It is now common knowledge that we are in the midst of a worldwide epidemic of type 2 diabetes (T2D). This disease occurs disproportionately in African Americans, Hispanic Americans, and other minorities. T2D is a major cause of cardiovascular disease morbidity and mortality, and these unfavorable outcomes are greatly increased in the presence of T2D (1–4). Impaired glucose tolerance (IGT) is itself a risk factor for cardiovascular disease (5). Obesity and IGT are each major risk factors for T2D, with yearly incidence of T2D in individuals with IGT ranging from 3.9 to 8.7% in various American populations, with an average of 5.7% (6), and averaging 6.5% in a meta-analysis of global studies (7). Both insulin resistance and diminished β-cell function (insulin secretion) are crucial pathogenetic factors in the progression from IGT to T2D, with the proportional impact of these defects varying in different reports and populations (8). The rate of progression can be reduced by lifestyle modification leading to weight loss (9) and the use of metformin (9), the thiazolidinedione drugs pioglitazone (PIO) (10) and rosiglitazone (11), α-glucosidase inhibitors (12), sulfonylureas (13), and insulin (14). Selection of therapeutic agents could be enhanced by matching their dominant actions on insulin resistance and/or β-cell dysfunction to the relative dominance of these abnormalities in specific individuals or populations with IGT.
For the above reasons, the study by DeFronzo et al. (15) in this issue is of particular interest. In the Actos Now for the prevention of diabetes (ACT NOW) study (10), which included 602 high-risk IGT patients, PIO reduced the progression to T2D over 2.4 years from an annual incidence of 7.6% in the placebo (PLAC) group by 72% (95% CI 51–84%); 48% of the PIO group reverted to normal glucose tolerance (NGT) compared with 28% in the PLAC group (P < 0.001). In the current report, 441 (73%) of these subjects had oral glucose tolerance tests at baseline and at study completion, with sampling of plasma glucose, insulin, and C-peptide. The protocol permitted calculation of insulin sensitivity by the Matsuda Index (16) as well as insulin secretion rate using plasma insulin and deconvolution of plasma C-peptide (17). Insulin sensitivity increased 92% in the PIO group compared with 17% in the PLAC group (P = 0.002), whereas insulin secretion rate increased 33% in the PLAC group compared with 11% in the PIO group (P < 0.001). The Disposition Index (insulin secretion rate/insulin resistance) increased 66% with PIO compared with 12% with PLAC (P < 0.001). The focus of DeFronzo et al., however, was on the relative changes from baseline of these derived parameters of glucose metabolism. Subjects were grouped into three categories according to their follow-up oral glucose tolerance tests: progression to T2D (N = 15 PIO and 45 PLAC), no change in IGT status, and reversion to NGT. In subjects who reverted to NGT, insulin sensitivity was increased 96% with PIO compared with PLAC, whereas insulin sensitivity changed little in those who progressed to T2D in either group. Insulin secretion in response to glucose declined with PIO and PLAC in those who progressed to T2D. Of importance, the Disposition Index fell slightly and similarly in both these groups.
In an analytical tour de force, a plot of incidence of T2D in the combined PIO and PLAC groups was negatively associated with octiles of the decrease in the natural logarithm of the Disposition Index with an extraordinary correlation coefficient of 0.99. The use of the somewhat unusual octiles reflected a curvilinear association between the variables. While this exceedingly strong association was driven by an outlier value at the lowest octile of the Disposition Index that corresponded to the highest incidence of diabetes, the remaining 7 points yielded a visually convincing but much more shallow curvilinear association. IGT subjects who experienced a >80% improvement in the Disposition Index had an approximate 2% incidence of diabetes whereas the incidence was 14% in those whose Disposition Index declined 60–80%.
Better final glucose tolerance status (NGT > IGT > T2D) was categorically associated with increase in insulin sensitivity (odds ratio [OR] = 0.61), insulin secretion (OR = 0.61), and Disposition Index (OR = 0.26). In multivariate analyses of factors associated with end of study outcome, NGT and IGT data were combined and compared with T2D. In the PIO group, the combined favorable outcome was associated with a 21% increase in insulin secretion rate, and a 70% increase in insulin secretion in response to glucose. There was a 109% increase in insulin sensitivity comparing only the group who reverted from IGT to NGT to the group that did not.
