Summary
Research on HIV infection and sexual behaviour in sub-Saharan Africa typically focuses on individuals aged 15-49 under the assumption that both become less relevant for older individuals. We test this assumption using data from rural Malawi to compare sexual behaviour and HIV infection for individuals aged 15-49 with individuals aged 50-64 and 65+. Although general declines with age were observed, levels of sexual activity and HIV remained considerable: 26.7% and 73.8% of women and men aged 65+ reported having sex in the last year; men's average number of sexual partners remained above 1; and HIV prevalence is significantly higher for men aged 50-64 (8.9%) than men aged 15-49 (4.1%). We conclude that older populations are relevant to studies of sexual behaviour and HIV risk. Their importance is likely to increase as access to ARVs in Africa increases. We recommend inclusion of adults over 49 in African HIV/AIDS research and prevention efforts.
Introduction
Although HIV prevalence data in sub-Saharan Africa (SSA) are typically presented only for individuals aged 15-49, recent research indicates that the level of HIV infection in populations over 50 is likely to be not-insubstantial.1,2 This is expected to reflect both the survival of adults infected earlier in life, and HIV infection acquired later in life. In addition to current levels of HIV infection at older ages, the increasing availability of antiretroviral therapy in SSA is expected to further increase prevalence. Despite the importance of HIV infection among older adults, with a few exceptions,3-7 research and policy related to the HIV epidemic in SSA continues to focus primarily on adults aged 15-49 and children infected during birth or via breastfeeding, in part reflecting the availability of data. Large sample population-based data on older adults' HIV prevalence or HIV risk behaviour in high prevalence settings like SSA have generally been lacking.
The small body of research in SSA suggests that levels and nature of sexual activity probably do not vary dramatically by age. Data from five cohorts in Uganda, Zimbabwe and South Africa indicates that the majority of ‘younger old’ men and a considerable minority of women aged 40-60 years remained sexually active.8 Data from ‘older old’ adults in Southwestern Nigeria shows that around a quarter of adults aged 65-102 had extra-marital sex at least once since turning 65.9 Furthermore, several studies have shown that older adults are less likely to use condoms than younger age groups.9,10 Using survey data collected across nine sites in West, East and Southern Africa, Negin et al. identified lower awareness and knowledge of HIV prevention measures among adults over 50 than those aged 25-49 years,5 mirroring findings using Demographic and Health Survey (DHS) data for men aged 50-64 from 39 counties in the region11 and the findings of earlier qualitative research among older men and women.3,4,7 Data collected from older adults living in two urban slums in Kenya's capital city identified low levels of perceived infection risk, especially among women.12
Efforts to prevent further new infection among adults in mid- and later life will require information about both, age patterns of HIV prevalence and sexual behaviour, the most likely mode of HIV transmission. Very few studies, however, have considered HIV infection or the extent and nature of sexual activity after age 49 years in this region. Of those that have, most have limitations. For example, many use relatively small sample sizes of older populations,13,14 which reduces statistical power. Others are limited to ‘younger old’ populations aged 50-65.8 Finally, several studies use indirect estimates of HIV infection (such as projections or verbal autopsy and case report data15,16) rather than actual HIV biomarkers.
In this research, we contribute to the sparse literature on HIV risk for populations over 50 in SSA by examining HIV prevalence, risk behaviour, and risk perception for older men and women living in Malawi. In doing so, we address many of the limitations of previous research. We use rich data from rural Malawi (with substantial sample size aged 65 and over) and several measures of HIV infection and risk.
Methods
We use data from the Malawi Longitudinal Study of Families and Health (MLSFH) (previously known as the Malawi Diffusion and Ideational Change Project (MDICP)), a longitudinal panel survey in rural Malawi. The first wave of MLSFH took place in 1998, for a sample of 1,541 ever-married women aged 15-49 and 1,065 of their spouses. The sample of men and women are relatively evenly spread across the three districts in which MLSFH conducts data collection, one in each region of Malawi. Follow-up surveys were conducted biennially. Between 2004 and 2008 MLSFH offered HIV testing and test results to all respondents, and response rates for HIV testing were over 90% for all years testing was offered. More detailed description and assessment of the MLSFH data and sample is presented elsewhere.17-19 The MLSFH study was approved by the University of Pennsylvania, and by the National Health Sciences Review Council in Malawi.
Several features of MLSFH make the data well-suited for our analysis. First, in 2008 MLSFH added a sample of approximately 800 parents of MLSFH respondents, many of which are aged over 49. To draw this sample, MLSFH asked respondents in 2006 to list characteristics of their parents, which MLSFH used in 2008 to include all parents who resided in a MLSFH sample village. In addition to this sample of parents, a substantial percentage of the original 1998 MLSFH sample is now aged over 49. Furthermore, the 2010 MLSFH survey contains extensive data on sexual behaviour and HIV infection. The survey refusal rate was less than 3% in all waves. Therefore, the large MLSFH sample of all-age adults, with no upper age limit, provides a rare opportunity to investigate HIV infection and sexual behaviour in older populations in SSA. Background characteristics for the sample are presented in Table 1.
