Abstract
Background/Aims
Central hypothyroidism (CH) in children is rare and may be due to a variety of genetic defects. Most of these defects, but not all, are associated with additional pituitary hormone deficits. In a young child presenting with CH, it is important to determine whether additional pituitary hormone deficiencies are present, but this may be difficult to establish clinically.
Methods
We describe the clinical characteristics of two young siblings, ages 6 months and 2 years, presenting with isolated CH. Whole exome sequencing was performed to determine the genetic basis of isolated CH.
Results
A homozygous frameshift mutation of PROP1 (296delGA) was identified in both probands. Defects in PROP1 cause progressive deficiency of multiple pituitary hormones. Based on this genetic diagnosis, further clinical testing was performed that demonstrated growth hormone deficiency in one sibling.
Conclusions
PROP1 deficiency may present as isolated central hypothyroidism at a very young age. In disorders with multiple potential causative genes, whole exome sequencing may facilitate rapid genetic diagnosis and lead to important changes in clinical management.
Keywords: central hypothyroidism, PROP-1, whole exome sequencing, gene diagnostics, genetics of endocrinopathies
Introduction
Central hypothyroidism (CH), the insufficient production of thyroid hormone due to lack of normal thyrotropin (TSH) secretion, is most often acquired through damage to the pituitary or hypothalamus. Congenital CH is rare, with an incidence traditionally estimated between 1:110,000 and 1:29,000 [1-3]; however, recent data suggest that the incidence may be as high as 1:16,000 [4] and that CH may account for up to 13% of permanent congenital hypothyroidism [5]. Because the majority of congenital CH occurs in the setting of combined pituitary hormone deficiency (CPHD), congenital CH in the absence of other pituitary hormone deficits (“isolated CH”) is very rare, occurring in no more than 1:92,000 infants [5].
When a child presents with apparently isolated CH, a primary concern is to identify whether the defect is limited to the thyrotroph lineage or affects multiple pituitary hormones, a distinction that has significant implications for clinical management and prognosis. However, the challenge of diagnosing pituitary hormone deficiencies in young children can make it difficult to distinguish true isolated CH from early or evolving CPHD. We report a case of two siblings with isolated CH in whom whole exome sequencing provided rapid genetic diagnosis of a homozygous frameshift mutation in PROP1, resulting in important changes in clinical management.
Patients and Methods
Case 1
A female infant was born at 32 weeks gestation due to spontaneous preterm labor. Her birth weight (1810 g; −0.1 SDS) and length (43.2 cm; +0.1 SDS) were normal for gestational age. Other than a brief need for nasogastric feeding, her neonatal course was unremarkable, with no hyperbilirubinemia or hypoglycemia. Newborn screening at 5 days of age showed normal TSH 3 mIU/L (normal, <20 mIU/L) and total T4 7.8 mcg/dL (normal, >6.0 mcg/dL). Newborn screening was repeated at 20 days of age (35 weeks corrected gestational age) and revealed normal TSH 2 mIU/L and slightly low total T4 5.4 mcg/dL (normal, >6.0 mcg/dL); nevertheless, these results were reported as normal by the newborn screening program and no follow up was recommended.
At age 2 years 3 months, the patient was noted to have a decrease in height SDS from −1.8 to −2.5, accompanied by a lesser decline in weight SDS (−0.4 to −1.2), with normal weight-for-length (+0.6 SDS). Developmentally, she began to sit up at 8 months (6 months corrected gestational age) and to crawl at 9 months, but began walking only at age 2 years. At the time of presentation, she did not yet speak two-word phrases. She had delayed tooth eruption, with only seven teeth present, and her bone age was delayed at 18 months. Laboratory evaluation showed low free T4 0.73 ng/dL (normal, 1.00–2.10 ng/dL) and normal TSH 1.78 mIU/L (normal, 0.70–5.70 mIU/L), consistent with CH. Of note, thyrotropin-releasing hormone (TRH) is not available for stimulation testing in the United States. Treatment was begun with levothyroxine 25 mcg daily. Magnetic resonance imaging (MRI) of the brain was normal, including normal pituitary size for age.
