Abstract
Microdialysis sampling in the brain is employed frequently in the chemical analysis of neurological function and disease. But, implanting the probes, which are substantially larger than the size and spacing of brain cells and blood vessels, is injurious and triggers ischemia, gliosis, and cell death at the sampling site. The nature of the interface between the brain and the microdialysis probe is critical to the use of microdialysis as a neurochemical analysis technique. The objective of the work reported here was to investigate the potential of two compounds, dexamethasone, a glucocorticoid anti-inflammatory agent, and XJB-5-131, a mitochondrially-targeted reactive oxygen species scavenger, to mitigate the penetration injury. Measurements were performed in the rat brain striatum, which is densely innervated by axons that release dopamine, an electroactive neurotransmitter. We used voltammetry to measure electrically evoked dopamine release next to microdialysis probes during the retrodialysis of dexamethasone or XJB-5-131. After the in vivo measurements, the brain tissue containing the microdialysis probe tracks was examined by fluorescence microscopy using markers for ischemia, neuronal nuclei, macrophages, and dopamine axons and terminals. Dexamethasone and XJB-5-131 each diminished the loss of evoked dopamine activity, diminished ischemia, diminished the loss of neuronal nuclei, diminished the appearance of extravasated macrophages, and diminished the loss of dopamine axons and terminals next to the probes. Our findings confirm the ability of dexamethasone and XJB-5-131 to mitigate, but not eliminate, the effects of the penetration injury caused by implanting microdialysis probes into brain tissue.
Keywords: Dexamethasone, Dopamine, Microdialysis, Voltammetry, XJB-5-131
INTRODUCTION
Microdialysis sampling is used extensively in the chemical analysis of brain tissues in animals and, in some cases, human patients.1-4 Microdialysis offers several advantages for in vivo studies because the dialysis membrane eliminates cellular debris, blood, and macromolecules from the sample stream, which might otherwise clog or damage analytical systems. This affords a high degree of flexibility in the coupling of analysis techniques to the sampling probes. Furthermore, the probes collect essentially all substances below the membrane’s molecular weight cutoff, which varies depending on the type of membrane.5-8 For these reasons, applications of brain microdialysis are very broad and continue to expand.8-12 Increasing the spatial resolution and temporal response of microdialysis sampling is a recent development, with temporal response approaching the timescale of a few seconds in some cases.13-15
Despite its several advantages, brain microdialysis suffers from a persistent and challenging problem stemming from the wound that is caused when the probes are inserted into brain tissues.16-21 Typical probes have a diameter of 220 μm or more, whereas the typical spacing between blood vessels in brain tissue is around 60 μm.22 Consequently, implanting a microdialysis probe damages blood vessels and causes ischemia in the surrounding tissue.18, 20 The penetration injury activates the brain’s astrocytes, which engulf the probe and eventually form a glial scar around it: scar formation is clearly evident 5 days after implanting the probe.19 Microglia, a second class of brain glial cell, respond to a penetration injury within a few minutes.23
The penetration injury results in a progressive disruption of the tissue adjacent to the probe. For example, Holson et al. described progressive declines in the response of dopamine (DA), a neurotransmitter, to several pharmacological challenges (methamphetamine, bupropion, haloperidol, and potassium ion) at 2, 4, and 6 h after implanting the probe.24, 25 The declines depended only on the time since probe implantation and not the type or sequence of the manipulations: the authors concluded that a progressive loss of DA activity occurs in the tissue surrounding the probe. We reached the same conclusion by ‘voltammetry next to microdialysis probes’. We used a carbon fiber microelectrode to record DA release in the rat striatum during electrical stimulation of the afferent DA pathway. Implanting a microdialysis probe next to the microelectrode either attenuated or abolished the DA response, providing a direct observation of a progressive decline in DA activity adjacent to the probe.26, 27
Penetration injury is not unique to microdialysis probes and is a matter of concern also in the case of neuron recording electrodes, such as those used for brain-machine interfaces.28-30 A number of laboratories have examined the effects of protective drugs, including dexamethasone, on the tissue response to neuron recording electrodes.31-35 Likewise, in a subcutaneous microdialysis study, dexamethasone suppressed the immune response to probes.36 Motivated by these positive findings, we investigated the retrodialysis of dexamethasone, a potent anti-inflammatory and immunosuppressant glucocorticoid, into the rat striatum. Dexamethasone exhibited a profound ability to restore blood flow and suppress gliosis but only negligibly affected DA microdialysis results.19 It is possible that the DA results are mainly dependent on the state of the brain tissue immediately adjacent to the probe, where the penetration injury is likely at its most severe.19 The goal of the present study was to investigate the consequences of the retrodialysis of protective compounds while using voltammetry to measure DA a short distance away from the probe (70-100 μm, see Methods), i.e. at a location where the penetration injury is possibly less severe and thus more amenable to mitigation but also sufficiently close to the probe for effective drug delivery via retrodialysis.
We used voltammetry next to microdialysis probes to record electrically evoked DA release in the rat striatum during the retrodialysis of dexamethasone (DEX) or XJB-5-131 (XJB). Whereas DEX is an anti-inflammatory drug, XJB is a reactive oxygen species (ROS) scavenger targeted with high selectivity to mitochondria.37-39 Recent studies show that XJB decreases lipid oxidation and improves neurocognitive function in rats after traumatic brain injury.40 Based on these observations, we hypothesized that XJB might also mitigate penetration injury. We further hypothesized that XJB might be particularly effective at protecting DA axons and terminals, which are highly sensitive to oxidative stress and mitochondrial dysfunction.41, 42 Following voltammetry next to the microdialysis probe, brain tissue containing the probe track was examined by fluorescence microscopy using markers for ischemia (fluorescent nanobeads), neuronal nuclei (Neu-N), blood born macrophages (ED-1), and DA axons and terminals (tyrosine hydroxylase, TH).
