Abstract
Background
Glioblastoma multiforme stem cells display a highly chemoresistant phenotype, whose molecular basis is poorly known. We aim to clarify this issue and to investigate the effects of temozolomide on chemoresistant stem cells.
Methods
A panel of human glioblastoma cultures, grown as stem cells (neurospheres) and adherent cells, was used.
Results
Neurospheres had a multidrug resistant phenotype compared with adherent cells. Such chemoresistance was overcome by apparently noncytotoxic doses of temozolomide, which chemosensitized glioblastoma cells to doxorubicin, vinblastine, and etoposide. This effect was selective for P-glycoprotein (Pgp) substrates and for stem cells, leading to an investigation of whether there was a correlation between the expression of Pgp and the activity of typical stemness pathways. We found that Wnt3a and ABCB1, which encodes for Pgp, were both highly expressed in glioblastoma stem cells and reduced by temozolomide. Temozolomide-treated cells had increased methylation of the cytosine–phosphate–guanine islands in the Wnt3a gene promoter, decreased expression of Wnt3a, disrupted glycogen synthase-3 kinase/β-catenin axis, reduced transcriptional activation of ABCB1, and a lower amount and activity of Pgp. Wnt3a overexpression was sufficient to transform adherent cells into neurospheres and to simultaneously increase proliferation and ABCB1 expression. On the contrary, glioblastoma stem cells silenced for Wnt3a lost the ability to form neurospheres and reduced at the same time the proliferation rate and ABCB1 levels.
Conclusions
Our work suggests that Wnt3a is an autocrine mediator of stemness, proliferation, and chemoresistance in human glioblastoma and that temozolomide may chemosensitize the stem cell population by downregulating Wnt3a signaling.
Keywords: ABCB1/P-glycoprotein, glioblastoma stem cells, temozolomide, Wnt3a
Glioblastoma multiforme (GBM) is the most malignant and frequent intra-axial tumor of the central nervous system. Despite improvements in neurosurgery, radiotherapy, and chemotherapy, no local control of the disease is today possible.1,2 The failure of chemotherapy is due to the tumor's polyclonality, to its modalities of invasion, and to resistance to therapies. The presence of so-called cancer stem cells (CSCs) within GBM has been suggested as another possible reason for chemoresistance.3–5
GBM CSCs are believed to originate from the transformation of neural stem cells of the embryonic matrix and the neurogenic zones of the adult brain6 or from the dedifferentiation of hemispheric astrocytes or tumor cells. The latter reacquire stemness through embryonic regression upon the progressive accumulation of mutations.7–10 GBM CSCs show self-renewal, clonogenicity, and tumorigenicity when transplanted into mice.11 In culture systems without a coated matrix, the occurrence of CSCs in a tumor is denoted by the generation of neurospheres (NSs) beside adherent cells (ACs) after the addition of epidermal growth factor (EGF) and basic fibroblast growth factor (bFGF).12 GBM CSCs share with neural stem cells stemness antigens, such as CD133, Musashi1, nestin, and Sox2,12,13 along with genetic alterations with primary tumors.10,14 They often show high activity of transforming growth factor/bone morphogenetic protein, Notch, Wnt, and sonic hedgehog homolog pathways, which are involved in self-renewal, proliferation, and invasion.15
The molecular basis of chemoresistance in GBM CSCs is under intensive investigation.3,16–18 The main drugs used in the therapy of GBM are DNA-damaging agents, such as the alkylating agents temozolomide (TMZ) and carmustine (BCNU), the topoisomerase II inhibitor etoposide, the topoisomerase I inhibitor irinotecan, and the DNA-intercalating drug doxorubicin.2 TMZ methylates the O6 position on guanine that mismatches with thymine in genomic DNA. The subsequent DNA aberration blocks replication and induces cell death. It is generally recognized that the overexpression of O6-methylguanine-DNA methyltransferase (MGMT), which removes the alkyl group from guanine, mediates the resistance to TMZ in GBM.3,19 However, MGMT alone is not always correlated with resistance to TMZ in GBM CSCs. A plethora of additional intrinsic factors (eg, the activity of DNA-repairing systems or the activity of specific signaling pathways involved in cell proliferation and apoptosis protection) or extrinsic factors (eg, the hypoxic environment or the presence of CSC niches rich in protective growth factors) may contribute to the chemoresistant phenotype of GBM CSCs.3,20
The role of membrane ATP-binding cassette (ABC) transporters—such as P-glycoprotein (Pgp/ABCB1), multidrug-resistance related protein 1 (MRP1/ABCC1), and breast cancer resistance protein (BCRP/ABCG2)—as inducers of the chemoresistant phenotype in GBM CSCs is still a matter of debate.3 Pgp,21,22 MRP1/3/5,21 and BCRP23,24 have been found highly expressed in GBM CSCs, either derived from primary tumors21,23 or obtained in vitro by growing ACs in the NS-generating medium.22 The overexpression of Pgp in NS U87MG cells is predictive of resistance to doxorubicin, etoposide, carboplatin, BCNU,22 and vincristine.25 TMZ has been reported to be a substrate of Pgp,26 although this observation is still controversial.
To our knowledge, the regulation of ABC transporters in GBM CSCs has not yet been investigated. In this work we found that Pgp levels in GBM CSCs are controlled by Wnt3a oncogene and that clinically achievable doses of TMZ interfere with Wnt3a-dependent signaling and decrease the expression of Pgp in this tumor population.
Materials and Methods
Chemicals
Plasticware for cell cultures was from Falcon (Becton Dickinson). TMZ was obtained from Sigma-Aldrich. WntA or 2-amino-4-(3,4-(methylenedioxy)benzylamino)-6-(3-methoxyphenyl)pyrimidine) was purchased from Calbiochem. Human recombinant Dickkopf-1 (Dkk-1) was from R&D Systems. Electrophoresis reagents were obtained from Bio-Rad Laboratories; the protein content of cell lysates was assessed with the bicinchoninic acid kit from Sigma-Aldrich. When not otherwise specified, all the other reagents were purchased from Sigma-Aldrich.
Cells
Primary human GBM cells (CV17, 010627, No3) were obtained from surgical samples of patients operated on at the Department of Neuroscience, Neurosurgical Unit, Universities of Turin and Novara, Italy, and from DIBIT San Raffaele, Milan, Italy (Dr Rossella Galli). The histological diagnosis was performed according to World Health Organization guidelines. Cells were cultured as NSs or ACs as previously described,27 with minor modifications. For NSs, Dulbecco's modified Eagle's medium (DMEM)–F12 was supplemented with 1M HEPES (4-(2-hydroxyethyl)-1-piperazine ethanesulfonic acid), 0.3 mg/mL glucose, 75 μg/mL NaHCO3, 2 mg/mL heparin, 2 mg/mL bovine serum albumin, 2 mM progesterone, 20 ng/mL EGF, and 10 ng/mL bFGF. For ACs, DMEM supplemented with 0.1 mL/mL fetal bovine serum (Lonza) was used. U87MG (American Type Culture Collection) was used as a GBM reference cell line for both NSs and ACs. ACs were obtained from dissociated NS cells, centrifuged at 1200 g for 5 min, and seeded in AC medium.
Morphologic analysis of GBM cells was performed by a Zeiss Axiovert 200M bright field microscope equipped with an AxioCam and ICc3 and coupled to an imaging system (Zeiss AxioVision Release 4.5). For phenotypic characterization, the following antibodies were used: anti-CD133 (Miltenyi Biotec), anti-nestin (Millipore), anti-Musashi1 (Millipore), anti-Sox2 (R&D Systems), anti-EGF receptor (Cell Signaling Technology), anti-p53 (Dako), anti–glial fibrillary acidic protein (Dako), anti-galactocerebroside (Millipore), and anti–βIII-tubulin (Millipore), followed by goat anti-rabbit fluorescein isothiocyanate (FITC)–conjugated immunoglobulin (Ig)G and rabbit anti-mouse tetramethyl rhodamine isothiocyanate–conjugated IgG antibodies. Nuclei were counterstained with 4′,6-diamidino-2-phenylindole (DAPI). Observations were made by immunofluorescence on a Zeiss Axioskop microscope equipped with an AxioCam5MRSc and coupled to an imaging system (Zeiss AxioVision Release 4.5) by using a 63× oil immersion objective (1.4 numerical aperture) and 10× ocular lens. For each experimental point, a minimum of 5 microscopic fields were examined.
Human doxorubicin-sensitive colon cancer HT29 cells and the doxorubicin-resistant counterpart HT29-dx, generated and cultured as previously reported,28 were chosen as models of chemosensitive and chemoresistant cells.
Clonogenic and Self-renewal Assays
For clonogenic assays, ACs and dissociated NSs were seeded in 48-well plates at a concentration of 100 cells/well in the respective media. Fresh medium was added weekly. At days 14, 28, and 42, the spheres or adherent colonies containing at least 50 cells were counted by bright field microscopy. Results were expressed as clonogenic index (number of spheres or adherent colonies/100 plated cells). When the clonogenic assay was performed with NSs treated with TMZ or other chemotherapeutic drugs, cells were treated at days 4, 11, 18, 25, 32, and 39 with 50μM TMZ for 72 h, with or without the second drug added in the last 24 h, then washed and reseeded in fresh medium. The spheres were counted at days 14, 28, and 42 as reported above.