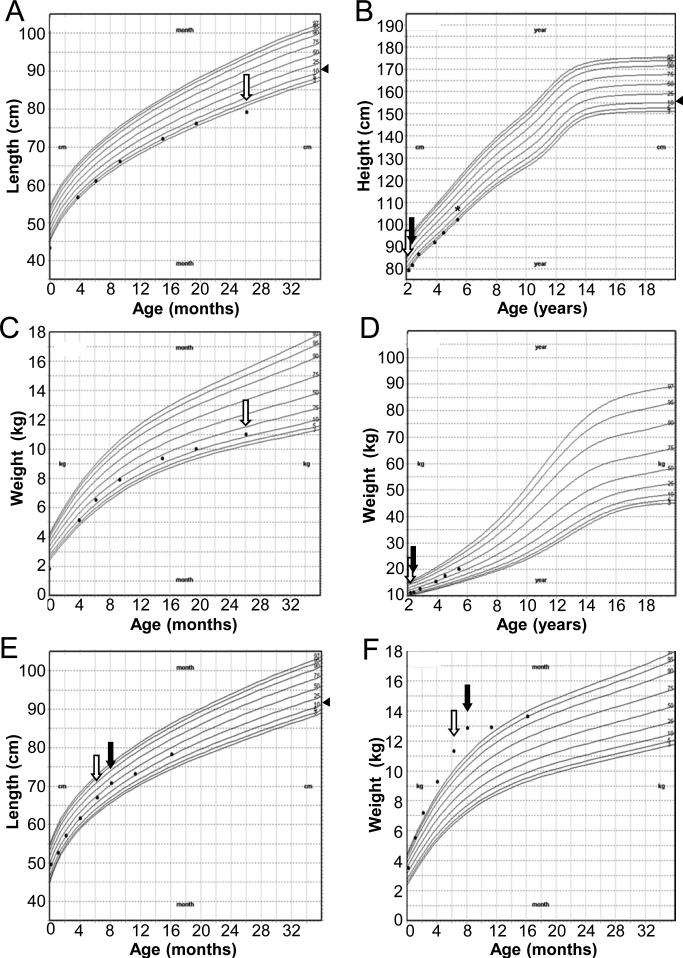
At the time of initial evaluation, insulin-like growth factor 1 (IGF-1) was low (<25 ng/mL; normal, 51–303 ng/mL) but IGF-binding protein 3 (IGFBP-3) was normal (1.4 mcg/mL; normal, 0.8–3.9 mcg/mL). Serum prolactin (PRL) was normal (8.3 ng/mL; normal, 3.2–25.9), as was a random serum cortisol (12.1 mcg/dL; normal, 5.0–25.0). LH and FSH levels were not tested, as the gonadotropin axis is normally quiescent at this age. Once treated with levothyroxine, the patient exhibited catch-up growth with an increase in height SDS by +0.9 (height −1.6 SDS; midparental height −1.2 SDS). Because low IGF-1 is not a reliable marker of growth hormone (GH) deficiency in young children [6] and may also be caused by decreased GH secretion due to hypothyroidism [7, 8], and because the patient's linear growth improved and then remained normal with restoration of euthyroidism, the patient was considered to have isolated CH. At age 5 years, the patient's growth velocity remained normal (6.3 cm/year; Figure 1). Despite her normal growth, GH stimulation testing was performed following the genetic diagnosis of PROP1 deficiency. This demonstrated profound GH deficiency, with a peak GH response of 0.23 ng/mL to arginine and glucagon, and GH treatment was started (25 mcg/kg/day).
Figure 1.
Growth curves (CDC) for Case 1 (A-D) and Case 2 (E, F). Measurements for Case 1 are uncorrected for gestational age. Arrowheads (◀) to the right of height or length curves indicate midparental height (B) or midparental height percentile (A,E). Open arrows indicate diagnosis of central hypothyroidism. Closed arrows indicate normalization of serum free T4. Asterisk (*) indicates diagnosis of growth hormone deficiency.
Case 2
A male infant, the younger brother of Case 1, was born at 40 weeks gestation with normal birth weight (3510 g; −0.1 SDS) and length (48 cm; −1.4 SDS). His neonatal course was notable for mild indirect hyperbilirubinemia that resolved without treatment and was attributed to glucose-6-phosphate dehydrogenase deficiency. Newborn screening at 2 days of age showed normal TSH < 2.5 mIU/L (normal, <20.0 mIU/L) and total T4 9.0 mcg/dL (normal, >5.0 mcg/dL). His subsequent medical and developmental history were normal.
At his six month well visit, the patient was noted to be severely obese (weight +3.1 SDS), despite a dietary history that was not consistent with excess caloric intake. His linear growth and physical examination were normal, including a normal size phallus and bilaterally descended testes. Based on his obesity and his sister's history of CH, thyroid function tests were obtained, showing low free T4 0.66 ng/dL (normal, 0.80–1.80 ng/dL) and normal TSH 3.57 mIU/L (normal, 0.70–5.70 mIU/L) consistent with CH. Treatment was begun with levothyroxine 25 mcg daily. MRI of the brain was normal. Like his sister, this patient had low IGF-1 (<25 ng/mL; normal, 55–327 ng/mL) and normal IGFBP-3 (1.6 mcg/mL; normal, 0.7–3.6 mcg/mL). Serum prolactin was normal (20.7 ng/mL; normal 3.2–25.9). Although a morning cortisol level at age 6 months was indeterminate (7.5 mcg/dL), he had no symptoms of adrenal insufficiency, and a subsequent morning cortisol was normal (16.3 mcg/dL) at age 18 months. The patient's rapid weight gain arrested after normalization of his serum free T4, and his growth velocity remained normal during subsequent follow up (Figure 1).
