Abstract
Salinity is among the environmental factors that affect plant growth and development and constrain agricultural productivity. Salinity stress triggers increases in cytosolic free Ca2+ concentration ([Ca2+]i) via Ca2+ influx across the plasma membrane. Salinity stress, as well as other stresses, induces the production of reactive oxygen species (ROS). It is well established that ROS also triggers increases in [Ca2+]i. However, the relationship and interaction between salinity stress-induced [Ca2+]i increases and ROS-induced [Ca2+]i increases remain poorly understood. Using an aequorin-based Ca2+ imaging assay we have analyzed [Ca2+]i changes in response to NaCl and H2O2 treatments in Arabidopsis thaliana. We found that NaCl and H2O2 together induced larger increases in [Ca2+]i in Arabidopsis seedlings than either NaCl or H2O2 alone, suggesting an additive effect on [Ca2+]i increases. Following a pre-treatment with either NaCl or H2O2, the subsequent elevation of [Ca2+]i in response to a second treatment with either NaCl or H2O2 was significantly reduced. Furthermore, the NaCl pre-treatment suppressed the elevation of [Ca2+]i seen with a second NaCl treatment more than that seen with a second treatment of H2O2. A similar response was seen when the initial treatment was with H2O2; subsequent addition of H2O2 led to less of an increase in [Ca2+]i than did addition of NaCl. These results imply that NaCl-gated Ca2+ channels and H2O2-gated Ca2+ channels may differ, and also suggest that NaCl- and H2O2-evoked [Ca2+]i may reduce the potency of both NaCl and H2O2 in triggering [Ca2+]i increases, highlighting a feedback mechanism. Alternatively, NaCl and H2O2 may activate the same Ca2+ permeable channel, which is expressed in different types of cells and/or activated via different signaling pathways.
Introduction
The presence of high salinity affects almost every aspect of plant growth and development, and causes enormous losses in agricultural production worldwide. It is estimated that about 10 million hectares of agricultural land is abandoned every year because of high salinity, and salt stress affects as much as a quarter to a third of global agricultural land, particularly land which has been irrigated [1-3]. Given the continued increase in human population occurring in the world, it is estimated that crop production must be increased 50% by 2025 to stave off large-scale food shortages [4]. Thus, it is crucial to understand how plants respond to salt stress.
Many studies have been carried out to dissect the molecular and genetic mechanisms of the plant response to salt (NaCl) stress, often using the model organism Arabidopsis thaliana [5-7]. Excess NaCl is toxic to plants, causing cellular ion imbalances and hyperosmotic stress [1-3,7]. NaCl stress also triggers a calcium signaling cascade in plants, leading to transcriptional regulation and subsequent physiological and developmental responses [1]. Although the molecular nature of initial perception of salt stress is unknown, it has been well established that salt stress triggers a transient increase in cytosolic Ca2+ concentration ([Ca2+]i) that lasts about 2 min [8,9]. This increase has been proposed to represent a salt sensory process in plants [3,10].
In plants, Ca2+ as a secondary messenger is a key element to understanding a sophisticated network of signaling pathways responding to a large array of abiotic and biotic stimuli, including salt stress [11-13]. These specific Ca2+ signatures are formed by the tightly regulated activities of Ca2+ channels and transporters in different tissues, organelles and membranes [13-16], and the changes in [Ca2+]i are detected by cytosolic Ca2+ sensors. More than 250 Ca2+-binding EF-hand proteins have been identified in Arabidopsis [17], including the calmodulin (CaM), the calmodulin-like (CML), the Ca2+-dependent protein kinase (CDPK), and the calcineurin B-like (CBL) protein families. These cytosolic Ca2+ sensors decode and relay the information encoded within [Ca2+]i signatures, allowing the plant to tightly bring about the appropriate adaptation to its ever-changing environment.
The salinity stress-induced increase in [Ca2+]i leads to the activation of SOS3/CBL4, which functions as the primary Ca2+ sensor of [Ca2+]i changes under salt stress [3]. Upon activation, SOS3/CBL4 interacts with the C-terminal region of a CBL-interacting protein kinase (CIPK) called SOS2/CIPK24, which in turn activates a plasma membrane Na+/H+ antiporter SOS1 that transports sodium ions out of the cell [3]. This salt signaling pathway reinforces the concept that the salt-induced [Ca2+]i increase is an essential component for bringing about the plant response to salt tress.
Interestingly, after salt stress treatment there is an overproduction of reactive oxygen species (ROS), such as hydrogen peroxide (H2O2) [18-22]. The time constants for salt-induced increases in [Ca2+]i and ROS are about 3 sec and 400 sec, respectively, as estimated from previous studies [8,21]. It appears that the increase in [Ca2+]i occurs earlier than the ROS elevation after salt stress treatment. Considering ROS have also been shown to trigger increases in [Ca2+]i [21,23-26], it is possible that ROS-induced [Ca2+]i increases might serve as a feed forward mechanism in the salt stress signal transduction pathway. However, less is known about the relationship and interaction between the salt stress-induced [Ca2+]i increases and the [Ca2+]i increases evoked by ROS, which are produced in response to either salt stress specifically or other stresses in general [1,27].
In this study, we have systematically analyzed the relationship and interaction between salt stress-induced [Ca2+]i increases and the ROS-induced [Ca2+]i increases in Arabidopsis. We found the increases in [Ca2+]i induced by both stimuli were higher than these induced by either single stress, suggesting that NaCl and H2O2 have an additive effect on [Ca2+]i. We have also found that NaCl-induced [Ca2+]i increases might inhibit both NaCl- and H2O2-gated channels by a feedback mechanism, but more NaCl-gated channels; a similar response was seen when the H2O2-induced [Ca2+]i increases were analyzed. These data suggest responses seen involve both feedback inhibitory mechanisms, as well as an interaction between two stimuli-mediated Ca2+ signaling pathways.
