Abstract
Generally, limited research is extended in studying stability and applicational properties of silver nanoparticles (Ag NPs) synthesized by adopting ‘green chemistry’ protocol. In this work, we report on the synthesis of stable Ag NPs using plant-derived materials such as leaf extract of Neem (Azadirachta indica) and biopolymer pectin from apple peel. In addition, the applicational properties of Ag NPs such as surface-enhanced Raman scattering (SERS) and antibacterial efficiencies were also investigated. As-synthesized nanoparticles (NPs) were characterized using various instrumentation techniques. Both the plant materials (leaf extract and biopolymer) favored the synthesis of well-defined NPs capped with biomaterials. The NPs were spherical in shape with an average particle size between 14-27 nm. These bio-NPs exhibited colloidal stability in most of the suspended solutions such as water, electrolyte solutions (NaCl; NaNO3), biological solution (bovine serum albumin), and in different pH solutions (pH 7; 9) for a reasonable time period of 120 hrs. Both the bio-NPs were observed to be SERS active through displaying intrinsic SERS signals of the Raman probe molecule (Nile blue A). The NPs were effective against the Escherichia coli bacterium when tested in nutrient broth and agar medium. Scanning and high-resolution transmission electron microscopy (SEM and HRTEM) images confirmed cellular membrane damage of nanoparticle treated E. coli cells. These environmental friendly template Ag NPs can be used as an antimicrobial agent and also for SERS based analytical applications.
Introduction
Metallic silver nanoparticles (Ag NPs) have gathered much attention due to their unique properties, which depend on their morphology, dimension and colloidal stability. Colloidal stability of the nanoparticles (NPs) in solvent other than water is desired in most of the biological applications and in several other analytical applications [1]. Hence, understanding the stability of Ag NPs in different environment is essential, which likely exhibits the fate of the NPs. Colloidal stability is a function of many factors including the type of capping agent, surrounding environmental conditions like pH, ionic strength and the background electrolyte composition [2,3]. Unstable nanoparticle (NP) aggregates may significantly contribute to dissolution of ions from NPs [4], which increases during particle storage [5]. Such coexistence of the NP and its ionic forms may induce toxic pathway [6,7]. The colloidal stability of the NPs is crucial, which determines their mobility, bioavailability and toxicity in any ecosystem [8,9]. Hence it is essential to synthesis stable NPs, which can minimize ion dissolution and retain its physicochemical properties.
The capping molecules bound to NP surface are the ones that give definite shape, size and stability to the NPs. Of the various synthesis methods, most of the stable NP syntheses have been achieved through wet chemical methods. Due to the growing interest in green chemistry methods in recent years, biological and polymer based synthesis of NPs has gained significant interest [10,11]. However, most of the green chemistry approaches for NP synthesis from sources such as microbial, plant extracts and polymers have stopped at the level of synthesis and basic characterization. Considering the wide application of NPs in various fields, the study on the stability and application of the NPs synthesized by ‘green chemistry’ has become very essential.
In the present work, we have used leaf extract of Neem (Azadirachta indica) and biopolymer pectin from apple peel for synthesis of Ag NPs. Polymers as well as the essential components in plant leaf extract such as proteins and sugars are known to provide stability to the NPs. The colloidal stability of Ag NPs in different electrolyte medium (NaCl and NaNO3), biological medium (bovine serum albumin [BSA]) and in different pH solutions (pH 7 and 9) was determined by measuring the changes in UV-Vis absorbance spectra [1], hydrodynamic diameter (HDD) and zeta potential value. In addition, the NPs were studied for their optical field enhancing properties through surface-enhanced Raman scattering (SERS) of Raman probe molecule Nile blue A (NBA), and antibacterial activity using Escherichia coli as the model organism. E. coli has been a major fecal coliform contaminant and, an indicator organism in water and also E. coli causes major foodborne diseases; hence it has been used as the model organism in the present study.
Materials and Methods
Materials
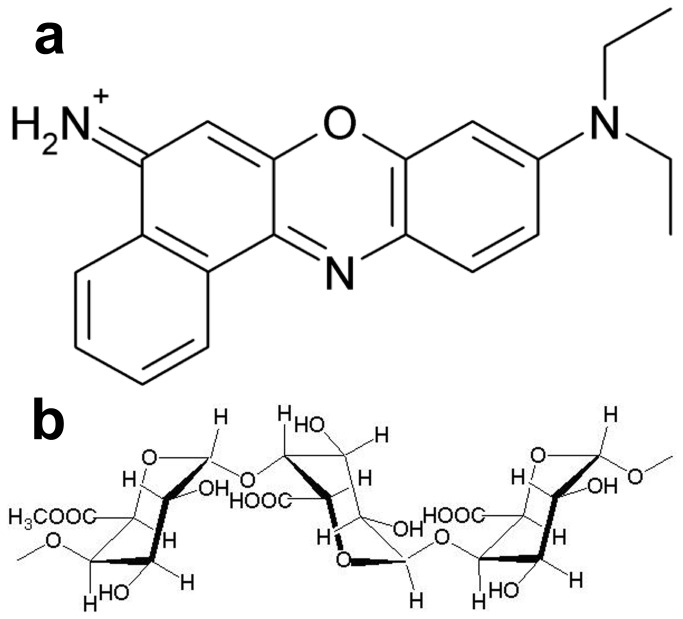
AgNO3, pectin (from apple peel), NBA, NaCl and NaNO3 were purchased from Sigma-Aldrich (Bangalore, India) and used as received. Molecular structure of NBA and pectin are shown in Figure 1. BSA and nutrient broth (NB) were purchased from HiMedia chemicals India Pvt. Ltd (Mumbai, India). Ultra pure water with a resistivity more than 18.0 MΩ (Millipore Milli-Q system) was used for preparation of all aqueous solutions. All the glasswares were cleaned with freshly prepared aqua regia (3:1, HCl:HNO3) and rinsed thoroughly with water. Then they were dry sterilized using hot air oven at 160 °C for 3 hrs, prior to use.
Figure 1. Molecular structure of biopolymer pectin and Raman analyte Nile Blue A.
Structure of (a) Nile blue A and (b) pectin.
Silver Nanoparticles Preparation and Characterization
Preparation of silver nanoparticles using plant leaf extract
Azadirachta indica leaves were collected freshly and washed thoroughly with sterile Milli-Q grade water to remove adsorbed impurities. 5 g of finely chopped leaves were boiled (~ 80 °C) with 50 mL of water for about 10 minutes and then brought to room temperature (~ 27 °C). The filtrate was then separated using Whatman grade I filter paper and stored at 4 °C until further use.
In a typical experiment, 1 mL of 0.1 M aqueous AgNO3 was added to 99 mL of aqueous leaf extract solution (10% leaf extract in final working solution) under gentle stirring. The pH of the working solution was adjusted to 7 using 0.5 M NaOH. The reaction was allowed to proceed under gentle stirring at room temperature for 12 hrs. The NPs were separated by centrifugation at 10,000 × g for 20 minutes at 20 °C and washed thrice with water to remove the impurities. Finally the NPs were filtered and concentrated using centrifugal filter devices (Amicon Ultra- 15 3K Millipore). The Ag NPs were stored at 4 °C before being used for further analysis.
