Abstract
Epithelial-msenchymal transition (EMT) contributes to posterior capsule opacification (PCO) type of cataract. Transcription factors Snail is a key trigger of EMT activated by transforming growth factor β (TGFβ). This study was done to investigate the effect of Snail targeting siRNA on TGFβ2-induced EMT in human lens epithelial cells. TGFβ2 treatment of cultured human epithelial cell line (HLEB3) upregulated the expression of Snail and the EMT relevant molecules such as vimentin and α-SMA but downregulated the expression of keratin and E-cadherin. After the stimulation of TGFβ2, the HLEB3 cells became fibroblast-like in morphology, and the junctions of cell-cell disappeared. TGFβ2 treatment also enhanced migration ability of HLEB3 cells. TGFβ2-induced Snail expression and EMT were significantly inhibited by Snail siRNA. By analyzing the response characteristics of HLEB3 in TGFβ2-induced EMT model with/without Snail-specific siRNA, we concluded that Snail is an element in the EMT of HLEB3 cells induced by TGFβ2. Snail siRNA targeting can block the induced EMT and therefore has the potential to suppress the development of PCO.
1. Introduction
Epithelial-mesenchymal transition (EMT) is a programmed development of biological cells characterized by loss of cell adhesion, repression of E-cadherin expression, increased cell mobility, and change of morphology. EMT is a highly conserved and fundamental process not only in development, but also in fibrosis, metastasis of tumor cells, and wound healing [1–4]. In cataract surgery, where entire lens content is removed, lens epithelial cells (LECs) can undergo EMT, migrate to the posterior capsular surface, and result in fibrosis of the posterior capsule as well as the residual anterior capsule [4–6]. Clinically, the EMT of LECs after cataract lens removal usually results in secondary cataract that can present as anterior polar cataracts and/or posterior capsular opacification [7, 8].
During EMT, epithelium cells undergo transdifferentiation toward a myofibroblastic phenotype. The two cell types have different skeletal proteins, keratin for epithelium and vimentin for myofibroblastic. The cells derived from surface ectoderm always express E-cadherin to form adherence to each other. The EMT process involves transcriptional reprogramming of a series of genes that include α-SMA known as a maker of myofibroblast cells. Therefore, except for the distinct expression of keratin and vimentin, the α-SMA expression is considered as the feature of LECs transdifferentiation as well as the loss of E-cadherin production [9–11].
TGFβ is composed of homodimeric polypeptides that regulate many aspects of cellular function, including cell growth, differentiation, inflammation, and wound healing [12–14]. Numerous in vitro and in vivo studies have indicated the role of active TGFβ in promoting an aberrant differentiation pathway and EMT of various epithelial tissues [15, 16]. Although five members of the TGFβ family have currently been identified, only TGFβ isoforms 1, 2, and 3 have been detected in mammals [17]. TGFβ1 and TGFβ2 are expressed in human lens and release abundantly in the ocular media [18]. The predominant form of TGFβ1 and TGFβ2 is in the latent [19] but can be activated under pathological conditions such as inflammation, fibrosis, trauma, and surgery after a 25-kDa dimer cleaved from its latent precursor [20]. The amount of TGFβ in aqueous humor after cataract surgery with intraocular lens implantation ranged from 2.3 to 8.1 ng/mL with 61% of it present in the active form [21]. Normally, the activity of TGFβ in the eye appears to be highly regulated by vitreous containing molecules [8]. TGFβ2 is expressed at much higher levels than the other isoforms in the aqueous humor and vitreous and thus is likely to be a major mediator of EMT in LECs in vivo [22, 23].
The Snail family members are a group of transcription factors that are involved in regulation of EMT induced by TGFβ during embryonic development and tumor progression [24–28]. They are involved in many embryonic processes, such as the ingression of the early mesodermal cells at gastrulation and the delamination of the neural crest from the neural tube [29]. In adult, Snail was mainly expressed in heart, lung, brain, and skeletal muscle, but there is no expression in most normal organs/tissues including eyes [30]. However, Snail can be expressed in ocular tissue under pathological conditions especially fibrotic diseases such as corneal scarring [31], subcapsular cataract [32], and proliferative vitreoretinopathy (PVR) [33]. Indeed, Snail is activated to induce EMT in mammalian cells and suppress the expression of E-cadherin [8, 34, 35]. Cho et al. have reported the role of Snail in ETM of mouse lens epithelial cells [36].
In the present study, we sought to confirm the involvement of Snail gene in TGFβ2-induced EMT of human LECs and to test a novel hypothesis that the inhibition of Snail expression by siRNA can block TGFβ2-induced EMT.
2. Material and Methods
2.1. Cells and Cell Culture
Human lens epithelial cell line HLEB3 was purchased from ATCC. Cells were cultured in Dulbecco's modified Eagle's medium (DMEM; Invitrogen, CA, USA) supplemented with 15% fetal bovine serum (FBS; Invitrogen, CA, USA). All culture medium contained no antibiotics. The TGFβ2 treatment was carried out after the cells were incubated in serum-free medium for 24 hours, and 10 ng/mL of TGFβ2 was added to the culture medium for the indicated times.
2.2. Reagents and Antibodies
Recombinant human TGFβ2 was purchased from Peprotech (Rocky, Hill, NJ, USA). Anti-E-cadherin and keratin antibodies were purchased from Cell Signaling (Beverly, MA, USA). Anti-Snail and vimentin antibodies were obtained from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Anti-α-SMA antibody was purchased from Abcam (Cambridge, MA, USA). CY3/FITC tagged secondary antibodies were from BOSTER (Wuhan, China).
