Abstract
Continuous deployment of antifungals in treating infections caused by dimorphic opportunistic pathogen Candida albicans has led to the emergence of drug resistance resulting in cross-resistance to many unrelated drugs, a phenomenon termed multidrug resistance (MDR). Despite the current understanding of major factors which contribute to MDR mechanisms, there are many lines of evidence suggesting that it is a complex interplay of multiple factors which may be contributed by still unknown mechanisms. Coincidentally with the increased usage of antifungal drugs, the number of reports for antifungal drug resistance has also increased which further highlights the need for understanding novel molecular mechanisms which can be explored to combat MDR, namely, ROS, iron, hypoxia, lipids, morphogenesis, and transcriptional and signaling networks. Considering the worrying evolution of MDR and significance of C. albicans being the most prevalent human fungal pathogen, this review summarizes these new regulatory mechanisms which could be exploited to prevent MDR development in C. albicans as established from recent studies.
1. Introduction
In the last decades, the incidence of fungal infections has increased dramatically due to the rise in the number of immunocompromised patients. The most prevalent fungal pathogen of humans is Candida albicans which ranks as the fourth most common cause of hospital acquired infectious disease and is the primary cause of systemic candidiasis, with mortality rates approaching 50% [1]. The dimorphic opportunistic pathogen, C. albicans, is normally a commensal organism in humans, but when the host is unable to mount an adequate immune response, as in AIDS, organ transplant, diabetes, or in cancer patients, it results in mucosal, cutaneous, or invasive mycoses [2, 3]. Prolonged usage of antifungals in treating infections caused by C. albicans has led to the emergence of azole resistance. This acquired azole resistance in clinical isolates of C. albicans mostly results in cross-resistance to many unrelated drugs, a phenomenon termed multidrug resistance (MDR) [4–6]. MDR is a serious complication during treatment of opportunistic fungal infections which poses grave concern given the limited number of clinically useful antifungal drugs available [7, 8]. Fungal species have evolved a multitude of mechanisms to survive exposure to antifungal drugs and some of them include an overexpression or mutations in ERG11, encoding the target enzyme of azoles lanosterol 14α-demethylase [4, 5, 9, 10], an over expression of the drug efflux pumps encoding genes such as CaCDR1 and CaCDR2 belonging to the ABC (ATP-binding cassette) [11–13] and CaMDR1 belonging to the MFS (major facilitator super family) transporters [14–16].
Although MDR is a complex manifestation of factors which are reasonably documented, there are reports to suggest that it may involve many unknown mechanisms which are yet to be elucidated. In the recent years, emerging evidence has demonstrated that there do exist such novel mechanisms which can be helpful in controlling MDR efficiently. Improved knowledge of such molecular mechanisms controlling MDR in pathogenic fungi should facilitate the development of novel therapies to combat these intransigent infections. This review further defines the focus on the exacerbated need of understanding such mechanisms (Figure 1) and attempts to highlight research areas that need to be investigated in greater detail.
Figure 1.
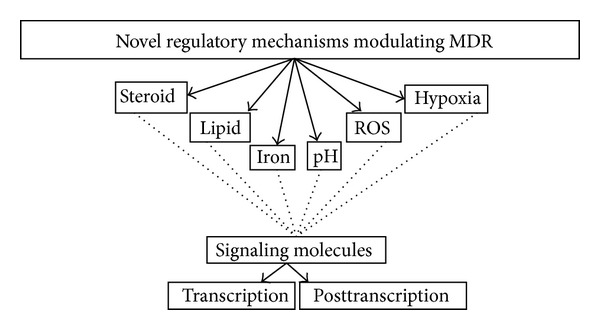
Novel regulatory mechanisms modulating MDR in Candida albicans.
2. ROS
In eukaryotic cells, mitochondria are common organelles that represent an important source of reactive oxygen species (ROS). ROS are not just a byproduct of mitochondrial disintegration but also a key regulator for yeast apoptosis [17]. As mediators in signal transduction pathways, ROS participate in early and late steps of the regulation of apoptosis. ROS produced by granulocytes or monocytes are known to exert activity against fungi [18]. Furthermore, C. albicans possesses a ROS scavenger, superoxide dismutase suggesting that fungi may require a cytoprotective mechanism against not only exogenous ROS but also endogenous ROS [19]. Kobayashi [20] demonstrated the role of ROS in azoles mediated drug sensitivity in C. albicans, thereby establishing a strong correlation between ROS and MDR. They showed that ROS production is directly involved in the cytostatic action of miconazole. In this study complete inhibition of miconazole-induced ROS production resulted in the restoration of 50 to 70% of cell viability, suggesting that ROS production is an important event, in addition to drug-induced inhibition of ergosterol synthesis. Similarly Wu [21] deciphered the antifungal action of plagiochin E (PLE) through mitochondrial-dysfunction-induced ROS accumulation in C. albicans. Xu [22] showed that endogenous ROS augmentation contributes to the synergistic action of fluconazole (FLC) and berberine against FLC-resistant C. albicans. Rosa [23] explored a mechanistic link between the drug sensitivity, gene expression, and pathogenesis phenotypes of C. albicans. They conclude that histone acetyltransferase, Rtt109, is particularly important for fungal pathogenicity, suggesting a unique target for therapeutic antifungal compounds. Recently, the fungicidal activity of Gemini-pyridinium salts and shikonin was found to be mediated through ROS generation only [24, 25]. Thus the fact that ROS production could either form the basis of antifungal action of the compound mentioned above or act in synergism to enhance the cytotoxicity of drugs has become a potential therapeutic strategy nowadays. Therefore, ROS signaling pathways need to be further elucidated in fungal pathogens to enhance the potency of such target.
3. Iron
Pathogens including C. albicans colonize various niches which are iron-limited, and iron, being an indispensable micronutrient, is required both by the host and by the microbial community residing within the host [26–31]. Availability of iron in host cells is tightly regulated, since iron is a transition metal and its ability to donate and accept electrons can indulge in the formation of toxic free radicals, and hence iron plays a key role in providing natural resistance to infections in humans [32]. Interestingly, studies suggest that there could be a correlation between intracellular iron and MDR phenomenon. For instance, role of iron in recurrent vulvovaginal candidiasis (RVVC) showed that this elements is not only important for pathogenic yeast, but also for normal function of host immunity [30]. Kuipers [33–35] showed that lactoferrin, an iron binding glycoprotein, is synergistic with antifungals against different Candida species. However, whether iron affects drug susceptibility of Candida cells was not demonstrated experimentally until Prasad [36] reported for the first time that availability of iron could have an impact on defense mechanisms of Candida against antifungal drugs. Interestingly, it was observed that iron deprivation enhanced drug susceptibility of Candida cells resulting in an increase in membrane fluidity, which in turn leads to enhanced passive diffusion of drugs. A link between changes in membrane fluidity and lowered ergosterol levels was established in iron deprived Candida cells probably due to downregulation of ERG11. However, the intricate relationship between cellular iron, calcineurin signaling, membrane lipid homeostasis, and drug susceptibility of Candida cells was first established by Hameed [37]. Even antifungal action of malachite green is mediated via depletion of labile iron pools as one of its mechanisms [38]. The synergism of lactoferrin with fluconazole has been reported to enhance the antifungal activity of fluconazole against Candida spp. [39]. Cap2/Hap43 is essential for C. albicans growth under iron-deprivation conditions and for virulence in mouse [40]. Moreover, the usage of fungicidal monoclonal antibodies seems to interfere with iron acquisition in C. albicans [41]. Many transcription factors governed by iron homeostasis namely, Sfu1, Hap43, Sef1, Cap2, and Aft2, and regulators of iron uptake genes have already been identified; however, sufficient knowledge of signaling pathways is still lacking, and therefore, understanding the role of these transcription factors which are governed by iron availability will better help in studying the relation between iron and MDR. Kaba [42] demonstrated a link between mitogen activated protein kinase HOG1 and iron availability. Dap1 is a heme binding protein that mediates a functional link between iron homeostasis and azole resistance in C. glabrata [43]. Since dap1 mutants show enhanced azole susceptibility and deceased ergosterol production, hence, its role in C. albicans could also be a possible target. Gene expression profiling of BCR1 transcription factor, known to have a role in biofilm formation, has reported that five out of eight genes have been implicated in iron homeostasis [44]. The influence of cellular iron on drug susceptibilities of Candida suggests iron to be yet another novel determinant of MDR which merits a closer look.