The parents of these siblings were of Hispanic ethnicity, originally from the Dominican Republic, and had no known consanguinity. The father's height was 165 cm (−1.6 SDS) and the mother's height was 160 cm (−0.5 SDS). Both the father and mother were obese (BMI 33 kg/m2 and 41 kg/m2, respectively) but were otherwise healthy and biochemically euthyroid. The occurrence of isolated CH in siblings of both genders with unaffected parents suggested an autosomal recessive genetic etiology. Whole exome sequencing of the two probands and their mother was performed as part of a research protocol approved by the Boston Children's Hospital IRB. Written informed consent was obtained, and DNA was extracted from peripheral blood mononuclear cells using standard techniques. Hybrid selection was performed using NimbleGen's SeqCapEZ Exome Version 3.0 (Roche NimbleGen Inc., Madison, WI). Samples were sequenced using the Illumina HiSeq 2000 platform (Illumina Inc., San Diego, CA). The resulting reads were aligned to the hg19 reference genome with the Burrows-Wheeler Aligner [9], Genome Analysis Toolkit [10] base quality score recalibration was applied, indel realignment was conducted, and single nucleotide polymorphism and indel discovery and genotyping were performed across all samples simultaneously using variant quality score recalibration [11]. Variants were annotated for functional effect using SnpEff 2.0.5 (http://snpeff.sourceforge.net/). As individuals with phenotypes of this severity are extremely uncommon, it was assumed that any causal genetic variants would be rare. Therefore, only variants with minor allele frequency less than 1% in the 1000 Genomes project (February 2012 release) [12] and the National Heart, Lung, and Blood Institute exome variant server [13] were considered. To investigate the recessive model, a search was conducted for autosomal genes containing either a homozygous or two heterozygous rare nonsynonymous variants. This analysis yielded 13 variants in nine genes, including a previously reported homozygous pathogenic mutation in PROP1 (296delGA). Conventional Sanger sequencing confirmed that both patients were homozygous for this mutation and that both parents were heterozygous. This mutation in PROP1 was considered the likely etiology of the patients’ CH.
Discussion
Congenital CH is a rare condition that may be caused by mutations in any of several identified genes involved in hypothalamic or pituitary development, differentiation, or function (Table 1) [3, 14-16]. In a minority of cases, including inactivating mutations of the TSH β-subunit (TSHB) [17], TRH receptor (TRHR) [18, 19], or IGSF1 [16], the functional defect is isolated to pituitary thyrotrophs, although defects in TRHR or IGSF1 usually cause combined TSH and prolactin deficiency. In over 75% of cases, congenital CH occurs as a component of CPHD [5], and in this setting other pituitary hormone deficiencies usually precede or manifest concurrently with CH. Thus, TSH deficiency is rarely the sole presenting feature of CPHD.
Table 1.
Genetic Causes of Central Hypothyroidism
| Gene | Inheritance | Laboratory Findings | Radiological and Clinical Findings |
|---|---|---|---|
| Isolated TSH Deficiency | |||
| TSHB | AR | Low or normal TSH, high α-GSU | |
| TRHR | AR | Low or normal TSH, subnormal PRL response to TRH | |
| Combined Pituitary Hormone Deficiency | |||
| Pituitary Primordium Factors | |||
| HESX1 | AR or AD | TSH*, PRL*, GH, LH/FSH*, ACTH* deficiency | Hypoplasia of pituitary, septum pellucidum, corpus callosum; ectopic posterior pituitary; optic nerve hypoplasia |
| OTX2 | AD | TSH, PRL*, GH, LH/FSH, ACTH* deficiency | Hypoplastic anterior pituitary, ectopic posterior pituitary, micro-/anophthalmia, seizures |
| LHX3 | AR | TSH, PRL, GH, LH/FSH, ACTH* deficiency | Normal or hypoplastic anterior pituitary, cervical spine/vertebral anomalies, deafness, hyperextensible joints |
| LHX4 | AD | TSH, GH, LH/FSH*, ACTH deficiency | Ectopic posterior pituitary, hypoplastic corpus callosum, pointed cerebellar tonsils |
| SOX3 | XLR | TSH, GH, LH/FSH, ACTH deficiency | Infundibular hypoplasia, ectopic posterior pituitary, absent corpus callosum, craniofacial abnormalities |
| Ventral Diencephalon Factors | |||
| FGF8 | AD | TSH, GH*, LH/FSH, ACTH* deficiency, DI* | Holoprosencephaly |
| FGFR1 | AD | TSH*, GH, LH/FSH, ACTH* deficiency, DI* | Hypoplasia of pituitary, corpus callosum; ocular defects |
| GLI2 | AD | TSH*, GH, LH/FSH*, ACTH* deficiency, DI* | Holoprosencephaly, ectopic posterior pituitary, craniofacial abnormalities, polydactyly |
| Pituitary Differentiation Factors | |||
| PROP1 | AR | TSH, PRL, GH, LH/FSH, ACTH* deficiency | Anterior pituitary hypo- or hyperplasia |
| POU1F1 | AR or AD | TSH, PRL, GH deficiency | Normal or hypoplastic anterior pituitary |
| Other Factors | |||
| IGSF1 | XLR | TSH, PRL*, GH* deficiency | Macroorchidism |
| LEPR | AR | TSH*, LH/FSH deficiency | Early-onset severe obesity |
In a patient presenting with CH, distinguishing an isolated thyrotroph defect from CPHD has important clinical implications, and evaluation for other pituitary hormone deficiencies is imperative. In infants or young children, however, diagnosis of pituitary hormone deficiencies can be difficult, making it challenging to differentiate true isolated CH from CPHD. Physical examination may detect midline defects (e.g., cleft palate) or neuroimaging may reveal structural abnormalities (e.g., ectopic posterior pituitary or absent septum pellucidum) that suggest a problem with hypothalamic or pituitary development that is likely to be associated with CPHD. Patients with certain genetic forms of CPHD may have additional clinical features that can assist diagnosis (Table 1), but the absence of such findings in a patient with CH does not exclude CPHD.