In assessing all of these impressive results, several points should be considered. The authors strongly assert that the overall conclusions indicate that improvement in β-cell function, i.e., the Disposition Index, is the dominant factor associated with final glucose tolerance status, and that the beneficial effect of PIO on reduced progression to T2D or increased reversion to NGT is largely attributable to the thiazolidinedione effect on this factor (OR = 0.26). This conclusion is consistent with most previous reports showing that insulin resistance is a factor in the earlier stages of IGT (8,18) whereas deteriorating insulin secretion in response to glucose is a later determinant of worsening glucose tolerance and severity of T2D (19). Hence, the decrease in conversion rate of IGT to T2D reflects less deterioration of β-cell function in the PIO group. However, the Disposition Index incorporates both of these functional parameters of glucose metabolism, and increases in both insulin sensitivity and insulin secretion were individually associated with the final impact of PIO therapy.
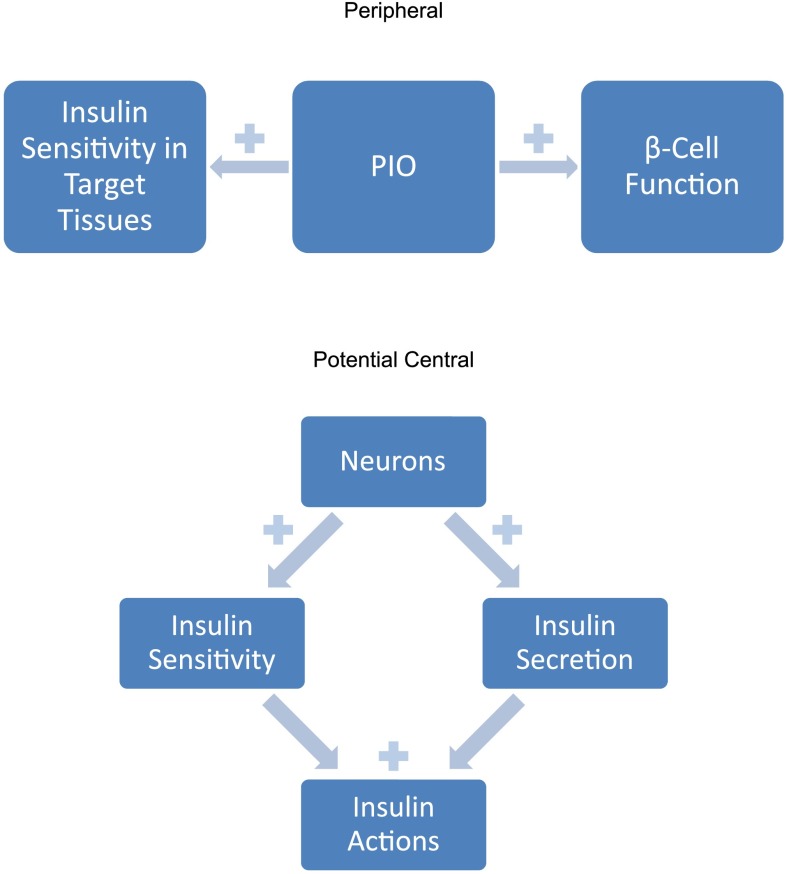
This should not be surprising. Although the temporal sequence of insulin resistance and diminished β-cell responsiveness cited above is widely accepted and appears logical, this does not mean that they are independent events. Several animal models support a homeostatic relationship between insulin sensitivity and insulin secretion (Fig. 1). Decreased sensitivity to insulin elicits production, especially by the liver, of betatrophin, a molecule that stimulates proliferation of β-cells, expansion of β-cell mass, and improvement in glucose tolerance in experimental animals (20). Moreover, insulin resistance—specifically at the level of the β-cell—impairs first phase insulin secretion in response to glucose (21).
FIG. 1.
Established and speculative beneficial effects of PIO.