For self-renewal assays, ACs and dissociated NS cells were serially diluted and seeded in 96-well plates at a concentration ranging from 0.5 to 3 cells/well in the respective media. Eighteen hours after seeding, the wells actually containing 1 cell were identified by microscope inspection and used for the assay. Fresh medium was added weekly. The cells per each well were counted at days 14, 28, and 42.
In Vivo Tumorigenicity
Tumorigenicity was tested by transplanting NSs and ACs into nonobese diabetic severe combined immunodeficient mice (Charles River). Two microliters of a 1 × 108 cells/mL suspension were stereotactically injected into the right striatum as previously described.11 Injections were carried out at the DIBIT San Raffaele, Milan. Formalin-fixed, paraffin-embedded brains were stained with hematoxylin/eosin.
Cell Cycle Analysis
Cells were washed twice with fresh phosphate buffered saline (PBS), incubated in 0.5 mL ice-cold ethanol (700 μL/mL) for 15 min, then centrifuged at 1200 g for 5 min at 4°C and rinsed with 0.3 mL citrate buffer (50 mM Na2HPO4, 25 mM sodium citrate, and 10μL/mL Triton X-100) containing 10 μg/mL propidium iodide and 1 mg/mL RNAse (from bovine pancreas). After a 15-min incubation in the dark, intracellular fluorescence was detected by a FACSCalibur flow cytometer (Becton Dickinson). For each analysis, 10 000 events were collected and analyzed by Cell Quest software (Becton Dickinson).
Cytotoxicity and Proliferation Assays
The release of lactate dehydrogenase (LDH) in cell supernatant, considered an index of cell damage and necrosis, was measured as described earlier.28 Briefly, the extracellular medium was centrifuged at 12 000 g for 15 min to pellet cellular debris, whereas the cells were washed with fresh medium, detached with 0.1 µL/mL trypsin/EDTA, resuspended in 0.2 mL of 82.3 mM triethanolamine phosphate-HCl (pH 7.6), and sonicated on ice with two 10-sec bursts. LDH activity was measured in the extracellular medium and in the cell lysate: 50 μL of supernatant from extracellular medium or 5μL of cell lysate was incubated at 37°C with 5 mM NADH. The reaction was started by adding 20 mM pyruvic acid and was followed for 6 min, measuring absorbance at 340 nm with a Packard EL340 microplate reader (Bio-Tek Instruments). The reaction kinetics were linear throughout the time of measurement. Both intracellular and extracellular enzyme activities were expressed as μmol NADH oxidized/min/dish, then extracellular LDH activity was calculated as percentage of the total LDH activity in the dish.
Cell proliferation was measured by growing cells for 24 h in a culture medium containing 1μCi/mL [3H]thymidine (62 Ci/mmol; Amersham Bioscience). At the end of the incubation, the cells were washed with ice-cold PBS, detached by trypsin/EDTA, and resuspended in 200μL PBS. A 50-μL aliquot was used for protein quantification, while the remaining part was counted by liquid scintillation. [3H]thymidine incorporated in each sample was expressed as pmol/mg cell proteins.
Wnt3a Overexpression and Silencing
Fifty thousand adherent 010627 cells were transfected with 2 μg Wnt3a-pCMV6-AC-GFP (green fluorescence protein) expression vector or pCMV6-AC-GFP empty vector as a control, using Turbofectin 8.0 (all Origene Technologies). Cells were sorted 24 h after transfection by detecting the GFP-positive cells (FACSCalibur system). Transfected cells (010627 AC Wnt3a+) were reseeded in fresh AC medium and used for experiments. For Wnt3a silencing, 300 000 NS-forming 010627 cells were treated with Turbofectin 8.0 and 1 μg pGFP-V-RS short hairpin (sh)RNA Wnt3a vector (Origene Technologies) or a 29-mer scrambled shRNA pGFP-V-RS vector (Origene Technologies) used as control. Twenty-four hours after transfection, green fluorescent cells were sorted by flow cytometry analysis. Cells (010627 NS Wnt3a−) were then seeded in fresh NS medium and used for the experiments.
Cells were monitored daily by bright field microscopy. Morphologic analysis was performed 72 h after transfection by an Olympus FV300 laser scanning confocal microscope equipped with a Blue Argon (488 nm) laser and FluoView 300 software (Olympus Biosystems). Nomarski images were obtained by differential interference contrast optical components installed on an IX71 inverted microscope. For each experimental point, a minimum of 5 microscopic fields were examined.
Subcultures of 010627 AC Wnt3a+ cells stably overexpressing Wnt3a and 010627 NS Wnt3a− cells stably silenced for Wnt3a were set up by selecting cells in neomycin- and puromycin-containing medium, respectively. Cells were cultured for up to 20 passages in the respective media. The persistence of the GFP signal was monitored weekly by microscope analysis: samples were rinsed with PBS, fixed with 40 μg/mL paraformaldehyde for 15 min, washed 3 times with PBS, and incubated with DAPI for 3 min at room temperature in the dark. Cells were washed 3 times with PBS and once with water, then the slides were mounted with 4 μL of Gel Mount Aqueous Mounting and examined with a Leica DC100 fluorescence microscope. For each experimental point, a minimum of 5 microscopic fields were examined. In parallel, a subculture of 010627 AC Wnt3a+ cells grown in NS medium and a subculture of 010627 NS Wnt3a− cells grown in AC medium were set up and maintained for 20 passages; cell morphology was analyzed as previously reported here.
Quantitative Real-time PCR
Total RNA was extracted and reverse-transcribed using the QuantiTect Reverse Transcription Kit (Qiagen). Quantitative real-time (qRT) PCR was carried out using IQ SYBR Green Supermix (Bio-Rad) according to the manufacturer's instructions. The same cDNA preparation was used for the quantitation of the genes of interest and β-actin, used as a housekeeping gene. Primer sequences are reported in Supplementary Table S1. The relative quantitation was performed by comparing each PCR product with the housekeeping PCR product, using the Bio-Rad Software Gene Expression Quantitation kit.
Promoter Methylation Assays
Wnt3a and ABCB1 promoter sequences were obtained from the University of California Santa Cruz Genome Browser (http://genome.ucsc.edu/). The localization of cytosine–phosphate–guanine (CpG) islands on Wnt3a and ABCB1 promoters and the design of primers for methylation-specific PCR (MSP) were performed with Methprimer software (http://www.urogene.org/methprimer). Primers specific for immunoprecipitation and ELISA of Wnt3a promoter were designed with Primer3 software (http://frodo.wi.mit.edu/primer3). The primer sequences are reported in Supplementary Table S1.
For immunoprecipitation and ELISA assays, 10 ng of extracted genomic DNA were amplified with specific primers for the CpG island–rich region of the Wnt3a promoter, using the KlenTaq LA DNA Polymerase kit (Sigma-Aldrich). The 1133-bp PCR product was purified with the GenElute PCR Clean-Up kit (Sigma-Aldrich) and divided into aliquots of 100 ng. One aliquot was immunoprecipitated overnight at 4°C with 1 μg of an anti–5 methyl cytosine (5mC) antibody (Active Motif) using the Magna ChIP A/G Chromatin Immunoprecipitation kit (Millipore). Samples were washed, the immunoprecipitated DNA was eluted using the GenElute Mammalian Genomic DNA Miniprep kit (Sigma-Aldrich) and resolved on agarose gel stained with ethidium bromide. A second aliquot was subjected to the quantification of 5mC, using the 5mC DNA ELISA kit (Zymo Research), following the manufacturer's instructions. The absorbance of each sample was read at 450 nm using a Packard EL340 microplate reader and converted into percentage of 5mC, according to a titration curve prepared with serial dilutions of the 5mC DNA positive control provided with the kit.
For MSP assays, 1 μg of extracted genomic DNA was subjected to bisulfite modification using the Methyl Easy Xceed kit (Human Genetics Signatures), following the manufacturer's instructions. MSP was performed with AmpliTaq Gold DNA Polymerase (Applied Biosystems), including universally methylated (CpGenome, Millipore) and unmethylated DNA samples (Human Genetics Signatures) as controls. PCR products were visualized on agarose gel stained with ethidium bromide.
Western Blot Analysis
Cells were rinsed with lysis buffer (50 mM Tris-HCl, 1 mM EDTA, 1 mM EGTA, 150 mM NaCl, and 10 μL/mL Triton-X100; pH 7.4), supplemented with the protease inhibitor cocktail set III (80 μM aprotinin, 5 mM bestatin, 1.5 mM leupeptin, 1 mM pepstatin; Calbiochem), 2 mM phenylmethanesulfonylfluoride, and 1 mM Na3VO4, then sonicated and centrifuged at 13 000 g for 10 min at 4°C. Twenty micrograms of protein extracts were subjected to sodium dodecyl sulfate–polyacrylamide gel electrophoresis and probed with the following antibodies: anti-MGMT (NeoMarkers), anti-Wnt3a (Abcam), anti–glycogen synthase-3 kinase (GSK3; BD Biosciences), anti-phospho(Tyr216)GSK3 (BD Biosciences), anti–β-catenin (BD Biosciences), anti–phospho(Ser33/37/Thr41)β-catenin (Cell Signaling Technology), anti–Pgp/ABCB1 (C219; Calbiochem), and anti-tubulin (Santa Cruz Biotechnology), followed by the peroxidase-conjugated secondary anti-mouse or anti-rabbit antibody (Bio-Rad). The membranes were washed with Tris-buffered saline/Tween, and proteins were detected by enhanced chemiluminescence (PerkinElmer).
