Abstract
Sensorineural deafness and retinitis pigmentosa (RP) are the hallmarks of Usher syndrome (USH) but are also prominent features in peroxisomal biogenesis defects (PBDs); both are autosomal recessively inherited. The firstborn son of unrelated parents, who both had sensorineural deafness and RP diagnosed as USH, presented with sensorineural deafness, RP, dysmorphism, developmental delay, hepatomegaly, and hypsarrhythmia and died at age 17 mo. The infant was shown to have a PBD, on the basis of elevated plasma levels of very-long- and branched-chain fatty acids (VLCFAs and BCFAs), deficiency of multiple peroxisomal functions in fibroblasts, and complete absence of peroxisomes in fibroblasts and liver. Surprisingly, both parents had elevated plasma levels of VLCFAs and BCFAs. Fibroblast studies confirmed that both parents had a PBD. The parents' milder phenotypes correlated with relatively mild peroxisomal biochemical dysfunction and with catalase immunofluorescence microscopy demonstrating mosaicism and temperature sensitivity in fibroblasts. The infant and both of his parents belonged to complementation group C. PEX6 gene sequencing revealed mutations on both alleles, in the infant and in his parents. This unique family is the first report of a PBD with which the parents are themselves affected individuals rather than asymptomatic carriers. Because of considerable overlap between USH and milder PBD phenotypes, individuals suspected to have USH should be screened for peroxisomal dysfunction.
Peroxisomal biogenesis disorders (PBDs [MIM 601539]) are caused by mutations in PEX genes encoding peroxins required for targeting of peroxisomal proteins from the cytosol to the peroxisome and for their subsequent importation into the organelle (Gould and Valle 2000). These autosomal recessively inherited disorders are characterized by absence of morphologically identifiable peroxisomes and deficiency of multiple peroxisomal metabolic functions. PBDs are multisystem disorders manifesting with craniofacial dysmorphism, hypotonicity, seizures, psychomotor retardation, vision and hearing impairment, and skeletal, renal, hepatic, and gastrointestinal disease. Their clinical spectrum ranges from the full-blown phenotype of Zellweger syndrome (ZS [MIM 214100]), which is fatal in infancy; through the intermediate form, adrenoleukodystrophy (NALD [MIM 202370]), which has predominant neurological features and survival for 1 or several years; to the milder form, infantile Refsum disease (IRD [MIM 266510]), which has predominant hepatic and gastrointestinal involvement and more-prolonged survival (Baumgartner et al. 1998; Wanders 1999; Suzuki et al. 2001).
Retinitis pigmentosa (RP) and sensorineural deafness are features common to all of the classic PBD phenotypes but are also the hallmarks of the Usher syndromes (USH [MIM 276900–276906, MIM 601067, MIM 602083, MIM 602097, and MIM 605472]), a clinically and genetically heterogeneous group of disorders classified into three distinct phenotypic subtypes, according to severity, age at onset, and presence or absence of vestibulary dysfunction (Petit 2001). Here, we report the diagnosis of a severe generalized PBD in an infant whose parents both have RP associated with congenital sensorineural deafness. Both parents were originally diagnosed as having USH but subsequently were shown to be suffering from a PBD.
The index patient was the firstborn son of unrelated parents (see below). He was born at term by Cesarean section, because of breech presentation and oligohydramnios. The Apgar score was 9 at 1 min and 10 at 5 min, birth weight was 2,720 g (3d percentile), and head circumference was 33 cm (5th percentile). At birth, he had a high-pitched cry, large anterior and posterior fontanels, widely open sutures, low-set, narrow external auditory canals, antimongoloid slanting of the eyes, horizontal nystagmus, grade I subcapsular cataracts, a webbed neck, widely spaced nipples, bilateral palmar Simian lines, right undescended testis, extreme laxity of the hips, and bilateral forefoot adduction. Karyotype analysis was normal (46,XY).
At age 4 mo, he displayed little or no response to visual and auditory stimuli, absence of smiling or social contact, wandering nystagmus, marked axial hypotonicity with dystonic limb hypertonicity, and hepatomegaly. Additional dysmorphic features included huge fontanels, dolichocephaly, high forehead, Brushfield spots, anteverted nares, a long philtrum, narrow hard palate, and mildly dysplastic auricles. By age 12 mo, he was in a vegetative state, with opisthotonus, dystonia, myoclonic seizures, and a hypsarrhythmia pattern on electroencephalography examination. Fundoscopy revealed peripheral pigmentary changes consistent with RP, as well as small optic disks. Electroretinography (ERG) examination revealed a flat response, and results of examination of visual evoked potential were compatible with RP. Free-field audiometry documented bilateral sensorineural deafness (left 55 dB, right 70 dB). Brain-stem evoked-response–audiometry revealed delayed waves III and V, with a hearing-level response (wave III) of 60 dB. His subsequent course was characterized by severe feeding difficulties necessitating nasogastric feeding, recurrent seizures, prolonged hospitalizations, and repeated episodes of bronchopneumonia and respiratory failure leading to death at age 17 mo.
