Abstract
Little is known about whether and how multimodal representations of the body (BRs) and of the space around the body (Peripersonal Space, PPS) adapt to amputation and prosthesis implantation. In order to investigate this issue, we tested BR in a group of upper limb amputees by means of a tactile distance perception task and PPS by means of an audio-tactile interaction task. Subjects performed the tasks with stimulation either on the healthy limb or the stump of the amputated limb, while wearing or not wearing their prosthesis. When patients performed the tasks on the amputated limb, without the prosthesis, the perception of arm length shrank, with a concurrent shift of PPS boundaries towards the stump. Conversely, wearing the prosthesis increased the perceived length of the stump and extended the PPS boundaries so as to include the prosthetic hand, such that the prosthesis partially replaced the missing limb.
Our physical body represents the interface between the self and the external world, in that it mediates every interaction with external stimuli. The physical body is represented at different levels in the human brain, from unimodal somatosensory, motor and visual body maps1,2,3, to multimodal areas representing the shape, dimensions and position of body parts4,5,6,7,8 and the space immediately around them (Peripersonal Space, PPS)9,10,11,12. Brain systems involved in body and PPS representation have been localized within a common fronto-parietal network, encompassing the ventral premotor cortex and the posterior parietal cortex, both in monkeys13,14,15 and in humans16,17,18,19,20; see21 for a review. Body representations in the brain depend on the structure of the physical body, and must dynamically update to changes of the physical body. While there is extensive evidence of plasticity in unimodal body representations following a sudden change in the physical body, such as in the case of traumatic amputation22,23,24,25, little is known about the extent to which multimodal body and PPS representations are dependent on the structure of the physical body and dynamically adapt to changes in body structure26,27,28.
Here we study a group of 10 upper limb amputees to show how a sudden change in the structure of the physical body affects a critical feature of body representations (BRs), i.e., the perceived dimension of the residual body part, and the extension of PPS around the affected body part. In addition, the effects of amputation are partially palliated by means of prostheses that at the same time physically replace the amputated body part and extend the functionality of the residual limb. Little is known on whether and how partially restoring the function and structure of the physical body by means of prosthesis implantation affects BRs and PPS representations. To study this issue, we selected amputee patients who underwent traumatic amputation of an upper limb at least 24 months before testing, and were implanted with and used a functional prosthesis. Patients performed a tactile distance perception task in order to assess the perceived length of the stump and of the healthy arm (see Experiment 1) and an audio-tactile interaction task in order to measure the extent of PPS representation around the stump and the healthy limb (see Experiment 2). The comparison between the results for the two hemisoma provided evidence about the effects of amputation. The same experiments were also run while patients were or were not wearing their prosthesis during testing, and the results from these two conditions were compared in order to study the effect of prosthesis implantation on body and PPS representation.
Results
Experiment 1 - amputation and prosthesis implantation affect BR
In order to assess the perceived length of the residual part of the upper limb and of the homologous region of the healthy limb, we used a tactile distance perception task. In each trial, subjects received two pairs of tactile stimuli, one pair on the forehead (serving as a reference body part) and one pair on the upper arm (target body part). Participants were asked to judge whether the distance between the two stimuli was longer on the forehead or on the arm. In the present experiment, we administered the tactile distance perception task with tactile stimuli longitudinally delivered on the upper arm, along the arm axis, in order to measure the perceived length of the arm. Amputees performed the task in 3 blocked conditions, run in counterbalanced between-subjects order, on the healthy limb and on the stump, with or without the prosthesis. For each subject, we calculated the mean probability of reporting the distance on the upper arm as longer for all combinations of inter-point distances (P-Arm). We predicted that P-Arm would vary depending on the perceived size of the stimulated arm, in line with a context dependent bias, well documented in the field of visual perception [e.g.29,30], and suggested also for haptic exploration [e.g.31; but see also32 for a different interpretation] and recently confirmed also for a visual33,34 and a tactile distance perception task34. According to this bias, higher or lower P-Arm would indicate, respectively, that the upper arm is perceived as shorter or as longer.
We first compared mean P-Arm between the amputees' healthy limb and the right arm of healthy controls, by means of an independent samples t-test. Data were normally distributed (Kolmogorov-Smirnov: p = .20). Scores did not differ significantly between the two groups [t(12) = 1.41, p = .29] suggesting that amputation of one upper limb did not affect the implicitly perceived length of the healthy arm.
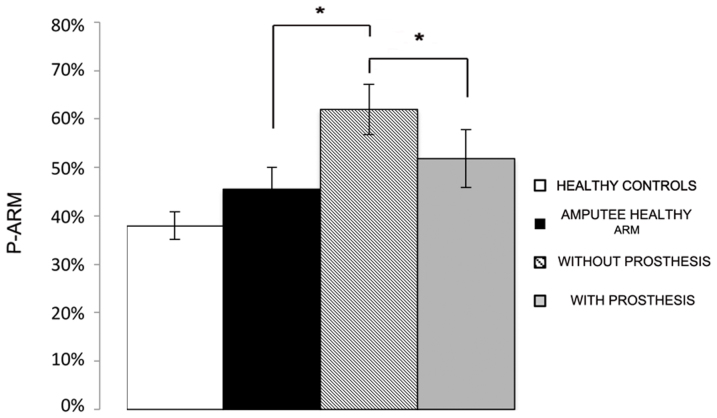
In order to study the effect of amputation and prosthesis implantation on perceived arm length, we compared mean P-Arm between the healthy and the amputated limb of patients, by means of a repeated measure ANOVA with Condition (Healthy limb, With Prosthesis and Without Prosthesis) as within-subjects factor. The main factor of Condition was significant [F(2,12) = 11.01; p < .01]. As shown in Figure 1, P-Arm was significantly higher when patients performed the task on the affected limb, while not wearing the prosthesis (62%, ±5%), as compared to the healthy limb (46%, ±4%; p < .001; Newman-Keuls corrected). In line with the context-dependent hypothesis, this result suggests that amputation reduced the perceived length of the remaining part of the affected limb. Critically, when patients performed the task on the stump, but while wearing their prosthesis, P-Arm was significantly lower (52%, ±6%) as compared to the Without Prosthesis condition (62%, ±5%; p < .05 - Newman-Keuls corrected). These results suggest that wearing the prosthesis increased the perceived length of the stump, making it more similar to the perception of the healthy limb, such that the prosthesis partially replaced the missing limb. P-Arm was not statistically different between the healthy limb and the stump while patients wore the prosthesis (p = .09).
