Summary
Acute inflammation is associated with cognitive deficits and alterations of cortical plasticity in multiple sclerosis (MS). We tested whether early treatment with high-dose interferon (IFN) beta-1a, known to reduce inflammatory activity, improves cortical function and cognitive deficits in MS.
Eighty treatment-naïve relapsing-remitting MS (RRMS) patients received IFN beta-1a (44 mcg) subcutaneously three times per week. Cognitive performance and cortical plasticity were measured through the paced auditory serial addition test (PASAT) and intermittent theta burst stimulation (iTBS) before and up to two years after IFN beta-1a initiation.
Before treatment, patients with gadolinium-enhancing lesions (Gd+) on MRI performed worse on the PASAT, and showed lower iTBS-induced plasticity, compared with Gd− patients. Six months after treatment initiation both PASAT and iTBS-induced plasticity improved in Gd+ and remained stable in Gd− patients.
These results suggest that cognitive and synaptic plasticity deficits may be rescued during high-dose IFN beta-1a treatment in newly-diagnosed RRMS patients with Gd+ lesions.
Keywords: cognition, inflammation, PASAT, synaptic plasticity, theta burst stimulation, transcranial magnetic stimulation
Introduction
Multiple sclerosis (MS) is associated with cognitive decline that includes alterations in memory, attention and executive functions, such as abstract and conceptual reasoning, fluency, planning and organisation (1). These factors contribute to the severe impairment of MS patients’ everyday functioning and quality of life (2). Cognitive symptoms can be present from the early stages of MS (3) and clinically isolated syndrome (4), and can indicate disease progression despite stable physical symptoms (5).
Learning and memory processes are imprinted in brain circuits as long-term synaptic changes established by experience and activity. In particular, long-term potentiation (LTP) is a use-dependent form of synaptic plasticity that is considered the starting point of learning and memory processes (6,7). This form of synaptic plasticity, initially described in animals, can be explored in a safe and non-invasive manner in humans using paradigms of repetitive transcranial magnetic stimulation (rTMS) (7,8). Among these paradigms, a novel LTP-inducing protocol has been developed to mimic the physiological activity of hippocampal neurons during learning episodes, and has been termed intermittent theta burst stimulation (iTBS) (9).
We recently demonstrated that during the inflammatory phases of relapsing-remitting MS (RRMS), amyloid-beta peptide metabolism is altered, showing a significantly lower detection rate in the cerebrospinal fluid of subjects with gadolinium-enhancing (Gd+) lesions on magnetic resonance imaging (MRI), and that this alteration leads to cognitive and cortical plasticity deficits explored by means of the paced auditory serial addition test (PASAT) and iTBS, respectively (10).
On the basis of these premises, a pharmaceutical approach that has been shown to be effective on Gd+ lesions could also have a positive effect in improving cortical function and cognitive deficits, especially if started at an early stage, when inflammation predominates over neurodegeneration and brain dysfunction might still be reversible. One of the therapies currently used for the treatment of MS is interferon (IFN) beta-1a. High-dose IFN beta-1a has a strong positive impact in preventing the formation of new Gd+ lesions in MS patients (11), possibly explaining its efficacy in improving cognitive abilities in patients with RRMS (12).
Given the efficacy of IFN beta-1a in reducing relapses and the formation of Gd+ lesions, we hypothesised that the treatment may be effective in improving cognitive function by restoring cortical synaptic plasticity disrupted during inflammatory events.
Materials and methods
This study complied with the principles of the Declaration of Helsinki, and was approved by the ethics committee of the Tor Vergata University Hospital in Rome. All the subjects gave their written informed consent to the study, which was performed between March 2008 and December 2011.
Subjects
Eighty treatment-naïve patients (16 males and 64 females, aged between 17 and 39 years) were enrolled. They were diagnosed with RRMS (13) in the six months before recruitment, and had an Expanded Disability Status Scale (EDSS) score of between 0 and 3. None of them had overt cognitive deficits or psychiatric disorders, including substance abuse.
