Abstract
Background:
Clinically, significant herb-drug interactions have been previously documented and can be pharmacodynamic and/or pharmacokinetic in nature. Pharmacokinetic interactions have been attributed to induction or inhibition of either metabolic enzymes or efflux transporters.
Objective:
The effect of gel and whole leaf materials from 3 different aloe species namely Aloe ferox, Aloe marlothii, and Aloe vera as well as polysaccharides precipitated from the A. vera materials on the bi-directional transport of cimetidine across rat intestinal tissue was investigated.
Materials and Methods:
Cimetidine transport studies were performed across excised rat intestinal tissue mounted in Sweetana-Grass diffusion chambers in both the apical-to-basolateral and basolateral-to-apical directions.
Results:
While A. vera gel and whole leaf materials did not inhibit the efflux of cimetidine, the polysaccharides precipitated from them did show a reduction of cimetidine efflux. On the other hand, both A. ferox and A. marlothii gel and whole leaf materials exhibited an inhibition effect on cimetidine efflux.
Conclusions:
This study identified a modulation effect of efflux transporters by certain aloe materials. This may cause herb-drug pharmacokinetic interactions when drugs that are substrates for these efflux transporters are taken simultaneously with aloe materials. On the other hand, these aloe materials may be used for drug absorption enhancement for drugs with low bioavailability due to extensive efflux.
Keywords: Aloe ferox, Aloe marlothii, Aloe vera, efflux, herb-drug interaction, in vitro transport
INTRODUCTION
Drugs crossing the intestinal epithelium membrane by passive diffusion may be substrates for efflux transporters that extrude compounds back into the gastro-intestinal lumen from within the epithelial cells. P-glycoprotein (P-gp), multi-drug resistance-associated protein-2 (MDRP2), and breast cancer resistance protein (BCRP) are members of different sub-families of ATP-binding cassette (ABC) efflux proteins. They lower intracellular drug concentrations of substrates by acting as energy-dependant drug efflux pumps.[1,2,3,4] Efflux is in some cases responsible for the low bioavailability of orally-administered drugs.[5]
Herb-drug interactions can be pharmacodynamic or pharmacokinetic in nature and in some cases, may be clinically significant. Pharmacokinetic herb-drug interactions have been attributed to induction or inhibition of either metabolic enzymes or efflux transporters by phytoconstituents present in the herbs.[6] Inhibition of drug efflux by co-administered herbs would result in higher uptake of the drug that may necessitate a reduction in drug dose to prevent toxic adverse effects.[7] Djuv and Nilson (2008)[8] reported that A. vera juice did not inhibit P-gp efflux transport of digoxin across Caco-2 cell monolayers. However, no further investigations on extracted polysaccharides or other species of aloe have been undertaken in terms of drug efflux inhibition or induction.
This study aimed at investigating the effect of A. vera, A. ferox, and A. marlothii gel and whole leaf materials on the efflux of cimetidine across excised rat intestinal tissues to identify potential interactions due to efflux modulation.
MATERIALS AND METHODS
Cimetidine and verapamil were donated by Sandoz (South Africa), and Krebs-ringer bicarbonate buffer was purchased from Sigma Aldrich (South Africa). Aloe vera (Aloe barbadensis Mill.), whole leaf extract (AVWL), and A. vera gel (AVG) powders were donations from the International Aloe Science Council (IASC051309, Texas, USA). A. vera dehydrated gel (Daltonmax 700® gel (DMG)) and whole leaf spray dried extract (Daltonmax700® whole leaf extract (DMWL)) were received as gifts from Improve USA Inc (Texas, USA). Fresh A. ferox whole leaves (AFWL) and A. ferox gel (AFG) fillets were received from Organic Aloe (Albertinia, South Africa). Fresh A. marlothii leaves were collected from the wild between Middelburg and Groblersdal, South Africa, and filleted manually. The whole leaves and fillets were each separately liquidized in a food processor and then lyophilized (VirTis, USA).
Polysaccharides were precipitated from A. vera Daltonmax700® gel (DMGP) and A. vera Daltonmax700® whole leaf extract (DMWLP) aqueous solutions with absolute ethanol (1:4; v:v). The polysaccharides were separated centrifugally (Jouan B4i, USA) at 4000 rpm for 10 min and then washed 4 times with 80% v/v ethanol, after which it was lyophilized (VirTis, USA).