4. Hypoxia
Organisms often encounter sites within the host during infection which have inadequate vascularization and irregular blood flow and thus present hypoxic areas. The transcription factor complex hypoxia inducible factor 1 (HIF-1) controls the expression of most genes involved in adaptation to hypoxic conditions and therefore represents an important regulator of MDR development. Hypoxia is known to cause resistance to chemotherapy by the induction of human MDR1 in growing tumors, via activation of hypoxia inducible factor-1 (HIF1) [45–48]. Although till date no homolog of MDR1 has yet been identified in C. albicans, as this organism encounters sites during infection which are hypoxic, it is indeed inevitable to understand the hypoxic responses of this pathogen. The relevance to explore hypoxic responses in C. albicans is depicted from a wide range of studies. For instance, transcriptome response of C. albicans under hypoxia reveals metabolic adaptation to scarce amount of oxygen availability [49]. Following hypoxic growth in vagina-simulative conditions, part of C. albicans proteome that is covalently linked to the cell wall has been determined [50]. Efg1 plays a major role in hypoxic responses of C. albicans as suggested by the fact that efg1 mutants, though inhibit hyphae formation during normoxia, are able to express filaments under microaerobic conditions if grown on or within (embedded) agar at temperatures from 25°C to 35°C [51, 52]. Another study revealed that efg1 mutants are hyperfilamentous as a response to low oxygen [49]. Role of Efg1p in biofilm formation, which is one of the major threat towards antifungal chemotherapy, under hypoxia has been established where Efg1p induces all the major classes of genes required for biofilm formation [53]. Moreover, adaptation to hypoxia forms an integral component of biofilm formation in C. albicans [54]. Carvalho [55] demonstrated that aspartyl protease activity could be modulated by oxygen availability in C. albicans. Another study revealed that regulation of gene expression in response to hypoxia in C. albicans could be signaled via lowered sterol levels and induction of filamentation under hypoxic conditions requires the Ras1- and Cdc35-dependent pathway [56]. Recently, it has been demonstrated that kinase Sch9 integrates both hypoxia and CO2 sensing to inhibit yeast to hyphal transition in C. albicans [57]. Responses to hypoxic conditions in pathogenic fungi including C. albicans have been already extensively reviewed [58, 59]. Certainly further research on the effect of hypoxia on infection by fungal pathogens should be focused. Antifungal compounds that act specifically on adaptive mechanisms of pathogens required for hypoxic adaptation could be promising alternatives to existing strategies.
5. Steroids
Steroids have been known to affect cell growth, germination, morphogenesis, and virulence in fungi [60–62]. Some lines of evidence for the presence of steroid binding proteins in yeast, such as estradiol binding protein [63], corticosteroid binding protein [64], and progesterone binding protein [65], have already been documented but their exact function in the steroid response is not known. Many studies have shown that steroids can induce a pleiotropic drug resistance (PDR) state in both pathogenic C. albicans and nonpathogenic Saccharomyces cerevisiae. Oestrogen regulation and its mechanism have been extensively studied [66, 67] and it is reported that the CDR1 promoter region does contain steroid responsive elements (SRE) and a drug response element (DRE). Oestrogen mediated binding of the transcription factor TAC1 to the DRE and induction of CDR1 expression have been studied [68]. Banerjee [69] for the first time demonstrated that yeast cells, which do not possess steroid receptor cascade, probably perceive steroids as a cellular stress. They examined the genome-wide changes in the gene expression profile following exposure to progesterone. Interestingly, an inverted CCAAT box which in combination with other conserved sequences is attributed towards human MDR1 responsiveness to cellular stresses is also present in steroid responsive region consensus sequences [70–72]. Banerjee [73] deciphered a more accurate evolutionary significance of the steroid response in yeasts by exposing S. cerevisiae and C. albicans cells to several doses of progesterone for different time periods. The study revealed the conserved and divergent features of PDR network in yeasts. Recently, antifungal potentials of steroidal quinolones and chalcones, respectively, have been reported and results seems to be quite promising [74, 75]. These studies showed that the steroid response in the absence of any known signaling cascade is a global phenomenon in yeast cells. Yet, in view of the importance of steroids in the physiology of both pathogenic and nonpathogenic yeasts, it would be interesting to examine the steroid-dependent regulatory cascade in these organisms and thereby decipher a new mechanism to combat MDR.
6. Morphogenesis
In response to various environmental stimuli, C. albicans is able to switch from the unicellular yeast form into either of the two distinct filamentous forms, that is, cells with pseudohyphae or true hyphae. This ability to switch is considered as an important virulence trait which is also coregulated with other virulence factors that are associated with cellular morphology [76, 77]. The morphological form of C. albicans is directly related to environmental conditions and these cues trigger separate signal transduction pathways which regulate common targets required to initiate hyphal growth [51, 76, 77]. The transcription factor Efg1p is a well known regulator of morphogenesis of C. albicans since it induces the yeast-to-hyphal transition and also regulates phenotypic switching and chlamydospore formation of this pathogen [52].
Lo [78] established that Efg1p is involved in drug resistance by regulating the expression of ERG3 gene of ergosterol biosynthetic pathway. Ergosterol is an important target for many antifungal drugs particularly on the plasma membrane. Considering the significance of Efg1p regulator in morphogenesis, Prasad [79] have evaluated if disruption in morphogenic signaling cascade would also affect MDR status of C. albicans cells. The study showed that null mutant of the morphogenic regulator EFG1 displayed enhanced drug sensitivity of C. albicans cells by a mechanism that is not dependent on the drug efflux pumps. This study establishes a convergence of EFG1 and MDR pathways and thus proposes an additional new role for this important morphogenic regulator of C. albicans. The role of a newly discovered regulator of hyphae formation, Rca1, in drug susceptibility of C. albicans was established recently [80]. Certainly more extensive analyses are required to elucidate the commonality between EFG1 and MDR signaling cascades to find newer targets for antifungal chemotherapy.
7. pH
The ability of microorganisms to sense and adapt to changes in the environment is essential for their survival. One environmental factor that microorganisms must respond to is extracellular pH. Environmental pH has dramatic effects on the cell, particularly at the plasma membrane, including effects on protein activity, maintenance of the proton gradient, and nutrient availability. Furthermore, in the opportunistic fungal pathogen C. albicans, environmental pH serves as one potent signal for morphological differentiation, although serum N-acetyl glucosamine and agar-embedding represent other several potent signals [2, 3]. Most chemotherapeutic drugs in use today are hydrophobic small, molecules that are also typically either weakly basic, weakly acidic, or charged. Thus, changes in the electrochemical parameters of microorganism's cell membranes have important effects on their transmembranous diffusion and cellular retention [81]. Changes in these parameters can also modulate the function of immunological agents and affect the signal transduction. The RIM101 pathway that has been identified in C. albicans governs pH responses, dimorphism, and pathogenesis [82]. The RIM101 pathway and pH responses, in general, play an intimate role in pathogenesis beyond simply allowing the organism to grow [83]. Apart from the major RIM101 pathway, some other pathways have also been identified in C. albicans, which act in parallel. For instance a novel RIM101 independent pH pathway was proposed which was mediated by PHR2 [83]. Phr1p and Phr2p are differentially expressed cell wall proteins in response to environmental pH. Phr1p is expressed under alkaline conditions and Phr2p is expressed under acidic conditions, and importantly both are required for the pathogenesis of the organism for causing systemic candidiasis (pH 7.4) and vaginal candidiasis (pH 4) [84]. Another signaling pathway that was identified for adaptation to neutral/alkaline pH is calcineurin signaling [85]. Calcineurin is a highly conserved calcium-dependent serine/threonine-specific protein phosphatase that mediates various stress responses inside the cell including conferring tolerance to alkaline pH in C. albicans [86]. Furthermore, Hameed [37] provided the first evidence to establish relationship between cellular iron, calcineurin signaling, membrane lipid homeostasis, and drug susceptibility of Candida cells. They showed that iron deficiency leads to downregulation of calcineurin signaling pathway leading to abrogated sensitivity at alkaline pH, salinity, and membrane stress. In general the ability of pathogenic fungi to adapt to host pH is critical for survival and disease progression. This highlights the importance of continuing studies of these fundamental pH response pathways in pathogenic fungi in order to understand how these pathogens are adapted to the mammalian host and potentially identify new approaches for preventing or treating infections.