Patients were prescribed subcutaneous IFN beta-1a (Rebif®, Merck Serono S.A., Geneva, Switzerland), 44 mcg three times per week, for the prevention of clinical relapses, and were monitored for up to two years, with scheduled visits that included PASAT at baseline (before starting IFN beta-1a) and at six and 24 months after initiation of IFN beta-1a, and TMS at baseline and every six months thereafter. The PASAT investigates cognitive domains frequently altered in MS, such as attention, concentration, and speed of information processing, and was assessed by applying the available Italian normative values (10,14).
MRI
Standard MRI was performed at baseline, i.e. within the two months preceding IFN beta-1a treatment initiation, and again at 12 and 24 months thereafter. Three-Tesla MRI scans consisted of dual-echo proton density, FLAIR T2-weighted spin-echo, and pre- and post-contrast T1-weighted spin-echo images. All images were acquired in the axial orientation with 3 mm-thick contiguous slices. The presence of Gd+ (0.2 ml/kg i.v.) lesions was evaluated by a neuroradiologist who was unaware of the patient’s clinical details (10).
TMS
In accordance with a previously reported procedure (10), iTBS was delivered over the motor ‘hot spot’ of the right first dorsal interosseous muscle (FDI) through a Magstim Rapid2 stimulator (Magstim, Whitland, UK). The active motor threshold (AMT) was defined as the minimum stimulation intensity required to evoke a motor potential from the FDI during voluntary contraction of about 200 mV. The stimulation intensity was 80% of the AMT. The iTBS protocol consisted of 10 bursts, each burst composed of three stimuli at 50 Hz, repeated at a theta frequency of 5 Hz every 10 s for a total of 600 stimuli (200 s). Sixty motor evoked potentials (MEPs) were collected before iTBS (baseline) and at two different time points (0 and 15 min) after the end of iTBS. Stimulation intensity was set to induce a stable MEP of ∼1mV peak-to-peak amplitude in the relaxed right FDI at baseline and remained unchanged until the end of recordings. MEP amplitudes were then averaged at each time point and normalized to the mean baseline amplitude.
The following tests were also performed (at baseline and 6, 12 and 24 months after IFN beta-1a initiation) through paired-pulse TMS (ppTMS), as reported elsewhere (10): short interval intracortical inhibition (SICI, mediated by intrinsic GABAA-ergic circuits) (15), intracortical facilitation (ICF, believed to follow the preferential recruitment of intrinsic excitatory fibres) (15), short intracortical facilitation (SICF, mediated by excitatory cortical interneurons) (16), and long-interval intracortical inhibition (LICI, mediated by local GABAB pathways) (17) of the left primary motor cortex. One figure-of-eight coil, external diameter 70 mm, was held tangentially to the scalp over the motor ‘hot spot’ for the right FDI muscle. SICI and ICF were tested using ppTMS with a sub-threshold conditioning stimulus (CS) preceding a suprathreshold test stimulus (TS) (15). Stimulation intensity for TS was adjusted in each experiment to evoke a MEP of ∼1mV peak-to-peak amplitude in the relaxed right FDI. The CS was set at 80% AMT. Three conditions were presented in a random order: control (TS given alone) and two paired-pulse (pp) conditions, i.e. TS preceded by CS at one of two different interstimulus intervals (ISIs) (2 and 10 ms). For SICF, the intensity of the CS was set at 90% resting motor threshold (RMT). Six conditions were presented in a random order: TS given alone and five conditions with the TS followed by the CS at one of five different ISIs (1.5, 2.1, 2.7, 3.7, and 4.5 ms) (18). For LICI, the intensity of the CS was set at 120% RMT. Two conditions were presented in a random order: control (TS given alone) and one pp condition (TS preceded by CS) at 100 ms ISI (17). For each experiment, 10 responses were collected for the TS alone and for the conditioned MEPs at each ISI. Changes in MEP amplitude at each ISI were expressed as the percentage of the mean unconditioned MEP amplitude.