Cytosol/nucleus separation was performed as reported.29 Ten micrograms of cytosolic or nuclear extracts were subjected to Western blot analysis using the anti–β-catenin antibody. To check the equal control loading in cytosolic and nuclear fractions, samples were probed respectively with an anti-tubulin or an anti–TATA-box binding protein (TBP/TFIID) antibody (Santa Cruz Biotechnology).
Flow Cytometry Analysis
Cells were washed with PBS, detached with Cell Dissociation Solution (Sigma-Aldrich), and resuspended at 5 × 105 cells/mL in culture medium containing 50 μL/mL fetal bovine serum. Samples were washed with 2.5 μg/mL bovine serum albumin–PBS, incubated with the primary antibodies for Frizzled (Santa Cruz Biotechnology) or low-density lipoprotein–receptor related protein (LRP)6 (Abcam) for 45 min at 4°C, then washed twice and incubated with the secondary FITC-conjugated antibody for 30 min at 4°C. After washing and fixation in 20 μg/mL paraformaldehyde, the surface amount of Frizzled or LRP6 was detected on 100 000 cells by a FACSCalibur system, using Cell Quest software (Becton Dickinson). Control experiments included incubation of cells with nonimmune isotypic antibody followed by secondary antibody.
Chromatin Immunoprecipitation
Chromatin immunoprecipitation (ChIP) experiments were performed using the Magna ChIP A/G Chromatin Immunoprecipitation kit. Samples were immunoprecipitated with 5 μg anti–β-catenin antibody or with no antibody. The immunoprecipitated DNA was then washed and eluted twice with 100μL elution buffer (0.1M NaHCO3, 10 μL/mL sodium dodecyl sulfate), cross-linking was reversed, incubating the samples at 65°C for 6 h, then samples were incubated with proteinase K for 1 h at 55°C. The DNA was eluted using the GenElute Mammalian Genomic DNA Miniprep kit and analyzed by PCR. The putative β-catenin site on the ABCB1 promoter was validated with MatInspector software (http://www.genomatix.de). The primer sequences, designed with Primer3 software (http://frodo.wi.mit.edu/primer3), are reported in Supplementary Table S1. PCR products were visualized on agarose gel stained with ethidium bromide. As negative internal controls, immunoprecipitated samples were subjected to PCR with primers matching 10 000 bp upstream of the ABCB1 promoter. In this condition, no PCR product was detected (not shown).
Rhodamine 123 Efflux
The efflux of rhodamine 123, a specific substrate of Pgp,30 was taken as the index of Pgp activity. Cells were washed with fresh PBS, detached with Cell Dissociation Solution, and resuspended at 5 × 105 cells/mL in 1 mL of culture medium. The samples were maintained at 37°C for 20 min in the presence of 1 μg/mL rhodamine 123. After this incubation time, cells were washed and resuspended in 0.5 mL PBS; the intracellular rhodamine content, which is inversely related to its efflux, was detected using the FACSCalibur system (Becton Dickinson). For each analysis, 100 000 events were collected. Data were analyzed by CellQuest software (Becton Dickinson).
Statistical Analysis
All data in text and figures are provided as means ± SD. The results were analyzed by a 1-way ANOVA. P < .05 was considered significant.
Results
Temozolomide Chemosensitizes GBM Stem Cells to Other Anticancer Drugs
We used 3 GBM cell cultures (CV17, 010627, and U87MG), grown both as ACs and NSs, depending on the culture conditions (Fig. 1A), and we analyzed clonogenicity, self-renewal, cell cycle distribution, and in vivo tumorigenicity. For all 3 cell lines, NSs were significantly more clonogenic than ACs (Fig. 1B); when plated at a concentration of 1 cell/well, ACs did not duplicate over a period of 42 days, different from NSs (Fig. 1C). The most active in clonogenic and self-renewal assay were 010627 NSs, followed by U87MG and CV17. Compared with NSs, ACs showed a significantly higher percentage of cells arrested in G2 phase and a significantly lower percentage of cells in S phase (Fig. 1D).
Fig. 1.
Characterization of GBM NS cells and ACs. (A) Morphologic analysis of CV17, 010627, and U87MG GBM cells, cultured as NSs or ACs and analyzed by a bright field microscope. Magnification: 100 × objective (0.52 numerical aperture); 10 × ocular lens. Bar: 20 μM. (B) Clonogenic assay. NSs or ACs were seeded at a density of 100 cells/well; the spheres or adherent colonies were counted at days 14, 28, and 42. Data are presented as means ± SD (n = 6). Significance of NS vs AC: *P < .01. (C) Self-renewal assay. NSs or ACs were diluted and seeded at a density of 1 cell/well; cells were counted at days 14, 28, and 42. Data are presented as means ± SD (n = 10). Since ACs had a value of 1 cell ± 0 at each time point, statistical analysis was not performed. (D) Cell cycle analysis. The distribution of NSs or ACs in sub-G1, G0/G1, S, and G2/M phases was analyzed by flow cytometry, as detailed under Materials and Methods. Data are presented as means ± SD (n = 4). Significance of NS vs AC: *P < .05.
The in vivo tumorigenicity of 010627 and U87MG NSs has been already reported.11 As far as CV17 cells are concerned, upon intracranial transplantation, ACs did not develop tumors (Supplementary Fig. S1A) or showed a reduced tumorigenicity in 2 out of 4 cases (data not shown), whereas NSs developed tumors (Supplementary Fig. S1B), demonstrating that NS populations included true GBM stem cells.
The distribution of stemness and differentiation markers revealed that 010627 NSs expressed more stemness markers (CD133, Musashi1, nestin, and Sox2) than the other NSs (Supplementary Table S2). MGMT was detected in CV17 NSs and ACs but not in 010627 and U87MG cells (Supplementary Fig. S2).
Treatment with TMZ for 72 h (Fig. 2A) or 120 h (not shown) induced a dose-dependent cell death—measured as release of intracellular LDH in the extracellular medium—in ACs but not in NSs, except for U87MG cells at the highest concentration (200 μM). Doxorubicin, a drug effective against GBM cells,31 was cytotoxic in ACs, as well as in doxorubicin-sensitive human colon cancer HT29 cells, but not in NSs and in the doxorubicin-resistant cell line HT29-dx (Fig. 2B). Surprisingly, preincubation with an apparently nontoxic (50 μM) concentration of TMZ for 72 h, followed by doxorubicin in the last 24 h, induced cell death in NSs not observed with each drug used alone (Fig. 2C). Weekly administration of TMZ or doxorubicin did not reduce the clonogenic potential of NSs (Fig. 2D); only pretreatment with TMZ for 72 h, with further addition of doxorubicin in the last 24 h, significantly decreased clonogenicity (Fig. 2D). Although devoid of effect in cytotoxicity and clonogenicity assays, 50 μM TMZ for 72 h was sufficient to induce G2 arrest, an effect already documented for this drug,32 and a significant increase of pre-G1 cells, suggestive of apoptotic cells, in the case of CV17 NSs (Fig. 2E). On the contrary, doxorubicin, which was also expected to induce G2 arrest in sensitive tumor cells,33 was not effective in NSs. The combination of TMZ followed by doxorubicin significantly increased the percentage of cells arrested in G2 phase and decreased the percentage of cells in S phase toward untreated NSs or NSs treated with TMZ alone (Fig. 2E).
Fig. 2.
Effects of TMZ and doxorubicin on GBM cells. (A–C) Drug-induced cell damage. NSs (open columns) and ACs (hatched columns) were incubated for 72 h with fresh medium (−) or TMZ at 50, 100, and 200 μM (T; panel A), 5 μM doxorubicin for 24 h (dox; panel B), or 50 μM TMZ for 72 h plus 5 μM doxorubicin for the last 24 h (panel C). Human doxorubicin-sensitive colon cancer HT29 cells and the doxorubicin-resistant counterpart HT29-dx cells were used in (B) as positive controls of drug-sensitive and drug-resistant cells, respectively. The culture medium was collected and analyzed in duplicate for the LDH release, as an index of cytotoxicity. Data are presented as means ± SD (n = 4). Significance vs untreated (−) cells: *P < .02; T + dox vs T alone: °P < .05. (D) NSs were seeded at a concentration of 100 cells/well, left untreated (ctrl) or treated at days 4, 11, 18, 25, 32, and 39 with 50 μM TMZ (T) for 72 h, 5 μM doxorubicin for 24 h, or TMZ for 72 h with the addition of doxorubicin in the last 24 h. At the end of each treatment, cells were washed and reseeded in fresh medium. The spheres formed in each well were counted at days 14, 28, and 42. Data are presented as means ± SD (n = 4). Significance vs untreated cells: *P < .02; T + dox vs T alone: °P < .005. (E) NSs incubated as reported in (C) were subjected to cell cycle analysis. Data are presented as means ± SD (n = 3). Significance vs untreated (−) cells: *P < .01; T + dox vs T alone: °P < .02.