Results
Dose-dependence and kinetics of NaCl- and H2O2-induced [Ca2+]i increases
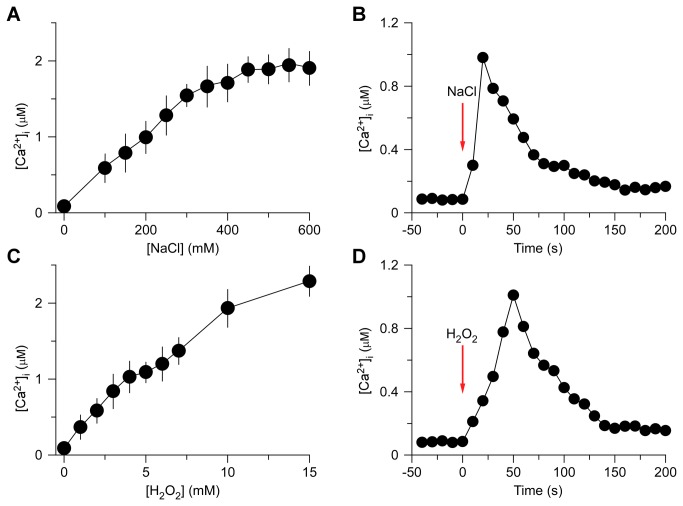
To analyze whether and how increases in [Ca2+]i induced by NaCl and H2O2 interact each other in Arabidopsis, we attempted initially to identify optimum concentrations of NaCl and H2O2, which ideally could be applied to generate about half of the maximum amplitude of [Ca2+]i for potential up- and down-regulation. In addition, we attempted to establish the kinetics of NaCl- and H2O2-induced increases in [Ca2+]i for administrating these stresses in different sequential combinations. First, to analyze NaCl-induced increases in [Ca2+]i, we treated Arabidopsis seedlings expressing aequorin with solutions containing 0 to 600 mM NaCl. Aequorin bioluminescence images were taken every 10 sec for 500 sec, and the peak [Ca2+]i was calculated and analyzed, as NaCl induces a transient increase in [Ca2+]i [8,9]. Plants grown on the half-strength MS medium had an average basal [Ca2+]i of 80 ± 21 nM (Figure 1A and C). As expected, the [Ca2+]i increased in response to NaCl treatment (Figure 1A). The magnitudes of [Ca2+]i increases were dependent on the concentration of NaCl, higher concentration of NaCl evoked a larger increase in [Ca2+]i. The NaCl concentration needed for a half-maximal response was ~200 mM, which was chosen as an optimum concentration to subsequently analyze the interaction with H2O2-induced increases in [Ca2+]i.
Figure 1. Increases in [Ca2+]i in response to NaCl and H2O2 treatments.
(A and C) Increases in [Ca2+]i induced by several concentrations of NaCl (A) and H2O2 (C) in Arabidopsis. Seedlings expressing aequorin and grown for 7 days were treated with solutions containing several concentrations of NaCl or H2O2, and aequorin images were taken every 10 sec for 500 sec. Data for four independent experiments are shown (mean ± sem; n = 64).
(B and D) Time courses of increases in [Ca2+]i induced by 200 mM NaCl (B) or 4 mM H2O2 (D). Seedlings grown for 7 days were treated with NaCl and H2O2 at time zero, and aequorin images were taken every 10 sec. Representative recordings from individual seedlings were shown. Similar results were seen in six independent experiments using 128 seedlings.
Then, we determined the temporal dynamics of NaCl-induced [Ca2+]i increases under the imposed experimental conditions as a control for further comparison (Figure 1B). We found that [Ca2+]i increased immediately after the application of 200 mM NaCl, reached a peak of ~1 µM at about 20 sec, and then declined gradually (Figure 1B). Note that, the peaking time might be shorter than 20 sec based on previous studies [8,9]. Nevertheless, imaging aequorin bioluminescence for less than 10 sec resulted in images with low a signal-noise ratio in our system. Thus, the temporal resolution was about 10 sec, which is sufficient for the current study. At about 200 sec, the [Ca2+]i was reduced to a new resting level of under 200 nM.
Similarly, we analyzed increases in [Ca2+]i in response to H2O2. Seedlings were treated with different concentrations of H2O2 from 0 to 15 mM and [Ca2+]i was analyzed. As expected, H2O2 induced increases in [Ca2+]i in a dose-dependent manner (Figure 1C). The H2O2 concentration for a half-maximal response was around 4 mM with the magnitude of [Ca2+]i similar to that induced by 200 mM NaCl. We then determined the temporal dynamics of the [Ca2+]i increase induced by 4 mM H2O2. Following treatment with 4 mM H2O2, [Ca2+]i increased and reached a peak of ~1 µM at 50 sec (Figure 1D). It took another 100 sec for the [Ca2+]i to reach a new basal level of just under 200 nM. Taken together, it seems that increases in [Ca2+]i occur faster in response to NaCl than H2O2, but are reset to a resting level 150 sec after treatment.