Preparation of silver nanoparticles using biopolymer pectin
Aqueous biopolymeric solution containing 10 g/L of pectin (from apple peel) was prepared by placing the mixture in warm water bath (~ 60 °C) for about 15 min for complete solubilization and then brought down to room temperature. To the biopolymeric solution, aqueous 0.1 M AgNO3 (1 mM final concentration) and NaOH solutions (final concentration 25 mM) were added rapidly under vigorous stirring. After 6 hrs of stirring at room temperature, the NPs were separated and washed thrice with water by centrifugation at 12,000 × g for 25 minutes at 25 °C. Finally the NPs were filtered and concentrated using centrifugal filter devices (Amicon Ultra- 15 3K Millipore). The concentrated stock colloidal NPs were stored at 4 °C.
Characterization of nanoparticles
UV-Vis absorption spectroscopy was performed on a PerkinElmer (LAMBDA 35 Singapore) UV/Vis spectrophotometer operating at 1 nm resolution. The size and morphology of NPs were examined using high-resolution transmission electron microscopy (HRTEM) JEOL JEM 2100 (Japan) and Philips CM20 TEM (Netherlands) operating at 200 kV. The samples were prepared by placing a drop of NPs on a carbon coated copper grid. The chemical analysis of the NPs was performed using energy dispersive X-ray spectroscopy (EDS) incorporated to the TEM. Powder X-ray diffraction (PXRD) measurements of Ag NPs were performed on a PANalytical X-ray diffractometer (XPERT PRO, Netherlands) using monochromatic Cu Kα radiation. Fourier transform infrared (FTIR) analysis was carried out using JASCO FT/IR 4100 (Japan) Fourier transform infrared spectrometer. The samples were recorded with KBr, at a spectral range 4000-400 cm-1, scanning speed of 2 mm sec-1 and resolution at 4 cm-1. DLS and zeta (ζ) potential measurements of NPs were performed with a zetasizer (Malvern Zetasizer Nano ZS90, UK) at room temperature.
Colloidal Stability of Silver Nanoparticles
For all stability experiments, a small volume of NP stock suspension was diluted with respective solutions. The stability of NP was determined in media such as water, NaCl and NaNO3 (electrolyte) solutions of concentrations 50 and 200 mM, 0.1 and 0.25% BSA (biological) solutions, and in pH 7 and 9. A precalculated amount of electrolyte solution or biological solution was added to the NP suspension to obtain the desired concentration making the final suspension volume of 50 mL. For pH stability, a small amount of NP stock solution was dispersed in 50 mL solutions of different pH (pH 7 and 9). The initial measurements were started almost immediately (0.5 hrs), and the changes in the UV-Vis absorbance spectra, HDD and electrophoretic mobility (surface charge) were recorded over 5 days at different time intervals. Small amount of the samples were retracted for analysis at different interval. All the analysis were conducted at a temperature of 25 ± 2 °C and the samples were stored at 25 ± 2 °C in dark, in an airtight container over the period of study.
SERS Activity of Silver Nanoparticles
For SERS experiments, 50 µl of the Ag colloid was mixed with 50 µl of the Raman analyte NBA solution (1 × 10−6 M final concentration), and then made up to a final volume of 0.3 mL using ultrapure water followed by vigorous mixing. The analyte-NPs mixture was dispersed thoroughly in water by sonication. SERS spectra for the samples were acquired using a PerkinElmer RamanFlexTM 400 Fibre Optic Analyzer (USA) at 532 nm at a power of ~ 32 mW. Spectra were accumulated for 1 second. Each spectrum was the average of 5 scans.
Antibacterial Activity of Silver Nanoparticles
Organism
E. coli was grown overnight in NB at 37 °C. Cells were diluted in NB, and optical density (OD) at 600 nm (OD600) was adjusted to 0.1 (OD of 0.1 corresponds to a concentration of 108 colony forming units [CFU]/mL at 600 nm).
Bacterial susceptibility to silver nanoparticles
To examine the bacterial susceptibility assay, the E. coli cells were cultured on a nutrient agar plate supplemented with Ag NPs (10-60 μg/ plate). Approximately 105 CFU of E. coli were cultured on nutrient agar plates supplemented with Ag NPs. The plates were incubated at 37 °C for 24 hrs and the number of colonies were manually counted [12,13]. Ag NPs free nutrient agar plates and plates supplemented with antibiotic (ampicillin; 10-60 µg) grown under same conditions were used for comparison.
Bacterial growth in the presence of silver nanoparticles
To study the bacterial growth rate in presence or absence of Ag NPs and antibiotic (ampicillin), E. coli were grown in 100 mL of NB medium supplemented with different doses of Ag NPs (10-60 µg) and ampicillin (10-60 µg) at 37 °C under continuous stirring on an orbital shaker. Growth rate was determined by measuring OD600 at various times. OD values were converted into CFU/ mL [12,13].
Electron microscopy analysis of Ag NPs treated bacteria
E. coli were grown in 20 mL NB medium overnight at 37 °C. 1 ml of 105 E. coli cells were inoculated into 100 mL fresh NB medium containing 20 µg of Ag NPs. The bacterial suspension was collected after 3 hrs of exposure to NPs and used for scanning electron microscopy (SEM) and HRTEM analysis.
For SEM sample preparation, 20 µL of NPs treated and untreated E. coli cells were each deposited on polycarbonate membrane and fixed in 2.5% (v/v) glutaraldehyde in 0.1 M potassium phosphate buffer (pH 7.2). These samples were dehydrated in a series of ethanol (35, 50, 70, 80, 90, 95 and 100% for 10 min each). After drying, the samples were mounted on aluminum stubs, sputter-coated with a layer of gold and viewed under SEM (LEO 1450VP, UK) operated at 10 kV.
For HRTEM, 10 mL of NPs treated and untreated bacteria were separated by centrifugation and washed thrice with sterile water. The washed bacterial isolates were stained with 0.2% phosphotungstic acid for 5 min. The stained cells were loaded onto 3 mm copper TEM holding grid and viewed under HRTEM (JEOL JEM 2100, Japan) operated at 80 kV.
