Abstract
The host response to infection is known to be influenced by many factors, including genetics, nutritional status, age, as well as drug and chemical exposures. Recent advances reveal that the aryl hydrocarbon receptor (AhR) modulates aspects of the innate and adaptive immune response to viral, bacterial, and parasitic organisms. Although many of these observations were made using the high affinity but poorly metabolized AhR agonist TCDD, not all of effects are detrimental to the host. Sometimes AhR activation, even with TCDD, was beneficial and improved host resistance and survival. A similar dichotomy is observed in infected AhR-deficient mice, wherein absence of functional AhR sometimes, but not always, alters host resistance. When examined in their totality, current data indicate that AhR controls multiple regulatory pathways that converge with infection-associated signals, and depending on the context (e.g., type of pathogen, site of infection) lead to distinct outcomes. This creates numerous exciting opportunities to harness the immunomodulatory action of AhR to transform host responses to infection. Moreover, since many of the mechanisms cued in response to infectious agents are pivotal in the context of other diseases, there is much to be learned about AhR's cellular targets and molecular mechanisms of action.
Keywords: AhR, immunity, host-pathogen interaction, virus, bacteria
Recent assessments of the global disease burden of infectious disease reveal a complicated scenario, in which some infectious diseases are in decline, primarily due to vaccination, whereas others are on the rise, and new ones have emerged [1, 2]. For example, over two dozen previously unknown, and currently incurable, infectious diseases have arisen in the past three decades, including HIV/AIDS, Ebola virus, Hepatitis C Virus, and severe acute respiratory syndrome (SARS). Other diseases, such as malaria and tuberculosis, persist despite many efforts to reduce their burden. Influenza viruses, which reassort across species bringing new pandemic strains, continue to exact a hefty toll on human health. Indeed, despite major improvements in health care and quality of life, lower respiratory tract infections remain particularly intransigent, and persist among top five leading causes of death worldwide [1, 3].
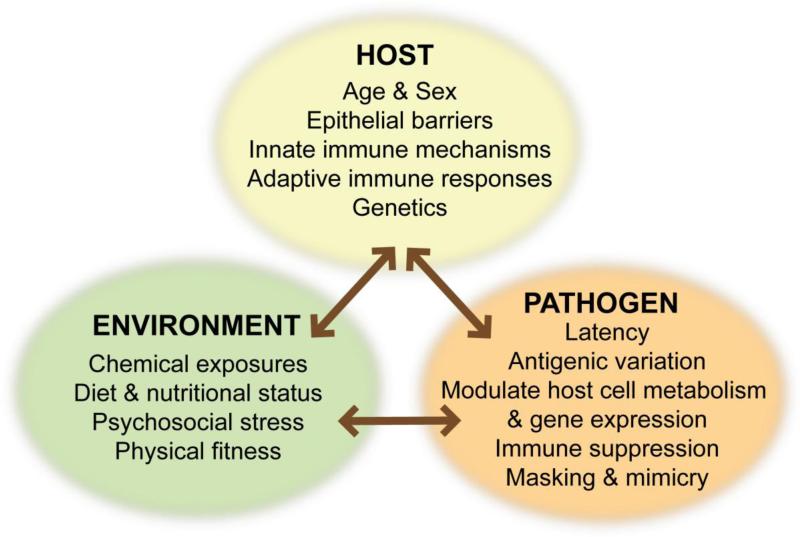
A central tenet of host-pathogen interactions is that, at the individual level, clinical outcome depends on the net result of two forces: resistance to damage caused by the pathogen, and resistance to damage caused by the immune response invoked to eliminate the pathogen. Differences in host gender, age, genetics, and nutritional status can explain some, but not all, differences in susceptibility to and pathophysiological consequences of infection. This leaves unresolved the identity of the other factors that contribute to differential clinical outcomes from infections in individuals and among populations. Recent epidemiology and animal studies suggest that exposure to chemicals from the environment are overlooked but likely very important contributors to host responses to infectious diseases [4-6] (Figure 1). The focus of this review will be on the current evidence that signaling through the aryl hydrocarbon receptor (AhR) has an important influence on host responses to infection, and will provide several recent examples of different immunoregulatory pathways affected by AhR in this context.
Figure 1. Host-Environment-Pathogen Interactions.
Differences in host genetics, age, and sex have long been known to influence outcomes following infection. Likewise, pathogen-specific mechanisms have evolved to evade host defenses, and continue to gain appreciation. Fewer studies have considered the impact of environmental differences on the host or the pathogen. Yet, when examined, emerging data provide compelling evidence that extrinsic factors, such as exposure to chemicals, psychosocial stress, and dietary constituents have a profound influence.
The AhR is an environment-sensing, inducible transcriptional regulator that has a diverse array of anthropogenic and naturally derived ligands [7-9]. While consensus regarding the identity of bona fide endogenous ligands has yet to be reached, it is evident from many studies that we are exposed to a diverse spectrum of AhR ligands in our daily lives. Exposure to AhR ligands is primarily via the food chain, but also occurs through inhalation and absorption across mucosal barriers, and the in vivo metabolism of certain compounds into AhR ligands [8-11]. Several years ago, there was an opinion that studies of common AhR binding pollutants, such as dioxins, polychlorinated biphenyls (PCBs) and polyaromatic hydrocarbons (PAHs), were obsolete. That is, since environmental levels of these toxicants were going down in the most economically developed countries, there was no compelling reason to study them further. However, contemporary environmental assessments indicate that while levels of these chemicals have plateaued and may even be declining in some affluent countries, they are on the rise in the developing world [12-15]. Moreover, contemporary epidemiology studies show that dioxin and PCBs levels are significantly and directly associated with more respiratory tract infections, increased incidence of wheezing, and poorer vaccine responses in infants and children [16-21]. Therefore, these AhR ligands remain a continued threat to public health. Moreover, the recent discovery that some pharmaceuticals, such as leflunomide and omeprazole, are AhR ligands further emphasizes the need to elucidate how AhR signaling alters critical innate and adaptive host responses during infection [22, 23].
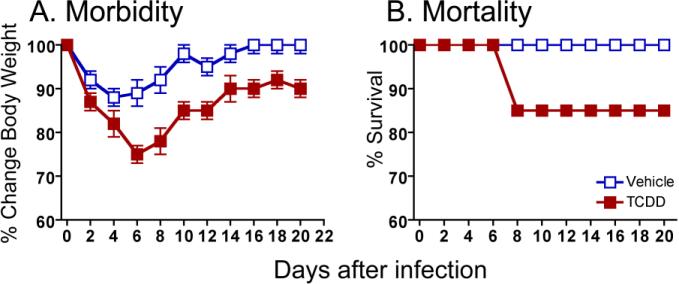
Although the AhR's role in normal physiology remains elusive, it is evident from numerous studies that it profoundly influences the function of the immune system [24]. Most recently, the AhR's remarkable capacity to modulate autoimmune diseases [25-29], allergic inflammation [30], and inflammatory bowel disease [31-33] has catalyzed new research to discover AhR ligands that can be used to treat these diseases. This is clearly an exciting line of inquiry. However, there is another facet of AhR immunobiology that has direct relevance to human health: the impact of AhR activation on host responses to infection. The first evidence that AhR activation influences host resistance to infection was reported over 30 years ago [34-37]. One of the most striking observations among these initial studies was that exposure to very low doses of the AhR binding pollutant 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD or dioxin) enhanced morbidity and mortality in rats and mice infected with lethal strains of influenza A virus [36, 37]. Since then, others have reported similar observations with other strains of influenza A virus; in particular even ones that cause sub-lethal infection in the absence of AhR activation [38, 39]. In other words, and as shown in Figure 2, simply activating AhR is sufficient to alter host resistance to an otherwise sub-lethal influenza virus infection.
Figure 2. AhR activation is sufficient to alter the course of viral infection.
C57Bl/6 mice were treated with a single oral dose of peanut oil vehicle (open squares) or TCDD (10 μg/kg body weight, closed squares) one day before intranasal infection with 0.05 hemagglutinating units (HAU) of influenza virus (A/PR/8/34, H1N1). Body weight and survival were monitored daily for 14 days. Similar observations have been made using other influenza A virus strains (not shown).
