Abstract
A comparative phylogeographic study on two economically important African tick species, Amblyomma hebraeum and Hyalomma rufipes was performed to test the influence of host specificity and host movement on dispersion. Pairwise AMOVA analyses of 277 mtDNA COI sequences supported significant population differentiation among the majority of sampling sites. The geographic mitochondrial structure was not supported by nuclear ITS-2 sequencing, probably attributed to a recent divergence. The three-host generalist, A. hebraeum, showed less mtDNA geographic structure, and a lower level of genetic diversity, while the more host-specific H. rufipes displayed higher levels of population differentiation and two distinct mtDNA assemblages (one predominantly confined to South Africa/Namibia and the other to Mozambique and East Africa). A zone of overlap is present in southern Mozambique. A mechanistic climate model suggests that climate alone cannot be responsible for the disruption in female gene flow. Our findings furthermore suggest that female gene dispersal of ticks is more dependent on the presence of juvenile hosts in the environment than on the ability of adult hosts to disperse across the landscape. Documented interspecific competition between the juvenile stages of H. rufipes and H. truncatum is implicated as a contributing factor towards disrupting gene flow between the two southern African H. rufipes genetic assemblages.
Introduction
Successful dispersal and the subsequent ability to reproduce with conspecifics are central to maintaining the integrity of sexually reproducing species [1]. The factors affecting the dispersal of parasites, and how genetic material is mixed among geographic regions, are not well documented. Ectoparasites in particular provide a case in point [2] and contemporary phylogeographic uncertainties can partly be ascribed to the complex mode of ectoparasite reproduction, coupled to an incredible diversity in life forms [3]. For example, blood-feeding ixodid ticks (Acari: Ixodidae) comprise approximately 700 extant species [4,5] characterized by divergent life histories (two or three hosts needed to complete the life cycle; host specialists versus generalists; mating on or off the host etc.). In multi-host taxa, the different life stages also often parasitize a variety of vertebrate hosts (mammals, birds, reptiles and amphibians) with different abilities for dispersal [1].
Identifying the factors influencing the dispersal and genetic connectivity among tick populations of different geographic origin is thus not a trivial exercise and several hypotheses have been proposed. Key to the discussions are host specificity and the mobility of the hosts of adult ticks [1,6,7], the number and type of host species needed to complete the life cycle [8,9], sex-biased dispersal of different life stages [6,10], parasite-host immunity interactions [11] and abiotic factors associated with biogeographic barriers and other environmental changes [12–16].
A better understanding of the dispersal ability of ticks is essential since they constitute an economically important group of arthropods that act as vectors of diseases to domestic livestock and wild animals [10,17,18]. In an attempt to address the paucity of data needed to explain some of the mechanisms responsible for tick dispersion and gene flow, we selected two economically important African species, Amblyomma hebraeum and Hyalomma rufipes. Amblyomma hebraeum is responsible for the transmission of Ehrlichia ruminantium (the cause of heartwater in bovine species) [19,20], Theileria mutans (causing benign theileriosis in cattle) [21,22], and Rickettsia africae (causing African tick bite fever in humans) [23,24]. In the southern African context, H. rufipes is probably the most important vector of Crimean-Congo hemorrhagic fever (CCHF) virus to humans [25,26]. It also transmits Anaplasma marginale, the causative organism of bovine anaplasmosis [27], Babesia occultans, the cause of benign babesiosis in cattle [17,28] and Ricketsia conorii, the cause of tick typhus in humans [29].
Hyalomma rufipes and A. hebraeum exhibit differences in life history traits, and when examined in a comparative fashion across a broadly speaking similar abiotic landscape, the data may help to explain some of the mechanisms involved in tick dispersion. The two species have partially overlapping distributions in southern Africa (Figure 1) and their adults mainly utilize highly mobile wild and domestic bovids as hosts [30–32]. It is thus predicted that the reproductively active adult stages of both species have the potential to frequently disperse over large tracts of land via natural host movement that could further also be facilitated by anthropogenic activities such as the trade in domestic animals. The life cycles of the two species differ, however, when the immature stages are taken into consideration. The larvae and nymphs of the three-host A. hebraeum feed on a wide variety of large and small mammals, including hares, ground-frequenting birds and sometimes tortoises [32–35] In contrast, the immature stages of the two-host tick, H. rufipes are host restricted and have only been recorded on hares and ground-frequenting birds [34,36–38]. In comparison, H. rufipes can thus be regarded as a more specialized parasite since the availability of wildlife is critical for the completion of its life cycle [37,39]. In A. hebraeum all three life stages can feed on the same adult host species [32,40] and this characteristic makes it a more generalist species with higher ecological plasticity [41].
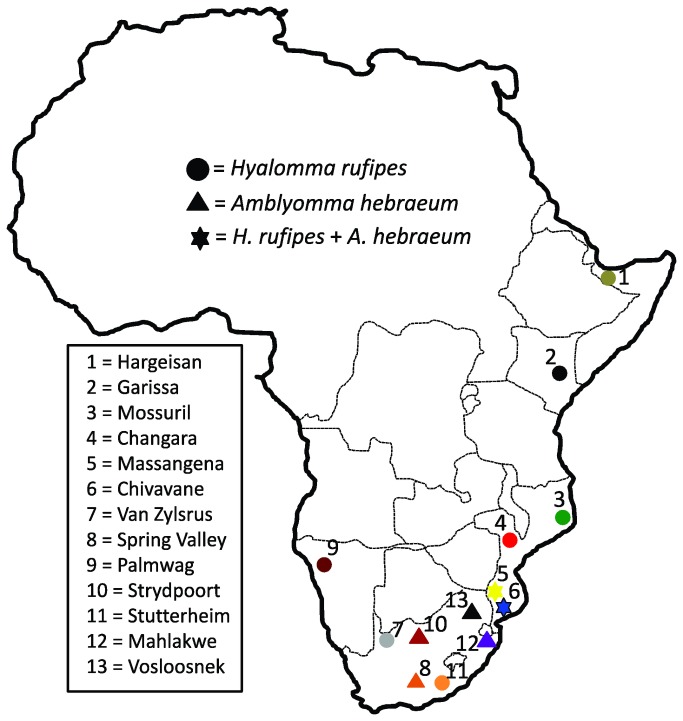
Figure 1. Sampling localities for A. hebraeum and H. rufipes used in the present study.
Locality names correspond to those provided in Table 1 and colours correspond to those provided in Figures 2 and 3.
Following from the above, it can be hypothesised that localized geographic populations of H. rufipes will exhibit stronger signals of population differentiation than A. hebraeum. To test this, we embarked on a comparative phylogeographic study to infer genetic connectivity among different sampling sites across the landscape. Ticks were sampled throughout southern and east Africa and were analyzed phylogeographically by making use of the mitochondrial COI gene, and in the case of H. rufipes, also the nuclear ITS-2 gene fragment. It was envisaged that the outcome of our investigation could provide new empirical data that can potentially be used to explain some of the mechanisms that play a role in the dispersal of ticks. This may in turn have direct implications for a better understanding of ectoparasite evolution and in particular speciation [42]. Should distinct genetic lineages be observed, the results of our study will also have medical/veterinary implications that will need further testing (for example the implementation of stricter control measures for the movement of domestic livestock especially if acaricide resistance can be coupled to genetic lineages) [18].
Materials and Methods
2.1 Specimen collections
A total of 115 adult A. hebraeum were collected from six localities in southern Africa and 162 adult H. rufipes were sampled from nine localities (Figure 1; Table 1). Most specimens were collected from cattle, Bos taurus/indicus hosts and in the rare event that sufficient numbers of ticks were not available at a specific site, they were collected from sheep, Ovis aries, or goats, Capra aegagrus. No ethical clearance was required to perform work on invertebrate parasites and all ticks were collected with permission from landowners. Ticks were stored in 70% ethanol, and their specific identification was confirmed by Ivan Horak.