EXPERIMENTAL SECTION
The procedures used herein have been employed in several prior studies.18-20, 26, 27 Therefore, only key aspects are described here and additional details are provided as Supplementary Information.
Reagents and Solutions
Artificial cerebrospinal fluid (aCSF: 142 mM NaCl, 1.2 mM CaCl2, 2.7 mM KCl, 1.0 mM MgCl2, 2.0 mM NaH2PO4, pH 7.40) was used for voltammetric DA calibration and as the perfusion fluid for microdialysis. Dexamethasone sodium phosphate (APP Pharmaceuticals LLC, Schaumburg, IL- and nomifensine maleate (Sigma Aldrich, St. Louis, MO) were used as received from their respective suppliers. XJB-5-131 was prepared as described by Wipf and coworkers.37 For retrodialysis, DEX (7.6 μM) and XJB (10 μM) were dissolved in aCSF and aCSF containing 1% DMSO, respectively (we lowered the concentration of DEX slightly compared to our previous study during which an adverse reaction to DEX was observed;19 the XJB concentration used here is near the solubility limit in 1% DMSO). Nomifensine was dissolved in phosphate-buffered saline (PBS: 155 mM NaCl, 100 mM NaH2PO4, pH 7.40) and administered at 20 mg/kg (i.p.). All solutions were prepared with ultrapure water (Nanopure; Barnstead, Dubuque, IA).
Voltammetry and Microdialysis
DA was measured in vivo by fast scan cyclic voltammetry (FSCV) with carbon fiber microelectrodes (7 μm in diameter, 400 μm in length). Concentricstyle microdialysis probes (300 μm in diameter, 4 mm in length) were built in-house using membranes with a 13 kDa MWCO (Spectra/Por hollow fiber, Spectrum Laboratories Inc., Rancho, Dominquez, CA). The probe inlet tubing (PE-20) was connected to a syringe pump (Harvard Apparatus, Holliston, MA) running at 0.610 μL/min. The probe outlet tubing (fused silica) was led to waste: the dialysate fluid was not analyzed during this work.
Voltammetry Next to a Microdialysis Probe
All procedures involving animals were approved by the University of Pittsburgh’s Animal Care and Use Committee. Voltammetry next to a microdialysis probe was performed in three groups of rats (n=6 rats per group). Each group underwent microdialysis with a different perfusion fluid: aCSF, aCSF with DEX (7.6 μM), or aCSF with XJB (10 μM and 1% DMSO: a fourth group was perfused with aCSF with 1% DMSO but the results are omitted because they were identical to those obtained with aCSF alone).
A carbon fiber voltammetric electrode was inserted into the striatum of an anesthetized rat and a stimulating electrode was lowered into the DA afferent pathway in the medial forebrain bundle (MFB). Electrically evoked release was recorded by FSCV during electrical stimulation of the MFB (stimulus waveform: biphasic, square, constant current pulses at 45 Hz, 300 μA pulse height, 2 ms pulse width, for 25 s). At least three stimulus responses were recorded to establish a stable, pre-probe response: at least 20 min intervals were inserted between stimulus procedures.
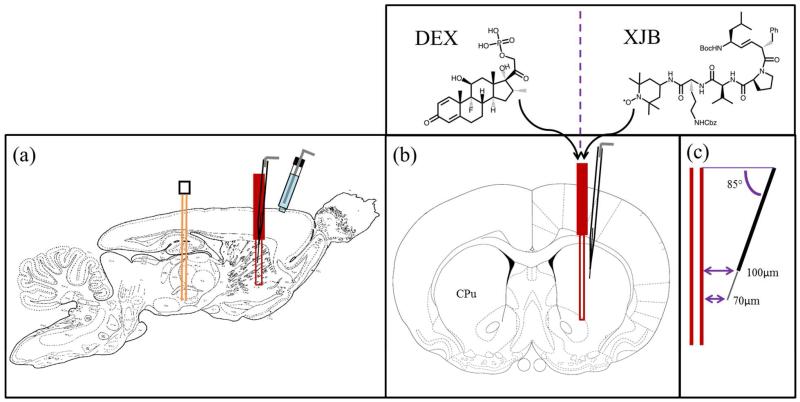
Next, a microdialysis probe was implanted in the same coronal plane as the microelectrode. The final position of the probe was such that the distance between the tip of the microelectrode and the surface of the probe was 70 μm and the distance between the top of the electrode and the probe was 100 μm (Fig 1). The relative position of the devices described here is nominal: it is based on adjustments made to the stereotaxic micropositioners (10 μm resolution). We relied on the nominal position because it is not possible to visualize the location of a carbon fiber track without making a lesion:18 the lesion destroys the tissue and would have prevented the immunohistochemical analysis described in the following section.
Figure 1.
A schematic of the placement of the devices in the rat brain. (a) A sagittal view of the stimulating electrode (orange) in the MFB, the microelectrode (black) and the microdialysis probe (red-in the striatum (CPu), and the Ag/AgCl reference electrode (blue) in contact with the brain surface. (b) A coronal view showing the microelectrode at a 5° angle from the probe. (c) The tip of the carbon fiber is 70 μm from the probe and the top of the fiber is 100 μm from the probe.
Next, 2 h after implanting the probe, three more stimulus responses were recorded: this is now ‘voltammetry next to the microdialysis probe’. Each rat then received a single dose of nomifensine (20 mg/kg i.p.), and a final stimulus response was recorded 25 min later.