Results
Silver Nanoparticles Characterization
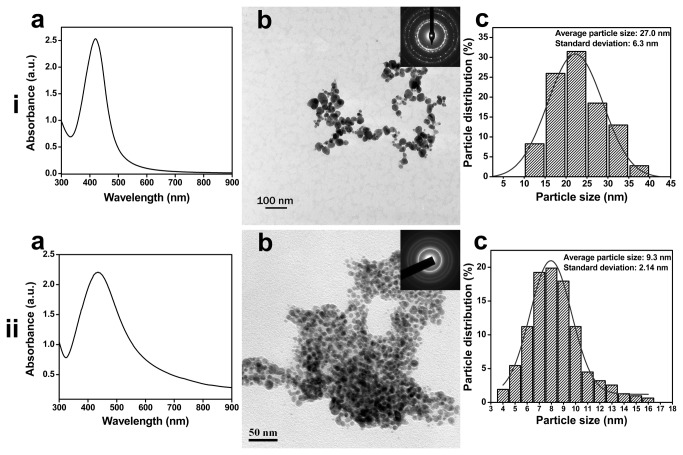
Ag NPs synthesized using A. indica leaf extract solution and biopolymer pectin were characterized using UV-Vis absorbance spectroscopy and TEM for optical and structural properties (Figure 2). A. indica and pectin Ag NPs showed absorption surface plasmon resonance (SPR) at 418 (Figure 2ia) and 434 nm (Figure 2iia), respectively. The morphology and dimension of the Ag NPs were determined using TEM (Figure 2ib and 2iib). From TEM images, the particles were observed to be almost spherical in shape. Inset in the TEM images are the corresponding selected area electron diffraction (SAED) patterns of the Ag NPs. The polycrystalline rings with plane distance around 2.36 Å, 2.04 Å, 1.45 Å and 1.23 Å (from inner to outer) can be assigned to the plane families (111), (200), (220) and (311) of pure fcc silver structure (see Figure S1 for corresponding powder XRD patterns). The average particle size measured by TEM was 27.0 (Figure 2ic) and 9.3 nm (Figure 2iic) for A. indica and pectin Ag NPs, respectively. However, the average HDD values for A. indica and pectin Ag NPs measured in water by DLS differed from their average diameters measured by TEM and were observed to be 33.42 and 199.2 nm (Table 1), respectively. This discrepancy may be attributed to the DLS measurement technique. In DLS, the HDD of NP include Ag core, capping agents and layers of the solvent molecules that are associated with the particle [14] while from the TEM image only the NP core size was scaled. One inherent limitation of the DLS method is taken into account; since the particle size is correlated with the scattered light intensity, presence of fewer larger diameter or aggregated NPs will dominate the intensity [15]. Both the A. indica and pectin Ag NPs had a negative ζ potential of −27.7 and −36.2 mV (Table 1), respectively in water, which were placed in the stable region (without aggregation) as noted in Derjaguin-Verwey Lundau-Overbeek (DVLO) theory [16,17]. Presence of elemental silver in the final NP product was evident by the EDS spectra (Figure S2).
Figure 2. Silver nanoparticles characterization.
(a) UV-Vis absorbance spectrum, (b) TEM image (inset SAED pattern) and (c) size distribution histogram pattern of (i) A. indica Ag NPs and (ii) pectin Ag NPs.
Table 1. TEM primary average size, HDD and ζ potential value (in water) of Ag NPs.
| Nanoparticles | TEM primary size (nm) | HDD in water (nm) | ζ value in water (mV) |
|---|---|---|---|
| A. indica Ag NPs | 27.0 ± 6.3 | 33.42 | −27.7 |
| Pectin Ag NPs | 9.3 ± 2.4 | 199.2 | −36.2 |
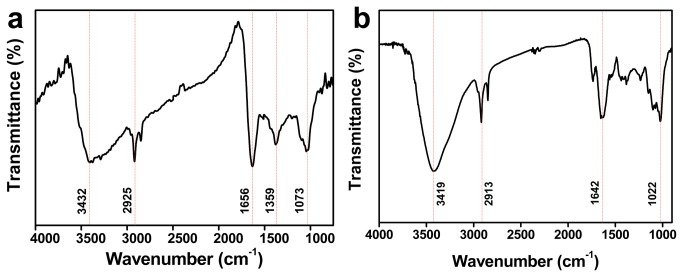
The surface functional groups present in A. indica and pectin Ag NPs were analyzed using FTIR (Figure 3). FTIR spectra of A. indica Ag NPs (Figure 3a) was dominated by IR peaks positioned at 3432 cm−1, 2925 cm−1, 1656 cm−1, 1359 cm−1 and 1073 cm−1 which correspond to functional groups of OH stretch; H−bonding of phenolic compounds, stretching modes of aldehydic C–H, stretching vibration of amide ν(C=O) of proteins, bending vibrations of geminal methyl or symmetric stretching of carbonyl group, and antisymmetric stretching of C–O group of polysaccharide and/or chlorophyll, respectively. Presence of these functional groups confirms the presence of bio-molecules on NPs’ surface, which may depict proteins, polyphenols, flavonoids, and carbohydrates present in the leaf extracts (see Figure S3 for leaf extract characterization).
Figure 3. Surface composition of bio-silver nanoparticles.
FTIR spectra of (a) A. indica and (b) pectin Ag NPs.
Figure 3b shows the FTIR spectra of pectin Ag NPs. It was observed from the IR spectra of pectin Ag NPs that most of the peaks appear at almost the same wavelength as seen for pure pectin (Figure S4). The peak at 1022 cm−1 can be attributed to C=O or C=C group of pectin. Stretching vibration at 1642 cm−1 is related to COO− group of pectin. The peak at about 2913 cm−1 arises from CH2 group and peak positioned at 3419 cm−1 can be indexed to ν(OH) group of pectin. The identified IR peaks corresponding to the functional groups of pure pectin confirms the presence of pectin molecules on the surface of Ag NPs.
Colloidal Stability of Silver Nanoparticles in Different Aqueous Environment
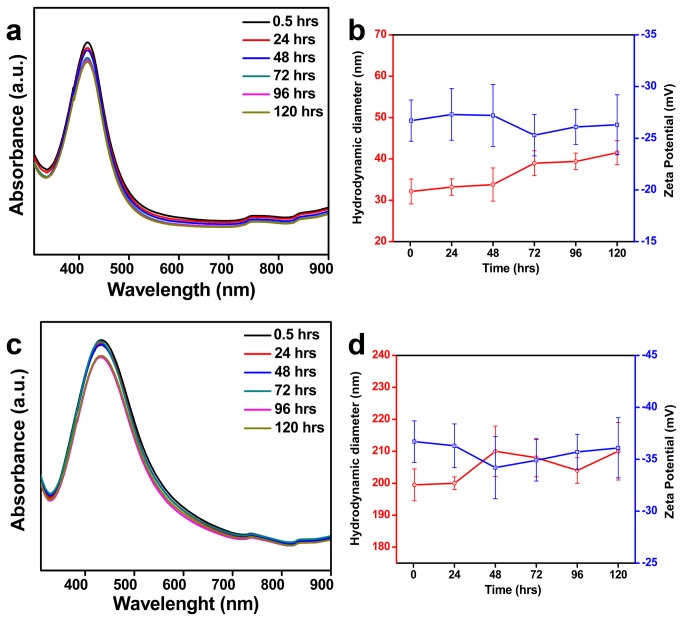
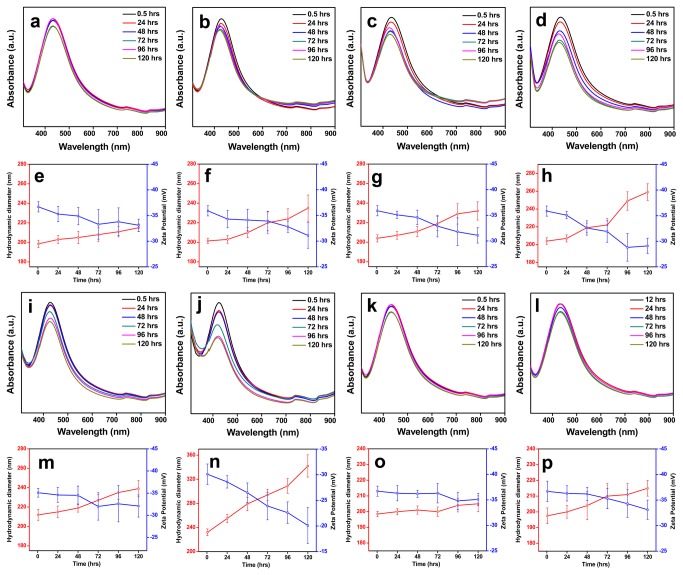
The stability of the NPs were studied in water, electrolyte environment, biological media and in different pH using UV-Vis absorbance spectroscopy by evaluating the changes in their SPR band, and also by studying their changes in HDD and surface charge using zeta measurements. Electrolyte environments - solutions of NaCl and NaNO3 with concentrations 50 and 200 mM; biological media -0.1 and 0.25% BSA solution; pH solutions - pH 7 and pH 9, were used to study the aggregation kinetics of the NPs synthesized using A. indica leaf extract and pectin solutions for a reasonable time period of 5 days (120 hrs) at temperature close to 25 °C. The UV-Vis absorbance spectra of both Ag NPs suspended in water were very similar and did not show any significant change during the entire study period of 120 hrs (Figure 4a and 4c). Similar to the absorbance spectra, the hydrodynamic size and ζ potential values of the NPs didn’t vary much indicating the stability of particle in water (Figure 4b and 4d).