Statistical analysis
A repeated measures ANOVA was used to look for differences before and after IFN beta-1a at each time point. In the event of non-parametric data, Friedman’s ANOVA was used. Results were reported using both the intention-to-treat (ITT) and per-protocol populations. ITT analysis used all evaluable subjects. Subjects who missed at least two consecutive scheduled visits were considered non-evaluable and thus excluded from the analysis. Relationships between MRI findings, neurophysiological and clinical tests were assessed using the Spearman correlation test for non-parametric values and Pearson’s correlation test for parametric values. Data, expressed as mean ± SEM, were considered significant at the 0.05 level.
Results
Eighty treatment-naïve RRMS patients were enrolled in the study and divided into two groups: those who, at baseline, presented gadolinium-enahancing lesions on MRI (Gd+) and those who did not (Gd−). A Student’s t-test revealed no significant differences in disease duration, age, EDSS score and relapse rate between the two groups at baseline (Table I). Sixty-five patients completed the two-year follow-up under IFN beta-1a. These patients were included in the per-protocol cohort and analysis. Five patients switched to second-line treatments because of poor clinical response. Six patients suspended IFN beta-1a because of common side effects. The 76 patients who completed the scheduled follow-up visits were included in the ITT analysis. Four patients abandoned the study before completion for personal reasons, and were excluded from the analysis because they missed at least two consecutive scheduled visits.
Table I.
Clinical and demographical characteristics of the studied sample at baseline.
| Group | Gender | Age | Disease duration* | EDSS | Relapse rate** |
|---|---|---|---|---|---|
| Gd+ | 30F 8M | 33±6.7 | 4.2±2.8 | 1.9±0.8 | 0.34±0.17 |
| Gd− | 34F 8M | 35±7.9 | 3.6±2.6 | 1.5±0.7 | 0.41±0.15 |
Abbreviations and symbols: Gd+=patients with gadolinium-enhancing lesions on MRI; Gd−=patients without gadolinium-enhancing lesions on MRI; EDSS=Expanded Disability Status Scale score;
*=time since the first clinical episode;
**=Baseline relapse rate was calculated as the time from first clinical episode to first clinical or MRI relapse.
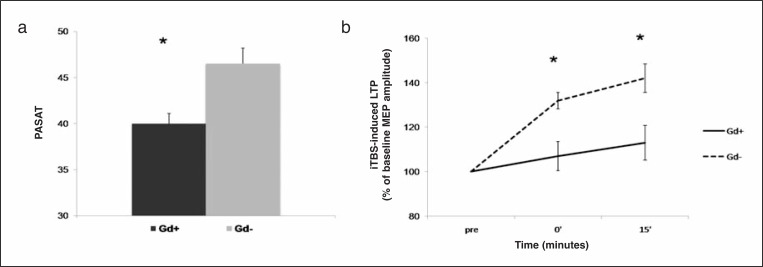
At baseline, the MS patients with Gd+ lesions on MRI (n=38) recorded significantly poorer performances on the PASAT compared with the Gd− MS subjects (n=42) (F=3.62; p=0.037; Fig. 1a). In parallel, the peak amplitude of iTBS-induced LTP was significantly reduced in the Gd+ compared with the Gd− patients (F=5.69; p=0.022; Fig. 1b), confirming previous observations that acute, focal inflammation causes cognitive deficits in MS patients (19,20), and that these deficits are associated with cortical plasticity alterations (10).
Figure 1.
Gd+ MS patients at baseline show lower PASAT scores (a) and iTBS-induced LTP (b) in comparison to Gd− patients, before starting treatment with IFN beta-1a.
* = p<0.05. Data points represent mean values; error bars represent standard error. PASAT=paced auditory serial addition test; iTBS=intermittent theta burst stimulation; LTP=long-term potentiation; GD=gadolinium enhancing lesions; IFN=interferon.