The infant’s mother is of Jewish Algerian and Ashkenazi origin. Her parents were unrelated, and there is no relevant family history. She was evaluated at age 3 mo, for cholestatic liver disease, which eventually resolved. She was noted to have strabismus and nystagmus, and RP was documented at age 12 mo. At age 24 mo, audiography performed because of speech delay revealed severe bilateral sensorineural hearing loss (65 dB). In view of the combination of RP and sensorineural deafness, she was diagnosed as suffering from USH. She uses hearing aids, has night blindness and restriction of peripheral visual fields to 100°, and exhibits a fine tremor in both hands, and her IQ has been assessed as borderline normal.
The infant’s father is of Yemenite Jewish origin. His parents were first cousins. Three of his maternal first cousins had RP and congenital deafness diagnosed as USH. The father has severe congenital sensorineural deafness (95 dB) that was presumed to be of infectious origin. However, after the birth of his son, he underwent ophthalmologic reevaluation and ERG examination, which revealed mild RP, and he too was diagnosed as having USH.
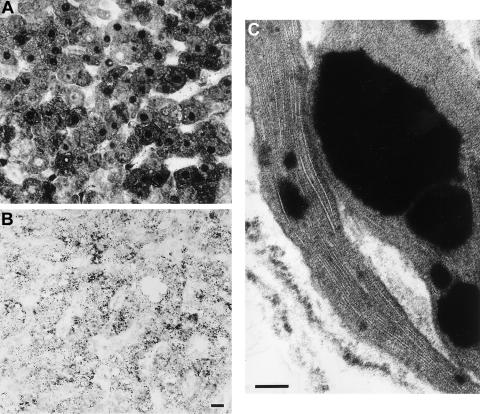
The diagnosis of a PBD in the infant was first suspected at age 7 mo, when GC/MS examination of urine organic acids revealed a distinctive dicarboxylic aciduria with reversal of the normal (C6 > C8) ratio and prominence of odd-chain-length dicarboxylic acids, 3-hydroxydicarboxylic acids, 3,6-epoxydicarboxylic acids, and 2-hydroxysebacic acid (Korman et al. 2000). This prompted examination of very-long-chain fatty acids (VLCFAs) in plasma (table 1), which revealed a marked elevation of both C26:0 and the C26:0/C22:0 ratio; in addition, there was a significant elevation of the branched-chain fatty acids (BCFAs)—phytanic acid and, particularly, pristanic acid. Biochemical investigations of fibroblasts (table 1) confirmed the elevation of VLCFAs and documented a deficiency in the following multiple additional peroxisomal functions: (i) impaired de novo plasmalogen synthesis (Schrakamp et al. 1988) and deficient dihydroxyacetonephosphate acyltransferase (DHAP-AT) activity (Wanders et al. 1995c); (ii) deficient β-oxidation of VLCFAs and pristanic acid (Wanders et al. 1995b); (iii) deficient α-oxidation of phytanic acid (Wanders and Van Roermund 1993); and (iv) abnormal immunoblot patterns (Wanders et al. 1995a) for the peroxisomal enzyme proteins acyl-CoA oxidase and peroxisomal thiolase. Finally, catalase-immunofluorescence analysis of fibroblasts (Wanders et al. 1989) demonstrated the complete absence of peroxisomes. Similarly, in liver-biopsy tissue (Espeel and Van Limbergen 1995; Roels et al. 1995), no peroxisomes were visible after staining for catalase activity, whereas immunolocalization catalase and alanine-glyoxylate aminotransferase (AGT) revealed antigen in the cytoplasm of parenchymal cells and in some nuclei, but no granules (fig. 1A and B). Absence of peroxisomes was confirmed by electron microscopy; no organelles were labeled by catalase or AGT antibodies. However, weak immunogold labeling for the 70-kD peroxisomal membrane protein was seen over rare and small vesicles of two types that have been reported in the livers of some other patients with PBD (Espeel et al. 1995b): dense core organelles and “empty” vesicles. Liver macrophages showed angulate lysosomes containing stacks of trilamellar structures (fig. 1C), detected as birefringent inclusions in polarized light. Macrophages also displayed small lipid droplets insoluble in acetone.