Table 1. Background Characteristics, 2010 MLSFH Men and Women.
| Women | Men Percentages (N) | Total | |
|---|---|---|---|
| N= | 2182 | 1537 | 3719 |
| 50+ years | 29.5% (644) | 33.4% (513) | 31.1% (1157) |
| 65+ years | 11.7 (256) | 13.5 (209) | 12.5 (465) |
| Education | |||
| No education | 27.2% (594) | 12.7% (195) | 21.2% (789) |
| Primary | 63.2 (1379) | 63.9 (982) | 63.5 (2361) |
| Secondary or higher | 9.6 (209) | 23.4 (360) | 15.3 (569) |
| Region of residence | |||
| Central | 32.6% (711) | 32.0% (492) | 32.3% (1203) |
| South | 34.9 (762) | 37.1 (570) | 35.9 (1332) |
| North | 32.5 (709) | 30.9 (475) | 31.8 (1184) |
| Marital characteristics | |||
| Currently married | 77.1% (1683) | 87.1% (1339) | 81.3% (3022 |
| Never married | 1.2 (26) | 7.6 (116) | 3.8 (142) |
| Divorced or separated | 9.7 (213) | 3.4 (52) | 7.1 (265) |
| Widowed | 11.9 (260) | 2.0 (30) | 7.8 (290) |
| 2008 HIV positive | 7.0% (104) | 4.7% (46) | 6.1% (150) |
| HIV prevalence by age group | |||
| 15-19 | 0.0% (0) | 0.0% (0) | 0.0% (0) |
| 20-29 | 5.9 (21) | 2.2 (5) | 4.4 (26) |
| 30-39 | 10.6 (39) | 2.6 (5) | 7.9 (44) |
| 40-49 | 8.9 (27) | 7.8 (16) | 8.5 (43) |
| 50-59 | 7.2 (15) | 10.7 (17) | 8.7 (32) |
| 60-69 | 0.9 (1) | 3.0 (3) | 1.9 (4) |
| 70-79 | 1.1 (1) | 0.0 (0) | 0.7 (1) |
| 80+ | 0.0 (0) | 0.0 (0) | 0.0 (0) |
Note: Not all respondents in the sample were tested for HIV in 2008. Due to refusals and some respondents not found in 2008 but found in 2010, 68% of women (N=1481) and 64% of men (N=985) in the sample were tested for HIV.
It is important to note that the MLSFH sample was not designed to be representative of rural Malawi. Despite this, comparisons of MLSFH sample characteristics of the rural population with nationally representative Malawi Demographic and Health Surveys (MDHS) showed that basic characteristics of MLSFH respondents closely matched the rural population in MDHS.19 Due to sample age restrictions of MDHS (which includes only women 15-49 and men 15-54), the elderly population of interest in this paper was not compared with the MDHS rural population.
Analysis
We conduct our analysis in three steps. First, we examine whether there are differences by age group in HIV prevalence, risk behaviours, and perceptions. We use chi-squared and t-tests to compare HIV prevalence, sexual behaviour and HIV risk perception for (1) men and women aged 15 to 49 years with those aged 50 to 64, and (2) those aged 15 to 64 years with the population aged 65 and older.
Secondly, we run multivariate regressions to identify differences in sexual behaviour, while controlling for background characteristics, like marital status, region of residence, and level of education. Specific dependent variables include HIV prevalence, recent sexual activity, number of sexual partners, suspected spousal sexual partners, and worry of HIV infection To measure differences by age group, we include a three-category age variable in which age 15-49 is the reference category, and ages (1) 50-64 and (2) 65 and older are each compared to ages 15-49.
Finally, in addition to examining differences in HIV prevalence and sexual behaviour by age group, we use non parametric weighted regressions to show patterns by age. These regressions smooth the sexual behaviours and HIV prevalence over the ages included in the MLSFH survey, facilitating a better understanding of age patterns and the identification of populations relevant for HIV and sexual behaviour data collection on this basis.
Results
As shown in Table 1, the age structure of the MLSFH sample includes a substantial proportion of individuals over age 49. Approximately one third of men and women are age 50 years or older (N=1157), and 12% (256) of women and 14% (209) of men are age 65 years or older. There are few differences in age structure by gender (not shown here): slightly more women are 30-39 years old, and more men are 50 years and older. 56% of women aged 50+ were currently married compared to 86% of younger women, and 93% of men aged 50+ were currently married compared to 84% of younger men.
The overall HIV prevalence for respondents interviewed in 2010 was approximately 6.1%; 7.0% (104) for women and 4.7% (46) of men tested HIV positive. Not all respondents in the sample were tested for HIV in 2008. Due to refusals and some respondents not found in 2008 but found in 2010, 68% of women (N=1481) and 64% of men (N=985) in the sample were tested for HIV. Since MLSFH did not collect HIV biomarkers in 2010, we use 2008 HIV biomarkers in this analysis. This limitation is not expected to dramatically affect our results, since previous analysis found that the HIV incidence rate among MLSFH respondents was very low (0.7 per 100 person years).20 As a result, using the 2008 MLSFH HIV data is likely to lead to a small underestimate of HIV prevalence in this population. Finally, since individuals with HIV aged 50 and older may have been infected before age 50, this research considers HIV prevalence rather than incidence.
Differences in HIV Prevalence, Sexual Behaviour and Perceptions by Age
Bivariate results in Table 2 (Panel A, top) show important differences by gender in age patterns of HIV prevalence. While HIV prevalence is higher among women aged 15-49 (8.3%) than women age 50 to 64 (5.4%), the difference is not significant at the (p<0.10 level). HIV prevalence is significantly higher for men aged between 50 and 64than those aged 15-49 (8.9% compared to 4.1%). HIV prevalence is, however, significantly lower for both men and women aged 65 and older than individuals of reproductive ages (15-49).