2.3. siRNA and Transfection
According to Elbashir's principle [37], four siRNAs (P1–P4) targeting human Snail and one negative control siRNA (P5) were designed using web-based software (http://www.ambion.com/techlib/misc/siRNA_finder.html) and synthesized chemically (Biomics, Nantong, China) (Table 1). The siRNAs were transfected into HLEB3 cells by liposome Lipofectamine 2000 according to the manufacturer's protocol (Invitrogen, CA, USA). The siRNA treatment was performed before the TGFβ2 stimulation.
Table 1.
siRNA sequences for snail targeting and negative control.
| siRNA duplex |
siRNA duplex sequences (5′-3′) | |
|---|---|---|
| P1 | Sense: | GAAUGUCCCUGCUCCACAAGCdTdT |
| Antisense: | GCUUGUGGAGCAGGGACAUUCdTdT | |
| P2 | Sense: | GCGAGCUGCAGGACUCUAAUCdTdT |
| Antisense: | GAUUAGAGUCCUGCAGCUCGCdTdT | |
| P3 | Sense: | CCUUCGUCCUUCUCCUCUACUdTdT |
| Antisense: | AGUAGAGGAGAAGGACGAAGGdTdT | |
| P4 | Sense: | CAGAUGUCAAGAAGUACCAGUdTdT |
| Antisense: | ACUGGUACUUCUUGACAUCUGdTdT | |
| P5 | Sense: | UUCUCCGAACGUGUCACGUdTdT |
| Antisense: | ACGUGACACGUUCGGAGAAdTdT | |
Four siRNAs (P1–P4) were designed from the coding sequence of the human Snail gene. The siRNA duplex sequences are listed. A nonspecific, scrambled siRNA duplex as negative control (P5) was used as a control.
2.4. Quantification of Snail mRNA
Total RNA of HLEB3 cells was extracted for cDNA synthesis using RISO reagent (RISO; Biomics, Nantong, China). cDNA was synthesized by MLV reverse transcriptase using 2 μg total RNA in a total volume of 20 μL (QuantiTect, Qiagen, Germany). The Snail transcript was detected by quantitative RT-PCR using iCycler iQ System (Bio-Rad Laboratories, Hercules, CA, USA) and SYBR Green QPCR Master Mix (Biomics, Nantong, China). The primers for snail are forward 5′-TCGTCCTTCTCCTCTACTTCAG-3′ and reverse 5′-CGTGTGGCTTCGGATGTG-3′, which amplify a 201 bp target. For the internal control, GAPDH was amplified using primers forward 5′-GAAGGTGAAGGTCGGAGTC-3′ and reverse 5′-GAAGATGGTGATGGGATTTC-3′, which amplify a 226 bp target. Following PCR, a thermal melt profile was performed for amplicon identification. The specificity of the amplification reactions was also confirmed by agarose gel electrophoresis. The relative expression was presented as fold changes after normalizing to the GAPDH control.
2.5. Immunofluorescent Staining
HLEB3 cells were grown on glass coverslips before siRNAs were transfected and then exposed to 10 ng/mL of TGFβ2 for 1 hour. Cells were fixed with 4% paraformaldehyde for 30 min at 4°C, followed by incubation with 0.1% Triton X-100 and 3% BSA for 2 h in room temperature for permeabilization and blocking. The primary antibodies (1 : 100) against Snail, vimentin, E-cadherin, keratin, or α-SMA diluted in PBS were placed on cells for overnight at 4°C, respectively, followed by incubation with CY3-conjugated goat anti-rabbit or FITC-conjugated goat anti-mouse immunoglobulin (1 : 200) for 2 hours at 37°C in the dark. The nuclei were counterstained with Hoechst 33258 (Invitrogen, CA, USA). Images were acquired with a fluorescence microscope (DM4000B, Leica, Germany).
2.6. Transwell Assay
Transwell apparatus with 8 μm pore size membrane (Costar, Cambridge, MA, USA) was used to detect the migration ability of HLEB3 cells. The siRNAs-treated HLEB3 cells were exposed to 10 ng/mL of TGFβ2 for 48 h. Serum-free DMEM containing 1 × 105 cells in 100 μL was added into the upper chamber; the lower chamber contained 500 μL of 15% FBS-containing medium. After incubation at 37°C for 24 h, membranes were swabbed with a cotton swab, soaked in 0.1% crystal violet for 10 min, and then washed with PBS. The number of cells attached to the lower surface of the polycarbonate filter was counted at 100x magnification under a light microscope.
2.7. Statistical Analysis
All results are expressed as the mean ± SD. The data were analyzed with ANOVA and SNK-q test using SPSS17.0. P < 0.05 was considered to be statistically significant.
3. Results
3.1. Expression of Snail Induced by TGFβ2
To determine whether the expression of Snail is regulated by TGFβ2, we examined the expression and intracellular localization of Snail in HLEB3 cells. RT-PCR results indicated that, in the absence of TGFβ2, there was no Snail expression in HLEB3 cells whereas the level of Snail mRNA was significantly elevated in cells stimulated with TGFβ2. TGFβ2-induced Snail expression was does dependent, and the expression was detected as early as 0.5 h after the treatment (Figures 1 and 2).
Figure 1.