8. Lipid
Recent lines of evidence have provided a comprehensive amount of data regarding the role of lipids and many other lipid derivatives in establishment of various infectious diseases. MDR in yeast is closely linked to the status of membrane lipids. It has been already established that the associated changes in membrane lipid composition (phospholipid and ergosterol), its order (fluidity), and asymmetry could be important determinants in the drug susceptibilities of Candida cells [87]. Similarly, changes in membrane lipid composition between sensitive and resistant could also influence the action of antifungal drugs like azoles and thereby can form one of the factors responsible for drug resistance [12]. Most strikingly it has been observed that clinical as well as adapted azole-resistant isolates of C. albicans exhibit altered membrane phospholipid and sterol compositions [88]. Mukhopadhyay [89] demonstrated that there is an interaction between membrane ergosterol and sphingolipids, and a reduction in the content of either of these two components results in a disruption of this interaction, which has deleterious effects on the drug susceptibilities of C. albicans cells. Thus the fact that lipid could also play an important role in drug susceptibilities is becoming apparent from a wide range of recent studies. The importance of lipid signaling molecules in the development and pathogenicity of clinically important fungi has been highlighted [90]. In C. albicans, exposure to the oxylipin farnesol causes the regulation of specific genes involved in hyphal development, drug resistance, and iron acquisition. Farnesol increases resistance to oxidative stress in C. albicans. Through technologies such as lipidomics, a wider vision has been obtained to know about the diversity of lipid molecule which is not only limited to cellular functions but also to pathogenesis of diseases. Nowadays, lipidome-wide quantification of individual molecular lipid species (molecules with defined chemical structure) by absolute quantification provided a new approach to relate lipidomics and functional genomics studies [91]. Hameed [37] demonstrated the role of lipid homeostasis in iron mediated drug susceptibility of C. albicans where iron deprivation leads to enhanced drug susceptibility due to lowered ergosterol levels and increases membrane fluidity. Similarly the antifungal effect of curcumin has also been shown to be influenced by membrane lipid composition depicting marked changes in phosphoglyceride (PGL) species and also ergosterol depletion [92]. Recently, a high throughput lipid profiling has revealed some differences in lipid composition between azole sensitive and azole-resistant isolates highlighting fluctuations in phosphatidyl serine, mannosylinositolphosphorylceramides, and sterol esters levels indicating their compensatory role in maintaining lipid homeostasis among most of the resistant Candida isolates [93]. Cross-talk between mitochondrial lipid homeostasis, cell wall integrity, and azole tolerance has been revealed showing significant changes in several lipid classes, particularly in plasma membrane microdomain-specific lipids such as mannosylinositolphosphorylceramides and ergosterol, and in a mitochondrial-specific phosphoglyceride, phosphatidyl glycerol [94]. The overall cellular lipid homeostasis is a critical factor in the observed FLC resistance [95]. Although the transcriptome, proteome, and interactome of several eukaryotic model organisms have been described in detail, lipidomes still remain relatively uncharacterized and will also improve our understanding of the molecular architecture of membrane domains and cellular organelles. Hence, the lipid associated changes of pathogenic fungi induced in response to infection might help in better understanding of the role of lipids in pathogenesis of C. albicans and thereby development of better therapeutic strategies.
9. Signaling
A focal point among the fungal pathogens is that signaling molecules have a key role in mediating cellular stress responses. Signal transduction pathways are crucial mechanisms that allow cells to sense and respond to diverse environmental cues. Exploiting these stress responses through blocking of signaling pathways may provide the foundation for new combination therapies to enhance the efficacy of our limited resources of clinically useful antifungal drugs. Jain [96] have examined the relationship between azole susceptibility and the cyclic AMP (cAMP) protein kinase A (PKA) signaling pathway. Likewise, a key regulator of cell signaling in all eukaryotes, calcineurin, provides a perfect example of the role of signaling molecules in mediating crucial responses to antifungal drugs [97]. Alonso-Monge [98] describe that MAPK cascades control most of the virulence factors characterized in C. albicans: cell morphology, superficial antigen (cell wall biogenesis), and response to oxidative and nitrosative stresses. These cascades allow opportunistic pathogens to recognize changes in their environment and take advantage of an impaired immunological system to cause infection. Recent studies reveal that Hsp90, a component of a chaperone complex induced by heat stress, governs drug resistance in fungi [99, 100]. Bastidas [101] showed the significance of signaling cascades in pathogenic fungi. They not only depicted the well known calcineurin pathway and that Hsp90 is an important antifungal target but also described the TOR signaling pathway in context with antifungal drug resistance. Thakur [102] showed mechanistically similar regulation of MDR like in vertebrates by the PXR nuclear receptor, revealing an unexpected functional analogy of fungal and metazoan regulators of MDR. Robbins [103] establish a novel role of nutrient signaling in azole resistance. They revealed that compromising the function of Tor kinase, a global regulator of growth and metabolism, could be an efficient strategy to control drug resistance. Another key cellular stress response pathway having implications in basal tolerance to azoles is the protein kinase C (PKC) mediated cell wall integrity pathway (CWI). Lafayette [104] established a novel role of CWI and calcineurin signaling pathway in C. albicans, while de Dios [105] revealed the relevance of MAPK signaling cascades in fungal virulence. The emerging roles of GlcNAc as an activator and mediator of cellular signaling in C. albicans have also been reviewed [106]. Recently, even the significance of metabolic pathways as therapeutic targets and how their disruption can have both physiological and regulatory consequences have also been demonstrated [107]. The diterpene acid, phorbasin H, affects the activity of the cAMP-Efg1 pathway leading to an alteration of C. albicans morphology as was demonstrated recently [108]. Likewise antidimorphism activity of catechin was depicted by interfering with Cek1 phosphorylation and cAMP synthesis [109]. Kaba [42], demonstrated the involvement of MAPK HOG1 pathway with iron availability, and as already discussed in the previous sections, iron plays a vital role if one considers drug susceptibility in C. albicans. A new study showed for the first time that even lower eukaryote like C. albicans possesses a two-component response regulator protein [110]. Thus a deeper knowledge of the mechanism and regulation of various signaling cascades could help in the control of candidiasis as well as in the development of effective therapeutics against these severe infections.
10. Transcription Factors
Transcriptional regulation can be of significant importance in the development of antifungal resistance. It is important to understand the regulatory network controlling drug resistance in fungal pathogens. Different strategies have been adopted for isolation of regulators of multidrug transporters in C. albicans. One such approach is through the analysis of cis-acting elements in genes encoding multidrug efflux transporters. For instance the basal expression element (BEE) responsible for basal expression, the drug-responsive element (DRE) required for the response to drugs such as fluphenazine and estradiol, two steroid-responsive element (SRE) involved in the response to steroid hormones and the negative regulatory element (NRE), have been already deciphered [66, 67, 111]. One of the major breakthroughs was the discovery of TAC1 regulator whose deletion abrogates the CDR1/CDR2 expression in C. albicans clinical isolates resistant to azoles, thus demonstrating that TAC1 was a major mediator of azole resistance due to the upregulation of the ABC transporter [68]. Apart from it other potential regulators of CDR1 have also been reported which were identified through functional complementation in S. cerevisiae. For instance functional homologue of PDR1/PDR3 in S. cerevisiae, namely, fluconazole resistance 1 (FCR1), was reported in C. albicans. Deletion of FCR1 in C. albicans resulted in reduced susceptibility to FLC [112]. Similarly, NDT80 inactivation in C. albicans, a gene similar to the S. cerevisiae NDT80 gene, resulted in a decreased basal CDR1 expression and a decreased CDR1 inducibility in the presence of drugs [113]. Genome-wide transcription profiling was employed for the identification of an MDR1 regulator or of factors binding to the MDR1 promoter. By comparing the transcriptional profiles of three different C. albicans clinical isolates over expressing MDR1 with azole-susceptible parents, one of the commonly upregulated genes in the three isolates was orf19.7372 which was subsequently known as MRR1 [114]. Another study reported that regulator of efflux pump 1 (REP1) acts as a negative regulator of MDR1 which belongs to the transcription factor family including NDT80 [115]. Similarly, C. albicans gene (UPC2) with homology to both S. cerevisiae genes has been identified and characterized [116]. Deletion of UPC2 in C. albicans abrogates ERG11 upregulation in response to azole drugs. A recent study has demonstrated the role of the mediator complex in the transcriptional response of multidrug transporter genes in S. cerevisiae and C. glabrata [102]; however, in C. albicans, the binding to the transcriptional activator of drug resistance genes (TAC1, MRR1, and UPC2) still remains hypothetical.