Figure 1. Experiment 1 results.
The graph shows mean P-Arm for healthy controls (white column), amputee healthy arm (black column), amputated limb without prosthesis (hatched column) and amputated limb with prosthesis (grey column). Error bars denote S.E.M.
Experiment 2 - amputation and prostheses implantation affect PPS representation
In order to investigate whether amputation and prosthesis implantation affected also the extent of the multisensory space surrounding the limb, we compared patients' performance in the auditory-tactile interaction task35. By using this task, we measured the critical distance at which a sound interacts with the processing of tactile stimuli at the upper limb. Participants were asked to answer as fast as possible to a tactile stimulus administered on the upper arm, while task-irrelevant sounds were presented, giving the impression of a sound source either approaching to (IN sound), or receding from (OUT sound) their limb. On different trials, tactile stimulation was delivered at one out of five possible different temporal delays from the onset of the sound (from T1 to T5). This way, tactile stimulation occurred when the sound source was perceived at one out of five possible distances (from D1, very far, to D5 very close) from the body (see Methods). The extent of PPS was assessed in a group of healthy subjects as a control in the patients' group, on the healthy limb and on the amputated limb, while patients were wearing or not their prosthesis.
As tactile stimulation was set clearly above threshold, false alarms and omissions were rare (on average: .86% and 2.87%, per subject, per condition, respectively) and therefore the performance was analysed in terms of RTs only. In order to study the relationship between RTs and perceived sound position as a proxy of PPS extension, we calculated mean RTs to tactile target both for IN and OUT sounds at the various temporal delays at which tactile stimulation was administered. RTs exceeding more than 2 standard deviations from the mean RTs were considered outliers and trimmed from the analyses (on average: .81% of trials per subject per condition). At every temporal delay, from T1 to T5, sounds were perceived as being at a different position in space with respect to the stimulated body part. Given the symmetric shape of the two waveforms for the IN and OUT sounds and the equivalent segmentation of the different temporal delays from T1 to T5, there was a spatial correspondence between the perceived position of IN and OUT sounds. Specifically, at T1 IN and T5 OUT (farthest distance from the body), T2 IN and T4 OUT (far distance), T3 IN and T3 OUT (intermediate distance), T4 IN and T2 OUT (close distance), and T5 IN and T1 OUT (closest distance). Therefore, we averaged tactile RTs for these couples of delays and analysed RTs as a unique function of the five possible perceived distances, from D1, farthest distance, to D5, closest distance, along a continuum.
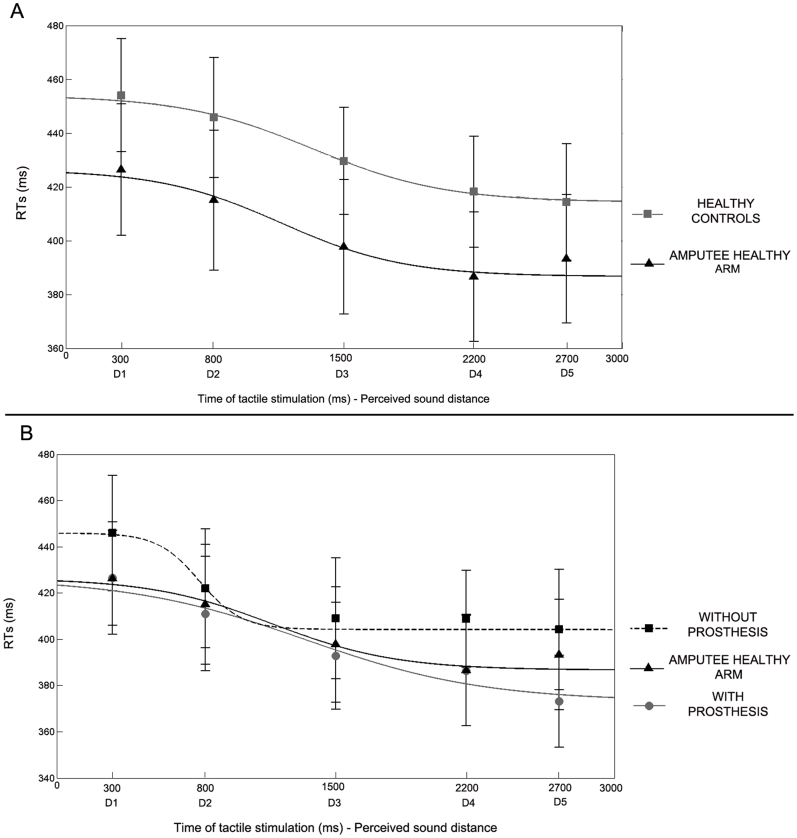
First of all, in order to study PPS representation around the healthy limb, we compared tactile RTs at each distance, from D1 to D5, when stimulation was administered to the amputees' healthy limb and to the arm of healthy controls. Data were normally distributed (Kolmogorov-Smirnov in 14 out of 15 conditions: p = .20). A repeated measures ANOVA with Distance (D1, D2, D3, D4, D5), as a within subject factor, and Group (Amputees healthy limb - Controls), as a between subject factor, revealed a significant main effect of Distance [F(4,72) = 17.31, p < .00001]. Results showed that tactile RTs progressively speeded up as the perceived sound approached the body (see Figure 2A). Newman-Keuls post-hoc tests confirmed that RTs at D1 (D1 = 440 ms, ±16) and D2 (430 ms, ±17) – when the sound was perceived as being far from the body – were significantly slower as compared to RTs at D3 (414 ms, ±16), D4 (402 ms, ±16) and D5 (404 ms, ±16; all ps < .01) – when the sound was perceived as being close to the body. This pattern of results was equivalent between patients and healthy controls. Indeed, neither the main effect of Group [F(1,18) = 0.81, p = .38] nor the Distance X Group interaction [F(4,72) = 0.32, p = .86] were significant. Taken together these results suggest that there is a critical spatial range (in this case between D2 and D3) within which auditory stimuli begin interacting with tactile stimuli administered on the body surface, resulting in quicker tactile RTs. This spatial range could be considered as the boundaries of the PPS. The present analysis suggests that the boundaries of PPS representation around the upper limb do not differ between healthy controls and amputees, for what concerned the non-affected side of their body.