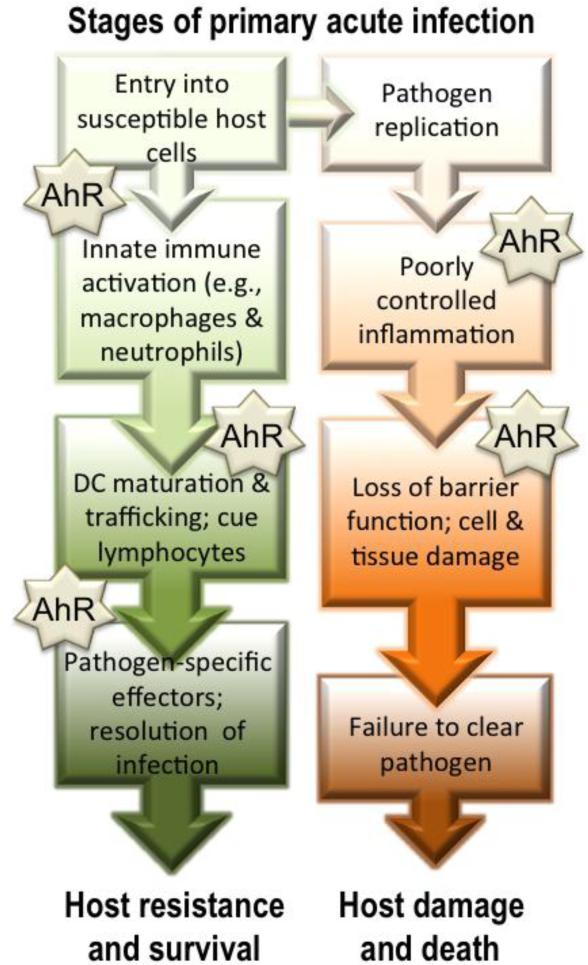
While several independent studies further support the idea that AhR plays an important role during infection, its precise function appears to vary with the type of pathogen and the target organ [39-46]. Furthermore, whether AhR signaling contributes to improved or worsened host resistance depends on the pathogen. For example, observations in AhR-deficient (AhR KO) mice show that they are less able to survive Citrobacter rodentium infection compared to wild-type mice; however, their ability to survive infection with Streptococcus pneumonaie is equivalent to wild-type controls [41, 44, 46]. AhR activation in wild-type rodent strains also shows different ‘directionality’ in host resistance. That is, there are examples of the same ligand increasing and diminishing morbidity and mortality to different pathogenic challenges [39, 41, 45]. These observations suggest the AhR has pathway-specific roles that modulate host defense mechanisms during infection, and also indicate there are multiple cellular targets of AhR ligands. However, the specific cell and gene targets of AhR that influence host responses to infection are not fully understood. Experimental evidence outlined in the following sections shows that the AhR affects multiple aspects of host responses to infection (Figure 3). However, it is worth noting that the majority of this evidence stems from studies conducted when AhR is activated using model high affinity ligands that are resistant to metabolism or elimination, or from observations in AhR-deficient mice. It remains to be determined what influence naturally derived AhR ligands or pharmaceuticals that also bind AhR will have on host responses to infectious agents. Yet, despite many unanswered questions, the studies to date reveal much about cellular and molecular targets of AhR, pointing to pathways that can be exploited in the future.
Figure 3. AhR modulates multiple aspects of host responses to infection.
Infection triggers an integrated series of responses by the host that seek to reduce pathogen proliferation, and ultimately eliminate invaders from the body. Host mechanisms include barrier cells and innate responses, such as promote inflammation, which contain and control infection while the adaptive immune system is activated. If an insufficient response is initiated or the response is not sustained, then complete pathogen elimination may not be achieved. Similarly, if the activated effector mechanisms are poorly controlled, then excessive damage to host tissue, and sometimes death, can ensue. AhR, either through its absence or activation by exogenous ligands, has been shown to influence multiple stages of the host response to acute primary infection.
Viral infections
The majority of information regarding how AhR modulates the immune response to virus infection stems from studies using mouse models of infection with human influenza A viruses. In the respiratory tract, influenza viruses initiate innate responses in epithelial cells, which recruit monocytes/macrophages and neutrophils to the lung. Using a variety of mechanisms, these innate mediators destroy infected host cells and release inflammatory and chemoattractive mediators that stimulate additional leukocyte recruitment to the infected lung [47, 48]. Respiratory dendritic cells (DCs) are another cell type activated very early during infection. DCs become infected or take up viral antigens, migrate to the draining lymph nodes, and present viral peptides to T cells [49]. This is important because the successful clearance of influenza virus ultimately depends on the adaptive immune system; specifically CD8+ T cells, CD4+ T cells, and B cells [50]. Host resistance during a primary influenza virus infection is largely mediated by virus-specific CD8+ cytotoxic T lymphocytes (CTL), which are activated in lymphoid tissues and then traffic to the lung and kill infected cells [50]. B cells produce virus-specific antibodies, which provide critical defenses from repeated infection with homotypic virus strains, but are not required for fighting primary infection [51]. Influenza virus-specific conventional CD4+ T cells (e.g., Th1 cells) are important for fine-tuning the production of virus-specific antibodies by B cells and promoting successful memory responses [51, 52]. Newly emerging data suggest roles for Foxp3+CD25+ regulatory CD4+ T cells (Tregs), Th17, and T follicular helper (TFH) cells during virus infection, but the specific functions of these lineages during influenza virus infection are not yet fully appreciated [53-57]. Upon successful viral clearance, a small pool of memory lymphocytes remains, providing protective immunity when the same or similar influenza viruses are encountered in the future. Proper regulation of all these pathways is required for surviving the primary infection. Uncontrolled stimulation of inflammatory responses can lead to excessive damage of healthy tissue, whereas insufficient immune activation could result in prolonged or more extensive viral replication.
As noted earlier, treatment with a single oral dose of TCDD increased morbidity and mortality in mice infected with influenza A virus. Studies to delineate how AhR activation alters host resistance to influenza virus reveal numerous changes in innate and adaptive responses to infection. For example, the adaptive immune response is severely suppressed upon AhR activation, with a striking reduction in the expansion and differentiation of conventional CD4+ and CD8+ T cells, lower virus-specific IgG titers, and diminished DC function [39, 58-60]. However, despite this profound suppression, there is no marked increase in the pulmonary viral burden of the survivors. That is, although AhR activation increases morbidity and mortality following influenza virus infection, the kinetics of viral growth and clearance is not significantly different between control and TCDD-treated mice [37, 38, 61]. Initially these data were puzzling, and made it difficult to explain the poorer survival. However, recent studies reveal that although components of the adaptive immune response are diminished, AhR activation enhances certain inflammatory responses in the infected lung. Specifically, AhR activation increases recruitment of neutrophils to the infected lung, and elevates pulmonary IFNγ and iNOS levels during infection [40, 62, 63], all of which may provide compensatory antiviral control mechanisms. However, these enhanced responses likely contribute, directly or indirectly, to more severe immunopathology and poorer survival. For instance, in vivo depletion of neutrophils markedly improves survival and reduces the enhanced bronchopulmonary inflammation observed when AhR is activated during acute influenza virus infection [40, 63].
Studies using mutant mice that express AhR proteins that either lack the nuclear translocation signal or DNA binding domain support that changes in the host response to influenza virus occur via a mechanism that involves AhR nuclear translocation and direct interaction with DNA via AhR response elements (AhRE), as opposed to cross-talk with other signaling molecules [60, 63]. However the specific gene targets of AhR that alter host responses to influenza virus remain to be determined. One barrier to identifying the AhR target genes directly responsible for modulating host responses to infection is that the cell types whose function is altered directly by cell intrinsic AhR mechanisms, versus indirectly by AhR activation in another cell type, are only now being defined. Using a combination of adoptive transfers, bone marrow transplantation, and conditional gene ablation, recent data reveal that AhR influences innate and adaptive immune responses to influenza virus through an amalgamation of AhR-mediated events within hematopoietic cells, endothelial cells, and lung epithelial cells [42, 62-64]. That is, impaired responses of virus-specific CD8+ T cells require that the AhR be present in bone marrow-derived cells, but not within CD8+ T cells themselves. This suggests AhR signaling in other immune cells drive the negative regulation of CD8+ T cell responses to influenza virus [42]. On the other hand, increased pulmonary neutrophilia and elevated iNOS and IFNγ levels in the lung do not require AhR in the hematopoietic compartment [62, 64]. Instead, studies with conditional AhR KO mice reveal that the increase in pulmonary neutrophilia requires AhR activation within the respiratory epithelium, and iNOS enhancement occurs through AhR activation in endothelial cells [63]. Thus, AhR-mediated alterations in different aspects of the host response to the same pathogen involve separate events in distinct tissue compartments. Moreover, not all changes in host responses to infection are due to AhR-driven signaling in hematopoietic cells.