Table 1. Locality information and genetic summary statistics for the geographic samples included in this study.
| Location | GPS Co-ordinates | N | Hn (Hs) | h (± SD) | π (± SD) | Fu’s Fs (P value) | τ |
|---|---|---|---|---|---|---|---|
| A. hebraeum | |||||||
| Massangena | 21°32'0.0"S 32°57'0.0"E | 20 | 6 (1) | 0.64 (±0.12) | 0.002 (0.001) | -2.394 (0.023) | 0.994 |
| Chivavane | 25°3'0.00"S 33°37'60.00"E | 20 | 7 (3) | 0.79 (±0.06) | 0.002 (0.001) | -2.908 (0.013) | 1.375 |
| Vosloosnek | 25°0’0.0"S 30.5°0’0.0"E | 18 | 5 (2) | 0.67 (±0.08) | 0.001 (0.001) | -1.822 (0.035) | 1.016 |
| Strydpoort | 27°0’0.0"S 26° 0’0.0"E | 18 | 7 (4) | 0.82 (±0.06) | 0.002 (0.001) | -2.521 (0.030) | 1.516 |
| Mahlakwe | 27.5°0’0.0"S 32.5°0’0.0"E | 20 | 4 (2) | 0.28 (±0.13) | 0.000 (0.000) | -2.749 (0.001) | 3.000 |
| Spring Valley | 32°17'0.0"S 26°25'0.0"E | 19 | 3 (2) | 0.20 (±0.12) | 0.000 (0.000) | -1.804 (0.012) | 3.0 |
| All | 115 | 22 (14) | 0.66 (±0.08) | 0.002 (0.001) | -23.366 (0.000) | 1.056 | |
| H. rufipes | |||||||
| Hargeisan | 9°33’44.60″N 44°04’37.25″E | 19 | 15 (9) | 0.96 (±0.04) | 0.013 (±0.007) | -4.163 (0.040) | 1.598 |
| Garissa | 0°27'28.83"S 39°39' 30.0"E | 20 | 16 (11) | 0.98 (±0.02) | 0.009 (±0.005) | -7.286 (±0.003) | 3.121 |
| Mussuril | 14°57'57.60"S 40° 39'39.97"E | 18 | 9 (7) | 0.80 (±0.09) | 0.012 (±0.007) | 1.035 (0.702) | 18.682 |
| Changara | 16°25'25.7″S 33°37'4.3"E | 20 | 16 (6) | 0.97 (±0.03) | 0.017 (±0.009) | -3.571 (±0.072) | 1.346 |
| Massangena | 21°32'0.0"S 32°57'0.0"E | 20 | 12 (2) | 0.94 (±0.03) | 0.009 (±0.005) | -1.879 (0.194) | 1.625 |
| Chivavane | 25°3'0.00"S 33°37'60.00"E | 19 | 12 (3) | 0.92 (±0.05) | 0.013 (±0.007) | -1.083 (0.310) | 1.830 |
| Stutterheim | 27°0’0.0"S 26° 0’0.0"E | 19 | 3 (0) | 0.51 (±0.12) | 0.001 (±0.001) | 1.211 (0.753) | 0.000 |
| Van Zylsrus | 26°20'18.28"S 22°36'44.03''E | 8 | 5 (3) | 0.86 (±0.11) | 0.002 (±0.002) | -1.358 (0.100) | 2.029 |
| Palmwag | 19°28'52.86"S 14°10'58.87''E | 19 | 10 (6) | 0.85 (±0.07) | 0.006 (±0.003) | -2.106 (0.137) | 2.879 |
| All | 162 | 64 (47) | 0.96 (±0.04) | 0.009 (±0.005) | -24.096 (0.000) | 0.486 |
Locality designations correspond to those given in Figure 1. N is the number of mtDNA sequences; Hn is the number of haplotypes followed by (Hs = singletons); h is the haplotype diversity (± standard deviation); π is the nucleotide diversity (± standard deviation); Fu’s Fs and τ is also indicated (see text for details).
2.2 DNA extraction, PCR and sequencing
Genomic DNA was extracted from individual ticks following the manufacturer’s protocol for animal tissues using a DNeasy® Blood & Tissue kit (QIAGENTM, Crawley, UK). Proteinase K (20mg/µl) digestion at 56°C was extended to 48 hours and the extracted DNA was eluted in 250µl of AE buffer and stored at -20°C.
A ~800 base pair stretch of the mitochondrial COI region was amplified by polymerase chain reaction (PCR), using a tick specific forward AR-U-COIa (5’-AAACTRTKTRCCTTCAAAG-3’) and reverse primer AR-L-COIa (5’-GTRTTAAARTTTCGATCSGTTA-3’), respectively (Ropiquet et al. unpubl. data). For the nuclear DNA, a portion of the ITS-2 was amplified by PCR from selected individuals of H. rufipes, using the tick specific forward primer RIB-8 (5’-GTCGTAGTCCGCCGTC-3’) and the reverse primer RIB-11 (5’-GAGTACGACGCCCTACC-3’) [43]. PCR reactions were performed following standard techniques with primer annealing at 40°C for COI and 62.5°C for ITS-2. PCR products were separated on 1% agarose gels stained with ethidium bromide and visualized using a UV light. After purification with the Nucleoafast 96 well plate-kit (Macherey-Nagel, Düren, Germany), PCR products were sequenced using the protocol prescribed by the BigDye terminator v 3.1 kit (Applied Biosystems, Warrington, UK)
2.3 Sequence data analyses
Sequences were manually inspected and edited with the software BioEdit v 7.0.5 [44]. The mitochondrial sequences were translated into proteins (http://www.ebi.ac.uk/Tools/emboss/transeq/index.htm) in order to ensure that there were no stop codons. Species authenticity was further confirmed with BlastN searches (http://blast.ncbi.nlm.nih.gov/Blast.cgi). The nuclear DNA haplotype reconstruction was conducted using DnaSP v 5.10 [45] and the default settings in PHASE v 2.1 [46] with 1 000 000 iterations.
Haplotype diversities (h), and nucleotide diversities (π) were estimated in Arlequin v 3.5 [47]. Uncorrected sequence divergences (distance matrix) were obtained using PAUP 4.0 [48]. Sequences were collapsed to haplotypes using DnaSP v 5.10 [45] and evolutionary relationships among haplotypes were depicted by statistical parsimony haplotype networks generated in TCS 1.21 [49]. Genetic differentiation between the various sampling populations was determined using an analyses of molecular variance (AMOVA) implemented in Arlequin v 3.5. By utilizing the outcome of the COI haplotype networks as priors, we also performed a hierarchical analysis of molecular variance (AMOVA [50]) to determine the level of variation within and among geographic groupings. Significance was estimated at the 0.05 level with 10 000 permutation steps.
As a complement to the traditional population genetic analysis (φST; Fst), a model-based Bayesian clustering method was used to investigate the genetic structure across the geographic range (BAPS v 5.3 [51]). Analysis of “spatial clustering of individuals” and “groups of individuals” were performed without any pre-defined assumptions on geographic group structure. Each analysis was performed 10 times with different k values (K=1-10). To investigate geographic distance as a potential isolating mechanism, the Mantel test [52], as implemented in Arlequin v 3.5 [47] was used to test for isolation-by-distance.
In order to obtain information about processes that could have caused observed genetic variation, historical demography of populations were analyzed using two approaches. Firstly, to test for selective neutrality, Fu’s Fs [53], was estimated in Arlequin v. 3.5 [47]. Mismatch distributions were then used to test for population expansion [54–56] in Arlequin v. 3.5 [47]. Approximate time of population expansion, t, was calculated by substituting values for τ and μ in the equation τ = 2µt, where τ is given by the mismatch analysis as an estimate of the time of occurrence of the hypothetical expansion and μ (the mutation rate per site per generation) of 0.75% between ancestor-descendent alleles (i.e., half of 1.5%, the average value for arthropod pairwise differences per million years [57,58]) was used. τ is expressed in generations (months), while the value of μ is measured in years.
