Abstract
Pellagra is a photosensitivity syndrome characterized by three “D's”: diarrhea, dermatitis, and dementia as a result of niacin deficiency. However, the molecular mechanisms of photosensitivity dermatitis, the hallmark abnormality of this syndrome, remain unclear. We prepared niacin deficient mice in order to develop a murine model of pellagra. Niacin deficiency induced photosensitivity and severe diarrhea with weight loss. In addition, niacin deficient mice exhibited elevated expressions of COX-2 and PGE syntheses (Ptges) mRNA. Consistently, photosensitivity was alleviated by a COX inhibitor, deficiency of Ptges, or blockade of EP4 receptor signaling. Moreover, enhanced PGE2 production in niacin deficiency was mediated via ROS production in keratinocytes. In line with the above murine findings, human skin lesions of pellagra patients confirmed the enhanced expression of Ptges. Niacin deficiency-induced photosensitivity was mediated through EP4 signaling in response to increased PGE2 production via induction of ROS formation.
Pellagra, first described nearly 250 years ago, is a remarkable chronic wasting disorder that results from a marked cellular deficiency of niacin, a water-soluble vitamin also known as vitamin B3. Pellagra is a photosensitivity syndrome characterized by a cardinal clinical feature - the often quoted three ‘Ds’: dermatitis, diarrhea, and dementia1. Pellagra continues to be a significant health issue in developing countries where corn and its products are the major food source or in ethnic populations with diets deficient in niacin2. In addition, this photosensitivity syndrome still exists in Europe and USA in chronic alcoholics, food faddists or people with drug addictions and patients suffering from malabsorption2.
Pellagra is derived from the Italian word, “pelle” meaning skin, and “agra” signifying rough, in reference to the thickened, rough skin of pellagra patients. Thus, the consistent clinical feature in pellagra is photosensitivity dermatitis and it begins as an erythema at the sun-exposed area. UV-induced dermatitis is characterized by erythema and edema as well as the induction of a wide variety of inflammatory mediators, including cytokines, nitric oxide (NO), and cyclooxygenase (COX) metabolites3,4,5. However, the pathogenic inflammatory mediators involved in photosensitivity dermatitis in pellagra have yet to be determined.
The prostanoid family is one of the mediators involved in UVB-induced skin inflammation. They are oxygenated metabolites of arachidonic acid produced by sequential catalysis of COX. Prostaglandin (PG) E2, PGD2, PGF2, PGI2, and thromboxane A2 (TXA2) comprise prostanoids, which exert a variety of physiological actions by acting on G protein-coupled receptors including the four subtypes of PGE receptors (EP1, EP2, EP3 and EP4)6,7. Previous studies showing that COX inhibitors attenuate UVB-induced skin inflammation suggest the involvement of prostanoids in this condition in humans8. Furthermore, PGE2 induces angiogenesis and vasodilation in the skin microenvironment9. Therefore, PGs are a potential cause of photosensitivity dermatitis in pellagra. In this study we have established a mouse model of photosensitivity dermatitis and diarrhea induced by niacin deficiency. We used pharmacological and genetic approaches to address the role of PGE2 in the pathogenesis of photosensitivity dermatitis in pellagra.
Results
A niacin antagonist 6-aminonicotinamide (6-AN) enhanced UVB-induced skin inflammation
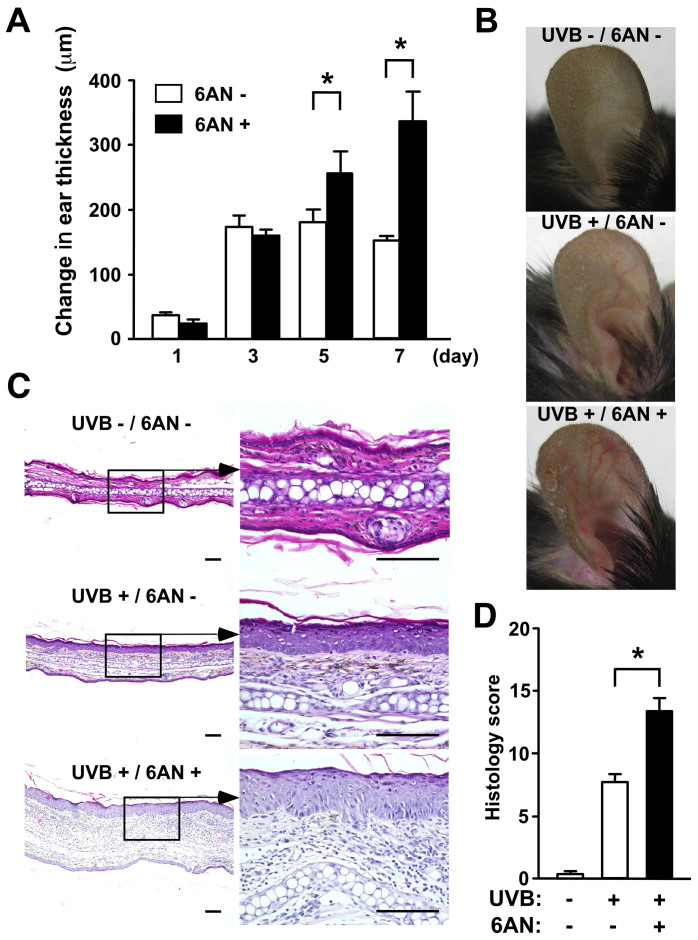
We treated C57BL/6 mice with intraperitoneal (i.p.) injections of 12.5 mg/kg of 6-AN and exposed them to UVB at a dose of 5 kJ/m2. UVB exposure-induced skin inflammation was measured by changes in ear thickness and histology. Mice treated with 6-AN 24 hours before UVB exposure exhibited enhanced ear swelling at 5 and 7 days after UVB exposure compared to control mice with no UVB or 6-AN treatments (Fig. 1A). The UVB-induced redness and vasodilation were prominent in 6-AN-treated mice (Fig. 1B). In addition, the skin of 6-AN-treated mice after UVB exposure exhibited hyperkeratosis, infiltration of neutrophils and lymphocytes, and dermal edema (Fig. 1C). Each histological finding was scored (Supplementary Table S1), and the sum of each score in 6-AN-treated mice was significantly higher than that in control mice (Fig. 1D). To confirm the 6-AN activity in the UVB irradiated pellagra model mice, we measured the levels of niacin-derived nicotinamide adenine dinucleotide (NAD) and NADH. NAD+ and NADH production induced by UVB irradiation was inhibited by intraperitoneal administration of 6-AN (Supplementary Fig. S1). These data indicate that intraperitoneal administration of 6-AN is active in the UVB irradiated skin and this antagonist modulates the production of NAD+ and NADH.
