In cucumber seedlings, phloem transport velocity fluctuates in response to developmental changes and varies among discrete vascular bundles.
Abstract
We use a novel dye-tracing technique to measure in vivo phloem transport velocity in cucumber (Cucumis sativus) plants during early seedling development. We focus on seedlings because of their importance in plant establishment and because they provide a simple source and sink model of phloem transport. The dye-tracing method uses a photodiode to track the movement of a bleach front of fluorescent dye traveling in the phloem from the cotyledons (source) to the roots (sink). During early seedling development, phloem transport velocity in this direction can change 2-fold depending on vascular connectivity and the number of actively growing sinks. Prior to leaf expansion, vascular bundles attached to the first developing leaf demonstrate a decline in basipetal phloem transport that can be alleviated by the leaf’s removal. At this stage, seedlings appear carbon limited and phloem transport velocity is correlated with cotyledon area, a pattern that is apparent both during cotyledon expansion and after source area manipulation. When the first leaf transitions to a carbon source, seedling growth rate increases and basipetal phloem transport velocity becomes more stable. Because bundles appear to operate autonomously, transport velocity can differ among vascular bundles. Together, these results demonstrate the dynamic and heterogeneous nature of phloem transport and underline the need for a better understanding of how changes in phloem physiology impact growth and allocation at this critical stage of development.
Many aspects of plant productivity, carbon allocation, and plant ecology are influenced by the ability of plants to efficiently transport sugars from carbon rich tissues, such as leaves, to carbon poor tissues, where growth and metabolic activity act as sinks. Carbohydrate transport occurs in the phloem and can be regulated by sink and source activity depending on carbon availability and environmental conditions (Marcelis, 1996; Minchin and Thorpe, 1996; Paul and Foyer, 2001). Although Münch (1930) first proposed a mechanism for phloem transport over 80 years ago, methodological limitations associated with studying phloem transport in vivo (Knoblauch and Peters, 2010) have prevented many facets of phloem physiology and anatomy from being understood. Recent work has clarified aspects of phloem loading and examined the implications of sap sugar concentration and phloem ultrastructure on phloem transport (Rennie and Turgeon, 2009; Froelich et al., 2011; Jensen et al., 2011), but what remains unknown is whether phloem transport is stable over space and time and how the structure of the phloem network impacts carbon movement in plants.
Limited, and sometimes conflicting, data exist on the extent that phloem transport changes temporally during a plant’s lifetime (Kallarackal et al., 2012). For example, diurnal variation in carbon assimilation is often predicted to influence phloem sugar concentration and/or carbon flux because of changes in source activity. While several studies have documented fluctuations in these parameters (Peel and Weatherley, 1962; Sharkey and Pate, 1976; Mitchell et al., 1992), it is unclear whether this variation results in changes in transport velocity. A recent study on four species found that three of them maintained a constant velocity over time despite potential changes in diurnal source and sink activity (Windt et al., 2006). One possible explanation for these results is that phloem transport is regulated at the level of velocity. This type of regulation is appealing because it would provide stable transport conditions for phloem-mobile molecular signals. However, more research is needed to determine whether transport velocity is determined by internal regulation and/or solely by modification of source and sink activity.
Transport within the phloem is often sectorial, with lower leaves supplying carbon to the roots and upper leaves supplying carbon to the apical meristem and nearby sinks (Sprugel et al., 1991). Within each sector, vascular connections are further subdivided according to their phyllotaxy, which is the arrangement of leaves around their stem (Watson and Casper, 1984; Marshall, 1996). For example, in potatoes (Solanum tuberosum), radiolabeled carbon (C-14) only travels within vascular bundles on the same side of the plant as the labeled source leaf (Oparka and Davies, 1985). A similar pattern is found in cottonwoods (Populus deltoides), where carbon is imported into the region of the sink closest to the marked source (Larson and Dickson, 1973). While some research suggests that, on a larger scale (e.g. branch or node level), plants can compensate for source and sink removal by transporting carbon between sectors (Watson and Casper, 1984; Marshall, 1996), it remains unclear whether phloem in individual sectors can operate autonomously, leading to variation in phloem transport velocity spatially, over short time periods.