2.4 Climate suitability analyses
We assessed the suitability of climate for permanent populations of H. rufipes from collection records obtained between 1985 and 2010. The ‘Maximum Entropy Approach’ within the Maxent computer program for modelling species geographic distributions (v.3.3.3k [59]) was employed. Maxent is a general-purpose program that generates inferences from incomplete information, estimating a target probability distribution by finding the probability distribution of maximum entropy, subject to a set of constraints that represent the incomplete information concerning the reported distribution. Maxent is a machine learning modelling method, which has recently attracted attention because of its favorable performance in comparison to other modelling methods [60]. All models were produced with default parameter settings that are suited to a range of presence-only datasets [61]. The documented distribution of the species was obtained from [62] and records were updated with the most recent taxonomic overview of taxa [63]. We downloaded a set of monthly values of temperature and the Normalized Derived Vegetation Index (NDVI), at 0.1° spatial resolution (NEO-NASA web server, March 2000 to September 2011). NDVI was used as a proxy for humidity since this factor has been shown to be important for the survival of ticks. Reliability of the model was determined using the area under the ROC curve (AUC [64]). Models with values above 0.75 are considered potentially useful [65].
Results
The 115 adult A. hebraeum individuals revealed 22 haplotypes for the mitochondrial COI gene fragment (Genbank Accession numbers: JX049245 - JX049266; Table 1; Figure 2). More than 50% of the haplotypes were unique to sampling localities resulting in a haplotype diversity (h) of 0.66 (Table 1). Marked differences in haplotypic diversity were detected among sampling sites and ranged from 0.20 in Spring Valley to 0.82 in a population from Strydpoort. The overall nucleotide diversity (π) was 0.002 (Table 1) and a similar low value was also reflected in the average sequence divergence value between the haplotypes (0.005%; ±0.0025). For H. rufipes, a total of 162 individuals were analyzed and 64 mtDNA COI haplotypes were identified (Genbank Accession numbers: JX049267 - JX049330; Table 1; Figure 3). The overall haplotype diversity (h) of 0.96 was much higher than that detected for A. hebraeum and ranged between 0.51 in a population from Stutterheim to 0.98 for the Garissa sampling site. Although low, the overall nucleotide diversity (π) of 0.009 was higher than that found for A. hebraeum and ranged between 0.001 at Stutterheim and 0.017 at Changara (Table 1).
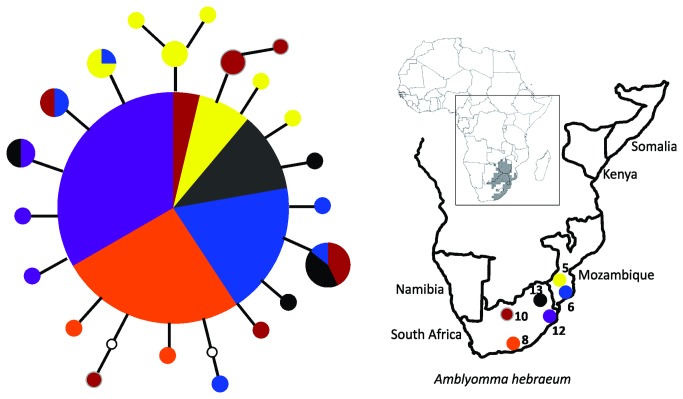
Figure 2. Haplotype network of the 22 mitochondrial COI haplotypes detected from A. hebraeum.
The size of the circles corresponds to the number of individuals characterized by the specific haplotype. Each line separating haplotypes represents one mutational step and missing/intermediate haplotypes are shown by an white circle. Each sampled haplotype is color coded according to the sampling sites indicated on the map inset. Distribution map for A. hebraeum taken from [41,81].
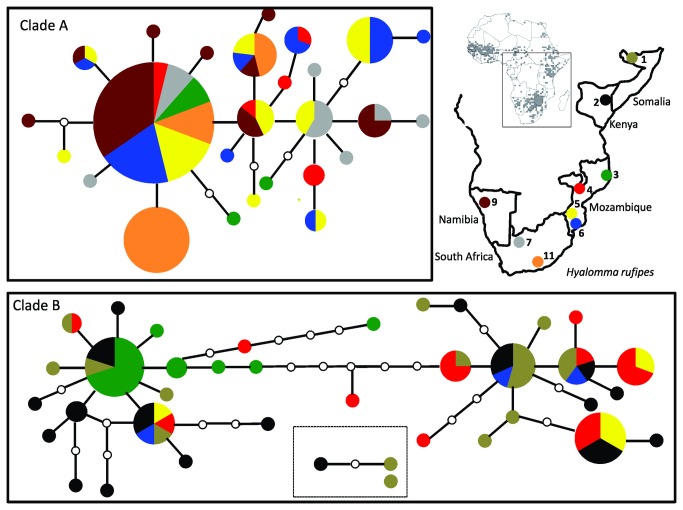
Figure 3. Haplotype network of the 64 mitochondrial COI haplotypes detected for H. rufipes.
The southern and northern clades are indicated by solid boxes and the three unconnected haplotypes from north Africa is indicated in the form of a dotted box. The size of the circles corresponds to the number of individuals characterized by the specific haplotype. Each line separating haplotypes represents one mutational step and missing/intermediate haplotypes are shown by an white circle. Each sampled haplotype is color coded according to the sampling sites indicated on the map inset. Distribution map for H. rufipes taken from [41,81].
A star-shaped statistical parsimony network for A. hebraeum was obtained reflecting a complete lack of geographic population structure, and a recent common ancestry for nearly all the maternal gene lineages (Figure 2). The most common haplotype was present in all sampled sites, representing 57% of the total number of individuals. In sharp contrast, the same analyses for H. rufipes resulted in two divergent statistically unconnected haplogroups (Clade A and B; Figure 3). Clade A will be referred to as Southern group and amongst others contains all the Namibian and South African individuals sampled at Palmwag, Van Zylsrus, and Stutterheim. Clade B will be referred to as the Northern group and contains haplotypes predominantly sampled at the Mozambique locations and further north in Africa. Interestingly, all the sample sites in Mozambique had some level of haplotype sharing between the two clades with the eastern Changara, Massangena and Chivane sampling sites showing larger amounts of admixture than the western Mussuril sampling site. No haplotypes belonging to Clade A were detected in Somalia and Kenya, and in fact, three haplotypes originating from Somalia and Kenya could not be connected to any of the other H. rufipes haplotypes detected in our study (indicated by the dotted line in Figure 3). The average uncorrected sequence divergence between the two clades is 3.17% (± 0.38%), with sequence diversity values of 0.70% (± 0.38%) within the Southern and 1.17% (±0.64%) within the Northern clade. Within the Southern clade there is no evidence for any geographic substructure, but within the Northern assemblage, two subgroups are present separated by at least 6 mutational steps from each other. The Bayesian analyses of population structure confirms the presence of a single lineage within A. hebraeum and two main assemblages within H. rufipes (K= 2). The analysis clusters one of the Mozambique sampling sites (Changara) within the Southern assemblage.
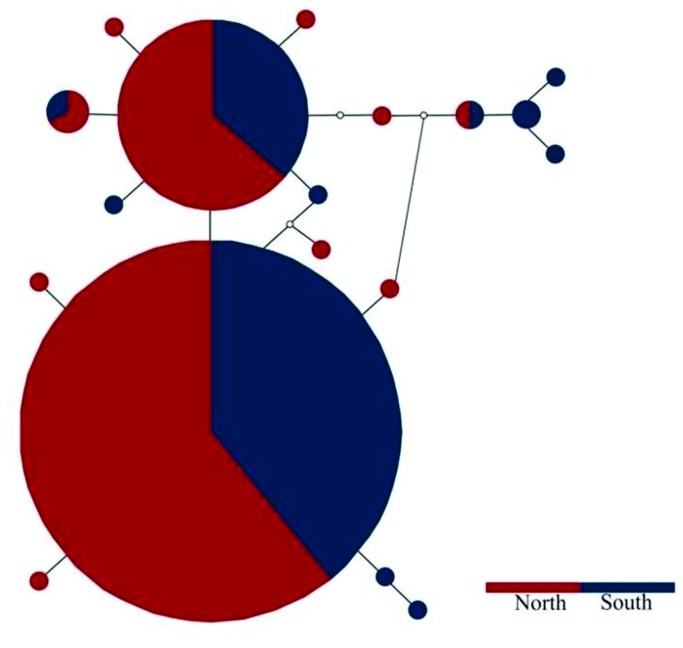
Because of the low level of variation among A. hebraeum haplotypes, and the complete absence of mtDNA geographic structure for this species, only H. rufipes was targeted for nuclear ITS-2 sequencing. The 64 H. rufipes haplotypes retrieved by the mitochondrial COI analyses were amplified. Three individuals failed to amplify resulting in a total of 122 nuclear alleles available for the analyses. Nineteen nuclear haplotypes were retrieved in DnaSP v 5 (Genbank Accession numbers, JX049226 - JX049244) with an overall haplotype diversity (h) of 0.618 (±0.039) and an overall nucleotide diversity of 0.005 (±0.003). Contrary to the mitochondrial DNA analysis, the statistical parsimony network resulted in one haplogroup for all the alleles with no evidence of geographic structure (Figure 4).