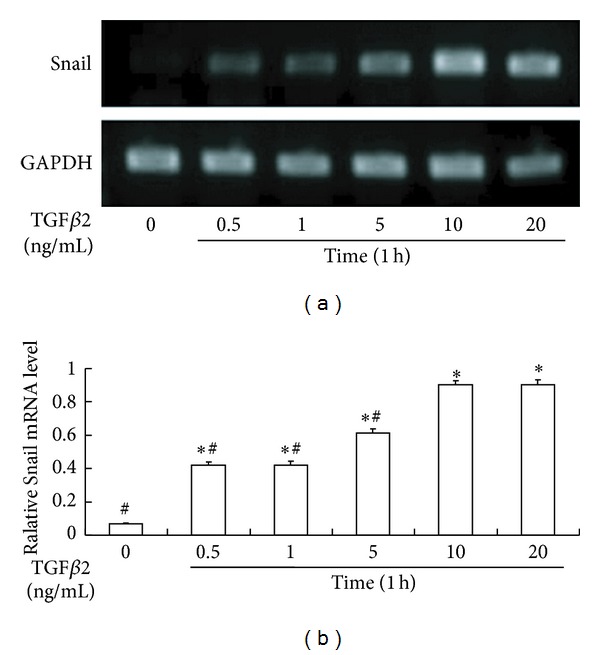
TGFβ2-induced expression of Snail mRNA in a dose-dependent manner. (a) Representative agarose gel electrophoresis images of Snail and house gene expression after TGFβ2 treatment. (b) The summary of triplicated experiments. *P < 0.05 compared with TGFβ2 (−) (0 ng/mL). # P < 0.05 compared with the group treated with 10 ng/mL TGFβ2.
Figure 2.
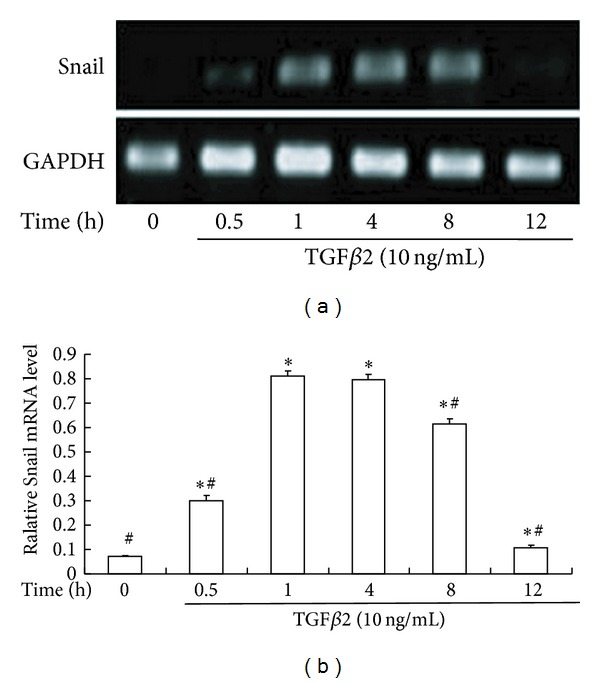
The time course of TGFβ2-induced expression of Snail mRNA. (a) Representative agarose gel electrophoresis images revealed TGFβ2-induced early expression of Snail. (b) The summary of triplicated experiments. *P < 0.05 compared with TGFβ2 (−) (0 h). # P < 0.05 compared with TGFβ2 (+) (1 h).
Consistent with the mRNA expression, Snail protein synthesis was induced after stimulation by TGFβ2. In the absence of TGFβ2, the cells showed no immunoreactivity for the protein. However, Snail protein production was greatly increased in the presence of TGFβ2, and immunostaining was detected mainly in the nucleus and nearby cytosol (Figure 3).
Figure 3.
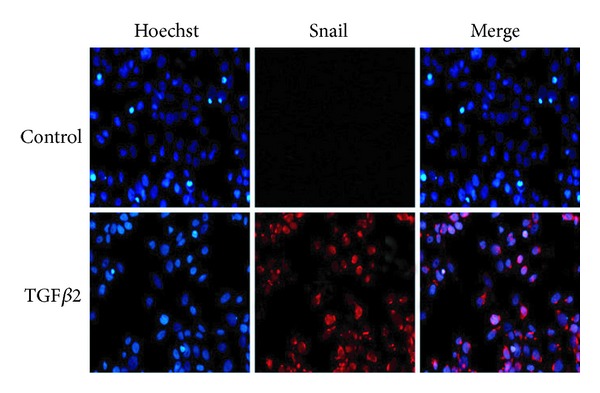
TGFβ2-induced expression of Snail protein. HLEB3 cells were incubated in the absence or presence of 10 ng/mL TGFβ2. After 8 hours of culture, cells were immunofluorescence stained with anti-Snail antibody (red) and counterstained with Hoechst (blue). Snail were expressed after TGFβ2 treatment and located in nuclear (400x).
3.2. Efficiency of siRNAs Inhibition of Snail Expression
Four Snail siRNAs (P1–P4) inhibited the expression of Snail mRNA expression after TGFβ2 treatment by 55.00% (P1), 74.85% (P2), 49.85% (P3), and 43.98% (P4), respectively (P < 0.05), while the negative control siRNA (P5) showed no effects (Figure 4). Because P2 was the most efficient in the inhibition, it was used in the following experiments.
Figure 4.