Table 1.
Biochemical Data on the Infant and His Parents
| Measure | Infant | Mother | Father | Control Value |
| Plasma VLCFAs: | ||||
| C22:0 (μg/ml) | 8.6 | 21.1 | 18.0 | 10–35 |
| C24:0/C22:0 ratio | 1.30 | .95 | .90 | .68 ± .15 |
| C26:0/C22:0 ratio | .303 | .124 | .054 | .018 ± .009 |
| Plasma branched-chain fatty acids (μg/ml): | ||||
| Phytanic acid | 4.1 | 4.8 | 4.5 | .5–3.5 |
| Pristanic acid | 1.97 | 1.95 | .62 | .01–0.40 |
| VLCFAs in fibroblasts: | ||||
| C22:0 μmol/g protein | 3.91 | 6.22 | 3.71 | 3.84–10.20 |
| C24:0 μmol/g protein | 9.83 | 13.00 | 11.93 | 7.76–17.66 |
| C26:0 μmol/g protein | 1.41 | .48 | .91 | .18–.38 |
| C24:0/C22:0 ratio | 2.51 | 2.09 | 3.21 | 1.55–2.30 |
| C26:0/C22:0 ratio | .36 | .08 | .24 | .03–.07 |
| De novo plasmalogen synthesis in fibroblasts:a | ||||
| pPE in PE (%) | 14.4 | 59.0 | 39.0 | 64.5–85.7 |
| pPC in PC (%) | .8 | 1.6 | 1.1 | 2.0–8.0 |
| 3H/14C ratio in alkenyl PE | 10.0 | 3.4 | 6.8 | .3–2.4 |
| 3H/14C ratio in alkenyl PC | 1.5 | 2.0 | 3.0 | .3–2.0 |
| DHAP-AT activity in fibroblasts: | ||||
| DHAP-AT level (nmol/mg/2h) | .8 | 7.4 | 5.6 | 10.9 ± 2.5 |
| DHAP-AT/GDH ratiob | .3 | 3.2 | 2.2 | 6.6 ± 2.2 |
| Fatty-acid oxidation in fibroblasts: | ||||
| C16:0 (palmitic) (pmol/h/mg) | 1,999 | 3,078 | 3,151 | 2,841 ± 681 |
| C26:0 (cerotic) (pmol/h/mg) | 229 | 1,131 | 824 | 1,937 ± 440 |
| Pristanic (pmol/h/mg) | 9 | 287 | 167 | 1,126 ± 267 |
| Phytanic (pmol/h/mg) | 8 | 22 | 23 | 68 ± 29 |
PE = ethanolamine glycerophospholipid; PC = choline glycerophospholipid.
GDH = glutamate dehydrogenase.
Figure 1.
AGT immunolocalization and ultrastructure of liver. A, AGT immunolocalization in liver of index patient. Label is present in cytoplasm and nuclei but not in granules. The same image is seen after staining for catalase (not shown). B, AGT immunolocalization in liver of control. Peroxisomes (granules) are well visualized, and cytoplasm and nuclei are unstained (Scale bar = 10 μm). C, Ultrastructure of liver of index patient, showing stacks of trilamellar inclusions in angulate lysosome of macrophage; drops of insoluble lipid are also shown. Both inclusions are seen in all PBD livers (Scale bar = 0.1 μm).
This combination of clinical and laboratory findings is diagnostic of a generalized PBD. All PBDs are autosomal recessively inherited, and obligate heterozygote carriers do not display any clinical or biochemical features (Moser et al. 1999); therefore, parents do not usually undergo biochemical investigations. In this case, however, it was decided to investigate the parents, in view of their auditory and visual handicaps. Surprisingly, both parents were found to have elevated levels of plasma VLCFAs and plasma BCFAs (table 1). Subsequent studies of fibroblasts confirmed that both parents have a PBD, albeit in a mild form. VLCFA levels were clearly elevated in the father’s fibroblasts but only mildly so in the mother. Both had mildly deficient β-oxidation of VLCFAs and pristanic acid, partially deficient α-oxidation of phytanic acid, abnormal de novo plasmalogen synthesis, and mildly abnormal DHAP-AT activity. Results of thiolase immunoblotting were mildly abnormal in the father but were normal in the mother.