The synergistic effects in terms of cell damage (Fig. 3A), reduced clonogenicity (Fig. 3B), and cell cycle arrest (Fig. 3D) were observed also when TMZ was followed by vinblastine or etoposide, but not by cisplatin (Supplementary Fig. S3), methotrexate (Supplementary Fig. S4), or mitoxantrone (Supplementary Fig. S5). Of note, the drugs that synergized with TMZ (doxorubicin, vinblastine, and etoposide) are all substrates of Pgp, whereas cisplatin, methotrexate, and mitoxantrone are mainly effluxed by MRP1/2 and BCRP.34
Fig. 3.
Effects of TMZ, vinblastine, and etoposide on NS GBM cells. (A) Drug-induced cell damage. NSs were incubated with fresh medium (−) or 50 μM TMZ for 72 h (T), 20nM vinblastine for 24 h (VBL), 10 μM etoposide for 24 h (ETO), or 50 μM TMZ for 72 h with the addition of vinblastine or etoposide in the last 24 h. The culture medium was collected and analyzed in duplicate for the LDH release as an index of cytotoxicity. Data are presented as means ± SD (n = 3). Significance vs untreated (−) cells: *P < .05; T + VBL/ETO vs T alone: °P < .05. (B) NSs were seeded in the plate at a concentration of 100 cells/well and left untreated (ctrl) or treated at days 4, 11, 18, 25, 32, and 39 with 50 μM TMZ (T) for 72 h, 20nM vinblastine for 24 h (VBL), 10 μM etoposide for 24 h (ETO), or 50 μM TMZ for 72 h plus vinblastine or etoposide in the last 24 h. At the end of each treatment, cells were washed and reseeded in fresh medium. The spheres formed in each well were counted at days 14, 28, and 42. Data are presented as means ± SD (n = 4). Significance vs untreated cells: *P < .05; T + VBL/ETO vs T alone: °P < .001. (C) NSs incubated as reported in (A) were subjected to cell cycle analysis. Data are presented as means ± SD (n = 3). Significance vs untreated (−) cells: *P < .05; T + dox vs T alone: °P < .05.
Temozolomide Downregulates Wnt3a in GBM Stem Cells
Since TMZ chemosensitizes specifically NSs to Pgp substrates, we first investigated whether in this population there was a correlation between the expression of typical stemness antigens, like Notch, sonic hedgehog, and Wnts, and the expression of Pgp. Notch1, 2, 3, 4 and SHH were variably present in CV17, 010627, and U87MG cells, but their expression did not change in a unidirectional way between ACs and NSs (Supplementary Fig. S6A and B). Among the Wnt antigens present in neural stem cells,35 Wnt5 mRNA was poorly detectable, whereas Wnt3a was higher in all the NS cultures compared with AC (Fig. 4A). NSs showed greater expression of ABCB1 (Fig. 4B), which encodes for Pgp, compared with ACs, and TMZ strongly decreased both Wnt3a and ABCB1 expression in NSs in a time-dependent way (Fig. 4C). NSs also had a higher expression of ABCC1 and ABCG2 (Supplementary Figure S7A and B), but they were not affected by TMZ (Supplementary Fig. S7C).
Fig. 4.
Expression levels of Wnts and ABCB1 genes in GBM cells. CV17, 010627, and U87MG GBM cells, cultured as ACs (open columns) or NSs (hatched columns), were subjected to RNA extraction and qRT-PCR to measure the expression of Wnt3a, Wnt5 (Wn, panel A), and ABCB1 (panel B). The expression level of CV17 ACs was considered “1” and used as the reference for all the other experimental conditions. Data are presented as means ± SD (n = 3). Versus ctrl: *P < .05. (C) Time-dependent expression of Wnt3a and ABCB1 genes in CV17, 010627, and U87MG NSs, incubated in the absence (−) or presence of 50 μM TMZ (T) for 24, 48, and 72 h. The expression level of CV17 cells was considered “1” and used as the reference for all the other experimental conditions. Data are presented as means ± SD (n = 3). Versus ctrl: *P < .05. (D) Methylation of Wnt3a promoter. Genomic DNA from CV17, 010627, U87MG, and No3 NSs, incubated in fresh medium (−) or with 50 μM TMZ (T) for 48 h was subjected to bisulfite modification, followed by PCR with specific primers for methylated (M) and unmethylated (UM) Wnt3a promoter. The figure is representative of 3 experiments with similar results. + : positive controls with a universally methylated or unmethylated genome sequence. bl: blank.
The decrease of Wnt3a started earlier (at 24 h) than the decrease of ABCB1 (Fig. 4C), leading us to hypothesize that a “priming” event induced by TMZ may occur on the Wnt3a gene rather than on the ABCB1 gene. Methylation of CpG islands in promoters is an effective mechanism of gene downregulation. Of note, Wnt3a promoter has many CpG islands, whereas ABCB1 promoter has none (Supplementary Fig. S8A and B). We thus analyzed the methylation of Wnt3a promoter in GBM stem cells untreated and treated with TMZ. The fragment rich with CpG islands in Wnt3a promoter was amplified by PCR from 010627 NSs and subjected to immunoprecipitation with a specific anti-5mC antibody: untreated cells did not show any detectable band of DNA containing 5mC, which instead became detectable after TMZ treatment (Supplementary Fig. S9A). Similarly, using an ELISA kit, which allowed the quantification of 5mC contained in Wnt3a promoter, the percentage of 5mC was below 1% in 010627 NSs but increased up to 25% in cells treated with TMZ (Supplementary Fig. S9B). Interestingly, coincubation with 5-aza-2′-deoxycytidine, an agent that inhibits the methylation of cytosine on position 5 exerted by DNA methyltransferases, significantly counteracted the TMZ effects (Supplementary Fig. S9A and B). In keeping with these experimental findings, in all the NS lines analyzed with MSP, we found a fully unmethylated promoter of Wnt3a, which became methylated in the presence of TMZ (Fig. 4D). The TMZ effect was drug specific. Another alkylating agent, BCNU, did not change the expression levels of Wnt3a and ABCB1 (Supplementary Fig. S10A), irrespective of its cytotoxicity (Supplementary Fig. S10B). Adherent 010627 cells had low Wnt3a basal expression, and we did not detect any further reduction or change in promoter methylation by TMZ (data not shown).
Wnt3a Controls Morphology, Proliferation, and ABCB1 Expression in GBM Stem Cells
To correlate the expression changes of Wnt3a with those of ABCB1, we overexpressed Wnt3a in 010627 ACs (creating the 010627 AC Wnt3a+ subline) and silenced Wnt3a in NS 010627 cells (generating the 010627 NS Wnt3a− subline), using specific vectors containing a reporter GFP. The transfected populations were identified by flow cytometry (Fig. 5A), and the effective overexpression or silencing of Wnt3a was checked by qRT-PCR (Fig. 5B). Surprisingly, 72 h after the transfection, 010627 Wnt3a+ ACs started to detach and grow as NSs, whereas 010627 Wnt3a− NSs progressively disaggregated and grew as a single-cell or few-cell suspension (Fig. 5C). The morphology (Supplementary Fig. S11A) and the expression levels of Wnt3a (Supplementary Fig. S11B) in 010627 Wnt3a+ ACs stably overexpressing Wnt3a and in 010627 Wnt3a− NS cells stably silenced for Wnt3a were checked in up to 20 passages, without observing any further modifications. Of note, 010627 Wnt3a+ ACs grew as NSs in NS and AC media, while 010627 Wnt3a− NSs grew as single-cell/few-cell suspensions in both media (data not shown). Wild-type NS 010627 cells grew faster than ACs (Fig. 5D); 010627 Wnt3a+ ACs proliferated even faster than wild-type 010627 NSs, whereas in 010627 NS Wnt3a− cells the proliferation rate was dramatically reduced (Fig. 5D). In keeping with this trend, 010627 Wnt3a+ ACs showed a greater clonogenic potential than wild-type 010627 ACs; the clonogenicity was even higher than that of wild-type 010627 NSs (Fig. 5E). On the contrary, 010627 Wnt3a− NSs had a dramatic decrease in their clonogenic potential and displayed a similar behavior to wild-type 010627 ACs (Fig. 5E). Wild-type 010627 ACs did not self-duplicate but acquired a self-renewal capacity—even higher than wild-type 010627 NSs—when overexpressing Wnt3a (Fig. 5F). By contrast, in 010627 Wnt3a− NSs, self-renewal, which was a typical feature of NSs, was dramatically reduced. However, 010627 Wnt3a− NSs did not lose completely the self-renewal potential: at day 42 we detected 1 cell in 5/10 wells, 2 cells in 3/10 wells, and 4 cells in 2/10 wells (Fig. 5F).
Fig. 5.