Additive effect of NaCl and H2O2 on triggering increases in [Ca2+]i
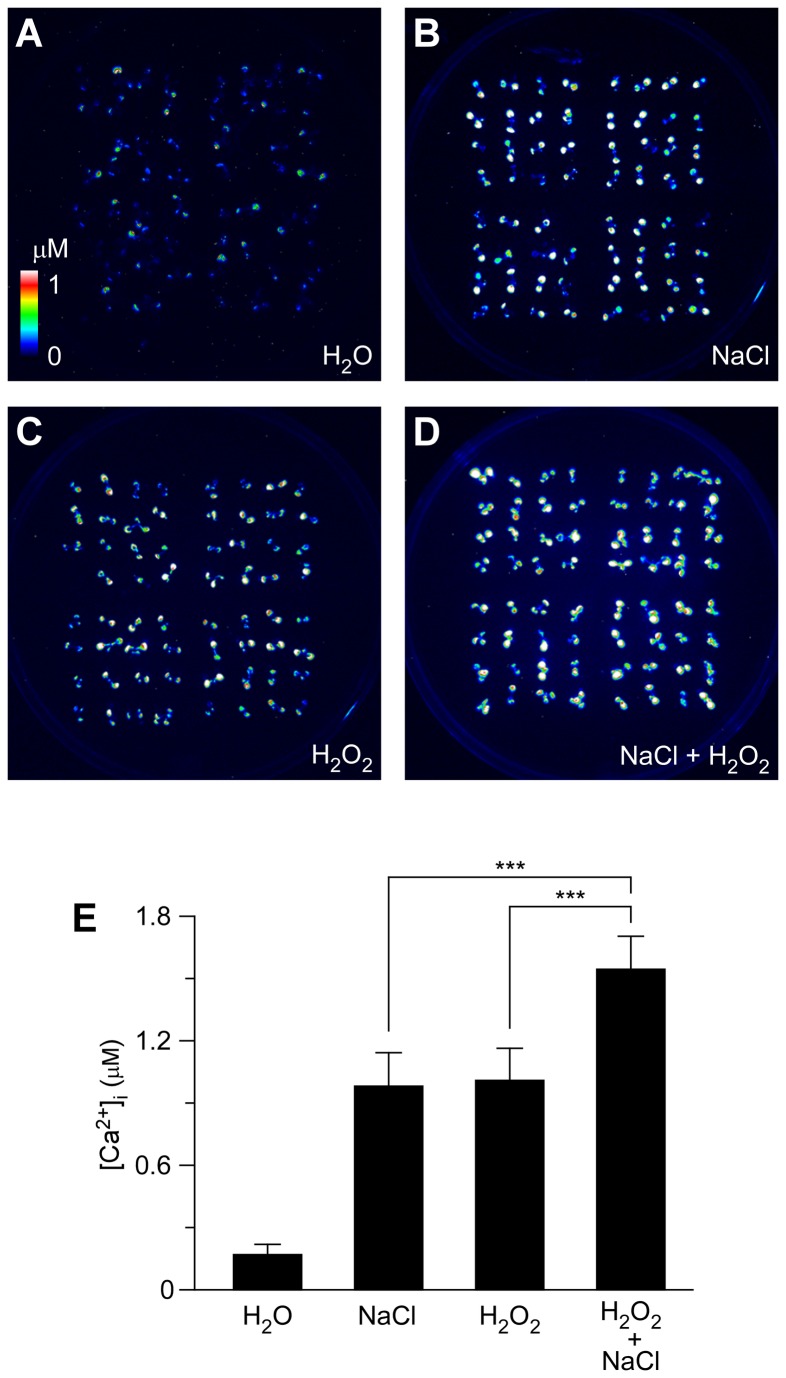
To investigate thoroughly the relationship and/or interaction between [Ca2+]i increases triggered by NaCl and H2O2, Arabidopsis seedlings were treated with 200 mM NaCl or 4 mM H2O2 separately, or 200 mM NaCl together with 4 mM H2O2. Note that, although salt-induced ROS production could be detected within ~2 min after salt treatment [21] the estimated half-time to the peak of ROS production is more than 5 min. Note also that, we did not detect a second peak of [Ca2+]i within 5 min after salt stress treatment (Figure S1A), suggesting that salt-induced ROS could not trigger a detectable increase in [Ca2+]i under the current experimental conditions. Because we measured [Ca2+]i changes within 5 min after salt treatment, the effect of salt-induced ROS on [Ca2+]i should not interfere apparently. The [Ca2+]i increases recorded after single treatments were consistent with the results described above (Figure 2A–C). NaCl and H2O2 induced similar increases in [Ca2+]i (Figure 2E). When plants were treated with both stimuli simultaneously, the peaks of [Ca2+]i were much larger than that induced by each individual stimulus (Figure 2D and E), showing an additive effect. To further analyze how salt-induced ROS affects [Ca2+]i increases in response to salt stress within 500 sec, we carried out an experiment by using the NADPH oxidase inhibitor DPI [28] and ROS scavengers ascorbic acid and glutathione [27], and found that neither of these reagents significantly affected [Ca2+]i increases induced by NaCl (Figure S1). These results suggest that the NaCl- and H2O2-induced [Ca2+]i increases may be largely independent events. In other words, NaCl and H2O2 might activate different Ca2+ permeable channels.
Figure 2. Increases in [Ca2+]i in response to NaCl and H2O2 individually or combined.
(A to D) Imaging of [Ca2+]i increases in response to the treatments of water (H2O; A), 200 mM NaCl (B), 4 mM H2O2 (A), and 200 mM NaCl and 4 mM H2O2 together (D) in Arabidopsis seedlings expressing aequorin. [Ca2+]i increases were analyzed by imaging bioluminescence and scaled by a pseudo-color bar.
(E) Quantification of [Ca2+]i increases from experiments as in (A) to (D). Data for four independent experiments are shown (mean ± sd; n = 64; *** P < 0.001; NS, not significant P > 0.05).
The crosstalk between NaCl- and H2O2-induced [Ca2+]i increases
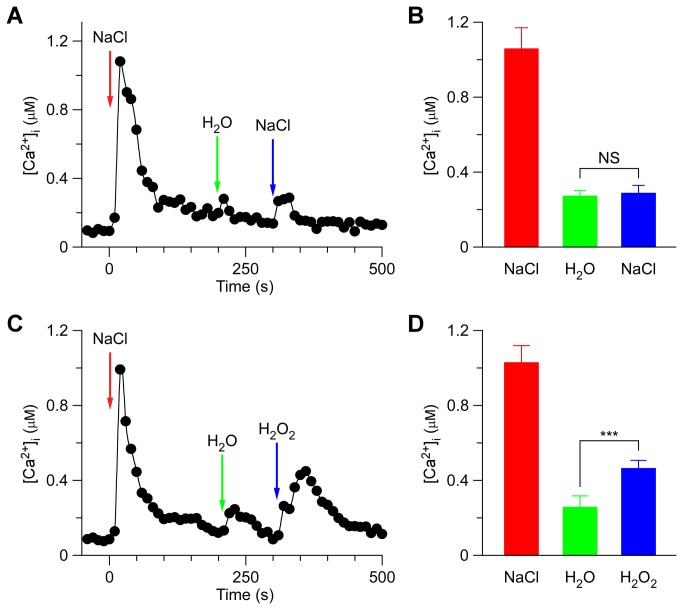
To further characterize the potential interaction between the two stimuli-triggered [Ca2+]i signals, plants were successively treated either with the same stimulus or the other. When the Arabidopsis seedlings were treated with 200 mM NaCl, the level of [Ca2+]i increased quickly to reach a peak and decreased to the new resting level after 150 sec (Figure 3A), as described in Figure 1B. A subtle increase in [Ca2+]i could be detected in seedlings after washing with deionized water at 200 sec (Figure 3A and B; green). Then, NaCl was added again, which caused a small increase in [Ca2+]i. It decayed from 300 sec to a level similar to the previous resting level (Figure 3A and B). Compared to the first NaCl treatment, which led to a large [Ca2+]i increase to ~1 µM, the 2nd NaCl treatment resulted in a [Ca2+]i increase that was only a fraction of the size of the first [Ca2+]i increase. This observation suggests that the NaCl-activated Ca2+ permeable channel (NaC) might be desensitized or adapted by unknown signaling elements upstream of NaC activation. To test whether the NaC is desensitized or adapted, we waited for 3 hr and were able to detect a normal (~1 µM) [Ca2+]i increase in response to NaCl, suggesting that the NaC is most likely desensitized (data not shown).