Tissue Fixation and Immunohistochemistry
After recording the stimulus responses, the probe was left in place for a total of 4 h. Then, the rats were systemically perfused through the heart with fixative followed by a suspension of fluorescent nanobeads (Invitrogen, Eugene, OR), after which the tissue was post-fixed.18, 20 The brain tissue containing the microdialysis probe track was sliced in a cryostat and the sections were immunolabeled with antibodies for NeuN (Chemicon, Temecula, CA), ED-1 (AbD Serotec, Raleigh, NC), or TH (Millipore, Temecula, CA).43 The secondary antibody was goat anti-rabbit IgG-CY3 (Invitrogen, Eugene, OR).
Fluorescence Microscopy and Image Processing
Fluorescence microscopy was performed with a 20× objective (Olympus BX61, Olympus; Melville, NY- and filter sets as appropriate for the nanobeads and IgG-CY3 (Chroma Technology; Rockingham, VT). Quantitative image processing was performed with Metamorph/Fluor 7.1 software (Universal Imaging Corporation; Molecular Devices) and OriginPro. A threshold value was established to eliminate background light from each image and a freehand tool was used to exclude the track and adjacent edge effects from the region of interest (see Supplementary Information for additional details). In the case of tissues labeled with nanobeads and anti-TH, the number of fluorescent pixels was expressed as a percentage of the total number of pixels in the region of interest (% fluorescent pixels). In the case of tissues labeled with anti-NeuN and anti-ED-1, the number of labeled cells was counted and normalized with respect to the area of the region of interest (cells/mm2). Please note: the procedure used here to quantify the images does not account for differences in the intensity (i.e. brightness) of the fluorescence: this is because the fluorescence intensity is influenced by several factors that are difficult to control, such as the scattering properties of the tissue section, the tissue penetration by the antibody and blocking reagent, the extent of photobleaching, the power of the laser, etc. Statistical analyses of the fluorescent counts from the images obtained with aCSF, DEX, or XJB were based on ANOVA. For comparison, images of non-implanted tissue obtained from the brain hemisphere opposite to the microdialysis probes were processed and counted in the same fashion: results from the opposite hemisphere were omitted from the statistical analyses, which were intended to assess the impact of perfusion conditions on the tissue at the probe tracks.
RESULTS AND DISCUSSION
Electrically Evoked DA Release
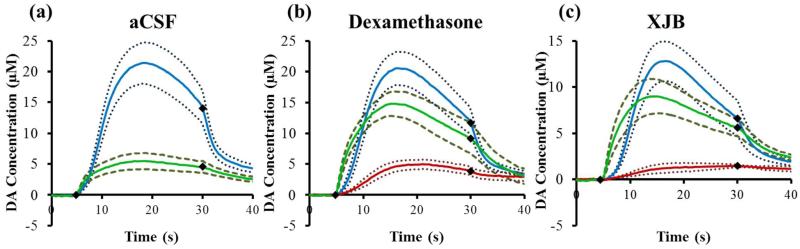
Before implanting the probe, electrical stimulation of the DA afferent pathway evoked robust DA release in the rat striatum (Fig 2, blue lines). The solid lines in Fig 2 are the average DA concentration measured in each group of animals and the dashed lines are confidence intervals based on the SEM of each data point.
Figure 2.
The effect of aCSF (a), DEX (b), or XJB (c) on electrically evoked DA responses measured before implanting the probe (blue lines), 2 h and 40 min after implanting the probe (red lines, the response was non-detectable in a), and 25 min after nomifensine (green lines). The solid lines are the average of the responses in each group of rats (n=6 per group) and the broken lines are confidence intervals based on the standard error of the mean of each data point. The black diamonds show when the stimulus begins and ends.
Microdialysis Probes Disrupt Evoked DA Release
In the case of microdialysis probes perfused with aCSF (i.e. no DEX, no XJB), implanting the probe next to the voltammetric electrode abolished all the electrically evoked DA responses. DA was not detected during any of the electrical stimuli applied after implanting the probe. Next, we gave the rats nomifensine (20 mg/kg i.p.), a DA reuptake inhibitor that increases the concentration of DA observed during electrical stimulation procedures.44, 45 Stimulated DA release was observed after the dose of nomifensine (Fig 2a, green).
The experiment in Fig 2a is similar, but not identical, to those reported previously by Borland et al. (see Fig 3b of Ref 26) and by Wang and Michael (see Fig 3 of Ref 27). In the current work, we placed the microelectrode 150 μm closer to the probe, so the responses in Fig 2 are smaller than those we reported before. We decreased the spacing during this work in order to increase the likelihood that the microelectrode would be in the diffusion zones of DEX and XJB in the following experiments.
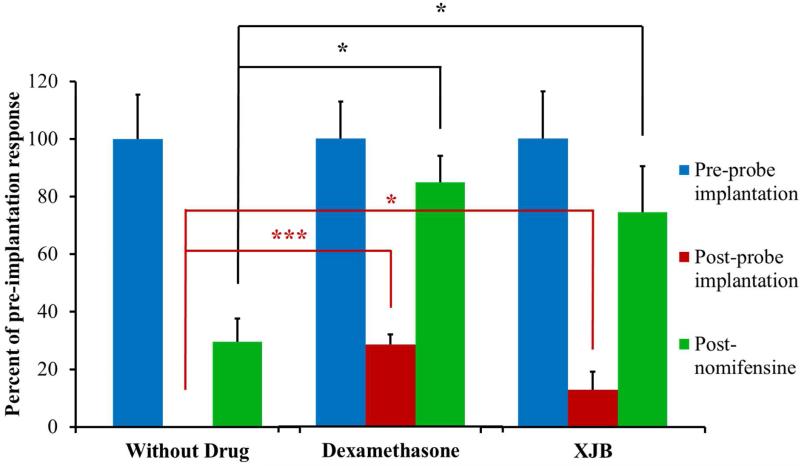
Figure 3.
