Abstract
Rationale
Idiopathic pulmonary fibrosis (IPF) is a fatal interstitial lung disease characterized by progressive scarring and matrix deposition. Recent reports highlight an autoimmune component in IPF pathogenesis. We have reported anti-col(V) immunity in IPF patients. The objective of our study was to determine the specificity of col(V) expression profile and anti-col(V) immunity relative to col(I) in clinical IPF and the efficacy of nebulized col(V) in pre-clinical IPF models.
Methods
Col(V) and col(I) expression profile was analyzed in normal human and IPF tissues. C57-BL6 mice were intratracheally instilled with bleomycin (0.025 U) followed by col(V) nebulization at pre-/post-fibrotic stage and analyzed for systemic and local responses.
Results
Compared to normal lungs, IPF lungs had higher protein and transcript expression of the alpha 1 chain of col(V) and col(I). Systemic anti-col(V) antibody concentrations, but not of anti-col(I), were higher in IPF patients. Nebulized col(V), but not col(I), prevented bleomycin-induced fibrosis, collagen deposition, and myofibroblast differentiation. Col(V) treatment suppressed systemic levels of anti-col(V) antibodies, IL-6 and TNF-α; and local Il-17a transcripts. Compared to controls, nebulized col(V)-induced tolerance abrogated antigen-specific proliferation in mediastinal lymphocytes and production of IL-17A, IL-6, TNF-α and IFN-γ. In a clinically relevant established fibrosis model, nebulized col(V) decreased collagen deposition. mRNA array revealed downregulation of genes specific to fibrosis (Tgf-β, Il-1β, Pdgfb), matrix (Acta2, Col1a2, Col3a1, Lox, Itgb1/6, Itga2/3) and members of the TGF-β superfamily (Tgfbr1/2, Smad2/3, Ltbp1, Serpine1, Nfkb/Sp1/Cebpb).
Conclusions
Anti-col(V) immunity is pathogenic in IPF, and col(V)-induced tolerance abrogates bleomycin-induced fibrogenesis and down regulates TGF- β-related signaling pathways.
Introduction
Idiopathic Pulmonary Fibrosis (IPF) is a fatal disease with no effective treatment and a three year mean survival rate [1]. The early IPF lesion is believed to be alveolitis that leads to thickening of the alveolar wall and epithelial cell death, eventually culminates in fibrogenic foci which is characterized by overexpression of collagen types I [col(I)] and V [col(V)] [2]. However, the relative quantities of total col(I) to col(V) and their respective individual chains in the IPF lung are unknown.
Type V collagen ([col(V)] is a minor collagen normally sequestered within the lung interstitium and therefore, hidden from the immune system. Remodeling, a characteristic feature of IPF, leads to col(V) exposure and overexpression which in turn is associated with the development of anti-col(V) immunity - indicating an autoimmune aspect in IPF pathogenesis. Specifically, up to 60% of IPF patients have anti-col(V) reactive T cells [3], and nearly half develop specific systemic antibody responses [4].
Persistent immune responses and resulting dysregulated tissue healing accelerates the pathogenic conditions towards fibrosis and accumulation of extracellular matrix components [5]. When autoimmune or other agents cause epithelial injury, matrix metalloproteinases [6] are expressed to gain access to damaged tissues. Subsequently, pro-inflammatory chemokines and growth factors are recruited to the site of injury. During this phase, the chemokine cocktail leads to myofibroblast accumulation and deposition of extracellular matrix components such as integrins [7]–[9], fibronectins [10] and collagens [11]. TGF-β, a master regulator of the fibrogenic process [12], is intricately involved in the crosstalk of other pro-fibrotic molecules, particularly PDGF [13] and IL-1β [14]. In this pro-fibrotic environment, intricate interactions which exist between specific key players, for example - TGF-β and integrins, accelerate fibrogenesis. Integrins, which are receptors for cell-cell and cell-matrix adhesion, has been reported to activate latent TGF-β [8], specifically αVβ6 integrins. Similarly, TGF-β has been reported to upregulate integrin expression [15]. Notably, collagens may bind to α3β1 integrins via the RGD binding site, since it is recognized in all ligands [16]. Collagen type I has been reported to bind to α3β1 integrins via the DGEA binding site [17]. Interestingly, the effects of col(V)-induced tolerance on the gene expression of integrins and other fibrosis related genes, in IPF are unknown.
Col(V)-induced tolerance reportedly downregulates TGF-β in an experimental model of systemic sclerosis [18]. Col(V)-induced tolerance by systemic administration abrogates obliterative bronchiolitis (OB) post lung transplant [19] and bleomycin-induced acute injury [20]. Since col(V) is overexpressed in IPF and associated with anti-col(V) immunity, then in the current study we tested the hypothesis that nebulizing col(V) to significantly scarred lungs will arrest progression of fibrosis and that col(V)-induced tolerance by inhalation will attenuate pro-fibrotic signaling in a pre-clinical model of IPF. Our findings suggest that col(V)-induced tolerance effectively mitigates lung fibrosis and down regulates TGF-β and related signaling pathways.
Materials and Methods
Human Studies
All protocols were approved by the Institutional Review Board, IUSM.
Plasma and frozen tissues from IPF patients were obtained from the Lung Tissue Research Consortium (LTRC) sponsored by NIH/NHLBI. The LTRC collected medical data and tissues from large cohorts of patients to study the cause, early detection and best treatment for IPF and other similar lung diseases. A written consent was obtained from patients by LTRC and this questionnaire is available at their public domain http://www.ltrcpublic.com/docs/LTRC_Consent_Jul_2010.pdf. Plasma from IPF patients were also provided by ImmuneWorks, Inc. The samples were collected by written consent from patients enrolled in a clinical trial. The patient selection criteria are available in the public domain: http://www.clinicaltrials.gov/ct2/show/NCT01199887?term=pulmonaryfibrosisimmuneworks&rank=1. Therefore, the individuals in this manuscript have given written informed consent (as outlined in PLOS consent form) to publish these research details. Pathologist-certified paraffin-embedded lung tissues and normal lung specimens were also procured from the Department of Pathology, Indiana University School of Medicine (IUSM).
Real Time PCR
Total RNA was isolated from two 20 µm scrolls of formalin-fixed paraffin embedded tissue using RecoverAll™ Total Nucleic Acid Isolation Kit (Ambion) according to manufacturer's instructions. cDNA was synthesized using the iScript cDNA synthesis kit (Bio-Rad, Hercules, CA). Alpha 1 and 2 chains for col(I) and col(V) and GAPDH were performed using the Assays-on-Demand™ gene expression kits (Applied Biosystems, Foster City, CA) on a Smartcycler (Cepheid, Sunnyvale, CA). qRT-PCR for Il-17a and β-actin in mouse whole lung homogenates were performed using the following real-time primer sequences: β-actin- FW:CAATAGTGATGACCTGGCCGT, RV:AGAGGGAAATCGTGCGTGAC, IL-17A- FW:CTGTGTCTCTGATGCTGTTG, RV:ATGTGGTGGTCCAGCTTTC.
Mouse Fibrosis PCR Microarrays
Murine lung mRNA was used to generate cDNA. The Mouse Fibrosis PCR array- RT2 Profiler PCR Arrays version 3.0 (SABiosciences, Qiagen, Valencia, CA) was used as per manufacturer's instructions and array data were analyzed using Qiagen PCR Array Data Analysis software. The array data is provided in Table S1.
Circulating col(V) and col(I) antibody detection
Known volumes of plasma (10 µl) were incubated with microsphere beads coated with col(V) or col(I) and the signal was detected by flow cytometry as previously described [21]. Results of the duplicate reactions were expressed as Mean Fluorescent Intensity (MFI) as a fold change compared to pooled normal plasma.
Pepsin digestion
Frozen IPF and normal lung tissues were homogenized and subjected to pepsin extraction by lysing the tissues in a buffer containing pepsin in 0.2M acetic acid. The lysates were then subjected to dialysis, lyophilization, followed by careful weighing and re-suspension in lysis buffer at a concentration of 1 mg/ml. Pepsin-digested samples were equally loaded at 15 µg in a fixed 5% concentration agarose gel and electrophoresed as previously described [22]. Densitometry measurements: Bands corresponding to the individual chains of col(V) or col(I) was measured in normal and IPF lungs using NIH Image J. The col(V) protein standard was used as a control on the exposure time. Values were then expressed as Mean ± SEM.