This family is unique in that a patient with PBD was born to parents who are themselves actually affected by—rather than simply being asymptomatic carriers of—a PBD. This fascinating observation indicates that deficiency of multiple peroxisomal functions in both the male partner and the female partner is not a barrier to normal conception and reproductive function. Such a union may be more common than a random occurrence, given that (a) deafness is a major feature of PBD and (b) there is a propensity for deaf individuals to marry within their own community.
The fact that both parents in this family are clinically and biochemically affected implies that they each must have mutations in both alleles of a PEX gene, rather than being heterozygous carriers of a single mutation. Furthermore, for their offspring to be affected, it could be deduced that both parents must be homozygous or compound heterozygous for mutations in the same PEX gene. This hypothesis was tested by complementation analysis, whereby fibroblasts from two different patients are fused to generate hybrid cells; restoration of function indicates involvement of different complementation groups (CG), reflecting mutations in different genes (Yajima et al. 1992). Using catalase immunofluorescence as the marker of complementation in fused cells, the analysis revealed that, indeed, the infant and his parents all belong to the same complementation group, CG-C (equivalent to CG4 in the U.S. and European classifications).
PBDs belonging to CG-C (i.e., CG4) are caused by mutations in the PEX6 gene [MIM 601498]. To determine the dysfunction of PEX6 in both the infant and his parents, we examined PEX6 cDNA from fibroblasts, by means of RT-PCR; all mutations found were confirmed at the gDNA level (table 2). As predicted by the complementation analysis, the infant and his parents were all found to have mutations on both alleles of their PEX6 gene, confirming that the parents are truly affected by—rather than only asymptomatic carriers for—a PBD. The infant was compound heterozygous for a 1715C→T mutation, leading to a T572I substitution at the protein level, and a IVS10+2T→C splice-site mutation in intron 10, leading to aberrant splicing involving the retention of intron 10, which probably does not lead to the synthesis of a functional protein. The infant’s father is homozygous for the 1715C→T mutation, whereas the infant’s mother is compound heterozygous for the IVS10+2T→C splice-site mutation and two missense mutations on the other allele (2426C→T and 2534T→C), leading to two amino acid substitutions (A809V and I845T, respectively). It is not clear whether both these missense mutations are disease causing or whether one of them represents a polymorphic variant. So far, neither has been identified after analysis of >50 PEX6 alleles. Analysis of the maternal grandparents’ DNA revealed that the mother inherited the splice-site mutation from her own mother and inherited the two missense mutations, on her second allele, from her father.
Table 2.
Primer Sets and Methods for PEX6 Mutation Analysis[Note]
| Amplicon | 5′ Primer (Forward) | 3′ Primer (Reverse) |
| cDNA analysis: | ||
| Fragment 1 | [−21M13]-ACTAGTCGTCTGGTTCTCTG | [M13rev]-GTGCCAGAAACCGCAAAGG |
| Fragment 1b | [−21M13]-TCCTCGTTGGTGTCCTGTC | [M13rev]-GTGCCAGAAACCGCAAAGG |
| Fragment 2 | [−21M13]-CCAGACTGTGTCCAGAGTC | [M13rev]-CACATAGAACATCCCCTTCC |
| Fragment 3 | [−21M13]-TGCCAGAGAGTTACACATCG | [M13rev]-ATGGCCTGCAGTTTTGTCTC |
| Fragment 4 | [−21M13]-TGGGAAGACCACAGTAGTTG | [M13rev]-TCCTCCTCAGTCAAGCCAC |
| Fragment 5 | [−21M13]-ACTTGGCACAGCTAGCACG | [M13rev]-TCCATCACTCCTCCAGAATC |
| Fragment 6 | [−21M13]-AAAGTGAGGAGAATGTGCGG | [M13rev]-TCTGTGGGCTATCAAGGTAC |
| gDNA analysis: | ||
| Exons 8 and 9 | [−21M13]-ACAAGGCAGTCCACAGGAG | [M13rev]-CCACCCACCCATCTACATC |
| Exons 10 and 11 | [−21M13]-ATGGGACGCTGATGGTGAG | [M13rev]-GAGCCGTCAGATGCACATAC |
| Exons 12 and 13 | [−21M13]-GTATGTGCATCTGACGGCTC | [M13rev]-TCTCTGGACTCTGAAGACTG |
| Exons 14 and 15 | [−21M13]-TAAAGAGAGGTACCACAGGC | [M13rev]-TGTTGCATGCATCCCCTAAG |
Note.— Total RNA or genomic DNA was isolated from primary skin fibroblasts by the Wizard RNA purification kit and the Wizard genomic DNA purification kit, respectively (Promega). For mutation analysis at the cDNA level, the coding region of PEX6 cDNA was amplified by PCR in six overlapping fragments from first-strand cDNA prepared from total RNA, as described elsewhere (IJlst et al. 1994), by use of the cDNA primer sets shown. For mutation analysis at the genomic level, the complete exons plus flanking intron sequences from the PEX6 gene were amplified by PCR using the gDNA primer sets shown. All forward and reverse primers used for mutation analysis were tagged with a −21M13 (5′-TGTAAAACGACGGCCAGT-3′) sequence and a M13rev (5′-CAGGAAACAGCTATGACC-3′) sequence, respectively. PCR fragments were sequenced in two directions, by “−21M13” and “M13rev” fluorescent primers, on an Applied Biosystems 277A automated DNA sequencer, according to the manufacturer's protocol (Perkin-Elmer).