Effects of Wnt3a overexpression and silencing on morphology and proliferation in 010627 GBM cells. (A) 010627 ACs (gray peak) were transfected with a pCMV6-AC-GFP empty vector (dotted line) or with Wnt3a-pCMV6-AC-GFP expression vector (Wnt3a+; continuous line). NS 010627 cells (gray peak) were transfected with a 29-mer scrambled shRNA pGFP-V-RS vector (dotted line) or with a pGFP-V-RS shRNA-Wnt3a vector (Wnt3a−; continuous line). The efficiency of transfection was checked by flow cytometry after 24 h. The figures shown here are representative of 3 similar experiments, each performed in triplicate. (B) Wnt3a expression was detected 48 h after the transfection in triplicate by qRT-PCR. The expression level of 010627 ACs was considered “1” and used as the reference for all the other experimental conditions. Data are presented as means ± SD (n = 3). Versus 010627 AC ctrl: *P < .02; vs 010627 NS ctrl: °P < .001. (C) Microscope analysis of Wnt3a-overexpressing 010627 ACs (010627 AC Wnt3a+) and of Wnt3a-silenced 010627 NSs (010627 NS Wnt3a−). The samples were analyzed by Nomarski differential interference contrast optics (DIC; left panel) or by FV300 laser scanning confocal microscope for GFP signal (right panel). The micrographs are representative of the cell morphology 72 h after transfection. Magnification: 60 × objective (1.4 numerical aperture); 10 × ocular lens. (D) Cell proliferation was measured in triplicate 96 h after transfection by the [3H]thymidine incorporation assay. Data are presented as means ± SD (n = 3). Versus 010627 AC ctrl: *P < .001; vs 010627 NS ctrl: °P < .001. (E) Clonogenic assay. Wild-type 010627 ACs (ctrl), 010627 ACs transfected with a pCMV6-AC-GFP empty vector (empty), AC 010627 cells stably overexpressing Wnt3a (Wnt3a+), wild-type NS 010627 cells (ctrl), NS 010627 transfected with a 29-mer scrambled shRNA pGFP-V-RS vector (scrambled), 010627 NSs stably silenced for Wnt3a (Wnt3a−) were seeded at a density of 100 cells/well; the spheres or adherent colonies were counted at days 14, 28, and 48. Data are presented as means ± SD (n = 4). Significance of Wnt3a+ or Wnt3a− vs the respective ctrl: *P < .002. (C) Self-renewal assay. NSs or ACs were diluted and seeded at a density of 1 cell/well; cells were counted at days 14, 28, and 48. Data are presented as means ± SD (n = 10). Since wild-type 010627 ACs and 010627 ACs transfected with the empty vector had a value of 1 cell ± 0 at each time point, statistical analysis was not performed.
Wnt signaling is canonically transduced by the Frizzled receptors and the LRP5 and LRP6 coreceptors; in the absence of Wnt, cytosolic β-catenin bound to the adenomatous polyposis coli/axin complex is phosphorylated by GSK3 and ubiquitinated. Wnt reduces the phosphorylation of β-catenin by GSK3 and allows its translocation into the nucleus,37,38 where it associates with T-cell factor/lymphoid enhancer factors and acts as a transcription activator of several target genes, including ABCB1.39 Both 010627 wild-type NSs and 010627 Wnt3a+ ACs had low or undetectable levels of active phospho(Tyr216)GSK3 and of phospho(Ser33/37/Thr41)β-catenin (Fig. 6A). On the contrary, in 010627 wild-type ACs and 010627 Wnt3a− NSs, phospho(Tyr216)GSK3 and phospho(Ser33/37/Thr41)–β-catenin were high (Fig. 6A). Of note for the aim of this work, transient (Fig. 6B) and stably transfected (Supplementary Fig. S11C) 010627 Wnt3a+ ACs had higher levels of ABCB1 than parental 010627 ACs, whereas 010627 Wnt3a− NSs had lower levels of ABCB1 than parental 010627 NSs (Fig. 6B; Supplementary Fig. S11C).
Fig. 6.
Effects of Wnt3a overexpression and silencing on the GSK3/β-catenin pathway and ABCB1 expression in 010627 GBM cells. 010627 ACs were transfected with a pCMV6-AC-GFP empty vector (empty) or with Wnt3a-pCMV6-AC-GFP expression vector (Wnt3a+). 010627 NSs were transfected with a 29-mer scrambled shRNA pGFP-V-RS vector (scrambled) or with a pGFP-V-RS shRNA-Wnt3a vector (Wnt3a−). (A) Western blot analysis (96 h after the transfection) of GSK3, phospho(Tyr216)GSK3 (p-GSK3), β-catenin, and phospho(Ser33/37/Thr41)β-catenin (p-catenin) in whole cell lysates. Tubulin expression was used as control of equal protein loading. The figure is representative of 3 experiments with similar results. (B) ABCB1 expression was detected in triplicate by qRT-PCR 96 h after the transfection. The expression level of 010627 AC was considered “1” and used as reference for all the other experimental conditions. Data are presented as means ± SD (n = 3). Versus 010627 AC ctrl: *P < .01; vs 010627 NS ctrl: °P < .001.
Temozolomide Downregulates ABCB1 Transcription by Interfering With the Wnt3a/GSK3/β-catenin Pathway in GBM Stem Cells
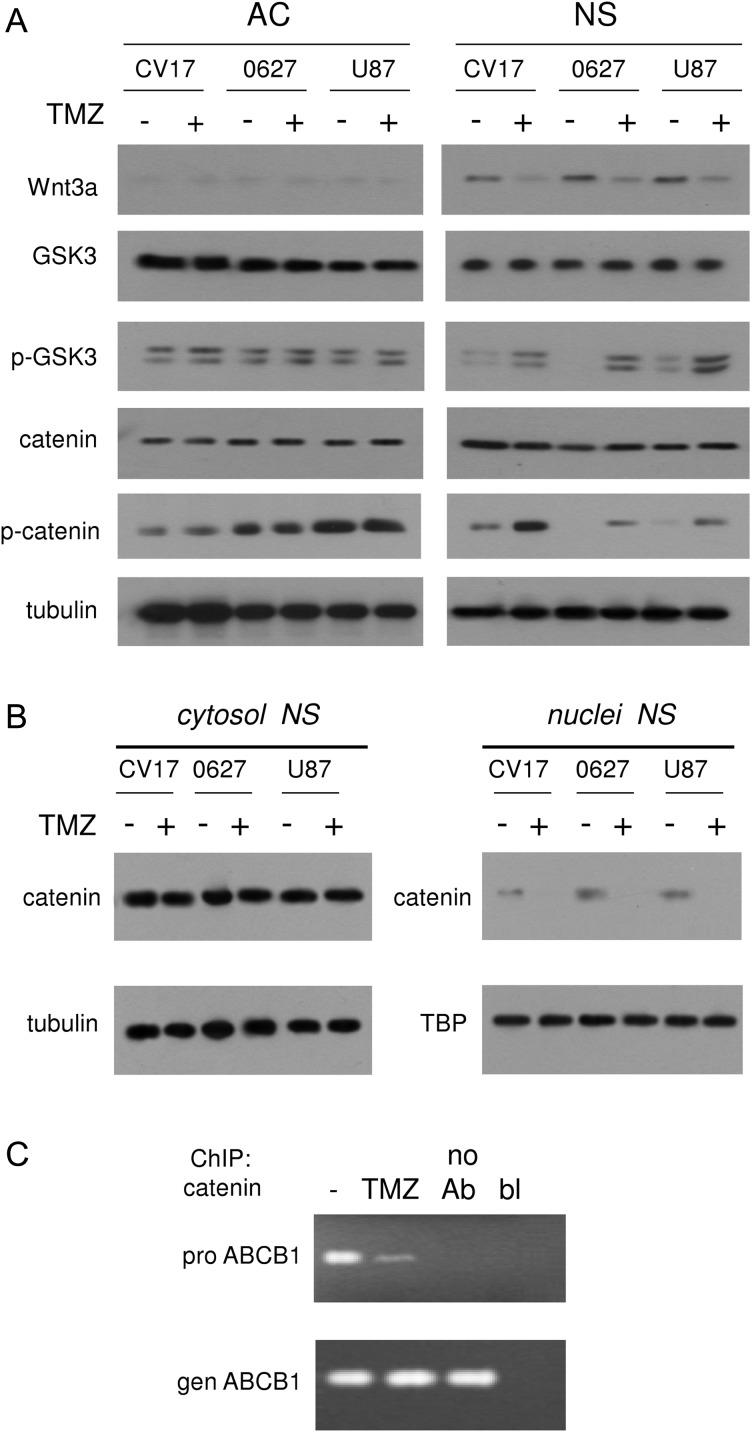
Since TMZ decreases ABCB1 levels, we investigated whether this effect was consequent to the disruption of the Wnt3a/GSK3/β-catenin axis. No ACs had any detectable amounts of Wnt3a and constitutive expression of phospho(Tyr216)GSK3 and phospho(Ser33/37/Thr41)β-catenin; TMZ did not change this expression pattern (Fig. 7A). In NSs, Wnt3a was basally expressed and was paralleled by low levels of phospho(Tyr216)GSK3 and phospho(Ser33/37/Thr41)β-catenin. TMZ, which reduced Wnt3a levels, increased the amount of phosphorylated GSK3 and β-catenin (Fig. 7A). NSs also had higher amounts of the Wnt3a receptor Frizzled (Supplementary Fig. S12A) and coreceptor LRP6 (Supplementary Fig. S12B) compared with ACs. Since TMZ did not modify the surface levels of Frizzled or LRP6 (Supplementary Fig. S12A and B), we excluded that the drug interfered at this point in the Wnt3a axis.
Fig. 7.