Animal Studies
The Indiana University School of Medicine Animal Care and Use Committee (IACUC) approved the animal protocols used in this study. Female C57BL6 mice (6–8 weeks of age) were purchased from Jackson Laboratory (Bar Harbor, ME). After a 5 day period of acclimatization, mice were administered intratracheal PBS or bleomycin (0.025 U) as previously described [23]. Based on previous studies, we used varying doses of col(V): 4.16, 5.5 and 8.33 mg/kg bodyweight, at a final volume of 100 µl, and nebulized it to a mouse of average body weight of 18 gm. Mice were nebulized in a conscious state using Aeroneb® Lab Nebulizer, which is designed to produce highly respirable aerosol particles with sizes averaging between 2.5 µm–4.0 µm. Historically, aerosolized drug particles with a size of ∼3.0 µm has been documented as optimal for penetration, retention and maximal therapeutic effectiveness in the lung [24]. Lungs were processed for immunohistochemical (IHC) staining or stored at −20°C until further analyses.
Statistical analysis
Statistical analysis was performed using Student's t test, one-way ANOVA with Bonferroni post hoc test using GraphPad Prism version 3.0 for Windows GraphPad Software (San Diego, CA, www.graphpad.com). Statistical significance was defined at p<0.05.
Additional descriptive material for methods used in this study is provided in File S1. The raw array data for the array analyses is provided in Table S1.
Results
Col(V) protein and mRNA overexpression in the fibrotic foci of lungs with IPF
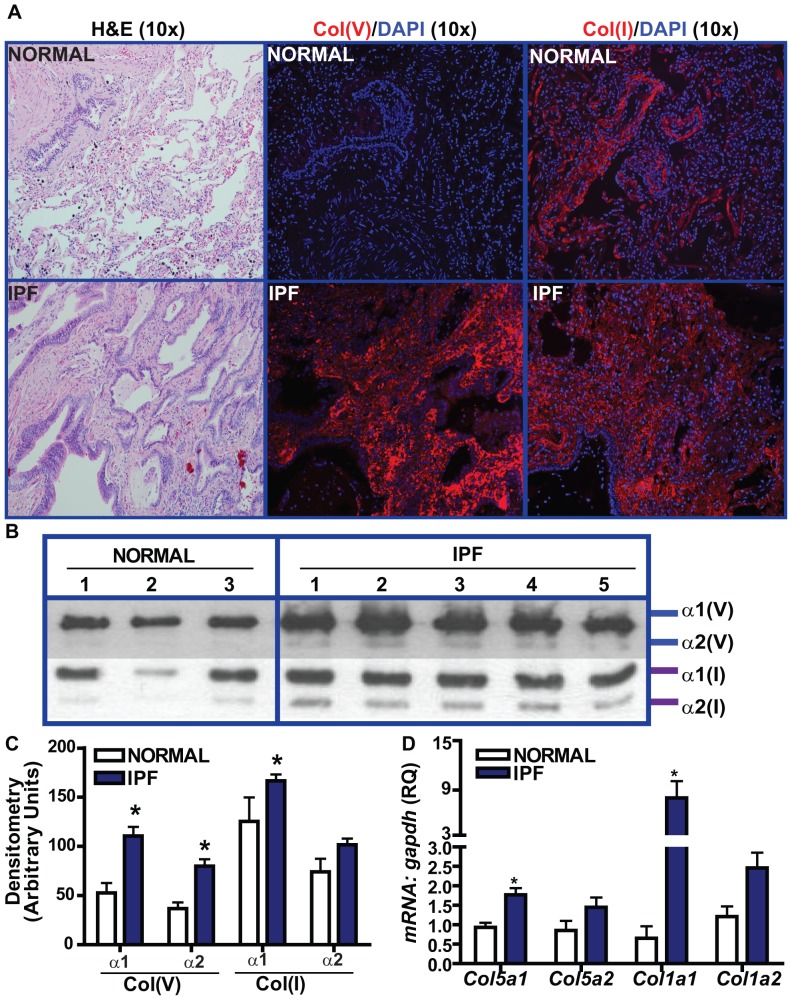
Col(V) exists in the lung as the predominant heterotrimer, [α1(V)]2. α2(V). The activity of this molecular form to bind to heparin at physiological salt concentrations may be attributed to a proteolytic NH2-terminal 30-kDa fragment of the α1(V) chain [25]. Parra and colleagues have demonstrated the distribution of col(V) in IPF [26]. However, the relative quantitative distribution of col(I) to col(V) and the expression pattern of the individual chains of col(V) and col(I) in the IPF lungs are largely uncertain. In contrast to normal lungs wherein we observed strong expression of total col(I), but not col(V), in the interstitium; both col(I) and col(V) were expressed heavily in IPF lungs (Figure 1A). We observed a similar trend when we analyzed pepsin-digested normal and IPF lungs for protein expression levels of alpha 1 [α1(V)] and alpha 2 [α2(V)] chains of col(V) (Figure 1B). The expression levels of α1(V) and α2(V) were ∼2.5–3 fold higher than the normal tissues. Notably, α1(V) levels were higher than α2(V) in the IPF lungs (p<0.01). Similarly, α1(I) levels were higher in IPF patients compared to normal donors. IPF lungs had higher α1(I) levels than α1(V) (p<0.0001) (Figure 1 B,C; Figure S1). At the transcript level, we observed higher expression of α1(V) (Col5a1) and α1(I) (Col1a1) in the IPF lungs compared to normal and that Col1a1 was ∼6 fold higher than Col5a1 in IPF (Figure 1D). Collectively, these studies demonstrate that col(V), particularly α1(V), is overexpressed at the transcript and protein level in IPF.
Figure 1. Relative expression of col(I) and col(V) in patients with UIP/IPF and pathologically normal specimens.
(A) Lung tissue sections from UIP/IPF patients and pathologically normal specimens were immunostained with col(I) and col(V) antibodies and their IgG, followed by incubation with rhodamine-anti-rabbit. Nuclei were counterstained with DAPI. (Original magnification, 10×, representative of 4 patients). Corresponding H&E staining is also shown. (B) Pepsin digested lung homogenates (15 µg) and corresponding standards run in a 5% gel and immunoblotted with antibodies against col(V) and col(I). Image shows representative 3 normal and 5 IPF tissues, (C) Densitometry of protein expressions of individual alpha chains of col(I) and col(V) obtained from IPF lung biopsies and pathologically normal specimens. Values represent mean ± SEM of5 normals and 20 IPF specimens (p<0.01; one-way ANOVA, post hoc test: Bonferroni), (D) mRNA expression were determined by qPCR of lung tissue sections of UIP/IPF and pathologically normal specimens. Values represent mean ± SEM; 3 normals and 4 UIP/IPF specimens; (p<0.01; one-way ANOVA, post hoc test: Bonferroni).
Circulating col(V)-specific antibody responses in IPF
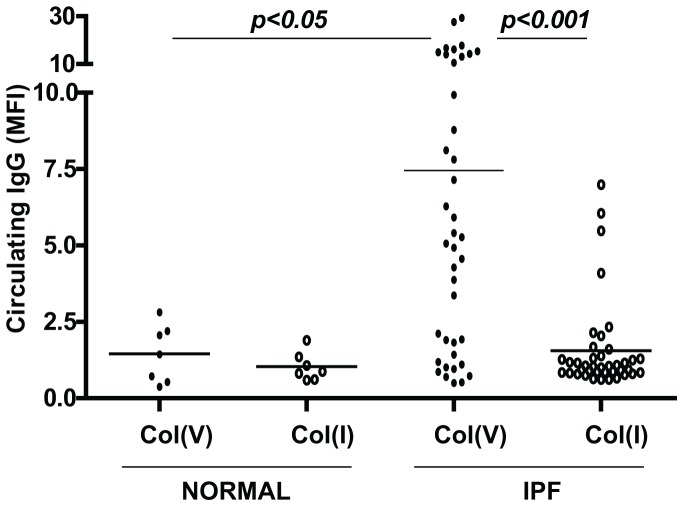
Although we have reported anti-col(V) cellular immunity in IPF [3], the relative circulating antibody levels of col(V) and col(I) are unknown. We investigated a cohort of 40 patients diagnosed with IPF as per ATS criteria as previously described [3]. The cohort has a gender distribution of 15 females and 25 males and their average age is 64.3 years with a standard deviation of 7.78. We observed that compared to normal healthy volunteers, anti-col(V) levels were higher in IPF patients (Figure 2) (p<0.05). Additionally, anti-col(I) antibodies were detected at very low levels in all patients studied. The above studies demonstrate that IPF is associated with systemic anti-col(V) antibodies specifically.
Figure 2. Auto-antibody responses of col(I) and col(V) in plasma of patients with UIP/IPF and normal volunteers.
