah et al. 2007). Among low‐income families in Malawi, the addition of nutrient‐dense foods to the child diet is essential but is often limited by seasonality and cost (Chirwa 2009). Children are most susceptible to growth faltering during the first 2 years of life (Victora et al. 2010), and nutritional insult during this critical developmental window can have lifelong health consequences including increased risk of child mortality, short adult stature, reduced intellectual development and economic productivity and low offspring birthweight (Grantham‐McGregor et al. 2007; Black et al. 2008; Victora et al. 2008). Considering the potential health risks associated with early breastfeeding cessation, we aimed to evaluate the effects of using early breastfeeding cessation as an HIV prevention strategy in a resource‐poor setting by investigating dietary adequacy and growth after early weaning.
We evaluated dietary intakes among a sample of 15–18‐month‐old children after exiting the Breastfeeding Antiretrovirals and Nutrition (BAN) Study to determine the prevalence of nutrient inadequacies. To determine if BAN‐exited children exhibited significantly greater growth faltering, we also compared the anthropometric status of BAN‐exits (non‐breastfeeding) with a sample of self‐reported HIV‐negative, community controls matched on age, gender and local health clinic. This study was conducted to inform policy regarding best infant feeding practices where concerns over breastfeeding increasing the risk of HIV transmission must be balanced by the risks of malnutrition.
Key messages
-
•
Non‐breastfed children consumed inadequate amounts of fat and the prevalence of inadequate micronutrient intakes was: 46% for vitamin A; 20% for vitamin B6; 69% for folate; 13% for vitamin C; 19% for iron; 23% for zinc.
-
•
The dietary intakes of non‐breastfeeding children differed significantly by gender. Boys consumed 24% of total calories from fat and 64% from carbohydrate sources; girls consumed only 20% from fat calories and 69% from carbohydrate. Boys were 4 times as likely to receive milk products.
-
•
The matched community comparison sample of girls (95% breastfeeding) had significantly greater weight velocity and length velocity scores than the sample of BAN‐exited girls who stopped breastfeeding at 6 months.
-
•
Early breastfeeding cessation as an HIV prevention method may have trade‐offs in resource‐poor settings such as inadequate dietary intakes and reduced infant growth.
Materials and methods
The BAN Study
The BAN study was a randomised controlled clinical trial conducted in Malawi between 2004 and 2010. Two thousand three hundred sixty‐nine HIV‐infected mothers were assigned to two main study interventions: a two‐arm maternal nutritional intervention to promote maternal health, and a three‐arm antiretroviral intervention – with drugs given for up to 28 weeks to the mother, the infant or neither – to prevent HIV transmission during breastfeeding (van der Horst et al. 2009; Chasela et al. 2010). All mothers were counselled to EBF to age 24 weeks, and then rapidly transition to a replacement diet, achieving early breastfeeding cessation by 28 weeks. To reduce the risk of malnutrition following early breastfeeding cessation, mothers were provided with a lipid‐based nutrient supplement (LNS) made locally from full‐cream powdered milk, peanut butter, sugar, oil and micronutrients (Briend 2001). This breast milk substitute (LNS) was designed to fulfil infant micronutrient requirements and replace the energy (400 kcal) and protein (9.5 g) that would have been provided by breast milk (12 months) (van der Horst et al. 2009). This product was based on the ‘Plumpynut’ standard recipe intended for short‐term use among children with severe acute malnutrition but was used by the BAN Study because of the current paucity of alternative LNS formulations for long‐term use (World Health Organization/World Food Programme/UNICEF 2007). LNSs are bacteria resistant, do not require water for preparation, and can be consumed directly from the container without cooking, making its use safe even in poor hygienic conditions (Briend 2002). LNS was provided after weaning until infants exited the study at 48 weeks (12 months).
Between 24 and 48 weeks, mothers were taught how to feed their children an adequate diet without breast milk using a Complementary Feeding Counseling Script that emphasised the appropriate age for each food introduction, serving sizes, nutrient density and variety, feeding frequency, food safety, feeding during illness, and responsive feeding (Ferguson et al. 2009; Bentley et al. 2011). Specifically, mothers were instructed to practice responsive feeding; feed frequently with patience to ensure a maximum amount was consumed; prepare soft, thick porridge; enrich porridge with cooking oil, groundnut flour, sugar, honey, groundnuts, milk, soybeans, meat, or fish; ensure diet diversity to prevent micronutrient deficiencies; avoid sweets and soft drinks; and feed four to five meals and two to three snacks per day.
Follow‐up study
The data for this feasibility study were collected by observing a subsample of HIV‐infected mothers and their infants after exiting the BAN Study using a cohort design. When infants exited the BAN Study, they no longer received LNS. Of concern were the dietary intakes and nutritional status of non‐breastfed children after LNS supplementation ceased, and this formed the basis of the current study. In particular, nutrients of concern for the non‐breastfed child in urban Malawi include energy, protein, fat, vitamin A, vitamin B6, folate, vitamin C, calcium, iron and zinc (Dewey et al. 2004; National Statistics Office [Malawi] and ORC Macro 2005).
The feasibility study was conducted at Bwaila Hospital in central Lilongwe, Malawi between April 2008 and March 2009. This study evaluated the dietary intakes of a sample of children after exiting the BAN Study (BAN‐exits) when they were between 15 and 18 months of age to determine the prevalence of nutrient inadequacies and anthropometric status among non‐breastfed children in comparison with a matched sample of non‐participants from the community. BAN‐exited children were EBF to 6 months with no breast milk thereafter; received intensive face to face nutrition counselling and close medical attention to 12 months; and received the local standard of care after 12 months. The matched community children were EBF for less than 6 months but continued breastfeeding to 18 months; received the local standard of care regarding nutrition counselling and medical care up to 18 months.
BAN‐exited children were eligible to participate in this follow‐up study if they: were singletons at birth, carried to term (>36 weeks), completed the BAN Study to 12 months, HIV negative, available at 15–16 months of age for their first study visit, and willing to return 2 months later for a follow‐up visit. For each child in the BAN‐exited group, a non‐participant child from the community was recruited into a comparison group matched on age, gender and local health centre. If a matched non‐participant child did not return for their second visit, they were withdrawn from the sample and another child was chosen from the community, matched on age, gender and local health clinic to the already enrolled BAN‐exited child. The mothers of all matched community children reported themselves and their children to be HIV negative. Reports of HIV‐negative status were most likely correct because all mothers would have been informed about their status during the HIV counselling and testing procedures conducted at all urban antenatal clinics; mothers also submitted to two confidential interviews, each approximately 1‐h in length, and answered in depth questions regarding the child's medical history and food practices thus making dishonesty difficult to sustain.
The sample size of 40 children per group was determined from power calculations described in the anthropometrics section, below. Recruitment occurred at the same three urban health clinics as the BAN Study and continued until a minimum of 80 children completed the study. Mother‐child pairs were recruited in one of the following ways: at the BAN clinic following the exit interview, at a catchment clinic following HIV‐positive support group meetings, at a catchment clinic following healthy baby clinic visits, or using home locator maps. Compensation to participants included 600MK (US $4) for transport and 1 kg fortified corn soya‐blended flour for both study visits.