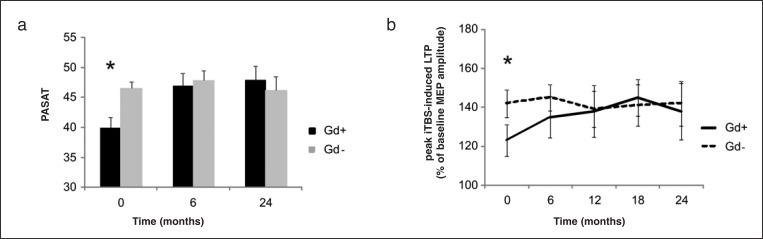
The per-protocol analysis showed that no contrast enhancement was detected on MRI scans during IFN beta-1a treatment in either group (baseline Gd+ and Gd−). During the 24 months of follow-up, the PASAT score improved in the baseline Gd+ patients. Cognitive improvement was already visible after six months of treatment and remained stable at two years (40.6±0.7 at baseline; 46.6±1.1, F=4.25, p=0.03, after six months; 46.8±0.9, F=5.11, p=0.027, after 24 months of IFN beta-1a therapy). In the baseline Gd− group, the PASAT score was stable (p>0.05) for the entire duration of the study (Fig. 2a, over). Peak iTBS-induced LTP was monitored every six months during the course of treatment, showing a marked and constant improvement of synaptic plasticity in the baseline Gd+ group from month 6 to month 18, when the maximal improvement was reached (123.3±1.9 at baseline; 134.6±2.2, F=3.21, p=0.038 after six months; 138±1.9, F=3.89, p=0.029, after 12 months; 145.2±2.3, F=5.69, p=0.022, after 18 months, and 138.8±1.7, F=5.92, p=0.021 after 24 months of IFN beta-1a therapy). In the baseline Gd− group, iTBS-induced LTP was unchanged from baseline to month 24 (Fig. 2b, over).
Figure 2.
PASAT scores (a) and iTBS-induced LTP (b) show a marked and constant improvement in the Gd+ group from month 6 after therapy with IFN beta-1a in comparison to baseline. In the Gd− group, PASAT and iTBS-induced LTP was unchanged from baseline to month 24.
* = p<0.05. Data points represent mean values; error bars represent standard error (SE).
The ITT analysis showed that contrast enhancement was detected on MRI scans during IFN beta-1a treatment in both groups: in six subjects in baseline Gd+, and in four subjects in Gd− at month 12. During the 24 months of follow-up, the PASAT score again showed an improvement in the baseline Gd+ patients and remained stable in the baseline Gd− group. In the baseline Gd+ group, cognitive improvement was again already visible after six months of treatment and remained stable after two years (39.9±0.9 at baseline; 45.9±1.6, F=5.21, p=0.023 after six months; 44.9±1.9, F=4.12, p=0.034 after 24 months of IFN beta-1a therapy). In the baseline Gd− group, the PASAT score was again found to be stable (p>0.05) for the entire duration of the study.
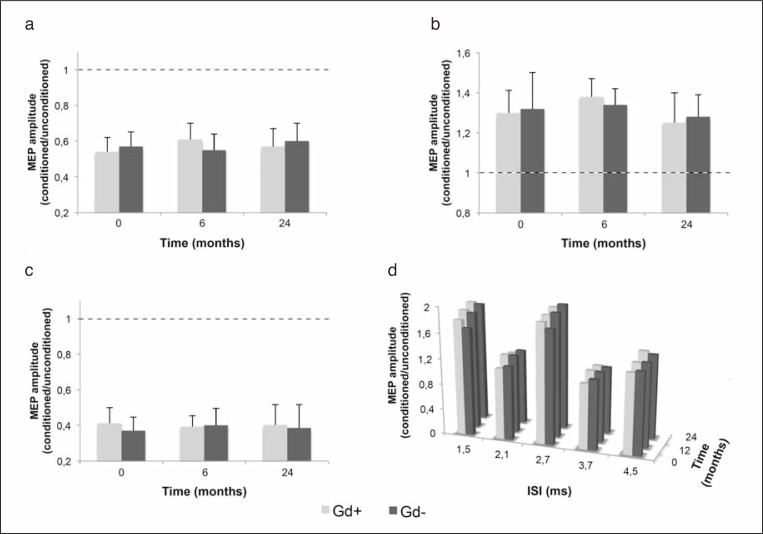
Peak iTBS-induced LTP again showed a marked and constant improvement of synaptic plasticity in the baseline Gd+ group from month 6 to month 18, when the maximal improvement was reached (117.1±2.4 at baseline; 128.7±2.8, F=4.34, p=0.031 after six months; 129.8±2.6, F=2.78, p=0.040, after 12 months; 132.4±2.8, F=4.21, p=0.033, after 18 months and 130.9±2.7, F=2.76, p=0.043 after 24 months of IFN beta-1a therapy). In the baseline Gd− group, iTBS-induced LTP was, again, unchanged from baseline to month 24. SICI, ICF, LICI and SICF were also stable, and indistinguishable, in the Gd+ and Gd− patients at baseline and at 6- and 24-month follow-up (Fig. 3, over).