Figure 4. Haplotype network of the 19 ITS-2 haplotypes detected for H. rufipes.
The size of the circles corresponds with the number of individuals represented by the haplotype. Each line separating haplotypes represents one mutational step and missing/intermediate haplotypes are shown by an white circle. For visual comparison between data sets, localities were color coded based on the outcome of the mtDNA analyses (southern clade = red; northern clade = blue) and these are also similarly indicated on the map inset.
AMOVA analysis for A. hebraeum, detected shallow but significant genetic structure, with an overall Φst value of 0.196 (p < 0.05). Contrary to the expectations based on the star-shaped haplotype network, pairwise Φst comparisons between geographic localities were significant in most instances supporting some degree of population differentiation across the range (Table 2). A similar suite of geographic population differentiation analyses for the more specialized H. rufipes also revealed significant genetic structure among most sampling sites (Table 3), and a higher degree of differentiation overall (Φst value of 0.43; p < 0.05). It is possible that the higher level of differentiation can simply be due to differences in sampling extent. When the analyses were performed partitioning the individuals in Southern and Northern assemblages (as defined by the BAPS analysis), 66% of the variation can still be ascribed to among populations within clades (p < 0.05). AMOVA analysis of the ITS-2 data was performed by specifying the populations according to the mtDNA groups and this resulted in a low (0.001) and non significant Φst value between groups.
Table 2. Population pairwise Φst matrix of A. hebraeum among sampling regions generated by AMOVA.
| Massangena | Chivavane | Vosloosnek | Strydpoort | Mahlakwe | Spring Valley | |
|---|---|---|---|---|---|---|
| Massangena | 0.0 | |||||
| Chivavane | 0.164 | 0.0 | ||||
| Vosloosnek | 0.055 | 0.230 | 0.0 | |||
| Strydpoort | 0.199 | 0.317 | 0.074 | 0.0 | ||
| Mahlakwe | 0.032 | 0.216 | 0.148 | 0.325 | 0.0 | |
| Spring Valley | 0.032 | 0.222 | 0.165 | 0.333 | 0.0 | 0.0 |
Significance values are in bold (p < 0.05). Locality names correspond to Table 1.
Table 3. Population pairwise Φst matrix of H. rufipes among sampling regions generated by AMOVA.
| Hargeisan | Garissa | Mussuril | Changara | Massangena | Chivavane | Stutterheim | Van Zylsrus | Palmwag | |
|---|---|---|---|---|---|---|---|---|---|
| Hargeisan | 0.0 | ||||||||
| Garissa | 0.0 | 0.0 | |||||||
| Mussuril | 0.076 | 0.067 | 0.0 | ||||||
| Changara | 0.121 | 0.195 | 0.104 | 0.0 | |||||
| Massangena | 0.532 | 0.613 | 0.493 | 0.228 | 0.0 | ||||
| Chivavane | 0.408 | 0.492 | 0.365 | 0.106 | 0.0 | 0.0 | |||
| Stutterheim | 0.735 | 0.807 | 0.714 | 0.489 | 0.231 | 0.291 | 0.0 | ||
| Van Zylsrus | 0.642 | 0.737 | 0.615 | 0.350 | 0.021 | 0.099 | 0.455 | 0.0 | |
| Palmwag | 0.638 | 0.716 | 0.607 | 0.353 | 0.011 | 0.094 | 0.217 | 0.0 | 0.0 |
Significance values are in bold (p < 0.05). Locality names correspond to Table 1.
The Mantel test (Mantel 1967) for the mtDNA data showed no relationship between genetic and geographic distances for A. hebraeum (r = 0.0, p = 0.6) and a similar scenario existed when each clade of H. rufipes was analyzed separately (Northern group: r = 0.25, p = 0.2; Southern group: r = 0.27, p = 0.1). For both A. hebraeum and H. rufipes Fu’s Fs was negative and significant (Table 1) and subsequent mismatch analysis for A. hebraeum revealed a unimodal distribution of pairwise differences that is consistent with a recent population expansion model (SSD = 0.002, p = 0.2; Table 1). Likewise, when the two H. rufipes assemblages were analyzed individually, each one produced a unimodal distribution of pairwise differences, and these were not significantly different from a population expansion model (Southern clade: SSD= 0.032, p= 0.4; Northern clade: SSD= 0.001, p= 0.6). To compare the potential effect of the demographic history on diversity estimates of the two tick species, the entire mtDNA data sets for the two species were used respectively to calculate the time since expansion for each. Given τ = 1.056 for A. hebraeum and τ = 0.486 for H. rufipes the time since expansion is 70 400 years ago for A. hebraeum (approximately 1 year of generation time [66]) and 64 800 years ago for H. rufipes (6 months of generation time [67]).
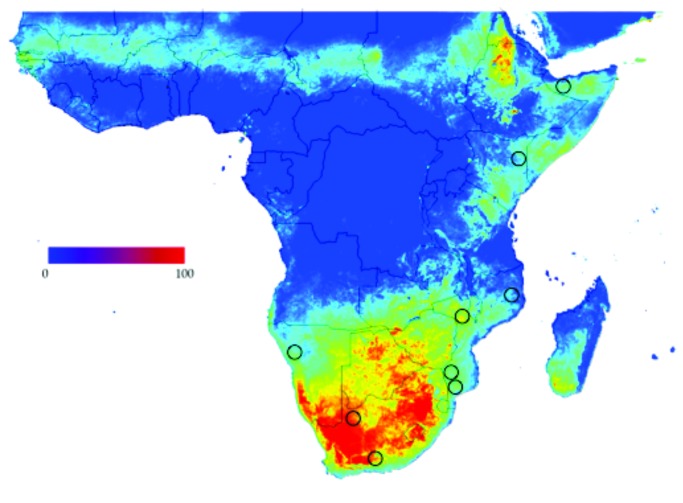
H. rufipes shows large areas of high climate suitability in Africa (Figure 5). The overall AUC for the best model is approximately 0.8. Areas of rain forests in central Africa and the Sahelian and sub-Saharian zones are not regarded as suitable climates for the tick. Pertinent to the focus of our study, however, the two H. rufipes clades are not separated by a strong zone of “non suitable” habitat, suggesting that some alternative factor plays a role in the disruption of mtDNA gene flow in the southern part of the species range.
Figure 5. Climate suitability of H. rufipes in Africa based on temperature and the Normalized Derived Vegetation Index (NDVI), at 0.1° spatial resolution.
Suitability are indicated in a sliding window where red are most suitable and blue are least suitable. Sampling localities are indicated by black circles.