Dietary intake
Dietary intake data were collected from BAN‐exited children because they had stopped breastfeeding and total intake could be easily calculated; dietary intake data was not collected from the matched community children because they were still breastfeeding and funding was not available to quantify breast milk intake using precise estimates (i.e. doubly labelled water). Two 24‐h diet recalls were collected from each child within 1 week between 15 and 16 months of age, and again between 17 and 18 months of age, for a total of four recalls from each of the BAN‐exited children. Diet recalls were conducted by BAN nutrition officers. Mothers were prompted to recall what their child had eaten over the past 24 h using food models, standard household dishes and utensils, and recording recipes. Nutrient consumption was calculated using a Malawi food composition table (FCT) developed by Rosalind Gibson (Ferguson et al. 1995). Additional information was added from a Tanzanian FCT (Lukmanji et al. 2008), the United States Department of Agriculture (USDA) nutrient database (USDA National Nutrient Database for Standard Reference 2010), or directly from the manufacturer. The resulting database had the nutritional value of individual food items as well as mixed dishes based on standard recipes (e.g. nsima).
Anthropometrics
Anthropometrics were obtained from each child, once between 15 and 16 months and again between 17 and 18 months. The sample size was calculated using Malawi's 2004 demographic and health survey and the mean height‐for‐age z‐score [and respective standard deviation (SD)] for infants 15–18 months. A convenience sample size of 40 per group was feasible to recruit within our time frame and allowed us to be able to detect a 0.92 difference in mean z‐scores between samples, with 80% power and significance set at 10% (National Statistics Office [Malawi] and ORC Macro 2005). If significance had been set at 5% while keeping sample size and power parameters constant, we could have only detected a difference greater than 1.03 in mean z‐scores.
Two trained Malawian research assistants recorded recumbent length following standardised protocol using a paediatric length board (to the nearest 1 mm) made locally based on UNICEF model specifications and a calibrated Tanita infant scale (to the nearest 10 g) (Maternal Child and Health Bureau 2000). All measurements were made in triplicate and averaged. Length‐for‐age z‐scores (LAZ), weight‐for‐age z‐scores (WAZ) and weight‐for‐length z‐scores (WLZ) were calculated in Microsoft Excel using the LMS method (Box‐Cox power, L(t); median, M(t); and coefficient of variation, S(t)) and the WHO 2006 standards for breastfed children (Cole & Green 1992). Stunting, underweight and wasting were respectively defined as having an LAZ, WAZ or WLZ score less than 2 SD below the WHO median.
Background questionnaire
A background survey was administered to obtain data from each mother on age, marital status, education and occupation of both parents, number of household members, household socio‐economic indicators and food availability. Ethical approval for this study was obtained from the institutional review boards at the US Centers for Disease Control and Prevention, UNC‐Chapel Hill and the National Health Sciences Research Committee in Malawi. Participants in the original BAN Study had signed a consent form to be contacted after the study was completed.
Seasonality
The Malawian climate is characterised by four seasons: rainy (November to April), post‐rainy (April to May), cool (May to mid‐August) and hot (mid‐August to November). The rainy season is known locally as the ‘hungry season’ because food is limited during this time. For each BAN‐exited child, a matched child was measured within the same month; however, high attrition among the matched comparison extended their enrolment period and reduced seasonal overlap between the samples. Because a child's growth velocity is affected by season, a variable was created to measure percent exposure to the hungry season during the 2 months prior to each study visit. All regression models comparing growth outcomes controlled for exposure to the hungry season. Diet recalls were collected from BAN‐exited infants between April 2008 and December 2008 and did not greatly overlap the hungry season.
Data analysis
Nutrient intake was analysed only within the BAN‐exited sample, since the comparison group was still receiving breast milk. Nutrient intake distributions were calculated from the 24‐h dietary recalls. The prevalence of inadequacy for each nutrient, approximated from the proportion of children consuming less than the estimated average requirement (EAR) cut‐point (Institute of Medicine 1997, 1998, 2000, 2001, 2002/2005), was estimated using C‐SIDE (C‐SIDE, version 1.0, 1996; Department of Statistics, Iowa State University) (Dodd 1996; Guenther et al. 1997; Carriquiry 2003). C‐SIDE requires multiple dietary recalls per individual to reduce within‐individual day‐to‐day variation and enable an estimation of within‐ and between‐individual variation (Dodd 1996; Dodd et al. 2006). The EAR equivalent for energy is expected energy requirement (EER) and can be calculated for each individual (15–18 months) based on body weight [EER = (89 × weight [kg]−100) + 20 kcal]. For calcium, only an adequate intake (AI) level exists preventing the calculation of inadequacy prevalence. Total protein, carbohydrate and fat intakes were compared with the Acceptable Macronutrient Distribution Ranges (AMDR) (Institute of Medicine 2002/2005). Typical foods consumed and sources of certain vitamins were also examined using the dietary recalls.
We used Stata 10 (StataCorp LP, College Station, TX, USA), longitudinal random effects sex‐stratified models to evaluate group differences in WAZ, LAZ and WLZ. The linear regression models accounted for exposure to the hungry season, maternal age, parity, visit (at 15 vs. 17 months) and an interaction variable between visit number and group. Child's age was not included because the age range was small and groups were age‐matched. The models were selected using a priori variable selection, checks for collinearity and sequential removal of variables that did not statistically confound the association of interest; final models were checked using residual analysis. Parity was dropped because of its collinearity with maternal age. Based on the findings from the longitudinal models, we did further group comparisons of weight and length velocity between the two visits using one‐way analysis of variance (Lepage et al. 1996). The prevalence of stunting, underweight and wasting were calculated for each child sample in each age group; a chi‐squared test was used to determine if the differences in proportions were significant between the two groups using Stata 10.
Results
Subjects
A total of 99 children were recruited into the study: 81 children completed the study and 78 were included in the analysis (Fig. 1). Thirteen of the matched community children were lost to follow‐up between the first and second visit; a comparison of socioeconomic characteristics revealed no differences of practical importance between the final sample and those lost to follow‐up. Three children were excluded because of suspected measurement error because they had anthropometric z‐scores more than 4 SDs above or below the WHO median. A sensitivity analysis found their exclusion did not change the direction of the relationships but attenuated the growth velocity differences between groups in girls as well as boys. The BAN‐exited group and the matched community group each contained 39 children. Of the 39 matched community children, 37 (95%) were still breastfeeding at 15 months. None of the 39 BAN‐exited children were breastfed past 7 months.
Figure 1.
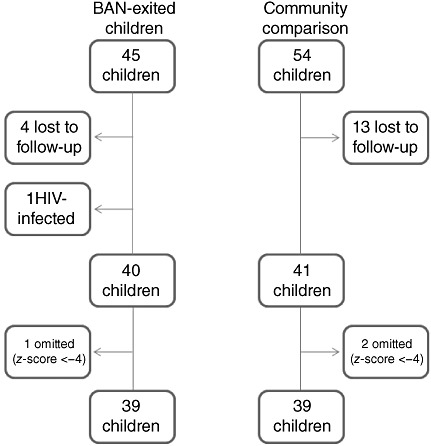
A flow chart illustrating subject enrolment during the feasibility study in Lilongwe, Malawi, April 2008 to March 2009. BAN, Breastfeeding Antiretrovirals and Nutrition Study.
Socio‐demographic characteristics
In both samples, most households were headed by males and had more than one child younger than 5 years. Selected socioeconomic and demographic characteristics of the sample are shown in Table 1. Three notable differences between groups were that the matched community mothers were younger, more likely to be married, and more were primiparous.