Table 2. Differences in HIV prevalence and sexual behaviour by age for 2010 MLSFH men and women.
| Women | Men | |||||||
|---|---|---|---|---|---|---|---|---|
| Panel A: Bivariate comparison of HIV prevalence and sexual behaviour by age category | ||||||||
| Age Group | 15-49 | 50-64 | 65+ | 15-49 | 50-64 | 65+ | ||
| N= | 1538 | 388 | 256 | 1024 | 304 | 209 | ||
| HIV positive | 8.3% | 5.4% | 1.3%** | 4.1% | 8.9%** | 0.7%** | ||
| Had sex in past year | 89.4% | 49.6%** | 26.7%** | 92.0% | 83.8%** | 73.8%** | ||
| More than one sexual partner in past year | 1.9% | 1.0% | 0.9% | 19.9% | 18.9% | 14.1% | ||
| Number sexual partners in past year | ||||||||
| 0 | 7.2% | 26.5%** | 64.9%** | 5.4% | 6.4% | 11.8%** | ||
| 1 | 91.0% | 72.4%** | 34.7%** | 74.7% | 71.8% | 74.1% | ||
| 2+ | 1.8% | 1.1% | 0.4% | 19.9% | 21.8% | 14.1%* | ||
| Suspected number of spouse's partners in past year | ||||||||
| 0 | 8.0% | 16.7%** | 36.7%** | 11.4% | 14.2% | 28.6%** | ||
| 1 | 58.8% | 52.7% | 44.9%** | 86.4% | 83.4% | 66.8%** | ||
| 2+ | 33.2% | 30.6% | 18.4%** | 2.2% | 2.4% | 4.6% | ||
| Worried a lot about HIV infection | 27.7% | 14.4%** | 8.0%** | 23.3% | 15.0%** | 16.2%* | ||
|
| ||||||||
| Panel B: Multivariate comparison of HIV prevalence and sexual behaviour by age category | ||||||||
| 50-64 | 65+ | 50-64 | 65+ | |||||
| Odds Ratio/Coef | 95% CI | Odds Ratio/Coef | 95% CI | Odds Ratio/Coef | 95% CI | Odds Ratio/Coef | 95% CI | |
|
| ||||||||
| HIV prevalence a | 0.47* | (0.26,0.86) | 0.07** | (0.02,0.29) | 2.01* | (1.06,3.80) | 0.14 | (0.02,1.05) |
| Had sex in past year a | 0.23** | (0.16,0.33) | 0.08** | (0.05,0.12) | 0.39** | (0.22,0.67) | 0.11** | (0.06,0.18) |
| Number sexual partners in past year b | -0.07** | (-0.12,-0.02) | -0.23** | (-0.30,-0.16) | -0.09 | (-0.21,0.02) | -0.19* | (-0.33,-0.04) |
| Suspected number of spouse's partners in past year b | -0.04 | (-0.26,0.18) | -0.56** | (-0.90,-0.22) | -0.04 | (-0.11,0.04) | -0.14** | (-0.22,-0.05) |
| Worried a lot about HIV infection a | 0.58** | (0.43,0.78) | 0.24** | (0.14,0.39) | 0.57** | (0.39,0.82) | 0.71 | (0.46,1.09) |
| More than one sexual partner in past yeara | 0.70 | (0.24,2.05) | 0.66 | (0.14,3.17) | 1.18 | (0.85,1.64) | 0.74 | (0.46,1.17) |
Notes: bivariate and multivariate both compare older age groups with the 15-49 age group; p-values significant at
1% level,
5% level;
regressions run are
=logistic,
=OLS; all multivariate models control for region of residence, level of education, and marital status
Both men and women reported declines in sexual activity by age. Individuals aged 50 and older are less likely to have had sex in the past 12 months than individuals aged 15-49. Almost half of the female sample (N=192) reported having recent sex at ages 50-64 and just over a quarter reported recent sex at ages 65 and older (N=68). For men, 83.8% (N=255) reported recent sex at ages 50-64 and almost three quarters reported recent sex at ages 65 and older (N=154). There were no significant differences in the number of sexual partners between men aged 15-49 and 50-64, but women aged 50-64 generally had fewer sexual partners than those aged 15-49. Both men and women aged 65 and older generally had fewer sexual partners than those aged 15-49. The likelihood of having more than one sexual partner did not significantly differ by age group. Women were less likely to have multiple sexual partners than men: 1.1% and 0.4% of women aged 50-64 and 65+ had 2 or more sexual partners, and 21.8% and 14.1% of men aged 50-64 and 65+ had 2 or more sexual partners.
In addition to reporting their number of sexual partners in the past year, respondents were asked to estimate the number of sexual partners their spouse had in the past year. There are few significant differences in suspected number of spouse's partners between men and women aged 15-49 and those aged 50-64. Men and women aged 65 and older however did suspect significantly fewer spousal partners than adults aged 15-49. Men and women over 50 are additionally less worried about HIV infection than those aged 15-49 years.