Sixteen PEX6 mutations—including missense, nonsense, frameshift, and splice-site mutations—have been reported elsewhere (Fukuda et al. 1996; Yahraus et al. 1996; Zhang et al. 1999; Imamura et al. 2000; Matsumoto et al. 2001). Pex6p, the protein encoded by PEX6, is a member of the AAA ATPase family and is involved in the terminal steps of peroxisomal matrix–protein import (Collins et al. 2000). PEX6-defective cells are thus deficient in the importation of peroxisomal matrix proteins but retain some capacity for peroxisomal membrane biogenesis. Accordingly, using antibodies against the 70-kD peroxisomal membrane protein PMP70, we were able to identify remnant peroxisomal membranous structures, termed “ghosts,” in the infant’s liver and in fibroblasts from the infant and from his parents (not shown).
A remarkable feature in this unique family is the striking disparity between the two affected generations, in severity of both the clinical and the biochemical phenotypes. Whereas the infant’s presentation was intermediate between the severe ZS and NALD phenotypes, the parents’ disease was even milder than the IRD phenotype. Such milder variants have been reported elsewhere (Moser et al. 1995; Baumgartner et al. 1998). Two observations made in catalase-immunofluorescence–microscopy studies correlate with the parents’ milder phenotypes: mosaicism and temperature sensitivity (TS). In both parents’ fibroblasts, catalase-immunofluorescence microscopy revealed a mosaic pattern, with peroxisomes present in some cells and absent in others. Whether the parents have a mosaic pattern also in hepatic tissue could not be determined, since biopsy of the liver was not justified. The fibroblasts and liver sample of the infant, who was more severely affected, did not display peroxisomal mosaicism. Mosaicism in livers of patients with PBD has been found to be associated with a milder clinical course (Espeel et al. 1995a; Giros et al. 1996; Pineda et al. 1999) and has been described in patients belonging to CG4 and CG6, which are associated with PEX6 mutations (Pineda et al. 1999, Matsumoto et al. 2001).
The second observation indicative of a milder phenotype is TS, the restoration of morphological peroxisome formation and biochemical function in PBD fibroblasts cultured at 30°C, rather than at 37°C. This has been demonstrated only in fibroblasts with the milder NALD and IRD phenotypes, but never in ZS cells. TS was evaluated by determination of the percentage of immunofluorescent-catalase–positive cells counted among 20 cells in each of five fields at ×1,000 magnification, after incubation of fibroblasts for 72 h at either 37°C or 30°C (Imamura et al. 1998). At 37°C, peroxisomes were not detected at all in fibroblasts from the infant or his mother and in only 7% of fibroblasts from his father; at 30°C, however, peroxisomes were partially biosynthesized in the infant (37% of fibroblasts), whereas peroxisomes were detected in most of the cells from his mother (84% of fibroblasts) and from his father (82% of fibroblasts). This finding of TS in the parents’ fibroblasts is consistent with their relatively mild clinical phenotype, whereas the partial TS in the infant’s fibroblasts correlates with his clinical phenotype, which is intermediate between that of classic ZS and classic IRD. In previous studies, TS has been described in a single PEX6 mutant (Imamura et al. 2000), as well as in PEX1, PEX2, and PEX13 mutants; all TS mutants identified thus far are missense mutations (Suzuki et al. 2001). The underlying basis for TS mutants has not been definitely resolved but probably is related to a more effective folding process at lower temperature.