1H-NMR fingerprinting of aloe materials
An amount of 50 mg of each of the aloe materials and precipitated polysaccharides together with 5 mg of the internal standard (nicotinic acid amide or NSA) were dissolved in 1 ml of D2O, and their 1H-NMR spectra were recorded with an Avance 300 Hz NMR spectrometer (Bruker). The percentage content of acetylated polymannose or aloverose was calculated by using the integral of the peak of this compound in relation to the integral of a known concentration of the internal standard.
Preparation of rat intestinal tissue
This research project that involved excised rat intestinal tissue was approved by the Ethics Committee of the North-West University, South Africa (project number: NWU-0018-09-A5). Un-fasted adult male Sprague-Dawley rats (350 - 450 g) obtained from the Laboratory Animal Center at the Potchefstroom campus of the North-West University, South Africa, was euthanized by halothane inhalation. A 20-30 cm intestinal segment was excised 10 cm distal from the pylorus valve. The excised intestinal segment was flushed and rinsed with ice-cold Krebs-Ringer bicarbonate buffer, after which the serosal layer was removed by blunt dissection. Before commencing with the diffusion studies, the rat intestinal tissue was kept in ice-cold Krebs-Ringer bicarbonate buffer. The excised rat intestinal segment was then cut along the mesenteric border to form a sheet, which was then cut further into 3 cm strips. The excised jejunum segments were mounted onto the half cells of a Sweetana-Grass diffusion apparatus[9] where after the cells were clamped together and inserted into the heating block. The surface area available for permeation was 1.78 cm2. The buffer was circulated by a gas-lift using 95% O2/5% CO2 at a flow rate of 15 – 20 ml/min. The cells were kept in the heating block at 37 °C for the entire study.
Cimetidine transport studies
Cimetidine, an H2-receptor antagonist, is a substrate for efflux transporters and has been used previously as a model compound to investigate efflux inhibition, including P-gp related efflux, using in vitro models.[2,10,11] Transport of cimetidine in the apical to basolateral (A-B) and basolateral to apical (B-A) direction across excised rat intestinal tissue was determined in the absence (control group) and presence of selected aloe materials (experimental groups) at a concentration of 2% (w/v) in Krebs-Ringer bicarbonate buffer adjusted to a pH of 7.4 by HCl and/or NaOH solutions. Verapamil, a known P-gp efflux inhibitor,[12,13,14] at a concentration of 100 μM (0.045 mg/ml) together with cimetidine was used as the positive control group. The concentration of cimetidine as model compound in the transport studies was 2.5 mg/ml. Prior to transport experiments, 7 ml Krebs-Ringer bicarbonate buffer (pH 7.4) was added to the apical and basolateral chambers of the diffusion chamber for 30 min to ensure equilibrium was reached, after which the buffer was removed from both sides. For the study in the A-B direction, 7 ml test solution was added to the apical side and 7 ml Krebs-Ringer bicarbonate buffer to the basolateral side of the excised rat intestinal tissue. For the study in the B-A direction, 7 ml test solution was added to the basolateral side and 7 ml Krebs-Ringer bicarbonate buffer to the apical side of the excised rat intestinal tissue. The compartment where the test solution was added is referred to as the donor compartment, and the compartment where the samples were withdrawn is referred to as the receptor/acceptor compartment.[14] Samples of 200 μl were taken at 20, 40, 60, 80, 100, and 120 min from the receptor compartment. The samples withdrawn from the receptor chamber were replaced with an equal volume of Krebs-Ringer bicarbonate buffer.
All the experiments were done in triplicate at 37 °C at a pH of 7.4. The TEER was measured before and after the study to ensure the integrity of the membrane was maintained throughout the transport study.
Cimetidine concentration in the samples was determined by means of reverse-phase high-performance liquid chromatography (RP-HPLC). The HPLC system consisted of an Agilent 1100 series HPLC equipped with a gradient pump, auto sampler, UV detector, and Chemstation Rev. A.0803 data acquisition and analysis software. Separation was achieved on a USP L1, Venusil XBP C18 column (150 mm × 4.6 mm; 5 μm, Agela Technologies, Newark, DE). The mobile phase consisted of acetonitrile/water with 0.005 M Na-heptane sulfonate adjusted to pH 3.5 with orthophosphoric acid (H3PO4) 20:80. The flow rate used was 1.0 ml/min. Cimetidine was detected at a wavelength of 220 nm and had a retention time of ± 4.5 min. The injection volume was 20 μl.