Multivariate results (Table 2, Panel B, bottom) comparing individuals aged 50-64 and 65 and older with the population aged 15-49 (controlling for level of education, marital status, and region of residence) are largely similar to the bivariate results above. HIV prevalence is significantly less for women in older age groups than the 15-49 age groups (0.47 at 50-64 and 0.07 at 65+), but is significantly higher among men aged 50-64 than men aged 15-49 (2.01). Older men and women in rural Malawi were less likely to have had sex in the past year, were less worried about HIV infection, had fewer sexual partners in the past year (with the exception of men aged 50-64), and those aged 65 and older suspected that their spouse had fewer partners in the past year. Similarly, there were no significant differences in self-reported multiple sexual partnerships by age group. Therefore the differences in HIV prevalence and sexual behaviour persist even after controlling for the fact that older Malawians are less likely to be currently married.
HIV Prevalence, Sexual Behaviour and Risk Perception over the Life Course
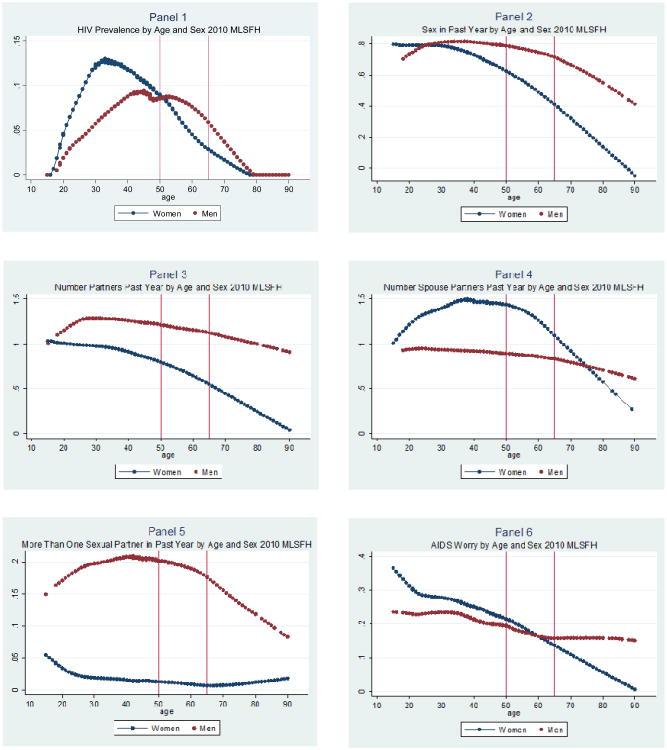
Next we present more detailed age patterns of sexual behaviour and HIV prevalence for rural Malawians. To do so, we discuss results for the non-parametric graphs of sexual behaviour by age and gender. Figure 1 has six panels that display HIV infection and risk behaviour by each age included in the sample.
Figure 1. Lowess Smoothing of HIV Prevalence and Sexual Risk Behaviour by Age, 2010 MLSFH Men and Women.
Panel 1 in Figure 1 shows the likelihood of HIV infection over each age for women and men. As the results above suggest, there are different patterns for men and women. Likelihood of HIV infection for women peaks in the early 30s and declines steadily thereafter. For men the greatest likelihood of HIV infection is more than 10 years later, and doesn't decline until the mid-50s. For both men and women, likelihood of HIV infection remains considerable over age 50.
Several differences in sexual activity by gender are shown in Panels 2 and 3. Women are more likely than men to be sexually active and have a partner at ages 15-24, likely to reflect that men are typically older than women at age of first marriage.21 The likelihood of having sex and the number of partners in the past 12 months declines with age, beginning around age 30 for women, but not until age 40 for men. Men's later age at marriage is again likely to account for this. Finally, the levels of sexual activity are higher for men by age 30 and remain higher. This is likely to reflect the greater likelihood of men being married and women being widowed or divorced in older age in this sample. It is important to point out that, although declines in sexual activity are evident over age 50, levels of activity remain considerable over age 49 for both men and women.
Panel 5 shows the smoothed likelihood of reporting multiple sexual partners across all ages. For men, the reporting of multiple sexual partnerships reaches a peak around age 40 and then declines with age. For women, reporting of multiple sexual partnerships remains almost constant after age 15-19. The number of women who report having multiple sexual partners is much lower than the number of men reporting having had multiple sexual partners.
Since many of the men and women in the MLSFH sample are married to others in the sample,21 a comparison of self-reported number of sexual partners with estimated spouses' number of partners provides insight into any discrepancies in what individuals know or report about their spouse's behaviour. Men and women's reports of their spouses' number of sexual partners are shown in Panel 4. Both men and women appear to have higher estimates of their spouse's number of partners than the number of partners claimed by men and women (Panel 3). Because MLSFH men are older than their wives by 6.6 years, women aged 50 are likely to be reporting on men aged around 50-60. Whilst women aged 50 report their spouses have around 1.45 partners, men aged 50-60 report having around 1.2-1.1 partners. Similarly, men aged 65 report their wives have around 0.85 partners, yet women aged 55-65 report having around 0.75-0.5 partners.
Worry about HIV infection declines for both men and women across the ages included in the sample but the decline is more pronounced among women (Panel 6). For women, worry about HIV infection is greatest at age 15-19 and declines steadily from age 20, in contrast to actual likelihood of HIV infection which doesn't decrease until around mid-30. For men, HIV worry declines very gradually until the early 60s when it plateaus.