Here we report two siblings with the unusual presentation of isolated CH, which strongly suggested an underlying genetic abnormality. Because CH has multiple genetic causes, we used whole exome sequencing to rapidly identify the causative mutation among the many candidate genes. Both siblings were found to be homozygous for a known pathogenic mutation of PROP1. This gene encodes a transcription factor critical for the normal development of multiple anterior pituitary cell lineages, and inactivating mutations of PROP1 cause deficiency of GH, TSH, PRL, LH, FSH, and in some cases ACTH. PROP1 deficiency is the most commonly identified genetic cause of CPHD, accounting for 30-50% of familial cases [20-22], although PROP1 mutations are found only rarely in sporadic CPHD [22-25]. The 296delGA mutation present in our patients is the most commonly identified PROP1 mutation, accounting for 50-72% of pathogenic mutant alleles [20, 26]. This two-base pair deletion causes a frameshift that leads to premature truncation of the PROP1 protein, loss of its DNA-binding homeodomain, and abrogation of its function [27].
These cases illustrate two important clinical aspects of PROP1 deficiency in children. First, children may present with isolated CH as the sole initial manifestation of PROP1 deficiency, in contrast to previous reports suggesting that loss of other anterior pituitary hormones generally precedes TSH deficiency [20]. The most common presentation of PROP1 deficiency is childhood growth failure due to GH deficiency. Gonadotropin deficiency may be apparent at birth as microphallus or cryptorchidism in a male, but more frequently manifests as delay or failure of normal pubertal progression. ACTH deficiency is a more variable feature that is more common with advancing age but may present during childhood, including with life-threatening adrenal crisis [21, 28-30]. Of note, both siblings in these cases had normal adrenal function at last follow up. Case 1 had a peak serum cortisol of 28.9 mcg/dL after glucagon injection (during GH stimulation testing), while Case 2 had a normal morning cortisol of 16.3 mcg/dL at age 18 months.
Second, these cases demonstrate that isolated CH due to PROP1 deficiency may occur at an earlier age than generally recognized. Two previous case series identified no children with PROP1 mutations who developed CH under about 3 years of age [21, 30], while another series reported CH in 11% of patients at one year of age and in 22% at three years; however, the prevalence of CH under one year and whether TSH was the only deficient hormone in these cases was not specified [20]. To our knowledge, there is only one well-documented case of PROP1 deficiency presenting with isolated CH under one year of age [31]. The current case therefore further supports the idea that isolated CH may be the presenting sign of PROP1 deficiency in infants and young children. Notably, although prolactin deficiency is reported to be common in PROP1 deficiency, both patients in these cases had normal basal prolactin levels. Although TRH-stimulated prolactin levels could not be measured, these cases suggest that prolactin deficiency may evolve over time and may not be present in very young children with PROP1 deficiency.
It is interesting to note that CH presented in Case 2 with severe obesity. Although hypothyroidism in infancy rarely leads to obesity, in this case the marked decrease in weight SDS after levothyroxine treatment strongly suggests a causative relationship. Although a similar growth pattern could be observed in a breastfed infant who weans from breastmilk, Case 2 was formula-fed from birth and had begun to eat pureed foods by age 6 months. Increased energy expenditure from walking, which began at age 14 months, cannot account for his decreased weight gain. This case serves as a reminder that evaluation for hypothyroidism should be considered in infants and young children with severe obesity, particularly if the dietary history does not suggest excess caloric intake.