Figure 1. Administration of the niacin antagonist (6-AN) enhanced UVB-induced ear skin swelling.
(A) Ear swelling responses were monitored and the increase in ear thickness was calculated. Data are presented as means ± SEM. *, P < 0.05. (B) Clinical photographs. (C) H&E staining of the skin after UVB exposure. Original magnification, x200. Scale bar 100 μm. (D) The histological samples were scored. Data are presented as means ± SEM.*, P < 0.05.
Another hallmark of pellagra is diarrhea. Mice treated with 6-AN exhibited weight loss in a dose dependent manner (Supplementary Fig. S2). Clinical and histological score obtained from 6-AN-treated mice were significantly higher than that from control mice. Therefore, diarrhea, one of the symptoms in pellagra, is reproduced by the niacin blockade in mice.
We also performed an additional experiment to rule out toxicity of 6-AN treatment. 25 mg/kg of 6-AN was administered with intraperitoneal injection to C57BL/6 mice, and peripheral blood samples were collected five days after the treatment. The number of T cells was comparable between by treatment with PBS (6.08 ± 0.72 × 104/ml) and 6-AN (6.27 ± 0.77 × 104/ml). These data provide some supporting evidence that intraperitoneal administration of 25 mg/kg of 6-AN was not toxic to mice at least in T cell subsets.
A deficiency in dietary niacin exaggerated UVB-induced skin inflammation
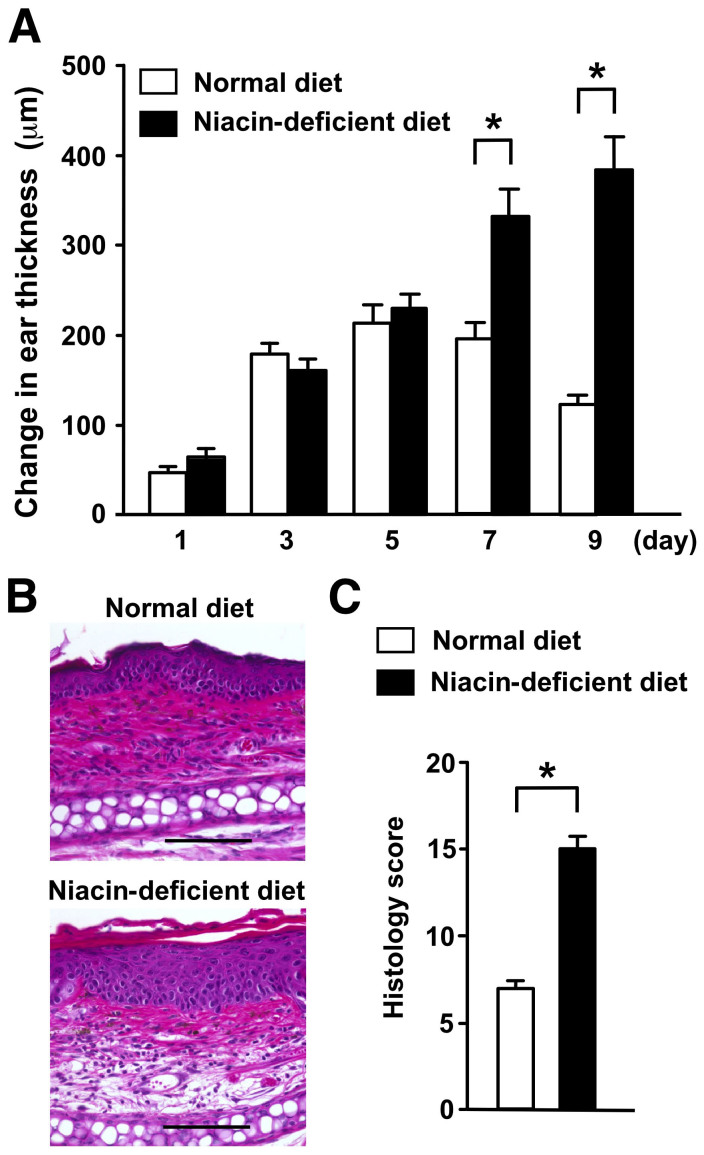
To confirm whether niacin deficiency can recapitulate the findings obtained using the niacin antagonist, we fed mice a niacin-deficient diet for two weeks and then treated them with 5 kJ/m2 UVB. Mice fed with a niacin-deficient diet showed markedly prolonged and enhanced ear swelling responses to UVB exposure compared with those fed with a normal diet (Fig. 2A). Histological examination of UVB-irradiated skin lesions of mice fed with a niacin deficient diet revealed more pronounced epidermal hyperplasia, hyperkeratosis and a perivascular inflammatory cell infiltrate compared to the skin of mice fed with a normal diet (Fig. 2B). Consistently, the histological score of skin lesions from UVB-treated niacin deficient mice was higher than that of irradiated mice fed with a normal diet (Fig. 2C). These histological findings were consistent with those of cutaneous pellagra lesions in humans and with those in mice treated with 6-AN. Thus, niacin deficiency reproduced the UVB-induced photosensitivity caused by treatment with the niacin antagonist 6-AN.
Figure 2. Mice fed with a niacin-deficient diet recapitulate enhanced UVB-induced skin inflammation.
(A) C57BL/6 mice fed with a niacin-deficient diet and or a normal control diet were exposed to UVB radiation and examined for ear swelling responses at the indicated time points. Data are presented as means ± SEM. *, P < 0.05. (B) H&E staining of ear samples collected from mice fed with a niacin-deficient or a control diet after UVB irradiation. Original magnification, x200. Scale bar 100 μm. (C) The histological findings were scored. Data are presented as means ± SEM. *, P < 0.05.