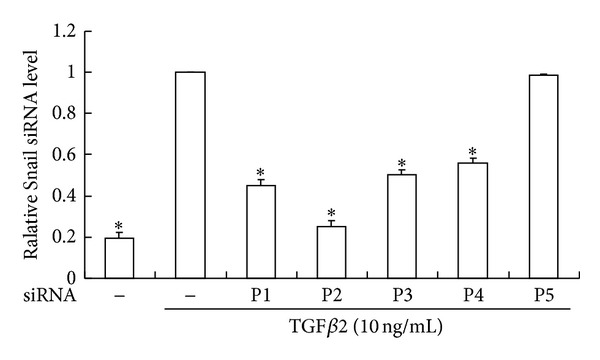
Efficiency of four siRNAs (P1–P4) on Snail expression. Serum starved HLEB3 cells were transfected with human Snail siRNAs (P1–P4) and negative control (P5) before being stimulated with TGFβ2 for 1 hour. Snail expressions were significantly decreased with the siRNA treatment. The data were collected from 3 experiments. *P < 0.05 compared with siRNA (−)/TGFβ2 (+) (10 ng/mL).
3.3. Role of Snail in TGFβ2-Induced EMT of HLEB3
The Snail siRNA (P2) reduced the Snail protein expression as well as the mRNA level induced by TGFβ2 (Figure 5). Although LECs are derived from surface ectoderm, they express vimentin [38] as well as the epithelial surface marker, keratin, and E-cadherin. The vimentin is expressed physiologically in an appropriate amount while overexpression is an evidence of EMT. Immunofluorescence analysis for EMT relevant proteins revealed that keratin, E-cadherin, and vimentin were expressed in normal HLEB3 cells but not α-SMA. The TGFβ2-induced repression of keratin and E-cadherin production was significantly abolished by the Snail targeting siRNA. The increase of vimentin and α-SMA by TGFβ2 was inhibited by the siRNA treatment (Figure 6).
Figure 5.
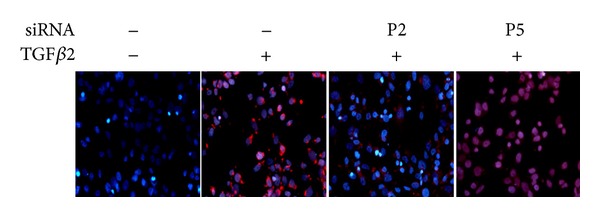
siRNA inhibition of Snail protein expression. Serum starved HLEB3 cells were transfected with human Snail siRNA (P2) and negative control (P5) before being stimulated with TGFβ2 for 8 hours. Cells were stained with anti-Snail antibody (red) and counterstained with Hoechst (blue). Images were taken by fluorescence microscope (400x).
Figure 6.
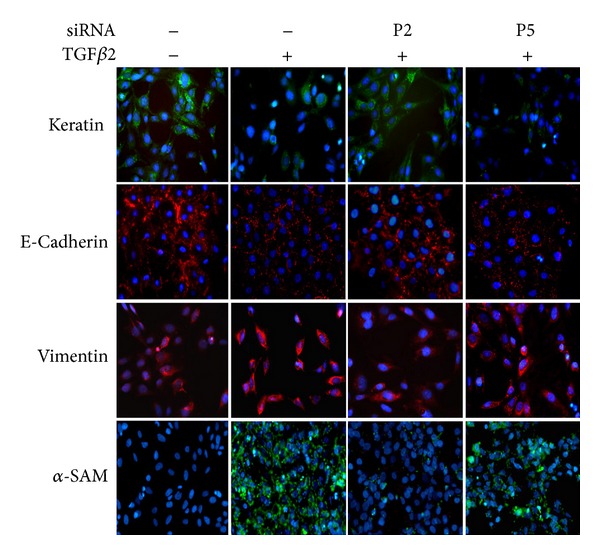
siRNA inhibition of EMT relevant molecules. Serum starved HLEB3 cells were transfected with human Snail siRNA (P2), negative control siRNA (P5). Then cells were stimulated with TGFβ2 for 24 hours. Various cellular proteins were detected by immunofluorescence staining. Images were taken by fluorescence microscope (400x).
The observation of the morphology of HLEB3 cells showed that untreated HLEB3 cells were polygonal with tight junction. After the stimulation of TGFβ2, the cells became longer and slimmer, spindly shaped as fibroblast, and the junctions of cell-cell were lost. Snail targeting siRNA reversed those morphological changes (Figure 7).
Figure 7.

Morphological changes of HLEB3 cells. Serum starved HLEB3 cells were transfected with human Snail siRNA (P2) and negative control siRNA (P5) before the cells were stimulated with TGFβ2. The morphology of the cells was observed under inverted microscope. (a) TGFβ2-induced cells became spindly shaped. (b) Snail targeting siRNA prevented the cells from the TGFβ2-induced morphological change (200x).
There were few untreated HLEB3 cells that passed through the polycarbonate. The migration of TGFβ2-treated cells was markedly higher than the untreated cells (P < 0.05). The treatment of Snail siRNA (P2) significantly blocked the increased migration stimulated by TGFβ2 (P < 0.05) (Figure 8).
Figure 8.