Table 1.
Demographic and socioeconomic characteristics of mothers participating in the Feasibility Study in Lilongwe, Malawi, 2008
| Breastfeeding Antiretrovirals and Nutrition Study‐exited sample | Matched community sample | |
|---|---|---|
| n = 39 | n = 39 | |
| Age in years (±standard deviation) | 28.8 (±5.9) | 23.6 (±3.8) |
| Marital status | ||
| Married | 75.6% | 93% |
| Separated | 17% | 7% |
| Divorced | 5% | 0 |
| Widowed | 2% | 0 |
| Education (years) | ||
| 0 | 2% | 0 |
| <8 | 39% | 46% |
| 8 | 27% | 27% |
| 8–12 | 15% | 10% |
| 12 | 17% | 17% |
| Occupation | ||
| Currently working | 45% | 39% |
| Self‐employed (non‐farming) | 30% | 24% |
| Husband's occupation | ||
| Salaried employment | 58% | 58% |
| Self‐employed (non‐farming) | 30% | 37% |
| Other (military, landlord) | 6% | 2.5% |
| Unemployed | 3% | 2.5% |
| Fertility | ||
| Proportion primiparous | 17% | 46% |
| Parity | 3.4 (±1.9) | 2.1 (±1.4) |
| Total number of living children | 2.9 (±1.4) | 2.0 (±1.3) |
| Number of children <5 years in household | 1.7 (±0.9) | 1.5 (±0.6) |
| Socio‐economic status | ||
| Refrigerator | 7% | 5% |
| Electricity | 22% | 12% |
| Personal water source | 29% | 17% |
| Traditional latrine with roof | 66% | 75% |
| Cement floor | 66% | 78% |
| Iron sheet roofing | 83% | 80% |
Nutrient adequacy
On average, the diet of the non‐breastfeeding BAN‐exited sample exceeded protein and carbohydrate requirements but was deficient in energy and fat (Table 2). Nutrient intakes at 15–16 months were similar to intakes at 17–18 months. Overall, energy intake was composed of carbohydrate (66%), protein (11%) and fat (22%). Approximately 65% of BAN‐exited children consumed energy‐deficient diets and 81% consumed an inadequate amount of fat (Table 2). Micronutrient intakes were not normally distributed (skewed right) and are therefore summarised by medians. Median intakes of vitamin B6, vitamin C, iron and zinc were above their respective EAR but median vitamin A and folate intake fell below. The prevalence of inadequacy was 46% for vitamin A, 20% for vitamin B6, 69% for folate, 13% for vitamin C, 19% for iron and 23% for zinc (Table 2). Mean calcium intake (172 mg) was below the AI and thus there is a reasonable likelihood that dietary intakes were inadequate (Table 2).
Table 2.
Macronutrient and select micronutrient intakes from Breastfeeding Antiretrovirals and Nutrition Study‐exited child participants in the feasibility study in Lilongwe, Malawi, 2008.
| Nutrient | Expected total intake* | Median intake | Mean (SD) intake | Inadequacy prevalence (adjusted) |
|---|---|---|---|---|
| Energy | EER = (89 × weight[kg]−100) + 20 kcal | 91.5% | 92% (±40%) | 65% |
| Protein | EAR = 0.87 g kg−1 per day | 250% | 221% (±105%) | 0.5% |
| Protein AMDR | 5–20% of energy | 11.2% | 11.5% (±2%) | 2.3% |
| Fat AMDR | 30–40% of energy | 21.7% | 22% (±6%) | 80.8% |
| Carbohydrate AMDR | 45–65% of energy | 66.7% | 66% (±7%) | 6.6% |
| Vitamin A | EAR = 210 µg day−1 | 143 µg | 235 µg (±228) | 46% |
| Vitamin B6 | EAR = 0.4 mg day−1 | 0.58 mg | 0.7 mg (±0.58) | 20% |
| Folate | EAR = 120 µg day−1 | 113 µg | 104 µg (±67) | 69% |
| Vitamin C | EAR = 13 mg day−1 | 25 mg | 38 mg (±39) | 13% |
| Iron | EAR = 3.0 mg day−1 | 4.7 mg | 4.4 mg (±2.4) | 19% |
| Zinc | EAR = 2.5 mg day−1 | 3.5 mg | 3.5 mg (±1.7) | 23% |
| Calcium | AI = 500 mg day−1 | 137 mg | 172 mg (±150) | – |
AMDR, Acceptable Macronutrient Distribution Ranges; EAR, estimated average requirement; EER, expected energy requirement; IOM, Institute of Medicine. *Expected total nutrient intake, derived from the IOM, was used as the comparison referent. Expected intakes of energy, protein, fat, and carbohydrate were obtained from IOM (2002/2005); expected intakes of vitamin A, zinc, and iron were obtained from IOM (2001); expected intake of vitamin C was obtained from IOM (2000); expected intakes of vitamin B6 and folate were obtained from IOM (1998); expected intake of calcium was obtained from IOM (1997).
In addition to the general diet description described earlier, we further explored if dietary differences existed between genders. The only difference was that girls consumed significantly less fat and more carbohydrate as a proportion of their total energy intake than boys (Student's t‐test, P < 0.05). Boys consumed 24% of total calories from fat and 64% from carbohydrate sources; girls consumed only 20% from fat calories and 69% from carbohydrate.
Typical food consumption
Diets were based primarily on maize flour and consumed in the form of porridge and nsima. Nsima is a stiff porridge made from maize flour and eaten with a relish, an accompanying vegetable, bean, fish or meat side dish. Together, porridge and nsima provided 60% of total energy. On average, children received 2.25 servings of porridge (200 g) per day providing 350 kcal (53 g carbohydrate, 12 g protein, 10 g fat). The typical recipe included maize flour, water, sugar and salt but the frequent addition of soya (26%) and groundnut flour (33%) increased protein and fat consumption. Child intakes were deficient in fat but surpassed the protein recommendation (Table 2). Protein was mainly consumed from porridge (10.5 g day−1) because of the presence of soya flour, groundnut flour and large quantity of maize flour.
Food choices differed by gender. Boys were 3.9 times as likely to receive fresh or powdered milk, 1.2 times as likely to receive animal products, 1.2 times as likely to consume vegetable oil in their mixed dishes. Girls were 1.44 times as likely to consume fruits. The frequency of groundnut and soya flour consumption was similar between genders. Overall, boys were fed slightly more frequently and slightly larger portion sizes.
Sources of vitamin A, folate, fat
Dietary intakes were deficient in fat, vitamin A and folate. Primary sources of vitamin A in the diet were leafy green vegetables and tomatoes (vitamin A precursors). Less frequent were animal products (e.g. liver, eggs and milk) and sweet potatoes. Folate was mostly consumed from groundnut flour and soya flour in porridge. Additional sources included fruits, other vegetables, beans and leafy green vegetables but small portion sizes limited intake. Fat was largely consumed from groundnut flour and soya flour.