Prior to chromatographic analysis of cimetidine in the transport samples, the RP-HPLC method was validated in terms of linearity, accuracy, precision, sensitivity (limit of detection and limit of quantification), specificity, and peak asymmetry.
Data analysis
The cimetidine concentrations in the transport samples were corrected for dilution, and the cumulative transport was plotted as a function of time. The apparent permeability coefficients (Papp) in cm/s were calculated from the transport results by using the following equation[15]:
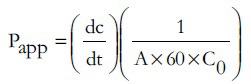
Where dc is the amount of cimetidine transported within a given time period (dt), A is the surface area of the tissue exposed to the transport medium, and C0 is the initial drug concentration.
Net permeability values (Papp net) were calculated by the following equation[16]:
Papp net = Papp (B-A) – Papp (A-B)
Where (Papp (A-B)) is the apparent permeability coefficient value in the apical to basolateral direction, and (Papp (B-A)) is the apparent permeability coefficient value in the basolateral to apical direction.
Efflux ratio values were used to indicate the extent of efflux. This ratio was calculated by the following equation[3]:
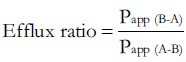
Where Papp (B-A) is the apparent permeability coefficient value in the basolateral to apical direction, and Papp (A-B) is the permeability coefficient value in the apical to basolateral direction.
The results were statistically analyzed by means of one-way repeated analysis of variance (ANOVA) to determine the differences between apparent permeability coefficients, efflux ratios, and net permeability values. Differences were considered statistically significant if P ≤ 0.05.
RESULTS AND DISCUSSION
Chemical fingerprinting of aloe materials
Aloverose (or acetylated polymannose) is a marker molecule for identification of fresh A. vera leaf material. According to the 1H-NMR spectra as previously published,[17] the A. vera gel materials namely AVG and DMG contained 12.7% and 15.2% aloverose of the dry mass, respectively. The A. vera whole leaf materials contained quantities of aloverose as follows: AVWL = 5.5% and DMWL = 4.9%.
The precipitated polysaccharides from the A. vera leaf materials contained aloverose as follows: DMGP = 8.0% and DMWLP = 25.0%.
Aloverose could not be detected in the A. ferox or A. marlothii gel and whole leaf materials by means of 1H-NMR spectroscopy, which is in line with previous findings on the chemical composition of A. ferox gel.[18,19] Quinic acid was found to be present in the A. ferox or A. marlothii gel and whole leaf materials as determined by 2D-COSY. Previous chemical analysis of the carbohydrate composition of A. ferox showed that arabinogalactan and rhamnogalacturonan make up the bulk of the carbohydrate component of both the inner and outer portions of its leave materials. The main monosaccharides obtained after hydrolysis of polysaccharides from A. ferox gel were glucose and galactose, while A. vera yields mainly mannose. It was further stated that the absence of acetylated compounds in A. ferox complicates the use of NMR spectroscopy for purposes of quality assurance for A. ferox materials, while it is successfully used for A. vera to identify marker molecules such as acetylated polymannose (aloverose).[18,19]
Cimetidine transport studies
Figure 1 shows the apparent permeability coefficient (Papp) values of cimetidine across rat intestinal tissue in the A-B and B-A directions in the presence of the different aloe materials and the control group. The net apparent permeability coefficient (Papp net) values and efflux ratio values are depicted in Table 1. A lower Papp net value indicates a higher movement of molecules in the absorptive direction (A-B direction) and, therefore, potentially also a higher bioavailability of the drug.[14]
Figure 1.
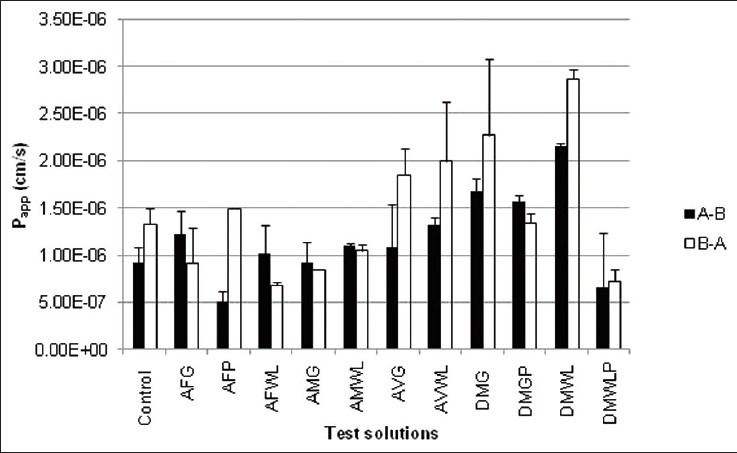
Permeability coefficient (Papp) values of cimetidine in A-B and B-A directions across rat intestinal tissues in the presence of different aloe materials and the control group. Closed bars indicate absorptive transport (A-B direction), while open bars represent secretory transport (B-A transport)
Table 1.