In this study, we examine how phloem transport velocity changes during cucumber (Cucumis sativus) seedling development and whether it is influenced by phyllotaxy. The main objective of this study was to determine whether phloem transport velocity is stable temporally and is homogenous spatially during early plant development. Seedlings present a relatively simple source-sink network for studying patterns of carbon allocation, and by examining their development over time, it is possible to gain insights into how the structure of the phloem network influences its transport. Early stages of plant growth, including the transition to autotrophy and the development of true leaves, are also critical to plant survival and establishment in nature (Lovell and Moore, 1971; Kitajima, 2002; Savage and Cavender-Bares, 2012). Although several methods exist for measuring phloem transport velocity (Minchin and Thorpe, 2003; Windt et al., 2006; Froelich et al., 2011), the spatial and time resolution required for this study necessitated the use of a new dye-tracing technique (Jensen et al., 2011). Slight modifications of this technique allowed us to target individual vascular bundles with minimal disturbance.
RESULTS
Early Seedling Development
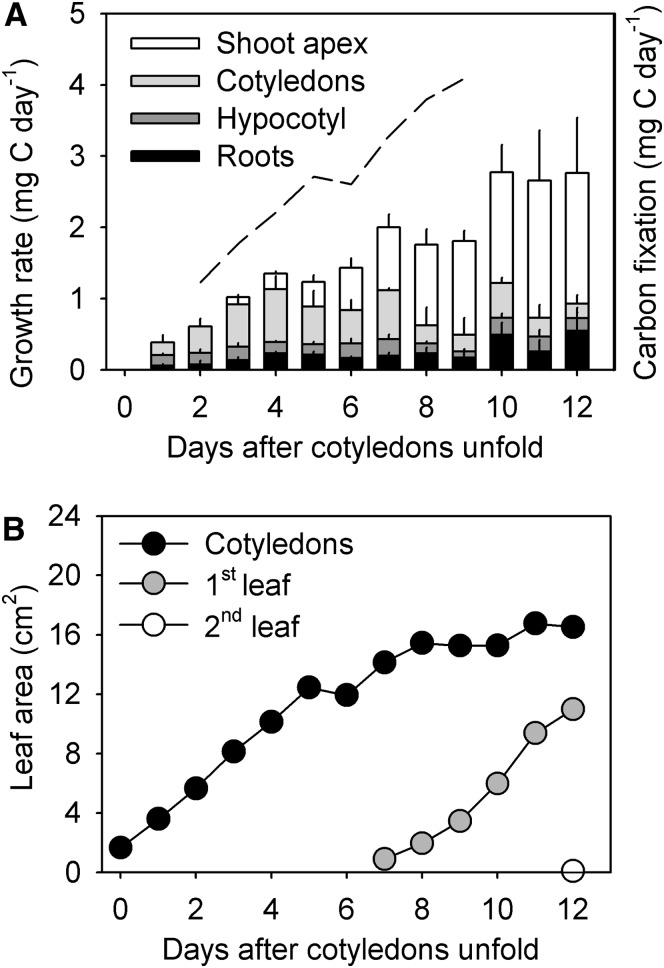
The first 2 weeks of early seedling development were marked by cotyledon and leaf expansion (Fig. 1). Both cotyledons and true leaves had a higher carbon content (39.8% ± 1.4% [se] and 42.1% ± 0.5% [se], respectively) than the hypocotyl (25.5% ± 0.7% [se]) and roots (36.2% ± 0.6% [se]). Because cucumbers are epigeal, their cotyledons serve as the primary site of carbon fixation during early development. Similar to studies on other cucurbits (Turgeon and Webb, 1975), we found that cucumbers did not demonstrate diurnal variation in carbon assimilation when grown under 24 h of constant light (repeated-measures ANOVA, F5,5 = 2.31, P = 0.20). There was also no difference in the rate of photosynthesis in early source leaves and cotyledons (two-sided Student’s t test, t16 = 0.27, P = 0.79). Moreover, the rate of carbon assimilation in cotyledons remained stable at 2.11 ± 0.07 (se) μmol CO2 m–2 s–1 during the first week of development (repeated-measures ANOVA, F7,3 = 4.0, P = 0.35), allowing us to estimate total carbon fixation and infer rates of respiration based on changes in leaf and cotyledon area (Fig. 1A). However, this estimate does not take into account the contribution of other tissues that may to a lesser extent act as carbon sources (e.g. petioles and hypocotyl).