Exacerbation of UVB-induced skin inflammation by 6-AN was dependent on COX-2 (Ptgs2)
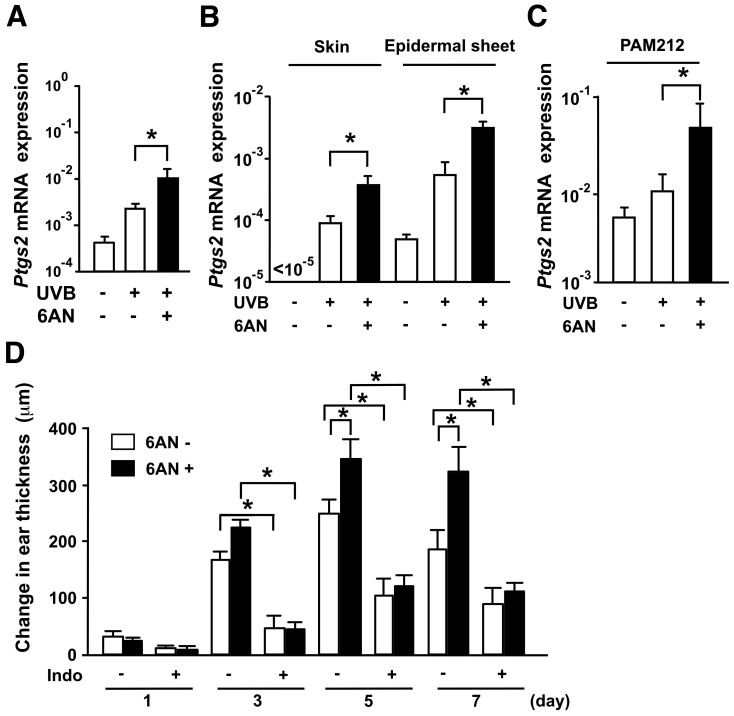
Next, we sought to address the underlying mechanism of pellagra using 6-AN-induced photosensitivity in mice as a model. UVB-induced skin inflammation is generated by chemical mediators including cytokines, NO and COX metabolites5. We first examined the mRNA levels of inducible nitric oxide synthase (iNOS) (Nos2), Ptgs2, interleukin (IL)-1, IL-8, IL-10 and TNF-α, all of which are known to be involved in UVB-induced skin inflammation5,10. Among these, Ptgs2 mRNA expression after UVB irradiation was enhanced when mice were treated with 6-AN 24 hours before UVB exposure (Supplementary Fig. S3). The effect of 6-AN was also observed 7 days after UVB exposure (Fig. 3A).
Figure 3. Quantitative PCR analysis of mRNA expression for Ptgs2 and attenuation of UVB-induced ear skin swelling response by Ptgs2 blockade.
(A) The amount of Ptgs2 mRNA is expressed as the relative amount of the mRNA normalized to Actb. Data are presented as means ± SEM. *, P < 0.05. (B) The amounts of mRNA for Ptgs2 in the whole skin and epidermal sheets were measured. Data are presented as means ± SEM. *, P < 0.05. (C) Ptgs2 mRNA expression in PAM212 was examined. Data are presented as means ± SEM. *, P < 0.05. (D) C57BL/6 mice were treated with or without 6-AN and pretreated with or without indomethacin (Indo), and then exposed to UVB. The increase in ear thickness was calculated. Data are presented as means ± SEM. *, P < 0.05.
To identify the source of Ptgs2 in 6-AN-induced photosensitivity, we measured the expression level of Ptgs2 mRNA in the whole skin and epidermal sheets isolated from the skin 7 days after 5 kJ/m2 UVB irradiation. Ptgs2 mRNA expression in response to UVB was enhanced by 6-AN treatment in both whole skin and epidermal sheets (Fig. 3B). Consistent with this observation, Ptgs2 mRNA expression after 30 mJ/cm2 UVB exposure in the keratinocyte cell line PAM 212 was markedly increased by treatment with 250 μM of 6-AN in vitro (Fig. 3C). These results suggest that UVB-induced Ptgs2 mRNA induction in epidermal keratinocytes is enhanced upon blockade of niacin signaling.
To confirm the direct involvement of Ptgs2 in the enhancement of UVB-induced skin inflammation by 6-AN, we treated UVB-irradiated mice with indomethacin, a COX inhibitor. Treatment with indomethacin significantly reduced ear swelling at each time point tested (Fig. 3D). Importantly, indomethacin completely inhibited the aggravating effect of 6-AN on UVB-induced dermatitis (Fig. 3D). This finding confirms the role of Ptgs2 in the development of photosensitivity induced by 6-AN. These findings suggest that enhancement of Ptgs2 expression might be the primary candidate of photosensitivity induced by niacin blockade.