Figure 3.
SICI (a), ICF (b), LICI (c) and SICF (d) did not differ between Gd+ and Gd− patients and between baseline and 6 and 24 months after IFN beta-1a.
Data points represent mean values; error bars represent standard error. SICI=short-interval intracortical inhibition; ICF=intracortical facilitation; LICI=long-interval intracortical inhibition; SICF=short-interval intracortical facilitation.
Discussion
Multiple sclerosis is a chronic, progressive and disabling disorder with a considerable social impact. The ultimate goal of the available disease-modifying treatments is to delay or halt disease progression, while trying to recover physical and cognitive functions as well as minimizing irreversible disability. For this reason early diagnosis and consequently early treatment are paramount for achieving better outcomes.
In the present study, we demonstrated that it is possible to rescue cognitive and synaptic plasticity deficits during two years of treatment with high-dose IFN beta-1a in newly-diagnosed RRMS patients with Gd+ lesions on the baseline MRI scan, as shown by the increase in both PASAT score and peak iTBS-induced LTP from basal level in this patient population.
High-dose IFN beta-1a is particularly effective in reducing new lesion formation in MS. For example, the EVIDENCE trial demonstrated that IFN beta-1a at a dose of 44 mcg given subcutaneously three times weekly was significantly more effective than IFN beta-1a at the lower dose (30 mcg given intramuscularly once weekly) in reducing relapses and MRI activity in RRMS patients (21). Another important study (REGARD) compared the efficacy of IFN beta-1a with another commonly prescribed drug for RRMS, glatiramer acetate, and found no significant difference between the two drugs on the primary outcome of the trial, i.e. time to relapse and relapse rate. However, patients treated with IFN beta-1a had a significantly lower number of Gd+ lesions, although the change in volume of these lesions was not different between the two treatment groups (11).
The demonstrated efficacy of high-dose IFN beta-1a on Gd+ lesions could explain the results of the present investigation in MS patients. In this respect, clear cognitive enhancing properties of high-dose IFN beta-1a were also demonstrated in the COGIMUS trial in RRMS patients (12), even in the secondary progressive phase of MS (SPMS) (22). In SPMS, the treatment was found to be beneficial in reducing disease progression, as measured by the change in the MS functional composite, which incorporates quantitative tests of ambulation, arm function and cognition, including the PASAT score (22). Long-term potentiation is a complex phenomenon, and changes in glutamate-mediated synaptic drive or GABA-mediated inhibition may alter its induction. Accordingly, blockade of NMDA glutamate receptors prevents LTP (23), as does enhancement of GABA-mediated inhibition (24–26). Thus, we set out to investigate whether the observed changes in LTP amplitude in Gd+ MS patients treated with IFN beta-1a might be secondary to changes in basal synaptic transmission in cortical neurons of MS patients. In ppTMS experiments, however, we found that the degree of SICI, ICF, SICF, and LICI was not affected by the presence of Gd+ lesions, since these parameters were similar at baseline in Gd+ and Gd− patients, suggesting that inflammation affects synaptic plasticity but not basal excitatory or inhibitory transmission.
In conclusion, recent reports emphasized that not only irreversible tissue destruction, but also acute and reversible focal inflammation results in cognitive deficits in MS patients, even in otherwise asymptomatic subjects (10,19,20). As a consequence, disease-modifying agents able to interfere with the formation of new inflammatory lesions have the potential not only to reduce disease progression, but also to improve cognitive abilities and thus possibly the quality of life of MS patients. Our results support the early initiation of disease-modifying therapy with high-dose IFN beta-1a in MS patients, to contrast the subtle cognitive deficits associated with inflammation that characterize the early stages of this disease.