Gain-of-function (GOF) mutations in alleles of the transcription factors (TAC1, MRR1, CgPDR1, and UPC2) from azole-resistant isolates cause constitutive high expression of their drug resistance gene targets and thus azole resistance when expressed in an azole-susceptible background [68, 114, 117–122]. Nowadays, genome-wide analysis is an important tool to yield regulons of selected transcription factors for the elucidation of transcriptional regulatory network of drug resistance. Moreover, systematic deletion of transcription factor genes could also be an alternative approach to reveal transcriptional circuits responsible for drug resistance. Genome-wide genetic screens for the identification of additional targets involved in drug resistance or participating in the response of fungal pathogens to drug exposure will likely result in elucidating regulatory mechanisms. Recently, it has been uncovered that a network comprises 800 target genes and a tightly knit transcriptional regulatory circuit indicating that many aspects of commensalism and pathogenicity are crosslinked [123]. Dhamgaye [124] deciphered through RNA sequencing some novel genes including the transcription factor CZF1 which are responsible for drug resistance.
Although the transcriptional regulation is considered to be the major step in MDR regulation, reports do exist nowadays revealing the significance of posttranscriptional regulation as well. Manoharlal [125] demonstrated that CDR1 mRNA half-life was increased in azole-resistant as compared to azole sensitive isolates. Moreover, Manoharlal [126] further dissected the molecular basis of the above observed increased mRNA stability where it was demonstrated that loss of heterozygosity at the PAP1 locus is linked to hyperadenylation and subsequent mRNA stability of CDR1 transcripts in azole resistant isolates. Recently, posttranscriptional regulation of transcription factor Sef1 has been demonstrated in controlling virulence of C. albicans [127]. Thus despite poor knowledge about the posttranscriptional regulation of MDR in C. albicans, certainly, this aspect needs to be further elucidated to gain deeper insights.
11. Concluding Remarks
Today, the rapidly evolving issue of MDR demands the obvious need for dissecting completely new regulatory mechanisms that could be targeted to control MDR. Novel mechanisms described earlier for pathogenic fungi clearly hold promise that can facilitate the development of better antifungal strategies to efficiently control the human fungal diseases. Elucidating these mechanisms may provide new foundations for antifungal chemotherapy and can present an exciting challenge for the future investigation.
Acknowledgment
The financial assistance in the form of Young Scientist Award (SR/FT/LS-12/2012) to Saif Hameed from Science and Engineering Research Board (SERB), New Delhi, is deeply acknowledged. The authors thank Professor S. M. Paul Khurana, Director, Amity Institute of Biotechnology for encouragement.
References
- 1.Pfaller MA, Diekema DJ. Epidemiology of invasive candidiasis: a persistent 873 public health problem. Clinical Microbiology Reviews. 2007;20(1):133–163. doi: 10.1128/CMR.00029-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Calderone RA. Candida and Candidiasis. Washington, DC, USA: American Society for Microbiology Press; 2002. [Google Scholar]
- 3.Odds FC. Candida and Candidosis: A Review and Bibliography. London, UK: 1988. [Google Scholar]
- 4.White TC, Marr KA, Bowden RA. Clinical, cellular, and molecular factors that contribute to antifungal drug resistance. Clinical Microbiology Reviews. 1998;11(2):382–402. doi: 10.1128/cmr.11.2.382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.White TC, Holleman S, Dy F, Mirels LF, Stevens DA. Resistance mechanisms in clinical isolates of Candida albicans . Antimicrobial Agents and Chemotherapy. 2002;46(6):1704–1713. doi: 10.1128/AAC.46.6.1704-1713.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Franz R, Kelly SL, Lamb DC, Kelly DE, Ruhnke M, Morschhäuser J. Multiple molecular mechanisms contribute to a stepwise development of fluconazole resistance in clinical Candida albicans strains. Antimicrobial Agents and Chemotherapy. 1998;42(12):3065–3072. doi: 10.1128/aac.42.12.3065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Anderson JB. Evolution of antifungal-drug resistance: mechanisms and pathogen fitness. Nature Reviews Microbiology. 2005;3(7):547–556. doi: 10.1038/nrmicro1179. [DOI] [PubMed] [Google Scholar]
- 8.Cowen LE, Steinbach WJ. Stress, drugs, and evolution: the role of cellular signaling in fungal drug resistance. Eukaryotic Cell. 2008;7(5):747–764. doi: 10.1128/EC.00041-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lamb DC, Kelly DE, Schunck W-H, et al. The mutation T315A in Candida albicans sterol 14α-demethylase causes reduced enzyme activity and fluconazole resistance through reduced affinity. Journal of Biological Chemistry. 1997;272(9):5682–5688. doi: 10.1074/jbc.272.9.5682. [DOI] [PubMed] [Google Scholar]
- 10.Prasad R, Gupta N, Gaur M. Molecular basis of antifungal resistance in pathogenic fungi. In: San-Blas G, Calderone RA, editors. Pathogenic Fungi—Host Interactions and Emerging Strategies For Control. Norfolk, UK: Caister Academic Press; 2004. pp. 357–414. [Google Scholar]
- 11.Albertson GD, Niimi M, Cannon RD, Jenkinson HF. Multiple efflux mechanisms are involved in Candida albicans fluconazole resistance. Antimicrobial Agents and Chemotherapy. 1996;40(12):2835–2841. doi: 10.1128/aac.40.12.2835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kohli A, Smriti S, Mukhopadhyay K, Rattan A, Prasad R. In vitro low-level resistance to azoles in Candida albicans is associated with changes in membrane lipid fluidity and asymmetry. Antimicrobial Agents and Chemotherapy. 2002;46(4):1046–1052. doi: 10.1128/AAC.46.4.1046-1052.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sanglard D, Ischer F, Monod M, Bille J. Cloning of Candida albicans genes conferring resistance to azole antifungal agents: characterization of CDR2, a new multidrug ABC transporter gene. Microbiology. 1997;143(2):405–416. doi: 10.1099/00221287-143-2-405. [DOI] [PubMed] [Google Scholar]
- 14.Ben-Yaacov R, Knoller S, Caldwell GA, Becker JM, Koltin Y. Candida albicans gene encoding resistance to benomyl and methotrexate is a multidrug resistance gene. Antimicrobial Agents and Chemotherapy. 1994;38(4):648–652. doi: 10.1128/aac.38.4.648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gupta V, Kohli A, Krishnamurthy S, et al. Identification of polymorphic mutant alleles of CaMDR1, a major facilitator of Candida albicans which confers multidrug resistance, and its in vitro transcriptional activation. Current Genetics. 1998;34(3):192–199. doi: 10.1007/s002940050385. [DOI] [PubMed] [Google Scholar]
- 16.Pao SS, Paulsen IT, Saier MH., Jr. Major facilitator superfamily. Microbiology and Molecular Biology Reviews. 1998;62:1–34. doi: 10.1128/mmbr.62.1.1-34.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Perrone GG, Tan S-X, Dawes IW. Reactive oxygen species and yeast apoptosis. Biochimica et Biophysica Acta. 2008;1783(7):1354–1368. doi: 10.1016/j.bbamcr.2008.01.023. [DOI] [PubMed] [Google Scholar]