The consequences of AhR activation during infection with other types of viruses have not been extensively characterized in vivo. Yet there is evidence that AhR activation affects host responses to other viruses [45, 65]. In mice infected with coxsackievirus B3, treatment with TCDD exacerbated mortality [65]. Similar to influenza virus, decreased host resistance did not correlate with increased viral burden; however, the underlying host mechanisms affected are not yet elucidated. Also, in an interesting twist on the way we think about these things, infection with coxsackievirus virus altered TCDD absorption in mice. This suggests that a broader consideration of host-pathogen-environment interactions should include exploration of the way in which infectious agents influence host metabolism and signaling pathways [66].
A more recent study shows that during ocular infection with herpes simplex virus (HSV), mice treated with TCDD exhibited reduced effector Th1 and Th17 cells, reduced neutrophilic inflammation, and an increase in the proportion of Foxp3+ Tregs [45]. Ocular HSV infection can result in a chronic immune-mediated inflammation of the cornea, that is thought to be driven by conventional CD4+ T cells and neutrophilia, and which ultimately leads to blindness. In this HSV model, TCDD-treated mice had higher virus titers, and many succumbed to herpes encephalitis if AhR was activated prior to infection. However, when AhR activation was initiated after HSV infection, herpes encephalitis was reduced and there was improved pathology in the eye tissue. Given that the pathology-inducing responses were reduced upon AhR activation, it suggests that AhR signaling is a potential new avenue for treatment options for this immune-mediated disease.
Bacterial Infections
Current knowledge of AhR activation and host responses to bacterial infection presents a somewhat mixed picture, depending on pathogen and model system examined. Streptococcus pneumoniae is a common commensal bacterium of the upper respiratory tract, but causes disease when it breaches protective boundaries and invades the lung, or penetrates into the systemic circulation. Using a model of intranasal infection with S. pneumoniae, AhR activation reduces bacterial invasion of the lung, and has a beneficial effect on host survival [41, 67]. More specifically, if administered prior to infection, TCDD treatment reduced or even prevented expansion of bacteria in the lung, and ultimately doubled the percentage of mice that survived an otherwise lethal infection. These protective effects against bacterial colonization were detectable within mere hours of infection, but were not associated with any enhanced activity or frequency of innate immune cells [67]. In fact, there was no concomitant increase in the number of phagocytic cells or inflammatory mediators that would explain bacterial clearance. Importantly, this protective effect was shown to be AhR-dependent in the host, and the AhR agonist used, TCDD, was not directly bactericidal or bacteriostatic for S. pneumoniae. Moreover, this improved outcome was highly dependent upon the timing of AhR activation relative to infection; survival was only improved when TCDD was administered before exposure to the bacteria. While the mechanism of host-protection in this model remains elusive, it is likely that AhR activation within lung parenchymal cells creates an inhospitable environment for this pathogen, perhaps by stimulating production of antimicrobial substances or down-regulating lung cell surface proteins the bacteria use to attach and invade the tissue.
In contrast to protective effects of AhR activation in the intranasal infection model, TCDD treatment had a profoundly negative impact on host survival when S. pneumoniae were administered systemically [68]. This supports the idea that AhR-mediated outcomes during infection are tissue compartment-dependent. However, different mouse and S. pneumoniae strains were used in the intranasal versus systemic infection models; therefore, host or pathogen-related differences could also contribute to different sensitivities. Interpretation of studies using another bacterium, Listeria monocytogenes, is also complicated by the different outcomes observed under different study designs. Depending on mouse strain and sex, as well as the administered route and dose of TCDD, animals were either more susceptible, unaffected, or potentially even protected against challenge with L. monocytogenes [34-36]. Thus, while AhR activation clearly influences aspects of the host response to bacterial infection, the outcome varies with pathogen and infection route, and no common set of pathways affected or central mechanism has yet to be elucidated.
Parasitic infections
Only a handful of studies have examined whether activation of AhR signaling pathways is involved in host responses to parasitic infections. While a common mechanism has yet to be identified, these reports consistently support the idea that AhR modulates immunoregulatory mechanisms involved in host defenses against parasitic protozoa and nematodes. For instance, in separate studies in rats and mice, treatment with TCDD impaired clearance of Trichinella spiralis, a common parasitic nematode that infects humans and other mammals [69, 70]. These are interesting observations because elimination of T. spiralis is antibody-mediated, and these data suggest that AhR engagement by TCDD impairs naïve lymphocyte activation leading to a reduced or less effective humoral response against parasitic worms.
Leishmania major is a parasitic protozoan that in humans causes leishmaniasis, a disease that affects at least 12 million people worldwide. Mouse models of L. major infection reveal distinctly skewed CD4+ T cell responses that depend on the mouse strain (e.g., BALB/c are Th2 prone, whereas a Th1 biased response to L. major is observed in C57Bl/6 mice). Using C57Bl/6 mice, AhR activation with TCDD reduced several aspects of the adaptive response to L. major infection, including suppressed lymphocyte numbers and antibody production. This is not unexpected as AhR activation typically suppresses conventional CD4+ T cell responses to antigen challenge. However, the authors were surprised to discover that AhR activation also decreased the parasite burden by 10- to greater than 100-fold [71]. The underlying reason for reduced parasite load has yet to be determined, but recent discoveries about AhR and other CD4+ T cell subsets provides a testable explanation for this (refer other articles within this issue here). As discussed in the following section, further ideas about a possible role for AhR signaling in host responses to parasitic infections are suggested from studies of AhR KO mice.
Insights from AhR KO mice
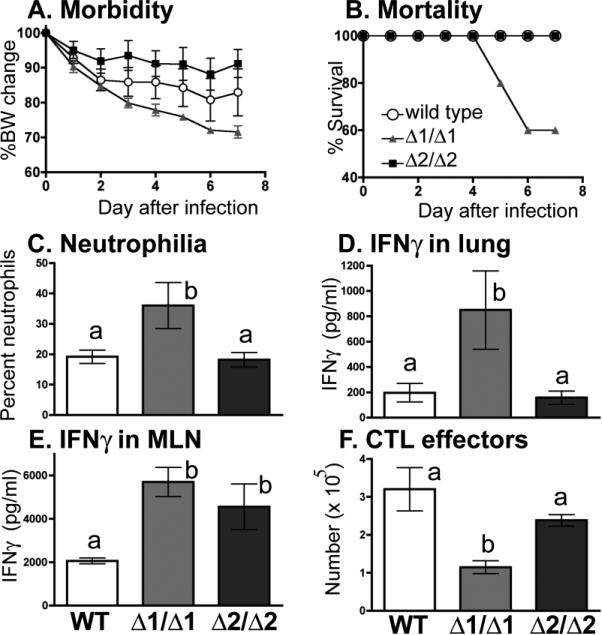
A classic tool for probing the role of a particular protein in complex biology is to remove it, such as by selectively preventing expression or translation of the gene that encodes it. When initially created, some modest changes in immune organ cellularity were reported in AhR KO mice [72, 73], but there were no patent deficits in the immune response to model antigens [74]. However, more recent studies have revealed that loss of AhR does affect some immune endpoints, and raise several issues that need to be considered when interpreting data using AhR KO mice. Using two independently generated AhR KO mouse strains, we compared several aspects of the immune response to influenza A virus (Figure 4). These mouse strains were created by either replacing exon 1 (Δ1/Δ1) or exon 2 (Δ2/Δ2) of the Ahr locus [72, 73], [75]. For this comparison, we deliberately selected an influenza infection that is generally sub-lethal in immunocompetent mice, and compared the response of wild type, Δ1/Δ1, and Δ2/Δ2 mice simultaneously. The difference in host resistance between the two KO strains was striking, as the Δ1/Δ1 mice were unable to survive the infection, and were all dead by the 8th day. In contrast, host resistance in the Δ2/Δ2 mice was comparable to WT, with no mortality observed using this virus strain. Further comparison of specific immunologic endpoints, measured in surviving mice on the 7th day after infection, revealed that Δ2/Δ2 and wild-type mice were generally similar. However, the infected Δ1/Δ1 mice had a significantly greater frequency of pulmonary neutrophils, higher IFNγ levels in the lung, and fewer CD8+ CTLs in their mediastinal lymph nodes. Interestingly, the amount of IFNγ produced by lymph node cells was similar in both AhR KO strains, and was significantly higher than IFNγ produced by cells from infected wild-type mice. Taken together, these results underscore that there are inherent differences in the response of these to AhR KO strains to infection with the same pathogen.
Figure 4. Two AhR KO mouse strains differ in their response to influenza virus infection.