Growth comparison
Both BAN‐exited and matched comparison children were below the WHO reference median but still of moderately healthy weight. Table 3 lists the mean WAZ scores of each subgroup adjusted for exposure to the hungry season, maternal age, visit number, and an interaction variable between visit number and BAN status; when the matching criteria were included as covariates in the model (e.g. child age, sex) the results were unchanged. The interaction term was significant (P = 0.076) implying that BAN‐exited girls had a different pattern of change in their WAZ scores over time. Weight velocity significantly differed between the groups among girls; BAN‐exited girls (n = 16) gained an average of 0.02 g kg−1 per day and the matched community girls (n = 18) gained an average of 0.05 g kg−1 per day (P < 0.05) (Table 3). The WAZ scores of BAN‐exited boys (n = 23) and community boys (n = 21) did not significantly differ; velocity of weight gain among BAN‐exited boys was 0.02 g kg−1 per day and community boys was 0.03 g kg−1 per day (Table 3).
Table 3.
Adjusted anthropometric means for BAN‐exited and matched community children in the feasibility study in Lilongwe, Malawi, April 2008 to March 2009.
| Group | Sample size (n) | WAZ* | LAZ † | WLZ ‡ |
|---|---|---|---|---|
| 15–16 months | ||||
| BAN‐exited girls | 16 | −0.41 (−0.98, 0.17) | −1.34 (−1.88, −0.79) | 0.30 (−0.36, 0.95) |
| Matched girls | 18 | −0.41 (−0.95, 0.13) | −1.52 (−2.03, −1.01) | 0.36 (−0.25, 0.97) |
| BAN‐exited boys | 23 | −0.10 (−0.58, 0.38) | −1.49 (−2.02, −0.95) | 0.80 (0.20, 1.39) |
| Matched boys | 21 | −0.56 (−1.1, −0.05) | −2.03 (−2.60, −1.47) | 0.50 (−0.12, 1.12) |
| 17–18 months | ||||
| BAN‐exited girls | 16 | −0.41 (−0.99, 0.16) | −1.53 (−2.07, −0.98) | 0.43 (−0.22, 1.08) |
| Matched girls | 18 | 0.07 (−0.48, 0.61) | −1.30 (−1.82, −0.79) | 0.90 (0.28, 1.52) |
| BAN‐exited boys | 23 | −0.01 (−0.50, 0.48) | −1.34 (−1.89, −0.80) | 0.83 (0.22, 1.44) |
| Matched boys | 21 | −0.53 (−1.05, 0.00) | −1.73 (−2.32, −1.15) | 0.36 (−0.29, 1.02) |
| Velocity | Weight gain § (g kg−1 per day) | Height gain ¶ (cm month−1) | ||
| BAN‐exited girls | 16 | 0.02 (0.01, 0.03)** | 0.73 (0.40, 1.06)** | |
| Matched girls | 18 | 0.05 (0.03, 0.07)** | 1.55 (0.98, 2.12)** | |
| BAN‐exited boys | 23 | 0.02 (0.01, 0.04) | 1.19 (0.79, 1.58) | |
| Matched boys | 21 | 0.03 (0.02, 0.04) | 1.47 (0.97, 1.98) |
BAN, Breastfeeding Antiretrovirals and Nutrition Study; WAZ, weight‐for‐age z‐score; LAZ, length for age z‐score; WLZ, weight‐for‐length z‐score. *WAZ, †LAZ and ‡WLZ scores were adjusted on exposure to the hungry season, maternal age, visit number and the interaction between visit number and BAN status. §Weight gain and ¶height gain were adjusted to the number of days the child was followed. **Significant difference (P < 0.05).
Both groups were on average shorter than the WHO 2006 reference median. Table 3 lists the mean LAZ scores of each subgroup adjusted for exposure to the hungry season, maternal age, visit number and an interaction variable between visit number and BAN status; when the matching criteria were included as covariates in the model (e.g. child age, sex) the results were unchanged. Overall, the LAZ and length velocity scores of BAN‐exited (n = 23) and matched community (n = 21) boys did not differ (Table 3). The LAZ scores of BAN‐exited (n = 16) and matched community (n = 18) girls did not differ, but the group by visit interaction variable was significant, suggesting BAN‐exited girls did worse over time (P = 0.076). This was further illustrated by the lower length velocity of BAN‐exited girls (P < 0.05) (Table 3).
The combination of short stature and moderately healthy weight status resulted in WLZ scores above the WHO median. Adjusted mean WLZ scores are listed in Table 3. WLZ scores did not significantly differ between girl samples or boy samples.
Malnutrition
At 15–16 months, 36% (14/39) of BAN‐exits and 49% (19/39) of community children were stunted; at 17–18 months, 31% (12/39) of BAN‐exits were stunted and 31% (12/39) of community children were stunted. At 15–16 months, 5% (2/39) of BAN‐exits and 13% (5/39) of the community comparison were underweight; at 17–18 months, 5% (2/39) and 3% (1/39) of BAN‐exited and community children were underweight, respectively. While none of the BAN‐exited children were wasted, 5% (2/39) of community children were wasted at 15–16 months and 5% (2/39) were wasted at 17–18 months. Table 4 illustrates these differences were not significant.
Table 4.
The prevalence of malnutrition among BAN‐exited and matched community children in the feasibility study in Lilongwe, Malawi, April 2008 to March 2009.
| BAN‐exited children | Matched children | P‐value | |
|---|---|---|---|
| % Stunted (15–16 m) | 36 | 49 | 0.25 |
| % Stunted (17–18 m) | 31 | 31 | 1.00 |
| % Underweight (15–16 m) | 5 | 13 | 0.24 |
| % Underweight (17–18 m) | 5 | 3 | 0.56 |
| % Wasted (15–16 m) | 0 | 5 | 0.15 |
| % Wasted (17–18 m) | 0 | 5 | 0.15 |
BAN, Breastfeeding Antiretrovirals and Nutrition Study.
Discussion
This study provides insight into the diets of 15–18‐month‐old children, living in a resource‐poor setting within Malawi, who were weaned from the breast at 6 months and who received LNS through 12 months as a breast milk substitute. After children stopped receiving LNS, their diet was maize‐based but enriched with purchased ingredients, such as groundnuts and soya. Overall, the diet without breast milk was rich in carbohydrate and protein but deficient in energy, fat and micronutrients. Among this sample, early breastfeeding cessation was associated with low growth velocity scores between 15 and 18 months when no substitute was provided after 12 months. In comparison with the matched community sample of breastfed girls, BAN‐exited girls had significantly reduced growth velocity outcomes over the 2‐month follow‐up.
The results of the present study are consistent with dietary modelling studies that reported difficulties constructing affordable child diets from traditional food sources to provide sufficient energy, 30% of calories from fat, and adequate amounts of vitamin A, thiamine, riboflavin, vitamin B6, folate, vitamin C, calcium, iron, and zinc (i.e. ‘problem nutrients’) (Dewey et al. 2004). A previous complementary diet study in Malawi found that young children were fed mainly maize flour, few animal‐source foods, and inadequate amounts of energy, calcium, iron and zinc (Ferguson et al. 1993; Hotz & Gibson 2001). The Malawi demographic and health survey reported that at least one serving of meat, fish, shellfish, poultry or eggs was consumed by children 16–19‐month‐old less than once a day, whether the child was breastfeeding or not, but small servings of fruits and vegetables were eaten three to four times a day. Also consistently reported were high rates of childhood anaemia and subclinical vitamin A deficiency, thus our findings of inadequate intakes of iron, vitamin C, folate and vitamin A are not surprising (National Statistics Office [Malawi] and ORC Macro 2005; Ministry of Health and Population [Malawi] 2003).