Col(I) and col(V) antibodies were detected in the plasma by flow cytometry in plasma of patients with UIP/IPF with varying degrees of disease severity (n = 40) and healthy normal volunteers (n = 7). Values are represented as mean ± SEM; *, p<0.05, **, p<0.001; one-way ANOVA, post hoc test: Bonferroni.
Bleomycin-induced fibrosis results in col(V) overexpression and production of anti-col(V) antibodies
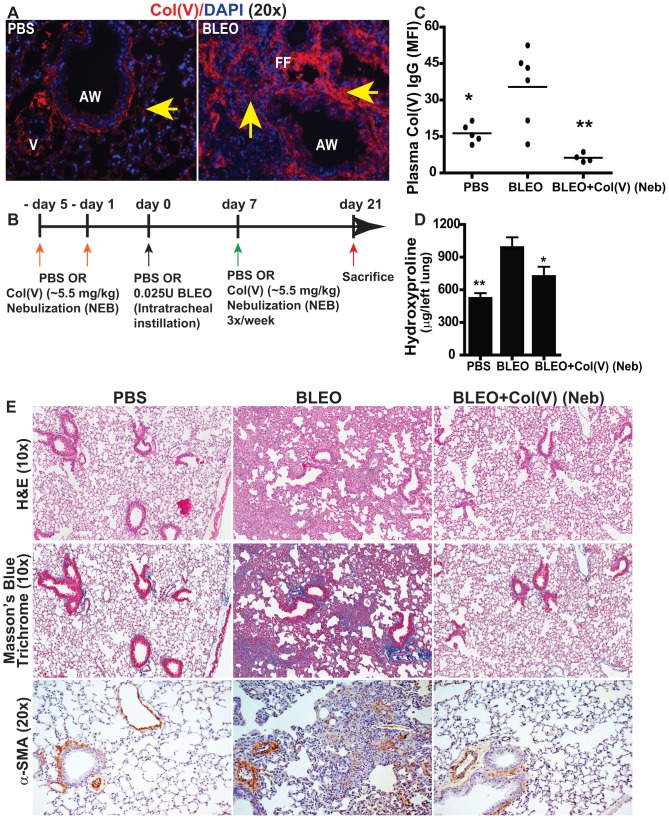
We next determined if the bleomycin-induced lung injury, as a model for IPF, results in col(V) overexpression, and anti-col(V) cellular and humoral immunity. Indeed, bleomycin-induced fibrosis was associated with overexpression of col(V) (Figure 3A), and development of anti-col(V) antibodies (Figure 3C) in a manner similar to that observed in clinical IPF. Since we have observed data showing that col(V) is the target of an autoimmune response in IPF in both pre-clinical and clinical studies, we next determined if col(V) could be utilized as a tolerogen to prevent fibrosis. Braun and colleagues have demonstrated that systemic administration of col(V) inhibits bleomycin-induced acute lung injury using measures of lung inflammation [20]. Since introducing antigens to the lung mucosa may result in tolerance, we nebulized col(V) into the lungs of mice using the experimental model of chronic bleomycin injury as illustrated in Figure 3B, followed by a systematic assessment of parameters associated with fibrosis. Compared to bleomycin-injured mice, nebulizing col(V) resulted in a reduction in anti-col(V) antibodies (Figure 3C), hydroxyproline concentration (Figure 3D), reduced connective tissue deposition as shown by trichrome staining and blunted myofibroblasts differentiation as observed by expression for α-smooth muscle actin (α-SMA) (Figure 3E).
Figure 3. Tolerance induction of Col(V) protects against bleomycin-induced fibrosis.
(A) Col(V) overexpression was detected in 21 day post bleomycin injury by labeling with rhodamine and counterstained with DAPI. (Original magnification, 10×). (B) Schematic illustration of the experimental design. (C) Circulating antibodies specific to col(V) were detected in 21 day post bleomycin injury mice. Values represent mean ± SEM; number of animals: PBS = 5, BLEO = 6 and BLEO+col(V) Neb = 4. Compared to bleomycin group, * p<0.001; ** p<0.01; one-way ANOVA, post hoc test: Bonferroni. (D) Collagen deposition was measured quantitatively by assaying for hydroxyproline concentrations from the whole left lung day 21 post bleomycin injury+col(V) treatment. Values represent mean ± SEM; n = 7 mice/group. Compared to bleomycin group, ** p<0.01; * p<0.05 one-way ANOVA, post hoc test: Newman-Keuhl's. (E) At day 21 post bleomycin injury, lung tissue sections were stained for H&E and Masson's blue trichrome staining. Extensive collagen deposition was observed with bleomycin injury while col(V) nebulized lungs, similar to normal lungs, had collagen deposition around airways and vasculature. Original magnifications: 10×. (Figure S2A: 1×). Lung tissue sections were immunostained against alpha-smooth muscle actin (α-SMA) or IgG. Streptavidin-conjugated horseradish peroxidase was used with 3,3′-diaminobenzidene as substrate (brown) and nuclei were counterstained with hematoxylin (blue). Original magnifications: 20×.
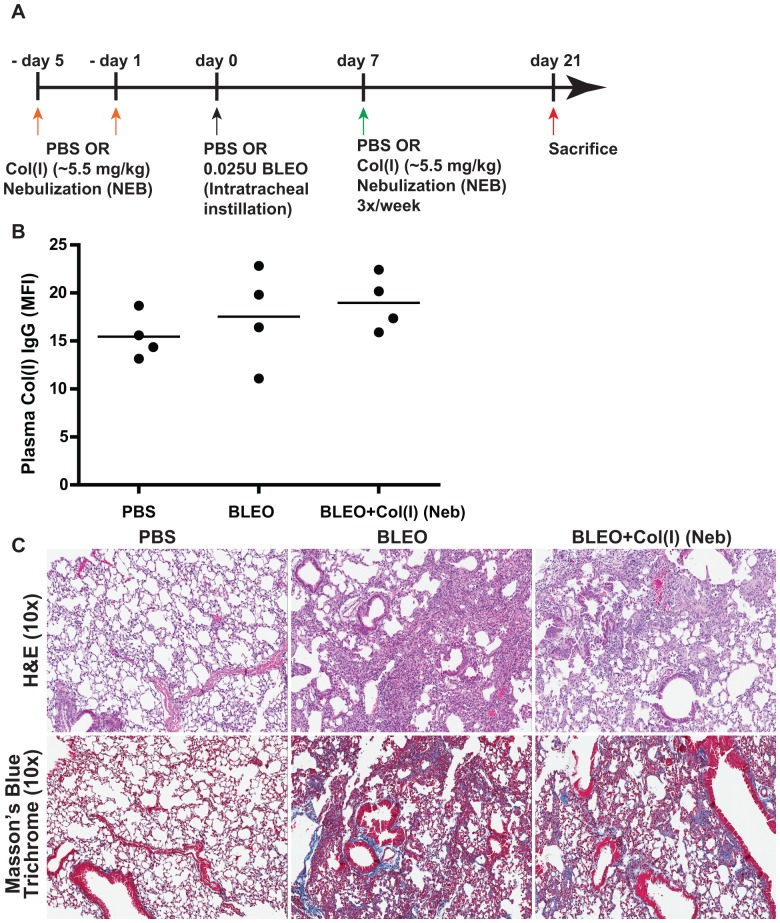
Type I collagen [col(I)], is the major lung collagen, and is highly expressed in IPF lungs [27]. However, high levels of circulating anti-col(I) antibodies were not detected in IPF patients (Figure 2) or in bleomycin-induced fibrosis (Figure 4B). Furthermore, nebulizing col(I) did not prevent fibrosis induced by bleomycin (Figure 4C). Our studies suggest that the fibro-protective effects of nebulizing a native lung protein, col(V) in particular, are a specific response.
Figure 4. Tolerance induction of Col(I) does not confer protection against bleomycin-induced fibrosis.
(A) Schematic illustration of the experimental design. (B) Circulating antibodies specific to col(I) were unchanged in 21 day post bleomycin injury mice. Values represent mean ± SEM; number of animals: PBS = 4, BLEO = 4 and BLEO+col(I) Neb = 4. (C) At day 21 post bleomycin injury, lung tissue sections were stained for H&E and Masson's blue trichrome staining. Extensive collagen deposition was observed with bleomycin injury and col(I) nebulized lungs. Original magnifications: 10×. (Figure S2B: 1×).