Mice (8-10 weeks of age) were infected with influenza virus (A/HKx31 (H3N2), 120 HAU, intranasally). A,B. Body weight (BW) change and survival were monitored daily. Open circles, wild-type C57Bl/6 mice; grey triangles, B6.129-Ahrtm1Gonz (Δ1/Δ1) mice; and black squares, B6.129-Ahrtm1Bra/J (Δ2/Δ2) mice. C-F. Remaining mice were sacrificed 7 days after infection. C. Bars represent the average percentage of neutrophils in the lung lavage fluid. The number of neutrophils was also significantly higher in airways of Δ1/Δ1 mice (data not shown). D,E. The mean IFNγ concentration in clarified lung lavage fluid (D), and produced by ex vivo re-stimulated mediastinal lymph node cells (E) was determined by ELISA [42, 62]. F. The number of CD8+ T cells in the MLN with an effector phenotype (CD44hiCD62Llo) was determined by flow cytometry [42]. Both AhR KO strains are on a C57Bl/6 genetic background. Error bars depict the SEM. Bars that share the same letter were not significantly different (p ≤ 0.05).
While we are unaware of other studies that have directly compared host resistance in both AhR KO strains, it is clear that the Δ2/Δ2 strain is not universally robust and comparable to wild type mice. For instance, Δ2/Δ2 mice are less able to cope effectively with Listeria monocytogenes [43] than WT animals, and both Δ2/Δ2 and Δ1/Δ1 are more susceptible to Citrobacter rodentium infections than WT [44, 46, 76]. Other host-resistance studies conducted in AhR KO mice reveal no apparent defect in host resistance to Streptococcus pneumoniae infection (Δ2/Δ2 strain) [41], further emphasizing that there are both mouse strain-dependent and pathogen-dependent variability, which may influence host outcome. Given that L. monocytogenes and S. pneumoniae are gram-positive bacteria, whereas C. rodentium are gram negative, it is unlikely simple differences in the outer wall explain these conflicting observations. Abnormalities in the development of intraepithelial lymphocytes in the gastrointestinal tract may explain reduced host resistance of AhR-deficient mice to C. rodentium, as this pathogen primarily colonizes the colon [44, 46, 76].
L. monocytogenes also replicates in the gastrointestinal tract, causing listeriosis, which is a major source of foodborne illness. Although disease is self-limiting in most people, vulnerable populations, such as pregnant women and patients receiving immunosuppressive therapies, are at higher risk for illness and disease. The host immune response to L. monocytogenes is quite well understood; however, what specific aspects of it are controlled or modulated by AhR remain to be identified. Following infection with L. monocytogenes, the Δ2/Δ2 AhR KO strain has higher bacterial titers in liver, spleen and blood, delayed pathogen clearance, and more severe lesions in the livers compared to AhR+/− and wild type mice [43]. However, for the endpoints measured, the AhR-deficient mice exhibited cytokine levels and adaptive responses to infection that were not different from L. monocytogenes-infected wild-type mice or AhR+/− littermates. Furthermore, no differences in L. monocytogenes uptake by or growth in bone marrow or peritoneal macrophages were found when comparing cells from AhR+/− and the Δ2/Δ2 mice, leaving the reason for higher bacterial burdens in vivo unresolved. In a follow up study, in vitro treatment of mouse embryonic hepatocytes with the AhR agonist beta-naphthoflavone (BNF) dose-dependently inhibited uptake of L. monocytogenes, whereas AhR activation with TCDD did not have this same effect [77]. Findings using pharmacological inhibitors with BNF treatment suggest that the distinct effects of BNF compared to TCDD may be explained by differences in reactive oxygen species production.
Host responses of Δ1/Δ1 AhR KO mice to parasites have also been examined, both in the context of L. major and Toxoplasma gondii infections. Following L. major infection, they exhibited a response that was quite distinct from wild-type littermates [78]. Specifically, Δ1/Δ1 AhR KO mice exhibited a faster onset of inflammation and reduced number of parasite lesions 8 weeks later. Other aspects of the immune response to L. major were also different in Δ1/Δ1 versus wild type mice, such as higher levels of circulating TNFα and IL-12, a shift in the kinetics of infection-stimulated increases in IL-10, higher titers of anti-L. major antibodies, and a reduced frequency of Foxp3+ Tregs. Since L. major infects phagocytic cells, it would be interesting to know if the response of different types of phagocytic leukocytes is altered in AhR-deficient mice; however this has not yet been directly examined. Another common parasite, Toxoplasma gondii, is carried by millions of people worldwide. AhR KO mice succumbed faster, and showed more liver damage than wild-type mice following infection with T. gondii [79]. Poorer survival appears to reflect an alteration in the host's response to infection, with changes in IFNγ, IL-10, nitric oxide, and other mediators described in AhR KO versus wild-type mice, although more characterization of these changes are needed.
Indeed, there is much still to learn about the distinctions and similarities between the different AhR KO strains that have been created, including the underlying reason for the differential response to infection with the same pathogen. Yet, these data make it very clear that when examining the potential role of AhR in immune function using global AhR KO mice, it is critically important to consider which mouse strain is being used.
Immunological memory
Immunological memory is an important feature of the immune system that has not been extensively studied from the perspective of AhR immunobiology. Yet, this is the foundation of protective immunity to repeated infections by the same or closely related pathogens, and underlies the efficacy of vaccination. Given the important contribution of these recall responses to human health, it is rather surprising that so few studies have examined whether and how AhR signaling affects the acquisition or maintenance of immunological memory. This issue warrants further attention because several contemporary epidemiological studies reveal that infants and children whose mothers have higher levels of dioxins and PCBs have lower antibody responses to routine childhood vaccinations [16-19]. Consistent with this, administration of a single dose of TCDD before primary influenza virus infection of mice decreased the antibody response upon reinfection with the same strain of virus [80]. Similarly, AhR activation at the time of primary infection resulted in about half as many virus-specific memory CD8+ T cells 40-60 days later. Moreover, after re-infection, the expansion of the virus-specific CD8+ T cell population was delayed by several days [80]. Despite these changes in the magnitude and kinetics of the recall response, at least for the strain of influenza virus used in these studies, host resistance during secondary infection was not impaired [42, 80]. Additionally, when AhR was activated after immunological memory was established (i.e., ligand was administered only after the primary infection was fully resolved), then there were no detectable effects on the magnitude of the recall antibody or CD8+ T cell responses during secondary infection [42]. Thus, it is possible that AhR preferentially modulates events critical for the activation of naïve T cells and B cells [60, 81]. Yet, the potential for AhR to impact the generation, maintenance, or recall of memory immune responses has not been thoroughly probed, and cross protective acquired immunity has not been evaluated.
Interaction with pathogen-derived molecules
While AhR is highly conserved evolutionarily, with homologs in non-vertebrate species, there are no known AhR homologs in microorganisms. Yet, it is possible that proteins or other constituents derived from microorganisms interact with the AhR in the host. Another possibility is that AhR molecules the expression of pathogen genes. This idea may sound a bit far fetched; however, in the few studies in which this has been considered, the data are persuasive and suggest that this is a fertile area for future research. Epstein-Barr virus (EBV) provides a groundbreaking example. A relationship between EBV and exposure to environmental AhR ligands has been proposed as a risk factor for non-Hodgkin lymphoma and other diseases [82, 83]. In separate studies, associations between AhR and EBV genes and/or proteins have been reported. One such EBV-specific molecule is the nuclear protein EBNA-3, which contributes to the transformation of EBV-infected B cells [84]. EBNA-3 interacts directly with AhR, and also with the AhR chaperone protein XAP-2 [85, 86]. The precise nature of this interaction, and how it ultimately contributes to diseases such as non-Hodgkin lymphoma, remains to be elucidated. Some data suggest that EBNA-3 associates with AhR irrespective of whether an exogenous AhR ligand is present; however, EBNA-3 enhances TCDD-induced transcription of an AhR-driven reporter gene suggesting ligands activate or change this interaction [85]. The relationship between EBNA-3 and AhR might be via XAP2, which is thought to retain the AhR in the cytoplasm in the absence of exogenous ligand. Yet, this may not be the only way in which they interact because in the presence of EBNA-3 XAP2 translocates to the nucleus, which suggests that EBNA-3 influences AhR-regulated genes, perhaps by stabilizing transcriptionally active AhR in the nucleus.
More recently, another EBV gene has been suggested to be a novel direct AhR target [87]. The EBV gene BZLF1 encodes a gene product, Zta, which triggers the EBV lytic cycle. This is particularly important in latently infected cells, where the expression of BZLF1 re-initiates viral replication [88]. While AhR activation alone was not sufficient to switch EBV from latency to the lytic cycle, AhR activation combined with phorbol ester treatment reactivated EBV in B cells. Moreover, AhR appears to activate EBV genes in human salivary gland epithelial cells [87], although AhREs within EBV-encoded genes have yet to be identified. These reports collectively demonstrate that AhR interacts directly with viral proteins and may even directly induce the expression of viral genes.