Acknowledgments
This study was supported by a grant from Merck Serono S.A., Geneva, Switzerland to DC.
References
- 1.Chiaravalloti ND, De Luca J. Cognitive impairment in multiple sclerosis. Lancet Neurol. 2008;7:1139–1151. doi: 10.1016/S1474-4422(08)70259-X. [DOI] [PubMed] [Google Scholar]
- 2.Amato MP, Ponziani G, Rossi F, Liedl CL, Stefanile C, Rossi L. Quality of life in multiple sclerosis: the impact of depression, fatigue and disability. Mult Scler. 2001;7:340–344. doi: 10.1177/135245850100700511. [DOI] [PubMed] [Google Scholar]
- 3.Amato MP, Ponziani G, Pracucci G, Bracco L, Siracusa G, Amaducci L. Cognitive impairment in early-onset multiple sclerosis. Pattern, predictors, and impact on everyday life in a 4-year follow-up. Arch Neurol. 1995;52:168–172. doi: 10.1001/archneur.1995.00540260072019. [DOI] [PubMed] [Google Scholar]
- 4.Feuillet L, Reuter F, Audoin B, et al. Early cognitive impairment in patients with clinically isolated syndrome suggestive of multiple sclerosis. Mult Scler. 2007;13:124–127. doi: 10.1177/1352458506071196. [DOI] [PubMed] [Google Scholar]
- 5.Lynch SG, Parmenter BA, Denney DR. The association between cognitive impairment and physical disability in multiple sclerosis. Mult Scler. 2005;11:469–476. doi: 10.1191/1352458505ms1182oa. [DOI] [PubMed] [Google Scholar]
- 6.Bliss TV, Collingridge GL. A synaptic model of memory: long-term potentiation in the hippocampus. Nature. 1993;361:31–39. doi: 10.1038/361031a0. [DOI] [PubMed] [Google Scholar]
- 7.Cooke SF, Bliss TV. Plasticity in the human central nervous system. Brain. 2006;129:1659–1673. doi: 10.1093/brain/awl082. [DOI] [PubMed] [Google Scholar]
- 8.Ziemann U, Ilić TV, Pauli C, Meintzschel F, Ruge D. Learning modifies subsequent induction of long-term potentiation-like and long-term depression-like plasticity in human motor cortex. J Neurosci. 2004;24:1666–1672. doi: 10.1523/JNEUROSCI.5016-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Huang YZ, Edwards MJ, Rounis E, Bhatia KP, Rothwell JC. Theta burst stimulation of the human motor cortex. Neuron. 2005;45:201–206. doi: 10.1016/j.neuron.2004.12.033. [DOI] [PubMed] [Google Scholar]
- 10.Mori F, Rossi S, Sancesario G, et al. Cognitive and cortical plasticity deficits correlate with altered amyloid-beta CSF levels in multiple sclerosis. Neuropsychopharmacology. 2011;36:559–568. doi: 10.1038/npp.2010.187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mikol DD, Barkhof F, Chang P, et al. REGARD study group Comparison of subcutaneous interferon beta-1a with glatiramer acetate in patients with relapsing multiple sclerosis (the REbif vs Glatiramer Acetate in Relapsing MS Disease [REGARD] study): a multicentre, randomised, parallel, open-label trial. Lancet Neurol. 2008;7:903–914. doi: 10.1016/S1474-4422(08)70200-X. [DOI] [PubMed] [Google Scholar]
- 12.Patti F, Amato MP, Bastianello S, et al. COGIMUS Study Group Effects of immunomodulatory treatment with subcutaneous interferon beta-1a on cognitive decline in mildly disabled patients with relapsing-remitting multiple sclerosis. Mult Scler. 2010;16:68–77. doi: 10.1177/1352458509350309. [DOI] [PubMed] [Google Scholar]
- 13.Polman CH, Reingold SC, Edan G, et al. Diagnostic criteria for multiple sclerosis: 2005 revisions to the “McDonald Criteria”. Ann Neurol. 2005;58:840–846. doi: 10.1002/ana.20703. [DOI] [PubMed] [Google Scholar]