Papp net values and efflux ratio values of the different aloe materials
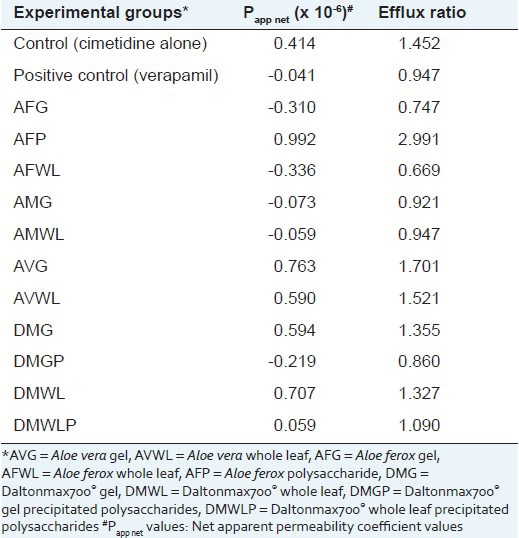
The bi-directional transport of cimetidine alone (control group) across the excised rat intestinal tissue model confirmed that it is a substrate for efflux transporters in this animal model because the Papp(A-B) value (0.916 × 10-6 cm/s) was lower than that in the B-A direction (Papp(B-A) = 1.33 × 10-6 cm/s). As expected, addition of verapamil (positive control group) resulted in a Papp net value of -0.041 × 10-6 cm/s, which is indicative of increased cimetidine transport in the absorptive direction compared to the secretory direction due to reduced efflux.
The permeability of cimetidine in both directions increased in the presence of the Aloe vera gel (AVG) and whole leaf (AVWL) materials. Similarly, the Daltonmax 700® Aloe vera gel and whole leaf products (DMG and DMWL) also exhibited increased cimetidine permeability in both directions. This increase in permeability may be attributed to opening of tight junctions as previously reported for A. vera gel and whole leaf materials[20] as well as for precipitated polysaccharides from A. vera gel.[17] None of the A. vera gel or whole leaf materials showed any inhibition of cimetidine efflux across the excised rat intestinal tissue when comparing Papp(A-B) values with that of Papp(B-A) for these experimental groups. This is in accordance with previously reported findings from a study that showed A. vera juice does not inhibit P-gp related efflux of digoxin across Caco-2 cell monolayers.[8]
However, the polysaccharides extracted from Daltonmax 700® Aloe vera materials (i.e. DMGP and DMWLP) had a reduction in Papp net values from 0.414 × 10-6 cm/s to -0.219 × 10-6 cm/s and 0.059 × 10-6 cm/s, respectively, indicating efflux inhibition. The Papp(A-B) value of cimetidine transport in the presence of DMGP increased from 0.916 × 10-6 cm/s (control group) to 1.561 × 10-6 cm/s, which was statistically significantly higher.
The results for the gel and whole leaf materials of the two aloe species indigenous to South Africa, namely A. ferox (AFG and AFWL) and A. marlothii (AMG), had negative Papp net values due to decreased permeation in the B-A direction (or efflux) of cimetidine, which was statistically significantly compared to that of the control group. In contrast to the A. vera materials, where the precipitated polysaccharides showed a reduction in the Papp net values, the polysaccharides extracted from A. ferox (AFP) demonstrated an increase in the Papp net value to 0.992 × 10-6 cm/s, which is an indication of an increase in the efflux of cimetidine. Permeability of cimetidine (A-B direction) was statistically significantly decreased to 0.498 × 10-6 cm/s by AFP while the permeability in the B-A direction was increased to 1.490 × 10-6 cm/s. A possible explanation for the different Papp net values obtained for the different experimental groups could be the chemical differences between the AFG and AFP (obtained through 1H-NMR spectra and described above). Phytoconstituents responsible for the efflux inducing effect in AFG might have been removed by the precipitation process, resulting in an inverse effect for the precipitated material.