Figure 4. Colloidal stability of bio-silver nanoparticles in water.
(a) UV-Vis absorbance spectra, and (b) HDD and ζ potential values of A. indica Ag NPs. (c) UV-Vis absorbance spectra and (d) HDD and ζ potential values pectin Ag NPs.
Figure 5 shows the SPR spectra, HDD and ζ values of A. indica Ag NPs dispersed in different solutions (NaCl, NaNO3, BSA and pH). Observing the SPR spectra of A. indica Ag NPs, it is clear that the plasmon bands look nearly similar when dispersed in all the solutions (Figures 5a-d, 5i-l), which indicates that the NPs were stable without any sign of particle aggregation. It was also seen, that there was no significant change in the HDD and ζ potential values of the particles when dispersed in the solutions (Figures 5e-h and 5m-p).
Figure 5. Colloidal stability of A. indica Ag NPs in different media.
Time dependent UV-Vis absorbance spectra and zeta (HDD and ζ potential value) measurements of A. indica Ag NPs dispersed in different media. A. indica Ag NPs dispersed in (a-b; e-f) NaCl, (c-d; g-h) NaNO3, (i-j; m-n) BSA, (k; o) pH 7 and (l; p) pH 9.
Pectin Ag NPs dispersed in the NaCl solutions (Figures 6a-b and 6e-f) and in pH 7 and 9 (Figures 6k-l and 6o-p) showed stability for a reasonable time period of 120 hrs with almost identical UV-Vis absorbance spectra and zeta measurements (HDD and ζ potential values). However, with prolonged exposure time the UV-Vis absorbance spectra were not the same when dispersed in NaNO3 (Figure 6c-d) and BSA (Figure 6i-j). There was a change in their absorbance intensity as time prolonged when they were dispersed in higher concentration of NaNO3 (200 mM) (Figure 6d) and BSA (0.25%) solutions (Figure 6j). Similar to the UV-Vis absorbance spectra, the NPs suspended in 200 mM NaNO3 (Figure 6h) and 0.25% BSA (Figure 6n) solutions showed increased hydrodynamic size (above 250 nm at 120 hrs) and low negative ζ values.
Figure 6. Colloidal stability of pectin Ag NPs in different media.
Time dependent UV-Vis absorbance spectra and zeta (HDD and ζ potential value) measurements of pectin Ag NPs dispersed in different media. Pectin Ag NPs dispersed in (a-b; e-f) NaCl, (c-d; g-h) NaNO3, (i-j; m-n) BSA, (k; o) pH 7 and (l; p) pH 9.
Raman Enhancement Efficiency of Bio-Silver Substrates
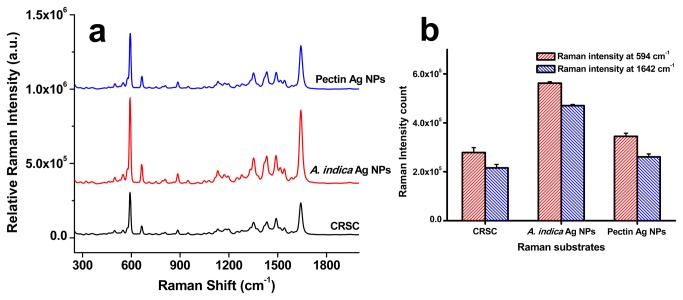
Optical field enhancing efficiencies of Ag NPs were determined using SERS by enhancing the signals of NBA dye molecules (Figure 7). Included for comparison are standard silver colloids prepared by a citrate reduction method [18]. Clearly the NPs do not give any significant background Raman spectral features (Figure S5). Figure 7a shows the average SERS spectra recorded of 1 × 10-6 M NBA solution with each of the silver samples. Also included are the neat NBA Raman spectrum and SERS spectrum recorded when standard citrate reduced silver colloid (CRSC) was probed. All the NBA-Ag NPs showed spectral signals of NBA dye molecule indicating that the substrates are SERS active. However, pure NBA at similar concentration (1 × 10−6 M) in water (without Ag substrate) didn’t hardly produce any characteristic spectral signals of NBA (Figure S6). The SERS spectra of NBA consist of several observable Raman peaks, which are intrinsic to chemical bonds seen in NBA [19]. Table 2 details the NBA peaks and their corresponding peak assignments. When the average spectra collected from the Ag substrates was normalized, it was obvious that A. indica and Pectin Ag NPs substrates out perform CRSC substrate (Figure 7a). The A. indica Ag NPs produced highest SERS signals of NBA followed by pectin Ag NPs and CRSC. Figure 7b shows the Raman intensity counts read at Raman 594 and 1642 cm-1. The Raman intensity counts obtained from the A. indica Ag NPs were almost two folds higher than that obtained from the CRSC substrate.
Figure 7. Optical enhancing properties of silver nanoparticles.
(a) SERS spectra of NBA adsorbed on A. indica Ag NPs, pectin Ag NPs and CRSC substrates. (b) Raman intensity count with respect to Ag substrates at 549 and 1642 cm-1.
Table 2. The Raman shifts and peak assignments of Nile blue A.