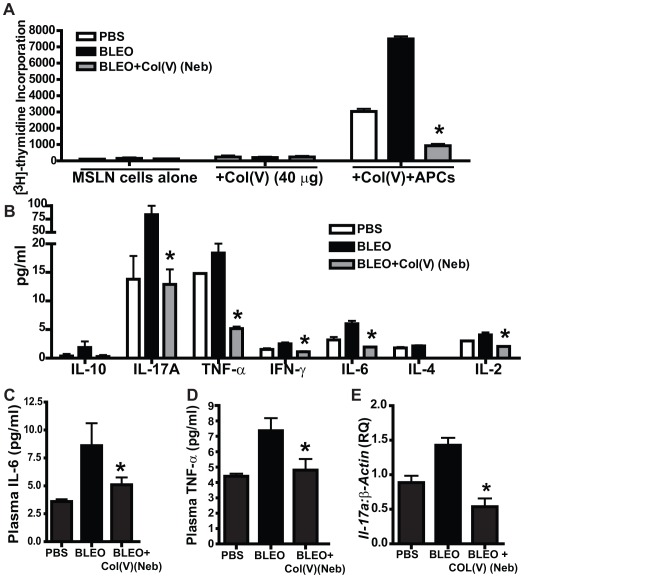
Tolerance induction of col(V) suppresses local T lymphocyte proliferation and pro-inflammatory/pro-fibrotic cytokine expression
We have previously demonstrated lung allograft rejection is in part mediated by anti-col(V) immunity and that col(V)-induced tolerance abrogated the rejection response [28] and Wilson and colleagues had reported that bleomycin-induced lung fibrosis may be T cell dependent [29]. We next determined if immune tolerance was the mechanism of col(V)-induced prevention of lung fibrosis we had earlier shown in Figure 3. Data in Figure 5 shows that mediastinal lymph node lymphocytes isolated from col(V)-treated mice do not proliferate in response to col(V) when presented by autologous antigen presenting cells, therefore indicating col(V)-induced tolerance (Figure 5A). IL-17, IL-6, and TNF-α have all been linked to fibrosis [29]–[31]. Therefore, we next examined the cytokines produced in the supernatants of the mixed leukocyte reactions reported in Figure 5A. Notably, col(V)-induced tolerance was associated with IL-17A, IL-6 and TNF-α (Figure 5B). We also noted a reduction in IFN-γ but not IL-10 (Figure 5B). These changes were also observed systemically by data showing lower serum levels of IL-6 and TNF-α (Figure 5C, D), as well as lower lung transcript levels for Il-17a in col(V)-treated mice (Figure 5E), albeit at an earlier time point at day 14 wherein we could detect higher mRNA expression of Il-17a in bleomycin-injured mice. Collectively, the above data indicates that immune tolerance induced by col(V) protects against bleomycin injury by blunting T cell activation and the associated systemic and local expression of pro-inflammatory and pro-fibrotic cytokines.
Figure 5. Tolerance induction of col(V) suppresses T lymphocytes activation and associated pro-inflammatory/pro-fibrotic cytokine expression.
(A) At day 21 post bleomycin injury, mediastinal lymphocytes were isolated and cultured alone or in the presence of autologous antigen presenting cells (APCs from naïve C57-BL/6 mice) for 48 h. The cells were then radiolabeled with tritiated thymidine for 16 h and assessed for proliferation rates. Values represent mean ± SEM. n = 5 mice/group; Compared to bleomycin group, * p<0.01; one-way ANOVA, post hoc test: Bonferroni. (B) Conditioned media from A was analyzed for Th1/Th2/Th17 cytokines. Values represent mean ± SEM. n = 5 mice/group; Compared to bleomycin group, * p<0.01; one-way ANOVA, post hoc test: Bonferroni. (C, D) Plasma samples from (A) were analyzed for Th1/Th2/Th17 cytokines. Values represent mean ± SEM. n = 5 mice/group; Compared to bleomycin group, * p<0.01; one-way ANOVA, post hoc test: Bonferroni. (E) At day 14 post bleomycin injury, whole lung homogenates from bleomycin-injured mice+col(V) treatment were quantified for il-17a mRNA expression and normalized for β-actin. Values represent mean ± SEM. n = 5 mice/group; compared to bleomycin group, * p<0.01; one-way ANOVA, post hoc test: Bonferroni.
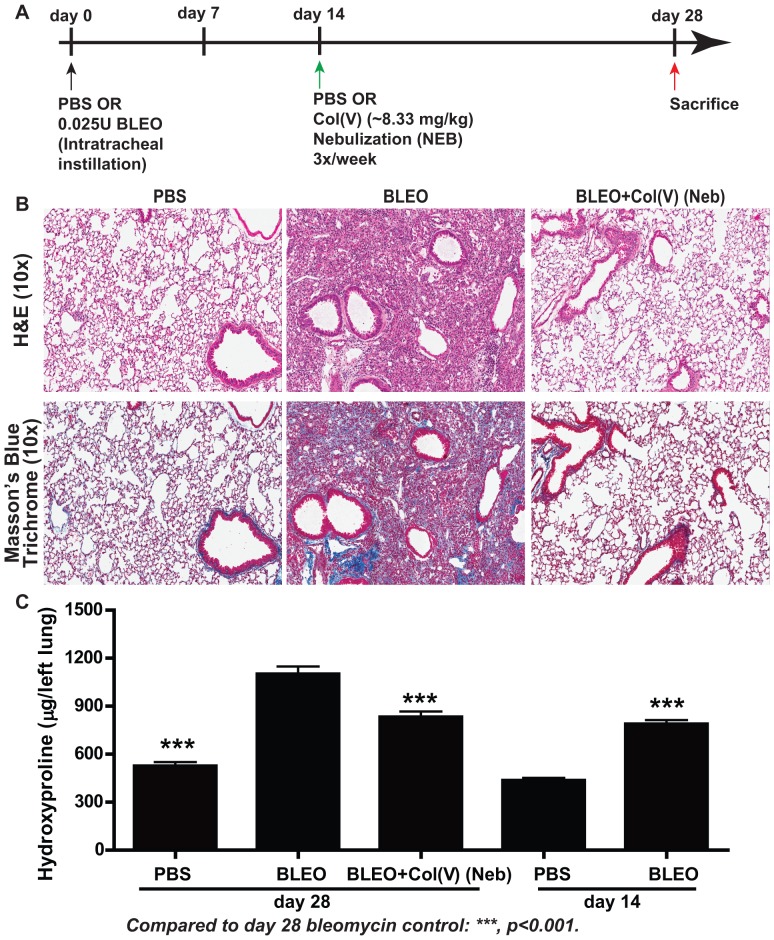
Col(V) prevents collagen accumulation in established fibrosis
We next determined if nebulized col(V) would arrest ongoing collagen deposition in a model of chronic fibrosis induced by bleomycin. Based on efficacy established in preliminary studies, we nebulized 8.33 mg/kg bodyweight of col(V) protein three times a week beginning at day 14 post bleomycin injury, a time when fibrosis is established [32], followed by sacrifice at day 28 as schematically presented in Figure 6A. Notably, we observed significant protection from fibrosis (Figure 6B; top panel) and attenuated collagen deposition as observed by Masson's trichrome staining for collagen (Figure 6B; lower panel) and hydroxyproline content (Figure 6C). Notably, at the time of nebulization of col(V), there was significantly higher hydroxyproline concentrations in the lung at day 14 post bleomycin instillation compared to PBS-instilled lungs (p<0.001). We then observed that compared to day 14 post bleomycin instillation, at day 28 post bleomycin, the lungs had higher hydroxyproline concentration (p<0.01). Compared to day 28 post bleomycin, col(V) nebulization significantly attenuated hydroxyproline concentrations in the lung (p<0.001). Through our studies, we conclude that delayed administration of col(V) in a nebulized form is effective in arresting the progression of established fibrosis.
Figure 6. Col(V) treatment protects against established fibrosis.
(A) Schematic illustration of the experimental design. (B) At day 28 post bleomycin injury, lung tissue sections were stained for H&E and Masson's blue trichrome staining. Extensive collagen deposition was observed with bleomycin injury while col(V) nebulized lungs, similar to normal lungs, had collagen deposition around airways and vasculature. Original magnifications: 10×. (Figure S2C: 1×). (C) Collagen deposition was measured quantitatively by assaying for hydroxyproline concentrations from the whole left lung day 28 and day 14 post bleomycin injury+col(V) treatment. Values represent mean ± SEM; n = 12 mice/group; * p<0.001 for day 28 bleomycin vs. day 28 PBS and day 28 col(V) (Neb); day 14 bleomycin vs. day 14 PBS; day 28 bleomycin vs. day 14 bleomycin; one-way ANOVA, post hoc test: Bonferroni.