Effects of TMZ on Wnt3a/GSK3/β-catenin/Pgp axis in GBM cells. CV17, 010627, and U87MG GBM cells, cultured as ACs or NSs, were incubated for 48 h in the absence (−) or presence (+) of 50 μM TMZ. (A) Western blot analysis of Wnt3a, GSK3, phospho(Tyr216)GSK3 (p-GSK3), β-catenin, and phospho(Ser33/37/Thr41)β-catenin (p-catenin) in whole cell lysates. Tubulin expression was used as control of equal protein loading. The figure is representative of 3 experiments with similar results. (B) Nuclear and cytosolic extracts prepared from NSs were analyzed for the amount of β-catenin. The expressions of tubulin and TBP (TATA-box binding protein) were used as control of equal protein loading for cytosolic and nuclear samples, respectively. The figure is representative of 2 experiments with similar results. (C) ChIP of β-catenin on ABCB1 promoter in 010627 NS cells. no Ab: precipitated samples without anti–β-catenin antibody. bl: blank. The figure is representative of 3 experiments with similar results.
Untreated NSs displayed β-catenin translocated into the nucleus (Fig. 7B), where it was bound to the ABCB1 promoter (Fig. 7C). In keeping with the increased activation of GSK3 and the phosphorylation of β-catenin, TMZ dramatically reduced the amount of β-catenin detectable in NS nuclear extracts (Fig. 7B) and decreased the binding on ABCB1 promoter (Fig. 7C).
To further confirm that TMZ decreased the transcription of ABCB1 by disrupting the Wnt3a/GSK3/β-catenin axis, we treated 010627 NSs with known activators and inhibitors of the Wnt3a/GSK3/β-catenin pathway and measured the expression of ABCB1 mRNA: the synthetic Wnt activator WntA and the GSK3 inhibitor LiCl indeed increased ABCB1 expression, whereas the recombinant Dkk-1 protein decreased it. TMZ counteracted the effects of both WntA and LiCl (Supplementary Fig. S13).
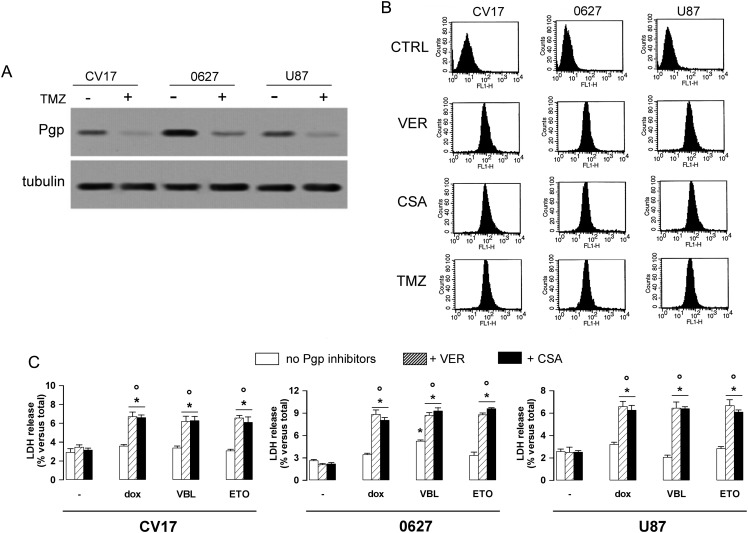
Temozolomide Decreases Expression and Activity of Pgp and Restores the Cytotoxicity of Pgp Substrates in GBM Stem Cells
CV17, 010627, and U87MG NSs had detectable amounts of Pgp protein, which was strongly reduced by TMZ (Fig. 8A). As a consequence, the intracellular accumulation of rhodamine 123, a specific substrate of Pgp, was basally low in NSs (Fig. 8B). Of note, TMZ was as effective as the Pgp inhibitors verapamil and cyclosporine A in reducing the efflux of rhodamine 123 (Fig. 8B). In keeping with this trend, verapamil and cyclosporine A, which were not cytotoxic in NSs, restored the cytotoxicity of the Pgp substrates doxorubicin, vinblastine, and etoposide (Fig. 8C), achieving the same efficacy of nontoxic doses of TMZ (Fig. 2C and 3A).
Fig. 8.
Effects of TMZ on Pgp expression and activity in GBM cells. (A) CV17, 010627, and U87MG GBM cells, cultured as NSs, were incubated for 72 h in the absence (−) or presence (+) of 50 μM TMZ. Cells were lysed and subjected to Western blot for Pgp protein. Tubulin expression was used as control of equal protein loading. The figure is representative of 2 experiments with similar results. (B) NSs were grown in fresh medium (CTRL), with 10 μM verapamil (VER) or cyclosporine A (CSA) for 24 h, or 50 μM TMZ for 72 h, then incubated with the Pgp substrate rhodamine 123. The activity of Pgp, measured as the rate of efflux of rhodamine, was evaluated by flow cytometry (see Materials and Methods). The figures shown here are representative of 3 similar experiments, performed in duplicate. (C) NSs were incubated for 24 h in fresh medium (−) or in the presence of 5 μM doxorubicin (dox), 20nM vinblastine (VBL), or 10 μM etoposide (ETO), alone (no Pgp inhibitors) or together with 10 μM verapamil or 10 μM cyclosporine A. The culture medium was collected and analyzed in duplicate for LDH release, as an index of cytotoxicity. Data are presented as means ± SD (n = 3). Significance vs untreated (−) cells: *P < .01; dox/VBL/ETO + VER or CSA vs dox/VBL/ETO alone: °P < .01.
Discussion
One of the most severe problems encountered by chemotherapy against GBM is the high chemoresistance of the CSC component. We addressed this point by investigating GBM cultures (derived from primary tumors or from commercially available cells) grown as ACs and NSs. The latter were used as models of GBM CSCs, according to our results of clonogenic and self-renewal assays, in vivo tumorigenicity, and the literature.9,11–13,40 A strong resistance toward a panel of drugs that have been variably used in GBM therapy2 differentiates NSs from ACs, suggesting that they may represent 2 different grades of GBM malignancy. Also our NSs showed a multidrug-resistant phenotype. When treated with TMZ, then washed and reseeded in fresh medium, NSs did not alter their proliferation rate. This suggests that adequate recovery periods after TMZ pulses allow cells surviving in TMZ treatment to restart proliferation. This observation is in accord with another work showing that 200 µM TMZ for 7 days, followed by a recovery period of 7 days in fresh medium, did not prevent the formation of secondary spheres of U87MG NSs and tumor formation in vivo.41 Of note, the resistance to TMZ in our models was independent of MGMT levels, since it occurred in both MGMT-positive and MGMT-negative cells, suggesting that several other factors are likely involved.3,20 Unexpectedly, however, noncytotoxic doses of TMZ chemosensitized NSs to apparently noncytotoxic doses of doxorubicin, vinblastine, and etoposide in terms of cell damage, decreased clonogenic potential, and cell cycle arrest.
Polychemotherapy protocols have been used in various experiments in GBM2; for instance, TMZ achieves an appreciable synergistic effect when used together with drugs (eg, inhibitors of poly-ADP ribosyltransferase) that hamper the DNA-repairing machinery.2 In our study, the drugs that synergized with TMZ had other targets than the DNA-repairing enzymes, indicating that the molecular basis of the synergism should rely on different mechanisms. Doxorubicin, vinblastine, and etoposide are all substrates of Pgp.34 By contrast, no synergism was observed between TMZ and drugs that are not substrates of Pgp. Overall, these results suggest that in our experimental conditions, the actual agent responsible for the decreased proliferation of NSs was not TMZ, but the second chemotherapeutic drug, when it was a substrate of Pgp. This observation led us to investigate how TMZ could sensitize GBM CSCs to Pgp substrates and how Pgp was regulated in GBM CSCs, an issue that has been poorly explored.
In keeping with findings in other studies,21,22 NSs showed a higher expression of ABCB1 than did ACs. Of note, TMZ downregulated ABCB1 levels without affecting other ABC transporters. Looking for factors highly expressed in GBM CSCs and putative controllers of ABCB1, we found that NSs had higher levels of Wnt3a compared with ACs. Intriguingly, TMZ reduced Wnt3a expression. The decreased expression of Wnt3a was associated with an increased amount of 5mC in the CpG islands of Wnt3a promoter. The cytotoxic effects of TMZ as alkylating agent are due to the methylation of guanine on the O6 position, an event that induces DNA strand breaks if not repaired by the MGMT enzyme. The vast majority of methylation events exerted by TMZ are, however, represented by other methylations, which are usually not cytotoxic.36 The consequences of these nontoxic methylations, as well as the molecular mechanisms involved, have been less investigated. To the best of our knowledge, there are no reports showing that TMZ directly methylates cytosine on the 5 position. Interestingly, we observed that the increase of 5mC induced by TMZ was reversed by the DNA methyltransferase inhibitor 5-aza-2′-deoxycytidine. This observation suggests that the methylation changes exerted by TMZ are likely due to indirect mechanisms; for instance, they might be consequent to changes in DNA methyltransferase activity, produced by TMZ or its metabolites. Our results pave the way for further investigations to clarify the molecular mechanism by which TMZ may modulate DNA methyltransferase activity or expression and to unveil the importance of intracellular factors (eg, the abundance of CpG islands, the accessibility of position 5 of cytosine after the chromatin folding process, the absence or presence of DNA-binding proteins on a specific promoter, and the type and activity of DNA methyltransferases and demethylating enzymes) in the determination of the rate and type of methylations exerted by TMZ.