- 18.Vázquez N, Walsh TJ, Friedman D, Chanock SJ, Lyman CA. Interleukin-15 augments superoxide production and microbicidal activity of human monocytes against Candida albicans . Infection and Immunity. 1998;66(1):145–150. doi: 10.1128/iai.66.1.145-150.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hwang C-S, Rhie G-E, Kim S-T, et al. Copper- and zinc-containing superoxide dismutase and its gene from Candida albicans . Biochimica et Biophysica Acta. 1999;1427(2):245–255. doi: 10.1016/s0304-4165(99)00020-3. [DOI] [PubMed] [Google Scholar]
- 20.Kobayashi D, Kondo K, Uehara N, et al. Endogenous reactive oxygen species is an important mediator of miconazole antifungal effect. Antimicrobial Agents and Chemotherapy. 2002;46(10):3113–3117. doi: 10.1128/AAC.46.10.3113-3117.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wu X-Z, Cheng A-X, Sun L-M, Sun S-J, Lou H-X. Plagiochin E, an antifungal bis(bibenzyl), exerts its antifungal activity through mitochondrial dysfunction-induced reactive oxygen species accumulation in Candida albicans . Biochimica et Biophysica Acta. 2009;1790(8):770–777. doi: 10.1016/j.bbagen.2009.05.002. [DOI] [PubMed] [Google Scholar]
- 22.Xu Y, Wang Y, Yan L, et al. Proteomic analysis reveals a synergistic mechanism of fluconazole and berberine against fluconazole-resistant Candida albicans: endogenous ROS augmentation. Journal of Proteome Research. 2009;8(11):5296–5304. doi: 10.1021/pr9005074. [DOI] [PubMed] [Google Scholar]
- 23.Da Rosa JL, Boyartchuk VL, Zhu LJ, Kaufman PD. Histone acetyltransferase Rtt109 is required for Candida albicans pathogenesis. Proceedings of the National Academy of Sciences of the United States of America. 2010;107(4):1594–1599. doi: 10.1073/pnas.0912427107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Shirai A, Ueta S, Maseda H, Kourai H, Omasa T. Action of reactive oxygen species in the antifungal mechanism of gemini-pyridinium salts against yeast. Biocontrol Science and Technology. 2012;17(2):77–82. doi: 10.4265/bio.17.77. [DOI] [PubMed] [Google Scholar]
- 25.Miao H, Zhao L, Li C, Shang Q, Lu H, Fu Z. Inhibitory effect of shikonin on Candida albicans growth. Biological & Pharmaceutical Bulletin. 2012;35(11):1956–1963. doi: 10.1248/bpb.b12-00338. [DOI] [PubMed] [Google Scholar]
- 26.Almeida RS, Wilson D, Hube B. Candida albicans iron acquisition within the host. FEMS Yeast Research. 2009;9(7):1000–1012. doi: 10.1111/j.1567-1364.2009.00570.x. [DOI] [PubMed] [Google Scholar]
- 27.Bullen JJ, Rogers HJ, Spalding PB, Ward CG. Natural resistance, iron and infection: a challenge for clinical medicine. Journal of Medical Microbiology. 2006;55(3):251–258. doi: 10.1099/jmm.0.46386-0. [DOI] [PubMed] [Google Scholar]
- 28.Fischbach MA, Lin H, Liu DR, Walsh CT. How pathogenic bacteria evade mammalian sabotage in the battle for iron. Nature Chemical Biology. 2006;2(3):132–138. doi: 10.1038/nchembio771. [DOI] [PubMed] [Google Scholar]
- 29.Nyilasi I, Papp T, Takó M, Nagy E, Vágvölgyi C. Iron gathering of opportunistic pathogenic fungi: a mini review. Acta Microbiologica et Immunologica Hungarica. 2005;52(2):185–197. doi: 10.1556/AMicr.52.2005.2.4. [DOI] [PubMed] [Google Scholar]
- 30.Spacek J, Jilek P, Buchta V, Forstl M, Hronek M, Holeckova M. The serum levels of calcium, magnesium, iron and zinc in patients with recurrent vulvovaginal candidosis during attack, remission and in healthy controls. Mycoses. 2005;48(6):391–395. doi: 10.1111/j.1439-0507.2005.01164.x. [DOI] [PubMed] [Google Scholar]
- 31.Weinberg ED. The role of iron in protozoan and fungal infectious diseases. Journal of Eukaryotic Microbiology. 1999;46(3):231–238. doi: 10.1111/j.1550-7408.1999.tb05119.x. [DOI] [PubMed] [Google Scholar]
- 32.Radisky D, Kaplan J. Regulation of transition metal transport across the yeast plasma membrane. Journal of Biological Chemistry. 1999;274(8):4481–4484. doi: 10.1074/jbc.274.8.4481. [DOI] [PubMed] [Google Scholar]
- 33.Kuipers ME, De Vries HG, Eikelboom MC, Meijer DKF, Swart PJ. Synergistic fungistatic effects of lactoferrin in combination with antifungal drugs against clinical Candida isolates. Antimicrobial Agents and Chemotherapy. 1999;43(11):2635–2641. doi: 10.1128/aac.43.11.2635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kuipers ME, Beljaars L, Van Beek N, et al. Conditions influencing the in vitro antifungal activity of lactoferrin combined with antimycotics against clinical isolates of Candida: impact on the development of buccal preparations of lactoferrin. APMIS. 2002;110(4):290–298. doi: 10.1034/j.1600-0463.2002.100403.x. [DOI] [PubMed] [Google Scholar]
- 35.Kuipers ME, Heegsma J, Bakker HI, et al. Design and fungicidal activity of mucoadhesive lactoferrin tablets for the treatment of oropharyngeal candidosis. Drug Delivery. 2002;9(1):31–38. doi: 10.1080/107175402753413154. [DOI] [PubMed] [Google Scholar]
- 36.Prasad T, Chandra A, Mukhopadhyay CK, Prasad R. Unexpected link between iron and drug resistance of Candida spp.: iron depletion enhances membrane fluidity and drug diffusion, leading to drug-susceptible cells. Antimicrobial Agents and Chemotherapy. 2006;50(11):3597–3606. doi: 10.1128/AAC.00653-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hameed S, Dhamgaye S, Singh A, Goswami SK, Prasad R. Calcineurin signaling and membrane lipid homeostasis regulates iron mediated multidrug resistance mechanisms in Candida albicans . PLoS ONE. 2011;6(4) doi: 10.1371/journal.pone.0018684.e18684 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Dhamgaye S, Devaux F, Manoharlal R, et al. In vitro effect of malachite green on Candida albicans involves multiple pathways and transcriptional regulators UPC2 and STP2. Antimicrobial Agents and Chemotherapy. 2012;56(1):495–506. doi: 10.1128/AAC.00574-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kobayashi T, Kakeya H, Miyazaki T, et al. Synergistic antifungal effect of lactoferrin with azole antifungals against Candida albicans and a proposal for a new treatment method for invasive candidiasis. Japanese Journal of Infectious Diseases. 2011;64(4):292–296. [PubMed] [Google Scholar]
- 40.Singh RP, Prasad HK, Sinha I, Agarwal N, Natarajan K. Cap2-HAP complex is a critical transcriptional regulator that has dual but contrasting roles in regulation of iron homeostasis in Candida albicans . Journal of Biological Chemistry. 2011;286(28):25154–25170. doi: 10.1074/jbc.M111.233569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Brena S, Cabezas-Olcoz J, Moragues MD, et al. Fungicidal monoclonal antibody C7 interferes with iron acquisition in Candida albicans . Antimicrobial Agents and Chemotherapy. 2011;55(7):3156–3163. doi: 10.1128/AAC.00892-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kaba HE, Nimtz M, Müller PP, Bilitewski U. Involvement of the mitogen activated protein kinase Hog1p in the response of Candida albicans to iron availability. BMC Microbiology. 2013;13, article 16 doi: 10.1186/1471-2180-13-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hosogaya N, Miyazaki T, Nagi M, Tanabe K, Minematsu A, Nagayoshi Y. The heme-binding protein Dap1 links iron homeostasis to azole resistance via the P450 protein Erg11 in candida glabrata. FEMS Yeast Research. 2013;13(4):411–421. doi: 10.1111/1567-1364.12043. [DOI] [PubMed] [Google Scholar]