|
Raman shifts (cm−1)
|
||||
|---|---|---|---|---|
| CRSC | A. indica Ag NPs | Pectin Ag NPs | Reported Raman peaks for NBA (cm−1) | Peak assignments |
| 592 | 592 | 594 | 590 | CCN; CNC |
| 663 | 662 | 666 | In plane CCC; NCC | |
| 885 | 886 | 886 | ||
| 1132 | 1132 | 1132 | 1141 | CH bending |
| 1352 | 1352 | 1352 | 1351 | Ring stretching |
| 1434 | 1432 | 1433 | ||
| 1490 | 1490 | 1490 | 1492 | |
| 1642 | 1642 | 1640 | 1640 | |
Bactericidal Activity of Bio-Silver Nanoparticles
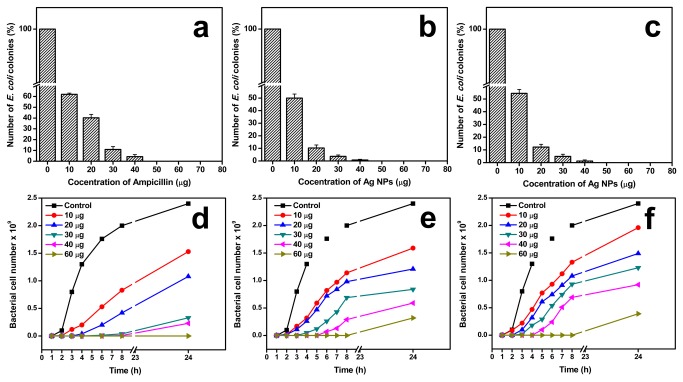
In order to study the bactericidal activity of the Ag NPs, E. coli was grown on nutrient agar plates and NB media supplemented with NPs. Figure 8b and 8c shows the number of bacterial colonies grown on NB agar plates as a function of NPs concentration. The Ag NPs inhibited the bacterial growth of almost 90% when supplemented with 30 µg of Ag NPs in the nutrient agar (Figure 8b and 8c). For both the NPs hardly any colonies of bacteria were found above 40 µg NP concentration, indicating 100% inhibition of growth (Figure 8b and 8c). However, the activity was not the same when identical doses of Ag NPs were studied with the NB liquid medium. Figure 8e and 8f shows the bacterial growth in NB liquid medium supplemented with different concentrations of Ag NPs. Most of the studied concentration of NPs showed bacterial growth and a major difference was the time delay in bacterial growth in the liquid medium. The delay of bacterial growth was seen to be proportionate to the increase in concentration of the NPs (Figure 8e and 8f). On comparison of bactericidal activity of Ag NPs with antibiotic, the ampicillin supplemented plates showed little higher number of bacterial colony, however, concentration above 40 µg completely inhibited the bacterial growth (Figure 8a) as observed with Ag NPs. In liquid culture, concentrations like 10 and 20 µg showed a considerable level of bacterial growth and increase in the concentration further affected the bacterial growth (30 and 40 µg ampicillin supplemented cultures contained < 0.2 × 109 cells after 24 hrs) with complete growth inhibition at the highest studied at a concentration of 60 µg (Figure 8d).
Figure 8. Effect of Ag NPs on bacterial growth.
Viability of E. coli cells in solid agar and NB liquid media on exposure to antibiotic (ampicillin) and Ag NPs. Percentage histogram plot of the number of E. coli colonies as a function of the concentration of (a) ampicillin, (b) A. indica Ag NPs and (c) pectin Ag NPs in agar plates. Growth curve of E. coli in NB medium supplemented with (d) ampicillin, (e) A. indica Ag NPs and (f) pectin Ag NPs.
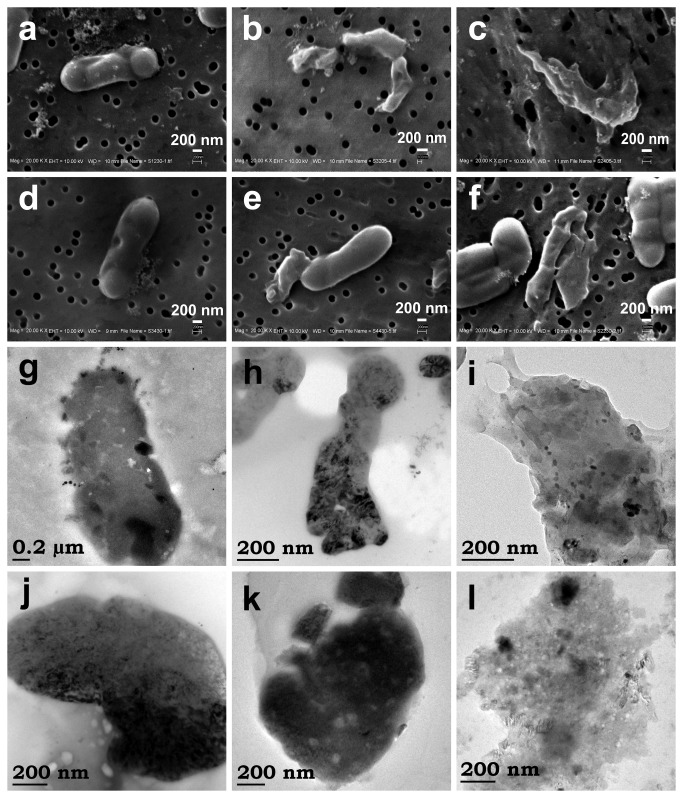
To study the cellular membrane structure of Ag NPs treated and untreated bacterial cells, electron microscopy (SEM and HRTEM) techniques were employed. Figure 9a-f shows the SEM images of cells treated with A. indica Ag NPs (Figure 9a-c) and pectin Ag NPs (Figure 9d-f) (see Figure S7 for SEM image of control bacterium). SEM images clearly indicate the anchoring of Ag NPs on the surface of the bacterial cells making a physical contact (Figure 9a and 9d). It was seen from the images that the cells were completely disintegrated without a definite cell structure evidencing the cell lyses (Figures 9b-c and 9e-f). To get a clear picture on the above parameters such as physical contact of Ag NPs with cellular membrane, membrane rupturing and lyses of cell, HRTEM images of Ag NPs treated E. coli were acquired. Physical interaction of the Ag NPs to the E. coli cellular membrane was evident with the HRTEM images (Figure 9g and 9j). The structural damage (Figure 9h and 9k) and complete disintegration (or degradation) of the cellular membrane structure were also observed (Figure 9i and 9l).
Figure 9. Electron microscopy images of bio-Ag NPs exposed E. coli cells.
SEM images of E. coli cells treated with (a-c) A. indica Ag NPs and (d-f) pectin Ag NPs. HRTEM images E. coli cells treated with (g-i) A. indica Ag NPs and (j-l) pectin Ag NPs.
Discussion
The use of plant-derived materials, in particular, the leaf extract has been extensively studied over the past years in synthesis of Ag NPs. The phytochemicals present in plant extracts such as water-soluble proteins, polyphenols, and sugars have been reported to play a significant role in reducing and stabilizing the NPs [20-22]. The presence of the above phytochemicals in the leaf extract have been determined using UV-Vis absorbance spectroscopy (Figure S3a) and through standard biochemical estimations (Figure S3b). However, it is obvious that determining the specific role of these phytochemicals is ruled-out, since most of them have dual function during synthesis. For example, proteins and sugars can actively participate in both reduction of metal ions and in stabilization of the NP formed [20]. The FTIR spectra of A. indica Ag NPs (Figure 3a) indicate the presence of functional groups corresponding to polyphenols, amides and carbohydrates on the NP surface proving that these molecules contribute to the stability of NPs. A. indica phytochemicals capping the NPs makes them colloidally stable in different media such as electrolyte solutions (NaCl; NaNO3), biological solution (BSA) and in pH solutions (pH 7 and pH 9). The UV-Vis spectra and zeta measurements remained almost same for NPs suspended in all media throughout the study (Figure 5). It is conceivable that the phytochemicals in A. indica leaf extract provide a strong coating on the surface of the NPs resulting in electrostatic repulsion between the NPs in the different aqueous environments leading to efficient stability.