The amplitude of evoked DA responses in the rat striatum in the presence of probes perfused with aCSF, DEX, or XJB. The response amplitudes observed after implanting the probes (red) and after nomifensine (green) are normalized with respect to the amplitude observed before implanting the probes (blue = 100%). The bars show the mean ± SEM (n=6 rats per group) of the normalized results. DEX significantly increased evoked DA after implanting the probe (one-sample t-test: t(5)=8.208, p<0.0005). XJB significantly increased evoked DA after implanting the probe (one-sample t-test: t(5)=2.015, p<0.05). Both DEX and XJB significantly increased evoked DA after nomifensine administration (one-way ANOVA: F(2,15)=7.43, p=0.006; and Tukey posthoc test: p<0.05. and p<0.05 respectively). *p<0.05, ***p<0.0005.
The response obtained after nomifensine administration (Fig 2a, green) has an interesting feature. That is, the DA signal is detected right away when the stimulus begins, i.e. during the first FSCV scan performed 250 ms after the stimulator is activated. Using the reported diffusion coefficient of DA in the rat striatum, 2.4×10−6 cm2/s,46 the average diffusion distance in 250 ms is ~11 μm. Thus, the DA detected when the stimulus begins was released from DA terminals in close proximity to the microelectrode. This supports a previous suggestion from our laboratory that some surviving DA terminals are present near the microelectrode, although they may be in a suppressed state.27 This motivates our on-going efforts to protect and preserve the activity of those surviving DA terminals.
DEX and XJB Preserve Evoked DA Activity Next to Microdialysis Probes
In the case of microdialysis probes perfused with DEX, implanting the probe next to the microelectrode diminished, but did not abolish, electrically evoked DA release (Fig 2b, red). Thus, retrodialysis of DEX diminished the loss evoked DA activity, possibly indicating improved survival of DA terminals in the tissue near the probe. To facilitate comparison with the results obtained using probes perfused with unmodified aCSF, we again recorded a stimulus response after treating the animals with nomifensine (Fig 2b, green): DEX substantially enhanced the amplitude of the post-nomifensine response. Implanting probes perfused with XJB also had beneficial effects on evoked DA release: similarly to DEX, XJB diminished but did not abolish evoked DA release next to the microdialysis probe (Fig 2c, red), although XJB was less effective than DEX in this regard (and see Fig 3, below, for statistical evaluation). However, similarly to DEX, XJB substantially enhanced the amplitude of the post-nomifensine response (Fig 2c, green). As was the case during aCSF perfusion, DA was rapidly detected at the start of the stimulus after nomifensine administration, indicating the presence of surviving DA terminals in close proximity to the voltammetric microelectrode. Overall, these results confirm that both DEX and XJB preserved DA activity in the tissue next to the microdialysis probes.
Statistics
To facilitate statistical analysis of these results, we prepared a histogram of the maximum DA response amplitudes normalized with respect to the maximum amplitude of the responses recorded before each probe was implanted (Fig 3). In the case of perfusion with aCSF, no DA was detected before nomifensine administration. Therefore, we used one-sample, one-tailed t-tests to compare the DEX and XJB (Fig 3, red bars) results to zero: both DEX and XJB significantly increased the evoked response amplitude. The normalized nomifensine results (Fig 3, green bars) were subjected to a one-way ANOVA and posthoc Tukey test. Both DEX and XJB significantly increased the post-nomifensine response amplitude. These findings demonstrate that perfusion with DEX or XJB preserves robust DA uptake activity in the tissue nearby the microdialysis probes. This robust DA uptake activity likely contributed to keeping the pre-nomifensine amplitudes small. The responses observed after uptake inhibition by nomifensine, therefore, more directly reflect the protection of DA release afforded by DEX and XJB. Based on Fig 3, we conclude that both DEX and XJB significantly attenuate the loss of DA activity in the tissue near the microdialysis probes.
DEX and XJB Mitigate the Histochemical Effects of Microdialysis Probes
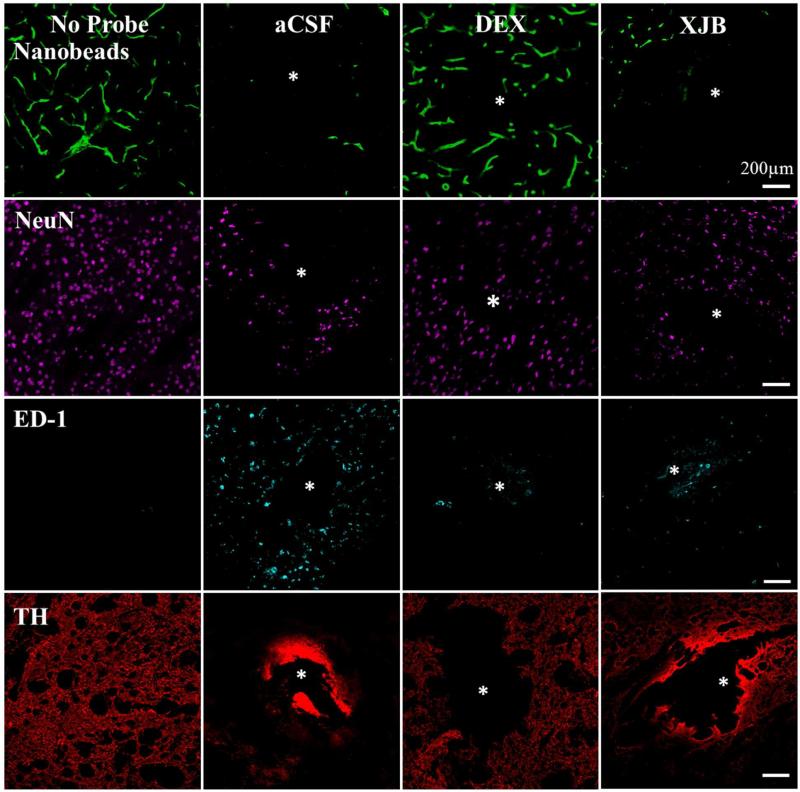
We used fluorescence microscopy to examine thin horizontal sections of brain tissue containing the tracks of the microdialysis probes perfused with aCSF, DEX, or XJB (Fig 4). For comparison, Fig 4 includes images of non-implanted control tissue (taken from the brain hemisphere opposite to the microdialysis probe) immunolabeled in the same fashion as the probe tracks. As we have reported before, probes perfused with unmodified aCSF caused profound ischemia (diminished blood flow), as indicated by a near-total absence of fluorescent nanobeads in the tissues surrounding the probe tracks.18, 20 Both DEX and XJB increased the presence of nanobeads near the probe tracks (see Fig 5, below, for statistical evaluation), indicating in both cases a decrease in ischemia. However, DEX was more effective in this regard, which is consistent with DEX’s classification as an anti-inflammatory drug.