Further evidence that AhR influences viral replication come from in vitro studies of various cell lines infected with other viruses, such as bovine herpes virus-1 (BHV-1), cytomegalovirus (CMV) and human immunodeficiency virus (HIV). AhR enhances replication of BHV-1 and up-regulates expression of a BHV-1 specific protein in MDCK cells [89]. Others have observed enhanced replication of latent CVM and CMV-related cytopathic effects in TCDD-treated human fibroblasts [90]. In other studies, in vitro treatment with TCDD, benzo[a]pyrene, and 3-methycholanthrene increased HIV gene expression and the level of secreted HIV p24 in several different cell lines [91-94]. Using mutant and dominant negative AhR constructs, this effect on HIV appears to be AhR-dependent [93]. Interactions between AhR and NF-κB have been implicated in some, but not all, of these studies [91-93], leaving the precise manner by which AhR impacts viral latency and viral replication uncertain. Another uncertainty is whether AhR ligands influence the replication of any of these viruses in vivo. One recent report [87] suggests that, at least for EBV, environmental AhR ligands may indeed contribute to virus reactivation and disease.
Finally, in an intriguing twist of this concept, a study of common probiotic, or commensal lactic acid bacteria found in many dietary supplements and foods, reveals that numerous strains release AhR ligands. In vivo tests using one strain, Lactobacillus bulgaricus OL1181, profoundly reduced pathology in a mouse model of acute, induced inflammation of the gastrointestinal tract [31]. While the factor from L. bulgaricus that activates AhR has yet to be identified, this intriguing study poses the idea that constituents from some bacteria activate the AhR. Thus, despite the fact that few reports have even considered interactions between AhR and pathogen-specific molecules, the studies that have been conducted to date provide compelling evidence for a convergence between AhR and pathogen-encoded factors. These interactions, if they occur in vivo, could profoundly alter cellular function and may serve to partially explain the complex, and at times even controversial, relationship between AhR ligand exposures and certain diseases
Conclusion
Collectively, these studies reveal that the AhR modulates responses to several different classes of common human viruses, including both RNA and DNA viruses, as well as to gram-negative and gram-positive bacterial species, parasitic protozoa, and even a pathogenic nematode. While the underlying cellular targets and precise molecular mechanisms have yet to be fully elucidated, it is clear that AhR profoundly influences host responses to infection, and the nature of the effect varies depending on the pathogen. When examined at the cellular level, AhR regulates both innate and adaptive immune responses; sometimes in opposite directions (e.g., enhancing inflammation but suppressing adaptive responses in the infected host). Moreover, the scope of cell types in which AhR signaling directly influences host responses, at least in the context of influenza virus, include immune cells, endothelial cells, and lung epithelial cells. This indicates that future mechanistic studies should not be limited to hematopoietic cells, but need to consider AhR regulation of events in cells of non-hematopoietic origin too.
The repercussions of AhR-driven changes in host responses to infection include the potential for environmentally derived AhR ligands to alter the balance between appropriate and excessive host defense mechanisms during infection. Given that many AhR ligands are common and abundant pollutants found around the globe, the influence of AhR signaling to the burden of infectious diseases requires considerably more attention. Another important area for future inquiry is to examine whether and how other AhR ligands including natural, food-derived compounds and pharmaceuticals, affect host responses to infection. By deciphering the cellular targets and molecular mechanisms by which AhR ligands, the good and the bad, alter host responses to infection, we will gain knowledge that will catalyze our ability to harness the power of this fascinating receptor to deliberately alter immune function to treat numerous diseases.
Major strides have been made in reducing the global burden of infectious diseases. Yet, despite improvements in medicine and public health, they remain a major source of illness. Advances in microbiology and immunology continue to expand our appreciation of the incredible complexity of immune responses to infectious agents, and the variety of ways pathogens attempt to circumvent these protective responses. We have also come to realize that poorly controlled inflammation, often initiated by infection, contributes to the progression or exacerbation of numerous chronic illnesses. We have much to learn about how AhR signaling networks modulate immune responses to infection, as well as how we can harness this fascinating receptor to improve disease outcomes and human health. Yet, the work summarized in this article reveals that AhR and its many ligands are overlooked but critically important modulators of innate and adaptive immune responses to infection.
Acknowledgements
On-going research is supported by the following grants from the National Institutes of Health: R01-ES017250 (BPL), R01-HL097141 (BPL), R15-AI82403 (BAV), T32-ES07026, and P30-ES01247. BPL is the recipient of an NIH Career Development Award (K02-ES012409).
Abbreviations
- AhR
aryl hydrocarbon receptor
- AhR deficient
AhR KO
- AhRE
AhR response element
- CMV
cytomegalovirus
- CTL
cytotoxic T lymphocyte
- EBNA-3
EBV nuclear antigen
- EBV
Epstein-Barr virus
- HAU
hemagglutinating unit
- iNOS
inducible nitric oxide synthase
- PAH
polyaromatic hydrocarbon
- PCB
polychlorinated biphenyl
- TCDD
2,3,7,8-tetrachlorodibenzo-p-dioxin
- Th17
IL-17 producing T helper cells
- Treg
CD4+ regulatory T cells
- XAP-2
X-associated protein 2
Footnotes
Conflict of Interest Statement
The authors declare that there are no conflicts of interest.
References
- 1.Lozano R, Naghavi M, Foreman K, Lim S, Shibuya K, Aboyans V, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2013;380:2095–2128. doi: 10.1016/S0140-6736(12)61728-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.World Health Organization The global burden of disease: 2004 update. 2008 http://www.who.int/healthinfo/global_burden_disease/2004_report_update/en/index.html.
- 3.World Health Organization Acute Respiratory Infections. 2005 url:www.who.int/vaccine_research/diseases/ari/en.
- 4.Colosio C, Birindelli S, Corsini E, Galli C, Maroni M. Low level exposure to chemicals and immune system. Toxicol Appl Pharmacol. 2005;207:S320–S328. doi: 10.1016/j.taap.2005.01.025. [DOI] [PubMed] [Google Scholar]
- 5.Singh V. The burden of pneumonia in children: An Asian perspective. Pediatr Resp Rev. 2005;6:88–93. doi: 10.1016/j.prrv.2005.03.002. [DOI] [PubMed] [Google Scholar]
- 6.Feingold B, Vegosen L, Davis M, Leibler J, Peterson A, Silbergeld EK. A niche for infectious disease in environmental health: Rethinking the toxicological paradigm. Environ Health Persp. 2010;118:1165–1172. doi: 10.1289/ehp.0901866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.McIntosh B, Hogenesch J, Bradfield CA. Mammalian Per-Arnt-Sim proteins in environmental adaptation. Annu Rev Physiol. 2010;72:625–645. doi: 10.1146/annurev-physiol-021909-135922. [DOI] [PubMed] [Google Scholar]
- 8.Denison M, Nagy S. Activation of the aryl hydrocarbon receptor by structurally diverse exogenous and endogenous chemicals. Annu Rev Phramacol Toxicol. 2003;43:309–334. doi: 10.1146/annurev.pharmtox.43.100901.135828. [DOI] [PubMed] [Google Scholar]