Figure 2. Experiment 2 results.
(A) Mean RTs at different perceived sound distances (from D1 - farthest - to D5 - closest), corresponding to different time of tactile stimulus delivery and best fitting sigmoidal functions describing the relationship between RTs and sound distance, for healthy controls (grey line) and for amputee healthy arm (black line). Error bars denote S.E.M. (B) Mean RTs at different perceived sound distances (from D1 - farthest - to D5 - closest), corresponding to different time of tactile stimulus delivery and best fitting sigmoidal functions describing the relationship between RTs and sound distance, for amputee healthy arm (black line), amputated limb without-prosthesis (dotted line) and amputated limb with-prosthesis (grey line). Error bars denote S.E.M.
In order to study the effect of amputation and prosthesis implantation on PPS representation, we compared the results between patients' healthy and amputated limb, while wearing or not wearing their prosthesis. We entered tactile RTs in a repeated measure ANOVA with Condition (Healthy Limb, With Prosthesis and Without Prosthesis) and Distance (D1, D2, D3, D4, D5) as the within subject factors. The main effect of Distance was significant [F(4,36) = 19.92, p < .00001], resembling the pattern of responses found for the healthy limb and for controls: RTs became faster when the sound was perceived as being closer to the body (see Figure 2B). Critically, the main effect of Condition was also significant [F(2,18) = 3.64, p < .05]. A Newman-Keuls post-hoc test showed that when patients performed the task without the prosthesis, RTs were slower (418 ms, ±29) as compared to when they performed the task with the healthy limb (404 ms, ±29; p < .05; one-tailed). When patients performed the task with the amputated limb, RTs were faster when they wore the prosthesis (398 ms, ±26) as compared to when they did not wear the prosthesis (p < .05). RTs were not significantly different between the Healthy limb and the With Prosthesis conditions (p = .45). Taken together these results suggest that amputation affected PPS representation around the stump, as compared to the non-affected limb. Wearing a prosthesis compensated this effect, making PPS representation around the stump more similar to PPS representation around the healthy limb.
Experiment 3 - prosthesis implantation shifts PPS coding from the stump to the prosthetic hand
In order to interpret the differential effect on RTs found between the with - and without prosthesis conditions in amputee patients, we hypothesized that, when patients did not wear the prosthesis, sound position was codified with respect to the stump, which represented the boundaries of the physical body. Instead, when patients wore their prosthesis, the perceived position of sound in space was re-calibrated with respect to the prosthetic hand, such that the prosthetic hand itself represented the new body boundary. In other words, audio-tactile interaction was coded with respect to the stump in the Without Prosthesis condition and with respect to the prosthetic hand in the With Prosthesis condition, resulting in a general reduction of RTs in every temporal delay (i.e., at each sound distance) when the prosthesis was on. In order to test this hypothesis, we ran a further experiment on healthy participants. The task was the same as for Experiment 2, in exception for a manipulation in the location of tactile stimuli. Sound positions were kept constant in relationship to the upper arm (i.e., the near loudspeaker was close to the upper arm and the far loudspeaker was at 100 cm). Tactile targets, however, were administered in two different experimental conditions either at the arm (Upper Arm condition), or at the hand (Hand conditions). This way we aimed at simulating, respectively, the stump stimulation in the Without Prosthesis condition and the recoding of auditory-tactile interaction to the prosthetic hand in the With Prosthesis condition in amputee patients.
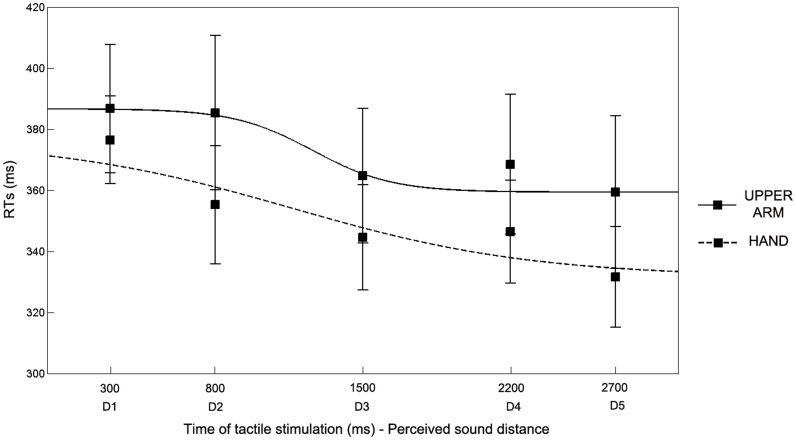
Subjects performed two blocks for each experimental condition (Upper Arm and Hand condition), ran in a counterbalanced order. False alarms and omissions were rare (on average: .45 and 1.89%, per subject, per condition, respectively). Data were normally distributed (Kolmogorov- Smirnov in 9 out of 10 conditions, p>.09). Mean RTs (after trimming outliers, .54% of trails per subject per condition) to tactile stimulation were entered in a repeated-measures ANOVA with Condition (Upper Arm – Hand) and Distance (from D1 to D5), as within subject factors, and Order of administration (Upper Arm – Hand; Hand – Upper Arm) as between subject factor. As in the previous experiment, the main effect of Distance was significant [F(4,40) = 11.64, p < .0001], replicating the modulation of tactile RTs depending on the position of sound in space (see Figure 3). The Condition X Distance interaction was not significant [F(4,40) = 1.56, p = .20]. Critically, also the main effect of Condition was significant [F(1,10) = 5.47, p < .05]. Newman-Keuls post-hoc tests showed that when subjects performed the task while receiving the tactile stimulation on their hand they were faster (351 ms, ±15) in every temporal delay as compared to when they received the tactile stimulation on their upper arm (373 ms, ±21). These results suggest that when the tactile stimulation was administered at the hand, while the near sound source was placed close to the upper arm, sounds were processed as if they were closer to the boundaries of the stimulated limb, i.e., the hand. This pattern of results evidenced a general reduction of tactile RTs in every temporal delay as compared to when tactile stimuli were administered at the upper arm. This effect suggests that the perceived position of sound was computed with respect to the part of the limb tactilely stimulated and clearly resembles the effect found in amputee patients when task was performed while wearing, as compared to not wearing, the prosthesis.