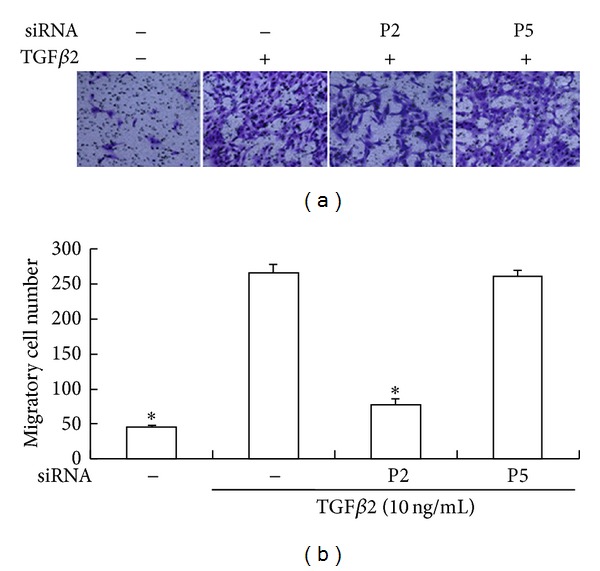
Inhibition of migration ability by Snail siRNA. Serum starved HLEB3 cells were transfected with human Snail siRNA (P2), negative control siRNA (P5) before the cells were stimulated with TGFβ2 for 48 h. Transwell assay was used to detect the migration ability of cells. (a) Crystal violet stained transmembrane cells under light microscope (100x). (b) The count of migrated HLEB3 cells from triplicated experiments.*P < 0.05 compared with siRNA (−)/TGFβ2 (+) (10 ng/mL).
4. Discussion
In this study, we successfully established a human LEC EMT model and found that Snail targeting siRNA can efficiently inhibit TGFβ2-induced EMT of human LECs, which has not been reported previously. The data indicated the potential to use siRNA approach to suppress development of PCO after cataract surgery.
At present, surgery is the only effective treatment of cataract to restore impaired vision. Unfortunately, many patients suffer a secondary loss of vision over time because of PCO. PCO is the most common long-term complication of cataract surgery. The incidence of PCO is approximately 50% in adults and 100% in children [39–42]. It usually causes a decrease in visual acuity by blocking the visual axis and striae or folds in the posterior capsule. In addition, traction-induced intraocular lens (IOL) malposition, which needed further corrective surgery, can occur during PCO.
PCO is usually caused by the proliferation, migration, EMT, collagen deposition, and lens fiber regeneration of residual LECs [43–46] because the surgery induces a wound-healing response in the lens. Usually, proliferation of the remaining LECs starts within a few hours after cataract surgery [47]. Proliferation and migration of LECs may precede EMT, and the two events are thought to be independently regulated [48, 49]. Therefore, postsurgical medical inhibition of LECs' proliferation, migration, and EMT would be an option for preventing PCO.
Myofibroblasts play a central role in the process of tissue fibrosis and scarring. This cell type is derived from both activated fibroblasts and epithelial cells including LECs. Expression of α-SMA, a marker for fibroblast-myofibroblast conversion, is mediated by Smads [50]. The transdifferentiation in which an epithelial cell changes its phenotype to a myofibroblast involves many transcription factors including ZEB (Sip1/dEF1), bHLH (E47/Twist), and Snail1/2 [51–54]. These transcription factors are upregulated by TGFβ and directly suppress E-cadherin promoter, which is essential in the maintenance of epithelial phenotype. Expression of Snail, the master transcription factor involved in an early step of the EMT, is considered as an important factor in the tissue fibrosis in the eye [7].
We focus on Snail because of its relation in cellular proliferation and differentiation. Snail is a member of a family of zinc finger-containing transcriptional repressors. Snail family is implicated in the transcriptional repression of E-cadherin by interacting with the E-box sequence in the proximal E-cadherin promoter. So, the function of the gene is associated with suppression of the epithelial phenotype [55]. The gene had been shown to be a master gene for early stage of EMT [51, 56, 57].
Cho et al. had reported that TGFβ induced Snail expression in mouse lens epithelial cells [36]. It is also reported that Slug (Snail2, another member of Snail superfamily) was expressed in anterior polar cataracts and human lens epithelial cell lines [58].
RNA interference has become a standard method for in vitro knockdown of any target gene of interest. siRNA can incorporate into a protein complex that recognizes and cleaves target mRNA [59]. Compared to small chemicals for the purpose of inhibition, siRNA mimics RNAi that is a common phenomenon in living creature and is believed to be safe and efficient in the inhibition of a specific gene expression. Four siRNAs against Snail were used to avoid off-target effects. Our data suggested that all the designed siRNAs inhibited the expression of Snail notably.
In this study, we have demonstrated that Snail is an early responder of TGFβ in EMT of human LECs. TGFβ2-treated HLEB3 cells lose their epithelium character and gain mesenchymal feature. Snails are implicated in the repression by interacting with the E-box sequence in the proximal E-cadherin promoter, which is associated with morphologic changes in cells that occur during EMT in embryonic development and in tumor cell invasion [27, 34, 35]. We confirmed the similar mechanism in HLEB3 cells. TGFβ2 changed the polygonal LECs to elongated shape and lost contact with their neighbors. These cells gained notable migration ability. We presumed that the loss of cells' junction is caused by Snail-induced E-cadherin's reduction and the contractive property of α-SMA contributes to the migration. We found that all these EMT relevant changes were blocked by targeting Snail.
In conclusion, our data indicated that TGFβ2 induces Snail expression and EMT of human LECs, and Snail is an essential factor in this process. Snail targeting siRNA inhibits Snail expression and EMT in human LECs, and might be a candidate strategy to prevent subcapsular cataract including PCO.
Conflict of Interests
The authors declare that there is no conflict of interests. The authors have no commercial interest in any materials discussed in this paper.
Acknowledgments
This research was supported by the Research Program of Nantong University. The authors greatly thank Dr. Yuan-yuan Zhu and his team of Biomics for scientific advice and technical assistance.