- 9.Nguyen LP, Bradfield CA. The search for endogenous activators of the aryl hydrocarbon receptor. Chem Res Toxicol. 2008;21:102–116. doi: 10.1021/tx7001965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Behnisch PA, Hosoe K, Sakai S. Combinatorial bio/chemical analysis of dioxin and dioxin-like compounds in waste recycling, feed/food, humans/wildlife and the environment. Environ Int. 2001;27:495–519. doi: 10.1016/s0160-4120(01)00029-0. [DOI] [PubMed] [Google Scholar]
- 11.Schecter A, Cramer P, Boggess K, Stanley J, Papke O, Olson J, et al. Intake of dioxins and related compounds from food in the US population. J Toxicol Environ Health, Part A. 2001;63:1–18. doi: 10.1080/152873901750128326. [DOI] [PubMed] [Google Scholar]
- 12.Chan J, Xing G, Xu Y, Liang Y, Chen L, Wu S, et al. Body loadings and health risk assessment of polychlorinated dibenzo-p-dioxins and dibenzofurans at an intensive electronics waste recycling site in China. Enviorn Sci Technol. 2007;41:7668–7674. doi: 10.1021/es071492j. [DOI] [PubMed] [Google Scholar]
- 13.Li H, Yu L, Sheng G, Fu J, Peng P. Severe PCDD/F and PBDD/F pollution in air around an electronic waste dismantling area in China. Environ Sci Technol. 2007;41:5641–5646. doi: 10.1021/es0702925. [DOI] [PubMed] [Google Scholar]
- 14.Shen C, Huang S, Wang Z, qiao M, X T, Yu C-J, et al. Identification of Ah Receptor agonists in soil of e-waste recycling sites from Taizhou area in China. Environ Sci Technol. 2008;42:49–55. doi: 10.1021/es071162z. [DOI] [PubMed] [Google Scholar]
- 15.Loutfy N, Fuerhacker M, Tundo P, Raccanelli S, El Dien AG, Ahmed MT. Dietary intake of dioxins and dioxin-like PCBs due to the consumption of dairy products, fish/seafood and meat from Ismailia city, Egypt. Sci Tot Env. 2006;370:1–8. doi: 10.1016/j.scitotenv.2006.05.012. [DOI] [PubMed] [Google Scholar]
- 16.Heilmann C, Grandjean P, Welhe P, Nielsen F. Budtz-Jorgensen. Reduced antibody responses to vaccinations in children exposed to polychlorinated biphenyls. PLOS Medicine. 2006;3:1352–1359. doi: 10.1371/journal.pmed.0030311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stolevik S, Nygaard U, Namork E, Haugen M, Kvalem H, Meltzer H, et al. Prenatal exposure to polychlorinated biphenyls and dioxins is associated iwth increased risk of wheeze and infections in infants. Food Chem Toxicol. 2011;49:1843–1848. doi: 10.1016/j.fct.2011.05.002. [DOI] [PubMed] [Google Scholar]
- 18.Hochstenbach K, van Leeuwen DM, Gmuender H, Gottschalk RW, Stolevik S, Nygaard U, et al. Toxicogenomic profiles in relation to maternal immunotoxic exposure and immune functionality in newborns. Toxicol Sci. 2012;129:315–324. doi: 10.1093/toxsci/kfs214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Stolevik S, Nygaard U, Namork E, Haugen M, Meltzer H, Alexander J, et al. Prenatal exposure to polychlorinated biphenyls and dioxins from the maternal diet may be associated with immunosuppressive effets that persist into early childhood. Food Chem Toxicol. 2013;51:165–172. doi: 10.1016/j.fct.2012.09.027. [DOI] [PubMed] [Google Scholar]
- 20.Dallaire F, Dewailly E, Muckle G, Vizena C, Jacobson S, Jacobson J, et al. Acute infections and environmental exposure to organochlorines in Inuit infants from Nunavik. Environ Health Perspect. 2004;112:1359–1364. doi: 10.1289/ehp.7255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Miyashita C, Sasaki S, Saijo Y, Washino N, Okada E, Kobayashi S, et al. Effects of prenatal exposure to dioxin-like compounds on allergies and infections during infancy. Environ Res. 2011;111:551–558. doi: 10.1016/j.envres.2011.01.021. [DOI] [PubMed] [Google Scholar]
- 22.O'Donnell E, Salli KS, Koch D, Kopparapu P, Farrer D, Bisson W, et al. The anti-inflammatory drug leflunomide is an agonist of the aryl hydrocarbon receptor. PLoS One. 2010 Oct 1;5(10):pii, e13128. doi: 10.1371/journal.pone.0013128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jin U, Lee S, Safe S. Aryl hydrocarbon receptor (AHR)-active pharmaceuticals are selective AHR modulators in MDA-MB-468 and BT474 breast cancer cells. J Pharmacol Exp Therapeut. 2012;343:333–341. doi: 10.1124/jpet.112.195339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Stevens EA, Mezrich JD, Bradfield CA. The aryl hydrocarbon receptor: A perspective on potential roles in the immune system. Immunology. 2009;127:299–311. doi: 10.1111/j.1365-2567.2009.03054.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Funatake C, Marshall N, Steppan LB, Mourich D, Kerkvliet NI. Cutting Edge: Activation of the aryl hydrocarbon receptor (AhR) by 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) generates a population of CD4+CD25+ cells with characteristics of regulatory T cells. J Immunol. 2005;175:4184–4188. doi: 10.4049/jimmunol.175.7.4184. [DOI] [PubMed] [Google Scholar]
- 26.Kerkvliet NI, Steppan LB, Vorachek W, Oda S, Farrer D, Wong CP, et al. Activaiton of aryl hydrocarbon receptor by TCDD prevents diabetes in NOD mice and increases Foxp3+ T cells in pancreatic lymph nodes. Immunotherapy. 2009;1:539–547. doi: 10.2217/imt.09.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Quintana FJ, Basso AS, Iglesias AH, Korn T, Farez MF, Bettelli E, et al. Control of Treg and TH17 cell differentiation by the aryl hydrocarbon receptor. Nature. 2008;453:65–71. doi: 10.1038/nature06880. [DOI] [PubMed] [Google Scholar]
- 28.Quintana FJ, Murugaiyan G, Farez MF, Mitsdoerffer M, Tukpah A-M, Burns EJ, et al. An endogenous aryl hydrocarbon receptor ligand acts on dendritic cells and T cells to suppress experimental autoimmune encephalomyelitis. Proc Natl Acad Sci U S A. 2010;107:20768–20773. doi: 10.1073/pnas.1009201107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Veldhoen M, Hirota K, Christensen JL, O'Garra A, Stockinger B. Natural agonists for aryl hydrocarbon receptor in culture medium are essential for optimal differentiation of Th17 T cells. J Exp Med. 2008;206:43–49. doi: 10.1084/jem.20081438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lawrence BP, Denison M, Novak H, Vorderstrasse BA, Harrer N, Neruda W, et al. Activation of the aryl hydrocarbon receptor is essential for mediating the anti-inflammatory effects of a novel low molecular weight compound. Blood. 2008;112:1158–1165. doi: 10.1182/blood-2007-08-109645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Takamura T, Harama D, Fukumoto S, Nakamura Y, Shimokawa N, Ishimaru K, et al. Lactobacillus bulgaricus OLL1181 activates the aryl hydrocarbon receptor pathway and inhibits colitis. Immunol Cell Biol. 2011;165:1–6. doi: 10.1038/icb.2010.165. doi:10.1038/icb.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Benson J, Shepherd DM. Aryl hydrocarbon receptor activaiton by TCDD reduces inflammation associated with Crohn's disease. Toxicol Sci. 2011;120:68–78. doi: 10.1093/toxsci/kfq360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chmill S, Kadow S, Winter M, Weighardt H, Esser C. 2,3,7,8-Tetrachlorodibenzo-p-dioxin impairs stable establishment of oral tolerance in mice. Toxicol Sci. 2010;118 doi: 10.1093/toxsci/kfq232. [DOI] [PubMed] [Google Scholar]
- 34.Vos J, Kreeftenberg J, Engel H, Minderhoud A, van Noorle Jansen L. Studies on 2,3,7,8-tetrachlorodibenzo-p-dioxin-induced immune suppression and decreased resistance to infection: endotoxin hypersensitivity, serum zinc concentrations and effect of thymosin treatment. Toxicology. 1978;9:75–86. doi: 10.1016/0300-483x(78)90033-1. [DOI] [PubMed] [Google Scholar]
- 35.Hinsdill R, Couch D, Speirs R. Immunosuppresssion in mice inductd by dioxin (TCDD) in feed. J Environ Pathol Toxicol. 1980;4:401–425. [PubMed] [Google Scholar]
- 36.House R, Lauer L, Murray M. Examination of immune parameters and host resistance mechanisms in B6C3F1 mice following adult exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin. JToxicol Environ Health. 1990;31:203–215. doi: 10.1080/15287399009531449. [DOI] [PubMed] [Google Scholar]