Figure 3. Experiment 3 results.
Mean RTs at different perceived sound distances (from D1 - farthest - to D5 - closest), corresponding to different time of tactile stimulus delivery and best fitting sigmoidal functions describing the relationship between RTs and sound distance, for Arm condition (black line) and Hand condition (dotted line). Error bars denote S.E.M.
Discussion
Two main results have been obtained by the present study. First, a modification in the physical structure of the body, such as limb loss due to traumatic amputation, affects high-order multisensory representations of the body and of the space around the body. Second, such effects are, at least partially, compensated by prosthesis implantation substituting the lost body part. Results from Experiment 1 show that, following amputation, the implicitly perceived length of the residual part of the upper arm decreased, such that patients perceived their stump as shorter as compared to the healthy arm. Wearing a prosthesis increased the perceived length of the arm, making the perception of the stump length more similar to that of the healthy limb. Amputation and prosthesis implantation also affected the representation of PPS around the stump. In Experiment 2, task-irrelevant sounds boosted tactile RTs in so far as they were perceived as being closer to the stimulated body part, both in healthy controls and in amputees tested on their healthy limb. This multisensory effect was reduced when amputated patients were tested on their amputated limb (without-prosthesis); in this condition patients showed slower RTs as compared to conditions involving healthy limb assessment or healthy controls, suggesting that, after amputation, the boundaries of PPS shifted towards the stump. However, when the task was administered on the stump while patients wore their prosthesis, there was again the same speeding effect on tactile RTs depending on the position of sounds in space, as for the healthy limb and in controls. These results suggested that prosthesis implantation restored the boundaries of PPS so that they included the prosthetic hand. Experiment 3 supports this conclusion, by showing that in healthy subjects, dynamic sounds coupled to tactile stimulation of the upper arm or of the hand resulted in the same modulation of tactile RT similar as that found in amputees respectively when they did not wear or they wore their prosthesis.
Until now, an extensive body of evidence has demonstrated that amputation modifies unimodal motor and somatosensory representations of the body in the brain, both in monkeys22,36,37 and in humans8,23,24,25 suggesting a strong dependency of unimodal body representations on the structure of the physical body. Phantom limb phenomena, such as tactile sensations arising from the missing limb or feeling of moving the amputated limb, occur at some point in almost every individual suffering amputation. These phenomena offer a striking example of the link between plasticity in primary cortical body representations and body experience38; see8 for review. Interestingly, phantom limb phenomena are not limited to sensory or motor precepts originating from the missing body part39, but are often referred by patients as conscious awareness of the presence - implying position, shape and size - of the missing limb39,40. The complexity and richness of these phantom limb phenomena is hardly explainable as resulting only from cortical reorganization in unimodal primary cortices. Rather, they suggest an involvement of multisensory body representations, which integrate the continuous flow of information from different sensory modalities in order to give raise to the experience of the body and its parts41,42. Surprisingly little is known yet about the extent to which multisensory body and space representations are dependent on the structure of the physical body and are affected by limb loss. By showing that upper limb amputation changes the perceived dimension of the residual limb and alters multisensory integration in the space surrounding the limb, the present study offers evidence of plasticity in multisensory body representations following a change in the physical structure of the body.
The present results also provide new insights about the direction of these effects. Indeed, shrinkage of a part of the physical body following amputation resulted in contraction of multisensory body and space representations. When participants did not wear their prosthesis, amputee patients perceived their stump as shorter and PPS representation around the stump shrunk. Previous studies provided evidence of plastic modifications of both body and PPS representations in the direction of extension34,43,44,45,46. Interestingly, there is much less evidence of contraction of body and PPS representation [e.g., see47,48]. Since development tends in the direction of growing and cannot normally be reversed, it makes sense that extension phenomena are more common and more easily demonstrated than contraction phenomena. Limb loss represents a rare means to study plasticity in body representations in the direction of shrinkage. Wearing a prosthesis, however, partially replaces the physical presence of the amputated limb and partially re-establishes its function of acting in space. In line with this, the second main finding of the present study is that while wearing a functional prosthesis, long-term prosthesis-users, such as the patients of the present sample, perceived their stump as longer and the boundaries of PPS representation shifted to include the region around their prosthetic hand.
In order to interpret these plasticity effects in multimodal body and space representations after amputation (in the direction of contraction) and prosthesis implantation (in the direction of extension) we refer to the well-documented sensory-to-motor functions of body and PPS representations. It is well known that both in monkeys10,49,50 and in humans51,52 multisensory fronto-parietal areas responding selectively to stimuli within PPS are directly linked to the motor system in order to trigger fast and appropriate motor reactions to stimuli potentially interacting with body. In the same way, information related to the size and position of different body parts is critical for action upon external objects. Consistent with this view, we showed that when the possibility of acting with a limb is limited, due to amputation, body and PPS representations contract. Instead, when such possibility is, at least partially, restored by using a functional prosthesis, BRs and PPS representations extend, such that they incorporate the prosthesis into the representation of the upper limb. These results are in line with a recent study from Makin and colleagues28 showing that hand amputation, by producing an asymmetry in action space, also caused a permanent distortion in visuo-spatial perception on the affected side of the body, as compared to the intact side.