The abnormalities noted on newborn screening in Case 1 suggest that CH may have been present shortly after birth, although interpretation is complicated by her prematurity. More typically, as in Case 2, thyroid function abnormalities are not detected in newborns with PROP1 deficiency, which has been interpreted as evidence that pituitary hormone deficiencies due to PROP1 mutations may not be present at birth but develop gradually during postnatal life. However, the fact that some individuals have microphallus and cryptorchidism at birth [21, 30] implies that elements of CPHD, such as gonadotropin deficiency, may be present at or before birth in at least a subset of patients. With this in mind, these cases raise the possibility that the incidence of neonatal CH in PROP1 deficiency has been underestimated. Such underestimation could be due in part to the limited sensitivity of newborn screening for CH, particularly in programs that measure TSH as the primary screening test, as remains the standard in many areas. Conversely, although up to 13% of congenital hypothyroidism detected in the newborn period is of central origin and the majority of these individuals have CPHD [5], it is unknown what proportion of CH detected on newborn screening is due to mutations in PROP1 or other genes associated with CPHD. These cases suggest that such patients may benefit from genetic testing to clarify their underlying defect.
Making a genetic diagnosis in these cases of apparently isolated CH had several important clinical consequences. First, had an isolated thyrotroph defect been discovered, it would have obviated the need for lifelong surveillance for and management of other pituitary hormone deficits. Instead, the finding of PROP1 deficiency clarified the siblings’ prognosis for developing GH, PRL, and gonadotropin deficiency, as well as indicating the need for careful monitoring of adrenal function even in childhood. The genetic diagnosis led directly to the diagnosis and treatment of profound GH deficiency in Case 1, in whom the diagnosis was otherwise obscured by her normal growth velocity. Although it is possible that GH deficiency may have been present since birth, despite the absence of neonatal hypoglycemia, this presentation reinforces that the diagnosis of GH deficiency in children may be complicated by the fact that some individuals display normal growth in childhood despite lack of normal GH secretion [32]. Nevertheless, growth hormone therapy may be indicated to address the adverse metabolic consequences of GH deficiency, including decreased lean body mass, increased fat mass, and decreased bone density [33]. As growth hormone replacement was recently initiated, we cannot yet comment on any response to therapy. Finally, identifying the causative mutation in these cases facilitated appropriate genetic counseling for the patients’ family and could provide the basis for pre-implantation or prenatal genetic testing in future pregnancies.
In summary, congenital CH may represent isolated TSH deficiency or the initial manifestation of CPHD, a distinction of profound clinical consequence. However, even a complete evaluation of pituitary function, which is mandatory in any child with CH, may not reliably make this distinction in a young child. We present a case of two siblings with isolated CH in whom whole exome sequencing rapidly identified a pathogenic mutation in PROP1. This genetic diagnosis, in turn, led to important changes in clinical management, including more aggressive evaluation for (and diagnosis of) associated pituitary deficiencies and the ability to provide more accurate prognostic information and genetic counseling to the family. These cases continue to broaden the clinical spectrum of PROP1 deficiency to include patients presenting in infancy or early childhood with CH that may not be accompanied by other pituitary hormone deficits. Furthermore, the marked benefit of securing a genetic diagnosis raises the possibility that infants and children diagnosed with CH may benefit from genetic testing for mutations in PROP1 or other genes associated with CH. In fact, the benefits of genetic diagnosis—including improved genetic counseling and directed screening for pituitary defects—may apply more generally to patients with other congenital pituitary hormone deficits such as isolated GH deficiency or hypogonadotropic hypogonadism [34-36]. This case demonstrates that whole exome sequencing may be an effective diagnostic tool in such patients, although a formal comparison of the utility of whole exome sequencing versus targeted candidate gene sequencing is necessary to evaluate the cost effectiveness of this approach. As it becomes less costly and more widely available clinically, whole exome sequencing will be a powerful tool for evaluating multiple candidate genes in these and other conditions.
Established facts.
Central hypothyroidism may be caused by a variety of genetic defects
In combined pituitary hormone deficiency due to mutations in PROP1, central hypothyroidism generally is preceded by other pituitary hormone deficiencies
Novel insights.
PROP1 deficiency may present with isolated central hypothyroidism in infants or young children
Whole exome sequencing may facilitate rapid genetic diagnosis and affect clinical management
Acknowledgments
We gratefully acknowledge Meghan Connolly and The Manton Center for Orphan Disease Research Gene Discovery Core at Boston Children's Hospital for facilitating patient DNA collection and for financial support for whole exome sequencing. We would like to thank Timothy Fennell, Charlotte Tolonen, Khalid Shakir, and Mark DePristo at the Broad Institute for technical assistance with variant calling. This work was also supported by National Institutes of Health Grants K12 HD052896-06 and K23 HD073351.