BAN‐exited children received intensive nutrition counselling and medical care to 12 months, which may have given them an advantage over the matched community comparison. Stunting is an indicator of chronic malnutrition and our data suggest that this sample of BAN‐exited children was less stunted at 15–16 months than the community comparison sample. Wasting is indicative of acute malnutrition and a greater proportion of the community comparison was measured during the hungry season, which may explain their greater wasting prevalence. While the prevalence rates of stunting and underweight improved by 18% and 10% among the matched comparison, respectively, little to no improvement was observed among the BAN‐exited sample. Although the difference between study groups was not statistically significant, these data could suggest that BAN‐exited children may have less ability to recover from malnutrition and thus may have greater prevalence malnutrition as they progress through childhood. The prevalence of stunting, wasting and underweight within these two child samples was lower than the national average for this age group in Malawi (National Statistics Office [Malawi] and ORC Macro 2005). Previous studies in Malawi found that height and weight velocity vary according to a seasonal pattern after 12 months of age (Maleta et al. 2003) possibly because of greater dependence on the external environment. Children typically experience negative weight velocity during the hungry season (December to January), peak weight gains following the harvest (May, June and July) and greatest height gains 3 months after the harvest (August to October).
In this region of the world, there is little evidence of gender preference; the anthropometric z‐scores of boys and girls under 5 years are similar within the Malawi Demographic and Health Survey (National Statistics Office [Malawi] and ORC Macro 2005). Both the length and weight velocity of BAN‐exited girls fell significantly below that of community girls, and the same difference was not observed between boy samples. We suspect the growth of BAN‐exited girls was negatively impacted by social factors such as traditional differences in diet quality (Ferguson et al. 1995). Families were four times as likely to purchase milk products for male children as for female children; suggesting households with boys allocated their resources to ensure boys continued receiving milk in the absence of breast milk; this finding may be restricted to low‐income, non‐breastfeeding, HIV‐infected populations. Girls consumed significantly less fat and more carbohydrate. While neither gender achieved the AMDR for fat, girls fell further from the 30–40% recommendation, which may have contributed to their reduced growth outcomes (Lifshitz & Moses 1989).
There is limited prior evidence of son preference in sub‐Saharan Africa (Kabeer 2003; Klasen & Wink 2003; Basu & de Jong 2006), especially Malawi, where vaccination coverage is equal, the percent of sick children receiving adequate health care does not differ by gender, and the under‐five child mortality rate is greater among boys than girls (166 vs. 149 per 1000) (National Statistics Office [Malawi] and ORC Macro 2005). Thus our findings could be the consequence of a small sample size, indicate differences between HIV‐infected families and the general population, or more likely be attributable to factors currently unknown. Poor growth outcomes among girls weaned early are particularly problematic because the deleterious effects of malnutrition can be passed on to their future offspring (Victora et al. 2008).
In Malawi, families place great value on complementary foods and believe they are essential for weight gain and growth (Parker et al. 2011) even when the staple food is not energy or nutrient dense. If families preferentially fed male children a greater ratio of complementary foods to breast milk, male growth would be impeded if the complementary foods were inadequate. While BAN‐exited children received enriched home‐made complementary foods between 12–15 months, matched community children likely received standard complementary foods in addition to breast milk. Because the growth rate of community comparison boys was similar to that of non‐breastfeeding boys, it is possible the matched community boys were primarily fed complementary foods.
All BAN‐exited children included in this study were HIV‐negative therefore we cannot attribute their lack of growth to HIV infection. We hypothesise that the community comparison had better growth outcomes because most were still breastfeeding between 15–18 months (mean intended duration = 28 months) and were buffered against seasonal changes. Non‐breastfeeding children are more vulnerable to dietary deficiencies and are not protected against infections by maternal antibodies thus their recovery from infection is impaired and could negatively impact their growth and recovery from growth faltering.
Breast milk provides a superior diet, contains anti‐infective properties, promotes gastrointestinal track development, a healthy immune system and lower morbidity incidence (Duncan et al. 1993; Goldman 1993; Dewey et al. 1995; Cushing et al. 1998; Hanson 1998; Bachrach et al. 2003). Early breastfeeding cessation as an HIV‐prevention method has been associated with increased risk of serious gastroenteritis (Onyango‐Makumbi et al. 2009), gastroenteritis‐associated mortality (Kafulafula et al. 2010), growth faltering (Arpadi et al. 2009), nutrient deficiencies (Lunney et al. 2008) and failed to improve the rate of HIV‐free survival (Kuhn et al. 2008). Infants who participated in the BAN Study were all indirectly exposed to antiretroviral medicines during labour, delivery and the first 2 weeks of life, a third received antiretrovirals (ARVs) directly during the 6‐month breastfeeding period and a third ingested their mother's ARVs indirectly through breast milk to 6 months (van der Horst et al. 2009). While maternal HIV infection is documented to impair prenatal growth (Bulterys et al. 1994) there is conflicting evidence on whether or not it negatively impacts postnatal growth (Hankin et al. 2005; Paul et al. 2005). Exposure to ARVs during pregnancy and delivery does not affect postnatal growth (Briand et al. 2006) and research to date has not evaluated if postnatal ARVs impact infant growth. It is possible that HIV‐exposed infants do not develop sufficient nutrient stores because of the combination of inadequate maternal intake or bodily stores and the increased energy and nutrient needs of HIV infection (Grinspoon et al. 1998; 2001a, 2001b). Such inadequate infant stores would be quickly depleted after birth if breast milk was only consumed for a short time and replaced by inadequate complementary foods, resulting in growth faltering.
The greatest limitation to our study was the accuracy and comparability of recumbent length measurements. Children consistently prove difficult to measure using a length board and we were forced to omit three because of suspected measurement error in an attempt to improve validity. Although the rapid decline in stunting prevalence among matched community children could suggest error, the measuring assistants were not changed over the course of the study thus any measurement error would have been random and thus could only have mitigated the differences between samples. While we recognise 13 of the matched community sample were lost to follow‐up, no socioeconomic differences were found between those lost and those retained and thus it is unlikely that such attrition introduced bias. Differences in the timing of participation between the two groups did create a systematic bias and limited the conclusions we could draw by comparing the growth of BAN‐exited children to community children. However, we do suspect that the difference in growth velocities documented here would have been further amplified if the entire matched community sample been measured during the harvest season rather than the hungry season. In retrospect, this study would have benefited from measuring morbidity incidence among both samples to examine if BAN‐exited children experienced more illness and if this difference impacted growth outcomes. While the lack of dietary information from the matched community comparison group and confirmed HIV‐negative status are also limitations, we must still recognise the possibility that other biases and unknown confounders may exist between the groups.
Previous studies among Malawian children found mean intakes derived from the 24‐h recall were within ±10% of values calculated from weighed food records (Ferguson et al. 1989). The 24‐h recall underestimates actual intake because of memory error reporting snack consumption, the use of average recipes and underestimations of the quantity consumed from the main meal (1989, 1995). In comparison, the present study was less affected by such errors because BAN Study mothers had 12 months experience reporting 24‐h dietary recalls, we did not use standard recipes for dishes with varying ingredients, and mothers were prompted to remember accurate intakes using standard household measures and food models.