The prosthesis' function of restoring the possibility of the body to act in its space in amputees resembles that of a tool in extending the reachable space of healthy subjects. However a prosthesis is more than a tool in that the majority of the prosthetic limbs, differentially from tools, also mimic the visual appearance of a limb. The prostheses used by the present sample of patients were artificial devices that faithfully reproduced the exterior appearance of a real arm and hand; they were also controlled myoelectrically or kinematically by residual muscles in order to allow quite complex limb movements. Since prosthetic limbs share more features with an anatomical limb as compared to tools, prosthesis-use and tool-use could have differential effects on the plasticity of BRs. In particular, distinction has been proposed between the effects of body extension (e.g., in the case of tool-use) and body incorporation (e.g., in the case of prosthesis-use)53,54. It is still not clear, however, whether and to what extent a prosthetic limb can be embodied such that it becomes in some sense indistinguishable from a real body part. We did not directly test the subjective experience of prosthesis embodiment, but the present results suggest that although the effect of prosthesis implantation might overcome that of tool-use, prosthetic limbs cannot be totally conceived as a real part the body, at least because they are known to be attachments that can be taken off. Accordingly, our data show that in amputee patients, two different body representations coexisted and were differentially activated when patients did or did not wear their prosthesis. Indeed, the perceived length of the stump and the extension of PPS immediately shrunk or elongated, depending on whether the prosthesis was respectively on or off. The coexistence of multiple body representations depending on different body states (with or without a tool) resembles other forms of plasticity shown after long-term tool-use experiences55,56.
Understanding the mechanisms of prostheses embodiment and identifying key features of prosthetic devices favouring prosthesis-use and acceptance are key issues for rehabilitation of limb loss and the new field of neuroprosthetics. The present study might contribute to research in this field: on the one hand, it demonstrates striking effects of amputation and prosthesis implantation on the perception of body part size and on multisensory integration in the space around the body. On the other hand, it proposes sensitive and easy-to-apply tasks to measure the effects of using prosthetic devices on BRs and PPS representations.
Methods
Amputee subjects
Ten volunteers participated in the study (8 males and 2 females, mean age 45 years, range 21–66 years), recruited at the INAIL Prostheses Centre, Budrio, Bologna (http://www.inail-ricerca.it/index.aspx). They were healthy except that they had all one upper limb amputated either below or above the elbow, following a traumatic accident. Before the accident all patients were right-handed. The inclusion criterion was that they must have been using a functional prosthesis, cosmetically designed, so they resembled arm appearance, at least 4–8 h daily for 5–7 days per week for at least 1 year. Patients' demographic and clinical data are reported in Table 1. All subjects gave their informed consent to participate in the study, which was approved by the Ethical commission of the INAIL Prostheses Centre and performed in accordance with the Declaration of Helsinki.
Table 1. Patients' demographic and clinical data. All patients but P1, P3 and P10 participated in Experiment 1. All patients participated in Experiment 2.
| Patient | Age | Gender | Handiness | Amputation side, level | Prosthesis type | Years since amputation | Phantom limb symptoms |
|---|---|---|---|---|---|---|---|
| P1 | 41 | F | Dx | Right, above elbow | Kinematic | 2 | Yes |
| P2 | 56 | M | Dx | Left, above elbow | Myoelectric | 21 | Yes |
| P3 | 50 | M | Dx | Right, above elbow | Myoelectric | 18 | No |
| P4 | 30 | M | Dx | Right, below elbow | Kinematic | 1.5 | No |
| P5 | 21 | F | Dx | Right, below elbow | Kinematic | 2.5 | Yes |
| P6 | 38 | M | Dx | Right, below elbow | Myoelectric | 18 | No |
| P7 | 66 | M | Dx | Right, below elbow | Myoelectric | 16 | Yes |
| P8 | 62 | M | Dx | Right, below elbow | Myoelectric | 42 | No |
| P9 | 41 | M | Dx | Right, below elbow | Kinematic | 4 | No |
| P10 | 43 | M | Dx | Left, above elbow | Kinematic | 3 | Yes |
Control subjects
Twenty-nine healthy volunteers participated in Experiment 1 (N = 7, 5 males, mean age = 33.7 years, range 25–62 years), Experiment 2 (N = 10, all females, mean age = 22.1 years, range 19–24 years) and Experiment 3 (N = 12, 2 males, mean age = 23.4 years, range 20–26 years) as control groups. All subjects gave their informed consent to participate in the study, which was approved by the Ethical commission of the INAIL Prostheses Centre and performed in accordance with the Declaration of Helsinki.
Experimental procedure: overall structure
Amputee patients participated in two experiments, assessing BRs (Experiment 1) and PPS representations (Experiment 2), performed in a single 2-hour and a half session. Seven out of ten patients participated in Experiment 1: two patients were excluded because of the very high level of the amputation (above the elbow; P1 and P10, see Table 1), thus not allowing administering tactile stimuli of sufficient dimensions (see Experiment 1 methods), because the prosthesis covered most of the stump surface. One patient could not perform the experiment for matter of time (P3, see Table 1). All patients participated in Experiment 2.
Patients performed Experiment 1 and Experiment 2 in three different experimental conditions: stimuli were administered on the healthy limb (Healthy limb condition), on the amputated limb without prosthesis (Without Prosthesis condition) and on the amputated limb with prosthesis (With Prosthesis condition). All healthy subjects from the control group performed the tasks in Experiment 1 and 2 on their right upper arm, on a skin region matching the site of stump stimulation used for amputee patients.
Procedure and materials
Experiment 1 - tactile distance perception task
In order to assess the perceived length of the residual part of the upper limb and of the homologous region of the healthy limb, we used a tactile distance perception task. On each trial of the tactile distance perception task, participants were touched with a pair of dots on the forehead and then with a pair of dots on the upper arm, longitudinally delivered along the arm axis. Participants made untimed two-alternative forced-choice judgments of whether the two points felt farther apart on the forehead or on the upper arm, responding verbally “forehead” or “arm”. Subjects were lain down with the tested arm resting in a prone position. In order to set the spatial distance between the stimuli administered to the forehead and the upper arm, we initially measured the two-point discrimination threshold (2 pdt) on the upper arm for longitudinal orientation by using a staircase method, as described in57. For each subject, the individual 2 pdt on the arm was used to set the distance between the pairs of dots used during the tactile distance task. Three different inter-point distances were used: at the 2 pdt; 1.5 the 2 pdt; and twice the 2 pdt. The task comprised a total of 36 trials. An experimenter administered the stimuli manually for approximately one second, with an inter-stimulus interval of one second between taps on the forehead and the arm. Subjects were blindfolded throughout the procedure.