References
- 1.Hay ED. An overview of epithelio-mesenchymal transformation. Acta Anatomica. 1995;154(1):8–20. doi: 10.1159/000147748. [DOI] [PubMed] [Google Scholar]
- 2.Savagner P. Leaving the neighborhood: molecular mechanisms involved during epithelial-mesenchymal transition. BioEssays. 2001;23(10):912–923. doi: 10.1002/bies.1132. [DOI] [PubMed] [Google Scholar]
- 3.Thiery JP. Epithelial-mesenchymal transitions in cancer onset and progression. Bulletin de l’Academie Nationale de Medecine. 2009;193(9):1969–1979. [PubMed] [Google Scholar]
- 4.Sun D, Baur S, Hay ED. Epithelial-mesenchymal transformation is the mechanism for fusion of the craniofacial primordia involved in morphogenesis of the chicken lip. Developmental Biology. 2000;228(2):337–349. doi: 10.1006/dbio.2000.9946. [DOI] [PubMed] [Google Scholar]
- 5.Saika S, Okada Y, Miyamoto T, Ohnishi Y, Ooshima A, McAvoy JW. Smad translocation and growth suppression in lens epithelial cells by endogenous TGFβ2 during wound repair. Experimental Eye Research. 2001;72(6):679–686. doi: 10.1006/exer.2001.1002. [DOI] [PubMed] [Google Scholar]
- 6.Saika S, Miyamoto T, Tanaka S, et al. Response of lens epithelial cells to injury: role of lumican in epithelial-mesenchymal transition. Investigative Ophthalmology and Visual Science. 2003;44(5):2094–2102. doi: 10.1167/iovs.02-1059. [DOI] [PubMed] [Google Scholar]
- 7.Saika S, Kono-Saika S, Ohnishi Y, et al. Smad3 signaling is required for epithelial-mesenchymal transition of lens epithelium after injury. American Journal of Pathology. 2004;164(2):651–663. doi: 10.1016/S0002-9440(10)63153-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.de Iongh RU, Wederell E, Lovicu FJ, McAvoy JW. Transforming growth factor-β-induced epithelial-mesenchymal transition in the lens: a model for cataract formation. Cells Tissues Organs. 2005;179(1-2):43–55. doi: 10.1159/000084508. [DOI] [PubMed] [Google Scholar]
- 9.Vaughan MB, Howard EW, Tomasek JJ. Transforming growth factor-β1 promotes the morphological and functional differentiation of the myofibroblast. Experimental Cell Research. 2000;257(1):180–189. doi: 10.1006/excr.2000.4869. [DOI] [PubMed] [Google Scholar]
- 10.Serini G, Bochaton-Piallat M, Ropraz P, et al. The fibronectin domain ED-A is crucial for myofibroblastic phenotype induction by transforming growth factor-β1. Journal of Cell Biology. 1998;142(3):873–881. doi: 10.1083/jcb.142.3.873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vernon A, LaBonne C. Tumor metastasis: a new twist on epithelial-mesenchymal transitions. Current Biology. 2004;14(17):R719–R721. doi: 10.1016/j.cub.2004.08.048. [DOI] [PubMed] [Google Scholar]
- 12.Massague J. TGF-beta signal transduction. Annual Review of Biochemistry. 1998;67:753–791. doi: 10.1146/annurev.biochem.67.1.753. [DOI] [PubMed] [Google Scholar]
- 13.Zimmerman CM, Padgett RW. Transforming growth factor β signaling mediators and modulators. Gene. 2000;249(1-2):17–30. doi: 10.1016/s0378-1119(00)00162-1. [DOI] [PubMed] [Google Scholar]
- 14.Miyazono K, Ten Dijke P, Heldin C. TGF-β signaling by Smad proteins. Advances in Immunology. 2000;75:115–157. doi: 10.1016/s0065-2776(00)75003-6. [DOI] [PubMed] [Google Scholar]
- 15.Moustakas A, Pardali K, Gaal A, Heldin C. Mechanisms of TGF-β signaling in regulation of cell growth and differentiation. Immunology Letters. 2002;82(1-2):85–91. doi: 10.1016/s0165-2478(02)00023-8. [DOI] [PubMed] [Google Scholar]
- 16.Ten Dijke P, Goumans MJ, Itoh F, Itoh S. Regulation of cell proliferation by Smad proteins. Journal of Cellular Physiology. 2002;191(1):1–16. doi: 10.1002/jcp.10066. [DOI] [PubMed] [Google Scholar]
- 17.Roberts AB, Sporn MB. Differential expression of the TGF-β isoforms in embryogenesis suggests specific roles in developing and adult tissues. Molecular Reproduction and Development. 1992;32(2):91–98. doi: 10.1002/mrd.1080320203. [DOI] [PubMed] [Google Scholar]
- 18.Gordon-Thomson C, de Iongh RU, Hales AM, Chamberlain CG, McAvoy JW. Differential cataractogenic potency of tgf-β1, β2, and -β3 and their expression in the postnatal rat eye. Investigative Ophthalmology and Visual Science. 1998;39(8):1399–1409. [PubMed] [Google Scholar]
- 19.Ashish T, Jonathan CKT, Ajay S, Rangan G, Rajiv RM. Role of transforming growth factor beta in corneal function, biology and pathology. Current Molecular Medicine. 2010;10(6):565–578. doi: 10.2174/1566524011009060565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ohta K, Yamagami S, Taylor AW, Streilein JW. IL-6 antagonizes TGF-β and abolishes immune privilege in eyes with endotoxin-induced uveitis. Investigative Ophthalmology and Visual Science. 2000;41(9):2591–2599. [PubMed] [Google Scholar]