Polymers are known to offer control over reduction rate, enabling NPs of different shapes and sizes, and also can provide an inelastic stability to NPs preventing their aggregation. Pectin used in the study is a non-toxic; naturally occurring plant polysaccharide (biopolymer) present in the peels of apple, citrus fruits and also found in some vegetables [23,24]. Structurally pectin is a linear polysaccharide consisting of D-galacturonic acid units joined in chain by means of α-(1-4) glycosidic linkage (Figure 1b) [25,26]. The presence of functional groups such as carboxyl and hydroxyl groups makes pectin an ideal molecule for complexing with various metals [27,28]. The presence of pectin molecules on the surface of the NPs is evident by the FTIR spectrum (Figure 3b). It was observed from the FTIR spectra that the intense peak at 1744 cm−1 for pure pectin (COOH group of pectin) is obviously weaker for the pectin Ag NPs (Figure 3b and Figure S4). The above changes in the FTIR pattern may indicate the participation of carboxyl group in formation of the NPs. The pectin-Ag NPs showed excellent level of colloidal stability in the electrolyte solutions (NaCl and NaNO3 [50mM]), in different pH solutions (pH 7 and 9) and in lower percentage of biological solution (BSA) (Figure 6). However, when pectin Ag NPs were dispersed in higher concentration of NaNO3 (200 mM) and BSA (0.25%) solutions, decrease in their SPR band intensities were observed. Difference in the absorption intensities of NPs can be due to the sedimentation of colloidal particles at higher strength of NaNO3 and BSA solutions [29,30], which might have influenced the availability of free NPs in the suspension media resulting in decrease in absorbance intensity.
A careful survey of literature indicates that in most of the NPs synthesized using plant-derived materials, research has stopped at the level of basic material characterization without exploring its application. Here, the NPs were studied for optical field enhancing property using SERS by enhancing the signals of NBA using the A. indica Ag NPs and pectin Ag NPs as the substrates. Additionally, antibacterial property was determined by treating E. coli with bio-NPs. In determining the SERS efficiency of the Ag NPs substrate, A. indica Ag NPs were observed to enhance the signals of NBA more than that of the pectin NPs. As reported elsewhere, the SERS efficiency is dependent on the surface charge and efficient adsorption of the analyte molecule on the surface of the NPs [31,32]. The higher negative surface charge of the pectin Ag NPs (ζ potential value of -36.2 mV) may prevent the particle aggregation and also influence the adsorption of analyte on the surface of the particles, hence affecting SERS enhancement [31,32]. Figure S8 shows the plot matching the SERS intensity count at Raman shift 594 cm−1 with ζ potential values of plant Ag substrates, clearly indicating dependable SERS activity with NP surface charge.
All studied test compounds (antibiotic and Ag NPs) showed almost similar level of bactericidal activity on agar plates. Concentrations above 40 µg completely inhibited the bacterial growth. However, in the liquid medium the bacterial growth was observed in all the studied NP concentrations, the only difference is the initial growth delay of the bacterial cells, which increased as the concentration of the NPs increased in the medium. On other hand, at concentration above 30 µg of ampicillin the bacterial growth was greatly affected with complete inhibition even after 24 hrs when supplemented with 60 µg of ampicillin. It was explained elsewhere, that the NPs in liquid medium only delays the bacterial growth [12,13]. It has been stated that, as the time prolongs, the concentration of the Ag NPs in the medium decreases due to the interaction of the NPs with intracellular components from the deceased bacterial cells causing particle coagulation, and allowing resumption of the bacterial growth [12,13]. Even though the pectin NPs possessed smaller grain size and higher negative ζ potential value (-36.2 mV), it displayed almost similar E. coli cell growth pattern in the liquid medium as that of A. indica Ag NPs. As observed by the behavior of pectin Ag NPs in BSA solution (Figure 6j and 6n), the pectin Ag NPs might have interacted with the intracellular components (peptides) from the lysed cells, thus minimizing the dispersion state of NPs in the NB media leading to ineffective antibacterial activity.
Although the exact mechanism of bactericidal action of Ag NPs has not been fully explained, the physical damage of the bacterial membrane caused by the NP interaction may lead to cell death. However, the mode of interaction (anchoring) between E. coli cell surface and NP (through any specific receptors or molecular species in cell wall) is not yet resolved fully [13,33]. In a recent study, effective anchoring of Ag NPs on E. coli cell surface was stated due to the adsorbed organic molecule on the NP surface from the guava leaf broth during synthesis, which aided in better interaction with the cell surface when compared to the chemically synthesized Ag NPs [33]. Polysaccharides (exopolysaccharide) surfaced Ag NPs are also reported for a higher adherence towards bacterial surface to provide an efficient bactericidal activity [34]. As stated in above literatures, the organic (phytochemicals) coat on A. indica Ag NPs and polysaccharide (pectin) coat on pectin Ag NPs might have been aided in their better interaction with the bacterial cell wall [33,34]. A good physical interaction between the NP and the bacterial cell wall is vital for the cell damage [35]. Physical interaction of the NPs with the cellular membrane may cause physical damage (through pitting) (Figures 9a, 9d, 9g and 9j) that ultimately leads to cellular death or pass through membrane [36,37]. Similarly Sondi and Sondi [13] reported that the Ag NPs causes ‘pits’ in the cellular membrane of bacteria, leading to increased membrane permeability and finally, causing cell death. Increase in the membrane permeability was observed by Stoimenov and co-workers [38] when E. coli cells were treated with highly reactive metal oxide. Increase in the permeability affects the bacterial cells to regulate transport through the plasma membrane and, finally, leading to cell death [38]. E. coli membrane is known to consist of lipopolysaccharids (LPS) in the outer membrane which serves as an effective permeability barrier [39,40]. The interaction of NPs with the membrane may affect the LPS and other membrane proteins, causing change in the membrane permeability (or degradation of membrane structure) (Figure 9) [13,33,34,41].
Conclusions
We herein have presented a facile green method for synthesis of Ag NPs using leaf extracts of A. indica and biopolymer pectin from apple peel. Both the NPs showed a considerable level of stability in most of the studied media for a reasonable time period of 120 hrs. On determining the SERS activity, both the studied Ag NPs displayed elegant SERS signals of Raman analyte NBA describing their SERS activity. SERS signals exhibited by the studied Ag NPs substrates were higher than that of the routinely synthesized CRSC; in particular the Ag NPs synthesized using A. indica leaf extract showed the highest SERS efficiency. On studying the bactericidal activity of the Ag NPs, it was found that above 40 µg of Ag NPs concentration in agar plates completely inhibited bacterial growth. However, in the liquid culture, the NPs caused a growth delay and the delay time increased in proportion to the increase in concentration of the Ag NPs in the media. The electron microscopy images evidenced the physical interaction between the NPs and cellular membrane and also the damages caused by NP to the bacterial cell wall integrity. These stable green Ag NPs can be applied in biology for detection based on SERS and in bacterial infection control.
Supporting Information
Crystalline phase of bio-Ag NPs. Powder XRD pattern of Ag NPs synthesized using A. indica plant leaf extract and biopolymer pectin.
(TIF)
Elemental analysis of the bio-Ag NPs. EDS of (a) A. indica and (b) pectin Ag NPs.