The perceived size of tactile stimuli touching the body depends on the perceived dimension of the body part tactilely stimulated6,7,58,59 and is influenced by visual33 or proprioceptive60 information about the stimulated body part. Thus, the tactile distance perception task can be used as an indirect measure of the internal representation of body part size [see also34].
Experiment 2 and 3- audio-tactile interaction task
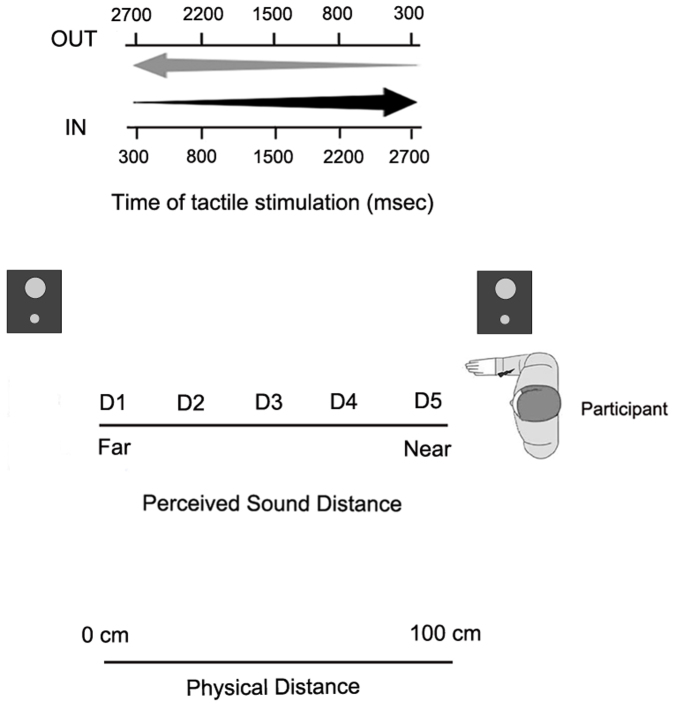
During the task, subjects were blindfolded and were seated with the tested arm resting prone on a table beside them. During each trial a sound (pink noise) was presented for 3000 ms. The sounds were generated by two loudspeakers, one placed on the table in proximity of the upper arm, the other one placed on the table, at a distance of ~100 cm from the near loudspeaker, thus far from the upper arm. Sound intensity was manipulated so that IN sounds had exponentially rising acoustic intensity, while OUT sounds had exponentially falling acoustic intensity. In this way, IN sounds gave the impression of a sound source moving from the far to the near loudspeaker, i.e., towards the subject's body, while OUT sounds gave the impression of a sound source moving in the opposite direction, i.e. receding from the body. Along with the auditory stimulation, in half of the trials subjects were also presented with a tactile stimulation, delivered by means of a constant-current electrical stimulator (DS7A, Digitimer, Hertfordshire, United Kingdom), via a pair of neurological electrodes (Neuroline, Ambu, Ballerup, Denmark), placed on the dorsal surface of the upper arm. The remaining trials were catch trials with auditory stimulation only. Subjects were asked to respond vocally to the tactile target, when present, saying “TAH” as rapidly as possible, trying to ignore the sound. Tactile RTs were recorded by means of a voice-activated relay. A PC running C.I.R.O. software (www.cnc.unibo.psice.unibo/ciro) was used to control the presentation of the stimuli and to record responses [see35].
The critical experimental manipulation was that the tactile stimulus was delivered at different temporal delays (from T1 to T5, see below) from the onset of the auditory stimulus, both for IN and OUT sounds. In this way, tactile stimulation occurred when the sound source was perceived at different locations with respect to the body: i.e., close to the body, at high temporal delays for the IN sound and at low temporal delays for the OUT sound; and far from the body, at low temporal delays for the IN sound and at high temporal delays for the OUT sound. For each trial, the sound was preceded and followed by 1000 ms of silence. Temporal delays from sound onset for the tactile stimulus were set as follows: T1, tactile stimulation administered at 300 ms, T2 at 800 ms, T3 at 1500 ms, T4 at 2200 ms and T5 at 2700 ms (see Figure 4).
Figure 4. Experimental set up for Experiment 2 and Experiment 3.
The task used for Experiment 2 and Experiment 3 consisted in a random combination of 8 target stimuli for each temporal delay, for the IN and OUT sounds, resulting in a total of 76 trials with a tactile target, randomly intermingled with 76 catch trials. Trials were equally divided into 2 blocks, each block lasting about 8 minutes.
Funding
This work was supported by a Volkswagen Stiftung grant (the UnBoundBody project, ref. 85 639) to AS and by a Spinner 2013 grant (Prosthesis and brain) by Regione Emilia Romagna to EC and AS.
Author Contributions
The experiment was conceived by AS and EC. Data were collected by EC and MM, and they have been analysed by EC. Recruiting of the patients was done by AA and GV. The manuscript text was written by EC and AS.
Acknowledgments
The authors thank patients for their participation in the study, Angelo Davalli for his support and collaboration, Francesca Baldassarri for patients recruitment, Valentina Tedeschi for her contribution in data collection, Adrian Smith and Ana Tajadura for their comments and Elisa Magosso for her help in data analysis.
References
- Penfield W. & Boldrey E. Somatic motor and sensory representation in the cerebral cortex of man as studied by electrical stimulation. Brain. 60, 389–443 (1937). [Google Scholar]
- Downing P. E., Jiang Y., Shuman M. & Kanwisher N. A cortical area selective for visual processing of the human body. Science. 293, 2470–2473 (2001). [DOI] [PubMed] [Google Scholar]
- Orlov T., Makin T. R. & Zohary E. Topographic representation of the human body in the occipitotemporal cortex. Neuron. 68, 586–600 (2010). [DOI] [PubMed] [Google Scholar]
- Head H. & Holmes G. Sensory disturbances from cerebral lesions. Brain. 34, 102 (1911). [Google Scholar]
- Gallagher S. How the Body Shapes the Mind. New York: Oxford University Press. 284, 0199271941 (2005).