Discussion
Our analysis of data collected from older Malawians shows that whilst HIV prevalence among both men and women declines at older old ages, the level of HIV infection after the typical 15-49 focus of HIV data collection remains considerable. Among men, likelihood of HIV in fact increases in the following 15 years. Prevalence of HIV among men aged 50-64 is 8.9% (compared to 4.1% among 15-49 year olds) and among women is 5.4% (compared to 8.3% among 15-49 year olds). This means that the population routinely excluded from data collection and prevention efforts contributes 16.3 and 43.5% of the female and male HIV disease burden in this sample.
This study is one of the first to collect HIV data for adults over 50 in a population-based survey. It reinforces and expands upon the limited number of previous estimates based on smaller samples and extrapolation by contributing robust, disaggregated data and building a more detailed picture of prevalence across Africa. Like the data presented here, previous these studies have identified considerable HIV prevalence at older ages and generally higher prevalence among older men than older women. A study in Cameroon in 2002 found prevalence of 2.7% among women and 2.5% among men aged 55-70, compared to 7.1% and 5.9% at ages 15-54.13 A study of northwestern Ethiopia in 2004 found much higher prevalence of 4.5% among women and 5.35% among men aged 50+, compared to only 6.3% among men and women aged 15-49 obtained by national sentinel surveillance.14
More recently, Negin and Cumming produced estimates of HIV at 50+ calculated from DHS, AIDS Indicators Survey and other population-based surveys for 43 SSA countries.11 They found HIV among men and women over 49 was 4%, compared to 5% at ages 15-49, accounting for 14.3% of infections. Using Malawian data, they derive estimates of HIV prevalence among men and women at 50+ of 12.7%, compared to 11.9% of 15-49. Although the age-pattern of infection mirrors those presented here, the discrepancy in magnitude of infection reflects the inclusion of both rural and urban residents and weighting of the sample in the DHS on which their extrapolations are based. Nevertheless, their calculations of the contribution of HIV at older ages to the total HIV burden are similar to those presented here in highlighting the importance of HIV at 50+. They find HIV at 50+ accounted for 18.6% of all infections, compared to the 32.7% of all infections identified here (see Table 1). Therefore although limited, both studies point to the need for the inclusion of adults aged beyond reproductive ages in the collection of HIV data.
HIV infection at older ages is an important public health concern, for several reasons. Data from high income countries indicate that HIV infected adults aged over 49 have poorer prognoses than their younger counterparts. For example, older adults in Europe are typically diagnosed with HIV later than younger adults, with much lower CD4 cell counts.22-24 There is evidence to suggest this pattern of late-diagnosis at older ages is likely to be mirrored in sub-Saharan African settings.3,5,25 The time of diagnosis is critical: older late presenters are substantially more likely to die within a year of diagnosis than older adults who are not diagnosed late or younger late presenters.26 Recent research in South Africa found that adults initiating treatment after age 49 were significantly more likely to die than adults initiating before aged 50, but that these differences were only evident in the first year of treatment, underlining the need for increased HIV testing, early ART initial and improved clinical care for older adults in SSA settings.27
In addition, developed country data indicate a high prevalence of HIV-related illnesses among adults over 49 years living with HIV. Studies have identified elevated cases of metabolic syndrome28 and reduced bone mass density29 among older infected adults, and compared to younger infected adults, higher risks of extrapyramidal motor signs,30 developing AIDS dementia complex,23,31 wasting syndrome, and of presenting multiple AIDS-defining illnesses.23 Older infected adults also experience higher rates of many non-AIDS-related illness than younger infected adults or older uninfected adults.24,32-38 In SSA, a recent study in Uganda found higher mortality among HIV-infected adults over 59 than younger adults after controlling for baseline CD4 cell count and time of ART initiation, also suggesting the need for clinical care beyond HIV treatment for these patients.39 In all settings, but particularly resource-poor settings, these factors make the management of HIV infection more complicated at older ages. Already overwhelmed health systems in SSA will struggle to provide the necessary geriatric care for HIV-infected adults, particularly at facilities in rural areas where the majority of older adults live.40
Effective prevention of further HIV infection among the older population requires information about sexual behaviour after childbearing. The level of sexual activity reported by MLSFH respondents declines steadily at older ages, but again remains considerable, particularly for men. Approximately half of women and almost 85% of men aged 50-64 reported having had sex in the year prior to the survey, as did one quarter of women and three quarters of men over age 64 . This is in-line with data from Uganda, Zimbabwe and South Africa for adults aged 50-6041 but extends analysis to a wider age range.
The sexual activity respondents are most likely to report is within marriage. Previous research has shown that the majority of HIV transmission takes place within marriage.42,43 Given sexual activity, as indicated here by reports of sex in the past year, remains considerable at older ages, and the average number of sexual partners in the past year for men remains above 1 until around aged 80 (likely representing sexual networks in which HIV can be transmitted),HIV risk from multiple sexual partners is likely to remain at older ages. Indeed, although these data indicate that women were aware of their spouses' additional sexual partners (perhaps even over-estimating their number), there is no evidence in these data to suggest that sexual activity within marriage was affected, and there is evidence from elsewhere that condom-use is especially hard to navigate within marital partnerships.44,45
Our data mirror previous findings regarding gender differences in perceptions of HIV risk at older age. However, the relatively high proportion (16%) of men and smaller proportion (8%) of women aged over 64 reported being “very worried” about HIV infection, is greater than found in prior research in urban Kenya.12 This level of worry is likely to in part reflect the nature of the sample: respondents were asked a number of questions about HIV, offered HIV testing by the MLSFH, exposed to regular HIV prevention messages by government and non-government actors in the field sites and were therefore repeatedly informed about the risk of HIV infection. However, considered alongside respondents' reports of their spouses' sexual partners, and older men's self-reported number of partners, their reports of being worried about HIV infection are also likely to indicate known or suspected HIV risk behaviours.