Figure 4.
DEX and XJB mitigate the histochemical impact of penetration injury. Separate columns provide representative images of tissue after retrodialysis of aCSF, DEX, and XJB for 4 h. The left-most column shows images of non-implanted control striatal tissue. Separate rows provide representative images of tissue labeled with markers for blood flow (nanobeads), neuronal nuclei (NeuN), macrophages (ED-1), and dopamine axons and terminals (TH). The probe track is in the center of the images and marked with an asterisk. Scale bars are 200 μm.
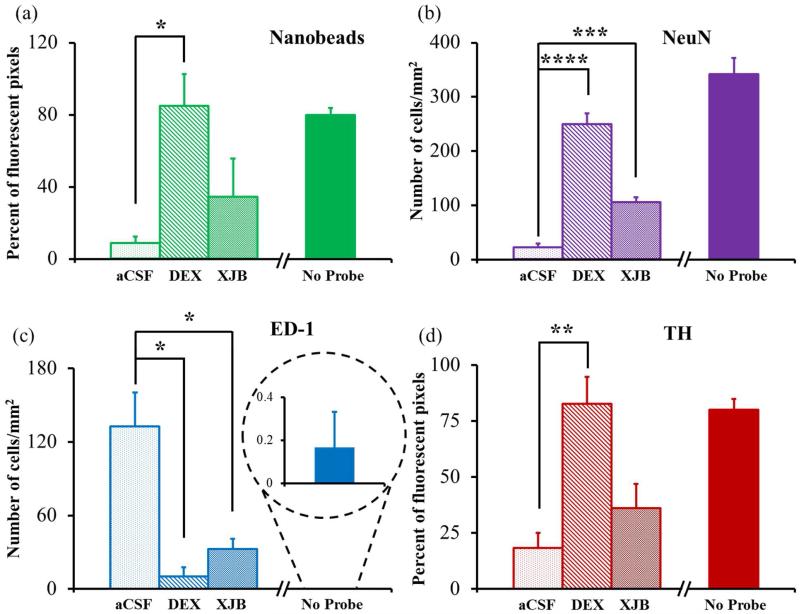
Figure 5.
Normalized counts of the individual histochemical markers were performed in the region of interest in horizontal tissue sections containing the track of the microdialysis probes. Nanobead (a) and TH (d) results are reported as the percentage of fluorescent pixels (mean± SEM). NeuN (b) and ED-1 (c) results are reported as the number of cells/mm2 (mean± SEM). Statistical analyses were performed by one-way ANOVA and Tukey or Tamhane T2 post hoc tests: *p<0.05, **p<0.005, ***p<0.00005, ****p<0.00000005. Images of non-implanted tissue from the brain hemisphere opposite the microdialysis probes were quantified by the same procedures (no-probe) but omitted from the ANOVAs.
Probes perfused with unmodified aCSF decreased NeuN labeling, indicating a loss of striatal neurons near the probes, and increased ED-1 labeling, indicating the infiltration and activation of macrophages near the probes. The ED-1 marker specifically labels blood-derived macrophages and so indicates an opening of the blood-brain barrier.47 Both DEX and XJB increased NeuN labeling and decreased ED-1 labeling, indicating that both compounds protected the brain tissue near the probes.
In non-implanted striatal tissue, TH labeling is punctate, corresponding to the size and distribution of DA axons and terminals. Probes perfused with unmodified aCSF eliminated the punctate labeling in the surrounding tissue. Intense labeling was observed at the edges of the probe track but this was not punctate and likely reflects non-specific binding.48 The loss of punctate TH labeling does not necessarily mean a loss of DA axons and terminals, but rather that the axons and terminals have lost their TH. Despite this caveat, the images confirm that probes perfused with aCSF profoundly disrupt DA axons and terminals. Both DEX and XJB diminished the non-specific edge effect and preserved punctate TH labeling.
Statistics
We quantified these images using Metamorph’s built-in tools. After setting a threshold fluorescence intensity for each image, the software quantified the percentage of pixels exhibiting nanobead and TH fluorescence, and counted the number of NeuN-positive and ED-1-positive cells in the region of interest: the quantitative results are reported as histograms in Fig 5. The numerical results for each marker were subjected to one-way ANOVA (details in the figure legend). Compared to probes perfused with unmodified aCSF, retrodialysis of DEX significantly increased nanobead, NeuN, and TH labeling, and significantly decreased ED-1 labeling. Retrodialysis of XJB significantly increased NeuN labeling and decreased ED-1 labeling. Retrodialysis of XJB also increased nanobead and TH labeling, but these effects were not statistically significant.