The drug absorption enhancement potential of aloe leaf materials by means of the mechanism of inhibition of drug efflux across intestinal epithelial was shown for the first time in this study. Since only the precipitated polysaccharides showed a drug efflux inhibition effect and not the A. vera gel and whole leaf extracts, it may indicate that certain phytoconstituent(s) responsible for the drug efflux inhibition effect are present in sufficient high concentrations in the precipitated polysaccharide fraction but not in the gel and whole leaf materials. Higher concentrations of A. vera gel and whole leaf materials should, therefore, be investigated in future for potential drug efflux inhibition.
The different results obtained between the aloe species indigenous to South-Africa and A. vera materials in terms of their effects on cimetidine efflux can also be explained by the different chemical composition of these species as indicated by 1H-NMR results described above. The results indicate that aloverose (present in A. vera gel, whole leaf, and precipitated materials) is not the only phytoconstituent in aloe leave materials that plays a role during efflux inhibition because it was absent in all of the A. ferox and A. marlothii materials that showed an efflux modulation effect.
Efflux ratio values of cimetidine in the absence and presence of the aloe materials and verapamil are shown in Table 1. As expected, the efflux ratio value of cimetidine transport decreased in the presence of the positive control (100 μM verapamil). Furthermore, the efflux ratio values decreased in the presence of all the aloe materials investigated, except AFP, AVG and AVWL. The reduction in efflux ratios based on the bi-directional transport of cimetidine suggests that these aloe materials might inhibit the function of efflux transporters such as P-gp. The increased efflux ratios of cimetidine in combination with AFP and AVG, respectively, might suggest that P-gp expression is stimulated by these products.
CONCLUSIONS
The A. vera gel and whole leaf materials did not inhibit cimetidine efflux across excised rat intestinal tissue, which is in accordance with previous findings already published. However, the precipitated polysaccharides from A. vera gel (Daltonmax 700®) did show an inhibition effect on cimetidine efflux. On the other hand, gel and whole leaf materials of A. ferox and A. marlothii showed cimetidine efflux inhibition effects, while A. ferox precipitated material showed a promoting effect on the efflux when compared to the control. The difference in effects on cimetidine efflux by the different aloe species and materials can be attributed to the difference in their chemical composition as indicated by 1H-NMR. Future studies should focus on isolating the specific molecules from the A. vera precipitated polysaccharides and other aloe leaf materials that are responsible for the efflux inhibition effect in order to be conclusive regarding the involvement of aloverose in this effect.
Footnotes
Source of Support: The National Research Foundation (South Africa) and Tshwane University of Technology (South Africa) are acknowledged for financial support
Conflict of Interest: The authors declare no conflict of interest.
REFERENCES
- 1.Saitoh H, Arashiki Y, Oka A, Oda M, Hatakeyama Y, Kobayashi M, et al. Arbekacin is actively secreted in the rat intestine via a different efflux system from P-glycoprotein. Eur J Pharm Sci. 2003;19:133–40. doi: 10.1016/s0928-0987(03)00072-1. [DOI] [PubMed] [Google Scholar]
- 2.Chan LM, Lowes S, Hirst BH. The ABCs of drug transport in intestine and liver: efflux proteins limiting drug absorption and bioavailability. Eur J Pharm Sci. 2004;21:25–51. doi: 10.1016/j.ejps.2003.07.003. [DOI] [PubMed] [Google Scholar]