Lack of niacin increased skin Ptges level in mice and humans
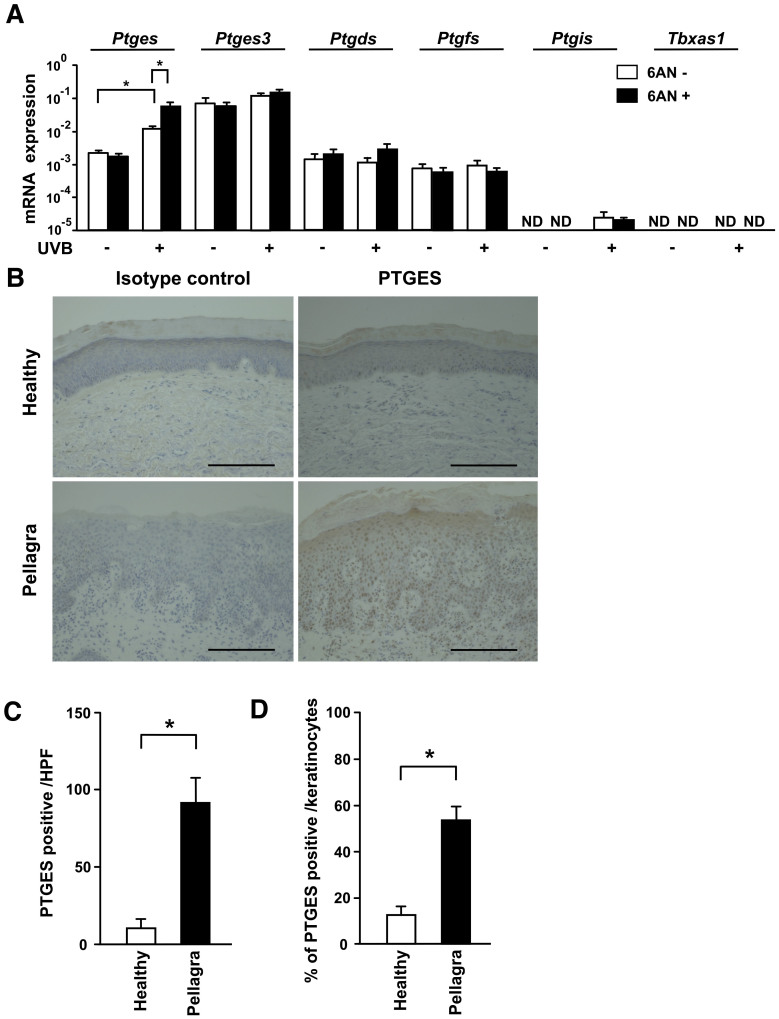
The above results suggest that 6-AN-induced photosensitivity is mediated by prostanoids produced by Ptgs2. These prostanoids are comprised of PGE2, PGD2, PGF2, PGI2 and TXA2, which are produced by PGE syntheses (Ptges and Ptges3), Ptgds, Ptgfs, Ptgis, and Tbxas1, respectively. We therefore sought to identify which prostanoid mediates the photosensitivity induced by 6-AN. Among the Ptges, Ptges3, Ptgds, Ptgfs, Ptgis and Tbxas1 mRNA levels in the skin 7 days after UVB exposure, Ptges and Ptgis mRNA was increased in the UVB-irradiated skin and the Ptges mRNA level was additionally augmented by 6-AN (Fig. 4A). Although treatment with 6-AN enhanced both Ptgs2 and Ptges mRNA expression in the gut (Supplementary Fig. S4), treatment with indomethacin (4 mg/kg/day) did not ameliorate 6-AN (12.5 mg/kg) induced diarrhea or weight loss (body weight: 6-AN, 17.1 ± 0.30; 6-AN plus indomethacin, 16.8 ± 0.46, mean ± SEM of five mice). Since treatment with indomethacin itself induces gastrointestinal ulceration and bleeding11, it is difficult to assess whether PGE2 mediate the development of diarrhea in our model. Together with the evaluation of dementia, further studies will be required in the future.
Figure 4. Increased expression of Ptges in the skin of mice and humans as a result of niacin deficiency.
(A) The levels of mRNA were measured and expressed as relative quantities normalized to Actb. ND, not detectable. (B) Skin specimens taken from healthy subjects (n = 3) and patients with pellagra (n = 3) were immunostained with anti-PTGES antibody. Representative samples are shown. Scale bar 100 μm. (C; D) The number and percentage of PTGES positive cells were counted. Data are presented as means ± SEM. *, P < 0.05. HPF, high power field.
To address whether the above findings in photosensitivity dermatitis in mice are applicable to human pellagra, we examined the expression level of the PGE synthase that is encoded by the PTGES gene in humans. Skin samples from patients with pellagra were immunostained with an anti-PTGES antibody. Keratinocytes isolated from the dorsal surface of the hands of pellagra patients were positive for PTGES but not keratinocytes at the same area from healthy subjects (Fig. 4B). The numbers and percentages of PTGES positive cells in keratinocytes from patients with pellagra were significantly higher than those in normal healthy controls, which in combination with the data above, suggests the possible involvement of PTGES in the development of photosensitivity in human pellagra (Fig. 4C and D).
PGE2 mediated photosensitivity induced by the niacin antagonist 6-AN
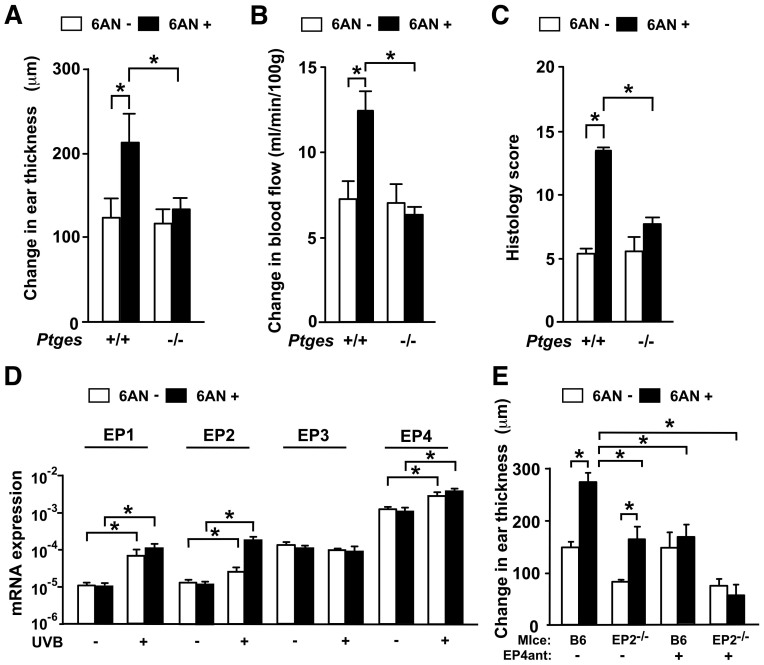
To further confirm the involvement of PGE2 in niacin antagonist-induced photosensitivity, we tested the ear swelling response in mice deficient for Ptges (Ptges−/− mice). The ear swelling induced by UVB was significantly attenuated in Ptges−/− mice 7 days after UVB exposure with the level of ear swelling almost similar to the baseline without 6-AN treatment (Fig. 5A).
Figure 5. Impaired UVB-induced ear skin swelling upon blockade of the PGE2-EP4 pathway.