There is evidence to support the concept of phenotype-genotype correlation for PBD caused by PEX1, PEX5, PEX7, PEX10, and PEX12 defects (Moser 1999), and this correlation recently has been suggested for PEX6 also (Gootjes et al. 2001). From the clinical presentations and corresponding genotypes in this family, it can be inferred that the IVS10+2T→C allele has the most severe—and that the A809V+I845T allele has the least severe—effect on the production and/or function of Pex6p protein. This implies that, in the infant, the T572I allele accounts for the partially temperature-sensitive phenotype. The difference, in severity of phenotypes, between the parents and the offspring cannot be fully assessed, however, until the functional consequences that these mutations have for importation are determined by expression studies in vitro (Zhang et al. 1999; Imamura et al. 2000).
The differential diagnosis of sensorineural deafness associated with retinopathy includes USH, PBD, intrauterine infections, perinatal complications, mitochondrial respiratory-chain disorders, Alstrom syndrome, Senior-Loken syndrome, and retinitis pigmentosa inversa with deafness. Since USH is the most commonly identified cause of combined deafness and blindness in developed countries, it is understandable that the parents in this study initially were labeled with this diagnosis. Deafness may be the predominant and presenting feature of mild varieties of PBD, and additional manifestations (such as, in this family, RP or transient neonatal cholestasis) may be subtle and easily missed. There is considerable overlap between the clinical manifestations of USH and those of milder PBD phenotypes. On the basis of both our own experience and additional reports of patients with PBD who have been misdiagnosed as having USH (Weleber et al. 1984; Bader et al. 2000), we advocate that, in patients with combined sensorineural deafness and RP and suspected USH, PBD should be excluded, starting with examination of plasma VLCFAs and BCFAs.
Acknowledgments
Antibodies against PMP70 were donated by Stefan Alexson (Karolinska Institutet, Huddinge, Sweden). This study was supported in part by European Concerted Action “Peroxisomal Leukodystrophy” grant BMH4 CT 96-1621.
Electronic-Database Information
Accession numbers and the URL for data in this article are as follows:
- Online Mendelian Inheritance in Man (OMIM), http://www.ncbi.nlm.nih.gov/Omim/ (for IRD [MIM 266510], NALD [MIM 202370], PBD [MIM 601539], PEX6 [MIM 601498], USH [MIM 276900–276906, MIM 601067, MIM 602083, MIM 602097, and MIM 605472], and ZS [MIM 214100])
References
- Bader PI, Dougherty S, Cangany N, Raymond G, Jackson CE (2000) Infantile Refsum disease in four Amish sibs. Am J Med Genet 90:110–114 [PubMed] [Google Scholar]
- Baumgartner MR, Poll-Thé BT, Verhoeven NM, Jakobs C, Espeel M, Roels F, Rabier D, Levade T, Rolland MO, Martinez M, Wanders RJA, Saudubray JM (1998) Clinical approach to inherited peroxisomal disorders: a series of 27 patients. Ann Neurol 44:720–730 [DOI] [PubMed] [Google Scholar]
- Collins CS, Kalish JE, Morrell JC, McCaffery JM, Gould SJ (2000) The peroxisome biogenesis factors Pex4p, Pex22p, Pex1p, and Pex6p act in the terminal steps of peroxisomal matrix protein import. Mol Cell Biol 20:7516–7526 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Espeel M, Mandel H, Poggi F, Smeitink JAM, Wanders RJA, Kerckaert I, Schutgens RBH, Saudubray JM, Poll-The BT, Roels F (1995a) Peroxisome mosaicism in the livers of peroxisomal deficiency patients. Hepatology 22:497–504 [PubMed] [Google Scholar]
- Espeel M, Roels F, Giros M, Mandel H, Peltier A, Poggi F, Poll-The BT, Smeitink JA, Van Maldergem L, Santos MJ (1995b) Immunolocalization of a 43 kDa peroxisomal membrane protein in the liver of patients with generalized peroxisomal disorders. Eur J Cell Biol 67:319–327 [PubMed] [Google Scholar]