- 3.Shen Q, Lin Y, Handa T, Doi M, Sugie M, Wakayama K, et al. Modulation of intestinal P-glycoprotein function by polyethylene glycols and their derivatives by in vitro transport and in situ absorption studies. Int J Pharm. 2006;313:49–56. doi: 10.1016/j.ijpharm.2006.01.020. [DOI] [PubMed] [Google Scholar]
- 4.Tarirai C, Viljoen AM, Hamman JH. Herb-drug pharmacokinetic interactions reviewed. Expet Opin Drug Metabol Toxicol. 2010;6:1–24. doi: 10.1517/17425255.2010.529129. [DOI] [PubMed] [Google Scholar]
- 5.Werle M. Polymeric and low molecular mass efflux pump inhibitors for oral drug delivery. J Pharm Sci. 2008;97:60–70. doi: 10.1002/jps.21090. [DOI] [PubMed] [Google Scholar]
- 6.Zhou S, Lim LY, Chowbay B. Herbal Modulation of P-Glycoprotein. Drug Metab Rev. 2004;36:57–104. doi: 10.1081/dmr-120028427. [DOI] [PubMed] [Google Scholar]
- 7.Darby RA, Callaghan R, McMahon RM. P-glycoprotein inhibition: The past, the present and the future. Curr Drug Metab. 2011;12:722–31. doi: 10.2174/138920011798357006. [DOI] [PubMed] [Google Scholar]
- 8.Djuv A, Nilsen OG. Caco-2 Cell methodology and inhibition of the P-glycoprotein transport of digoxin by Aloe vera Juice. Phytother Res. 2008;22:1623–8. doi: 10.1002/ptr.2536. [DOI] [PubMed] [Google Scholar]
- 9.Grass GM, Sweetana SA. In Vitro Measurement of gastrointestinal tissue permeability using a new diffusion cell. Pharm Res. 1988;5:372–6. doi: 10.1023/a:1015911712079. [DOI] [PubMed] [Google Scholar]
- 10.Endres CJ, Hsiao P, Chung FS, Unadkat JD. The role of transporters in drug interactions. Eur J Pharm Sci. 2006;27:501–17. doi: 10.1016/j.ejps.2005.11.002. [DOI] [PubMed] [Google Scholar]
- 11.Taur JS, Rodriguez Proteau R. Effects of dietary flavonoids on the transport of cimetidine via P-glycoprotein and cationic transporters in Caco-2 and LLC-PK1 cell models. Xenobiotica. 2008;38:1536–50. doi: 10.1080/00498250802499467. [DOI] [PubMed] [Google Scholar]
- 12.Hunter J, Hirst BH. Intestinal secretion of drugs. The role of P-glycoprotein and related drug efflux systems in limiting oral drug absorption. Adv Drug Del Rev. 1997;25:129–57. [Google Scholar]
- 13.Choi JS, Li X. The effect of verapamil on the pharmacokinetics of paclitaxel in rats. Eur J Pharm Sci. 2005;24:95–100. doi: 10.1016/j.ejps.2004.10.002. [DOI] [PubMed] [Google Scholar]
- 14.Bansal T, Mishra G, Jaggi M, Khar RK, Talegaonkar S. Effect of P-glycoprotein inhibitor, verapamil, on oral bioavailability and pharmacokinetics of irinotecan in rats. Eur J Pharm Sci. 2009;36:580–90. doi: 10.1016/j.ejps.2008.12.005. [DOI] [PubMed] [Google Scholar]
- 15.Kotzé AF, Lueßen HL, De Leeuw BJ, De Boer BG, Verhoef CJ, Junginger HE. Comparison of the effect of different chitosan salts and N-trimethyl chitosan chloride on the permeability of intestinal epithelial cells (Caco-2) J Controlled Release. 1998;51:35–46. doi: 10.1016/s0168-3659(97)00154-5. [DOI] [PubMed] [Google Scholar]
- 16.Collett A, Higgs NB, Sims E, Rowland M, Warhurst G. Modulation of the Permeability of H 2 Receptor Antagonists Cimetidine and Ranitidine by P-Glycoprotein in Rat Intestine and the Human Colonic Cell Line Caco-2. J Pharmacol Exp Ther. 1999;288:171–8. [PubMed] [Google Scholar]
- 17.Beneke CE, Viljoen AM, Hamman JH. In vitro drug absorption enhancement effects of Aloe vera and Aloe ferox. Sci Pharm. 2012;80:475–86. doi: 10.3797/scipharm.1202-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mabusela WT, Stephen AM, Botha MC. Carbohydrate polymers from Aloe ferox leaves. Phytochemistry. 1990;29:3555–8. [Google Scholar]
- 19.O’Brien C, Van Wyk BE, Van Heerden FR. Physical and chemical characteristics of Aloe ferox gel. S Afr J Bot. 2011;77:988–95. [Google Scholar]
- 20.Chen W, Lu Z, Viljoen A, Hamman J. Intestinal drug transport enhancement by Aloe vera. Planta Med. 2009;75:587–95. doi: 10.1055/s-0029-1185341. [DOI] [PubMed] [Google Scholar]