(TIF)
Characterization of leaf extracts. (a) UV-Vis absorbance spectrum of leaf extract. Strong absorption band at ~ 272 nm is assigned to the aromatic side group of amino acid residue of protein. The accompanying peak at absorbance ~ 325 nm could be arisen from the water-soluble phenolic compounds in the extract. (b) Concentration of total soluble proteins (TSP), total sugars and total phenolic content (TPC) in final leaf filtrate.
(TIF)
Chemical composition of pectin. FTIR spectrum of pure pectin.
(TIF)
Background Raman spectra of Ag NPs. Raman spectra of blank silver nanoparticles (without Raman analyte molecule NBA).
(TIF)
Neat Raman spectrum of NBA dye molecule. Raman spectrum of 1 × 10−6 M Nile Blue A in water without silver substrate.
(TIF)
Electron microscopy photo of E. coli. SEM image of Ag NPs untreated E. coli cells.
(TIF)
Comparison of zeta potential values of bio-Ag NPs with their respective SERS intensities. Plot of SERS intensity count at 594 cm−1 versus zeta potential values of Ag NPs.
(TIF)
Acknowledgments
The authors thank the Central research facility, IIT Kharagpur and Ganab Jaffar, Nancy-University, France for HRTEM and TEM analysis. Dr R. Selvakumar, Department of Chemistry, Bharathiar University, is thankful for helping us in FTIR.
Funding Statement
No current external funding source for this study.
References
- 1. Doty RC, Tshikhudo TR, Brust M, Fernig DG (2005) Extremely stable water-soluble Ag nanoparticles. Chem Mater 17: 4630-4635. doi: 10.1021/cm0508017. [DOI] [Google Scholar]
- 2. Chen KL, Elimelech M (2006) Aggregation and deposition kinetics of fullerene (C60) nanoparticles. Langmuir 22: 10994-11001. doi: 10.1021/la062072v. PubMed: 17154576. [DOI] [PubMed] [Google Scholar]
- 3. Tielemans M, Roose P, De Groote P, Vanovervelt S (2006) Colloidal stability of surfactant-free radiation curable polyurethane dispersions. Prog Org Coat 55: 128-136. doi: 10.1016/j.porgcoat.2005.08.010. [DOI] [Google Scholar]
- 4. Zhang W, Yao Y, Sullivan N, Chen Y (2011) Modeling the primary size effects of citrate-coated silver nanoparticles on their ion release kinetics. Environ Sci Technol 45: 4422-4428. doi: 10.1021/es104205a. PubMed: 21513312. [DOI] [PubMed] [Google Scholar]
- 5. Kittler S, Greulich C, Diendrof J, Koller M, Epple M (2010) Toxicity of silver nanoparticles increases during storage because of slow dissolution under release of silver ions. Chem Mater 22: 4548-4554. doi: 10.1021/cm100023p. [DOI] [Google Scholar]
- 6. Fabrega J, Fawcett SR, Renshaw JC, Lead JR (2009) Silver nanoparticle impact on bacterial growth: Effect of pH, concentration, and organic Matter. Environ Sci Technol 43: 7285-7290. doi: 10.1021/es803259g. PubMed: 19848135. [DOI] [PubMed] [Google Scholar]
- 7. Lubick N (2008) Nanosilver toxicity: Ions, nanoparticles--or both? Environ Sci Technol 42: 8617. doi: 10.1021/es8026314. PubMed: 19192768. [DOI] [PubMed] [Google Scholar]
- 8. Auffan M, Rose J, Wiesner MR, Bottero JY (2009) Chemical stability of metallic nanoparticles: a parameter controlling their potential cellular toxicity in vitro. Environ Pollut 157: 1127-1133. doi: 10.1016/j.envpol.2008.10.002. PubMed: 19013699. [DOI] [PubMed] [Google Scholar]
- 9. Jiang J, Oberdorster G, Biswas P (2009) Characterization of size, surface charge, and agglomeration state of nanoparticle dispersions for toxicological studies. J Nanopart Res 11: 77-89. doi: 10.1007/s11051-008-9446-4. [DOI] [Google Scholar]
- 10. Kumar V, Yadav SK (2008) Plant-mediated synthesis of silver and gold nanoparticles and their applications. J Chem Technol Biotechnol 84: 151-157. [Google Scholar]
- 11. Iravani S (2011) Green synthesis of metal nanoparticles using plants. Green Chem 13: 2638-2650. doi: 10.1039/c1gc15386b. [DOI] [Google Scholar]
- 12. Pal S, Tak YK, Song JM (2007) Does the antibacterial activity of silver nanoparticles depend on the shape of the nanoparticle? A study of the gram-negative bacterium Escherichia coli . Appl Environ Microbiol 73: 1712-1720. doi: 10.1128/AEM.02218-06. PubMed: 17261510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Sondi I, Salopek-Sondi B (2004) Silver nanoparticles as antimicrobial agent: a case study on E. coli as a model for gram-negative bacteria. J Colloid Interface Sci 275: 177-182. doi: 10.1016/j.jcis.2004.02.012. PubMed: 15158396. [DOI] [PubMed] [Google Scholar]
- 14. MacCuspie RI, Rogers K, Patra M, Suo Z, Allen AJ, et al. (2011) Challenges for physical characterization of silver nanoparticles under pristine and environmentally relevant conditions. J Environ Monit 13: 1212-1226. doi: 10.1039/c1em10024f. PubMed: 21416095. [DOI] [PubMed] [Google Scholar]
- 15. Handy RD, von der Kammer F, Lead J, Handy RD, von der Kammer F (2008) The ecotoxicology and chemistry of manufactured nanoparticles. Ecotoxicology 17: 287-314. doi: 10.1007/s10646-008-0199-8. PubMed: 18351458. [DOI] [PubMed] [Google Scholar]
- 16. Derjaguin BV, Landau L (1941) Theory of the stability of strongly charged lyophobic sols and adhersion of strongly charged particles in solutions of Electrolytes. Acta Physicochim URSS 14: 633-652; (1993) Prog Surf Sci 43: 30-59 [Google Scholar]
- 17. Verwey EJW, Overbeek JTG (1948) Theory of the Stability of Lyophobic Colloids: 1st ed. Amsterdam: Elsevier. [Google Scholar]
- 18. Lee PC, Meisel D (1982) Adsorption and surface enhanced Raman of dyes on silver and gold solutions. J Phys Chem 86: 3391-3395. doi: 10.1021/j100214a025. [DOI] [Google Scholar]
- 19. Lawless MK, Mathies RA (1992) Excited-state structure and electronic dephasing time of Nile blue from absolute resonance Raman intensities. J Chem Phys 96: 8037-8045. doi: 10.1063/1.462355. [DOI] [Google Scholar]
- 20. Shukla R, Nune SK, Chanda N, Katti K, Mekapothula S et al. (2008) Soybeans as a phytochemical reservoir for the production and stabilization of biocompatible gold nanoparticles. Small 4: 1425-1436. doi: 10.1002/smll.200800525. PubMed: 18642250. [DOI] [PubMed] [Google Scholar]
- 21. Li S, Shen Y, Xie A, Yu X, Qiu L et al. (2007) Green synthesis of silver nanoparticles using Capsicum annuum L. extract. Green Chem 9: 852-858. doi: 10.1039/b615357g. [DOI] [Google Scholar]