Thus, there are two ways to interpret PIO’s effects on the Disposition Index and its individual components (Fig. 1). The most straightforward is that PIO is a thiazolidinedione and has positive actions on both insulin sensitivity in its target tissues as well as on β-cell function (22). Plasma glucose homeostasis is thereby maintained at an acceptable level because PIO only rarely causes hypoglycemia. A second speculative construct is to posit a brain regulating center with overall control of total body insulin action (Fig. 1). When there is need for more total body insulin action on all its target tissues, in addition to its effects on glucose metabolism, this center would simultaneously increase insulin sensitivity and insulin secretion. Of course, there would be need for a negative feedback plasma/intracellular glucose brake on the output of such neurons to prevent hypoglycemia. PIO has been shown to affect glucose metabolism in brain cells (23). The possibility that the ultimate seat of T2D could lie in the brain has also been suggested before (24), given the importance of insulin in regulating energy expenditure and storage through central as well as peripheral effects. PIO could presumably act by stimulating the output of such putative neurons. These two alternative mechanisms of PIO action, peripheral and central, need not be mutually exclusive.
These speculative ruminations aside, the main importance of the observations by DeFronzo et al. may be to encourage further development of T2D treatments that have ameliorative effects on glucose dysmetabolism, but less severe and less frequent adverse effects on the risks of congestive heart failure (25), fractures (26), and possibly bladder cancer (27). Their observations on the natural history of progression or reversion of IGT could also be applied to modification or development of other therapeutic agents. From bedside to bench and back to bedside should be a productive therapeutic path to follow.
ACKNOWLEDGMENTS
No potential conflicts of interest relevant to this article were reported.
Footnotes
See accompanying original article, p. 3920.
REFERENCES
- 1.Laakso M. Hyperglycemia and cardiovascular disease in type 2 diabetes. Diabetes 1999;48:937–942 [DOI] [PubMed] [Google Scholar]
- 2.Wilson PW. Diabetes mellitus and coronary heart disease. Endocrinol Metab Clin North Am 2001;30:857–881 [DOI] [PubMed] [Google Scholar]
- 3.Stamler J, Vaccaro O, Neaton JD, Wentworth D. Diabetes, other risk factors, and 12-yr cardiovascular mortality for men screened in the Multiple Risk Factor Intervention Trial. Diabetes Care 1993;16:434–444 [DOI] [PubMed] [Google Scholar]
- 4.Khaw KT, Wareham N, Bingham S, Luben R, Welch A, Day N. Association of hemoglobin A1c with cardiovascular disease and mortality in adults: the European prospective investigation into cancer in Norfolk. Ann Intern Med 2004;141:413–420 [DOI] [PubMed] [Google Scholar]
- 5.Ford ES, Zhao G, Li C. Pre-diabetes and the risk for cardiovascular disease: a systematic review of the evidence. J Am Coll Cardiol 2010;55:1310–1317 [DOI] [PubMed] [Google Scholar]
- 6.Edelstein SL, Knowler WC, Bain RP, et al. Predictors of progression from impaired glucose tolerance to NIDDM: an analysis of six prospective studies. Diabetes 1997;46:701–710 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gerstein HC, Santaguida P, Raina P, et al. Annual incidence and relative risk of diabetes in people with various categories of dysglycemia: a systematic overview and meta-analysis of prospective studies. Diabetes Res Clin Pract 2007;78:305–312 [DOI] [PubMed] [Google Scholar]
- 8.Kahn SE. The relative contributions of insulin resistance and beta-cell dysfunction to the pathophysiology of type 2 diabetes. Diabetologia 2003;46:3–19 [DOI] [PubMed] [Google Scholar]
- 9.Knowler WC, Barrett-Connor E, Fowler SE, et al. Diabetes Prevention Program Research Group Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 2002;346:393–403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.DeFronzo RA, Tripathy D, Schwenke DC, et al. ACT NOW Study Pioglitazone for diabetes prevention in impaired glucose tolerance. N Engl J Med 2011;364:1104–1115 [DOI] [PubMed] [Google Scholar]