To our knowledge, this is the first study to investigate growth outcomes 1 year after performing early breastfeeding cessation as an HIV prevention method, and in comparison with a matched community sample. By conducting this assessment, we were able to investigate the dietary deficits of non‐breastfeeding children and confirm that the traditional child diet was in fact deficient in macronutrients and micronutrients, the most concerning of which were fat, vitamin A and folate. This information will help tailor future complementary feeding guides used to counsel HIV‐infected mothers choosing to not breastfeed in resource‐poor settings.
This study has raised concerns about the safety of breastfeeding cessation as an HIV prevention method. The most recent revisions to the WHO HIV and infant feeding guidelines now advise mothers to continue breastfeeding for the first 12 months of life, with receipt of maternal or infant antiretroviral medications, and perform breastfeeding cessation only once a nutritionally adequate and safe diet without breast milk can be provided (World Health Organization 2009). Because early breastfeeding cessation appears to have trade‐offs in resource‐poor settings, we believe it is essential that HIV prevention programmes provide dietary counselling to participating families to ensure they understand what components are necessary to provide an adequate child diet and help them evaluate if they are capable of providing those components. This information will help families to make an informed decision regarding whether or not they should stop breastfeeding at 12 months.
Source of funding
This research was funded by the Prevention Research Centers Special Interest Project SIP 13–01 U48‐CCU409660‐09 and SIP 26–04 U48‐DP000059‐01, Centers for Disease Control and Prevention; supported by the NIAID P30‐AI50410 UNC Center for AIDS Research; DHHS/NIH/FIC 2‐D43 Tw01039‐06 AIDS International Training and Research Program and the National Institutes of Health, National Institute of Child Health and Human Development (NICHD, 1R03HD057775‐01), Nutrition Obesity Research Center (NIH DK56350) and Fogarty International Clinical Research Fellows Program at Vanderbilt (R24 TW007988).
Conflict of interest statement
None of the above authors have any financial or personal interest in the above results and have no conflicts of interest to declare.
Contributions
MEP, MEB, LA, EGP, CMV, DJJ and SE conceived the research question and designed the study; MEP, CC, DK, AS, CM, MT and FM conducted research; MEP and LA analysed data; MEP, MEB and LA drafted the initial version, and EGP, CMV, DJJ, SE, CC, DK, AS, CM, MT and FM each performed multiple revisions to produce the final version. All authors read and approved the final manuscript.
Acknowledgements
This follow‐up study would not have been possible without the dedicated efforts of Tasila Zulu, Chrissie Chilima, Chalimba Lusewa, Tonse Gauti and Joyce Mhone. We would also like to thank Valerie Flax for her detailed review and editorial suggestions. We are extremely grateful for the dedicated efforts of the BAN Study Team at University of North Carolina Chapel Hill, Centers for Disease Control and Prevention, Atlanta, and UNC Project team in Lilongwe including: Linda Adair, Yusuf Ahmed, Mounir Ait‐Khaled, Sandra Albrecht, Shrikant Bangdiwala, Ronald Bayer, Margaret Bentley, Brian Bramson, Emily Bobrow, Nicola Boyle, Sal Butera, Charles Chasela, Charity Chavula, Joseph Chimerang'ambe, Maggie Chigwenembe, Maria Chikasema, Norah Chikhungu, David Chilongozi, Grace Chiudzu, Lenesi Chome, Anne Cole, Amanda Corbett, Amy Corneli, Ann Duerr, Henry Eliya, Sascha Ellington, Joseph Eron, Sherry Farr, Yvonne Owens Ferguson, Susan Fiscus, Shannon Galvin, Laura Guay, Chad Heilig, Irving Hoffman, Elizabeth Hooten, Mina Hosseinipour, Michael Hudgens, Stacy Hurst, Lisa Hyde, Denise Jamieson, George Joaki (deceased), David Jones, Zebrone Kacheche, Esmie Kamanga, Gift Kamanga, Coxcilly Kampani, Portia Kamthunzi, Deborah Kamwendo, Cecilia Kanyama, Angela Kashuba, Damson Kathyola, Dumbani Kayira, Peter Kazembe, Caroline C. King, Rodney Knight, Athena P. Kourtis, Robert Krysiak, Jacob Kumwenda, Edde Loeliger, Misheck Luhanga, Victor Madhlopa, Maganizo Majawa, Alice Maida, Cheryl Marcus, Francis Martinson, Navdeep Thoofer, Chrissie Matiki (deceased), Douglas Mayers, Isabel Mayuni, Marita McDonough, Joyce Meme, Ceppie Merry, Khama Mita, Chimwemwe Mkomawanthu, Gertrude Mndala, Ibrahim Mndala, Agnes Moses, Albans Msika, Wezi Msungama, Beatrice Mtimuni, Jane Muita, Noel Mumba, Bonface Musis, Charles Mwansambo, Gerald Mwapasa, Jacqueline Nkhoma, Richard Pendame, Ellen Piwoz, Byron Raines, Zane Ramdas, John Rublein, Mairin Ryan, Ian Sanne, Christopher Sellers, Diane Shugars, Dorothy Sichali, Wendy Snowden, Alice Soko, Allison Spensley, Jean‐Marc Steens, Gerald Tegha, Martin Tembo, Roshan Thomas, Hsiao‐Chuan Tien, Beth Tohill, Charles van der Horst, Esther Waalberg, Jeffrey Wiener, Cathy Wilfert, Patricia Wiyo, Innocent Zgambo Chifundo Zimba. We would also like to thank Project Peanut Butter and Valid Nutrition who produced the LNS. Finally and most especially, all the women and infants that have agreed to participate in the study.
The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the US Centers for Disease Control and Prevention.
References
- Adu‐Afarwuah S., Lartey A., Brown K.H., Zlotkin S., Briend A. & Dewey K.G. (2007) Randomized comparison of 3 types of micronutrient supplements for home fortification of complementary foods in Ghana: effects on growth and motor development. American Journal of Clinical Nutrition 86, 412–420. [DOI] [PubMed] [Google Scholar]
- Arpadi S., Fawzy A., Aldrovandi G.M., Kankasa C., Sinkala M., Mwiya M. et al (2009) Growth faltering due to breastfeeding cessation in uninfected children born to HIV‐infected mothers in Zambia. American Journal of Clinical Nutrition 90, 344–353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bachrach V., Schwarz E. & Bachrach L.R. (2003) Breastfeeding and the risk of hospitalization for respiratory disease in infancy: a meta‐analysis. Archives of Pediatrics & Adolescent Medicine 157, 237–243. [DOI] [PubMed] [Google Scholar]
- Basu D. & de Jong R. (2006) Son Preference and Gender Inequality . Available at: http://www.econ.ohio-state.edu/dejong/gender2.pdf.
