Abstract
Sucralose is a widely-used artificial high-intensity sweetener. Although doubts have been raised about the safety of sucralose by several researchers, it can still be found in a broad range of foods and beverages worldwide, including in baked goods. Sucralose may decompose at high temperatures, and participate in chlorination reactions, generating highly toxic compounds. Here, we demonstrate that heating sucralose at high temperatures in stainless steel or other metal utensils in the presence of rust (Al2O3, Fe2O3, and CuO) produces polychlorinated dibenzo-p-dioxins and dibenzofurans (PCDD/Fs). PCDD/Fs were found in smoke generated during the heating of sucralose and in the residues after heating. CuO enhanced the PCDD/F yield in comparison with Al2O3 and Fe2O3.
Sucralose (4,1′,6′-trichloro-4,1′,6′-trideoxygalactosucrose, TGS) is an artificial sweetener which results from the selective substitution of three of the sucrose hydroxyl groups with chlorine atoms, involving inverting the configuration at the 4-position from the gluco- to the galacto-analogue, resulting in a product 600 times sweeter than sugar1,2. Sucralose is used as a food additive in more than 80 countries, and is present in over 4,000 products, including baked goods3,4,5,6. However, several studies have shown that sucralose has metabolic effects that may negatively impact human health7,8,9,10,11,12,13. In addition, it has been found that sucralose is not stable at high temperatures14,15.
Using thermoanalytical techniques, Bannach et al. (2009) found that sucralose is thermally stable at temperatures up to 119°C, but above this temperature, and up to 550°C, thermal decomposition takes place in three steps without the sucralose melting14. Rahn & Yaylayan (2010) reported that the thermal degradation of sucralose at 250°C resulted in the generation of toxic chloropropanols15. Our previous studies showed that highly toxic polychlorinated aromatic compounds, such as polychlorinated dibenzo-p-dioxins and dibenzofurans (PCDD/Fs), dioxin-like polychlorinated biphenyls, and polychlorinated naphthalenes, could be generated during the heating of foodstuffs in the presence of sucralose16,17,18. It still remains unclear whether these highly toxic compounds could be produced when pure sucralose is heated, and the formation mechanisms for these compounds have not yet been elucidated.
The effects of temperature, utensil materials, and rusts (metal oxides) on the formation of PCDD/Fs when sucralose is heated were investigated in the study presented here. The concentrations of tetra- through octa-chlorinated PCDD/Fs and 17 toxic 2,3,7,8-substituted PCDD/F congeners, collected from the smoke generated by heated sucralose and from the residues after heating, were measured.
Results
Large amounts of sucralose (5 g) were used in the experiments, and the PCDD/Fs produced were quantified. PCDD/Fs were not detected in the smoke or residues when sucralose was heated in quartz, aluminium, or copper utensils (the metal utensils were polished and rust was removed before use) at temperatures between 200 and 400°C. However, PCDD/Fs were detected in both the smoke and the residues when sucralose was heated at 350°C in stainless steel utensils, and it should be noted that stainless steel is commonly used for cooking utensils.
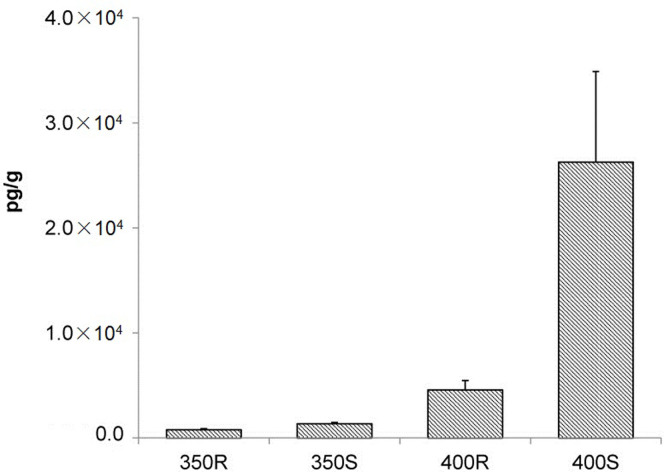
The PCDD/F concentrations in the smoke and residues were calculated on the basis of the original weight of sucralose (5 g) used in the experiments. Figure 1 shows that the PCDD/F concentrations increased in both the smoke and the residues with increasing temperature. The PCDD/F concentrations were higher in the smoke than in the residues. The total PCDD/F concentrations produced from sucralose (5 g) heated at 400°C reached 3.1 × 104 pg g−1, which was approximately 25 times higher than the concentrations produced at 350°C. The 17 toxic 2,3,7,8-substituted PCDD/F congeners contributed approximately 30% of the total amount of PCDD/Fs in the samples.