- Espeel M, Van Limbergen G (1995) Immunocytochemical localization of peroxisomal proteins in human liver and kidney. In: Roels F, De Bie S, Ruud BH, Schutgens RBH, Besley GTN (eds) Diagnosis of human peroxisomal disorders: a handbook. J Inherit Metab Dis 18 Suppl 1:135–154 [DOI] [PubMed] [Google Scholar]
- Fukuda S, Shimozawa N, Suzuki Y, Zhang Z, Tomatsu S, Tsukamoto T, Hashiguchi N, Osumi T, Masuno M, Imaizumi K, Kuroki Y, Fujiki Y, Orii T, Kondo N (1996) Human peroxisome assembly factor-2 (PAF-2): a gene responsible for group C peroxisome biogenesis disorder in humans. Am J Hum Genet 59:1210–1220 [PMC free article] [PubMed] [Google Scholar]
- Giros M, Roels F, Prats J, Ruiz M, Ribes A, Espeel M, Wanders RJ, Schutgens RB, Pampols T (1996) Long survival in a case of peroxisomal biogenesis disorder with peroxisome mosaicism in the liver. Ann NY Acad Sci 804:747–749 [DOI] [PubMed] [Google Scholar]
- Gootjes J, Prins GT, Barth P, Poll-The BT, Waterham HR, Wanders RJA (2001) Molecular basis of Zellweger syndrome and other peroxisome biogenesis disorders: mutation analysis of human PEX6 gene. J Inherit Metab Dis 24 Suppl 1:90 [Google Scholar]
- Gould SJ, Valle D (2000) Peroxisome biogenesis disorders: genetics and cell biology. Trends Genet 16:340–345 [DOI] [PubMed] [Google Scholar]
- IJlst L, Wanders RJ, Ushikubo S, Kamijo T, Hashimoto T (1994) Molecular basis of long-chain 3-hydroxyacyl-CoA dehydrogenase deficiency: identification of the major disease-causing mutation in the alpha-subunit of the mitochondrial trifunctional protein. Biochim Biophys Acta 1215:347–350 [DOI] [PubMed] [Google Scholar]
- Imamura A, Shimozawa N, Suzuki Y, Zhang Z, Tsukamoto T, Fujiki Y, Orii T, Osumi T, Wanders RJA, Kondo N (2000) Temperature-sensitive mutation of PEX6 in peroxisome biogenesis disorders in complementation group C (CG-C): comparative study of PEX6 and PEX1. Pediatr Res 48:541–545 [DOI] [PubMed] [Google Scholar]
- Imamura A, Tsukamoto T, Shimozawa N, Suzuki Y, Zhang Z, Imanaka T, Fujiki Y, Orii T, Kondo N, Osumi T (1998) Temperature-sensitive phenotypes of peroxisome-assembly processes represent the milder forms of human peroxisome-biogenesis disorders. Am J Hum Genet 62:1539–1543 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korman SH, Mandel H, Gutman A (2000) Characteristic urine organic acid profile in peroxisomal biogenesis disorders. J Inherit Metab Dis 23:425–428 [DOI] [PubMed] [Google Scholar]
- Matsumoto N, Tamura S, Moser A, Moser HW, Braverman N, Suzuki Y, Shimozawa N, Kondo N, Fujiki Y (2001) The peroxin Pex6p gene is impaired in peroxisomal biogenesis disorders of complementation group 6. J Hum Genet 46:273–277 [DOI] [PubMed] [Google Scholar]
- Moser AB, Kreiter N, Bezman L, Lu S, Raymond GV, Naidu S, Moser HW (1999) Plasma very long chain fatty acids in 3,000 peroxisome disease patients and 29,000 controls. Ann Neurol 45:100–110 [DOI] [PubMed] [Google Scholar]
- Moser AB, Rasmussen M, Naidu S, Watkins PA, McGuinness M, Hajra AK, Chen G, Raymond G, Liu A, Gordon D, Garnaas K, Walton DS, Skjeldal OH, Guggenheim MA, Jackson LG, Elias RE, Moser HW (1995) Phenotypes of patients with peroxisomal disorders subdivided into sixteen complementation groups. J Pediatr 127:13–22 [DOI] [PubMed] [Google Scholar]
- Moser HW (1999) Genotype-phenotype correlations in disorders of peroxisome biogenesis. Mol Genet Metab 68:316–327 [DOI] [PubMed] [Google Scholar]
- Petit C (2001) Usher syndrome: from genetics to pathogenesis. Annu Rev Genomics Hum Genet 2:271–297 [DOI] [PubMed] [Google Scholar]