(A) Ptges+/+ and Ptges−/− mice were treated with 6-AN and exposed to UVB. Their ear swelling responses were examined. Data are presented as means ± SEM. *, P < 0.05. (B) Blood flow in the ears of Ptges+/+ and Ptges−/− mice at day 5 after UVB irradiation. Data are presented as means ± SEM. *, P < 0.05. (C) The histological findings in Ptges+/+ and Ptges−/− mice were scored. Data are presented as means ± SEM. *, P < 0.05. (D) Expression of EP1, EP2, EP3 and EP4 mRNAs after UVB exposure. Data are presented as means ± SEM. *, P < 0.05. (E) EP4 receptor signaling blockade impairs 6-AN-induced photosensitivity. Data are presented as means ± SEM. *, P < 0.05.
After UVB exposure, an increase in local microenvironmental blood flow is essential to establish UVB-induced skin inflammation. Therefore, we assessed the blood flow in the abdominal skin after UVB irradiation using a laser doppler device. The blood flow reached its peak 5 days after UVB exposure. Consistent with the kinetics of the swelling response, the niacin antagonist increased the blood flow about twofold (Fig. 5B). Such an enhancement was not observed in Ptges−/− mice (Fig. 5B). In agreement with these findings, the exacerbated histological findings induced by 6-AN were attenuated in Ptges−/− mice (Supplementary Table S2 and Fig. 5C). These data suggest that PGE2 enhance the blood flow and the accumulation of inflammatory cells in the microenvironment to establish photosensitivity induced by the niacin antagonist.
PGE2 elicits its physiological functions by binding to four different types of G protein-coupled receptors, known as EP1, EP2, EP3 and EP4. All four receptors are expressed in the mouse ear7. The levels of EP1, EP2 and EP4 mRNAs were enhanced 7 days after UVB irradiation while that of EP3 was unchanged (Fig. 5D).
To identify the responsible PGE2 receptors in 6-AN-induced photosensitivity, we irradiated UVB to EP1−/−, EP2−/−, EP3−/− and control C57BL/6 mice in the presence or absence of 6-AN. Since EP4−/− mice cannot be backcrossed to C57BL/6 mice because of neonatal death by patent ductus arteriosus12, we used the EP4-selective antagonist, 4-(4-Cyano-2-(2-(4-fluoronaphthalen-1-yl) propionylamino) phenyl) butyric acid (AE3-208) instead. C57BL/6 mice treated with or without the EP4-selective antagonist AE3-208 were exposed to UVB in the presence or absence of 6-AN. 7 days after UVB exposure, the amounts of ear swelling responses of EP1−/− and EP3−/− mice were comparable to that of control C57BL/6 mice. The UVB-induced ear swelling in EP2−/− mice was attenuated compared to that in control mice although the ear swelling in EP2−/− mice was augmented by treatment with 6-AN (Fig. 5E). This indicates that EP2 is involved in the UVB-induced skin inflammation, but is unlikely to mediate photosensitivity induced by blockade of niacin signaling. On the other hand, 6-AN-induced photosensitivity was not observed in C57BL/6 mice by treatment with the EP4 antagonist AE3-208 (Fig. 5E). To clarify whether EP4 expression in response to PGE2 contributes to 6-AN-induced photosensitivity, we neutralized both EP2 and EP4 by administering the EP4 antagonist AE3-208 to EP2−/− mice. The UVB-induced ear swelling in EP2−/− mice treated with AE3-208 was comparable to that in EP2−/− mice, but the 6-AN-induced photosensitivity seen in EP2−/− mice was completely abrogated by the additional treatment with the EP4 antagonist at 7 days after UVB exposure (Fig. 5E). These findings suggest that enhanced production of PGE2 acts on EP4 to induce the niacin-antagonist-induced photosensitivity.
PGE2 signaling via reactive oxygen species (ROS) formation mediated 6-AN-induced photosensitivity
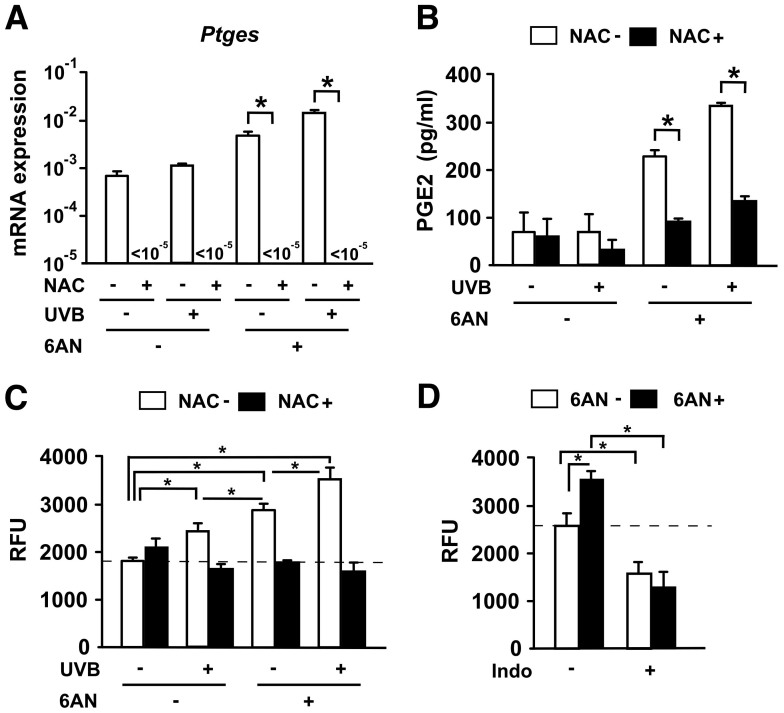
Our next question is how production of PGE2 is enhanced under niacin deficient conditions. It has been reported that niacin affects not only PGs but also intracellular redox potential13, and that ROS mediates skin inflammation through modulating oxidative stress14. Therefore, it is possible that ROS generated at the skin of pellagra modulate the production of PGE2. We analyzed the mRNA expression of Ptges in keratinocytes cultured primarily from C57BL/6 mice. Keratinocytes were incubated for 24 hours with or without a 20 mM of ROS inhibitor, N-acetylcysteine (NAC) (Sigma-Aldrich, St. Louis, MO), in the presence or absence of 250 μM of 6-AN after 30 mJ/cm2 UVB exposure. Intriguingly, the level of Ptges was significantly attenuated by treatment with NAC (Fig. 6A). The culture supernatants were subjected to enzyme-linked immunosorbent assay (ELISA) to quantify PGE2. Consistently, the production of PGE2 was significantly reduced by NAC treatment (Fig. 6B). We observed that ROS formation in keratinocytes increased significantly under stimulation with both UVB and 6-AN, and that the treatment with the ROS inhibitor, NAC, resulted in a reduction in the levels of ROS (Fig. 6C). These results suggest that niacin deficiency was mediated via induction of ROS formation in keratinocytes. To further confirm the involvement of ROS production in vivo, UVB-irradiated mice were given indomethacin (4 mg/kg/day) and the skin samples were collected 7 days after the treatment. The skin samples were homogenized and then the supernatants were analyzed. The production of ROS after UVB irradiation was significantly increased in vivo (Fig. 6D). In addition, this production was completely inhibited by treatment with indomethacin. These data raise the possibility that niacin deficiency promotes PGE2 production via ROS formation, resulting in the enhancement of UVB-induced skin inflammation.