References
- 1.Fisher DA, Dussault JH, Foley TP, Jr., Klein AH, LaFranchi S, Larsen PR, Mitchell ML, Murphey WH, Walfish PG. Screening for congenital hypothyroidism: results of screening one million North American infants. J Pediatr. 1979;94:700–705. doi: 10.1016/s0022-3476(79)80133-x. [DOI] [PubMed] [Google Scholar]
- 2.Hanna CE, Krainz PL, Skeels MR, Miyahira RS, Sesser DE, LaFranchi SH. Detection of congenital hypopituitary hypothyroidism: ten-year experience in the Northwest Regional Screening Program. J Pediatr. 1986;109:959–964. doi: 10.1016/s0022-3476(86)80276-1. [DOI] [PubMed] [Google Scholar]
- 3.Persani L. Central hypothyroidism: pathogenic, diagnostic, and therapeutic challenges. J Clin Endocrinol Metab. 2012;97:3068–3078. doi: 10.1210/jc.2012-1616. [DOI] [PubMed] [Google Scholar]
- 4.Lanting CI, van Tijn DA, Loeber JG, Vulsma T, de Vijlder JJ, Verkerk PH. Clinical effectiveness and cost-effectiveness of the use of the thyroxine/thyroxine-binding globulin ratio to detect congenital hypothyroidism of thyroidal and central origin in a neonatal screening program. Pediatrics. 2005;116:168–173. doi: 10.1542/peds.2004-2162. [DOI] [PubMed] [Google Scholar]
- 5.van Tijn DA, de Vijlder JJ, Verbeeten B, Jr., Verkerk PH, Vulsma T. Neonatal detection of congenital hypothyroidism of central origin. J Clin Endocrinol Metab. 2005;90:3350–3359. doi: 10.1210/jc.2004-2444. [DOI] [PubMed] [Google Scholar]
- 6.Rosenfeld RG, Wilson DM, Lee PD, Hintz RL. Insulin-like growth factors I and II in evaluation of growth retardation. J Pediatr. 1986;109:428–433. doi: 10.1016/s0022-3476(86)80112-3. [DOI] [PubMed] [Google Scholar]
- 7.MacGillivray MH, Aceto T, Jr., Frohman LA. Plasma growth hormone responses and growth retardation of hypothyroidism. Am J Dis Child. 1968;115:273–276. doi: 10.1001/archpedi.1968.02100010275018. [DOI] [PubMed] [Google Scholar]
- 8.Baxter RC, Brown AS, Turtle JR. Radioimmunoassay for somatomedin C: comparison with radioreceptor assay in patients with growth-hormone disorders, hypothyroidism, and renal failure. Clin Chem. 1982;28:488–495. [PubMed] [Google Scholar]
- 9.Li H, Durbin R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics. 2009;25:1754–1760. doi: 10.1093/bioinformatics/btp324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, Kernytsky A, Garimella K, Altshuler D, Gabriel S, Daly M, DePristo MA. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 2010;20:1297–1303. doi: 10.1101/gr.107524.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.DePristo MA, Banks E, Poplin R, Garimella KV, Maguire JR, Hartl C, Philippakis AA, del Angel G, Rivas MA, Hanna M, McKenna A, Fennell TJ, Kernytsky AM, Sivachenko AY, Cibulskis K, Gabriel SB, Altshuler D, Daly MJ. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat Genet. 2011;43:491–498. doi: 10.1038/ng.806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Abecasis GR, Altshuler D, Auton A, Brooks LD, Durbin RM, Gibbs RA, Hurles ME, McVean GA. A map of human genome variation from population-scale sequencing. Nature. 2010;467:1061–1073. doi: 10.1038/nature09534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.National Heart Lung and Blood Institute . Exome variant server [homepage on the Internet] Seattle, WA: [2012 May 23]. c2012. Available from http://evs.gs.washington.edu/EVS/ [Google Scholar]
- 14.Kelberman D, Rizzoti K, Lovell-Badge R, Robinson IC, Dattani MT. Genetic regulation of pituitary gland development in human and mouse. Endocr Rev. 2009;30:790–829. doi: 10.1210/er.2009-0008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cohen LE. Genetic disorders of the pituitary. Curr Opin Endocrinol Diabetes Obes. 2012;19:33–39. doi: 10.1097/MED.0b013e32834ed639. [DOI] [PubMed] [Google Scholar]