Discussion
The broad-scale phylogeographic patterns obtained for A. hebraeum and H. rufipes is markedly contrasting. The absence of any geographic structure in the host generalist, A. hebraeum, suggests a single geographic/taxonomic unit while the strong mtDNA genetic partitioning in the host specialist, H. rufipes, provides new evidence for allopatric evolutionary trajectories. The latter is supported by the complete mtDNA reciprocal monophyly of the two H. rufipes clades (Figure 2), coupled to a relatively narrow zone of overlap in the south east of Mozambique (predominantly at localities 4, 5 and 6; Figure 1). The lack of noticeable morphological differences among individuals belonging to the two mtDNA clades (Horak pers observation), and the lack of resolution at the more conservative nuclear ITS-2 level (Figure 4), could reflect an incipient speciation processes (with the retention of ancestral polymorphisms [68]). Our study, however, also suggests that a third H. rufipes lineage may exist in North Africa (3 individuals from Kenya and Somalia could not be linked to the geographically proximate Northern clade; Figure 3). More intensive sampling from West and North Africa will be needed to reach any firm taxonomic conclusions, especially in the light of the report that H. rufipes can hybridize with H. truncatum and H. dromedarii in this region [43].
The comparative phylogeographic patterns obtained in this study are useful towards gaining greater insights into the factors influencing the dispersal of ticks. A striking difference between the two species involves haplotypic and nucleotide diversity estimates (A. hebraeum: h = 0.66; π= 0.002; H. rufipes: h = 0.96, π= 0.009; Table 1). The differences in genetic diversity between the two lineages are also well illustrated when only the region of geographic overlap between the two species is considered (see structured complex haplotype network for clade A of H. rufipes versus the starlike haplotype network of A. hebraeum, cf. Figures 2 and 3). Numerous factors can be advanced as contributing towards this discrepancy. It is for example possible that differences in the evolutionary rate between the two species exist and or different selection pressures operate on the mtDNA lineages [69–71]. It can, however, also be argued that these results are simply due to the larger geographic range occupied by H. rufipes, but in the absence of significant isolation by distance (also see 72), this seems not to be the case. A more recent founder event for A. hebraeum can also result in low diversity values (also see 73), but the estimated times of population expansion for both species are roughly similar (70 400 years ago for A. hebraeum and 64 800 years ago for H. rufipes). We argue that the more pronounced genetic diversity and geographic structure obtained for H. rufipes is rather a result of the restrictions in their abilities to disperse (two host specialist needing wildlife to complete its life cycle) while the less structured pattern obtained in A. hebraeum supports a higher level of dispersal among geographic sampling sites (three-host generalist which can complete its entire life cycle on the same host).
Despite the marked differences in genetic diversity estimates, individual populations of both species exhibit a fairly high degree of differentiation among sampling sites (Tables 2 and 3; also see 72,74–77). This finding is counterintuitive given the wide variety of highly mobile hosts that can facilitate the dispersal of adult ticks of both species [32–38,78]. In addition, domestic cattle are frequently moved over large distances for anthropogenic reasons [31,76,79]. Furthermore, the immature stages of both species are often reported on birds [34,37], of which several species that may be infested with H. rufipes are migratory [36,38]. Host movement is thus not the main contributing factor driving the differentiation among sampling sites. It seems more plausible to suggest that the perceived pattern is rather due to capacity of the immature stages of the ticks to survive off the host in the various microhabitats [80–82]. Since the engorged female ticks detach from their hosts, and lay eggs in a sheltered environment, the availability of suitable hosts for the immature stages seems crucial towards ensuring the completion of the life cycle. Indeed, the structure obtained in the more specialized two-host tick, H. rufipes (which is dependent on wildlife to complete its life cycle [83],; and is more sensitive to desiccation in the environment [84],) is more pronounced than that found in the more habitat tolerant generalist A. hebraeum (where all three life stages can utilize the same domestic or wild host to complete its life cycle [32]).
If the physical environment, and particularly the availability of suitable hosts for the immature stages, play an important role in the phylogeographic structure of these two tick species, it is interesting to speculate on the reason/s for the two distinct mtDNA genetic clades found in H. rufipes (Figure 2). The observation that the co-distributed species, A. hebraeum, does not show a similar genetic break would support the idea that the two H. rufipes assemblages are probably not the result of a strong abiotic isolating event in the region (vicariance; also see 85). The projected range of spatial distribution of H. rufipes (based on temperature and the Normalized Derived Vegetation Index) suggest that the two genetic clades cannot be explained by unsuitable climate. Local host associations for different hare species, however, has been documented as an important factor for the successful completion of the life cycle of certain tick species [10] and even if suitable hosts are available, larvae well-adapted to harsh conditions can die from desiccation and starvation if a host is not found in time [86]. Since A. hebraeum seems to be more robust in withstanding harsh environmental conditions [84], and juveniles can survive on adult hosts also, the key to the difference in phylogeographic structure is probably in the availability of suitable hosts for the immature stages of H. rufipes.
At first site, the availability of a suitable host to the juvenile stages of H. rufipes does not seem to provide a plausible for obtaining two genetic assemblages in southern Africa. Hares acting as hosts for juvenile H. rufipes are abundant within the region where the genetic break has been observed (Kruger Park Region in South Africa and southern part of Mozambique). We thus propose that the structure could have been facilitated by a secondary enigmatic complexity related to competition between different tick species [37,87,88]. In the case of A. hebraeum its geographic distribution seems to be limited because of interspecific competition with A. variegatum [41,82,89]. For H. rufipes, the immature stages have to compete with H. truncatum for the predilection attachment site around the neck of hares [90]. This competition appears to be more intense in the western regions of South Africa [35,90,91]. On the other hand, in the south-eastern region of South Africa, H. rufipes is virtually exclusively present on hares as hosts for their immature stages [90]. However, in the Kruger National Park (bordering Mozambique) there is a complete absence of immature stages of H. rufipes larvae on scrub hares and in this this region, they are replaced by H. truncatum [35,90,92]. Given that the Kruger National Park is situated parapatrically to the zone of contact between the two H. rufipes assemblages, it is thus plausible to speculate that competitive habitat exclusion on hosts for immature stages may severely limit gene exchange across this region, and thereby play a further role in the genetic differentiation between the two H. rufipes assemblages.
Acknowledgments
Anne Ropiquet is thanked for providing tick specific COI primer sequences.