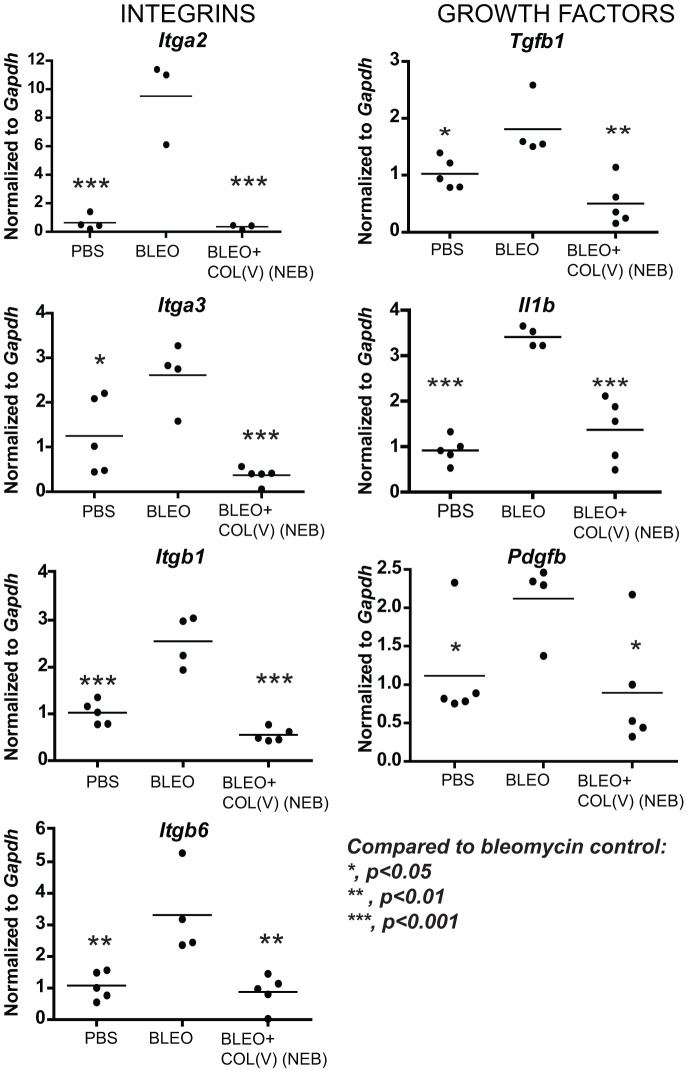
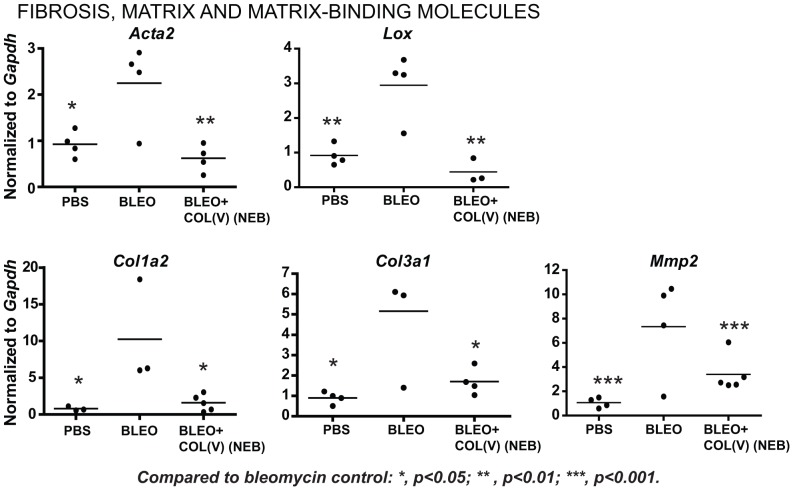
Nebulized col(V) downregulates local fibrosis-related transcripts
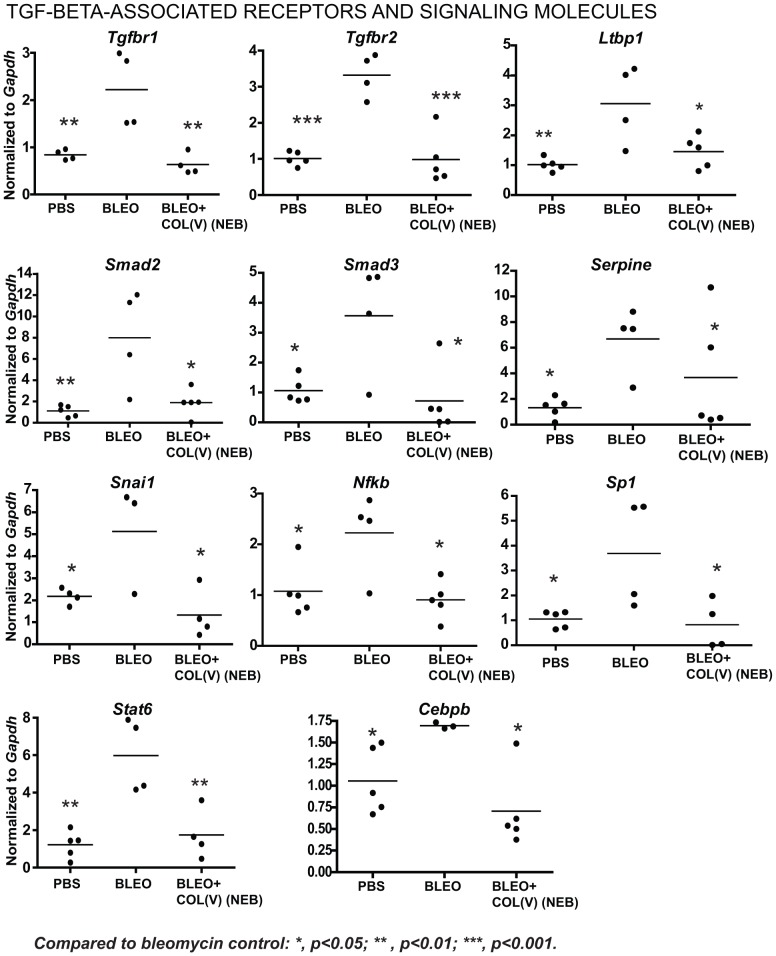
To further investigate mechanisms underlying the beneficial effects of col(V) in established fibrosis, we next analyzed transcript expression for fibrosis-related genes in the bleomycin model. In a comparison of PBS or bleomycin-instilled lung, we observed upregulation of genes from five major functional classes implicated as fibrogenic factors in IPF (Table 1). Nebulized delivery of col(V), suppressed bleomycin-induced genes listed in the following functional categories: integrins (Itga1, Itga2, Itgb1, Itgb6, Figure 7), pro-fibrotic growth factors (Tgfb, Il1b, Pdgfb, Figure 7), fibrosis and matrix-related molecules (Acta2, Col1a2, Col3a1,Mmp2, Lox, Figure 8) and members of the TGF-beta superfamily (Tgfbr1/2, Smad2/3, Ltbp1, Serpine1, Nfkb/Sp1/Cebpb, Figure 9) in addition to Snai-1 and Stat6 (Figure 9). These data suggest that treatment with col(V) may lead to attenuation of multiple pro-fibrotic genes involved in the pathogenesis of IPF.
Table 1. List of bleomycin-induced genes modulated by col(V) treatment.