Figure 1. Polychlorinated dibenzo-p-dioxin and dibenzofuran (PCDD/F) concentrations produced from 5 g of sucralose heated in stainless steel utensils at 350°C and 400°C. R = residues, S = smoke.
The surface of metal cooking utensils can become oxidized, so rust, which is composed of metal oxides, is commonly found on utensil surfaces. The effects of different rusts (Al2O3, Fe2O3, and CuO), chosen based on metals typically used for cooking utensils, on PCDD/F formation during the heating of sucralose at 400°C were investigated. Figure 2 shows the PCDD/F concentrations produced in the smoke and residues when sucralose (5 g) was heated in the presence of Al2O3, Fe2O3, and CuO (1 g) at 400°C. CuO enhanced the yield of PCDD/Fs in comparison with Al2O3 and Fe2O3. The total PCDD/F concentrations reached 4.2 × 106 pg g−1 when sucralose was heated in the presence of CuO. Fe2O3 also had a strong effect on PCDD/F formation (giving PCDD/F concentrations of up to 1.2 × 106 pg g−1) in comparison with Al2O3 (which gave PCDD/F concentrations of up to 9.7 × 104 pg g−1). PCDD/F concentrations produced when sucralose was heated in the presence of Al2O3, Fe2O3, and CuO were significantly higher than the concentrations produced when sucralose was heated in stainless steel utensils (which gave PCDD/F concentrations of up to 3.1 × 104 pg g−1). No significant amounts of PCDD/Fs were detected when sucralose was heated in the presence of the metal oxides at 300°C.
Figure 2. Polychlorinated dibenzo-p-dioxin and dibenzofuran (PCDD/F) concentrations produced from 5 g of sucralose heated in the presence of 1 g of Al2O3, Fe2O3, or CuO at 400°C.
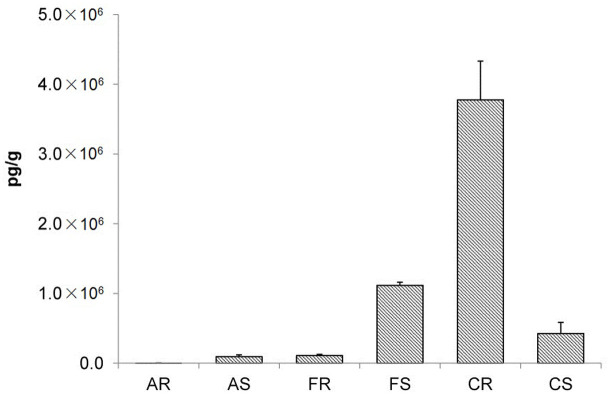
A = Al2O3, F = Fe2O3, C = CuO.
Sucralose (5 g) was heated in the presence of small amounts of CuO (1 g, 0.1 g, and 0.01 g) at 400°C to study the effects of the amount of metal oxide present on PCDD/F formation. The PCDD/F concentrations in the smoke and residues decreased as the amount of CuO present decreased. Smaller amounts of sucralose were also tested, and PCDD/F concentrations of approximately 3.0 × 106 pg g−1 were produced from 1 g of sucralose and 0.1 g of CuO at 400°C.