- Bentley M.E., Wasser H.M. & Creed‐Kanashiro H.M. (2011) A review of responsive feeding and child undernutrition in low and middle countries. Journal of Nutrition 141, 490–494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Black R.E., Allen L.H., Bhutta Z.A., Caulfield L.E., de Onis M., Ezzati M. et al (2008) Maternal and child undernutrition: global and regional exposures and health consequences. Lancet 371, 243–260. [DOI] [PubMed] [Google Scholar]
- Briand N., Le Coer S., Traisathit P., Karnchanamayul V., Hansudewechakul R., Ngampiyasakul C. et al (2006) Growth of human immunodeficiency virus‐uninfected children exposed to perinatal zidovudine for the prevention of mother‐to‐child human immunodeficiency virus transmission. Pediatric Infectious Disease Journal 25, 325–332. [DOI] [PubMed] [Google Scholar]
- Briend A. (2001) Highly nutrient‐dense spreads: a new approach to delivering multiple micronutrients to high‐risk groups. British Journal of Nutrition 85 (Suppl. 2), S175–S179. [PubMed] [Google Scholar]
- Briend A. (2002) Possible use of spreads as a FOODlet for improving the diets of infants and young children. Food and Nutrition Bulletin 23, 239–243. [DOI] [PubMed] [Google Scholar]
- Bulterys M., Chao A., Munyemana S., Kurawige J.B., Nawrocki P., Habimana P. et al (1994) Maternal human immunodeficiency virus 1 infection and intrauterine growth: a prospective cohort study in Butare, Rwanda. Pediatric Infectious Disease Journal 13, 94–100. [DOI] [PubMed] [Google Scholar]
- Carriquiry A.L. (2003) Estimation of usual intake distributions of nutrients and foods. Journal of Nutrition 133, 601S–608S. [DOI] [PubMed] [Google Scholar]
- Chasela C., Hudgens M.G., Jamieson D.J., Kayira D., Hosseinipour M., Kourtis A.P. et al (2010) Maternal antiretrovirals or infant nevirapine to reduce HIV‐1 transmission. New England Journal of Medicine 362, 2271–2281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chirwa E. (2009) The 2007‐2008 Food Price Swing: impact and Policies in Malawi Policies for Good Economic Management of Food Price Swings in Africa. FAO Trade and Markets Division Available at: http://www.fao.org/es/esc/foodpriceswing/papers/Price%20Swings_Malawi.pdf.
- Cole T.J. & Green P.J. (1992) Smoothing reference centile curves: the LMS method and penalized likelihood. Statistics in Medicine 11, 1305–1319. [DOI] [PubMed] [Google Scholar]
- Corneli A.L., Piwoz E.G., Bentley M.E., Moses A., Nkhoma J.R., Tohill B.C. et al (2007) Involving communities in the design of clinical trial protocols: the BAN Study in Lilongwe, Malawi. Contemporary Clinical Trials 28, 59–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cushing A.H., Samet J.M., Lambert W.E., Skipper B.J., Hunt W.C., Young S.A. et al (1998) Breastfeeding reduces risk of respiratory illness in infants. American Journal of Epidemiology 147, 863–870. [DOI] [PubMed] [Google Scholar]
- Dewey K.G. & Brown K.H. (2003) Update on technical issues concerning complementary feeding of young children in developing countries and implications for intervention programs. Food and Nutrition Bulletin 24, 5–28. [DOI] [PubMed] [Google Scholar]
- Dewey K.G., Heinig M.J. & Nommsen‐Rivers L.A. (1995) Differences in morbidity between breast‐fed and formula‐fed infants. Journal of Pediatrics 126, 696–702. [DOI] [PubMed] [Google Scholar]
- Dewey K.G., Cohen R.J., Rollins N.C. & Informal Working Group on Feeding of Nonbreastfed Children (2004) WHO technical background paper: feeding of nonbreastfed children from 6 to 24 months of age in developing countries. Food and Nutrition Bulletin 25, 377–402. [DOI] [PubMed] [Google Scholar]
- Dodd K.W. (1996) A user's guide to C‐SIDE: Software for intake distribution estimation version 1.0. Ames, IA: Center for Agriculture and Rural Development, Iowa State University.
- Dodd K.W., Guenther P.M., Freedman L.S., Subar A.F., Kipnis V., Midthune D. et al (2006) Statistical methods for estimating usual intake of nutrients and foods: a review of the theory. Journal of the American Dietetic Association 106, 1640–1650. [DOI] [PubMed] [Google Scholar]
- Duncan B., Ey J., Holberg C.J., Wright A.L., Martinez F.D. & Taussig L.M. (1993) Exclusive breast‐feeding for at least 4 months protects against otitis media. Pediatrics 91, 867–872. [PubMed] [Google Scholar]
- Ferguson E.L., Gibson R.S., Ounpuu S. & Sabry J.H. (1989) The validity of the 24 hour recall for estimating the energy and selected nutrient intakes of a group of rural Malawian preschool children. Ecology of Food and Nutrition 23, 273–285. [Google Scholar]
- Ferguson E.L., Gibson R.S., Opare‐Obisaw C., Ounpuu S., Thompson L.U. & Lehrfeld J. (1993) The zinc nutriture of preschool children living in 2 African countries. Journal of Nutrition 123, 1487–1496. [DOI] [PubMed] [Google Scholar]
- Ferguson E.L., Gadowsky S.L., Huddle J.M., Cullinan T.R., Lehrfeld J. & Gibson R.S. (1995) An interactive 24‐h recall technique for assessing the adequacy of trace mineral intakes of rural Malawian women; its advantages and limitations. European Journal of Clinical Nutrition 49, 565–578. [PubMed] [Google Scholar]
- Ferguson Y.O., Eng E., Bentley M., Sandelowski M., Steckler A., Randall‐David E. et al (2009) Evaluating nurses' implementation of an infant‐feeding counseling protocol for HIV‐infected mothers: the Ban Study in Lilongwe, Malawi. AIDS Education and Prevention 21, 141–155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friis H., Gomo E., Koestel P., Ndhlovu P., Nyazema N., Krarup H. et al (2001a) HIV and other predictors of serum folate, serum ferritin, and hemoglobin in pregnancy: a cross‐sectional study in Zimbabwe. American Journal of Clinical Nutrition 73, 1066–1073. [DOI] [PubMed] [Google Scholar]
- Friis H., Gomo E., Koestel P., Ndhlovu P., Nyazema N., Krarup H. et al (2001b) HIV and other predictors of serum beta‐carotene and retinol in pregnancy: a cross‐sectional study in Zimbabwe. American Journal of Clinical Nutrition 73, 1058–1065. [DOI] [PubMed] [Google Scholar]
- Goldman A.S. (1993) The immune system of human milk: antimicrobial, antiinflammatory and immunomodulating properties [review]. Pediatric Infectious Disease Journal 12, 664–672. [DOI] [PubMed] [Google Scholar]
- Grantham‐McGregor S., Cheung Y.B., Cueto S., Glewwe P., Richter L. & Strupp B. (2007) Developmental potential in the first 5 years for children in developing countries. Lancet 369, 60–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grinspoon S., Corcoran C., Miller K., Wang E., Hubbard J., Schoenfeld D. et al (1998) Determinants of increased energy expenditure in HIV‐infected women. American Journal of Clinical Nutrition 68, 720–725. [DOI] [PubMed] [Google Scholar]
- Guenther P.M., Kott P.S. & Carriquiry A.L. (1997) Development of an approach for estimating usual nutrient intake distributions at the population level. Journal of Nutrition 127, 1106–1112. [DOI] [PubMed] [Google Scholar]
- Habicht J.P., DaVanzo J. & Butz W.P. (1986) Does breastfeeding really save lives, or are apparent benefits due to biases? American Journal of Epidemiology 123, 279–290. [DOI] [PubMed] [Google Scholar]
- Hankin C., Thorne C., Newell M.L. & Collaborative E. (2005) Does exposure to antiretroviral therapy affect growth in the first 18 months of life in uninfected children born to HIV‐infected women? Journal of Acquired Immune Deficiency Syndromes 40, 364–370. [DOI] [PubMed] [Google Scholar]