- 44.Srikantha T, Daniels KJ, Pujol C, Kim E, Soll DR. Identification of genes upregulated by the transcription factor Bcr1 that are involved in impermeability, impenetrability and drug-resistance of Candida albicans a/α biofilms. Eukaryot Cell. 2013 doi: 10.1128/EC.00071-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Comerford KM, Wallace TJ, Karhausen J, Louis NA, Montalto MC, Colgan SP. Hypoxia-inducible factor-1-dependent regulation of the multidrug resistance (MDR1) gene. Cancer Research. 2002;62(12):3387–3394. [PubMed] [Google Scholar]
- 46.Comerford KM, Colgan SP. Assessing oxygen sensitivity of the multidrug resistance (MDR) gene. Methods in Enzymology. 2004;381:376–387. doi: 10.1016/S0076-6879(04)81025-5. [DOI] [PubMed] [Google Scholar]
- 47.Comerford KM, Cummins EP, Taylor CT. c-Jun NH2-terminal kinase activation contributes to hypoxia-inducible factor 1α-dependent P-glycoprotein expression in hypoxia. Cancer Research. 2004;64(24):9057–9061. doi: 10.1158/0008-5472.CAN-04-1919. [DOI] [PubMed] [Google Scholar]
- 48.Mukhopadhyay CK, Mazumder B, Fox PL. Role of hypoxia-inducible factor-1 in transcriptional activation of ceruloplasmin by iron deficiency. Journal of Biological Chemistry. 2000;275(28):21048–21054. doi: 10.1074/jbc.M000636200. [DOI] [PubMed] [Google Scholar]
- 49.Setiadi ER, Doedt T, Cottier F, Noffz C, Ernst JF. Transcriptional response of Candida albicans to hypoxia: linkage of oxygen sensing and Efg1p-regulatory networks. Journal of Molecular Biology. 2006;361(3):399–411. doi: 10.1016/j.jmb.2006.06.040. [DOI] [PubMed] [Google Scholar]
- 50.Sosinska GJ, de Groot PWJ, de Mattos MJT, et al. Hypoxic conditions and iron restriction affect the cell-wall proteome of Candida albicans grown under vagina-simulative conditions. Microbiology. 2008;154(2):510–520. doi: 10.1099/mic.0.2007/012617-0. [DOI] [PubMed] [Google Scholar]
- 51.Brown AJP, Gow NAR. Regulatory networks controlling Candida albicans morphogenesis. Trends in Microbiology. 1999;7(8):333–338. doi: 10.1016/s0966-842x(99)01556-5. [DOI] [PubMed] [Google Scholar]
- 52.Sonneborn A, Bockmühl DP, Ernst JF. Chlamydospore formation in Candida albicans requires the Efg1p morphogenetic regulator. Infection and Immunity. 1999;67(10):5514–5517. doi: 10.1128/iai.67.10.5514-5517.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Stichternoth C, Ernst JF. Hypoxic adaptation by Efg1 regulates biofilm formation by Candida albicans . Applied and Environmental Microbiology. 2009;75(11):3663–3672. doi: 10.1128/AEM.00098-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Bonhomme J, Chauvel M, Goyard S, Roux P, Rossignol T, D’Enfert C. Contribution of the glycolytic flux and hypoxia adaptation to efficient biofilm formation by Candida albicans . Molecular Microbiology. 2011;80(4):995–1013. doi: 10.1111/j.1365-2958.2011.07626.x. [DOI] [PubMed] [Google Scholar]
- 55.Carvalho AP, Gursky LC, Rosa RT, et al. Non-steroidal anti-inflammatory drugs may modulate the protease activity of Candida albicans . Microbial Pathogenesis. 2010;49(6):315–322. doi: 10.1016/j.micpath.2010.07.007. [DOI] [PubMed] [Google Scholar]
- 56.Synnott JM, Guida A, Mulhern-Haughey S, Higgins DG, Butler G. Regulation of the hypoxic response in Candida albicans . Eukaryotic Cell. 2010;9(11):1734–1746. doi: 10.1128/EC.00159-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Stichternoth C, Fraund A, Setiadi E, Giasson L, Vecchiarelli A, Ernst JF. Sch9 kinase integrates hypoxia and CO2 sensing to suppress hyphal morphogenesis in Candida albicans . Eukaryotic Cell. 2011;10(4):502–511. doi: 10.1128/EC.00289-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ernst JF, Tielker D. Responses to hypoxia in fungal pathogens. Cellular Microbiology. 2009;11(2):183–190. doi: 10.1111/j.1462-5822.2008.01259.x. [DOI] [PubMed] [Google Scholar]
- 59.Grahl N, Shepardson KM, Chung D, Cramer RA. Hypoxia and fungal pathogenesis: to air or not to air? Eukaryotic Cell. 2012;11(5):560–570. doi: 10.1128/EC.00031-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Romani L, Bistoni F, Puccetti P. Adaptation of Candida albicans to the host environment: the role of morphogenesis in virulence and survival in mammalian hosts. Current Opinion in Microbiology. 2003;6(4):338–343. doi: 10.1016/s1369-5274(03)00081-x. [DOI] [PubMed] [Google Scholar]
- 61.Gujjar PR, Finucane M, Larsen B. The effect of estradiol on Candida albicans growth. Annals of Clinical and Laboratory Science. 1997;27(2):151–156. [PubMed] [Google Scholar]
- 62.Zhang X, Essmann M, Burt ET, Larsen B. Estrogen effects on Candida albicans: a potential virulence-regulating mechanism. Journal of Infectious Diseases. 2000;181(4):1441–1446. doi: 10.1086/315406. [DOI] [PubMed] [Google Scholar]
- 63.Madani ND, Malloy PJ, Rodriguez-Pombo P, Krishnan AV, Feldman D. Candida albicans estrogen-binding protein gene encodes an oxidoreductase that is inhibited by estradiol. Proceedings of the National Academy of Sciences of the United States of America. 1994;91(3):922–926. doi: 10.1073/pnas.91.3.922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Malloy PJ, Zhao X, Madani ND, Feldman D. Cloning and expression of the gene from Candida albicans that encodes a high-affinity corticosteroid-binding protein. Proceedings of the National Academy of Sciences of the United States of America. 1993;90(5):1902–1906. doi: 10.1073/pnas.90.5.1902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Das M, Datta A. Steroid binding protein(s) in yeasts. Biochemistry International. 1985;11(2):171–176. [PubMed] [Google Scholar]
- 66.De Micheli M, Bille J, Schueller C, Sanglard D. A common drug-responsive element mediates the upregulation of the Candida albicans ABC transporters CDR1 and CDR2, two genes involved in antifungal drug resistance. Molecular Microbiology. 2002;43(5):1197–1214. doi: 10.1046/j.1365-2958.2002.02814.x. [DOI] [PubMed] [Google Scholar]
- 67.Kamani N, Akhtar Gaur N, Jha S, et al. SRE1 and SRE2 are two specific steroid-responsive modules of candida drug resistance gene I (CDRI) promoter. Yeast. 2004;21(3):219–239. doi: 10.1002/yea.1067. [DOI] [PubMed] [Google Scholar]
- 68.Coste AT, Karababa M, Ischer F, Bille J, Sanglard D. TAC1, transcriptional activator of CDR genes, is a new transcription factor involved in the regulation of Candida albicans ABC transporters CDR1 and CDR2. Eukaryotic Cell. 2004;3(6):1639–1652. doi: 10.1128/EC.3.6.1639-1652.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Banerjee D, Pillai B, Karnani N, Mukhopadhyay G, Prasad R. Genome-wide expression profile of steroid response in saccharomyces cerevisiae. Biochemical and Biophysical Research Communications. 2004;317(2):406–413. doi: 10.1016/j.bbrc.2004.03.053. [DOI] [PubMed] [Google Scholar]
- 70.Barnes KM, Dickstein B, Cutler GB, Jr., Fojo T, Bates SE. Steroid transport, accumulation, and antagonism of P-glycoprotein in multidrug-resistant cells. Biochemistry. 1996;35(15):4820–4827. doi: 10.1021/bi952380k. [DOI] [PubMed] [Google Scholar]
- 71.Sukhai M, Piquette-Miller M. Regulation of the multidrug resistance genes by stress signals. Journal of Pharmacy & Pharmaceutical Sciences. 2000;3(2):268–280. [PubMed] [Google Scholar]
- 72.Labialle S, Gayet L, Marthinet E, Rigal D, Baggetto LG. Transcriptional regulators of the human multidrug resistance 1 gene: recent views. Biochemical Pharmacology. 2002;64(5-6):943–948. doi: 10.1016/s0006-2952(02)01156-5. [DOI] [PubMed] [Google Scholar]