Figure 3. NaCl-induced [Ca2+]i increases inhibits NaCl-activated channels more than H2O2-activated channels.
(A and C) Arabidopsis seedlings were subjected to a 200 mM NaCl treatment once at 0 sec, and the solution was perfused by deionized water at 200 sec. Then, a second 200 mM NaCl (A), or 4 mM H2O2 (C) treatment was applied around 300 sec. Aequorin luminescence was recorded continuously through the treatments in the dark.
(B and D) Quantification of [Ca2+]i increases for the 2nd NaCl (B) or 2nd H2O2 treatment (C) from experiments as in (A) to (C), respectively. Data for four independent experiments are shown (mean ± sd; n = 64; NS, not significant P > 0.05; *** P < 0.001).
Subsequently we analyzed whether the hydrogen peroxide-activated Ca2+ permeable channel (HpC) was affected by the initial NaCl treatment. The second NaCl treatment was replaced by 4 mM H2O2 at 300 sec (Figure 3C). Interestingly, the peak of [Ca2+]i induced by H2O2 was clearly greater than that induced by 200 mM NaCl (P < 0.001). After 450 sec, the [Ca2+]i decreased to a new basal level under 200 µM (Figure 3C and D). The lower inhibition of HpC than NaC by the initial NaCl treatment suggests that the initial high level of [Ca2+]i, which resulted from NaC activation (called NaC[Ca2+]i microdomain/puff) subsequently inhibited NaC more than HpC (Figure 3).
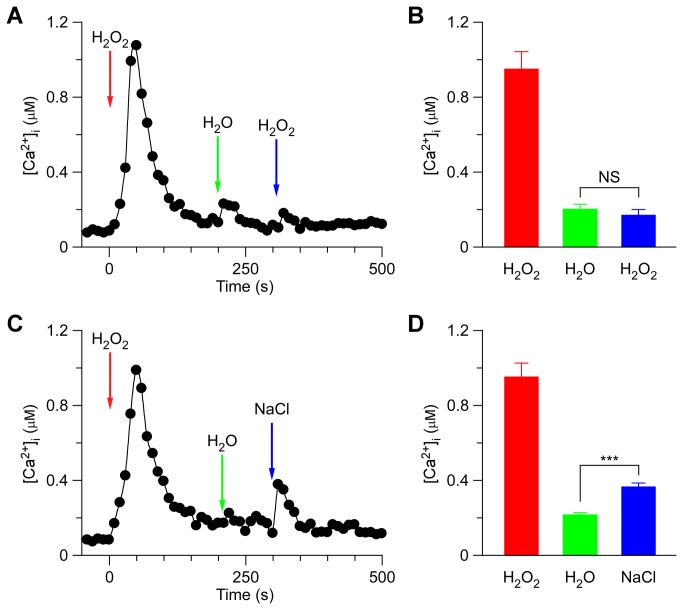
By analogy, we used H2O2 as the first stimuli to treat the seedlings and then analyzed the second treatment using H2O2 or NaCl (Figure 4). When H2O2 was added to the Petri dish, at 300 sec after the first H2O2 treatment, the [Ca2+]i level stabilized at 178 ± 32 nM, similar to previous resting levels (Figure 4A and B). But when we used 200 mM NaCl to replace H2O2 at 300 sec, the peak values of [Ca2+]i were 381 ± 23 nM, small but significantly higher than that seen with the second H2O2 treatment (Figure 4B and D). Similarly, our results suggest that the high [Ca2+]i, which resulted from the initial HpC activation (called HpC[Ca2+]i microdomain), inhibited HpC more than NaC (Figures 4 and 5).
Figure 4. H2O2-induced [Ca2+]i increases inhibits H2O2-activated channels more than NaCl-activated channels.
(A and C) Arabidopsis seedlings were subjected to a 4 mM H2O2 treatment once at 0 sec, and the solution was perfused by deionized water at 200 sec. Then, a second 4 mM H2O2 (A), or 200 mM NaCl (C) treatment was applied around 300 sec. Aequorin luminescence was recorded continuously through the treatments in the dark.
(B and D) Quantification of [Ca2+]i increases for the 2nd H2O2 (B) or 2nd NaCl treatment (C) from experiments as in (A) to (C), respectively. Data for four independent experiments are shown (mean ± sd; n = 64; NS, not significant P > 0.05; *** P < 0.001).
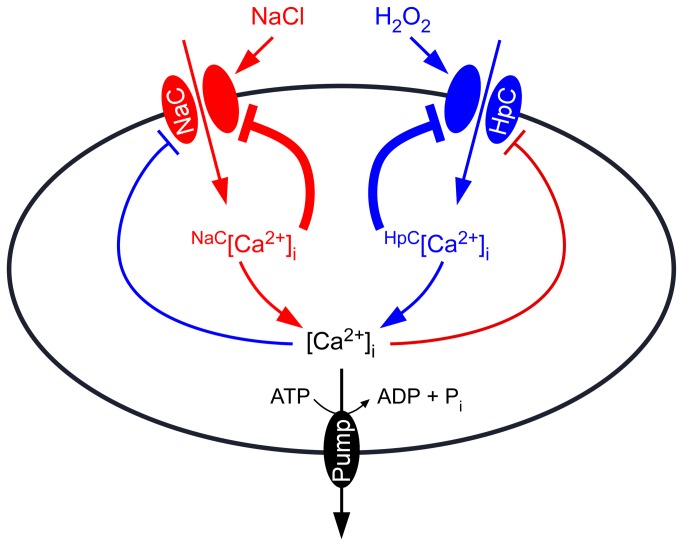
Figure 5. Model for the interaction between NaCl- and H2O2-induced [Ca2+]i increases.