- 37.Burleson G, Lebrec H, Yang Y, Ibanes J, Pennington K, Birnbaum L. Effect of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) on influenza virus host resistance in mice. Fund App Toxicol. 1996;29:40–47. doi: 10.1006/faat.1996.0004. [DOI] [PubMed] [Google Scholar]
- 38.Luebke R, Copeland C, Bishop L, Daniels M, Gilmour M. Mortality in dioxin-exposed mice infected with influenza: Mitochondrial toxicity (Reye's like syndrome) versus enhanced inflammation as the mode of action. Toxicol Sci. 2002;69:109–116. doi: 10.1093/toxsci/69.1.109. [DOI] [PubMed] [Google Scholar]
- 39.Warren TK, Mitchell KA, Lawrence BP. Exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin suppresses the cell-mediated and humoral immune response to influenza A virus without affecting cytolytic activity in the lung. Toxicol Sci. 2000;56:114–123. doi: 10.1093/toxsci/56.1.114. [DOI] [PubMed] [Google Scholar]
- 40.Teske S, Bohn AA, Regal JF, Neumiller JJ, Lawrence BP. Exploring mechanisms that underlie aryl hydrocarbon receptor-mediated increases in pulmonary neutrophilia and diminished host resistance to influenza A virus. Am J Physiol Lung Cell Mol Physiol. 2005;289:111–124. doi: 10.1152/ajplung.00318.2004. [DOI] [PubMed] [Google Scholar]
- 41.Vorderstrasse BA, Lawrence BP. Protection against lethal challenge with S. pneumoniae is conferred by aryl hydrocarbon receptor activation, but is not associated with enhanced inflammation. Infect Immun. 2006;74:5679–5686. doi: 10.1128/IAI.00837-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lawrence BP, Roberts AD, Neumiller JJ, Cundiff JA, Woodland DL. Aryl hydrocarbon receptor activation impairs the priming but not the recall of influenza virus-specific CD8+ T cells in the lung. J Immunol. 2006;177:5819–5858. doi: 10.4049/jimmunol.177.9.5819. [DOI] [PubMed] [Google Scholar]
- 43.Shi LZ, Faith NG, Nakayama Y, Suresh M, Steinberg H, Czupyrynski C. The aryl hydrocarbon receptor is required for optimal resistance to Listeria monocytogenes infection in mice. J Immunol. 2007;179:6952–6962. doi: 10.4049/jimmunol.179.10.6952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kiss E, Vonarbourg C, Kopfmann S, Hobeika E, Finke D, Esser C, et al. Natural aryl hydrocarbon receptor ligands control organogenesis of intestinal lymphoid follicles. Sciencexpress. 2011 doi: 10.1126/science.1214914. [DOI] [PubMed] [Google Scholar]
- 45.Veiga-Parga T, Suryawanshi A, Rouse Controlling viral immuno-inflammatory lesions by modulating aryl hydrocarbon receptor signaling. PLoS Pathog. 2011 Dec;7(12):e1002427. doi: 10.1371/journal.ppat.1002427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lee J, Cella M, McDonald K, Garlanda C, Kennedy GD, Nukaya M, et al. AHR drives the development of ILC22 and post-natal lymphoid tissues through Notch-dependent and independent pathways. Nat Immunol. 2012 doi: 10.1038/ni.2187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Julkunen I, T S, Pirhonen J, Ronni T, Melen K, Matikainen S. Molecular pathogenesis of influenza A virus infection and virus-induced regulation of cytokine gene expression. Cytokine Growth Factor Reviews. 2001;12:171–180. doi: 10.1016/s1359-6101(00)00026-5. [DOI] [PubMed] [Google Scholar]
- 48.Wang JP, Kurt-Jones EA, Finberg RW. Innate immunity to respiratory viruses. Cell Microbiol. 2007;9:1641–1646. doi: 10.1111/j.1462-5822.2007.00961.x. [DOI] [PubMed] [Google Scholar]
- 49.Waithman J, Mintern J. Dendritic cells and influenza A virus infection. Virulence. 2012;3:603–609. doi: 10.4161/viru.21864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kohlmeier JE, Woodland DL. Immunity to respiratory viruses. Annu Rev Immunol. 2009;27:61–82. doi: 10.1146/annurev.immunol.021908.132625. [DOI] [PubMed] [Google Scholar]
- 51.Hikono H, Kohlmeier J, Ely KH, Scott I, Roberts AD, Blackman MA, et al. T cell memory and recall responses to respiratory virus infections. Immunol Rev. 2006;211:119–132. doi: 10.1111/j.0105-2896.2006.00385.x. [DOI] [PubMed] [Google Scholar]
- 52.Belz G, Wodarz D, Diaz G, Nowak M, Doherty P. Compromised influenza virus-specific CD8+ T cell memory in CD4+ T cell deficient mice. J Virol. 2002;76:12388–12393. doi: 10.1128/JVI.76.23.12388-12393.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Brown D, Roman E, Swain S. CD4 T cell responses to influenza infection. Semin Immunol. 2004;16:171–177. doi: 10.1016/j.smim.2004.02.004. [DOI] [PubMed] [Google Scholar]
- 54.Boyden A, Legge KL, Waldschmidt T. Pulmonary infection with influenza A virus induces site-specific germinal center and T follicular helper cell responses. PLOS One. 2012;7:e40733. doi: 10.1371/journal.pone.0040733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Betts RJ, Prabhu N, Ho AWS, Hutchinson PE, Rotzschke O, Macary PA, et al. Influenza A virus infection results in a robust, antigen-responsive, and widely disseminated Foxp3+ regulatory T cell response. J Virol. 2012;86:2817–2825. doi: 10.1128/JVI.05685-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Antunes I, Kassiotis G. Suppression of innate immune pathology by regulatory T cells during Influenza A virus infection of immunodeficient mice. J Virol. 2010;84:12564–12575. doi: 10.1128/JVI.01559-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Almansa R, Socias L, Ramirez J, Martin-Loeches I, Valles J, Loza A, et al. Imbalanced pro-and anti-Th17 responses (IL-17/granulocyte colony-stimulating factor) predict fatal outcome in 2009 pandemic influenza. Crit Care. 2011;15 doi: 10.1186/cc10426. doi: 10.1186/cc10426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Vorderstrasse B, Bohn A, Lawrence BP. Examining the relationship between impaired host resistance and altered immune function in mice treated with TCDD. Toxicology. 2003;188:15–28. doi: 10.1016/s0300-483x(02)00749-7. [DOI] [PubMed] [Google Scholar]
- 59.Mitchell K, Lawrence BP. Exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) renders influenza virus-specific CD8+ T cells hyporesponsive to antigen. Toxicol Sci. 2003;74:74–84. doi: 10.1093/toxsci/kfg110. [DOI] [PubMed] [Google Scholar]
- 60.Jin GB, Moore AJ, Head JL, Neumiller JJ, Lawrence BP. Aryl hydrocarbon receptor activation reduces dendritic cell function during influenza virus infection. Toxicol Sci. 2010;116:514–522. doi: 10.1093/toxsci/kfq153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Neff-LaFord HD, Vorderstrasse BA, Lawrence BP. Fewer CTL, not enhanced NK cells, are sufficient for viral clearance from the lungs of immunocompromised mice. Cell Immunol. 2003;226:54–64. doi: 10.1016/j.cellimm.2003.11.005. [DOI] [PubMed] [Google Scholar]
- 62.Neff-LaFord HD, Teske S, Bushnell TP, Lawrence BP. Aryl hydrocarbon receptor activation during influenza virus infection unveils a novel pathway of IFNγ production by phagocytic cells. J Immunol. 2007;179:247–255. doi: 10.4049/jimmunol.179.1.247. [DOI] [PubMed] [Google Scholar]
- 63.Wheeler J, Martin K, Lawrence BP. Novel cellular targets of AhR underlie alterations in neutrophilic inflammation and iNOS expression during influenza virus infection. J Immunol. 2012;190 doi: 10.4049/jimmunol.1201341. doi: 10.4049/jimmunol.1202324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Teske S, Bohn AA, Hogaboam JP, Lawrence BP. Aryl hydrocarbon receptor targets pathways extrinsic to bone marrow cells to enhance neutrophil recruitment during influenza virus infection. Toxicolog Sci. 2008;102:89–99. doi: 10.1093/toxsci/kfm282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Funseth E, Wesslen L, Lindh UF, G, Ilback N-G. Effect of 2,3,7,8-tetrachlorodibenzo-p-dioxin on trace elements, inflammation and viral clearance in the myocardium during Coxsackievirus B3 infection in mice. Sci Tot Env. 2002;284:135–147. doi: 10.1016/s0048-9697(01)00874-9. [DOI] [PubMed] [Google Scholar]
- 66.Funseth E, Ilback N. Coxsackievirus B3 infection alters the update of 2,3,7,8-tetrachlorodibenzo-p-dioxin into various tissues of the mouse. Toxicology. 1994;90:29–38. doi: 10.1016/0300-483x(94)90202-x. [DOI] [PubMed] [Google Scholar]