Correlation of Voltammetry and Immunohistochemistry
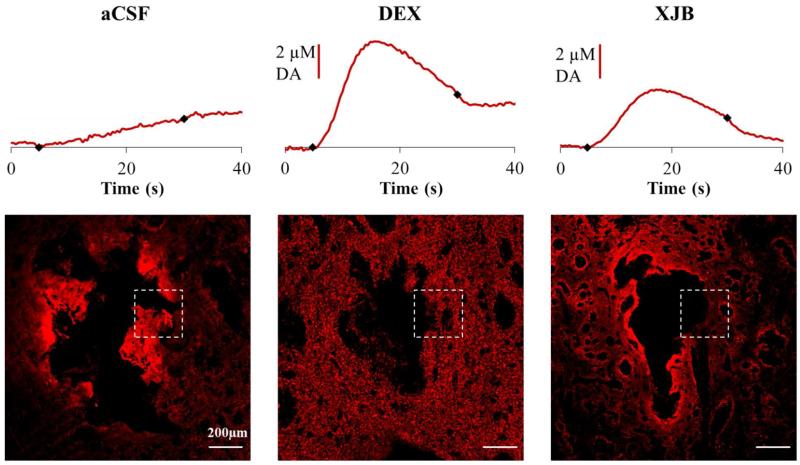
Our findings reveal, for the first time, a general correlation between the ability of DEX and XJB to preserve DA activity as assessed by voltammetry next to microdialysis probes and their ability to protect the tissue near the probes as assessed by immunohistochemical labeling. The most direct correlation can be expected between DA activity and punctate TH labeling, since DA activity derives from DA axons and terminals. Fig 6 compares TH images with the DA responses taken from the same tissue: the white box in each image identifies the nominal location of the microelectrode. Even from this small sampling, it is clear that the intensity of the punctate TH labeling near the probe correlates with the amplitude of the stimulus response. Note that in the case of the probe perfused with unmodified aCSF, the bright TH labeling is non-punctate: it is the non-specific edge effect explained above.
Figure 6.
Correlation of voltammetry next to a microdialysis probe with TH immunohistochemistry. Individual DA responses recorded after implanting the probe (without nomifensine) are compared with the TH immunohistochemistry from the same rat. The white box indicates the nominal position of the microelectrode. Columns represent retrodialysis of aCSF, DEX, and XJB for 4 h. Scale bars are 200 μm.
CONCLUSION
The present study confirms that retrodialysis of dexamethasone or XJB-5-131 mitigates penetration injury during brain microdialysis. Both compounds protected striatal DA activity as assessed by voltammetry next to microdialysis probes and both also mitigated the histochemical effects of the penetration injury. The general correlation between the effects of DEX and XJB on DA activity and observed histochemical changes is potentially significant, because reports on the impact of neuroprotection strategies on the tissue responses to implanted devices are expanding rapidly.49-52
The protective effects observed during this study were partial, as neither the evoked DA activity nor the histochemical attributes of the tissue near the probe were preserved in their normal, completely uninjured state. Nevertheless, the mitigation observed during this study is substantially larger than during our previous observations of DA no-net-flux, which were negligibly affected by DEX.19 Thus, as we anticipated, the tissue even a small distance from the probe (70-100 μm) appears to be more amenable to protection by DEX and XJB. However, it is important to appreciate that measuring DA via microdialysis is an especially challenging task due to DA reuptake, which rapidly removes DA from the extracellular space and limits how far it can diffuse. This challenge is illustrated by the large impact of the DA reuptake inhibitor, nomifensine, on the evoked DA responses. Accordingly, we conclude that even though DEX and XJB protected the tissue 70-100 μm from the probe, DA’s ability to diffuse across the gap to the microdialysis probe was constrained, as usual, by reuptake. It is important to appreciate this point because microdialysis has applications to numerous other substances of interest that are not subject to such avid reuptake. It will be of great interest in the future to examine the effect of DEX, XJB, and other candidates on the microdialysis of substances other than DA.
Finally, this investigation focused on acute implants only 4 h in duration. Tremendous interest in chronic implants exists as well, and in particular, XJB has demonstrated significant efficacy in long-term neuroprotective studies.53 We reason that any protective strategy must be effective during the acute phase of the implant in order to preserve the tissue into the chronic phases, i.e. cell death in the acute phase is unlikely to be reversible. Accordingly, we envision that successful acute mitigation is needed as a precursor to successful chronic mitigation of penetration injury effects.
Supplementary Material
ACKNOWLEDGMENT
This work was funded by the NIH (MH 075989, NS 081744) and the NIH/NIGMS CMLD program (P50 GM067082).
Footnotes
ASSOCIATED CONTENT
Supporting Information. Additional information as noted in text. This material is available free of charge via the Internet at http://pubs.acs.org.