Funding Statement
The National Research Foundation (NRF), South African Biosystematics Initiative (SABI) and Stellenbosch University are thanked for financial support. There was a South African National Research Foundation grant to Conrad Matthee. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Labruna MB, Naranjo V, Mangold AJ, Thompson C, Estrada-Peña A et al. (2009) Allopatric speciation in ticks: genetic and reproductive divergence between geographic strains of Rhipicephalus (Boophilus) microplus . BMC Evol Biol 9: 46. doi: 10.1186/1471-2148-9-46. PubMed: 19243585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Poulin R, Krasnov BR, Mouillot D (2011) Host specificity in phylogenetic and geographic space. Trends Parasitol 27: 355-361. doi: 10.1016/j.pt.2011.05.003. PubMed: 21680245. [DOI] [PubMed] [Google Scholar]
- 3. Morand S, Krasnov BR (2010) The Biogeography of Host-Parasite Interactions. New-York: Oxford University Press. [Google Scholar]
- 4. Barker SC, Murrell A (2008) Systematics and evolution of ticks with a list of valid genus and species names. In Nuttall P, Bowman AS. Ticks: biology, disease and control. Cambridge University Press; pp. 1-39. [Google Scholar]
- 5. Guglielmone AA, Robbins RG, Apanaskevich DA, Petney TN, Estrada-Peña A et al. (2010) The Argas idae, Ixodidae and Nuttalliellidae (Acari: Ixodida) of the world: a list of valid species names. Zootaxa 2528: 1-28. [Google Scholar]
- 6. De Meeûs T, Renaud F (2002) Parasites within the new phylogeny of eukaryotes. Trends Parasitol 18: 247-251. doi: 10.1016/S1471-4922(02)02269-9. PubMed: 12036736. [DOI] [PubMed] [Google Scholar]
- 7. Krakowetz CN, Dergousoff SJ, Chilton NB (2010) Genetic variation in the mitochondrial 16S rRNA gene of the American dog tick, Dermacentor variabilis (Acari: Ixodidae). J Vector Ecol 35: 163-173. doi: 10.1111/j.1948-7134.2010.00073.x. PubMed: 20618663. [DOI] [PubMed] [Google Scholar]
- 8. Barker SC, Murrell A (2002) Phylogeny, evolution and historical zoogeography of ticks: a review of recent progress. Exp Appl Acarol 28: 55-68. doi: 10.1023/A:1025333830086. PubMed: 14570116. [DOI] [PubMed] [Google Scholar]
- 9. Kempf F, Boulinier T, De Meeûs T, Arnathau C, McCoy KD (2009) Recent evolution of host-associated divergence in the seabird tick Ixodes uriae . Mol Ecol 18: 4450-4462. doi: 10.1111/j.1365-294X.2009.04356.x. PubMed: 19793353. [DOI] [PubMed] [Google Scholar]
- 10. Kempf F, McCoy KD, De Meeûs T (2010) Wahlund effects and sex-biased dispersal in Ixodes ricinus, the European vector of Lyme borreliosis: New tools for old data. Infect Genet Evol 10: 989-997. doi: 10.1016/j.meegid.2010.06.003. PubMed: 20601167. [DOI] [PubMed] [Google Scholar]
- 11. Rahman MH (1984) Effects of host-immunity on the life cycle of Hyalomma rufipes Koch, 1844 (Ixodoidea: Ixodidae). Folia Parasitol 31: 63-67. PubMed: 6714847. [PubMed] [Google Scholar]
- 12. Kain DE, Sperling FA, Daly HV, Lane RS (1999) Mitochondrial DNA sequence variation in Ixodes pacificus (Acari: Ixodidae). Heredity 83: 378-386. doi: 10.1038/sj.hdy.6886110. PubMed: 10583539. [DOI] [PubMed] [Google Scholar]
- 13. Gylfe A, Yabuki M, Drotz M, Bergström S, Fukunaga M et al. (2001) Phylogeographic relationships of Ixodes uriae (Acari: Ixodidae) and their significance to transequatorial dispersal of Borrelia garinii . Hereditas 134: 195-199. PubMed: 11833281. [DOI] [PubMed] [Google Scholar]
- 14. Song S, Shao R, Atwell R, Barker S, Vankan D (2011) Phylogenetic and phylogeographic relationships in Ixodes holocyclus and Ixodes cornuatus (Acari: Ixodidae) inferred from COX1 and ITS2 sequences. Int J Parasitol 41: 871-880. doi: 10.1016/j.ijpara.2011.03.008. PubMed: 21540032. [DOI] [PubMed] [Google Scholar]
- 15. Noureddine R, Chauvin A, Plantard O (2011) Lack of genetic structure among Eurasian populations of the tick Ixodes ricinus contrasts with marked divergence from north-African populations. Int J Parasitol 41: 183-192. doi: 10.1016/j.ijpara.2010.08.010. PubMed: 20946897. [DOI] [PubMed] [Google Scholar]
- 16. Porretta D, Mastrantonio V, Mona S, Epis S, Montagna M, Sassera D, Bandi C, Urbanelli S (2013) The integration of multiple independent data reveals an unusual response to Pleistocene climatic changes in the hard tick Ixodes ricinus . Mol Ecol 22: 1666-1682. doi: 10.1111/mec.12203. PubMed: 23398505. [DOI] [PubMed] [Google Scholar]
- 17. Norval RAI, Horak IG (2004) Vectors: ticks. In Coetzer JAW, Tustin RC. Infection diseases of livestock. Cape Town: Oxford University Press; pp. 3-42. [Google Scholar]
- 18. Criscione CD, Poulin R, Blouin MS (2005) Molecular ecology of parasites: elucidating ecological and microevolutionary processes. Mol Ecol 14: 2247-2257. doi: 10.1111/j.1365-294X.2005.02587.x. PubMed: 15969711. [DOI] [PubMed] [Google Scholar]
- 19. Andrew HR, Norval RAI (1989) The carrier status of sheep, cattle and African buffalo recovered from heartwater. Vet Parasitol 34: 261-266. doi: 10.1016/0304-4017(89)90056-3. PubMed: 2617830. [DOI] [PubMed] [Google Scholar]
- 20. Deem SL (1998) A review of heartwater and the threat of introduction of Cowdria ruminantium and Amblyomma spp. ticks to the American mainland. J Zoo Wildlife Med 29: 109-113. [PubMed] [Google Scholar]
- 21. Lawrence JA (1979) The differential diagnosis of the bovine theilerias of Southern Africa. J SA Vet Assoc 50: 311-313. PubMed: 122000. [PubMed] [Google Scholar]
- 22. De Vos AJ, Roos JA (1981) Observations on the transmission of Theileria mutans in South Africa. Onderstepoort J Vet Res 48: 1-6. PubMed: 6792579. [PubMed] [Google Scholar]
- 23. Kelly PJ, Beati L, Mason PR, Matthewman LA, Roux V, Raoult D (1996) Rickettsia africae sp. nov., the etioloical agent of African tick-bite fever. Int J Syst Bacteriol 46: 611-614. doi: 10.1099/00207713-46-2-611. PubMed: 8934912. [DOI] [PubMed] [Google Scholar]
- 24. Kelly PJ (2001) Amblyomma hebraeum is a vector of Rickettsia africae and not R. conorii . J SA Vet Assoc 72: 182. [DOI] [PubMed] [Google Scholar]
- 25. Rechav Y, Zeederberg ME, Zeller DA (1987) Dynamics of African tick (Acari: Ixodoidea) populations in a natural Crimean-Congo Hemorrhagic fever focus. J Med Entomol 24: 575-583. PubMed: 3669030. [DOI] [PubMed] [Google Scholar]
- 26. Horak IG, Fourie LJ, Heyne H, Walker JB (2002) Ixodid ticks feeding on humans in South Africa: with notes on preferred hosts, geographic distribution, seasonal occurrence and transmission of pathogens. Exp Appl Acarol 27: 113-136. doi: 10.1023/A:1021587001198. PubMed: 12593517. [DOI] [PubMed] [Google Scholar]
- 27. Mtshali MS, De la Fuente J, Ruybal P, Kocan KM, Vicente J, Mbati PA, Shkap V, Blouin EF, Mohale NE, Moloi TP, Spickett AM, Latif AA (2007) Prevalence and genetic diversity of Anaplasma marginale strains in cattle in South Africa. Zoonoses. Public Health 54: 23-30. [DOI] [PubMed] [Google Scholar]
- 28. Uilenberg G (2006) Babesia – A historical overview. Vet Parasitol 138: 3-10. doi: 10.1016/j.vetpar.2006.01.035. PubMed: 16513280. [DOI] [PubMed] [Google Scholar]
- 29. Parola P, Inokuma H, Camicas JL, Brouqui P, Raoult D (2001) Detection and identification of spotted fever group Rickettsiae and Ehrlichiae in African ticks. Emerg Infect Diss 7: 1014-1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Horak IG (1982) Parasites of domestic and wild animals in South Africa. XV. The seasonal prevalence of ectoparasites on impala and cattle in the Northern Transvaal. Onderstepoort J Vet Res 49: 85-93. PubMed: 7177586. [PubMed] [Google Scholar]