| Gene | Bonferroni-adjusted P value | ||||
| Functional Class | Unigene | Symbol | Gene Description | Upregulation | Downregulation |
| PBS vs BLEO | BLEO vs BLEO+COL(V) | ||||
| Integrins | |||||
| Mm.5007 | Itga2 | Integrin alpha 2 | 0.001 | 0.001 | |
| Mm.57035 | Itga3 | Integrin alpha 3 | 0.05 | 0.001 | |
| Mm.263396 | Itb1 | Integrin beta 1 (fibronectin receptor beta) | 0.001 | 0.001 | |
| Mm.98193 | Itb6 | Integrin beta 6 | 0.01 | 0.01 | |
| Cytokines/Growth Factors | |||||
| Mm.248380 | Tgfb1 | Transforming growth factor, beta 1 | 0.05 | 0.01 | |
| Mm.222830 | Il1b | Interleukin 1 beta | 0.001 | 0.001 | |
| Mm.144089 | Pdgfb | Platelet derived growth factor, B polypeptide | 0.05 | 0.05 | |
| Fibrosis, matrix and matrix-binding molecules | |||||
| Mm.213025 | Acta2 | Actin, alpha 2, smooth muscle, aorta | 0.05 | 0.01 | |
| Mm.277792 | Col1a2 | Collagen, type I, alpha 2 | 0.05 | 0.05 | |
| Mm.249555 | Col3a1 | Collagen, type III, alpha 1 | 0.05 | 0.05 | |
| Mm.172 | Lox | Lysyl oxidase | 0.01 | 0.01 | |
| Mm.29564 | Mmp2 | Matrix metallopeptidase 2 | 0.05* | 0.05* | |
| Mm.439656 | Cebpb | CCAAT/enhancer binding protein (C/EBP), beta, latency associated peptide (LAP) | 0.05* | 0.05* | |
| Members of the TGF-beta superfamily | |||||
| Mm.197552 | Tgfbr1 | Transforming growth factor, beta receptor I, ALK5 | 0.05 | 0.01 | |
| Mm.172346 | Tgfbr2 | Transforming growth factor, beta receptor II | 0.001 | 0.001 | |
| Mm.269747 | Ltbp1 | Latent transforming growth factor beta binding protein 1 | 0.01 | 0.01 | |
| Mm.391091 | Smad2 | MAD homolog 2 (Drosophila) | 0.01 | 0.05 | |
| Mm.7320 | Smad3 | MAD homolog 3 (Drosophila) | 0.05 | 0.05 | |
| Mm.250422 | Serpine1 | Serine (or cysteine) peptidase inhibitor, clade E, member 1, PAI-1 | 0.05 | 0.05 | |
| Transcription factors | |||||
| Mm.2093 | Snai1 | Snail homolog 1 (Drosophila) | 0.05* | 0.01* | |
| Mm.256765 | Nfkb | Nuclear factor of kappa light polypeptide gene enhancer in B-cells 1, p105 | 0.05 | 0.05 | |
| Mm.4618 | Sp1 | Trans-acting transcription factor 1 | 0.05 | 0.05 | |
| Mm.121721 | Stat6 | Signal transducer and activator of transcription 6 | 0.01 | 0.01 | |
indicates Newman-Keul's-adjusted P value.
Figure 7. Col(V) treatment downregulates integrins, TGF-β and other pro-fibrotic cytokines.
At day 28 post bleomycin injury, lung tissues were homogenized and cDNA was analyzed for fibrosis-specific gene expression analyses.
Figure 8. Col(V) treatment downregulates matrix molecules.
At day 28 post bleomycin injury, lung tissues were homogenized and cDNA was analyzed for fibrosis-specific gene expression analyses.
Figure 9. Col(V) treatment downregulates TGF-β associated receptors, signaling molecules and transcription factors.
At day 28 post bleomycin injury, lung tissues were homogenized and cDNA was analyzed for fibrosis-specific gene expression analyses.
Discussion
Col(V), a minor collagen in the lung, is sequestered within the fibrils of type I collagen, the major pulmonary collagen. Our studies demonstrate the systemic production of anti-col(V) autoantibodies as a specific response and associated local expression of individual alpha chains of col(V) in clinical IPF specimens. Accordingly, tolerizing mice with nebulized col(V), but not col(I), prevented lung fibrosis and lower production of inflammatory cytokines. The current study also shows that nebulized col(V) protein arrests further development of bleomycin-induced fibrosis by the suppression of multiple fibrosis-related genes, specifically members of the TGF-β superfamily. To the best of our knowledge, the current study is the first to show the efficacy of nebulized therapy of a native protein in the treatment of established fibrosis. mRNA transcript data indicate that the beneficial effect of col(V) likely involves multiple pathways involved in fibrogenesis.
Previous reports by Parra and colleagues describe the role of col(V) in the pathogenesis of IPF by illustrating the distributions of col(V) in two different lung compartments and by using three-dimensional reconstruction to evaluate the amount of collagen [26]. The potential caveat therein may possibly be the green auto fluorescence emitted by lungs resulting in interference of measurements. Our studies demonstrate the overexpression of the individual chains of col(V) proteins in IPF lungs, which was determined by pepsin digestion of the lungs followed by careful systematic extraction of total collagen [22]. The mRNA expression of col(V) was analyzed in IPF tissue sections with confirmed fibrotic lesions. Similarly, our pre-clinical studies provide evidence that nebulized col(V), but not col(I), blunts circulating autoantibodies and renders col(V)-specific immunosuppression with attenuation of associated cytokines. Braun and colleagues examined effects of systemically tolerized col(V) in a bleomycin model of acute lung injury by using measures of inflammation [20]. We present effects of nebulized col(V) in a bleomycin model of chronic fibrosis using hallmarks of bleomycin-induced fibrosis/IPF: a) T cell activation, b) collagen deposition and c) myofibroblast differentiation.
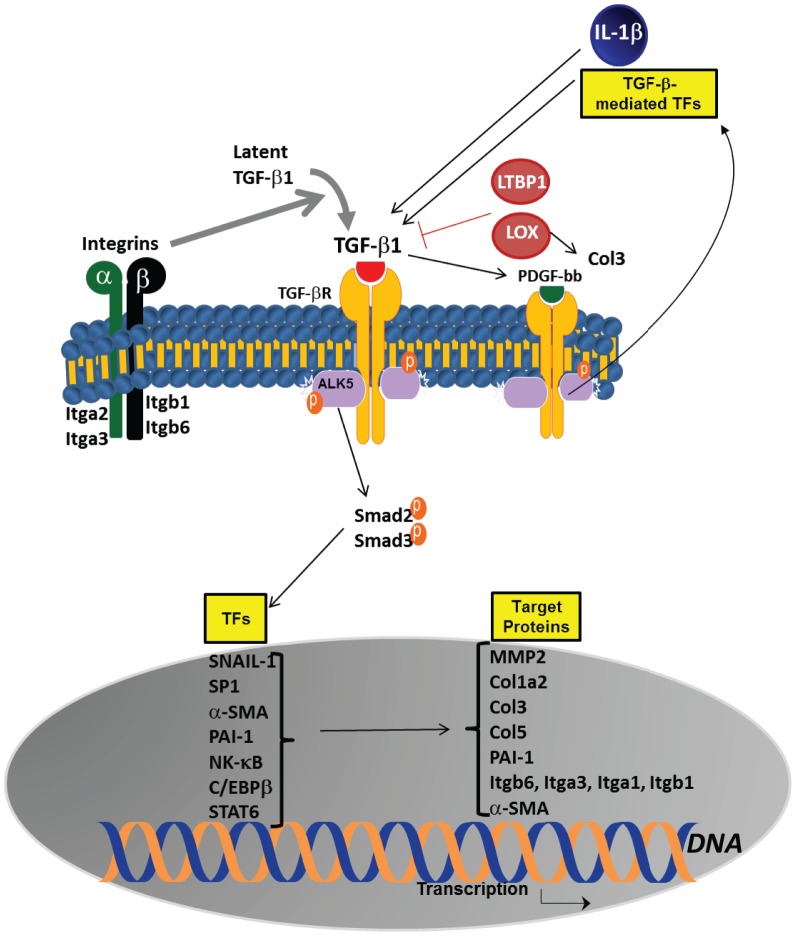
In order to simulate the relevant clinical setting wherein lungs of IPF patients at the time of diagnosis are significantly scarred, we used the bleomycin model of established fibrosis. Henderson and colleagues have previously demonstrated the effectiveness of using a therapeutic model examining bleomycin-induced lung fibrosis in which an intervention was administered at the time of established fibrosis to determine if fibrosis could be reversed [32]. Our demonstrate that nebulized col(V) arrests the progression of established fibrosis possibly via mitigation of multiple key fibrotic pathways (Figure 10). However, these pathways appear to be centrally revolving around TGF-β and its associated downstream signaling molecules. Even more significantly, a few integrins, the major receptors for cell adhesion to extracellular matrix proteins and other cells, are downregulated in response to col(V) treatment and are discussed here. αVβ6 integrin (Itgb6) activates latent TGF-β and hence lack of this gene [33] or its inhibition [34] protects against lung fibrosis. α3β1 integrin (Itgb1) has been implicated in activation of latent TGF-β and hence epithelial-mesenchymal transition [35] and additionally, it facilitates β-catenin-Smad3 crosstalk to promote myofibroblast formation and survival [9]. α2β1 integrin (Itga2), a receptor for type I collagen, is reportedly upregulated in response to TNF-α in IPF lung-derived fibroblasts [36].
Figure 10. Schematic representation of pathways affected by intrapulmonary instillation of col(V).