The importance of including adults aged beyond 49 in research on sexual behaviour and HIV prevalence in SSA is expected to grow in the future. As access to HIV antiretroviral drugs spreads throughout SSA, it is likely that HIV positive individuals will live to older ages. As a result, the sexual behaviour of older individuals may be of increasing significance in determining the future of the HIV/AIDS epidemic in SSA.
Our study also challenges the view that sexual behaviour and HIV data collection is difficult at older ages.46-48 We demonstrate that in Malawi questions about sexuality and HIV testing can be offered in population based studies without the risk of high non-response bias. This is consistent with previous findings in Thailand49 and therefore suggests that this may be the case in a number of quite different settings. This, coupled with our findings of considerable levels of HIV infection and sexual activity among older Malawians, suggests that individuals aged beyond 49 should and can be included in HIV/AIDS research and prevention efforts in SSA.
Acknowledgments
Sources of support: MLSFH/MDICP has received support through grants from the Rockefeller Foundation; NICHD (R01-HD4173, R01 HD372-276); NIA (AG1236-S3); and the Center for AIDS Research and the Center on the Demography of Aging, both at the University of Pennsylvania. The authors are grateful to the anonymous reviewers for their insightful comments.
Emily Freeman has received support to conduct analysis of MLSFH data from the Economic and Social Research Council, UK (ES/F022174/1)
Footnotes
Meetings at which parts of the data were presented: Presentation at 20th World Congress for Sexual Health, Glasgow, June 12-16, 2011
Poster presentation at Population Association of America Annual Meeting, Washington, DC, March 31-April 12, 2011
References
- 1.UNAIDS. Report on the global AIDS epidemic: A UNAIDS 10th anniversary special edition. Geneva: UNAIDS; 2006. [Google Scholar]
- 2.UNAIDS. Understanding the latest estimates of the 2006 report on the global AIDS epidemic. Geneva: UNAIDS; 2006. 19 December 2007. [Google Scholar]
- 3.HAI. The cost of love Older people in the fight against AIDS in Tanzania: HelpAge International. 2004. [Google Scholar]
- 4.Ingstad B, Bruun FJ, Tlou S. AIDS and the elderly Tswana: the concept of pollution and consequences for AIDS prevention. Journal of cross-cultural gerontology. 1997 Dec;12(4):357–72. doi: 10.1023/a:1006501414850. [DOI] [PubMed] [Google Scholar]
- 5.Negin J, Nemser B, Cumming R, Lelerai E, Ben Amor Y, Pronyk P. HIV Attitudes, Awareness and Testing Among Older Adults in Africa. AIDS and Behavior. 2011:1–6. doi: 10.1007/s10461-011-9994-y. [DOI] [PubMed] [Google Scholar]
- 6.UNAIDS. Global report: UNAIDS report on the global AIDS epidemic 2010. Geneva: UNAIDS; 2010. [Google Scholar]
- 7.Williams A, Tumwekwase G. Multiple impacts of the HIV/AIDS epidemic on the aged in rural Uganda. Journal of cross-cultural gerontology. 2001;16(3):221–36. doi: 10.1023/a:1011953126460. [DOI] [PubMed] [Google Scholar]
- 8.Todd J, Cremin I, McGrath N, Bwanika JB, Wringe A, Marston M, et al. Reported number of sexual partners: comparison of data from four African longitudinal studies. Sexually Transmitted Infections. 2009 Apr 1;85(Suppl 1):i72–i80. doi: 10.1136/sti.2008.033985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kinga O, Issacb O, Ademolab A, Nnennad I. Condom Use and Perceived HIV Infection Among Geriatrics Aged 65-102 Years in Nigeria. Outliers, a collection of Essays and Creative Writing on Sexuality in Africa. 2010;3(Spring) Non-peer reviewed journal? [Google Scholar]
- 10.McGrath N, Isingo R, Kasamba I, Lopman B, Maher D, Marston M, et al. Older people's sexual behaviour and HIV risk in five high prevalence sub-Saharan African populations. IUSSP XXVI International Population Conference 2009; Morroco. 2009. [Google Scholar]
- 11.Negin J, Cumming RG. Bullentin of the World Health Organization. 2010. HIV infection in older adults in sub-Saharan Africa: Extrapolating prevalence from existing data. Available online 7 September 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chepngeno-Langat G. ‘Bury the Head in the Sand’: Older People in Sub-Saharan Africa and Sexual Risk Perception. Population Association of America 2011 Annual Meeting; March 31 - April 2; Washington, DC. 2011. [Google Scholar]