Discussion
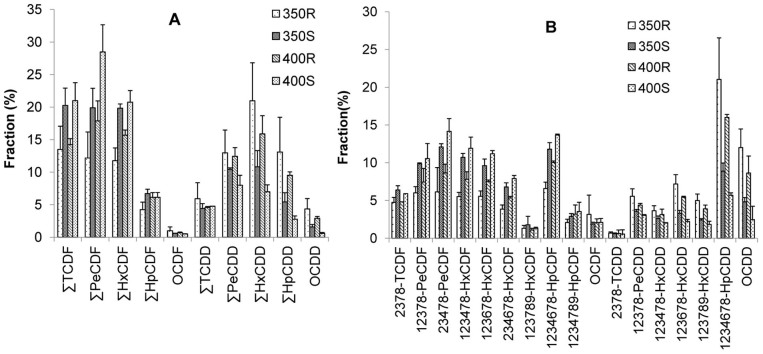
The PCDD/F concentrations were higher in the smoke than in the residues (Fig. 1). The PCDD/F distribution agreed with that described in previous reports, the PCDD/Fs produced being mainly in the gas phase17,18. The PCDD/F homologue and congener profiles in the smoke and residues were similar (Fig. 3), but they were different from those observed in previous studies17. This indicates that the PCDD/F formation pathways when sucralose is heated could be altered if foodstuffs (e.g., beef and vegetable oil) are added. Foodstuffs could provide a carbon source for the PCDD/F backbone formation. Sucralose was found to mainly act as a chlorine source in previous studies, but it acted as both a chlorine source and a carbon source in the study presented here. Furthermore, the addition of foodstuffs could cause PCDD/Fs to be produced at lower temperatures18.
Figure 3. Polychlorinated dibenzo-p-dioxin and dibenzofuran (PCDD/F) (A) homologue profiles and (B) congener profiles found in the smoke and residues formed when sucralose was heated in stainless steel utensils at 350°C and 400°C. TCDF = tetrachlorodibenzofuran, PeCDF = pentachlorodibenzofuran, HxCDF = hexachlorodibenzofuran, HpCDF = heptachlorodibenzofuran, OCDF = octachlorodibenzofuran, TCDD = tetrachlorodibenzo-p-dioxin, PeCDD = pentachlorodibenzo-p-dioxin, HxCDD = hexachlorodibenzo-p-dioxin, HpCDD = heptachlorodibenzo-p-dioxin, OCDD = octachlorodibenzo-p-dioxin.
CuO was more effective than the other metal oxides as a catalyst for PCDD/F formation19. Sucralose thermally decomposes at 119°C, liberating HCl14. Hisham and Benson (1995) investigated several metal oxides and found that they could absorb HCl and release Cl2 (the Deacon reaction)20, and they found that CuO was the most efficient of the metal oxides that were studied in releasing Cl2 at approximately 375°C. This reaction would allow Cl2 to be able to participate in the chlorination of aromatic structures, which could then be followed by the formation of dual ring structures in a second metal-catalysed reaction21,22.
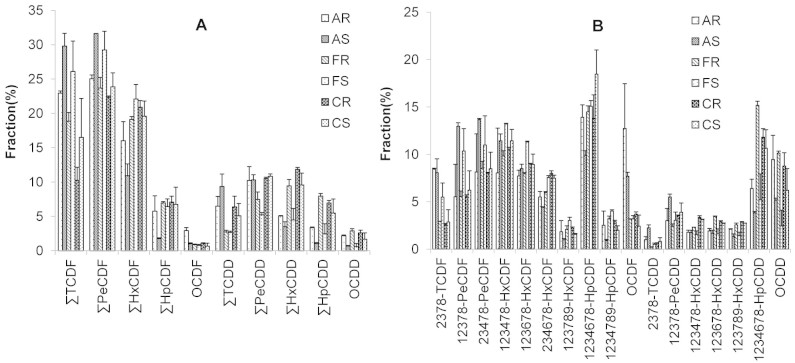
Figure 2 also shows that higher concentrations of PCDD/Fs were found in the residues than in the smoke when CuO was present. However, the PCDD/F distributions produced in the presence of Al2O3 and Fe2O3 were different, the PCDD/F concentrations being higher in the smoke than in the residues. We concluded, therefore, that the metal-catalysed PCDD/F formation reaction occurred on the metal oxide surfaces19. The PCDD/F homologue and congener profiles produced from sucralose heated in the presence of metal oxides were similar to the PCDD/F profiles formed from sucralose heated in stainless steel utensils (Fig. 4). This suggests that the metal oxides acted as catalysts, promoting PCDD/F formation, but did not alter the PCDD/F formation pathways.