In the specific case of Wnt3a, its decrease is achieved by TMZ concentrations that are not sufficient to induce significant cell death in NSs but are compatible with the levels of TMZ circulating in blood.42 We can speculate that also low doses of TMZ have a benefit, such as downregulation of a gene like Wnt3a, which is crucial for cell growth, tumorigenesis, and stemness maintenance in GBM.43,44
As suggested by Wnt3a silencing and overexpression experiments, changes in amounts of Wnt3a produced wide phenotypic changes in GBM CSCs. Since in our model the overexpression of Wnt3a was sufficient to transform the 010627 ACs into NSs, this finding supports the hypothesis that GBM CSCs may derive from a dedifferentiation of tumor cells.7,8 Moreover, since 010627 Wnt3+ ACs grew as NSs independently from the culture medium, this observation suggests that the transition from AC to NS is driven mainly by endogenous tumor-dependent factors, like Wnt3a signaling. On the other hand, the transition from NS to AC appears more complex, as pointed out by 010627 NS Wnt3a− cells, which in our experiment lost the ability to form neurospheres but did not acquire the morphologic feature of ACs even if cultured in the AC medium. Therefore, for the transition from NS to AC, intracellular rather than extracellular factors seem to have the predominant role. Wnt3a is a major determinant in the acquisition or maintenance of stemness because when overexpressed, it conferred to AC cells a “stemness phenotype” in terms of clonogenicity and self-renewal, even more pronounced than that of NS. However, Wnt3a cannot be regarded as the only factor important for stemness, since its silencing was not sufficient to abrogate the self-renewal property or induce a complete transition from NS into AC.
Besides the changes in proliferation and morphology, the decrease of Wnt3a, obtained either by gene silencing or by TMZ, lowered the expression of ABCB1. Time-dependence experiments with TMZ suggested that the decrease of Wnt3a was temporally antecedent to that of ABCB1, raising 2 questions: (i) Does Wnt3a control Pgp expression in GBM CSCs? and (ii) Does TMZ chemosensitize GBM CSCs to Pgp substrates by triggering the Wnt3a decrease?
Compared with ACs, our GBM NSs had higher expression of Frizzled and LRP6. This may favor the basal higher activity of the Wnt/β-catenin pathway and the higher basal expression of Pgp. TMZ did not modify the levels of Frizzled and LRP6 but reduced the production of Wnt3a protein by NSs themselves. We suggest that by doing so it disrupts a sort of autocrine loop, which may be important for stemness, proliferation, and chemoresistance at the same time. To the best of our knowledge, this is the first evidence showing that Wnt3a simultaneously controls these features in GBM and that TMZ interferes with this putative Wnt3a loop.
Some experimental evidence correlated Wnt signaling with chemoresistance.39,45,46 In human multiple myeloma, a tumor that is strongly refractory to several anticancer drugs, autocrine Wnt3a is responsible for chemoresistance; in this case the so-called noncanonical Wnt3a pathway (which recognizes RhoA/RhoA-kinase as intracellular transducers) has a predominant role compared with the GSK3/β-catenin pathway.46 This work suggests that the downstream effectors may vary, whereas Wnt3a appears to be a common upstream inducer of chemoresistance in different tumors. The Wnt antagonist Dkk-1 chemosensitized U87MG cells to alkylating agents such as BCNU and cisplatin, increasing the sensitivity to DNA damage.45 A specific effect on Pgp was not described in the work of Shou et al.,45 which was entirely performed on ACs. In our hands ACs revealed a very low expression of ABCB1. So far, the Pgp-independent effect of Dkk-1 reported by Shou et al45 is not surprising and can be compatible with our findings on NSs, where Pgp levels were high and Dkk-1 downregulated the expression of ABCB1.
Overall, we suggest that TMZ has the peculiar effect of decreasing the autocrine production of Wnt3a in GBM CSCs, to increase the phosphorylation of β-catenin and to reduce the β-catenin–induced expression of Pgp. As a consequence, in cells primed by TMZ, Pgp substrates (like doxorubicin, vinblastine, and etoposide) can accumulate within resistant tumor cells and reach sufficient concentrations to exert cytotoxic and antiproliferative effects. Of note, TMZ achieved the same efficacy of Pgp inhibitors, such as verapamil and cyclosporine A, in chemosensitizing GBM CSCs to Pgp substrates. The use of Pgp inhibitors is a useful tool to demonstrate in vitro the role of Pgp as a critical factor that makes GBM CSCs resistant to several anticancer drugs. Most of the classical Pgp inhibitors have low specificity and high toxicity in vivo and failed in clinical trials. TMZ may represent a valid alternative to these inhibitors, because it has the dual property of being a chemotherapeutic drug and a chemosensitizer agent. Notably, it shows this property at apparently noncytotoxic and clinically achievable doses. Thus, new treatment schemes, based on the association of TMZ with drugs such as doxorubicin, vinblastine, and etoposide, can be hypothesized. The added value of such associations relies on the specific targeting of GBM CSCs, which are the most resistant and unresponsive component of the tumor.
Supplementary Material
Conflict of interest statement. None declared.
Funding
This work was supported by Compagnia di San Paolo “Programma Neuroscienze” (grant 2008.1136), the Italian Association for Cancer Research (MFAG 11475), and the Italian Ministry of University and Research (Programma “Futuro in Ricerca” FIRB 2012). J.K. is the recipient of a Mario and Valeria Rindi Fellowship from the Italian Foundation for Cancer Research.
Supplementary Material
Acknowledgments
We are grateful to Costanzo Costamagna (Department of Oncology, University of Turin) for the technical assistance provided, to Dr Erika Ortolan (Department of Medical Sciences, University of Turin) for the assistance in confocal microscope analysis, and to Dr Oriana Monzeglio (Neuro-bio-oncology Center, Policlinico di Monza Foundation), for the help with methylation promoter assay. We are indebted to Dr Michele Lanotte (Department of Neuroscience, Neurosurgical Unit, University of Turin) and to Dr Rossella Galli (San Raffaele Scientific Institute, Milan) for having provided the primary glioblastoma samples.
References
- 1.Adamson C, Kanu OO, Mehta AI, et al. GBM multiforme: a review of where we have been and where we are going. Expert Opin Investig Drugs. 2009;18:1061–1083. doi: 10.1517/13543780903052764. [DOI] [PubMed] [Google Scholar]
- 2.Bai RY, Staedke V, Riggins GJ. Molecular targeting of GBM: drug discovery and therapies. Trends Mol Med. 2011;17:301–312. doi: 10.1016/j.molmed.2011.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Beier D, Schulz JB, Beier CP. Chemoresistance of glioblastoma cancer stem cells—much more complex than expected. Mol Cancer. 2011;10:128. doi: 10.1186/1476-4598-10-128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gong X, Schwartz PH, Linskey ME, Bota DA. Neural stem/progenitors and glioma stem-like cells have differential sensitivity to chemotherapy. Neurology. 2011;76:1126–1113. doi: 10.1212/WNL.0b013e318212a89f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yamada R, Nakano I. Glioma stem cells: their role in chemoresistance. World Neurosurg. 2012;77:237–240. doi: 10.1016/j.wneu.2012.01.004. [DOI] [PubMed] [Google Scholar]
- 6.Walton NM, Snyder GE, Park D, Kobeissy F, Scheffler B, Steindler DA. Gliotypic neural stem cells transiently adopt tumorigenic properties during normal differentiation. Stem Cells. 2009;27:280–289. doi: 10.1634/stemcells.2008-0842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Stiles CD, Rowitch DF. Glioma stem cells: a midterm exam. Neuron. 2008;58:832–845. doi: 10.1016/j.neuron.2008.05.031. [DOI] [PubMed] [Google Scholar]
- 8.Vescovi AL, Galli R, Reynolds BA. Brain tumour stem cells. Nat Rev Cancer. 2006;6:425–436. doi: 10.1038/nrc1889. [DOI] [PubMed] [Google Scholar]
- 9.Germano I, Swiss V, Casaccia P. Primary brain tumors, neural stem cell, and brain tumor cancer cells: where is the link? Neuropharmacology. 2010;58:903–910. doi: 10.1016/j.neuropharm.2009.12.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Caldera V, Mellai M, Annovazzi L, et al. Antigenic and genotypic similarity between primary GBMs and their derived neurospheres. J Oncol. 2011;2011:314962. doi: 10.1155/2011/314962. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 11.Galli R, Binda E, Orfanelli U, et al. Isolation and characterization of tumorigenic, stem-like neural precursors from human GBM. Cancer Res. 2004;64:7011–7021. doi: 10.1158/0008-5472.CAN-04-1364. [DOI] [PubMed] [Google Scholar]