- 21.Jampel HD, Roche N, Stark WJ, Roberts AB. Transforming growth factor-β in human aqueous humor. Current Eye Research. 1990;9(10):963–969. doi: 10.3109/02713689009069932. [DOI] [PubMed] [Google Scholar]
- 22.Cousins SW, McCabe MM, Danielpour D, Streilein JW. Identification of transforming growth factor-beta as an immunosuppressive factor in aqueous humor. Investigative Ophthalmology and Visual Science. 1991;32(8):2201–2211. [PubMed] [Google Scholar]
- 23.Kita T, Hata Y, Arita R, et al. Role of TGF-β in proliferative vitreoretinal diseases and ROCK as a therapeutic target. Proceedings of the National Academy of Sciences of the United States of America. 2008;105(45):17504–17509. doi: 10.1073/pnas.0804054105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kokudo T, Suzuki Y, Yoshimatsu Y, Yamazaki T, Watabe T, Miyazono K. Snail is required for TGFβ-induced endothelial-mesenchymal transition of embryonic stem cell-derived endothelial cells. Journal of Cell Science. 2008;121(20):3317–3324. doi: 10.1242/jcs.028282. [DOI] [PubMed] [Google Scholar]
- 25.Côme C, Arnoux V, Bibeau F, Savagner P. Roles of the transcription factors Snail and slug during mammary morphogenesis and breast carcinoma progression. Journal of Mammary Gland Biology and Neoplasia. 2004;9(2):183–193. doi: 10.1023/B:JOMG.0000037161.91969.de. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rosivatz E, Becker I, Specht K, et al. Differential expression of the epithelial-mesenchymal transition regulators Snail, SIP1, and twist in gastric cancer. American Journal of Pathology. 2002;161(5):1881–1891. doi: 10.1016/S0002-9440(10)64464-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Batlle E, Sancho E, Franci C, et al. The transcription factor Snail is a repressor of E-cadherin gene expression in epithelial tumour cells. Nature Cell Biology. 2000;2(2):84–89. doi: 10.1038/35000034. [DOI] [PubMed] [Google Scholar]
- 28.Pálmer HG, Larriba MJ, García JM, et al. The transcription factor Snail represses vitamin D receptor expression and responsiveness in human colon cancer. Nature Medicine. 2004;10(9):917–919. doi: 10.1038/nm1095. [DOI] [PubMed] [Google Scholar]
- 29.Murray SA, Gridley T. Snail1 gene function during early embryo patterning in mice. Cell Cycle. 2006;5(22):2566–2570. doi: 10.4161/cc.5.22.3502. [DOI] [PubMed] [Google Scholar]
- 30.Paznekas WA, Okajima K, Schertzer M, Wood S, Jabs EW. Genomic organization, expression, and chromosome location of the human Snail gene (SNAI1) and a related processed pseudogene (SNAI1P) Genomics. 1999;62(1):42–49. doi: 10.1006/geno.1999.6010. [DOI] [PubMed] [Google Scholar]
- 31.Aomatsu K, Arao T, Sugioka K, et al. TGF-β induces sustained upregulation of SNAI1 and SNAI2 through smad and non-smad pathways in a human corneal epithelial cell line. Investigative Ophthalmology and Visual Science. 2011;52(5):2437–2443. doi: 10.1167/iovs.10-5635. [DOI] [PubMed] [Google Scholar]
- 32.Shirai K, Saika S, Tanaka T, et al. A new model of anterior subcapsular cataract: involvement of TGFβ/Smad signaling. Molecular Vision. 2006;12:681–691. [PubMed] [Google Scholar]
- 33.Abu El-Asrar AM, Missotten L, Geboes K. Expression of myofibroblast activation molecules in proliferative vitreoretinopathy epiretinal membranes. Acta Ophthalmologica. 2011;89(2):e115–e121. doi: 10.1111/j.1755-3768.2010.01916.x. [DOI] [PubMed] [Google Scholar]
- 34.Cano A, Pérez-Moreno MA, Rodrigo I, et al. The transcription factor Snail controls epithelial-mesenchymal transitions by repressing E-cadherin expression. Nature Cell Biology. 2000;2(2):76–83. doi: 10.1038/35000025. [DOI] [PubMed] [Google Scholar]
- 35.Batlle E, Sancho E, Franci C, et al. The transcription factor Snail is a repressor of E-cadherin gene expression in epithelial tumour cells. Nature Cell Biology. 2000;2(2):84–89. doi: 10.1038/35000034. [DOI] [PubMed] [Google Scholar]
- 36.Cho HJ, Baek KE, Saika S, Jeong M, Yoo J. Snail is required for transforming growth factor-β-induced epithelial-mesenchymal transition by activating PI3 kinase/Akt signal pathway. Biochemical and Biophysical Research Communications. 2007;353(2):337–343. doi: 10.1016/j.bbrc.2006.12.035. [DOI] [PubMed] [Google Scholar]
- 37.Elbashir SM, Lendeckel W, Tuschl T. RNA interference is mediated by 21- and 22-nucleotide RNAs. Genes and Development. 2001;15(2):188–200. doi: 10.1101/gad.862301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sax CM, Farrell FX, Zehner ZE, Piatigorsky J. Regulation of vimentin gene expression in the ocular lens. Developmental Biology. 1990;139(1):56–64. doi: 10.1016/0012-1606(90)90278-q. [DOI] [PubMed] [Google Scholar]
- 39.Clark DS. Posterior capsule opacification. Current Opinion in Ophthalmology. 2000;11(1):56–64. doi: 10.1097/00055735-200002000-00009. [DOI] [PubMed] [Google Scholar]