Figure 6. Niacin deficiency promotes PGE2 production via ROS formation.
(A) The level of mRNA for Ptges in keratinocytes was measured. Data are presented as means ± SEM. *, P < 0.05. (B) Keratinocyte production of PGE2 in culture supernatants were quantified. Data are presented as means ± SEM. *, P < 0.05. (C) Keratinocytes were collected and ROS production was measured (represented as relative fluorescence units, RFU). Data are presented as means ± SEM. *, P < 0.05. (D) UVB-irradiated skin with or without 6-AN treatment were administered with indomethacin (Indo), and then collected at day 7 after UVB exposure for measurement of ROS production. Data are presented as means ± SEM. *, P < 0.05.
Discussion
Using either a niacin antagonist 6-AN and a niacin-deficient diet, we have established the first mouse model of pellagra which manifests photosensitivity dermatitis - one of the most striking clinical manifestations of pellagra in humans. Because our animal pellagra model involves photosensitivity dermatitis, it is especially useful for the analysis of its pathogenesis. Consistent with findings in human pellagra15, we demonstrate that UVB-induced skin inflammation is enhanced and prolonged in C57BL/6 mice treated with the niacin antagonist 6-AN. Consistently, the histology findings in these mice were fundamentally the same as those found in human pellagra15. In addition, we demonstrated that COX-2 (Ptgs2) expression was enhanced in 6-AN-induced photosensitivity, and that the photosensitivity was attenuated by a COX inhibitor indomethacin. Moreover, Ptges−/− mice or mice treated with the EP4 antagonist AE3-208 did not exhibit 6-AN-induced photosensitivity. The above findings suggest that PGE2 mediates photosensitivity dermatitis in pellagra through EP4 signaling. It has been reported that Ptgs2 affects neutrophil adhesion and recruitment in several inflammatory diseases16,17. Therefore, we speculate that neutrophil infiltration enhanced by niacin deficiency in our model is derived from Ptgs2 at least in part.
Niacin is required for the synthesis of two coenzyme molecules: NAD and Nicotinamide adenine dinucleotide phosphate (NADP)18. These coenzyme deficiency is considered to induce photosensitivity in pellagra by reduction of photorepair2. Therefore, tissues with a rapid cell turnover, such as the skin and the intestine, would be expected to be the sites of major metabolic derangement if cellular energy transfer were impaired19,20. Since the niacin antagonist 6-AN converts NAD into 6-aminonicotinamide adenine dinucleotide (6-ANAD) that adversely affects cellular energy transfer reactions21, severe skin inflammation was observed in neonatal mice following treatment with 6-AN even without UVB irradiation as reported previously22. This effect seen in neonates was, however, not observed in our adult pellagra model. It remains unclear why the dermatitis in these adult animals depends on UVB irradiation. It is possible that the increase of 6-ANAD as well as a decrease in NAD and NADP may be required to develop UVB-mediated photosensitivity dermatitis in our adult pellagra model.
Xeroderma pigmentosum group A (XPA) gene-deficient mice develop skin cancer upon UVB exposure as a result of the inability to repair UVB-induced DNA damage23. Interestingly, in an XPA mouse model, expression of Ptgs2 in the skin was increased after UVB irradiation. Niacin is a precursor of NAD and NADP, both of which are important cofactors for the repair of UVB-induced DNA damage in the epidermis and are involved in nonredox reactions such as DNA repair24. Therefore, a deficiency in NAD and NADP in pellagra may also contribute to DNA damage possibly through the induction of Ptgs2, which may share some mechanism in common with the pathogenesis of XPA.
Initial uncertainty about the role of sun exposure in the pathogenesis of pellagra has been addressed by a number of authors with clinical photoprovocation studies. These studies show that sun exposure plays a central etiological role in the exposed site dermatitis of pellagra. However, no research group has fully investigated the action spectrum of the photosensitivity reaction in pellagra using in vivo phototesting methods2. There have been four case reports describing the photo-tests in pellagra patients2. The conclusions on action spectrum were different between them and there has been no compelling evidence on the character of light responsible for photosensitivity in pellagra. Therefore, clinical studies on phototesting in pellagra have yet to establish the action spectrum for this photosensitivity disorder. It was interesting to determine the action spectrum (UVB, UVA, UVB plus UVA, and visible light) of pellagra in our model.
Niacin is generally effective in the treatment of pellagra. On the other hand, niacin administration sometimes induces adverse effects, including niacin flushing, as a result of excessive production of PGD2 by mast cells and Langerhans cells25,26. On the other hand, a COX-2 inhibitor was effective in the control of our pellagra model without any side effect. Thus, the regulation of prostanoid production seems to be involved in several diseases related to niacin metabolism; however, it remains unclear how niacin metabolism controls PGE2 production. In this study, we hypothesized that ROS generated at the skin of pellagra modulates the production of PGE2, and found that ROS inhibitor suppressed the production of PGE2 induced by treatment with 6-AN and UVB irradiation. In line with this, previous evidence supports that ROS positively regulates proinflammatory gene expression in various immune signaling pathways27.