- 12.Gunther HS, Schmidt NO, Phillips HS, et al. GBM-derived stem cell–enriched cultures form distinct subgroups according to molecular and phenotypic criteria. Oncogene. 2008;27:2897–2909. doi: 10.1038/sj.onc.1210949. [DOI] [PubMed] [Google Scholar]
- 13.Christensen K, Schroder HD, Kristensen BW. CD133 identifies perivascular niches in grade II-IV astrocytomas. J Neurooncol. 2008;90:157–170. doi: 10.1007/s11060-008-9648-8. [DOI] [PubMed] [Google Scholar]
- 14.Lee J, Kotliarova S, Kotliarov Y, et al. Tumor stem cells derived from GBMs cultured in bFGF and EGF more closely mirror the phenotype and genotype of primary tumors than do serum-cultured cell lines. Cancer Cell. 2006;9:391–403. doi: 10.1016/j.ccr.2006.03.030. [DOI] [PubMed] [Google Scholar]
- 15.Clark P, Treisman DM, Ebben J, Kuo JS. Developmental signaling pathways in brain tumor–derived stem-like cells. Dev Dyn. 2007;236:3297–3308. doi: 10.1002/dvdy.21381. [DOI] [PubMed] [Google Scholar]
- 16.Eramo A, Ricci-Vitiani L, Zeuner A, et al. Chemotherapy resistance of GBM stem cells. Cell Death Differ. 2006;13:1238–1241. doi: 10.1038/sj.cdd.4401872. [DOI] [PubMed] [Google Scholar]
- 17.Ghods AJ, Irvin D, Liu G, et al. Spheres isolated from 9L gliosarcoma rat cell line possess chemoresistant and aggressive cancer stem-like cells. Stem Cells. 2007;25:1645–1653. doi: 10.1634/stemcells.2006-0624. [DOI] [PubMed] [Google Scholar]
- 18.Johannessen TCA, Wang J, Skaftnesmo KO, et al. Highly infiltrative brain tumours show reduced chemosensitivity associated with a stem cell-like phenotype. Neuropathol Appl Neurobiol. 2009;35:380–393. doi: 10.1111/j.1365-2990.2008.01008.x. [DOI] [PubMed] [Google Scholar]
- 19.Kaina B, Christmann M, Naumann S, Roos WP. MGMT: key node in the battle against genotoxicity, carcinogenicity and apoptosis induced by alkylating agents. DNA Repair (Amst) 2007;6:1079–1099. doi: 10.1016/j.dnarep.2007.03.008. [DOI] [PubMed] [Google Scholar]
- 20.Johannessen TCA, Prestegarden L, Grudic A, Hegi ME, Bølge Tysnes B, Bjerkvig R. The DNA repair protein ALKBH2 mediates temozolomide resistance in human glioblastoma cells. Neuro Oncol. 2013;15:269–278. doi: 10.1093/neuonc/nos301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Salmaggi A, Boiardi A, Gelati M, et al. GBM-derived tumorospheres identify a population of tumor stem-like cells with angiogenic potential and enhanced multidrug resistance phenotype. Glia. 2006;54:850–860. doi: 10.1002/glia.20414. [DOI] [PubMed] [Google Scholar]
- 22.Nakai E, Park K, Yawata T, et al. Enhanced MDR1 expression and chemoresistance of cancer stem cells derived from GBM. Cancer Invest. 2009;27:901–908. doi: 10.3109/07357900801946679. [DOI] [PubMed] [Google Scholar]
- 23.Liu G, Yuan X, Zeng Z, et al. Analysis of gene expression and chemoresistance of CD133+ cancer stem cells in GBM. Mol Cancer. 2006;5:e67. doi: 10.1186/1476-4598-5-67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bleau AM, Huse JT, Holland EC. The ABCG2 resistance network of GBM. Cell Cycle. 2009;8:2937–2945. [PubMed] [Google Scholar]
- 25.Balik V, Mirossay P, Bohus P, Sulla I, Mirossay L, Sarissky M. Flow cytometry analysis of neural differentiation markers expression in human GBMs may predict their response to chemotherapy. Cell Mol Neurobiol. 2009;29:845–858. doi: 10.1007/s10571-009-9366-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Schaich M, Kestel L, Pfirrmann M, et al. A MDR1 (ABCB1) gene single nucleotide polymorphism predicts outcome of temozolomide treatment in GBM patients. Annal Oncol. 2009;20:175–181. doi: 10.1093/annonc/mdn548. [DOI] [PubMed] [Google Scholar]
- 27.Reynolds BA, Tetzlaff W, Weiss S. A multipotent EGF-responsive striatal embryonic progenitor cell produces neurons and astrocytes. J Neurosci. 1992;12:4565–4574. doi: 10.1523/JNEUROSCI.12-11-04565.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Riganti C, Miraglia E, Viarisio D, et al. Nitric oxide reverts the resistance to doxorubicin in human colon cancer cells by inhibiting the drug efflux. Cancer Res. 2005;65:516–525. [PubMed] [Google Scholar]
- 29.Campia I, Gazzano E, Pescarmona G, Ghigo D, Bosia A, Riganti C. Digoxin and ouabain increase the synthesis of cholesterol in human liver cells. Cell Mol Life Sci. 2009;66:1580–1594. doi: 10.1007/s00018-009-9018-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sarkadi B, Homolya L, Szakacs G, Varadi A. Human multidrug resistance ABCB and ABCG transporters: participation in a chemoimmunity defense system. Physiol Rev. 2006;86:1179–1236. doi: 10.1152/physrev.00037.2005. [DOI] [PubMed] [Google Scholar]
- 31.Hau P, Fabel K, Baumgart U, et al. Pegylated liposomal doxorubicin-efficacy in patients with recurrent high-grade glioma. Cancer. 2004;100:1199–1207. doi: 10.1002/cncr.20073. [DOI] [PubMed] [Google Scholar]
- 32.Chalmers AJ, Ruff EM, Martindale C, Lovegrove N, Short SC. Cytotoxic effects of temozolomide and radiation are additive- and schedule-dependent. Int J Radiation Oncology Biol Phys. 2009;75:1511–1519. doi: 10.1016/j.ijrobp.2009.07.1703. [DOI] [PubMed] [Google Scholar]
- 33.Ling YE, El-Naggar AK, Priebe W, Perz-Soler R. Cell cycle-dependent cytotoxicity, G2/M phase arrest, and disruption of p34cdc2/cyclin B1 activity induced by doxorubicin in synchronized P388 cells. Mol Pharmacol. 1996;49:832–841. [PubMed] [Google Scholar]
- 34.Gottesman MM, Fojo T, Bates SE. Multidrug resistance in cancer: role of ATP-dependent transporters. Nat Rev Cancer. 2002;2:48–58. doi: 10.1038/nrc706. [DOI] [PubMed] [Google Scholar]
- 35.Yu JM, Kim JH, Song GS, Jung JS. Increase in proliferation and differentiation of neural progenitor cells isolated from postnatal and adult mice brain by Wnt-3a and Wnt-5a. Mol Cell Biochem. 2006;288:17–28. doi: 10.1007/s11010-005-9113-3. [DOI] [PubMed] [Google Scholar]
- 36.Wick W, Plattan M, Weller M. New (alternative) temozolomide regimens for the treatment of glioma. Neuro Oncol. 2009;11:69–79. doi: 10.1215/15228517-2008-078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Katoh M, Katoh M. WNT signalling pathway and stem cells signaling network. Clin Cancer Res. 2007;13:4042–4045. doi: 10.1158/1078-0432.CCR-06-2316. [DOI] [PubMed] [Google Scholar]
- 38.MacDonald BT, Tamai K, He X. Wnt/β-catenin signaling: components, mechanisms, and diseases. Dev Cell. 2009;17:9–26. doi: 10.1016/j.devcel.2009.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Flahaut M, Meier R, Coulon A, et al. The Wnt receptor FZD1 mediates chemoresistance in neuroblastoma through activation of the Wnt/b-catenin pathway. Oncogene. 2009;28:2245–2256. doi: 10.1038/onc.2009.80. [DOI] [PubMed] [Google Scholar]
- 40.Bexell D, Gunnarsson S, Siesj P, Bengzon J, Darabi A. CD133+ and nestin+ tumor-initiating cells dominate in N29 and N32 experimental gliomas. Int J Cancer. 2009;125:15–22. doi: 10.1002/ijc.24306. [DOI] [PubMed] [Google Scholar]
- 41.Gilbert CA, Daou MC, Moser RP, Ross AH. Gamma-secretase inhibitors enhance temozolomide treatment of human gliomas by inhibiting neurosphere repopulation and xenograft recurrence. Cancer Res. 2010;70:6870–6879. doi: 10.1158/0008-5472.CAN-10-1378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sankar A, Thomas DG, Darling JL. Sensitivity of short-term cultures derived from human malignant glioma to the anti-cancer drug temozolomide. Anticancer Drugs. 1999;10:179–185. doi: 10.1097/00001813-199902000-00006. [DOI] [PubMed] [Google Scholar]
- 43.Zheng H, Ying H, Wiedemeyer R, et al. PLAGL2 regulates Wnt signaling to impede differentiation in neural stem cells and gliomas. Cancer Cell. 2010;17:497–509. doi: 10.1016/j.ccr.2010.03.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhang N, Wei P, Gong A, et al. FoxM1 promotes β-catenin nuclear localization and controls Wnt target expression and glioma tumorigenesis. Cancer Cell. 2011;20:427–442. doi: 10.1016/j.ccr.2011.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shou J, Ali-Osman F, Multani AS, Pathak S, Fedi P, Srivenugopal KS. Human Dkk-1, a gene encoding a Wnt antagonist, responds to DNA damage and its overexpression sensitize. Oncogene. 2002;21:878–889. doi: 10.1038/sj.onc.1205138. [DOI] [PubMed] [Google Scholar]
- 46.Kobune M, Chiba H, Kato J, et al. Wnt3/RhoA/ROCK signaling pathway is involved in adhesion-mediated drug resistance of multiple myeloma in an autocrine mechanism. Mol Cancer Ther. 2007;6:1774–1784. doi: 10.1158/1535-7163.MCT-06-0684. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.