- 67.Wang T, Wyrick KL, Pecka MR, WIlls TB, Vorderstrasse BA. Mechanistic exploration of AhR-mediated host protection against Streptococcus pneumoniae infection. Int Immunopharm. 2012;13:490–498. doi: 10.1016/j.intimp.2012.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.White K, Lysy H, McCay J, Anderson AC. Modulation of serum complement levels following exposure to polychlorinated dibenzo-p-dioxins. Toxicol Appl Pharmacol. 1986;84:209–219. doi: 10.1016/0041-008x(86)90128-6. [DOI] [PubMed] [Google Scholar]
- 69.Luebke RW, Copeland C, Dilberto JJ, Akubue PI, Andrews DL, Riddle MM, et al. Assessment of Host Resistance to Trichinella spiralis in Mice Following Preinfection Exposure to 2,3,7,8-TCDD. Toxicol Appl Pharmacol. 1994;125:7–16. doi: 10.1006/taap.1994.1043. [DOI] [PubMed] [Google Scholar]
- 70.Luebke RW, Copeland C, Andrews DL. Host Resistance to Trichinella spiralis Infection in Rats Exposed to 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD). Fund Appl Toxicol. 1995;24:285–289. doi: 10.1006/faat.1995.1031. [DOI] [PubMed] [Google Scholar]
- 71.Bowers OJ, Sommersted KB, Sowell RT, Boling GE, Hanneman WH, Titus RG, et al. 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) reduces Leishmania major burdens in C57BL/6 mice. Am J Trop Med Hyg. 2006;75:749–752. [PubMed] [Google Scholar]
- 72.Fernandez-Salguero P, Pineau T, Hilbert D, McPhail T, Lee S, Kimura S, et al. Immune system impairment and hepatic fibrosis in mice lacking the dioxin-binding Ah receptor. Science. 1995;268:722–726. doi: 10.1126/science.7732381. [DOI] [PubMed] [Google Scholar]
- 73.Schmidt JV, Su GH, Reddy JK, Simon MC, Bradfield CA. Characterization of a murine Ahr null allele: involvement of the Ah receptor in hepatic growth and development. Proc Natl Acad Sci U S A. 1996;93:6731–6736. doi: 10.1073/pnas.93.13.6731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Vorderstrasse B, Steppan L, Silverstone A, Kerkvliet N. Aryl hydrocarbon receptor-deficient mice generate normal immune responses to model antigens and are resistant to TCDD-induced immune suppression. Toxicol Appl Pharmacol. 2001;171:157–164. doi: 10.1006/taap.2000.9122. [DOI] [PubMed] [Google Scholar]
- 75.Lahvis G, Bradfield CA. Ahr null alleles: Distinctive or Different? Biochem Pharmacol. 1998;56:781–787. doi: 10.1016/s0006-2952(98)00134-8. [DOI] [PubMed] [Google Scholar]
- 76.Qiu J, Heller JJ, Guo X, Chen ZE, Fish K, Fu Y-X, et al. The aryl hydrocarbon receptor regulates gut immunity through modulation of innate lymphoid cells. Immunity. 2012;36:92–104. doi: 10.1016/j.immuni.2011.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Shi LZ, Czuprynski CJ. Beta-naphthoflavone causes an AhR-independent inhibition of invasion and intracellular multiplication of Listeria monocytogenes in murine hepatocytes. Microb Pathog. 2009;47:258–266. doi: 10.1016/j.micpath.2009.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Elizondo G, Rodriquez-Sosa M, Estrada-Muniz E, Gonzalez FJ, Vega L. Deletion of the aryl hydrocarbon receptor enhances the inflammatory response to Leishmania major infection. Int J Biol Sci. 2011;7:1220–1229. doi: 10.7150/ijbs.7.1220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Sanchez Y, de Dios Rosado J, Vega L, Elizondo G, Estrada-Muniz E, Saavedra R, et al. The unexpected role for the aryl hydrocarbon receptor on susceptibiity to experimental toxoplasmosis. J Biomed Biotechnol. 2010;2010 doi: 10.1155/2010/505694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lawrence BP, Vorderstrasse BA. Activation of the aryl hydrocarbon receptor diminishes the memory response to homotypic influenza virus infection but does not impair host resistance. Toxicol Sci. 2004;79:304–314. doi: 10.1093/toxsci/kfh094. [DOI] [PubMed] [Google Scholar]
- 81.Kerkvliet N, Baecher-Steppan L, Shepherd D, Oughton J, Vorderstrasse B, DeKrey G. Inhibition of TC-1 cytokine production, effector cytotoxic T lymphocyte development and alloantibody production by 2,3,7,8-tetrachlorodibenzo-p-dioxin. J Immunol. 1996;157:2310–2319. [PubMed] [Google Scholar]
- 82.Hardell L, Lindstrom G, van Bavel B, Hardell K, Linde A, Carlberg M, et al. Adipose tissue concentrations of dioxins and dibenzofurans, titers of antibodies to Epstein-Barr virus early antigen and the risk for Non-Hodgkin lymphoma. Environ Res. 2001;87:99–107. doi: 10.1006/enrs.2001.4295. [DOI] [PubMed] [Google Scholar]
- 83.Stancek D, Kosecka M, Oltman M, Keleova A, Johnova E. Links between prolonged exposure to xenobiotics, increased incidence of hepatopathies, immunological disturbances and exacerbation of latent Epstein-barr virus infections. Int J Immunopharmacol. 1995;17:321–328. doi: 10.1016/0192-0561(95)00006-n. [DOI] [PubMed] [Google Scholar]
- 84.Kuppers R. B cells under influence: Transformation of B cells by Epstein-Barr virus. Nat Rev Immunol. 2003;3:801–812. doi: 10.1038/nri1201. [DOI] [PubMed] [Google Scholar]
- 85.Kashuba E, Gradin K, Isaguliants M, Szekely L, Poellinger L, Klein G, et al. Regulation of transactivation function of the aryl hydrocarbon receptor by the Epstein Barr virus-encoded EBNA-3 protein. J Biol Chem. 2006;281:1215–1223. doi: 10.1074/jbc.M509036200. [DOI] [PubMed] [Google Scholar]
- 86.Kashuba E, Kashuba V, Pokrovskaja K, Klein G, Szekely L. Epstein-Barr virus encoded nuclear protein EBNA-3 binds XAP2, a protein associated iwth Hepatitis B virus X antigen. Oncogene. 2000;19:1801–1806. doi: 10.1038/sj.onc.1203501. [DOI] [PubMed] [Google Scholar]
- 87.Inoue H, Mishima K, Yamamoto-Yoshida S, Ushikoshi-Nakayama R, Nakagawa Y, Yamamoto K, et al. Aryl hydrocarbon receptor-mediated induction of EBV reactivation as a risk factor for Sjogren's syndrome. J Immunol. 2012;188:4654–4662. doi: 10.4049/jimmunol.1101575. [DOI] [PubMed] [Google Scholar]
- 88.Chen C-J, Li D, Guo N. Regulation of cellular and viral protein expression by the Epstein-Barr virus transcriptional regulator Zta: implications for therapy of EBV associated tumors. Cancer Biol Ther. 2009;8:987–995. doi: 10.4161/cbt.8.11.8369. [DOI] [PubMed] [Google Scholar]
- 89.Fiorito F, Marfe G, Granato GE, Ciarcia R, De Blasio E, Tafani M, et al. 2,3,7,8-Tetrachlorodibenzo-p-dioxin modifies expression and nuclear/cytosolic localization of bovine herpesvirus 1 immediate-early protein (bICPO) during infection. J Cell Biochem. 2010;111:333–342. doi: 10.1002/jcb.22700. [DOI] [PubMed] [Google Scholar]
- 90.Murayama T, Inoue M, Nomura T, Mori S, Eizuru Y. 2,3,7,8-Tetrachlorodibenzo-p-dioxin is a possible activator of human cytomegalovirus replication in a human fibroblast cell line. Biochem Biophys Res Comm. 2002;296:651–656. doi: 10.1016/s0006-291x(02)00921-x. [DOI] [PubMed] [Google Scholar]
- 91.Yao Y, Hoffer A, Chang C, Puga A. Dioxin activates HIV-1 gene expression by an oxidative stress pathway requiring a functional cytochrome P450 CYP1A1 enzyme. Environ Health Persp. 1995;103:366–371. doi: 10.1289/ehp.95103366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Gollapudi S, Kim CH, Patel A, Sindhu R, Gupta S. Dioxin activates human immunodeficiency virus-1 expression in chronically infected promonocytic U1 cells by enhancing NF-kappa B activity and production of tumor necrosis factor-alpha. Biochem Biophys Res Commun. 1996;226:889–894. doi: 10.1006/bbrc.1996.1445. [DOI] [PubMed] [Google Scholar]
- 93.Ohata H, Tetsuka T, Hayashi H, Onozaki K, Okamoto T. 3-Methylcholanthrene activates human immunodeficiency virus type 1 replication via aryl hydrocarbon receptor. Microbiol Immunol. 2003;47:363–370. doi: 10.1111/j.1348-0421.2003.tb03408.x. [DOI] [PubMed] [Google Scholar]
- 94.Tsyrlov I, Pokrovsky A. Stimulatory effect of the CYP1A1 inducer 2,3,7,8-tetrachlordibenzo-p-dioxin on the reproduction of HIV-1 in human lymphoid cell cultures. Xenobiotica. 1993;23:457–467. doi: 10.3109/00498259309057034. [DOI] [PubMed] [Google Scholar]