REFERENCES
- (1).Bosche B, Dohmen C, Graf R, Neveling M, Staub F, Kracht L, Sobesky J, Lehnhardt FG, Heiss WD. Stroke. 2003;34:2908–2915. doi: 10.1161/01.STR.0000100158.51986.EB. [DOI] [PubMed] [Google Scholar]
- (2).Feuerstein D, Manning A, Hashemi P, Bhatia R, Fabricius M, Tolias C, Pahl C, Ervine M, Strong AJ, Boutelle MG. J. Cereb. Blood Flow Metab. 2010;30:1343–1355. doi: 10.1038/jcbfm.2010.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (3).Parkin MC, Hopwood SE, Jones DA, Hashemi P, Landolt H, Fabricius MF, Lauritzen M, Boutelle MG, Strong AJ. J. Cereb. Blood Flow Metab. 2005;25:402–413. doi: 10.1038/sj.jcbfm.9600051. [DOI] [PubMed] [Google Scholar]
- (4).Sakowitz OW, Stover JF, Sarrafzadeh AS, Unterberg AW, Kiening KL. J. Trauma. 2007;62:292–298. doi: 10.1097/01.ta.0000203560.03937.2d. [DOI] [PubMed] [Google Scholar]
- (5).Gill C, Parkinson E, Church MK, Skipp P, Scott D, White AJ, O’Connor CD, Clough GF. AAPS J. 2011;13:309–317. doi: 10.1208/s12248-011-9269-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (6).Klede M, Schmitz H, G□en T, Fartasch M, Drexler H, Schmelz M. Exp. Dermatol. 2005;14:103–108. doi: 10.1111/j.0906-6705.2005.00227.x. [DOI] [PubMed] [Google Scholar]
- (7).Vidal-Torres A, Carceller A, Zamanillo D, Merlos M, Vela JM, Fernández-Pastor B. J Pharmacol Sci. 2012;120:129–132. doi: 10.1254/jphs.12105sc. [DOI] [PubMed] [Google Scholar]
- (8).Westerink BH, Cremers TIFH, editors. Handbook of Microdialysis: Methods, Applications and Perspectives. Academic Press; London: 2007. [Google Scholar]
- 9.Badiani A, Oates MM, Day HE, Watson SJ, Akil H, Robinson TE. J. Neurosci. 1998;18:10579. doi: 10.1523/JNEUROSCI.18-24-10579.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (10).Hopwood SE, Parkin MC, Bezzina EL, Boutelle MG, Strong AJ. J. Cereb. Blood Flow Metab. 2005;25:391–401. doi: 10.1038/sj.jcbfm.9600050. [DOI] [PubMed] [Google Scholar]
- (11).Watson CJ, Venton BJ, Kennedy RT. Anal. Chem. 2006;78:1391–1399. doi: 10.1021/ac0693722. [DOI] [PubMed] [Google Scholar]
- (12).Yang H, Thompson AB, McIntosh BJ, Altieri SC, Andrews AM. ACS Chem. Neurosci. 2013;4:790–798. doi: 10.1021/cn400072f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (13).Lee WH, Slaney TR, Hower RW, Kennedy RT. Anal. Chem. 2013;85:3828–3831. doi: 10.1021/ac400579x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (14).Liu Y, Zhang J, Xu X, Zhao MK, Andrews AM, Weber SG. Anal. Chem. 2010;82:9611–9616. doi: 10.1021/ac102200q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (15).Wang M, Slaney T, Mabrouk O, Kennedy RT. J. Neurosci. Methods. 2010;190:39–48. doi: 10.1016/j.jneumeth.2010.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (16).Benveniste H, Diemer NH. Acta Neuropathol. 1987;74:234–238. doi: 10.1007/BF00688186. [DOI] [PubMed] [Google Scholar]
- (17).Clapp-Lilly KL, Roberts RC, Duffy LK, Irons KP, Hu Y, Drew KL. J. Neurosci. Methods. 1999;90:129–142. doi: 10.1016/s0165-0270(99)00064-3. [DOI] [PubMed] [Google Scholar]
- (18).Jaquins-Gerstl A, Michael AC. J. Neurosci. Methods. 2009;183:127–135. doi: 10.1016/j.jneumeth.2009.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (19).Jaquins-Gerstl A, Shu Z, Zhang J, Liu Y, Weber SG, Michael AC. Anal. Chem. 2011;83:7662–7667. doi: 10.1021/ac200782h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (20).Mitala CM, Wang Y, Borland LM, Jung M, Shand S, Watkins S, Weber SG, Michael AC. J. Neurosci. Methods. 2008;174:177–185. doi: 10.1016/j.jneumeth.2008.06.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (21).Zhou F, Zhu X, Castellani RJ, Stimmelmayr R, Perry G, Smith MA, Drew KL. Am. J. Pathol. 2001;158:2145–2151. doi: 10.1016/S0002-9440(10)64686-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (22).Peters JL, Miner LH, Michael AC, Sesack SR. J. Neurosci. Methods. 2004;137:9–23. doi: 10.1016/j.jneumeth.2004.02.006. [DOI] [PubMed] [Google Scholar]
- (23).Davalos D, Grutzendler J, Yang G, Kim JV, Zuo Y, Jung S, Littman DR, Dustin ML, Gan WB. Nat. Neurosci. 2005;8:752–758. doi: 10.1038/nn1472. [DOI] [PubMed] [Google Scholar]
- (24).Holson RR, Bowyer JF, Clausing P, Gough B. Brain Res. 1996;739:301–307. doi: 10.1016/s0006-8993(96)00837-2. [DOI] [PubMed] [Google Scholar]
- (25).Holson RR, Gazzara RA, Gough B. Brain Res. 1998;808:182–189. doi: 10.1016/s0006-8993(98)00816-6. [DOI] [PubMed] [Google Scholar]
- (26).Borland LM, Shi G, Yang H, Michael AC. J. Neurosci. Methods. 2005;146:149–158. doi: 10.1016/j.jneumeth.2005.02.002. [DOI] [PubMed] [Google Scholar]
- (27).Wang Y, Michael AC. J. Neurosci. Methods. 2012;208:34–39. doi: 10.1016/j.jneumeth.2012.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (28).Skousen JL, Merriam SM, Srivannavit O, Perlin G, Wise KD, Tresco PA. Prog. Brain Res. 2011;194:167–180. doi: 10.1016/B978-0-444-53815-4.00009-1. [DOI] [PubMed] [Google Scholar]