- 73.Banerjee D, Lelandais G, Shukla S, et al. Responses of pathogenic and nonpathogenic yeast species to steroids reveal the functioning and evolution of multidrug resistance transcriptional networks. Eukaryotic Cell. 2008;7(1):68–77. doi: 10.1128/EC.00256-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Gogoi S, Shekarrao K, Duarah A, Bora TC, Gogoi S, Boruah RC. A microwave promoted solvent-free approach to steroidal quinolines and their in vitro evaluation for antimicrobial activities. Steroids. 2012;77(13):1438–1445. doi: 10.1016/j.steroids.2012.08.008. [DOI] [PubMed] [Google Scholar]
- 75.Kakati D, Sarma RK, Saikia R, Barua NC, Sarma JC. Rapid microwave assisted synthesis and antimicrobial bioevaluation of novel steroidal chalcones. Steroids. 2013;78(3):321–326. doi: 10.1016/j.steroids.2012.12.003. [DOI] [PubMed] [Google Scholar]
- 76.Ernst JF, Schmidt A. Dimorphism in Human Pathogenic and Apathogenic Yeasts. Bern, Switzerland: Karger; 2000. [Google Scholar]
- 77.Enrst JF. Transcription factors in Candida albicans-environmental control of morphogenesis. Microbiology. 2000;146(8):1763–1774. doi: 10.1099/00221287-146-8-1763. [DOI] [PubMed] [Google Scholar]
- 78.Lo H-J, Wang J-S, Lin C-Y, et al. Efg1 involved in drug resistance by regulating the expression of ERG3 in Candida albicans . Antimicrobial Agents and Chemotherapy. 2005;49(3):1213–1215. doi: 10.1128/AAC.49.3.1213-1215.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Prasad T, Hameed S, Manoharlal R, et al. Morphogenic regulator EFG1 affects the drug susceptibilities of pathogenic Candida albicans . FEMS Yeast Research. 2010;10(5):587–596. doi: 10.1111/j.1567-1364.2010.00639.x. [DOI] [PubMed] [Google Scholar]
- 80.Vandeputte P, Pradervand S, Ischer F, Coste AT, Ferrari S, Harshman K. Identification and functional characterization of Rca1, a transcription factor involved in both antifungal susceptibility and host response in Candida albicans . Eukaryot Cell. 2012;11(7):916–931. doi: 10.1128/EC.00134-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Roepe PD. pH and multidrug resistance. Novartis Foundation Symposium. 2001;240:232–247. doi: 10.1002/0470868716.ch16. [DOI] [PubMed] [Google Scholar]
- 82.Davis D. Adaptation to environmental pH in Candida albicans and its relation to pathogenesis. Current Genetics. 2003;44(1):1–7. doi: 10.1007/s00294-003-0415-2. [DOI] [PubMed] [Google Scholar]
- 83.Davis D, Wilson RB, Mitchell AP. RIM101-dependent and-independent pathways govern pH responses in Candida albicans . Molecular and Cellular Biology. 2000;20(3):971–978. doi: 10.1128/mcb.20.3.971-978.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ghannoum MA, Spellberg B, Saporito-Irwin SM, Fonzi WA. Reduced virulence of Candida albicans PHR1 mutants. Infection and Immunity. 1995;63(11):4528–4530. doi: 10.1128/iai.63.11.4528-4530.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Serrano R, Ruiz A, Bernal D, Chambers JR, Ariño J. The transcriptional response to alkaline pH in saccharomyces cerevisiae: Evidence for calcium-mediated signalling. Molecular Microbiology. 2002;46(5):1319–1333. doi: 10.1046/j.1365-2958.2002.03246.x. [DOI] [PubMed] [Google Scholar]
- 86.Bader T, Bodendorfer B, Schröppel K, Morschhäuser J. Calcineurin is essential for virulence in Candida albicans . Infection and Immunity. 2003;71(9):5344–5354. doi: 10.1128/IAI.71.9.5344-5354.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Mukhopadhyay K, Kohli A, Prasad R. Drug susceptibilities of yeast cells are affected by membrane lipid composition. Antimicrobial Agents and Chemotherapy. 2002;46(12):3695–3705. doi: 10.1128/AAC.46.12.3695-3705.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Löffler J, Einsele H, Hebart H, Schumacher U, Hrastnik C, Daum G. Phospholipid and sterol analysis of plasma membranes of azole-resistant Candida albicans strains. FEMS Microbiology Letters. 2000;185(1):59–63. doi: 10.1111/j.1574-6968.2000.tb09040.x. [DOI] [PubMed] [Google Scholar]
- 89.Mukhopadhyay K, Prasad T, Saini P, Pucadyil TJ, Chattopadhyay A, Prasad R. Membrane sphingolipid-ergosterol interactions are important determinants of multidrug resistance in Candida albicans . Antimicrobial Agents and Chemotherapy. 2004;48(5):1778–1787. doi: 10.1128/AAC.48.5.1778-1787.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Shea JM, Del Poeta M. Lipid signaling in pathogenic fungi. Current Opinion in Microbiology. 2006;9(4):352–358. doi: 10.1016/j.mib.2006.06.003. [DOI] [PubMed] [Google Scholar]
- 91.Ejsinga CS, Sampaioa JL, Surendranatha V, Duchoslavb E, Ekroosc K, Klemma RW. Global analysis of the yeast lipidome by quantitative shotgun mass spectrometry. Proceedings of the National Academy of Sciences. 2009;106(7):2136–2141. doi: 10.1073/pnas.0811700106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Sharma M, Dhamgaye S, Singh A, Prasad R. Lipidome analysis reveals antifungal polyphenol curcumin affects membrane lipid homeostasis. Frontiers in Bioscience. 2012;(4):1195–1209. doi: 10.2741/451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Singh A, Prasad R. Comparative lipidomics of azole sensitive and resistant clinical isolates of Candida albicans reveals unexpected diversity in molecular lipid imprints. PLoS ONE. 2011;6(4) doi: 10.1371/journal.pone.0019266.e19266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Singh A, Yadav V, Prasad R. Comparative lipidomics in clinical isolates of Candida albicans reveal crosstalk between mitochondria, cell wall integrity and azole resistance. PLoS One. 2012;7(6) doi: 10.1371/journal.pone.0039812.e39812 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Singh A, Mahto KK, Prasad R. Lipidomics and in vitro azole resistance in Candida albicans . OMICS. 2013;17(2):84–93. doi: 10.1089/omi.2012.0075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Jain P, Akula I, Edlind T. Cyclic AMP signaling pathway modulates susceptibility of candida species and Saccharomyces cerevisiae to antifungal azoles and other sterol biosynthesis inhibitors. Antimicrobial Agents and Chemotherapy. 2003;47(10):3195–3201. doi: 10.1128/AAC.47.10.3195-3201.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Steinbach WJ, Reedy JL, Cramer RA, Jr., Perfect JR, Heitman J. Harnessing calcineurin as a novel anti-infective agent against invasive fungal infections. Nature Reviews Microbiology. 2007;5(6):418–430. doi: 10.1038/nrmicro1680. [DOI] [PubMed] [Google Scholar]
- 98.Alonso-Monge R, Román E, Arana DM, Pla J, Nombela C. Fungi sensing environmental stress. Clinical Microbiology and Infection. 2009;15(1):17–19. doi: 10.1111/j.1469-0691.2008.02690.x. [DOI] [PubMed] [Google Scholar]
- 99.Cowen LE, Lindquist S. Cell biology: Hsp90 potentiates the rapid evolution of new traits: drug resistance in diverse fungi. Science. 2005;309(5744):2185–2189. doi: 10.1126/science.1118370. [DOI] [PubMed] [Google Scholar]
- 100.Cowen LE, Singha SD, Köhlerb JR, Collinsa C, Zaasc AK, Schellc WA. Harnessing Hsp90 function as a powerful, broadly effective therapeutic strategy for fungal infectious disease. Proceedings of the National Academy of Sciences. 2009;106(8):2818–2823. doi: 10.1073/pnas.0813394106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Bastidas RJ, Reedy JL, Morales-Johansson H, Heitman J, Cardenas ME. Signaling cascades as drug targets in model and pathogenic fungi. Current Opinion in Investigational Drugs. 2008;9(8):856–864. [PMC free article] [PubMed] [Google Scholar]