- 16.Sun Y, Bak B, Schoenmakers N, van Trotsenburg AS, Oostdijk W, Voshol P, Cambridge E, White JK, le Tissier P, Gharavy SN, Martinez-Barbera JP, Stokvis-Brantsma WH, Vulsma T, Kempers MJ, Persani L, Campi I, Bonomi M, Beck-Peccoz P, Zhu H, Davis TM, Hokken-Koelega AC, Del Blanco DG, Rangasami JJ, Ruivenkamp CA, Laros JF, Kriek M, Kant SG, Bosch CA, Biermasz NR, Appelman-Dijkstra NM, Corssmit EP, Hovens GC, Pereira AM, Dunnen JT, Wade MG, Breuning MH, Hennekam RC, Chatterjee K, Dattani MT, Wit JM, Bernard DJ. Loss-of-function mutations in IGSF1 cause an X-linked syndrome of central hypothyroidism and testicular enlargement. Nat Genet. 2012;44:1375–1381. doi: 10.1038/ng.2453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bonomi M, Proverbio MC, Weber G, Chiumello G, Beck-Peccoz P, Persani L. Hyperplastic pituitary gland, high serum glycoprotein hormone alpha-subunit, and variable circulating thyrotropin (TSH) levels as hallmark of central hypothyroidism due to mutations of the TSH beta gene. J Clin Endocrinol Metab. 2001;86:1600–1604. doi: 10.1210/jcem.86.4.7411. [DOI] [PubMed] [Google Scholar]
- 18.Collu R, Tang J, Castagne J, Lagace G, Masson N, Huot C, Deal C, Delvin E, Faccenda E, Eidne KA, Van Vliet G. A novel mechanism for isolated central hypothyroidism: inactivating mutations in the thyrotropin-releasing hormone receptor gene. J Clin Endocrinol Metab. 1997;82:1561–1565. doi: 10.1210/jcem.82.5.3918. [DOI] [PubMed] [Google Scholar]
- 19.Bonomi M, Busnelli M, Beck-Peccoz P, Costanzo D, Antonica F, Dolci C, Pilotta A, Buzi F, Persani L. A family with complete resistance to thyrotropin-releasing hormone. N Engl J Med. 2009;360:731–734. doi: 10.1056/NEJMc0808557. [DOI] [PubMed] [Google Scholar]
- 20.Deladoey J, Fluck C, Buyukgebiz A, Kuhlmann BV, Eble A, Hindmarsh PC, Wu W, Mullis PE. “Hot spot” in the PROP1 gene responsible for combined pituitary hormone deficiency. J Clin Endocrinol Metab. 1999;84:1645–1650. doi: 10.1210/jcem.84.5.5681. [DOI] [PubMed] [Google Scholar]
- 21.Vallette-Kasic S, Barlier A, Teinturier C, Diaz A, Manavela M, Berthezene F, Bouchard P, Chaussain JL, Brauner R, Pellegrini-Bouiller I, Jaquet P, Enjalbert A, Brue T. PROP1 gene screening in patients with multiple pituitary hormone deficiency reveals two sites of hypermutability and a high incidence of corticotroph deficiency. J Clin Endocrinol Metab. 2001;86:4529–4535. doi: 10.1210/jcem.86.9.7811. [DOI] [PubMed] [Google Scholar]
- 22.Turton JP, Mehta A, Raza J, Woods KS, Tiulpakov A, Cassar J, Chong K, Thomas PQ, Eunice M, Ammini AC, Bouloux PM, Starzyk J, Hindmarsh PC, Dattani MT. Mutations within the transcription factor PROP1 are rare in a cohort of patients with sporadic combined pituitary hormone deficiency (CPHD). Clin Endocrinol (Oxf) 2005;63:10–18. doi: 10.1111/j.1365-2265.2005.02291.x. [DOI] [PubMed] [Google Scholar]
- 23.Dateki S, Fukami M, Uematsu A, Kaji M, Iso M, Ono M, Mizota M, Yokoya S, Motomura K, Kinoshita E, Moriuchi H, Ogata T. Mutation and gene copy number analyses of six pituitary transcription factor genes in 71 patients with combined pituitary hormone deficiency: identification of a single patient with LHX4 deletion. J Clin Endocrinol Metab. 2010;95:4043–4047. doi: 10.1210/jc.2010-0150. [DOI] [PubMed] [Google Scholar]
- 24.de Graaff LC, Argente J, Veenma DC, Drent ML, Uitterlinden AG, Hokken-Koelega AC. PROP1, HESX1, POU1F1, LHX3 and LHX4 mutation and deletion screening and GH1 P89L and IVS3+1/+2 mutation screening in a Dutch nationwide cohort of patients with combined pituitary hormone deficiency. Horm Res Paediatr. 2010;73:363–371. doi: 10.1159/000308169. [DOI] [PubMed] [Google Scholar]
- 25.Nystrom HF, Saveanu A, Barbosa EJ, Barlier A, Enjalbert A, Glad C, Palming J, Johannsson G, Brue T. Detection of genetic hypopituitarism in an adult population of idiopathic pituitary insufficiency patients with growth hormone deficiency. Pituitary. 2011;14:208–216. doi: 10.1007/s11102-010-0278-8. [DOI] [PubMed] [Google Scholar]