- 13.Nyambi P, Zekeng L, Kenfack H, Tongo M, Nanfack A, Nkombe I, et al. HIV Infection in Rural Villages of Cameroon. JAIDS Journal of Acquired Immune Deficiency Syndromes. 2002;31(5):506–13. doi: 10.1097/00126334-200212150-00008. [DOI] [PubMed] [Google Scholar]
- 14.Kassu A, Mekonnen A, Bekele A, Abseno N, Melese E, Moges F, et al. HIV and syphilis infection among elderly people in northwest Ethiopia. Japenese Journal of Infectious Disease. 2004;57(6):264–7. [PubMed] [Google Scholar]
- 15.Knodel J, Watkins SC, VanLandingham M. AIDS and Older Persons: An International Perspective. USA: Population Studies Centre, Institute for Social Resarch, University of Michigan; 2002. Report No.: Report No. 02-495. [Google Scholar]
- 16.Negin J, Wariero J, Cumming RG, Mutuo P, Pronyk PM. High Rates of AIDS-Related Mortality Among Older Adults in Rural Kenya. JAIDS Journal of Acquired Immune Deficiency Syndromes. 2010 doi: 10.1097/QAI.0b013e3181e9b3f2. Publish Ahead of Print:10.1097/QAI.0b013e3181e9b3f2. [DOI] [PubMed] [Google Scholar]
- 17.Anglewicz P, adams j, Obare F, Kohler HP, Watkins S. The Malawi Diffusion and Ideational Change Project 2004-06: Data collection, data quality, and analysis of attrition. Demographic Research. 2009 May 05;20(21):503–40. doi: 10.4054/demres.2009.20.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bignami-Van Assche S, Smith K, Reniers G, Anglewitz P, Thornton R, Chao LW, et al. Protocol for biomarker testing in the 2004 Malawi Diffusion and Ideational Change Project: Social Networks Project. University of Pennsylvania; 2004. Jun, 2004 Contract No.: 6. [Google Scholar]
- 19.Watkins SC, Zulu EM, Kohler HP, Behrman JR. Introduction to Social Interactions and HIV/AIDS in Rural Africa. Demographic Research. 2003 Sep 19;1(1) Special Collection. [Google Scholar]
- 20.Obare F, Flemming P, Anglewicz P, Thornton R, Martinson F, Kapatuka A, et al. Acceptance of repeat population-based voluntary counselling and testing for HIV in rural Malawi. Sexually Transmitted Infections. 2009;85(2):139–44. doi: 10.1136/sti.2008.030320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Reniers G. Divorce and remarriage in rural Malawi. Demographic Research. 2003 Sep 19;1:176–200. Special Collection. [Google Scholar]
- 22.Health Protection Agency. HIV in the United Kingdom: 2010 Report. London: Health Protection Agency; Nov 26, 2010. [Google Scholar]
- 23.Longo B, Camoni L, Boros S, Suligoi B. Increasing proportion of AIDS diagnoses among older adults in Italy. AIDS Patient Care and STDS. 2008 May;22(5):365–71. doi: 10.1089/apc.2007.0168. [DOI] [PubMed] [Google Scholar]
- 24.Mothe B, Perez Ia, Domingo P, Podzamczer D, Ribera E, Curran A, et al. HIV-1 Infection in Subjects Older than 70: A Multicenter Cross-Sectional Assessment in Catalonia, Spain. Current HIV Research. 2009;7(6):597–600. doi: 10.2174/157016209789973691. [DOI] [PubMed] [Google Scholar]
- 25.Fylkesnes K, Siziya S. A randomized trial on acceptability of voluntary HIV counselling and testing. Tropical Medicine and International Health. 2004;9(5):566–72. doi: 10.1111/j.1365-3156.2004.01231.x. [DOI] [PubMed] [Google Scholar]
- 26.Smith R, Delpech V, Brown A, Rice B. HIV transmission and high rates of late diagnoses among adults aged 50 years and over. AIDS. 2010 Aug 24;24(13):210–2115. doi: 10.1097/QAD.0b013e32833c7b9c. [DOI] [PubMed] [Google Scholar]
- 27.Mutevedzi PC, Lessells RJ, Rodger AJ, Newell ML. Association of Age with Mortality and Virological and Immunological Response to Antiretroviral Therapy in Rural South African Adults. PLoS ONE. 2011;6:e21795. doi: 10.1371/journal.pone.0021795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Adeyemi O, Rezai K, Bahk M, Badri S, Thomas-Gossain N. Metabolic Syndrome in Older HIV-Infected Patients: Data from the CORE50 Cohort. AIDS Patient Care & STDs. 2008;22(12):941–5. doi: 10.1089/apc.2008.0119. [DOI] [PubMed] [Google Scholar]
- 29.Arnsten J, Freeman R, Howard A, Floris-Moore M, Lo Y, Klein R. Decreased bone mineral density and increased fracture risk in aging men with or at risk for HIV infection. AIDS. 2007;21(5):617–23. doi: 10.1097/QAD.0b013e3280148c05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Valcour V, Watters M, Williams A, Sacktor N, McMurtray A, Shikuma C. Aging exacerbates extrapyramidal motor signs in the era of highly active antiretroviral therapy. Journal of Neurovirology. 2008;14(5):362–7. doi: 10.1080/13550280802216494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Larussa D, Lorenzini P, Cingolani A, Bossolasco S, Grisetti S, Bongiovanni M, et al. Highly active antiretroviral therapy reduces the age-associated risk of dementia in a cohort of older HIV-1-infected patients. AIDS Research and Human Retroviruses. 2006;22(5):386–92. doi: 10.1089/aid.2006.22.386. [DOI] [PubMed] [Google Scholar]