- Hanson L.A. (1998) Breastfeeding provides passive and likely long‐lasting active immunity. Annals of Allergy, Asthma & Immunology 81, 523–537. [DOI] [PubMed] [Google Scholar]
- van der Horst C., Chasela C., Ahmed Y., Hoffman I., Hosseinipour M., Knight R. et al (2009) Modifications of a large HIV prevention clinical trial to fit changing realities: a case study of the Breastfeeding, Antiretroviral, and Nutrition (BAN) protocol in Lilongwe, Malawi. Contemporary Clinical Trials 30, 24–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hotz C. & Gibson R.S. (2001) Complementary feeding practices and dietary intakes from complementary foods amongst weanlings in rural Malawi. European Journal of Clinical Nutrition 55, 841–849. [DOI] [PubMed] [Google Scholar]
- Institute of Medicine (IOM) (1997) Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride. National Academy Press: Washington DC. [PubMed] [Google Scholar]
- Institute of Medicine (IOM) (1998) Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. National Academy Press: Washington DC. [PubMed] [Google Scholar]
- Institute of Medicine (IOM) (2000) Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. National Academy Press: Washington DC. [PubMed] [Google Scholar]
- Institute of Medicine (IOM) (2001) Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc. National Academy Press: Washington DC. [PubMed] [Google Scholar]
- Institute of Medicine (IOM) (2002/2005) Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids. National Academy Press: Washington DC. [DOI] [PubMed] [Google Scholar]
- Kabeer N. (2003) Gender mainstreaming in poverty eradication and the millennium development goals: A Handbook for Policy‐makers and Other Stakeholders. Commonwealth Secretariat/IDRC/CIDA: London. [Google Scholar]
- Kafulafula G., Hoover D.R., Taha T.E., Thigpen M., Li Q., Fowler M.G., Kumwenda N.I. et al (2010) Frequency of gastroenteritis and gastroenteritis‐associated mortality with early weaning in HIV‐1‐uninfected children born to HIV‐infected women in Malawi. Journal of Acquired Immune Deficiency Syndromes 53, 6–13. [DOI] [PubMed] [Google Scholar]
- Klasen S. & Wink C. (2003) Missing women: revisiting the debate. Feminist Economics 9, 263–299. [Google Scholar]
- Kuhn L., Aldrovandi G.M., Sinkala M., Kankasa C., Semrau K., Mwiya M. et al (2008) Effects of early, abrupt weaning on HIV‐free survival of children in Zambia. New England Journal of Medicine 359, 130–141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lepage P., Msellati P., Hitimana D.G., Bazubagira A., Van Goethem C., Simonon A. et al (1996) Growth of human immunodeficiency type 1‐infected and uninfected children: a prospective cohort study in Kigali, Rwanda, 1988 to 1993. Pediatric Infectious Disease Journal 15, 479–485. [DOI] [PubMed] [Google Scholar]
- Lifshitz F. & Moses N. (1989) Growth failure. A complication of dietary treatment of hypercholesterolemia. American Journal of Diseases of Children 143, 537–542. [PubMed] [Google Scholar]
- Lukmanji Z., Hertzmark E., Mlingi N., Assey V., Ndossi G. & Fawzi W. (2008) Tanzania Food Composition Tables MUHAS‐ TFNC, HSPH, Dar es Salaam, Tanzania. Available at: http://www.hsph.harvard.edu/nutritionsource/files/tanzania-food-composition-tables.pdf.
- Lunney K.M., Jenkins A.L., Tavengwa N.V., Majo F., Chidhanguro D., Iliff P. et al (2008) HIV‐positive poor women may stop breast‐feeding early to protect their infants from HIV infection although available replacement diets are grossly inadequate. Journal of Nutrition 138, 351–357. [DOI] [PubMed] [Google Scholar]
- Maleta K., Virtanen S.M., Espo M., Kulmala T. & Ashorn P. (2003) Seasonality of growth and the relationship between weight and height gain in children under three years of age in rural Malawi. Acta Paediatrica 92, 491–497. [DOI] [PubMed] [Google Scholar]
- Maternal Child and Health Bureau (2000) Accurately weighing and measuring: technique: US Department of Health and Human Services, Health Resources and Services Administration, Maternal and Child health Bureau. Available from: http://depts.washington.edu/growth.
- Ministry of Health and Population [Malawi] (2003) Report of the National Micronutrient Survey 2001. Ministry of Health and Population: Lilongwe, Malawi.
- Onyango‐Makumbi C., Bagenda D., Mwatha A., Omer S.B., Musoke P., Mmiro F. et al (2009) Early weaning of HIV‐exposed uninfected infants and risk of serious gastroenteritis: findings from two perinatal HIV prevention trials in Kampala, Uganda. Journal of Acquired Immune Deficiency Syndromes 53, 20–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker M.E., Bentley M.E., Chasela C., Adair L., Piwoz E.G., Jamieson D.J. et al (2011) The acceptance and feasibility of replacement feeding at 6 months as an HIV prevention method in Lilongwe, Malawi: results from the BAN Study. AIDS Education and Prevention 23, 281–2f95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paul M.E., Chantry C.J., Read J.S., Frederick M.M., Lu M., Pitt J. et al (2005) Morbidity and mortality during the first two years of life among uninfected children born to human immunodeficiency virus type 1‐infected women: the women and infants transmission study. Pediatric Infectious Disease Journal 24, 46–56. [DOI] [PubMed] [Google Scholar]
- National Statistics Office [Malawi] & ORC Macro (2005) Malawi Demographic and Health Survey 2004. NSO and ORC Macro: Calverton, Maryland: Available from http://www.measuredhs.com
- USDA National Nutrient Database for Standard Reference (2010) Release 22 United States Department of Agriculture.
- Victora C.G., Adair L., Fall C., Hallal P.C., Martorell R., Richter L. et al (2008) Maternal and child undernutrition: consequences for adult health and human capital. Lancet 371, 340–357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Victora C.G., de Onis M., Hallal P.C., Blössner M. & Shrimpton R. (2010) Worldwide timing of growth faltering: revisiting implications for interventions. Pediatrics 125, e473–e480. [DOI] [PubMed] [Google Scholar]
- Victora C.G., Smith P.G., Vaughan J.P., Nobre L.C., Lombardi C., Teixeira A.M. et al (1987) Evidence for protection by breast‐feeding against infant deaths from infectious diseases in Brazil. Lancet 330, 319–322. [DOI] [PubMed] [Google Scholar]
- World Health Organization (WHO)/World Food Programme (WFP)/UNICEF (2007) Community‐based Management of Severe Acute Malnutrition A Joint Statement by the World Health Organization, the World Food Programme, the United Nations System Standing Committee on Nutrition and the United Nations Children's Fund. Available at: http://www.who.int/nutrition/publications/severemalnutrition/9789280641479/en/index.html.
- World Health Organization (WHO) (2002) Infant and Young Child Nutrition Fifty‐fifth World Health Assembly. http://apps.who.int/gb/archive/pdf_files/WHA55/ea5515.pdf.
- World Health Organization (WHO) (2006) WHO HIV and infant feeding technical consultation. Consensus Statement. Geneva, Switzerland.
- World Health Organization (WHO) (2009) New WHO Recommendations: Infant feeding in the context of HIV. Key messages. Geneva, Switzerland.