Figure 4. Polychlorinated dibenzo-p-dioxin and dibenzofuran (PCDD/F) (A) homologue patterns and (B) congener profiles found in the smoke and residues formed when sucralose was heated in the presence of 1 g of Al2O3, Fe2O3, or CuO at 400°C.
The PCDFs were found at higher concentrations than the PCDDs in all cases (Figs. 3 and 4), suggesting that PCDD/Fs might be produced when a carbon matrix is broken down, in a series of oxidation and chlorination reactions, called de novo pathways23. The de novo synthesis of PCDD/Fs mostly occurred between 200 and 400°C, giving a PCDF/PCDD ratio of more than one24. It has also been reported that the oxidation of chlorobenzenes (CBzs) produces more PCDFs than PCDDs25. Polycyclic aromatic hydrocarbons (PAHs) and CBzs are considered to be precursors for PCDD/F formation19,26. Although no PAHs or CBzs were detected in the sucralose before it was heated, both PAHs and CBzs were found, along with PCDD/Fs, in the smoke. CBzs were also detected in the residues. This indicates that compounds containing benzene rings can be formed, chlorinated, and oxidized when sucralose is heated in the presence of metal oxides.
Our data indicate that sucralose may produce significant amounts of PCDD/Fs when heated in stainless steel utensils or in metal utensils in the presence of rust (Al2O3, Fe2O3, or CuO) at high temperatures. Occupational exposure to PCDD/Fs has attracted concern27. The improper use of sucralose at high temperatures may result in human exposure to PCDD/Fs.
Methods
Pesticide-grade hexane, dichloromethane, and acetone were purchased from J. T. Baker (Phillipsburg, NJ, USA). Sucralose (purity > 98%, HPLC grade) was purchased from Sigma-Aldrich, Inc. (St. Louis, MO, USA). Al2O3, Fe2O3, and CuO were purchased from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China). 13C-labelled PCDD/F standards and deuterated PAHs (d-PAHs) (catalogue no. L429) were purchased from Wellington Laboratories Inc. (Guelph, ON, Canada), and 13C-labelled CBz standards (catalogue no. EM-1725A) were purchased from Cambridge Isotope Laboratories, Inc. (Andover, MA, USA).
The experimental system and heating procedures used have been described previously by Dong et al. (2011, 2013)16,18. The tetra- to octa-chlorinated PCDD/F homologue groups and 17 individual 2,3,7,8-substituted PCDD/F congeners in the smoke and the residues were analysed following the U.S. Environmental Protection Agency method 1613. The sucralose residues after the heat tests were treated with 1 mol L−1 HCl and left to equilibrate overnight, washed with deionized water until the washings had a neutral pH, then freeze-dried. The sample extraction, clean-up, and instrumental analysis for the PCDD/Fs were conducted following the methods published by Wu et al. (2011)17. The ions monitored in the gas chromatograph-high resolution mass spectrometer analysis of PCDD/Fs are given in the Supplementary Table S1.
The smoke samples were analysed for PAHs following the Californian Environmental Protection Agency method 429. Known amounts of d-PAHs were added to the sample extracts, cleaned using gel permeation chromatography and then using a multilayer column filled with silica gel and neutral alumina. The extracts were then analysed for PAHs using an Agilent 6890/5973 N gas chromatograph-mass spectrometer (Agilent Technologies Inc., Santa Clara, CA, USA) equipped with an HP-5MS capillary column (30 m long, 0.32 mm i.d., 0.25 μm film thickness; Agilent Technologies Inc.) and operated in selected ion monitoring (SIM) mode. The ions monitored in the gas chromatograph-mass spectrometer analysis of PAHs are given in the Supplementary Table S2.