- 11.Gerstein HC, Yusuf S, Bosch J, et al. DREAM (Diabetes REduction Assessment with ramipril and rosiglitazone Medication) Trial Investigators Effect of rosiglitazone on the frequency of diabetes in patients with impaired glucose tolerance or impaired fasting glucose: a randomised controlled trial. Lancet 2006;368:1096–1105 [DOI] [PubMed] [Google Scholar]
- 12.Chiasson JL, Josse RG, Gomis R, Hanefeld M, Karasik A, Laakso M, STOP-NIDDM Trial Research Group Acarbose for prevention of type 2 diabetes mellitus: the STOP-NIDDM randomised trial. Lancet 2002;359:2072–2077 [DOI] [PubMed] [Google Scholar]
- 13.Eriksson JG, Lehtovirta M, Ehrnström B, Salmela S, Groop L. Long-term beneficial effects of glipizide treatment on glucose tolerance in subjects with impaired glucose tolerance. J Intern Med 2006;259:553–560 [DOI] [PubMed] [Google Scholar]
- 14.Bolli GB, Consoli A, Giaccari A. Early insulin treatment in type 2 diabetes: ORIGINal sin or valuable choice as ORIGINal treatment? An open debate on the ORIGIN study results. Nutr Metab Cardiovasc Dis 2012;22:1007–1012 [DOI] [PubMed] [Google Scholar]
- 15.DeFronzo RA, Tripathy D, Schwenke DC, et al.; for the ACT NOW Study. Prevention of diabetes with pioglitazone in ACT NOW: physiologic correlates. Diabetes 2013;62:3920–3926 [DOI] [PMC free article] [PubMed]
- 16.Matsuda M, DeFronzo RA. Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care 1999;22:1462–1470 [DOI] [PubMed] [Google Scholar]
- 17.Van Cauter E, Mestrez F, Sturis J, Polonsky KS. Estimation of insulin secretion rates from C-peptide levels. Comparison of individual and standard kinetic parameters for C-peptide clearance. Diabetes 1992;41:368–377 [DOI] [PubMed] [Google Scholar]
- 18.Weyer C, Bogardus C, Mott DM, Pratley RE. The natural history of insulin secretory dysfunction and insulin resistance in the pathogenesis of type 2 diabetes mellitus. J Clin Invest 1999;104:787–794 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Turner RC, Cull CA, Frighi V, Holman RR, UK Prospective Diabetes Study (UKPDS) Group Glycemic control with diet, sulfonylurea, metformin, or insulin in patients with type 2 diabetes mellitus: progressive requirement for multiple therapies (UKPDS 49). JAMA 1999;281:2005–2012 [DOI] [PubMed] [Google Scholar]
- 20.Yi P, Park JS, Melton DA. Betatrophin: a hormone that controls pancreatic β cell proliferation. Cell 2013;153:747–758 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 21.Kulkarni RN, Brüning JC, Winnay JN, Postic C, Magnuson MA, Kahn CR. Tissue-specific knockout of the insulin receptor in pancreatic beta cells creates an insulin secretory defect similar to that in type 2 diabetes. Cell 1999;96:329–339 [DOI] [PubMed] [Google Scholar]
- 22.Yki-Jarvinen H. Drug therapythiazolidinediones N Engl J Med 2004;351:1106–1118 [DOI] [PubMed] [Google Scholar]
- 23.Dello Russo C, Gavrilyuk V, Weinberg G, et al. Peroxisome proliferator-activated receptor gamma thiazolidinedione agonists increase glucose metabolism in astrocytes. J Biol Chem 2003;278:5828–5836 [DOI] [PubMed] [Google Scholar]
- 24.Porte D, Jr, Seeley RJ, Woods SC, Baskin DG, Figlewicz DP, Schwartz MW. Obesity, diabetes and the central nervous system. Diabetologia 1998;41:863–881 [DOI] [PubMed] [Google Scholar]
- 25.Erdmann E, Charbonnel B, Wilcox RG, et al. PROactive investigators Pioglitazone use and heart failure in patients with type 2 diabetes and preexisting cardiovascular disease: data from the PROactive study (PROactive 08). Diabetes Care 2007;30:2773–2778 [DOI] [PubMed] [Google Scholar]
- 26.Dormandy J, Bhattacharya M, van Troostenburg de Bruyn AR, PROactive investigators Safety and tolerability of pioglitazone in high-risk patients with type 2 diabetes: an overview of data from PROactive. Drug Saf 2009;32:187–202 [DOI] [PubMed] [Google Scholar]
- 27.Lewis JD, Ferrara A, Peng T, et al. Risk of bladder cancer among diabetic patients treated with pioglitazone: interim report of a longitudinal cohort study. Diabetes Care 2011;34:916–922 [DOI] [PMC free article] [PubMed] [Google Scholar]