While bleomycin-induced TGF-β and members of the TGF-β superfamily are suppressed by col(V) nebulization, other pro-fibrotic molecules which crosstalk with TGF-β are suppressed by col(V) and they are discussed below: (i) IL-1β causes acute lung injury via integrin-dependent mechanisms, including αVβ6 integrin [7] and (ii) PDGF signaling pathways [37]. Increased levels of lysyl oxidase (LOX), an amine oxidase critical for the initiation of collagen and elastin cross-linking [38], increased mRNA levels of collagen type I (Col1a2), collagen type III (Col3a1) and matrix metalloproteinase (Mmp2) have been reported in murine models of lung fibrosis [39]. Interestingly, Serpine1 - also known as the TGF-β-regulated protein PAI-1 – has been reported to directly correlate with collagen accumulation and that the TGF-β/PAI-1 signaling axis plays a vital role in myofibroblast survival [40]. The nuclear transcription factors which are blocked by col(V) treatment and implicated in pulmonary fibrosis are NF-κB [41], Sp1 [42] and CCAAT/enhancer-binding protein β (Cebpb) [43], SNAIL [44] and STAT6 [45]. While Velosa et al [18] have demonstrated that col(V)-induced tolerance downregulates TGF-β in an experimental model of systemic sclerosis, they had not investigated other related pathways which might likely fuel the pro-fibrotic signaling. Our studies suggest that col(V) likely confers its fibroprotective effects by targeting integrins, in addition to TGF-β and its associated-signaling pathways. Notably, we detected some bleomycin-induced genes which were unchanged by col(V) treatment and other genes which were unchanged due to bleomycin induction at the determined time of analyses (Figure S3).
In this report, we present evidence which identifies col(V) in an emerging class of target molecules combating specific autoimmune/fibrotic responses. Recent investigations have focused on certain pathologic inflammatory responses which may be due to autoimmunity wherein self-reactive circulating autoantibodies have been detected among patients and associated with acute disease process [46], [47]. While early studies show presence of circulating immune complexes [48]; a recent report has identified mutations in autoimmune regulator gene as a link between autoimmune responses and defective central immunotolerance in IPF [49]. Hence, circulating antibodies to self-antigens have been widely documented but their roles as potential immunotherapy has not been demonstrated. A prior study from our laboratory has identified the localization of col(V) in the apical surface of airway epithelial cells [21]. Interestingly, most of the documented evidence on auto-antibodies has been related to proteins specific to the epithelium which is the primary line of defense against injury due to pro-inflammatory/pro-fibrotic cytokines, pathogenic microbes and other agents such as the reactive oxygen species.
The limitations in this study would call for longitudinal studies in clinical tissues to understand the trend of antibody response relative to disease progression. Similarly, in our mouse models, elucidation of the functional and phenotypic effects of col(V) antibodies and their products on key cell types involved in the pathogenesis of IPF, such as epithelial, endothelial or mesenchymal cells. The interaction site between col(V) and integrins coupled with their functional and phenotypic effects in epithelial and mesenchymal cells are unknown. Therefore, the role of potential signaling molecules unraveled in this study, in addition to the effect of col(V) antibodies on impaired epithelialization, tissue repair and matrix remodeling, which are typical features of IPF, will be the target of our future studies in understanding this antigen.
In summary, we present evidence that autoimmunity to col(V) has a role in the pathogenesis of IPF. These studies also provide further insight for potential mechanisms, particularly TGF-β-related signaling molecules, which may be driving the auto-antigen specific immunosuppression for IPF.
Supporting Information
Pepsin digested lung homogenates (15 µg) and corresponding standards run in a 5% gel and Coomassie stained. Image shown here is representative of 3 normal and 5 IPF tissues.
(TIF)
A. Tolerance induction of col(V) protects against bleomycin-induced fibrosis. H&E and trichrome images of data presented in Figure 3. B. Tolerance induction of col(I) does not protect against bleomycin-induced fibrosis. H&E and trichrome images of data presented in Figure 4. C. Col(V) treatment protects against bleomycin-induced fibrosis. H&E and trichrome images of data presented in Figure 4. Original magnifications: 1×.
(TIF)
Hierarchical clustergram of all 80 genes modulated by col(V) treatment.
(TIF)
Excel file with raw data from the PCR-based gene array is provided in Table S1 (Supplemental Information-5).
(XLS)
Supplementary Methods.
(DOCX)
Acknowledgments
Bovine amnion collagen type V protein was a generous donation by ImmuneWorks, Inc. Bovine amnion collagen type I protein was a generous donation by Dr. David Brand, Vanderbilt University.
Funding Statement
This work was supported by NIH-NHLBI-Lung Tissue Research Consortium and NIH-NHLBI-HL109288 to RV; NIH-NCAATS-KL2 TR000163 (A. Shekhar, PI; Co-investigator: RV); and by the NIH-NHLBI-HL067177 and NIAID P01AI084853 to DSW. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Crystal RG, Bitterman PB, Rennard SI, Hance AJ, Keogh BA (1984) Interstitial lung diseases of unknown cause. Disorders characterized by chronic inflammation of the lower respiratory tract (first of two parts). N Engl J Med 310: 154–166. [DOI] [PubMed] [Google Scholar]
- 2. Strieter RM, Gomperts BN, Keane MP (2007) The role of CXC chemokines in pulmonary fibrosis. J Clin Invest 117: 549–556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Bobadilla JL, Love RB, Jankowska-Gan E, Xu Q, Haynes LD, et al. (2008) Th-17, monokines, collagen type V, and primary graft dysfunction in lung transplantation. Am J Respir Crit Care Med 177: 660–668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Burlingham WJ, Love RB, Jankowska-Gan E, Haynes LD, Xu Q, et al. (2007) IL-17-dependent cellular immunity to collagen type V predisposes to obliterative bronchiolitis in human lung transplants. J Clin Invest 117: 3498–3506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Wilson MS, Wynn TA (2009) Pulmonary fibrosis: pathogenesis, etiology and regulation. Mucosal Immunol 2: 103–121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Greenlee KJ, Werb Z, Kheradmand F (2007) Matrix metalloproteinases in lung: multiple, multifarious, and multifaceted. Physiol Rev 87: 69–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Ganter MT, Roux J, Miyazawa B, Howard M, Frank JA, et al. (2008) Interleukin-1beta causes acute lung injury via alphavbeta5 and alphavbeta6 integrin-dependent mechanisms. Circ Res 102: 804–812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Katsumoto TR, Violette SM, Sheppard D (2011) Blocking TGF-beta via Inhibition of the alphavbeta6 Integrin: A Possible Therapy for Systemic Sclerosis Interstitial Lung Disease. Int J Rheumatol 2011: 208219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Kim KK, Wei Y, Szekeres C, Kugler MC, Wolters PJ, et al. (2009) Epithelial cell alpha3beta1 integrin links beta-catenin and Smad signaling to promote myofibroblast formation and pulmonary fibrosis. J Clin Invest 119: 213–224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Muro AF, Moretti FA, Moore BB, Yan M, Atrasz RG, et al. (2008) An essential role for fibronectin extra type III domain A in pulmonary fibrosis. Am J Respir Crit Care Med 177: 638–645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Sueblinvong V, Neujahr DC, Mills ST, Roser-Page S, Ritzenthaler JD, et al. (2012) Predisposition for disrepair in the aged lung. Am J Med Sci 344: 41–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Sime PJ, Xing Z, Graham FL, Csaky KG, Gauldie J (1997) Adenovector-mediated gene transfer of active transforming growth factor-beta1 induces prolonged severe fibrosis in rat lung. J Clin Invest 100: 768–776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Fujimoto H, Gabazza EC, Taguchi O, Nishii Y, Nakahara H, et al. (2006) Thrombin-activatable fibrinolysis inhibitor deficiency attenuates bleomycin-induced lung fibrosis. Am J Pathol 168: 1086–1096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Wilson MS, Madala SK, Ramalingam TR, Gochuico BR, Rosas IO, et al. (2010) Bleomycin and IL-1beta-mediated pulmonary fibrosis is IL-17A dependent. J Exp Med 207: 535–552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Munger JS, Sheppard D (2011) Cross talk among TGF-beta signaling pathways, integrins, and the extracellular matrix. Cold Spring Harb Perspect Biol 3: a005017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Hynes RO (1992) Integrins: versatility, modulation, and signaling in cell adhesion. Cell 69: 11–25. [DOI] [PubMed] [Google Scholar]