The samples were extracted and cleaned-up for the analysis of the tetra-, penta-, and hexa-chlorinated CBzs following a procedure for “dioxin-like” polychlorinated biphenyl analysis that has been published previously9, and known amounts of 13C-labelled CBzs were added to the samples before being extracted. The CBzs were analysed using an Agilent 6890/5973 N instrument equipped with an HP-5MS capillary column (30 m long, 0.32 mm i.d., 0.25 μm film thickness) and operated in selected ion monitoring mode, monitoring the two most abundant ions in the molecular ion clusters. The CBz isomers were quantified using the 13C-labelled CBzs as isotope-dilution internal standards. The ions monitored in the gas chromatograph-mass spectrometer analysis of CBzs are given in the Supplementary Table S3.
All of the experiments were run at least in duplicate. The relative percentage differences between the target compound concentrations in the duplicate experiments were less than 37%. The recoveries of the 13C-labelled PCDD/F internal standards were 60–110%, the d-PAH recoveries were 68–92%, and the 13C-labelled CBz recoveries were 60–85%. The limits of detection (LODs; defined as the concentration giving a signal-to-noise ratio of 3) for the PCDD/Fs, PAHs, and CBzs were 0.01–0.4 pg g−1, 4–30 pg g−1, and 4–15 pg g−1, respectively. The concentrations of all of the analytes in the blanks and the unheated sucralose were near or below the LODs, so no blank correction was performed.
Author Contributions
S.D. and G.L. conceived and designed the experiments. S.D. performed the experiments. S.D. and J.H. analysed the data. S.D. wrote and M.Z. reviewed the manuscript.
Supplementary Material
Supplementary Information
Acknowledgments
This study was supported by the Project of Academy (Chinese Academy of Sciences) –Location (Guizhou province) Cooperation launched in 2012 and the National Natural Science Foundation of China (20777086 and 20921063).
References
- Knight I. The development and applications of sucralose, a new high-intensity sweetener. Can. J. Physiol. Pharm. 72, 435–439 (1994). [DOI] [PubMed] [Google Scholar]
- Grotz V. L. & Munro I. C. An overview of the safety of sucralose. Regul. Toxicol. Pharm. 55, 1–5 (2009). [DOI] [PubMed] [Google Scholar]
- Torres C. I. et al. Fate of sucralose during wastewater treatment. Environ. Eng. Sci. 28, 325–331 (2011). [Google Scholar]
- Tollefsen K. E., Nizzetto L. & Huggett D. B. Presence, fate and effects of the intense sweetener sucralose in the aquatic environment. Sci. Total Environ. 438, 510–516 (2012). [DOI] [PubMed] [Google Scholar]
- Barndt R. L. & Jackson G. Stability of Sucralose in Baked Goods. Food Technol. 44, 62–66 (1990). [Google Scholar]
- Chapello W. The use of sucralose in baked goods and mixes. Cereal Food World 43, 716–717 (1998). [Google Scholar]
- Brown R. J., Walter M. & Rother K. I. Ingestion of diet soda before a glucose load augments glucagon-like peptide-1 secretion. Diabetes Care 32, 2184–2186 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown R. J., Walter M. & Rother K. I. Effects of diet soda on gut hormones in youths with diabetes. Diabetes Care 35, 959–964 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pepino M. Y., Tiemann C. D., Patterson B. W., Wice B. M. & Klein S. Sucralose affects glycemic and hormonal responses to an oral glucose load. Diabetes Care April 30, 10.2337/dc12-2221 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abou-Donia M. B., El-Masry E. M., Abdel-Rahman A. A., McLendon R. E. & Schiffman S. S. Splenda alters gut microflora and increases intestinal P-glycoprotein and cytochrome P-450 in male rats. J. Toxicol. Environ. Heal. A 71, 1415–1429 (2008). [DOI] [PubMed] [Google Scholar]