- 31. Rechav Y, Dauth J, Els DA (1990) Resistance of Brahman and Simmentaler cattle to southern African ticks. Onderstepoort J Vet Res 57: 7-12. PubMed: 2339000. [PubMed] [Google Scholar]
- 32. Horak IG, Golezardy H, Uys AC (2007) Ticks associated with the three largest wild ruminant species in southern Africa. Onderstepoort J Vet Res 74: 231-242. PubMed: 17933365. [DOI] [PubMed] [Google Scholar]
- 33. Walker JB, Schulz KCA (1984) Records of the bont tick, Amblyomma hebraeum, from the angulate tortoise, Chersina angulata and the leopard tortoise, Geochelone pardalis . Onderstepoort J Vet Res 51: 171-173. PubMed: 6533508. [PubMed] [Google Scholar]
- 34. Horak IG , Fourie LJ, Novellie PA, Williams EJ (1991) Parasites of domestic and wild animals in South Africa. XXVI. The mosaic of ixodid tick infestations on birds and mammals in the Mountain Zebra National Park. Onderstepoort J Vet Res 58: 125-136. [PubMed] [Google Scholar]
- 35. Horak IG, Spickett AM, Braack LEO, Penzhorn BL (1993) Parasites of domestic and wild animals in South Africa. XXXII. Ixodid ticks on scrub hares in the Transvaal. Onderstepoort J Vet Res 60: 163-174. PubMed: 7970571. [PubMed] [Google Scholar]
- 36. Van Niekerk DJ, Fourie LJ, Horak IG (2006) Birds as hosts of immature ixodid ticks in Free State province, South Africa. Onderstepoort J Vet Res 73: 123-130. PubMed: 16958263. [DOI] [PubMed] [Google Scholar]
- 37. Uys AC, Horak IG (2005) Ticks on crested francolins, Francolinus sephaena, and on the vegetation on a farm in Limpopo Province, South Africa. Onderstepoort J Vet Res 72: 339-343. PubMed: 16562738. [PubMed] [Google Scholar]
- 38. Hasle G, Horak IG, Grieve G, Leinaas HP, Clarke F (2009) Ticks collected from birds in the northern provinces of South Africa, 2004–2006. Onderstepoort J Vet Res 76: 167-175. PubMed: 20698437. [DOI] [PubMed] [Google Scholar]
- 39. Horak IG, MacIvor KMDeF (1987) The scrub hare, a reliable indicator of the presence of Hyalomma ticks in the Cape Province. J S Afr Vet Assoc 58: 15-19. [PubMed] [Google Scholar]
- 40. Kuhnert F, Diehl PA, Guerin PM (1995) The life-cycle of the bont tick Amblyomma hebraeum in vitro. Int J Parasitol 25: 887-896. doi: 10.1016/0020-7519(95)00009-Q. PubMed: 8550288. [DOI] [PubMed] [Google Scholar]
- 41. Walker AR, Bouattour A, Camicas J-L, Estrada Peña A, Horak IG et al. (2003) Ticks of Domestic Animals in Africa: a Guide to Identification of Species. UK: Bioscience Reports. [Google Scholar]
- 42. Poulin R, Krasnov BR, Mouillot D (2011) Host specificity in phylogenetic and geographic space. Trends Parasitol 27: 355-361. doi: 10.1016/j.pt.2011.05.003. PubMed: 21680245. [DOI] [PubMed] [Google Scholar]
- 43. Rees DJ, Dioli M, Kirkendall LR (2003) Molecules and morphology: evidence for cryptic hybridization in African Hyalomma (Acari: Ixodidae). Mol Phylogenet Evol 27: 131-142. doi: 10.1016/S1055-7903(02)00374-3. PubMed: 12679078. [DOI] [PubMed] [Google Scholar]
- 44. Hall T (2005) BioEdit v7.0.5. Available: http://www.mbio.ncsu.edu/bioedit/page2.html. Accessed 8 April 2010.
- 45. Librado P, Rozas J (2009) DnaSP v5.10: A software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25: 1451-1452. doi: 10.1093/bioinformatics/btp187. PubMed: 19346325. [DOI] [PubMed] [Google Scholar]
- 46. Stephens M, Donnelly P (2003) A comparison of bayesian methods for haplotype reconstruction from population genotype data. Am J Hum Genet 73: 1162-1169. doi: 10.1086/379378. PubMed: 14574645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Excoffier L, Lischer HEL (2010) Arlequin suite ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour 10: 564-567. doi: 10.1111/j.1755-0998.2010.02847.x. PubMed: 21565059. [DOI] [PubMed] [Google Scholar]
- 48. Swofford DL (2003) PAUP*. Phylogenetic Analysis Using Parsimony (* and Other Methods), version 4. Sunderland, MA: Sinauer Associates. [Google Scholar]
- 49. Clement M, Posada D, Crandall KA (2000) TCS: a computer program to estimate gene genealogies. Mol Ecol 9: 1657-1660. doi: 10.1046/j.1365-294x.2000.01020.x. PubMed: 11050560. [DOI] [PubMed] [Google Scholar]
- 50. Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 131: 479-491. PubMed: 1644282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Corander J, Marttinen P, Sirén J, Tang J (2008) Enhanced Bayesian modeling in BAPS software for learning genetic structures of populations. BMC Bioinformatics 9: 539. doi: 10.1186/1471-2105-9-539. PubMed: 19087322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Manly BFJ (1991) Randomization and Monte Carlo Methods in Biology. London: Chapman & Hall. [Google Scholar]
- 53. Fu YX (1997) Statistical tests of neutrality of mutations against population growth, hitchhiking and background selection. Genetics 147: 915-925. PubMed: 9335623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Slatkin M, Hudson RR (1991) Pairwise comparisons of mitochondrial DNA sequences in stable and exponentially growing populations. Genetics 129: 555-562. PubMed: 1743491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Rogers AR, Harpending H (1992) Population growth makes waves in the distribution of pairwise genetic differences. Mol Biol Evol 9: 552-569. PubMed: 1316531. [DOI] [PubMed] [Google Scholar]
- 56. Schneider S, Excoffier L (1999) Estimation of demographic parameters from the distribution of pairwise differences when the mutation rates vary among sites: Application to human mitochondrial DNA. Genetics 152: 1079-1089. PubMed: 10388826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Quek SP, Davies SJ, Itino T, Pierce NE (2004) Codiversification in an ant-plant mutualism: stem texture and the evolution of host use in Crematogaster (Formicidae: Myrmicinae) inhabitants of Macaranga (Euphorbiaceae). Evolution 58: 554-570. doi: 10.1111/j.0014-3820.2004.tb01678.x. PubMed: 15119439. [DOI] [PubMed] [Google Scholar]
- 58. Lohman DJ, Peggie D, Pierce NE, Meier R (2008) Phylogeography and genetic diversity of a widespread Old World butterfly, Lampides boeticus (Lepidoptera: Lycaenidae). BMC Evol Biol 8: 301. doi: 10.1186/1471-2148-8-301. PubMed: 18973689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Phillips SJ, Anderson RP, Schapire R (2006) Maximum entropy modeling of species geographic distribution. Ecol Modell 190: 231-259. doi: 10.1016/j.ecolmodel.2005.03.026. [DOI] [Google Scholar]
- 60. Elith J, Graham CH, Anderson RP, Dudík M, Ferrier S et al. (2006) Novel methods improve prediction of species’ distributions from occurrence data. Ecography 29: 129-151. doi: 10.1111/j.2006.0906-7590.04596.x. [DOI] [Google Scholar]
- 61. Phillips SJ, Anderson RP, Schapire R (2006) Maximum entropy modeling of species geographic distribution. Ecol Modell 190: 231-259. doi: 10.1016/j.ecolmodel.2005.03.026. [DOI] [Google Scholar]
- 62. Cumming GS (2002) Comparing climate and vegetation as limiting factors for species ranges of African ticks. Ecology 83: 255-268. doi:10.1890/0012-9658(2002)083[0255:CCAVAL]2.0.CO;2. [Google Scholar]