- 32.Adeyemi OM, Badri SM, Max B, Chinomona N, Barker D. HIV Infection in Older Patients. Clinical Infectious Diseases. 2003;36(10):1347. doi: 10.1086/374871. [DOI] [PubMed] [Google Scholar]
- 33.Goulet JL, Fultz SL, Rimland D, Butt A, Gibert C, Rodriguez-Barradas M, et al. Do Patterns of Comorbidity Vary by HIV Status, Age, and HIV Severity? Clinical Infectious Diseases. 2007 Dec 15;45(12):1593–601. doi: 10.1086/523577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hughes A, Ward B, Stuart-Buttle C, Asboe D, Sullivan A, Barton S, et al. The Second Joint Conference of the British HIV Association and the British Association for Sexual Health and HIV. Manchester, UK; Apr 20–23, 2010. Over 50? It's time for a dual energy x-ray absorptiometry (DEXA) scan. [Google Scholar]
- 35.Magalhaes MG. Comorbidities in older patients with HIV: A retrospective study. Journal of the American Dental Association. 2007;138(11):1468–75. doi: 10.14219/jada.archive.2007.0083. [DOI] [PubMed] [Google Scholar]
- 36.Power L, Bell M, Freemantle I. A National Study of People over 50 with HIV. London: Joseph Roundtree Foundation; Sep, 2010. [Google Scholar]
- 37.Stuart-Buttle C, Randell P, Gedela K, Boffito M, Sullivan A, Asboe D. Screening for bone disease in HIV patients; The Second Joint Conference of the British HIV Association and the British Association for Sexual Health and HIV; 20–23 April 2010; Manchester, UK; 2010. [Google Scholar]
- 38.Ward B, Hughes A, Asboe D, Barton S, Gazzard B, Baber L, et al. Ageing and HIV/AIDS: evaluation of a dedicated clinical service for HIV-infected individuals over 50 years of age; The Second Joint Conference of the British HIV Association and the British Association for Sexual Health and HIV; 20–23 April 2010; Manchester, UK; 2010. [Google Scholar]
- 39.Bakanda C, Birungi J, Mwesigwa R, Ford N, Cooper C, Au-Yeung C, et al. Association of aging and survival in a large HIV-infected cohort on antiretroviral therapy. AIDS. 2011;25(5):701–5. doi: 10.1097/QAD.0b013e3283437ed7. [DOI] [PubMed] [Google Scholar]
- 40.Mills EJ, Rammohan A, Awofeso N. Ageing faster with AIDS in Africa. The Lancet. 2011;377(9772):1131–3. doi: 10.1016/S0140-6736(10)62180-0. [DOI] [PubMed] [Google Scholar]
- 41.McGrath N, Hosegood V, Newell M, editors. Sexual behaviour in older people (40+ years) in rural KwaZulu Natal, South Africa: implications for prevention and surveys. South African AIDS conference; 5-8 June 2007; Durban. [Google Scholar]
- 42.Dunkle KL, Stephenson R, Karita E, Chomba E, Kayitenkore K, Vwalika C, et al. New heterosexually transmitted HIV infections in married or cohabiting couples in urban Zambia and Rwanda: an analysis of survey and clinical data. The Lancet. 2008;371(9631):2183–91. doi: 10.1016/S0140-6736(08)60953-8. [DOI] [PubMed] [Google Scholar]
- 43.Quigley M, Munguti K, Grosskurth H, Todd J, Mosha F, Senkoro K, et al. Sexual behaviour patterns and other risk factors for HIV infection in rural Tanzania: a case-control study. AIDS. 1997;11(2):237–48. doi: 10.1097/00002030-199702000-00015. [DOI] [PubMed] [Google Scholar]
- 44.Maharaj P, Cleland J. Condom Use Within Marital and Cohabiting Partnerships in KwaZulu-Natal, South Africa. Studies in Family Planning. 2004;35(2):116–24. doi: 10.1111/j.1728-4465.2004.00013.x. [DOI] [PubMed] [Google Scholar]
- 45.de Walque D, Kline R. Variations in Condom Use by Type of Partner in 13 Sub-Saharan African Countries. Studies in Family Planning. 2011;42(1):1–10. doi: 10.1111/j.1728-4465.2011.00259.x. [DOI] [PubMed] [Google Scholar]
- 46.Levy J, Albrecht G. Methodological considerations in research on sexual behavior and AIDS among older people. In: Riley MW, Ory MG, Zablotsky D, editors. AIDS in an Aging Society. New York: Springer; 1989. pp. 96–123. [Google Scholar]
- 47.Pointon S. Myths and negative attitudes about sexuality in older people. Generations Review. 1997;7(4):6–8. [Google Scholar]
- 48.Kaye RA. Sexuality in the Later Years. Ageing and Society. 1993;13:415–426. 1993;13(3):416-26. [Google Scholar]
- 49.Knodel J, Chayovan N. Sexual activity among older Thais: the influence of age, gender and health. Journal of cross-cultural gerontology. 2001;16(2):173–200. doi: 10.1023/a:1010608226594. [DOI] [PubMed] [Google Scholar]