- Pineda M, Girós M, Roels F, Espeel M, Ruiz M, Moser A, Moser HW, Wanders RJA, Pavia C, Conill J, Aracil A, Amat L, Pampols T (1999) Diagnosis and follow-up of a case of peroxisomal disorder with peroxisomal mosaicism. J Child Neurol 14:434–439 [DOI] [PubMed] [Google Scholar]
- Roels F, De Prest B, De Pestel G (1995) Liver and chorion cytochemistry. In: Roels F, De Bie S, Ruud BH, Schutgens RBH, Besley GTN (eds) Diagnosis of human peroxisomal disorders: a handbook. J Inherit Metab Dis 18 Suppl 1:155–171 [DOI] [PubMed] [Google Scholar]
- Schrakamp G, Schalkwijk CG, Schutgens RB, Wanders RJA, Tager JM, van den Bosch H (1988) Plasmalogen biosynthesis in peroxisomal disorders: fatty alcohol versus alkylglycerol precursors. J Lipid Res 29:325–334 [PubMed] [Google Scholar]
- Suzuki Y, Shimozawa N, Imamura A, Fukuda S, Zhang Z, Orii T, Kondo N (2001) Clinical, biochemical and genetic aspects and neuronal migration in peroxisome biogenesis disorders. J Inherit Metab Dis 24:151–165 [DOI] [PubMed] [Google Scholar]
- Wanders RJA (1999) Peroxisomal disorders: clinical, biochemical, and molecular aspects. Neurochem Res 24:565–580 [DOI] [PubMed] [Google Scholar]
- Wanders RJ, Dekker C, Ofman R, Schutgens RB, Mooijer P (1995a) Immunoblot analysis of peroxisomal proteins in liver and fibroblasts from patients. In: Roels F, De Bie S, Ruud BH, Schutgens RBH, Besley GTN (eds) Diagnosis of human peroxisomal disorders: a handbook. J Inherit Metab Dis 18 Suppl 1:101–112 [DOI] [PubMed] [Google Scholar]
- Wanders RJ, Denis S, Ruiter JP, Schutgens RB, van Roermund CW, Jacobs BS (1995b) Measurement of peroxisomal fatty acid beta-oxidation in cultured human skin fibroblasts. In: Roels F, De Bie S, Ruud BH, Schutgens RBH, Besley GTN (eds) Diagnosis of human peroxisomal disorders: a handbook. J Inherit Metab Dis 18 Suppl 1:113–124 [DOI] [PubMed] [Google Scholar]
- Wanders RJ, Ofman R, Romeijn GJ, Schutgens RB, Mooijer PA, Dekker C, van den Bosch H (1995c) Measurement of dihydroxyacetone-phosphate acyltransferase (DHAPAT) in chorionic villous samples, blood cells and cultured cells. In: Roels F, De Bie S, Ruud BH, Schutgens RBH, Besley GTN (eds) Diagnosis of human peroxisomal disorders: a handbook. J Inherit Metab Dis 18 Suppl 1:90–100 [DOI] [PubMed] [Google Scholar]
- Wanders RJA, Van Roermund CWT (1993) Studies on phytanic acid alpha-oxidation in rat liver and cultured human skin fibroblasts. Biochim Biophys Acta 1167:345–350 [DOI] [PubMed] [Google Scholar]
- Wanders RJA, Wiemer EA, Brul S, Schutgens RBH, van den Bosch H, Tager JM (1989) Prenatal diagnosis of Zellweger syndrome by direct visualization of peroxisomes in chorionic villus fibroblasts by immunofluorescence microscopy. J Inherit Metab Dis 12 Suppl 2:301–304 [DOI] [PubMed] [Google Scholar]
- Weleber RG, Tongue AT, Kennaway NG, Budden SS, Buist NRM (1984) Ophthalmic manifestations of infantile phytanic acid storage diseases. Arch Ophthalmol 102:1317–1321 [DOI] [PubMed] [Google Scholar]
- Yahraus T, Braverman N, Dodt G, Kalish JE, Morrell JC, Moser HW, Valle D, Gould SJ (1996) The peroxisome biogenesis disorder group 4 gene, PXAAA1, encodes a cytoplasmic ATPase required for stability of the PTS1 receptor. EMBO J 15:2914–2923 [PMC free article] [PubMed] [Google Scholar]
- Yajima S, Suzuki Y, Shimozawa N, Yamaguchi S, Orii T, Fujiki Y, Osumi T, Hashimoto T, Moser HW (1992) Complementation study of peroxisome-deficient staining and characterization of fused cells. Hum Genet 88:491–499 [DOI] [PubMed] [Google Scholar]
- Zhang Z, Suzuki Y, Shimozawa N, Fukuda S, Imamura A, Tsukamoto T, Osumi T, Fujiki Y, Orii T, Wanders RJA, Barth PG, Moser HW, Paton BC, Besley GT, Kondo N (1999) Genomic structure and identification of 11 novel mutations of the PEX6 (peroxisome assembly factor-2) gene in patients with peroxisome biogenesis disorders. Hum Mutat 13:487–496 [DOI] [PubMed] [Google Scholar]