- 40.Schaumberg DA, Dana MR, Christen WG, Glynn RJ. A systematic overview of the incidence of posterior capsule opacification. Ophthalmology. 1998;105(7):1213–1221. doi: 10.1016/S0161-6420(98)97023-3. [DOI] [PubMed] [Google Scholar]
- 41.McDonnell PJ, Zarbin MA, Green WR. Posterior capsule opacification in pseudophakic eyes. Ophthalmology. 1983;90(12):1548–1553. doi: 10.1016/s0161-6420(83)34350-5. [DOI] [PubMed] [Google Scholar]
- 42.Dewey S. Posterior capsule opacification. Current Opinion in Ophthalmology. 2006;17(1):45–53. doi: 10.1097/01.icu.0000193074.24746.e6. [DOI] [PubMed] [Google Scholar]
- 43.Cobo LM, Ohsawa E, Chandler D. Pathogenesis of capsular opacification after extracapsular cataract extraction. An animal model. Ophthalmology. 1984;91(7):857–863. doi: 10.1016/s0161-6420(84)34225-7. [DOI] [PubMed] [Google Scholar]
- 44.Wormstone IM. Posterior capsule opacification: a cell biological perspective. Experimental Eye Research. 2002;74(3):337–347. doi: 10.1006/exer.2001.1153. [DOI] [PubMed] [Google Scholar]
- 45.Frezzotti R, Caporossi A, Mastrangelo D, et al. Pathogenesis of posterior capsular opacification. Part II: histopathological and in vitro culture findings. Journal of Cataract and Refractive Surgery. 1990;16(3):353–360. doi: 10.1016/s0886-3350(13)80708-0. [DOI] [PubMed] [Google Scholar]
- 46.Kappelhof JP, Vrensen GF. The pathology of after-cataract. A minireview. Acta ophthalmologica. 1992;(supplement 205):13–24. doi: 10.1111/j.1755-3768.1992.tb02176.x. [DOI] [PubMed] [Google Scholar]
- 47.Awasthi N, Wagner BJ. Suppression of human lens epithelial cell proliferation by proteasome inhibition, a potential defense against posterior capsular opacification. Investigative Ophthalmology and Visual Science. 2006;47(10):4482–4489. doi: 10.1167/iovs.06-0139. [DOI] [PubMed] [Google Scholar]
- 48.Wormstone IM, Liu CSC, Rakic J, Marcantonio JM, Vrensen GFJM, Duncan G. Human lens epithelial cell proliferation in a protein-free medium. Investigative Ophthalmology and Visual Science. 1997;38(2):396–404. [PubMed] [Google Scholar]
- 49.Walker JL, Wolff IM, Zhang L, Menko AS. Activation of Src kinases signals induction of posterior capsule opacification. Investigative Ophthalmology and Visual Science. 2007;48(5):2214–2223. doi: 10.1167/iovs.06-1059. [DOI] [PubMed] [Google Scholar]
- 50.Javelaud D, Mauviel A. Crosstalk mechanisms between the mitogen-activated protein kinase pathways and Smad signaling downstream of TGF-β: implications for carcinogenesis. Oncogene. 2005;24(37):5742–5750. doi: 10.1038/sj.onc.1208928. [DOI] [PubMed] [Google Scholar]
- 51.Nieto MA. The Snail superfamily of zinc-finger transcription factors. Nature Reviews Molecular Cell Biology. 2002;3(3):155–166. doi: 10.1038/nrm757. [DOI] [PubMed] [Google Scholar]
- 52.Kalluri R, Neilson EG. Epithelial-mesenchymal transition and its implications for fibrosis. Journal of Clinical Investigation. 2003;112(12):1776–1784. doi: 10.1172/JCI20530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Huber MA, Azoitei N, Baumann B, et al. NF-κB is essential for epithelial-mesenchymal transition and metastasis in a model of breast cancer progression. Journal of Clinical Investigation. 2004;114(4):569–581. doi: 10.1172/JCI21358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Lee JM, Dedhar S, Kalluri R, Thompson EW. The epithelial-mesenchymal transition: new insights in signaling, development, and disease. Journal of Cell Biology. 2006;172(7):973–981. doi: 10.1083/jcb.200601018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Nieto MA. Epithelial-Mesenchymal Transitions in development and disease: old views and new perspectives. International Journal of Developmental Biology. 2009;53(8–10):1541–1547. doi: 10.1387/ijdb.072410mn. [DOI] [PubMed] [Google Scholar]
- 56.Barrallo GA, Nieto MA. The Snail genes as inducers of cell movement and survival: implications in development and cancer. Development. 2005;132(14):3151–3161. doi: 10.1242/dev.01907. [DOI] [PubMed] [Google Scholar]
- 57.De Craene B, Van Roy F, Berx G. Unraveling signalling cascades for the Snail family of transcription factors. Cellular Signalling. 2005;17(5):535–547. doi: 10.1016/j.cellsig.2004.10.011. [DOI] [PubMed] [Google Scholar]
- 58.Choi J, Sun YP, Joo C. Transforming growth factor-β1 represses E-cadherin production via Slug expression in lens epithelial cells. Investigative Ophthalmology and Visual Science. 2007;48(6):2708–2718. doi: 10.1167/iovs.06-0639. [DOI] [PubMed] [Google Scholar]
- 59.McManus MT, Sharp PA. Gene silencing in mammals by small interfering RNAs. Nature Reviews Genetics. 2002;3(10):737–747. doi: 10.1038/nrg908. [DOI] [PubMed] [Google Scholar]