It has been shown that 10 mg/kg of 6-AN injected intraperitoneally results in cerebellar degeneration without any abnormality in internal organs28. This feature is consistent with the histological findings in necropsy cases of pellagra presented with various mental and neurological symptoms29. The dosage of 6-AN (12.5 mg/kg) contributes to dermatitis and diarrhea without toxicity in our model. Although we did not address whether 6-AN induces dementia that is another element of pellagra, this study raises the potential of this model as a useful tool to address the pathogenesis of pellagra.
Taken together, we have established a mouse model of pellagra and demonstrated that UVB-induced skin inflammation in a murine pellagra model is mediated through PGE2 via ROS production. In addition, our observation indicates the accumulation of PGE2 even in the skin lesion of human pellagra. Blockades of PGE2 and ROS production may be the new strategy for the treatment of pellagra.
Methods
Mice
Female C57BL/6 and BALB/c mice were purchased from Japan SLC (Hamamatsu, Japan). Ptges−/− mice were backcrossed to C57BL/6 mice for at least five generations30,31. Their wild-type littermates (Ptges+/+ mice) were used as controls. EP2−/− mice were generated as described32. EP2−/− mice were backcrossed 10 times to C57BL/6 mice. Mice were maintained on a 12 hours light/dark cycle under specific pathogen-free conditions. All protocols were approved by the Institutional Animal Care and Use Committee of the University of Occupational and Environmental Health.
Reagents
The niacin antagonist (6-AN) was obtained from MP Biomedicals (Solon, OH). 6-AN is an analogue of nicotinamide adenine dinucleotide phosphate (NADP) that impairs the conversion of tryptophan to niacin33. Mice were injected i.p. with 12.5 mg/kg of 6-AN or an equivalent volume of normal saline as a control 24 hours before UVB exposure. Indomethacin (Sigma-Aldrich, St. Louis, MO) was added to the drinking water at a dose of 4 mg/kg/day and administered to the animals throughout the entire experimental period, as reported previously34. This dose of indomethacin inhibits PGE2 production in mice in vivo35. An EP4-selective antagonist, AE3-208 was kindly provided by Ono Pharmaceutical Co., Osaka, Japan36. AE3-208 was administered (10 mg/kg/day) orally in the drinking water two days before UVB exposure and throughout the experiment. Niacin-deficient diet food and normal control diet food were purchased from the Oriental Yeast Co.,Ltd, Tokyo Japan. 20 mM of NAC was added to freshly isolated keratinocytes from C57BL/6 mice. Culture supernatants from keratinocytes were examined for PGE2 using a PGE2 express EIA kit-monoclonal from Cayman Chemical (Ann Arbor, MI).
UVB-induced skin inflammation model
For UVB-induced skin inflammation, mice were exposed to 5 kJ/m2 of UVB by a bank of four UVB lamps (Toshiba FL 20S, Toshiba Medical Supply, Tokyo, Japan) with an emission peak at 313 nm3,37. The irradiation was measured using an UVR-305/365 digital radiometer (Tokyo Kogaku Kikai K.K., Tokyo, Japan). The ear swelling was measured using an ear thickness gauge at the indicated time points after irradiation, and the ear thickness change was recorded3,37.
A statement identifying the institutional and/or licensing committee experimental approval
This study was approved by the Institutional Review Boards of our institutions (University of Occupational and Environmental Health, and Kyoto University Graduate School of Medicine), and informed written consent was obtained from all the subjects.
Author Contributions
K.S., A.S. and Y.N. performed most of the experiments with partial contributions from H.Y., T.N., J.S., K.N. and E.K.; S.U., J.N., S.A., M.N., S.N., Y.M., Y.T. and K.K. designed and supervised the study; K.S., T.N., M.N., Y.T. and K.K. wrote the manuscript.
Supplementary Material
Supplementary Information
Acknowledgments
This work was supported in part by grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
References
- Mercieri M. & Mercieri A. Images in clinical medicine: A photosensitive dermatitis in the intensive care unit. N Engl J Med 364, 361 (2011). [DOI] [PubMed] [Google Scholar]
- Wan P., Moat S. & Anstey A. Pellagra: a review with emphasis on photosensitivity. Br J Dermatol 164, 1188–1200 (2011). [DOI] [PubMed] [Google Scholar]
- Kabashima K. et al. Prostaglandin E2 is required for ultraviolet B-induced skin inflammation via EP2 and EP4 receptors. Lab Invest 87, 49–55 (2007). [DOI] [PubMed] [Google Scholar]
- Chang H. R., Tsao D. A., Wang S. R. & Yu H. S. Expression of nitric oxide synthases in keratinocytes after UVB irradiation. Arch Dermatol Res 295, 293–296 (2003). [DOI] [PubMed] [Google Scholar]
- Kiss F. & Anstey A. V. A review of UVB-mediated photosensitivity disorders. Photochem Photobiol Sci 12, 37–46 (2013). [DOI] [PubMed] [Google Scholar]
- Honda T., Tokura Y., Miyachi Y. & Kabashima K. Prostanoid receptors as possible targets for anti-allergic drugs: recent advances in prostanoids on allergy and immunology. Curr Drug Targets 11, 1605–1613 (2010). [DOI] [PubMed] [Google Scholar]
- Kabashima K. & Miyachi Y. Prostanoids in the cutaneous immune response. J Dermatol Sci 34, 177–184 (2004). [DOI] [PubMed] [Google Scholar]
- Gledhill K. et al. Prostaglandin-E2 is produced by adult human epidermal melanocytes in response to UVB in a melanogenesis-independent manner. Pigment Cell Melanoma Res 23, 394–403 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Syeda M. M. et al. Prostaglandin transporter modulates wound healing in diabetes by regulating prostaglandin-induced angiogenesis. Am J Pathol 181, 334–346 (2012). [DOI] [PubMed] [Google Scholar]
- Kuhn A., Ruland V. & Bonsmann G. Photosensitivity, phototesting, and photoprotection in cutaneous lupus erythematosus. Lupus 19, 1036–1046 (2010). [DOI] [PubMed] [Google Scholar]