- US FDA (United States Food and Drug Administration). Food additives permitted for direct addition to food for human consumption; sucralose. 21CFR Part 172 [Docket No. 87F-0086]. Federal Register Vol. 63 (No. 64) April 3, 16417–16433 (1998). [Google Scholar]
- Sasaki Y. F. et al. The comet assay with 8 mouse organs: results with 39 currently used food additives. Mutat. Res. 519, 103–119 (2002). [DOI] [PubMed] [Google Scholar]
- Schiffman S. S. Rationale for further medical and health research on high-potency sweeteners. Chem. Senses 37, 671–679 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bannach G., Almeida R. R., Lacerda L., Schnitzler E. & Ionashiro M. Thermal stability and thermal decomposition of sucralose. Eclética Química 34, 21–26 (2009). [Google Scholar]
- Rahn A. & Yaylayan V. A. Thermal degradation of sucralose and its potential in generating chloropropanols in the presence of glycerol. Food Chem. 118, 56–61 (2010). [Google Scholar]
- Dong S., Wu J., Liu G., Zhang B. & Zheng M. Unintentionally produced dioxin-like polychlorinated biphenyls during cooking. Food Cont. 22, 1797–1802 (2011). [Google Scholar]
- Wu J., Dong S., Liu G., Zhang B. & Zheng M. Cooking process: A new source of unintentionally produced dioxins? J. Agric. Food Chem. 59, 5444–5449 (2011). [DOI] [PubMed] [Google Scholar]
- Dong S., Liu G., Zhang B., Gao L. & Zheng M. Formation of polychlorinated naphthalenes during the heating of cooking oil in the presence of high amounts of sucralose. Food Cont. 31, 1–5 (2013). [Google Scholar]
- Altarawneh M., Dlugogorski B. Z., Kennedy E. M. & Mackie J. C. Mechanisms for formation, chlorination, dechlorination and destruction of polychlorinated dibenzo-p-dioxins and dibenzofurans (PCDD/Fs). Prog. Energ. Combust. 35, 245–274 (2009). [Google Scholar]
- Hisham M. W. M. & Benson S. W. Thermochemistry of the Deacon process. J. Phys. Chem. 99, 6194–6198 (1995). [Google Scholar]
- Gullett B. K., Bruce K. R., Beach L. O. & Drago A. M. Mechanistic steps in the production of PCDD and PCDF during waste combustion. Chemosphere 25, 1387–1392 (1992). [Google Scholar]
- Gullett B., Sarofim A., Smith K. & Procaccini C. The role of chlorine in dioxin formation. Process Saf. Environ. 78, 47–52 (2000). [Google Scholar]
- Iino F., Imagawa T., Takeuchi M. & Sadakata M. De novo synthesis mechanism of polychlorinated dibenzofurans from polycyclic aromatic hydrocarbons and the characteristic isomers of polychlorinated naphthalenes. Environ. Sci. Technol. 33, 1038–1043 (1999). [Google Scholar]
- Everaert K. & Baeyens J. The formation and emission of dioxins in large scale thermal processes. Chemosphere 46, 439–448 (2002). [DOI] [PubMed] [Google Scholar]
- Sommeling P. M., Mulder P. & Louw R. Formation of PCDFs during chlorination and oxidation of chlorobenzene in chlorine/oxygen mixtures around 340°C. Chemosphere 29, 2015–2018 (1994). [Google Scholar]
- Weber R. et al. Formation of PCDF, PCDD, PCB, and PCN in de novo synthesis from PAH: Mechanistic aspects and correlation to fluidized bed incinerators. Chemosphere 44, 1429–1438 (2001). [DOI] [PubMed] [Google Scholar]
- Wen S. et al. Elevated levels of urinary 8-hydroxy-2′-deoxyguanosine in male electrical and electronic equipment dismantling workers exposed to high concentrations of polychlorinated dibenzo-p-dioxins and dibenzofurans, polybrominated diphenyl ethers, and polychlorinated biphenyls. Environ. Sci. Technol. 42, 4202–4207 (2008). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Information