- 14.Amato MP, Portaccio E, Goretti B, et al. The Rao’s Brief Repeatable Battery and Stroop Test: normative values with age, education and gender corrections in an Italian population. Mult Scler. 2006;12:787–793. doi: 10.1177/1352458506070933. [DOI] [PubMed] [Google Scholar]
- 15.Kujirai T, Caramia MD, Rothwell JC, et al. Corticocortical inhibition in human motor cortex. J Physiol. 1993;471:501–519. doi: 10.1113/jphysiol.1993.sp019912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ziemann U, Tergau F, Wischer S, Hildebrandt J, Paulus W. Pharmacological control of facilitatory I-wave interaction in the human motor cortex. A paired transcranial magnetic stimulation study. Electroencephalogr Clin Neurophysiol. 1998;109:321–330. doi: 10.1016/s0924-980x(98)00023-x. [DOI] [PubMed] [Google Scholar]
- 17.Valls-Solé J, Pascual-Leone A, Wassermann EM, Hallett M. Human motor evoked responses to paired transcranial magnetic stimuli. Electroencephalogr Clin Neurophysiol. 1992;85:355–364. doi: 10.1016/0168-5597(92)90048-g. [DOI] [PubMed] [Google Scholar]
- 18.Hanajima R, Ugawa Y, Terao Y, et al. Mechanisms of intracortical I-wave facilitation elicited with paired-pulse magnetic stimulation in humans. J Physiol. 2002;538:253–261. doi: 10.1113/jphysiol.2001.013094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Foong J, Rozewicz L, Quaghebeur G, Thompson AJ, Miller DH, Ron MA. Neuropsychological deficits in multiple sclerosis after acute relapse. J Neurol Neurosurg Psychiatry. 1998;64:529–532. doi: 10.1136/jnnp.64.4.529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bellmann-Strobl J, Wuerfel J, Aktas O, et al. Poor PASAT performance correlates with MRI contrast enhancement in multiple sclerosis. Neurology. 2009;73:1624–1627. doi: 10.1212/WNL.0b013e3181c1de4f. [DOI] [PubMed] [Google Scholar]
- 21.Panitch H, Goodin D, Francis G, et al. EVIDENCE (EVidence of Interferon Dose-response: European North American Comparative Efficacy) Study Group and the University of British Columbia MS/MRI Research Group Benefits of high-dose, high-frequency interferon beta-1a in relapsing-remitting multiple sclerosis are sustained to 16 months: final comparative results of the EVIDENCE trial. J Neurol Sci. 2005;239:67–74. doi: 10.1016/j.jns.2005.08.003. [DOI] [PubMed] [Google Scholar]
- 22.Cohen JA, Cutter GR, Fischer JS, et al. IMPACT Investigators Benefit of interferon beta-1a on MSFC progression in secondary progressive MS. Neurology. 2002;59:679–687. doi: 10.1212/wnl.59.5.679. [DOI] [PubMed] [Google Scholar]
- 23.Cooke SF, Bliss TV. Plasticity in the human central nervous system. Brain. 2006;129:1659–1673. doi: 10.1093/brain/awl082. [DOI] [PubMed] [Google Scholar]
- 24.Matsuyama S, Taniguchi T, Kadoyama K, Matsumoto A. Long-term potentiation-like facilitation through GABAA receptor blockade in the mouse dentate gyrus in vivo. Neuroreport. 2008;19:1809–1813. doi: 10.1097/WNR.0b013e328319ab94. [DOI] [PubMed] [Google Scholar]
- 25.Gong N, Li Y, Cai GQ, et al. GABA transporter-1 activity modulates hippocampal theta oscillation and theta burst stimulation induced long-term potentiation. J Neurosci. 2009;29:15836–15845. doi: 10.1523/JNEUROSCI.4643-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pan BX, Dong Y, Ito W, Yanagawa Y, Shigemoto R, Morozov A. Selective gating of glutamatergic inputs to excitatory neurons of amygdala by presynaptic GABAb receptor. Neuron. 2009;61:917–929. doi: 10.1016/j.neuron.2009.01.029. [DOI] [PMC free article] [PubMed] [Google Scholar]