- 17. Staatz WD, Fok KF, Zutter MM, Adams SP, Rodriguez BA, et al. (1991) Identification of a tetrapeptide recognition sequence for the alpha 2 beta 1 integrin in collagen. J Biol Chem 266: 7363–7367. [PubMed] [Google Scholar]
- 18. Velosa AP, Teodoro WR, dos Anjos DM, Konno R, Oliveira CC, et al. (2010) Collagen V-induced nasal tolerance downregulates pulmonary collagen mRNA gene and TGF-beta expression in experimental systemic sclerosis. Respir Res 11: 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Braun RK, Molitor-Dart M, Wigfield C, Xiang Z, Fain SB, et al. (2009) Transfer of tolerance to collagen type V suppresses T-helper-cell-17 lymphocyte-mediated acute lung transplant rejection. Transplantation 88: 1341–1348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Braun RK, Martin A, Shah S, Iwashima M, Medina M, et al. (2010) Inhibition of bleomycin-induced pulmonary fibrosis through pre-treatment with collagen type V. J Heart Lung Transplant [DOI] [PubMed] [Google Scholar]
- 21. Iwata T, Philipovskiy A, Fisher AJ, Presson RG Jr, Chiyo M, et al. (2008) Anti-type V collagen humoral immunity in lung transplant primary graft dysfunction. J Immunol 181: 5738–5747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Smith GN Jr, Hasty KA, Yu LP Jr, Lamberson KS, Mickler EA, et al. (1991) Cleavage of type XI collagen fibers by gelatinase and by extracts of osteoarthritic canine cartilage. Matrix 11: 36–42. [DOI] [PubMed] [Google Scholar]
- 23. Hecker L, Vittal R, Jones T, Jagirdar R, Luckhardt TR, et al. (2009) NADPH oxidase-4 mediates myofibroblast activation and fibrogenic responses to lung injury. Nat Med 15: 1077–1081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Labiris NR, Dolovich MB (2003) Pulmonary drug delivery. Part I: physiological factors affecting therapeutic effectiveness of aerosolized medications. Br J Clin Pharmacol 56: 588–599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Delacoux F, Fichard A, Geourjon C, Garrone R, Ruggiero F (1998) Molecular features of the collagen V heparin binding site. J Biol Chem 273: 15069–15076. [PubMed] [Google Scholar]
- 26. Parra ER, Teodoro WR, Velosa AP, de Oliveira CC, Yoshinari NH, et al. (2006) Interstitial and vascular type V collagen morphologic disorganization in usual interstitial pneumonia. J Histochem Cytochem 54: 1315–1325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Scotton CJ, Chambers RC (2007) Molecular targets in pulmonary fibrosis: the myofibroblast in focus. Chest 132: 1311–1321. [DOI] [PubMed] [Google Scholar]
- 28. Yamada Y, Sekine Y, Yoshida S, Yasufuku K, Petrache I, et al. (2009) Type V collagen-induced oral tolerance plus low-dose cyclosporine prevents rejection of MHC class I and II incompatible lung allografts. J Immunol 183: 237–245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Wilson MS, Madala SK, Ramalingam TR, Gochuico BR, Rosas IO, et al. (2009) Bleomycin and IL-1beta-mediated pulmonary fibrosis is IL-17A dependent. J Exp Med 207: 535–552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Moodley YP, Misso NL, Scaffidi AK, Fogel-Petrovic M, McAnulty RJ, et al. (2003) Inverse effects of interleukin-6 on apoptosis of fibroblasts from pulmonary fibrosis and normal lungs. Am J Respir Cell Mol Biol 29: 490–498. [DOI] [PubMed] [Google Scholar]
- 31. Chapman HA (1999) A Fas pathway to pulmonary fibrosis. J Clin Invest 104: 1–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Henderson WR Jr, Chi EY, Ye X, Nguyen C, Tien YT, et al. (2010) Inhibition of Wnt/beta-catenin/CREB binding protein (CBP) signaling reverses pulmonary fibrosis. Proc Natl Acad Sci U S A 107: 14309–14314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Munger JS, Huang X, Kawakatsu H, Griffiths MJ, Dalton SL, et al. (1999) The integrin alpha v beta 6 binds and activates latent TGF-beta 1: a mechanism for regulating pulmonary inflammation and fibrosis. Cell 96: 319–328. [DOI] [PubMed] [Google Scholar]
- 34. Puthawala K, Hadjiangelis N, Jacoby SC, Bayongan E, Zhao Z, et al. (2008) Inhibition of integrin alpha(v)beta6, an activator of latent transforming growth factor-beta, prevents radiation-induced lung fibrosis. Am J Respir Crit Care Med 177: 82–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Kim KK, Kugler MC, Wolters PJ, Robillard L, Galvez MG, et al. (2006) Alveolar epithelial cell mesenchymal transition develops in vivo during pulmonary fibrosis and is regulated by the extracellular matrix. Proc Natl Acad Sci U S A 103: 13180–13185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Vancheri C, Sortino MA, Tomaselli V, Mastruzzo C, Condorelli F, et al. (2000) Different expression of TNF-alpha receptors and prostaglandin E(2)Production in normal and fibrotic lung fibroblasts: potential implications for the evolution of the inflammatory process. Am J Respir Cell Mol Biol 22: 628–634. [DOI] [PubMed] [Google Scholar]
- 37. Lo Re S, Lecocq M, Uwambayinema F, Yakoub Y, Delos M, et al. (2011) Platelet-derived growth factor-producing CD4+ Foxp3+ regulatory T lymphocytes promote lung fibrosis. Am J Respir Crit Care Med 184: 1270–1281. [DOI] [PubMed] [Google Scholar]
- 38. Yamaguchi Y, Takihara T, Chambers RA, Veraldi KL, Larregina AT, et al. (2012) A peptide derived from endostatin ameliorates organ fibrosis. Sci Transl Med 4: 136ra171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Kenyon NJ, Ward RW, McGrew G, Last JA (2003) TGF-beta1 causes airway fibrosis and increased collagen I and III mRNA in mice. Thorax 58: 772–777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Horowitz JC, Rogers DS, Simon RH, Sisson TH, Thannickal VJ (2008) Plasminogen activation induced pericellular fibronectin proteolysis promotes fibroblast apoptosis. Am J Respir Cell Mol Biol 38: 78–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Lin X, Sime PJ, Xu H, Williams MA, LaRussa L, et al. (2011) Yin yang 1 is a novel regulator of pulmonary fibrosis. Am J Respir Crit Care Med 183: 1689–1697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Sysa P, Potter JJ, Liu X, Mezey E (2009) Transforming growth factor-beta1 up-regulation of human alpha(1)(I) collagen is mediated by Sp1 and Smad2 transacting factors. DNA Cell Biol 28: 425–434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Hu B, Wu Z, Nakashima T, Phan SH (2012) Mesenchymal-specific deletion of C/EBPbeta suppresses pulmonary fibrosis. Am J Pathol 180: 2257–2267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Ulsamer A, Wei Y, Kim KK, Tan K, Wheeler S, et al. (2012) Axin pathway activity regulates in vivo pY654-beta-catenin accumulation and pulmonary fibrosis. J Biol Chem 287: 5164–5172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Liu T, Baek HA, Yu H, Lee HJ, Park BH, et al. (2011) FIZZ2/RELM-beta induction and role in pulmonary fibrosis. J Immunol 187: 450–461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Feghali-Bostwick CA, Tsai CG, Valentine VG, Kantrow S, Stoner MW, et al. (2007) Cellular and humoral autoreactivity in idiopathic pulmonary fibrosis. J Immunol 179: 2592–2599. [DOI] [PubMed] [Google Scholar]
- 47. Taille C, Grootenboer-Mignot S, Boursier C, Michel L, Debray MP, et al. (2011) Identification of periplakin as a new target for autoreactivity in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 183: 759–766. [DOI] [PubMed] [Google Scholar]
- 48. Nagaya H, Elmore M, Ford CD (1973) Idiopathic interstitial pulmonary fibrosis. An immune complex disease? Am Rev Respir Dis 107: 826–830. [DOI] [PubMed] [Google Scholar]
- 49. Shum AK, DeVoss J, Tan CL, Hou Y, Johannes K, et al. (2009) Identification of an autoantigen demonstrates a link between interstitial lung disease and a defect in central tolerance. Sci Transl Med 1: 9ra20. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Pepsin digested lung homogenates (15 µg) and corresponding standards run in a 5% gel and Coomassie stained. Image shown here is representative of 3 normal and 5 IPF tissues.
(TIF)
A. Tolerance induction of col(V) protects against bleomycin-induced fibrosis. H&E and trichrome images of data presented in Figure 3. B. Tolerance induction of col(I) does not protect against bleomycin-induced fibrosis. H&E and trichrome images of data presented in Figure 4. C. Col(V) treatment protects against bleomycin-induced fibrosis. H&E and trichrome images of data presented in Figure 4. Original magnifications: 1×.
(TIF)
Hierarchical clustergram of all 80 genes modulated by col(V) treatment.
(TIF)
Excel file with raw data from the PCR-based gene array is provided in Table S1 (Supplemental Information-5).
(XLS)
Supplementary Methods.
(DOCX)