A Ca2+ channel activated by NaCl (NaC) results in localized [Ca2+]i increases, called NaC-related [Ca2+]i microdomain (NaC[Ca2+]i). NaC[Ca2+]i feedback inhibits the activity of NaC. HpC, a Ca2+ channel activated by hydrogen peroxide, leads to localized [Ca2+]i increases, called HpC[Ca2+]i microdomain. HpC[Ca2+]i also feedback inhibits HpC activity. The [Ca2+]i mircodomain-mediated inhibition of Ca2+ channels is the major feedback inhibitory pathways (thick lines). In addition, both NaC[Ca2+]i and HpC[Ca2+]i might contribute to a global [Ca2+]i increase, which further inhibits both NaC and HpC, serving as global feedback inhibitory pathways (thin lines). [Ca2+]i is reset to the resting level by plasma membrane Ca2+ pumps.
Discussion
Calcium is a universal second messenger that plays an important role in signal transduction in animals and plants [25,29-31]. In the past 20 years, tremendous progress has been made in understanding the changes in [Ca2+]i that appear in response to various abiotic and biotic stresses in plants, including salt stress, oxidative stress, drought, high and low temperatures, and pathogen elicitors [13,25,26,32]. It is known that a specific stimulus can trigger unique temporal and spatial patterns of [Ca2+]i, also known as [Ca2+]i signatures [33]. The [Ca2+]i signature encodes information from the environmental stimulus which will be decoded subsequently by intracellular Ca2+ sensors, such as calmodulins (CaMs) and calcineurin B-like proteins (CBLs), leading to the activation of downstream events [10]. It is also known that the basal [Ca2+]i is maintained at a concentration about 10,000-fold below the extracellular Ca2+ concentration [29,31,34]. In general, Ca2+ channels in the plasma membrane and/or endomembranes are activated in response to environmental stimuli, leading to increases in [Ca2+]i [30,32]. Salt stress-induced increases in [Ca2+]i have long been proposed as being involved in the process perceiving the salt signal although the properties of the salt-activated Ca2+ permeable channel are poorly understood and its molecular nature remains to be identified [1,2,7].
In addition, various abiotic and biotic stresses lead to the production of ROS and oxidative stresses, which control many different processes in plants [27,35-37]. It has been well established that salt stress enhances the production of reactive oxygen species (ROS) in plants [18-22]. Interestingly, ROS has also been shown to activate Ca2+ permeable channels in the plasma membrane, which in turn lead to Ca2+ influx into the cell and thus increases in [Ca2+]i [19,24,38]. Note that, the salt stress-induced [Ca2+]i increases precede the production of H2O2 signaling molecule [39]. Nevertheless, little is known about the molecular mechanisms underlying ROS perception in plant cells, and it is possible that ROS activation of Ca2+ permeable channels may serve as a ROS perception process.
The decay of the increases in [Ca2+]i induced by both NaCl and H2O2 seen in this study (Figure 1B and D) as well as previous studies [8,9,15] indicates that the stimulus-activated Ca2+ permeable channels may be inactivated via a feedback inhibitory mechanism, i.e. elevated [Ca2+]i inhibits these channels, a desensitization process commonly seen for receptor ion channels in animals [31,40]. It remains to be addressed whether the localized increases in [Ca2+]i induced by one stimulus, called [Ca2+]i microdomain [29,31], inhibit the other stimulus-activated Ca2+ permeable channels. It is known that that NaCl induces multiple peaks of [Ca2+]i under certain conditions [9], possibly because the same NaCl-sensitive channels are repetitively activated, or that NaCl might trigger H2O2 production which subsequently activates another Ca2+ channel, different from the NaCl-sensitive channel. Under our experimental conditions, we did not observe multiple peaks of [Ca2+]i after salt treatment (Figure S1A).
NaCl and H2O2 together induced larger increases in [Ca2+]i than either NaCl or H2O2 alone (Figure 2), suggesting that NaCl and H2O2 may activate distinct Ca2+ permeable channels, NaC and HpC (Figure 5). NaC and HpC are likely regulated by feedback inhibition (Figure 5), considering their desensitization seen in this study (Figure 1B and D) as well as previous reports [8,9]. We demonstrated that repetitive NaCl treatments failed to trigger repetitive [Ca2+]i increases (Figure 3A and B). This indicates that the NaC cannot be activated repetitively within a short period of time, i.e. NaC is possibly desensitized. We propose that a feedback inhibition may be involved in the desensitization (Figure 5). Upon NaCl treatment, the NaC opens, leading to the localized increase of [Ca2+]i, NaC[Ca2+]i microdomain/puff. NaC[Ca2+]i in turn signals the channel to close, which prevents further [Ca2+]i increases and allows the basal [Ca2+]i to be reset via Ca2+ pumps. This feedback inhibition avoids the excessive increase of [Ca2+]i which could be disastrous to plant cells. The same phenomenon was also observed with the activation of HpC (Figure 5), i.e. HpC[Ca2+]i microdomain inhibits HpC via a feedback mechanism. Clearly, the most significant effect is that after the initial treatment by either NaCl or H2O2, [Ca2+]i increases induced by both NaCl and H2O2 are reduced (Figures 3 and 4). It is most likely that localized NaC[Ca2+]i and HpC[Ca2+]i merge to form a relatively global [Ca2+]i, which then feedback inhibits both NaC and HpC (Figure 5). We observed that Arabidopsis was unable to recover from 200 mM NaCl treatment 5 min after an initial stimulation. Similar results were observed after 4 mM H2O2 treatment. In contrast, a previous study has shown that Arabidopsis is able to recover its ability to respond almost fully to cold shock 3 min after an initial cold shock [41]. Note that, our work does not prove that HpC and NaC are localized in discreet and different microdomains in the plasma membrane, rather we have shown HpC and NaC may differ and interact via [Ca2+]i microdomains. It is also possible that NaCl and H2O2 may activate the same Ca2+ permeable channel, which is expressed in different types of cells and/or activated via different signaling pathways, leading to the differential changes in [Ca2+]i.