- (29).Szarowski DH, Andersen MD, Retterer S, Spence AJ, Isaacson M, Craighead HG, Turner JN, Shain W. Brain Res. 2003;983:23–35. doi: 10.1016/s0006-8993(03)03023-3. [DOI] [PubMed] [Google Scholar]
- (30).Turner AM, Dowell N, Turner SW, Kam L, Isaacson M, Turner JN, Craighead HG, Shain W. J. Biomed. Mater. Res. 2000;51:430–441. doi: 10.1002/1097-4636(20000905)51:3<430::aid-jbm18>3.0.co;2-c. [DOI] [PubMed] [Google Scholar]
- (31).Grand L, Wittner L, Herwik S, Gothelid E, Ruther P, Oscarsson S, Neves H, Dombovari B, Csercsa R, Karmos G, Ulbert I. J. Neurosci. Methods. 2010;189:216–229. doi: 10.1016/j.jneumeth.2010.04.009. [DOI] [PubMed] [Google Scholar]
- (32).Shain W, Spataro L, Dilgen J, Haverstick K, Retterer S, Isaacson M, Saltzman M, Turner JN. IEEE Trans. Neural Syst. Rehabil. Eng. 2003;11:186–188. doi: 10.1109/TNSRE.2003.814800. [DOI] [PubMed] [Google Scholar]
- (33).Spataro L, Dilgen J, Retterer S, Spence AJ, Isaacson M, Turner JN, Shain W. Exp. Neurol. 2005;194:289–300. doi: 10.1016/j.expneurol.2004.08.037. [DOI] [PubMed] [Google Scholar]
- (34).Zhong Y, Bellamkonda RV. J. Control. Release. 2005;106:309–318. doi: 10.1016/j.jconrel.2005.05.012. [DOI] [PubMed] [Google Scholar]
- (35).Zhong Y, Bellamkonda RV. Brain Res. 2007;1148:15–27. doi: 10.1016/j.brainres.2007.02.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (36).Mou X, Lennartz MR, Loegering DJ, Stenken JA. J. Diabetes Sci. Technol. 2011;5:619–631. doi: 10.1177/193229681100500316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (37).Fink MP, Macias CA, Xiao J, Tyurina YY, Delude RL, Greenberger JS, Kagan VE, Wipf P. Crit. Care Med. 2007;35:S461–467. doi: 10.1097/01.CCM.0000279192.96303.E7. [DOI] [PubMed] [Google Scholar]
- (38).Jiang J, Kurnikov I, Belikova NA, Xiao J, Zhao Q, Amoscato AA, Braslau R, Studer A, Fink MP, Greenberger JS, Wipf P, Kagan VE. J. Pharmacol. Exp. Ther. 2007;320:1050–1060. doi: 10.1124/jpet.106.114769. [DOI] [PubMed] [Google Scholar]
- (39).Wipf P, Xiao J, Jiang J, Belikova NA, Tyurin VA, Fink MP, Kagan VE. J. Am. Chem. Soc. 2005;127:12460–12461. doi: 10.1021/ja053679l. [DOI] [PubMed] [Google Scholar]
- (40).Ji J, Kline AE, Amoscato A, Samhan-Arias AK, Sparvero LJ, Tyurin VA, Tyurina YY, Fink B, Manole MD, Puccio AM, Okonkwo DO, Cheng JP, Alexander H, Clark RS, Kochanek PM, Wipf P, Kagan VE, Bayir H. Nat. Neurosci. 2012;15:1407–1413. doi: 10.1038/nn.3195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (41).Hunter RL, Dragicevic N, Seifert K, Choi DY, Liu M, Kim HC, Cass WA, Sullivan PG, Bing G. J. Neurochem. 2007;100:1375–1386. doi: 10.1111/j.1471-4159.2006.04327.x. [DOI] [PubMed] [Google Scholar]
- (42).Rabinovic AD, Lewis DA, Hastings TG. Neuroscience. 2000;101:67–76. doi: 10.1016/s0306-4522(00)00293-1. [DOI] [PubMed] [Google Scholar]
- (43).Card JP, Sved JC, Craig B, Raizada M, Vazquez J, Sved AF. J. Comp. Neurol. 2006;499:840–859. doi: 10.1002/cne.21140. [DOI] [PubMed] [Google Scholar]
- (44).Garris PA, Wightman RM. Synapse. 1995;20:269–279. doi: 10.1002/syn.890200311. [DOI] [PubMed] [Google Scholar]
- (45).Taylor IM, Jaquins-Gerstl A, Sesack SR, Michael AC. J. Neurochem. 2012;122:283–294. doi: 10.1111/j.1471-4159.2012.07774.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (46).Nicholson CD, Rice ME. Diffusion of ions and transmitters in the brain cell microenvironment. In: Fuxe K, Agnati LF, editors. Volume Transmission in the Brain: Novel Mechanisms for Neural Transmission. Raven Press; New York: 1991. pp. 279–294. [Google Scholar]
- (47).Rinaman L, Card JP, Enquist LW. J. Neurosci. 1993;13:685–702. doi: 10.1523/JNEUROSCI.13-02-00685.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (48).Sauer M, Hofkens J, Enderlein J. Handbook of Fluorescence Spectroscopy and Imaging: From Ensemble to Single Molecules. Germany; 2011. [Google Scholar]
- (49).Han N, Rao SS, Johnson J, Parikh KS, Bradley PA, Lannutti JJ, Winter JO. Front. Neuroeng. 2011;4:1–8. doi: 10.3389/fneng.2011.00002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (50).Hsiao TW, Swarup VP, Kuberan B, Tresco PA, Hlady V. Acta Biomater. 2013;9:7200–7208. doi: 10.1016/j.actbio.2013.02.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (51).Karumbaiah L, Norman SE, Rajan NB, Anand S, Saxena T, Betancur M, Patkar R, Bellamkonda RV. Biomaterials. 2012;33:5983–5996. doi: 10.1016/j.biomaterials.2012.05.021. [DOI] [PubMed] [Google Scholar]
- (52).Lewitus DY, Smith KL, Shain W, Bolikal D, Kohn J. Biomaterials. 2011;32:5543–5550. doi: 10.1016/j.biomaterials.2011.04.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (53).Xun Z, Rivera-Sanchez S, Ayala-Pena S, Lim J, Budworth H, Skoda EM, Robbins PD, Niedernhofer LJ, Wipf P, McMurray CT. Cell Rep. 2012;2:1–6. doi: 10.1016/j.celrep.2012.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.