- 26.Kelberman D, Turton JP, Woods KS, Mehta A, Al-Khawari M, Greening J, Swift PG, Otonkoski T, Rhodes SJ, Dattani MT. Molecular analysis of novel PROP1 mutations associated with combined pituitary hormone deficiency (CPHD). Clin Endocrinol (Oxf) 2009;70:96–103. doi: 10.1111/j.1365-2265.2008.03326.x. [DOI] [PubMed] [Google Scholar]
- 27.Wu W, Cogan JD, Pfaffle RW, Dasen JS, Frisch H, O'Connell SM, Flynn SE, Brown MR, Mullis PE, Parks JS, Phillips JA, 3rd, Rosenfeld MG. Mutations in PROP1 cause familial combined pituitary hormone deficiency. Nat Genet. 1998;18:147–149. doi: 10.1038/ng0298-147. [DOI] [PubMed] [Google Scholar]
- 28.Agarwal G, Bhatia V, Cook S, Thomas PQ. Adrenocorticotropin deficiency in combined pituitary hormone deficiency patients homozygous for a novel PROP1 deletion. J Clin Endocrinol Metab. 2000;85:4556–4561. doi: 10.1210/jcem.85.12.7013. [DOI] [PubMed] [Google Scholar]
- 29.Pernasetti F, Toledo SP, Vasilyev VV, Hayashida CY, Cogan JD, Ferrari C, Lourenco DM, Jr., Mellon PL. Impaired adrenocorticotropin-adrenal axis in combined pituitary hormone deficiency caused by a two-base pair deletion (301-302delAG) in the prophet of Pit-1 gene. J Clin Endocrinol Metab. 2000;85:390–397. doi: 10.1210/jcem.85.1.6324. [DOI] [PubMed] [Google Scholar]
- 30.Bottner A, Keller E, Kratzsch J, Stobbe H, Weigel JF, Keller A, Hirsch W, Kiess W, Blum WF, Pfaffle RW. PROP1 mutations cause progressive deterioration of anterior pituitary function including adrenal insufficiency: a longitudinal analysis. J Clin Endocrinol Metab. 2004;89:5256–5265. doi: 10.1210/jc.2004-0661. [DOI] [PubMed] [Google Scholar]
- 31.Voutetakis A, Maniati-Christidi M, Kanaka-Gantenbein C, Dracopoulou M, Argyropoulou M, Livadas S, Dacou-Voutetakis C, Sertedaki A. Prolonged jaundice and hypothyroidism as the presenting symptoms in a neonate with a novel Prop1 gene mutation (Q83X). Eur J Endocrinol. 2004;150:257–264. doi: 10.1530/eje.0.1500257. [DOI] [PubMed] [Google Scholar]
- 32.Arroyo A, Pernasetti F, Vasilyev VV, Amato P, Yen SS, Mellon PL. A unique case of combined pituitary hormone deficiency caused by a PROP1 gene mutation (R120C) associated with normal height and absent puberty. Clin Endocrinol (Oxf) 2002;57:283–291. doi: 10.1046/j.1365-2265.2002.01550.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Boot AM, Engels MA, Boerma GJ, Krenning EP, De Muinck Keizer-Schrama SM. Changes in bone mineral density, body composition, and lipid metabolism during growth hormone (GH) treatment in children with GH deficiency. J Clin Endocrinol Metab. 1997;82:2423–2428. doi: 10.1210/jcem.82.8.4149. [DOI] [PubMed] [Google Scholar]
- 34.Reynaud R, Gueydan M, Saveanu A, Vallette-Kasic S, Enjalbert A, Brue T, Barlier A. Genetic screening of combined pituitary hormone deficiency: experience in 195 patients. J Clin Endocrinol Metab. 2006;91:3329–3336. doi: 10.1210/jc.2005-2173. [DOI] [PubMed] [Google Scholar]
- 35.Alatzoglou KS, Dattani MT. Phenotype-genotype correlations in congenital isolated growth hormone deficiency (IGHD). Indian J Pediatr. 2012;79:99–106. doi: 10.1007/s12098-011-0614-7. [DOI] [PubMed] [Google Scholar]
- 36.Raivio T, Avbelj M, McCabe MJ, Romero CJ, Dwyer AA, Tommiska J, Sykiotis GP, Gregory LC, Diaczok D, Tziaferi V, Elting MW, Padidela R, Plummer L, Martin C, Feng B, Zhang C, Zhou QY, Chen H, Mohammadi M, Quinton R, Sidis Y, Radovick S, Dattani MT, Pitteloud N. Genetic overlap in Kallmann syndrome, combined pituitary hormone deficiency, and septo-optic dysplasia. J Clin Endocrinol Metab. 2012;97:E694–699. doi: 10.1210/jc.2011-2938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Dateki S, Kosaka K, Hasegawa K, Tanaka H, Azuma N, Yokoya S, Muroya K, Adachi M, Tajima T, Motomura K, Kinoshita E, Moriuchi H, Sato N, Fukami M, Ogata T. Heterozygous orthodenticle homeobox 2 mutations are associated with variable pituitary phenotype. J Clin Endocrinol Metab. 2010;95:756–764. doi: 10.1210/jc.2009-1334. [DOI] [PubMed] [Google Scholar]