In general, when plants are exposed to one stress, their resistance to other stresses can be enhanced. It is most likely that stress-evoked [Ca2+]i increases as well as stress-stimulated overproduction of ROS function as key integrators, possibly mediating stress signal perception and signal transduction. Our results demonstrate the inhibitory interaction of NaCl- and H2O2-induced [Ca2+]i increases, and may predict distinct Ca2+ permeable channels activated by NaCl and H2O2, respectively (Figure 5). In the future, it is important to analyze the pharmacological properties of these putative Ca2+ permeable channels activated by NaCl and H2O2 as described previously for MAMP-activated channels [42]. Obviously, the identification of these channels or sensors will be a hallmark in the study of plant salt resistance in the future. In addition, how NaC and HpC interact to contribute to the [Ca2+]i signatures and other downstream events can be further analyzed when their molecular nature is identified.
Materials and Methods
Plant materials and growth conditions
Arabidopsis thaliana ecotype Columbia-0 (Col-0) constitutively expressing intracellular aequorin (pMAQ2, a kind gift from Dr. M. Knight) under the control of the cauliflower mosaic virus 35S promoter was used [8,33]. Arabidopsis plants were grown in 150 mm x 15 mm round Petri dishes in half-strength Murashige and Skoog salts (MS; Gibco), supplemented with 1.5% (w/v) sucrose (Sigma), and 0.8% (w/v) agar (Becton Dickinson) adjusted to pH 6.0 with KOH in controlled environmental rooms at 21 ± 2°C. The fluency rate of white light was ~110 µmol m-2 sec-1. The photoperiods were 16 h light/8 h dark cycles. Seeds were sterilized with 2.5% PPM (Plant preservative mixture; Caisson Laboratories) and stratified at 4°C for 3 days in the dark, and then transferred to growth rooms.
Aequorin reconstitution and measurement of [Ca2+]i
Arabidopsis thaliana plants expressing cytosolic apoaequorin were used for [Ca2+]i measurements [33,43]. Seedlings were grown on half-strength Murashige and Skoog medium for 7 days. Reconstitution of aequorin was performed in vivo by spraying seedlings with 240 µL of 10 µM coelenterazine per Petri dish followed by incubation at 21°C in the dark for 8 hr. Treatments and aequorin luminescence imaging were performed at room temperature using a ChemiPro HT system that includes a cryogenically cooled and back-illuminated CCD camera, liquid nitrogen autofiller, camera controller, and computer-equipped WinView/32 software (Roper Scientific) as described previously [33]. The CCD camera has a 1300 × 1340 pixel resolution and is cooled to −110°C by the cryogenic cooler system prior to image recording. The recording was started 50 s prior treatments and luminescence images were taken every 10 sec. The total remaining aequorin was estimated by treating plants with a discharging solution containing 0.9 M CaCl2 in 10% (v/v) ethanol and recorded for 5 min until values were within 1% of the highest discharge value [8,15,33]. WinView/32 and Meta Morph 6 were used to analyze recorded luminescence images. Experiments were carried out at room temperature (22 to 24°C).
NaCl and H2O2 treatments
For stress treatments, Petri dishes were placed individually into the ChemiPro HT chamber and luminescence images were taken at 10 sec intervals starting 50 sec prior the treatment. The treatment solution (100 mL) at described concentrations of NaCl or H2O2 was added into Petri dish in the dark, and luminescence was recorded continuously. For changes in bath solution, a four-channel peristaltic pump (Dynamax RP-1, Rainin) was used to perfuse Petri dish with water as indicated in the figures. Then, additional stress treatment was applied by adding 100 mL solution into Petri dish.
Calibration of calcium measurements
The cytosolic free cytosolic Ca2+ concentrations were calculated based on the calibration equation described previously [41] with modification to the ChemiPro system. The wild-type Arabidopsis expressing aequorin were placed individually in each well in 96-well plates containing ½ MS medium, 1.5% (w/v) sucrose, and 0.8% (w/v) agar for 10 days. Kinetic luminescence measurements were performed with an automated microplate luminescence reader (Mithras LB 940, Berthold Technologies). After automatic injection of 0.2 ml of solution into the each well bioluminescence counts were integrated every 1 sec as described previously [41]. The solutions containing a range of [NaCl] from 0 to 600 mM were used to treat the plants, and the peak values of [Ca2+]i were calculated used the equation described previously [41]. Similar measurements were carried out using the ChemiPro HT system as described above to obtain L/Lmax values for each treatment, where L is luminescence and Lmax is the total remaining counts for bioluminescence. Then, we fit these data to the previously describe equation pCa = a * (−log (L/Lmax)) + b, and obtained the equation pCa = 0.9057209 * (−log (L/Lmax)) + 4.7712743. Note that, the calculated Ca2+ concentrations presented in the current study are similar to those reported previously [8,41].
Supporting Information
H2O2 levels do not affect [Ca2+]i increases in response to NaCl treatment. (A) Arabidopsis seedlings were treated with water (Control), the NADPH oxidase inhibitor DPI (15 µM), and ROS scavenger ascorbic acid (5 mM) and glutathione (5 mM) two hours prior to the NaCl treatment. The seedlings were then subjected to a 200 mM NaCl treatment, and aequorin luminescence was recorded continuously through the treatments in the dark. (B) Quantification of peak [Ca2+]i increases from experiments as in (A). Data for three independent experiments are shown (mean ± sd; n = 35 to 62; NS, not significant, P > 0.05).
(PDF)
Acknowledgments
We thank Marc R. Knight for Arabidopsis seeds expressing aequorin, James N. Siedow for discussion and critical reading of the manuscript, Douglas M. Johnson and Gary B. Swift for maintenance of the ChemiPro system, and the Pei lab members for discussion and support.