- Medina J. & Coslett H. B. From maps to form to space: touch and the body schema. Neuropsychologia. 48, 645–654 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Longo M. R., Azanon E. & Haggard P. More than skin deep: body representation beyond primary somatosensory cortex. Neuropsychologia. 48, 655–668 (2010). [DOI] [PubMed] [Google Scholar]
- Serino A. & Haggard P. Touch and the body. Neurosci Biobehav Rev. 34, 224–236 (2010). [DOI] [PubMed] [Google Scholar]
- Rizzolatti G., Fadiga L., Fogassi L. & Gallese V. The space around us. Science. 277, 190–191 (1997). [DOI] [PubMed] [Google Scholar]
- Graziano M. S. & Cooke D. F. Parieto-frontal interactions, personal space, and defensive behavior. Neuropsychologia. 44, 845–859 (2006). [DOI] [PubMed] [Google Scholar]
- Làdavas E. Functional and dynamic properties of visual peripersonal space. Trends Cogn Sci. 6, 17–22 (2002). [DOI] [PubMed] [Google Scholar]
- Làdavas E. & Serino A. Action-dependent plasticity in peripersonal space representations. Cogn Neuropsychol. 25, 1099–1113 (2008). [DOI] [PubMed] [Google Scholar]
- Duhamel J. R., Colby C. L. & Goldberg M. E. Ventral intraparietal area of the macaque:congruent visual and somatic response properties. J Neurophysiol 79, 126–136 (1998) [DOI] [PubMed] [Google Scholar]
- Graziano M. S. & Cooke D. F. Parieto-frontal interactions, personal space, and defensive behavior. Neuropsychologia 44, 845–859 (2006). [DOI] [PubMed] [Google Scholar]
- Graziano M. S. A. & Botvinick M. M. How the brain represents the body: insights from neurophysiology and psychology. In: Common Mechanisms in Perception and Action: Attention and Performance XIX. Eds. Prinz, W. and Hommel, B. Oxford England, pp. 136–157 (2002).
- Bremmer F., Schlack A., Shah N. J., Zafiris O., Kubischik M., Hoffmann K. P. et al. Polymodal Motion Processing in Posterior Parietal and Premotor Cortex: A Human fMRI Study Strongly Implies Equivalencies between Humans and Monkeys. Neuron 29, 287–296 (2001). [DOI] [PubMed] [Google Scholar]
- Filimon F., Nelson J. D., Huang R. S., Sereno M. I. Multiple parietal reach regions in humans: cortical representations for visual and proprioceptive feedback during on-line reaching. J Neurosci 29, 2961–71 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makin T. R., Holmes N. P., Zohary E. Is That Near My Hand? Multisensory Representation of Peripersonal Space in Human Intraparietal Sulcus. J Neurosci 27, 731–740 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sereno M. I. & Huang R. S. A human parietal face area contains aligned head-centered visual and tactile maps. Nat Neurosci 9, 1337–1343 (2006). [DOI] [PubMed] [Google Scholar]
- Serino A., Canzoneri E., Avenanti A. Fronto-parietal Areas Necessary for a Multisensory Representation of Peripersonal Space in Humans: An rTMS Study. J Cogn Neurosci 23, 2956–2967 (2011). [DOI] [PubMed] [Google Scholar]
- Blanke O. Multisensory brain mechanisms of bodily self-consciousness. Nat Neurosci Rev 13, 556–571 (2012). [DOI] [PubMed] [Google Scholar]
- Buonomano D. V. & Merzenich M. M. Cortical plasticity: from synapses to maps. Annu Rev Neurosci. 21, 149–186 (1998). [DOI] [PubMed] [Google Scholar]
- Chen R., Corwell B., Yaseen Z., Hallett M. & Cohen L. G. Mechanisms of cortical reorganization in lower-limb amputees. J Neurosci. 18, 3443–3450 (1998). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reilly K. T. & Sirigu A. The motor cortex and its role in phantom limb phenomena. Neuroscientist. 14, 195–202 (2008). [DOI] [PubMed] [Google Scholar]
- Borsook D., Becerra L., Fishman S., Edwards A., Jennings C. L. et al. Acute plasticity in the human somatosensory cortex following amputation. Neuroreport. 9, 1013–1017 (1998). [DOI] [PubMed] [Google Scholar]
- Nico D., Daprati E., Rigal F., Parsons L. & Sirigu A. Left and right hand recognition in upper limb amputees. Brain. 127, 120–132 (2004). [DOI] [PubMed] [Google Scholar]
- Ehrsson H. H., Rosen B., Stockselius A., Ragno C., Kohler P. et al. Upper limb amputees can be induced to experience a rubber hand as their own. Brain. 131, 3443–3452 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makin T. R., Wilf M., Schwartz I., Zohary E. Amputees “neglect” the space near their missing hand. Psychol Sci. 21, 55–7 (2010). [DOI] [PubMed] [Google Scholar]
- Ebbinghaus H. Memory: A Contribution to Experimental Psychology. New York, NY: Dover Publications (1987). [DOI] [PMC free article] [PubMed]
- Linkenauger S. A., Ramenzoni V. & Proffitt D. R. Illusory shrinkage and growth: body-based rescaling affects the perception of size. Psychol Sci. 21, 1318–25 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linkenauger S. A., Witt J. K. & Proffitt D. R. Taking a hands-on approach: apparent grasping ability scales the perception of object size. J Exp Psychol Hum Percept Perform. 37, 1432–41 (2011). [DOI] [PubMed] [Google Scholar]
- Bruno N. & Bertamini M. Haptic perception after a change in hand size. Neuropsychologia. 48, 1853–6 (2010) [DOI] [PubMed] [Google Scholar]
- Taylor-Clarke M., Jacobsen P. & Haggard P. Keeping the world a constant size: object constancy in human touch. Nat Neurosci. 7, 219–220 (2004). [DOI] [PubMed] [Google Scholar]