- Imaoka H. et al. Exacerbation of indomethacin-induced small intestinal injuries in Reg I-knockout mice. Am J Physiol Gastrointest Liver Physiol 299, G311–319 (2010). [DOI] [PubMed] [Google Scholar]
- Segi E. et al. Patent ductus arteriosus and neonatal death in prostaglandin receptor EP4-deficient mice. Biochem Biophys Res Commun 246, 7–12 (1998). [DOI] [PubMed] [Google Scholar]
- Ganji S. H., Qin S., Zhang L., Kamanna V. S. & Kashyap M. L. Niacin inhibits vascular oxidative stress, redox-sensitive genes, and monocyte adhesion to human aortic endothelial cells. Atherosclerosis 202, 68–75 (2009). [DOI] [PubMed] [Google Scholar]
- Honda T., Egawa G., Grabbe S. & Kabashima K. Update of immune events in the murine contact hypersensitivity model: toward the understanding of allergic contact dermatitis. J Invest Dermatol 133, 303–315 (2013). [DOI] [PubMed] [Google Scholar]
- Sato M. et al. Pellagra-like erythema on sun-exposed skin of patients with anorexia nervosa. J Dermatol 38, 1037–1040 (2011). [DOI] [PubMed] [Google Scholar]
- Lemos H. P. et al. Prostaglandin mediates IL-23/IL-17-induced neutrophil migration in inflammation by inhibiting IL-12 and IFNgamma production. Proc Natl Acad Sci U S A 106, 5954–5959 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Profita M. et al. Chronic obstructive pulmonary disease and neutrophil infiltration: role of cigarette smoke and cyclooxygenase products. Am J Physiol Lung Cell Mol Physiol 298, L261–269 (2010). [DOI] [PubMed] [Google Scholar]
- Benavente C. A., Jacobson M. K. & Jacobson E. L. NAD in skin: therapeutic approaches for niacin. Curr Pharm Des 15, 29–38 (2009). [DOI] [PubMed] [Google Scholar]
- Hegyi J., Schwartz R. A. & Hegyi V. Pellagra: dermatitis, dementia, and diarrhea. Int J Dermatol 43, 1–5 (2004). [DOI] [PubMed] [Google Scholar]
- Brown T. M. Pellagra: an old enemy of timeless importance. Psychosomatics 51, 93–97 (2010). [DOI] [PubMed] [Google Scholar]
- Walker D. L. et al. Murine pharmacokinetics of 6-aminonicotinamide (NSC 21206), a novel biochemical modulating agent. Biochem Pharmacol 58, 1057–1066 (1999). [DOI] [PubMed] [Google Scholar]
- Aikawa H. & Suzuki K. Lesions in the skin, intestine, and central nervous system induced by an antimetabolite of niacin. Am J Pathol 122, 335–342 (1986). [PMC free article] [PubMed] [Google Scholar]
- Kuwamoto K. et al. Possible involvement of enhanced prostaglandin E2 production in the photosensitivity in xeroderma pigmentosum group A model mice. J Invest Dermatol 114, 241–246 (2000). [DOI] [PubMed] [Google Scholar]
- Halliday G. M. Inflammation, gene mutation and photoimmunosuppression in response to UVR-induced oxidative damage contributes to photocarcinogenesis. Mutat Res 571, 107–120 (2005). [DOI] [PubMed] [Google Scholar]
- Papaliodis D. et al. Niacin-induced “flush” involves release of prostaglandin D2 from mast cells and serotonin from platelets: evidence from human cells in vitro and an animal model. J Pharmacol Exp Ther 327, 665–672 (2008). [DOI] [PubMed] [Google Scholar]
- Dunbar R. L. & Gelfand J. M. Seeing red: flushing out instigators of niacin-associated skin toxicity. J Clin Invest 120, 2651–2655 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bauernfeind F. et al. Cutting edge: reactive oxygen species inhibitors block priming, but not activation, of the NLRP3 inflammasome. J Immunol 187, 613–617 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penkowa M. & Hidalgo J. IL-6 deficiency leads to reduced metallothionein-I + II expression and increased oxidative stress in the brain stem after 6-aminonicotinamide treatment. Exp Neurol 163, 72–84 (2000). [DOI] [PubMed] [Google Scholar]
- Ishii N. & Nishihara Y. Pellagra among chronic alcoholics: clinical and pathological study of 20 necropsy cases. J Neurol Neurosurg Psychiatry 44, 209–215 (1981). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uematsu S., Matsumoto M., Takeda K. & Akira S. Lipopolysaccharide-dependent prostaglandin E(2) production is regulated by the glutathione-dependent prostaglandin E(2) synthase gene induced by the Toll-like receptor 4/MyD88/NF-IL6 pathway. J Immunol 168, 5811–5816 (2002). [DOI] [PubMed] [Google Scholar]
- Kuroda E. et al. Silica crystals and aluminum salts regulate the production of prostaglandin in macrophages via NALP3 inflammasome-independent mechanisms. Immunity 34, 514–526 (2011). [DOI] [PubMed] [Google Scholar]
- Hizaki H. et al. Abortive expansion of the cumulus and impaired fertility in mice lacking the prostaglandin E receptor subtype EP(2). Proc Natl Acad Sci U S A 96, 10501–10506 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rawat D. K. et al. Glucose-6-phosphate dehydrogenase and NADPH redox regulates cardiac myocyte L-type calcium channel activity and myocardial contractile function. PLoS One 7, e45365 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kabashima K. et al. The prostaglandin receptor EP4 suppresses colitis, mucosal damage and CD4 cell activation in the gut. J Clin Invest 109, 883–893 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohn S. M., Schloemann S., Tessner T., Seibert K. & Stenson W. F. Crypt stem cell survival in the mouse intestinal epithelium is regulated by prostaglandins synthesized through cyclooxygenase-1. J Clin Invest 99, 1367–1379 (1997). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kabashima K. et al. Prostaglandin E2-EP4 signaling initiates skin immune responses by promoting migration and maturation of Langerhans cells. Nat Med 9, 744–749 (2003). [DOI] [PubMed] [Google Scholar]
- Yoshiki R. et al. The mandatory role of IL-10-producing and OX40 ligand-expressing mature Langerhans cells in local UVB-induced immunosuppression. J Immunol 184, 5670–5677 (2010). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Information