Funding Statement
ZJ and SZ were supported by fellowships from China Scholarship Council, Pandeng Project funds (PD11001008001; PD11002002004002) from Hangzhou Normal University, and a grant from Zhejiang NSF (Z3110443). This work is supported by grants from USDA (CSREES-2006-35100-17304) and from NSF (IOS-0848263) to ZMP. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59: 651-681. doi: 10.1146/annurev.arplant.59.032607.092911. PubMed: 18444910. [DOI] [PubMed] [Google Scholar]
- 2. Zhu JK (2001) Plant salt tolerance. Trends Plant Sci 6: 66-71. doi: 10.5363/tits.6.11_66. PubMed: 11173290. [DOI] [PubMed] [Google Scholar]
- 3. Zhu JK (2003) Regulation of ion homeostasis under salt stress. Curr Opin Plant Biol 6: 441-445. doi: 10.1016/S1369-5266(03)00085-2. PubMed: 12972044. [DOI] [PubMed] [Google Scholar]
- 4. Flowers TJ (2004) Improving crop salt tolerance. J Exp Bot 55: 307-319. doi: 10.1093/jxb/erh003. PubMed: 14718494. [DOI] [PubMed] [Google Scholar]
- 5. Qiu QS, Guo Y, Dietrich MA, Schumaker KS, Zhu JK (2002) Regulation of SOS1, a plasma membrane Na+/H+ exchanger in Arabidopsis thaliana, by SOS2 and SOS3. Proc Natl Acad Sci U S A 99: 8436-8441. doi: 10.1073/pnas.122224699. PubMed: 12034882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Shi H, Ishitani M, Kim C, Zhu JK (2000) The Arabidopsis thaliana salt tolerance gene SOS1 encodes a putative Na+/H+ antiporter. Proc Natl Acad Sci U S A 97: 6896-6901. doi: 10.1073/pnas.120170197. PubMed: 10823923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Luan S, Lan W, Chul Lee S (2009) Potassium nutrition, sodium toxicity, and calcium signaling: connections through the CBL-CIPK network. Curr Opin Plant Biol 12: 339-346. doi: 10.1016/j.pbi.2009.05.003. PubMed: 19501014. [DOI] [PubMed] [Google Scholar]
- 8. Knight H, Trewavas AJ, Knight MR (1997) Calcium signalling in Arabidopsis thaliana responding to drought and salinity. Plant J 12: 1067-1078. doi: 10.1046/j.1365-313X.1997.12051067.x. PubMed: 9418048. [DOI] [PubMed] [Google Scholar]
- 9. Tracy FE, Gilliham M, Dodd AN, Webb AA, Tester M (2008) NaCl-induced changes in cytosolic free Ca2+ in Arabidopsis thaliana are heterogeneous and modified by external ionic composition. Plant Cell Environ 31: 1063-1073. doi: 10.1111/j.1365-3040.2008.01817.x. PubMed: 18419736. [DOI] [PubMed] [Google Scholar]
- 10. Luan S (2009) The CBL-CIPK network in plant calcium signaling. Trends Plant Sci 14: 37-42. doi: 10.1016/j.tplants.2008.10.005. PubMed: 19054707. [DOI] [PubMed] [Google Scholar]
- 11. Hetherington AM, Brownlee C (2004) The generation of Ca(2+) signals in plants. Annu Rev Plant Biol 55: 401-427. doi: 10.1146/annurev.arplant.55.031903.141624. PubMed: 15377226. [DOI] [PubMed] [Google Scholar]
- 12. Pandey GK, Cheong YH, Kim KN, Grant JJ, Li L et al. (2004) The calcium sensor calcineurin B-like 9 modulates abscisic acid sensitivity and biosynthesis in Arabidopsis . Plant Cell 16: 1912-1924. doi: 10.1105/tpc.021311. PubMed: 15208400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Dodd AN, Kudla J, Sanders D (2010) The language of calcium signaling. Annu Rev Plant Biol 61: 593-620. doi: 10.1146/annurev-arplant-070109-104628. PubMed: 20192754. [DOI] [PubMed] [Google Scholar]
- 14. Kudla J, Batistic O, Hashimoto K (2010) Calcium signals: the lead currency of plant information processing. Plant Cell 22: 541-563. doi: 10.1105/tpc.109.072686. PubMed: 20354197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Rentel MC, Knight MR (2004) Oxidative stress-induced calcium signaling in Arabidopsis . Plant Physiol 135: 1471-1479. doi: 10.1104/pp.104.042663. PubMed: 15247375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Batistič O, Kudla J (2012) Analysis of calcium signaling pathways in plants. Biochim Biophys Acta 1820: 1283-1293. doi: 10.1016/j.bbagen.2011.10.012. PubMed: 22061997. [DOI] [PubMed] [Google Scholar]
- 17. Day IS, Reddy VS, Shad Ali G, Reddy AS (2002) Analysis of EF-hand-containing proteins in Arabidopsis. Genome Biol 3: RESEARCH0056 PubMed: 12372144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Borsani O, Zhu J, Verslues PE, Sunkar R, Zhu JK (2005) Endogenous siRNAs derived from a pair of natural cis-antisense transcripts regulate salt tolerance in Arabidopsis . Cell 123: 1279-1291. doi: 10.1016/j.cell.2005.11.035. PubMed: 16377568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Miller G, Suzuki N, Ciftci-Yilmaz S, Mittler R (2010) Reactive oxygen species homeostasis and signalling during drought and salinity stresses. Plant Cell Environ 33: 453-467. doi: 10.1111/j.1365-3040.2009.02041.x. PubMed: 19712065. [DOI] [PubMed] [Google Scholar]
- 20. Vaidyanathan H, Sivakumar P, Chakrabarty R, Thomas G (2003) Scavenging of reactive oxygen species in NaCl-stressed rice (Oryza sativa L.) - differential response in salt-tolerant and sensitive varieties. Plant Sci 165: 1411-1418. doi: 10.1016/j.plantsci.2003.08.005. [DOI] [Google Scholar]
- 21. Leshem Y, Seri L, Levine A (2007) Induction of phosphatidylinositol 3-kinase-mediated endocytosis by salt stress leads to intracellular production of reactive oxygen species and salt tolerance. Plant J 51: 185-197. doi: 10.1111/j.1365-313X.2007.03134.x. PubMed: 17521408. [DOI] [PubMed] [Google Scholar]
- 22. Valderrama R, Corpas FJ, Carreras A, Gómez-Rodríguez MV, Chaki M et al. (2006) The dehydrogenase-mediated recycling of NADPH is a key antioxidant system against salt-induced oxidative stress in olive plants. Plant Cell Environ 29: 1449-1459. doi: 10.1111/j.1365-3040.2006.01530.x. PubMed: 17080966. [DOI] [PubMed] [Google Scholar]