- Canzoneri E., Ubaldi S., Rastelli V., Finisguerra A., Bassolino M. & Serino A. Tool-use reshapes the boundaries of body and peripersonal space representations. Exp Brain Res. 228, 25–42 (2013). [DOI] [PubMed] [Google Scholar]
- Canzoneri E., Magosso E. & Serino A. Dynamic sounds capture the boundaries of peripersonal space representation in humans. PLoS One. 7, e44306 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu C. W. & Kaas J. H. Reorganization in primary motor cortex of primates with long-standing therapeutic amputations. J Neurosci. 19, 7679–7697 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merzenich M. M., Nelson R. J., Stryker M. P., Cynader M. S., Schoppmann A. et al. Somatosensory cortical map changes following digit amputation in adult monkeys. J Comp Neurol. 224, 591–605 (1984). [DOI] [PubMed] [Google Scholar]
- Karl A., Birbaumer N., Lutzenberger W., Cohen L. G. & Flor H. Reorganization of motor and somatosensory cortex in upper extremity amputees with phantom limb pain. J Neurosci. 21, 3609–3618 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunter J. P., Katz J. & Davis K. D. The effect of tactile and visual sensory inputs on phantom limb awareness. Brain. 126, 579–589 (2003). [DOI] [PubMed] [Google Scholar]
- Flor H., Nikolajsen L. & Staehelin Phantom limb pain: a case of maladaptive CNS plasticity? Nat Rev Neurosci. 7, 873–881 (2006). [DOI] [PubMed] [Google Scholar]
- Ehrsson H. H. The Concept of Body Ownership. In: Stein, B. E., editor. The New Handbook of Multisensory Processing. MIT Press (in press).
- Ionta S., Gassert R. & Blanke O. Multi-sensory and sensorimotor foundation of bodily self-consciousness - an interdisciplinary approach. Front Psychol. 2, 383 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maravita A., Spence C., Kennett S. & Driver J. 2002 Tool-use changes multimodal spatial interactions between vision and touch in normal humans. Cognition. 83, B25–34 (2002). [DOI] [PubMed] [Google Scholar]
- Holmes N. P., Calvert G. A. & Spence C. Extending or projecting peripersonal space with tools? Multisensory interactions highlight only the distal and proximal ends of tools. Neurosci Lett. 372, 62–67 (2004). [DOI] [PubMed] [Google Scholar]
- Farnè A. & Làdavas E. Dynamic size-change of hand peripersonal space following tool use. Neuroreport. 11, 1645–1649 (2000). [DOI] [PubMed] [Google Scholar]
- Maravita A., Husain M., Clarke K. & Driver J. Reaching with a tool extends visual-tactile interactions into far space: evidence from cross-modal extinction. Neuropsychologia. 39, 580–585 (2001). [DOI] [PubMed] [Google Scholar]
- Di Russo F., Committeri G., Pitzalis S., Spitoni G., Piccardi L. et al. Cortical plasticity following surgical extension of lower limbs. NeuroImage. 30, 172–183 (2006). [DOI] [PubMed] [Google Scholar]
- Longo M. R., Kammers M. P., Gomi H., Tsakiris M. & Haggard P. Contraction of body representation induced by proprioceptive conflict. Curr Biol. 19, R727–8 (2009). [DOI] [PubMed] [Google Scholar]
- Graziano M. S., Taylor C. S. & Moore T. Complex movements evoked by microstimulation of precentral cortex. Neuron. 34, 841–851 (2002). [DOI] [PubMed] [Google Scholar]
- Stepniewska I., Fang P. C. & Kaas J. H. Microstimulation reveals specialized subregions for different complex movements in posterior parietal cortex of prosimian galagos. Proc Natl Acad Sci USA. 102, 4878–4883 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serino A., Annella L. & Avenanti A. Motor properties of peripersonal space in humans. PLoS One. 4, e6582 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makin T. R., Holmes N. P., Brozzoli C., Rossetti Y. & Farnè A. Coding of visual space during motor preparation: Approaching objects rapidly modulate corticospinal excitability in hand-centered coordinates. J Neurosci. 29, 11841–11851 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Preester H. & Tsakiris M. 2009 Body-extension versus body-incorporation: Is there a need for a body-model? Phenom Cogn Sci. 8, 307–319. [Google Scholar]
- Giummarra M. J., Gibson S. J., Georgiou-Karistianis N. & Bradshaw J. L. Mechanisms underlying embodiment, disembodiment and loss of embodiment. Neurosci Biobehav Rev. 32, 143–60 (2008). [DOI] [PubMed] [Google Scholar]
- Serino A., Bassolino M., Farnè A. & Làdavas E. 2007 Extended multisensory space in blind cane users. Psychol Sci. 18, 642–648 (2007). [DOI] [PubMed] [Google Scholar]
- Bassolino M., Serino A., Ubaldi S. & Làdavas E. Everyday use of the computer mouse extends peripersonal space representation. Neuropsychologia. 48, 803–811 (2010). [DOI] [PubMed] [Google Scholar]
- Serino A., Padiglioni S., Haggard P. & Làdavas E. 2009 Seeing the hand boosts feeling on the cheek. Cortex. 45, 602–9. [DOI] [PubMed] [Google Scholar]
- de Vignemont F., Majid A., Jola C. & Haggard P. Segmenting the body into parts: evidence from biases in tactile perception. Q J Exp Psychol (Colchester). 62, 500–512 (2009). [DOI] [PubMed] [Google Scholar]
- Longo M. R. & Haggard P. 2011 Weber's illusion and body shape: Anisotropy of tactile size perception on the hand. J Exp Psychol Hum Percept Perform. 37, 720–726 (2011). [DOI] [PubMed] [Google Scholar]
- de Vignemont F., Ehrsson H. H. & Haggard P. Bodily illusions modulate tactile perception. Curr Biol. 15, 1286–1290 (2005). [DOI] [PubMed] [Google Scholar]