- 22. Jacob JA, Mahal HS, Biswas N, Mukherjee T, Kapoor S (2008) Role of phenol derivatives in the formation of silver nanoparticles. Langmuir 24: 528-533. doi: 10.1021/la702073r. PubMed: 18095719. [DOI] [PubMed] [Google Scholar]
- 23. May CD (1990) Industrial pectins: Sources, production and applications. Carbohydr Polym 12: 79-99. doi: 10.1016/0144-8617(90)90105-2. [DOI] [Google Scholar]
- 24. Thakur BR, Singh RK, Handa AK (1997) Chemistry and uses of pectin--a review. Crit Rev Food Sci Nutr 37: 47-73. doi: 10.1080/10408399709527767. PubMed: 9067088. [DOI] [PubMed] [Google Scholar]
- 25. Sriamornsak P (2003) Chemistry of pectin and its pharmaceutical uses: A review Silpakorn University International Journal 3: 206-228. [Google Scholar]
- 26. Mukhiddinov ZK, Khalikov DK, Abdusamiev FT, Avloev CC (2000) Isolation and structural characterization of a pectin homo and ramnogalacturonan. Talanta 53: 171-176. doi: 10.1016/S0039-9140(00)00456-2. PubMed: 18968102. [DOI] [PubMed] [Google Scholar]
- 27. Kong JM, Wong CV, Gao ZQ, Chen XT (2008) Preparation of silver nanoparticles by microwave- hydrothermal technique. Synth React Inorg Metal-Organic Nano-Metal Chem 38: 186-188. [Google Scholar]
- 28. Sutton A, Franc G, Kakkar A (2009) Silver metal nanoparticles: Facile dendrimer-assisted size-controlled synthesis of selective catalytic reduction of chloronitrobenzenes. Journal of Polymer Sciences: Part A: Polymer Chemistry 47: 4482-4493. doi: 10.1002/pola.23501. [DOI] [Google Scholar]
- 29. Zamiri R, Azmi BZ, Ahangar HA, Zamiri G, Shahril Husin M et al. (2012) Preparation and characterization of silver nanoparticles in natural polymer using laser ablation. Bull Mater Sci 35: 727-731. doi: 10.1007/s12034-012-0360-0. [DOI] [Google Scholar]
- 30. El Badawy AM, Luxton TP, Silva RG, Scheckel KG, Suidan MT et al. (2010) Impact of environmental conditions (pH, ionic strength, and electrolyte type) on the surface charge and aggregation of silver nanoparticles suspensions. Environ Sci Technol 44: 1260-1266. doi: 10.1021/es902240k. PubMed: 20099802. [DOI] [PubMed] [Google Scholar]
- 31. Dos Santos DS Jr, Alvarez-Puebla RA, Oliveira ON Jr, Aroca RF (2005) Controlling the size and shape of gold nanoparticles in fulvic acid colloidal solutions and their optical characterization using SERS. J Mater Chem 15: 3045-3049. doi: 10.1039/b506218g. [DOI] [Google Scholar]
- 32. Alvarez-Puebla RA, Arceo E, Goulet PJ, Garrido JJ, Aroca RF (2005) Role of Nanoparticle Surface Charge in Surface-Enhanced Raman Scattering. J Phys Chem B 109: 3787-3792. doi: 10.1021/jp045015o. PubMed: 16851426. [DOI] [PubMed] [Google Scholar]
- 33. Parashar UK, Kumar V, Bera T, Saxena PS, Nath G et al. (2011) Study of mechanism of enhanced antibacterial activity by green synthesis of silver nanoparticles. Nanotechnology 22: 415104-415116. doi: 10.1088/0957-4484/22/41/415104. PubMed: 21918296. [DOI] [PubMed] [Google Scholar]
- 34. Selvakumar R, Aravind S, Anuradha MA, Balachandran YL (2013) A facile synthesis of silver nanoparticle with SERS and antimicrobial activity using Bacillus subtilis exopolysaccharides. J Experimental Nanosciences. doi: 10.1080/17458080.2013.778425. [DOI] [Google Scholar]
- 35. El Badawy AM, Silva RG, Morris B, Scheckel KG, Suidan MT et al. (2011) Surface charge-dependent toxicity of silver nanoparticles. Environ Sci Technol 45: 283-287. doi: 10.1021/es1034188. PubMed: 21133412. [DOI] [PubMed] [Google Scholar]
- 36. Choi O, Deng KK, Kim NJ, Ross L, Surampalli RY et al. (2008) The inhibitory effects of silver nanoparticles, silver ions, and silver chloride colloids on microbial growth. Water Res 42: 3066-3074. doi: 10.1016/j.watres.2008.02.021. PubMed: 18359055. [DOI] [PubMed] [Google Scholar]
- 37. Fabrega J, Renshaw JC, Lead JR (2009) Interactions of silver nanoparticles with Pseudomonas putida biofilms. Environ Sci Technol 43: 9004-9009. doi: 10.1021/es901706j. PubMed: 19943680. [DOI] [PubMed] [Google Scholar]
- 38. Stoimenov PK, Klinger RL, Marchin GL, Klabunde KJ (2002) Metal oxide nanoparticles as bactericidal agents. Langmuir 18: 6679-6686. doi: 10.1021/la0202374. [DOI] [Google Scholar]
- 39. Nikaido H, Vaara M (1985) Molecular basis of bacterial outer membrane permeability. Microbiol Rev 49: 1-32. PubMed: 2580220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Raetz CR (1990) Biochemistry of Endotoxins. Annu Rev Biochem 59: 129-170. doi: 10.1146/annurev.bi.59.070190.001021. PubMed: 1695830. [DOI] [PubMed] [Google Scholar]
- 41. Amro NA, Kotra LP, Wadu-Mesthrige K, Bulychev A, Mobashery S et al. (2000) High-resolution atomic force microscopy studies of the Escherichia coli outer membrane: structural basis for permeability. Langmuir 16: 2789-2796. doi: 10.1021/la991013x. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Crystalline phase of bio-Ag NPs. Powder XRD pattern of Ag NPs synthesized using A. indica plant leaf extract and biopolymer pectin.
(TIF)
Elemental analysis of the bio-Ag NPs. EDS of (a) A. indica and (b) pectin Ag NPs.
(TIF)
Characterization of leaf extracts. (a) UV-Vis absorbance spectrum of leaf extract. Strong absorption band at ~ 272 nm is assigned to the aromatic side group of amino acid residue of protein. The accompanying peak at absorbance ~ 325 nm could be arisen from the water-soluble phenolic compounds in the extract. (b) Concentration of total soluble proteins (TSP), total sugars and total phenolic content (TPC) in final leaf filtrate.
(TIF)
Chemical composition of pectin. FTIR spectrum of pure pectin.
(TIF)
Background Raman spectra of Ag NPs. Raman spectra of blank silver nanoparticles (without Raman analyte molecule NBA).
(TIF)
Neat Raman spectrum of NBA dye molecule. Raman spectrum of 1 × 10−6 M Nile Blue A in water without silver substrate.
(TIF)
Electron microscopy photo of E. coli. SEM image of Ag NPs untreated E. coli cells.
(TIF)
Comparison of zeta potential values of bio-Ag NPs with their respective SERS intensities. Plot of SERS intensity count at 594 cm−1 versus zeta potential values of Ag NPs.
(TIF)