- 23. McAinsh MR, Clayton H, Mansfield TA, Hetherington AM (1996) Changes in stomatal behavior and guard cell cytosolic free calcium in response to oxidative stress. Plant Physiol 111: 1031-1042. PubMed: 12226345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Pei ZM, Murata Y, Benning G, Thomine S, Klüsener B et al. (2000) Calcium channels activated by hydrogen peroxide mediate abscisic acid signalling in guard cells. Nature 406: 731-734. doi: 10.1038/35021067. PubMed: 10963598. [DOI] [PubMed] [Google Scholar]
- 25. McAinsh MR, Pittman JK (2009) Shaping the calcium signature. New Phytol 181: 275-294. doi: 10.1111/j.1469-8137.2008.02682.x. PubMed: 19121028. [DOI] [PubMed] [Google Scholar]
- 26. Ma W, Berkowitz GA (2011) Ca2+ conduction by plant cyclic nucleotide gated channels and associated signaling components in pathogen defense signal transduction cascades. New Phytol 190: 566-572. doi: 10.1111/j.1469-8137.2010.03577.x. PubMed: 21166809. [DOI] [PubMed] [Google Scholar]
- 27. Suzuki N, Koussevitzky S, Mittler R, Miller G (2012) ROS and redox signalling in the response of plants to abiotic stress. Plant Cell Environ 35: 259-270. doi: 10.1111/j.1365-3040.2011.02336.x. PubMed: 21486305. [DOI] [PubMed] [Google Scholar]
- 28. Marino D, Dunand C, Puppo A, Pauly N (2012) A burst of plant NADPH oxidases. Trends Plant Sci 17: 9-15. doi: 10.1016/j.tplants.2011.10.001. PubMed: 22037416. [DOI] [PubMed] [Google Scholar]
- 29. Clapham DE (2007) Calcium signaling. Cell 131: 1047-1058. doi: 10.1016/j.cell.2007.11.028. PubMed: 18083096. [DOI] [PubMed] [Google Scholar]
- 30. Ward JM, Mäser P, Schroeder JI (2009) Plant ion channels: gene families, physiology, and functional genomics analyses. Annu Rev Physiol 71: 59-82. doi: 10.1146/annurev.physiol.010908.163204. PubMed: 18842100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Berridge MJ, Bootman MD, Roderick HL (2003) Calcium signalling: dynamics, homeostasis and remodelling. Nat Rev Mol Cell Biol 4: 517-529. doi: 10.1038/nrm1155. PubMed: 12838335. [DOI] [PubMed] [Google Scholar]
- 32. Hetherington AM, Brownlee C (2004) The generation of Ca2+ signals in plants. Annu Rev Plant Biol 55: 401-427. doi: 10.1146/annurev.arplant.55.031903.141624. PubMed: 15377226. [DOI] [PubMed] [Google Scholar]
- 33. Tang RH, Han S, Zheng H, Cook CW, Choi CS et al. (2007) Coupling diurnal cytosolic Ca2+ oscillations to the CAS-IP3 pathway in Arabidopsis . Science 315: 1423-1426. doi: 10.1126/science.1134457. PubMed: 17347443. [DOI] [PubMed] [Google Scholar]
- 34. Swanson SJ, Choi WG, Chanoca A, Gilroy S (2011) In vivo imaging of Ca2+, pH, and reactive oxygen species using fluorescent probes in plants. Annu Rev Plant Biol 62: 273-297. doi: 10.1146/annurev-arplant-042110-103832. PubMed: 21370977. [DOI] [PubMed] [Google Scholar]
- 35. Vranová E, Inzé D, Van Breusegem F (2002) Signal transduction during oxidative stress. J Exp Bot 53: 1227-1236. doi: 10.1093/jexbot/53.372.1227. PubMed: 11997371. [DOI] [PubMed] [Google Scholar]
- 36. Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7: 405-410. doi: 10.1016/S1360-1385(02)02312-9. PubMed: 12234732. [DOI] [PubMed] [Google Scholar]
- 37. Wise RR, Naylor AW (1987) Chilling-Enhanced Photooxidation : The Peroxidative Destruction of Lipids during Chilling Injury to Photosynthesis and Ultrastructure. Plant Physiol 83: 272-277. doi: 10.1104/pp.83.2.272. PubMed: 16665235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Waring P (2005) Redox active calcium ion channels and cell death. Arch Biochem Biophys 434: 33-42. doi: 10.1016/j.abb.2004.08.001. PubMed: 15629106. [DOI] [PubMed] [Google Scholar]
- 39. Yang T, Poovaiah BW (2002) Hydrogen peroxide homeostasis: activation of plant catalase by calcium/calmodulin. Proc Natl Acad Sci U S A 99: 4097-4102. doi: 10.1073/pnas.052564899. PubMed: 11891305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Traynelis SF, Wollmuth LP, McBain CJ, Menniti FS, Vance KM et al. (2010) Glutamate receptor ion channels: structure, regulation, and function. Pharmacol Rev 62: 405-496. doi: 10.1124/pr.109.002451. PubMed: 20716669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Knight H, Trewavas AJ, Knight MR (1996) Cold calcium signaling in Arabidopsis involves two cellular pools and a change in calcium signature after acclimation. Plant Cell 8: 489-503. doi: 10.2307/3870327. PubMed: 8721751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Kwaaitaal M, Huisman R, Maintz J, Reinstädler A, Panstruga R (2011) Ionotropic glutamate receptor (iGluR)-like channels mediate MAMP-induced calcium influx in Arabidopsis thaliana . Biochem J 440: 355-365. doi: 10.1042/BJ20111112. PubMed: 21848515. [DOI] [PubMed] [Google Scholar]
- 43. Knight MR, Campbell AK, Smith SM, Trewavas AJ (1991) Transgenic plant aequorin reports the effects of touch and cold-shock and elicitors on cytoplasmic calcium. Nature 352: 524-526. doi: 10.1038/352524a0. PubMed: 1865907. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
H2O2 levels do not affect [Ca2+]i increases in response to NaCl treatment. (A) Arabidopsis seedlings were treated with water (Control), the NADPH oxidase inhibitor DPI (15 µM), and ROS scavenger ascorbic acid (5 mM) and glutathione (5 mM) two hours prior to the NaCl treatment. The seedlings were then subjected to a 200 mM NaCl treatment, and aequorin luminescence was recorded continuously through the treatments in the dark. (B) Quantification of peak [Ca2+]i increases from experiments as in (A). Data for three independent experiments are shown (mean ± sd; n = 35 to 62; NS, not significant, P > 0.05).
(PDF)