- 63. Apanaskevich D, Horak I (2006) The genus Hyalomma Koch, 1844. Reinstatement of Hyalomma (Euhyalomma) glabrum Delpy, 1949 (Acari: Ixodidae) as a valid species with a redescription of the adults, the first description of its immature stages and notes on its biology. Onderstepoort J Vet Res Res 73: 1-12. [DOI] [PubMed] [Google Scholar]
- 64. Fielding AH, Bell JF (1997) A review of methods for the assessment of prediction errors in conservation presence/absence models. Environ Conserv 24: 38-49. doi: 10.1017/S0376892997000088. [DOI] [Google Scholar]
- 65. Elith J, Burgman MA (2002) Predictions and their validation: rare plants in the Central Highlands, Victoria, Australia. In Scott JM, Heglund PJ, Morrison M, Raphael M, Haufler J. Predicting species occurrences: issues of accuracy and scale. Island Press; pp. 303-314. [Google Scholar]
- 66. Kuhnert F, Diehl PA, Guerin PM (1995) The life-cycle of the bont tick Amblyomma hebraeum in vitro. Int J Parasitol 25: 887-896. doi: 10.1016/0020-7519(95)00009-Q. PubMed: 8550288. [DOI] [PubMed] [Google Scholar]
- 67. Chen Z, Li Y, Liu Z, Yang J, Yin H (2012) The life cycle of Hyalomma rufipes (Acari: Ixodidae) under laboratory conditions. Exp Appl Acarol 56: 85-92. doi: 10.1007/s10493-011-9490-0. PubMed: 21913002. [DOI] [PubMed] [Google Scholar]
- 68. Maddison WP, Knowles LL (2006) Inferring phylogeny despite incomplete lineage sorting. Syst Biol 55: 21-30. doi: 10.1080/10635150500354928. PubMed: 16507521. [DOI] [PubMed] [Google Scholar]
- 69. Rechav Y (1987) Resistance of Brahman and Hereford cattle to African ticks with reference to serum gamma globulin levels and blood composition. Exp Appl Acarol 3: 219-232. doi: 10.1007/BF01270458. PubMed: 2456184. [DOI] [PubMed] [Google Scholar]
- 70. Brown SJ (1988) Characterization of tick antigens inducing host immune resistance. II. Description of rabbit-acquired immunity to Amblyomma americanum ticks and identification of potential tick antigens by Western blot analysis. Vet Parasitol 28: 245-259. doi: 10.1016/0304-4017(88)90112-4. PubMed: 3291384. [DOI] [PubMed] [Google Scholar]
- 71. Mitchell M (1996) Acaricide resistance - back to basics. Trop Anim Health Prod 28: 53S-58S. doi: 10.1007/BF02310700. PubMed: 8809993. [DOI] [PubMed] [Google Scholar]
- 72. Mixson TR, Lydy SL, Dasch GA, Real LA (2006) Inferring the population structure and demographic history of the tick, Amblyomma americanum Linnaeus. J Vector Ecol 31: 181-192. doi:10.3376/1081-1710(2006)31[181:ITPSAD]2.0.CO;2. PubMed: 16859107. [DOI] [PubMed] [Google Scholar]
- 73. Fauvelot C, Bernardi G, Planes S (2003) Reductions in the mitochondrial dna diversity of coral reef fish provide evidence of population bottlenecks resulting from holocene sea-level change. Evolution 57: 1571-1583. doi: 10.1554/02-173. PubMed: 12940362. [DOI] [PubMed] [Google Scholar]
- 74. De Meeûs T, Renaud F (2002) Parasites within the new phylogeny of eukaryotes. Trends Parasitol 18: 247-251. doi: 10.1016/S1471-4922(02)02269-9. PubMed: 12036736. [DOI] [PubMed] [Google Scholar]
- 75. Qiu WG, Dykhuizen DE, Acosta MS, Lu ſ t BJ (2002) Geographic uniformity of the Lyme disease spirochete (Borrelia burgdorferi) and its shared history with tick vector (Ixodes scapularis) in the northeastern United States. Genetics 160: 833-849. PubMed: 11901105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. McCoy KD, Boulinier T, Tirard C, Michalakis Y (2003) Host-dependent genetic structure of parasite populations: differential dispersal of seabird tick host races. Evolution 57: 288-296. doi:10.1554/0014-3820(2003)057[0288:HDGSOP]2.0.CO;2. PubMed: 12683525. [DOI] [PubMed] [Google Scholar]
- 77. Mixson TR, Fang QQ, McLain DK, Oliver JH Jr (2004) Population structure of the blacklegged tick Ixodes scapularis revealed by SSCP data using the mitochondrial Cyt b and the nuclear ITS1 markers. Acta Zool Sin 50: 176-186. [Google Scholar]
- 78. Horak IG, Boomker J, Spickett AM, De Vos V (1992) Parasites of domestic and wild animals in South Africa. XXVII. Ticks on helmeted guineafowls in the Eastern Cape Province and Eastern Transvaal Lowveld. Onderstepoort J Vet Res 58: 137-143. [PubMed] [Google Scholar]
- 79. Fourie LJ, Horak IG (1991) The seasonal activity of adult ixodid ticks on Angora goats in the south-western Orange Free State. J SA Vet Assoc 62: 104-106. [PubMed] [Google Scholar]
- 80. Needham GR, Teel PD (1986) Water balance by ticks between blood meals. In: Sauer JR, Hair JAeditors. Morphology, physiology, and behavioral biology of ticks. Chichester (UK): Ellis Horwood Limited; . pp. 100-151 [Google Scholar]
- 81. Cumming GS (1999) Host distributions do not limit the species ranges of most African ticks (Acari: Ixodida). Entomol Res 89: 303-332. [Google Scholar]
- 82. Estrada-Peña A, Horak IG, Petney T (2008) Climate changes and suitability for the ticks Amblyomma hebraeum and Amblyomma variegatum (Ixodidae) in Zimbabwe (1974-1999). Vet Parasitol 151: 256-267. doi: 10.1016/j.vetpar.2007.11.014. PubMed: 18083309. [DOI] [PubMed] [Google Scholar]
- 83. Apanaskevich DA, Horak IG (2008) The genus Hyalomma Koch, 1844: V. Re-evaluation of the taxonomic rank of taxa comprising the H. (Euhyalomma) marginatum Koch of species (Acari: Ixodidae) with redescription of all parasitic stages and notes on biology. Int J Acarol 43: 13-42. [Google Scholar]
- 84. Fielden LJ, Rechav Y (1996) Survival of six species of African ticks in relation to saturation déficits. Exp Appl Acarol 20: 625-637. doi: 10.1007/BF00053326. PubMed: 9022266. [DOI] [PubMed] [Google Scholar]
- 85. Matthee CA, Flemming AF (2002) Population fragmentation in the southern rock agama, Agama atra: more evidence for vicariance in Southern Africa. Mol Ecol 11: 465-471. doi: 10.1046/j.0962-1083.2001.01458.x. PubMed: 11918781. [DOI] [PubMed] [Google Scholar]
- 86. Anderson JF, Magnarelli LA (2008) Biology of Ticks. Infect Dis Clin North Am 22: 195-215. doi: 10.1016/j.idc.2007.12.006. PubMed: 18452797. [DOI] [PubMed] [Google Scholar]
- 87. Norval RAI, Short NJ (1984) Interspecific competition between Boophilus decoloratus and Boophilus microplus in South Africa. In Griffiths DA, Bowman CE. Acarology. Chichester: Ellis Horwood Limited. [Google Scholar]
- 88. Berkvens DL, Geysen DM, Chaka G, Madder M, Brandt JRA (1998) A survey of the ixodid ticks parasitising cattle in the Eastern Province of Zambia. Med Vet Entomol 12: 234-240. doi: 10.1046/j.1365-2915.1998.00106.x. PubMed: 9737594. [DOI] [PubMed] [Google Scholar]
- 89. Norval RAI (1983) Ticks of ZimbabweVII: The genus Amblyomma. Zimbabwe. Vet J 23: 1-15. [Google Scholar]
- 90. Horak IG, Fourie LJ (1991) Parasites of domestic and wild animals in South Africa. XXIX. Ixodid ticks on hares in the Cape Province and on hares and red rock rabbits in the Orange Free State. Onderstepoort J Vet Res 58: 261-270. PubMed: 1780126. [PubMed] [Google Scholar]
- 91. Horak IG, Spickett AM, Braack LEO, Penzhorn BL, Bagnall RJ, Uys AC (1995) Parasites of domestic and wild animals in South Africa. XXXIII. Ixodid ticks on scrub hares in the north-eastern regions of Northern and Eastern Transvaal and KwaZulu-Natal. Onderstepoort J Vet Res 62: 123-131. PubMed: 8600436. [PubMed] [Google Scholar]
- 92. Horak IG, Boomker J, Spickett AM, De Vos V (1992) Parasites of domestic and wild animals in South Africa. XXX. Ectoparasites of kudus in the eastern Transvaal Lowveld and the eastern Cape Province. Onderstepoort J Vet Res 59: 259-273. PubMed: 1297956. [PubMed] [Google Scholar]