- 102.Thakur JK, Arthanari H, Yang F, et al. A nuclear receptor-like pathway regulating multidrug resistance in fungi. Nature. 2008;452(7187):604–609. doi: 10.1038/nature06836. [DOI] [PubMed] [Google Scholar]
- 103.Robbins N, Collins C, Morhayim J, Cowen LE. Metabolic control of antifungal drug resistance. Fungal Genetics and Biology. 2010;47(2):81–93. doi: 10.1016/j.fgb.2009.07.004. [DOI] [PubMed] [Google Scholar]
- 104.Lafayette SL, Collins C, Zaas AK, et al. PKC signaling regulates drug resistance of the fungal pathogen Candida albicans via circuitry comprised of mkc1, calcineurin, and hsp90. PLoS Pathogens. 2010;6(8):79–80. doi: 10.1371/journal.ppat.1001069.e1001069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.de Dios CH, Román E, Monge RA, Pla J. The role of MAPK signal transduction pathways in the response to oxidative stress in the fungal pathogen Candida albicans: implications in virulence. Current Protein and Peptide Science. 2010;11(8):693–703. doi: 10.2174/138920310794557655. [DOI] [PubMed] [Google Scholar]
- 106.Konopka JB. N-acetylglucosamine (GlcNAc) functions in cell signaling. Scientifica. 2012;2012:15 pages. doi: 10.6064/2012/489208.489208 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Desai JV, Bruno VM, Ganguly S, Stamper RJ, Mitchell KF, Solis N. Regulatory role of glycerol in Candida albicans biofilm formation. MBio. 2013;4(2) doi: 10.1128/mBio.00637-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Lee SH, Jeon JE, Ahn CH, Chung SC, Shin J, Oh KB. Inhibition of yeast-to-hypha transition in Candida albicans by phorbasin H isolated from phorbas sp. 2013;97(7):3141–3148. doi: 10.1007/s00253-012-4549-3. [DOI] [PubMed] [Google Scholar]
- 109.Saito H, Tamura M, Imai K, Ishigami T, Ochiai K. Catechin inhibits Candida albicans dimorphism by disrupting Cek1 phosphorylation and cAMP synthesis. Microbial Pathogenesis. 2013;56:16–20. doi: 10.1016/j.micpath.2013.01.002. [DOI] [PubMed] [Google Scholar]
- 110.Mavrianos J, Berkow EL, Desai C, Pandey A, Batish M, Rabadi MJ. Mitochondrial two-component signaling systems in Candida albicans . Eukaryot Cell. 2013 doi: 10.1128/EC.00048-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Gaur NA, Puri N, Karnani N, Mukhopadhyay G, Goswami SK, Prasad R. Identification of a negative regulatory element which regulates basal transcription of a multidrug resistance gene CDR1 of Candida albicans . FEMS Yeast Research. 2004;4(4-5):389–399. doi: 10.1016/S1567-1356(03)00204-6. [DOI] [PubMed] [Google Scholar]
- 112.Talibi D, Raymond M. Isolation of a putative Candida albicans transcriptional regulator involved in pleiotropic drug resistance by functional complementation of a pdr1 pdr3 mutation in saccharomyces cerevisiae. Journal of Bacteriology. 1999;181(1):231–240. doi: 10.1128/jb.181.1.231-240.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Chen C-G, Yang Y-L, Shih H-I, Su C-L, Lo H-J. CaNdt80 is involved in drug resistance in Candida albicans by regulating CDR1. Antimicrobial Agents and Chemotherapy. 2004;48(12):4505–4512. doi: 10.1128/AAC.48.12.4505-4512.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Morschhäuser J, Barker KS, Liu TT, Blaß-Warmuth J, Homayouni R, Rogers PD. The transcription factor Mrr1p controls expression of the MDR1 efflux pump and mediates multidrug resistance in Candida albicans . PLoS Pathogens. 2007;3(11, article e164) doi: 10.1371/journal.ppat.0030164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Chen C-G, Yang Y-L, Tseng K-Y, et al. Rep1p negatively regulating MDR1 efflux pump involved in drug resistance in Candida albicans . Fungal Genetics and Biology. 2009;46(9):714–720. doi: 10.1016/j.fgb.2009.06.003. [DOI] [PubMed] [Google Scholar]
- 116.Silver PM, Oliver BG, White TC. Role of Candida albicans transcription factor Upc2p in drug resistance and sterol metabolism. Eukaryotic Cell. 2004;3(6):1391–1397. doi: 10.1128/EC.3.6.1391-1397.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Coste AT, Turner V, Ischer F, et al. A mutation in Tac1p, a transcription factor regulating CDR1 and CDR2, is coupled with loss of heterozygosity at chromosome 5 to mediate antifungal resistance in Candida albicans . Genetics. 2006;172(4):2139–2156. doi: 10.1534/genetics.105.054767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Tsai H-F, Krol AA, Sarti KE, Bennett JE. Candida glabrata PDR1, a transcriptional regulator of a pleiotropic drug resistance network, mediates azole resistance in clinical isolates and petite mutants. Antimicrobial Agents and Chemotherapy. 2006;50(4):1384–1392. doi: 10.1128/AAC.50.4.1384-1392.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Vermitsky J-P, Earhart KD, Smith WL, Homayouni R, Edlind TD, Rogers PD. Pdr1 regulates multidrug resistance in candida glabrata: gene disruption and genome-wide expression studies. Molecular Microbiology. 2006;61(3):704–722. doi: 10.1111/j.1365-2958.2006.05235.x. [DOI] [PubMed] [Google Scholar]
- 120.Dunkel N, Blaß J, Rogers PD, Morschhäuser J. Mutations in the multi-drug resistance regulator MRR1, followed by loss of heterozygosity, are the main cause of MDR1 overexpression in fluconazole-resistant Candida albicans strains. Molecular Microbiology. 2008;69(4):827–840. doi: 10.1111/j.1365-2958.2008.06309.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Dunkel N, Liu TT, Barker KS, Homayouni R, Morschhäuser J, Rogers PD. A gain-of-function mutation in the transcription factor Upc2p causes upregulation of ergosterol biosynthesis genes and increased fluconazole resistance in a clinical Candida albicans isolate. Eukaryotic Cell. 2008;7(7):1180–1190. doi: 10.1128/EC.00103-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Ferrari S, Ischer F, Calabrese D, et al. Gain of function mutations in CgPDR1 of candida glabrata not only mediate antifungal resistance but also enhance virulence. PLoS Pathogens. 2009;5(1) doi: 10.1371/journal.ppat.1000268.e1000268 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Pérez JC, Kumamoto CA, Johnson AD. Candida albicans commensalism and pathogenicity are intertwined traits directed by a tightly knit transcriptional regulatory circuit. PLOS Biology. 2013;11(3) doi: 10.1371/journal.pbio.1001510.e1001510 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Dhamgaye S, Bernard M, Lelandais G, Sismeiro O, Lemoine S, Coppée JY. RNA sequencing revealed novel actors of the acquisition of drug resistance in Candida albicans . BMC Genomics. 2012;13, article 396 doi: 10.1186/1471-2164-13-396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Manoharlal R, Gaur NA, Panwar SL, Morschhäuser J, Prasad R. Transcriptional activation and increased mRNA stability contribute to overexpression of CDR1 in azole-resistant Candida albicans . Antimicrobial Agents and Chemotherapy. 2008;52(4):1481–1492. doi: 10.1128/AAC.01106-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Manoharlal R, Gorantala J, Sharma M, Sanglard D, Prasad R. PAP1 [poly(A) polymerase 1] homozygosity and hyperadenylation are major determinants of increased mRNA stability of CDR1 in azole-resistant clinical isolates of Candida albicans . Microbiology. 2010;156(2):313–326. doi: 10.1099/mic.0.035154-0. [DOI] [PubMed] [Google Scholar]
- 127.Chen C, Noble SM. Post-transcriptional regulation of the Sef1 transcription factor controls the virulence of Candida albicans in its mammalian host. PLOS Pathogens. 2012;8(11) doi: 10.1371/journal.ppat.1002956.e1002956 [DOI] [PMC free article] [PubMed] [Google Scholar]