Figure 1.
A, Growth rate on an organ basis during early cucumber development. Growth was estimated based on dry weight and tissue carbon content. The dashed line is an estimate of total carbon fixation based on gas exchange for 1 week. Respiration accounts for the difference between the bars and the dashed line. Shoot apex biomass includes the apical bud and the first true leaves. B, Change in seedling source (cotyledons and true leaves) area over time.
Developmental Changes in Phloem Transport
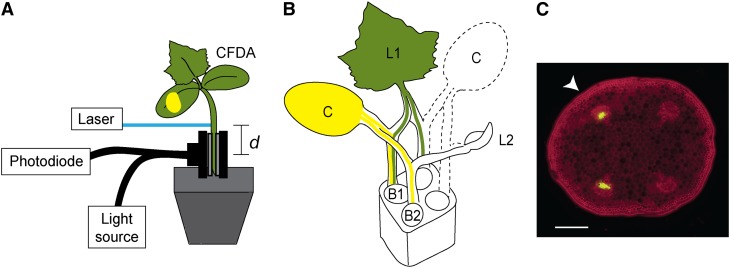
Basipetal phloem transport toward the roots was measured by tracing the movement of a phloem-mobile dye (carboxyfluorescein [CF]) in the hypocotyl toward the roots (Fig. 2A). Seedlings were grown under low-light conditions to guarantee that their hypocotyls were an adequate length for the procedure (>3 cm). Because phloem loading in cucumbers depends on polymer trapping (Turgeon, 1991; Haritatos et al., 1996), dye added directly to the cotyledons readily enters the phloem through symplastic connections with the mesophyll. In young seedlings, there are typically four vascular bundles that span the hypocotyl: two connected to each cotyledon, one of which is attached to the first true leaf (first bundle), and one that is attached to the second true leaf (Fig. 2B, second bundle). The average translocation area in each seedling did not change during the first 2 weeks of development (ANOVA, F11,5 = 5.1, P = 0.62) and did not vary among vascular bundles (two-sided Student’s t test, T = 1.58, P = 0.12). There was also no evidence that median lumen area of the sieve tubes, which averaged 56.8 ± 2.0 [se] μm2, differed between the bundles attached to the first and second developing leaves (two-sided Student’s t test, t6 = –0.42, P = 0.68). Because the development of these leaves occurred in series (Fig. 1B), the first bundles had two major sinks (roots and leaf primordium), while the second bundles had only one major sink (roots) during the first week after the cotyledons expanded. To separate the effects of the different sinks on phloem translocation, velocity was measured on individual vascular bundles (Fig. 2C).
Figure 2.
A, Setup for measuring phloem transport velocity. A phloem-mobile tracer is applied to a cotyledon and travels into the hypocotyl, where it is bleached with a laser. The bleach front is tracked using a photodiode attached to a fiber-optic cable and an excitation light. Transport velocity is estimated based on the time it takes for the bleach front to move the distance (d) from the laser to the sensor. B, Connections between organs and vascular bundles in the hypocotyl. The fate of the dye added to the first leaf (L1) and cotyledon (C), which are marked with dark and light gray (green and yellow, online), respectively. L2 is the second true leaf, and the vascular bundles are numbered based on their attachment to the two leaves (B1 and B2). C, Cross section of hypocotyl after tracer applied to one cotyledon. Cell walls are stained with safranin (magenta, online) and the phloem with CF (green, online). Individual vascular bundles can be targeted by orienting the sensor, as indicated by the white arrow. Bar = 400 µm. [See online article for color version of this figure.]
Carbon Limitation and Source Area
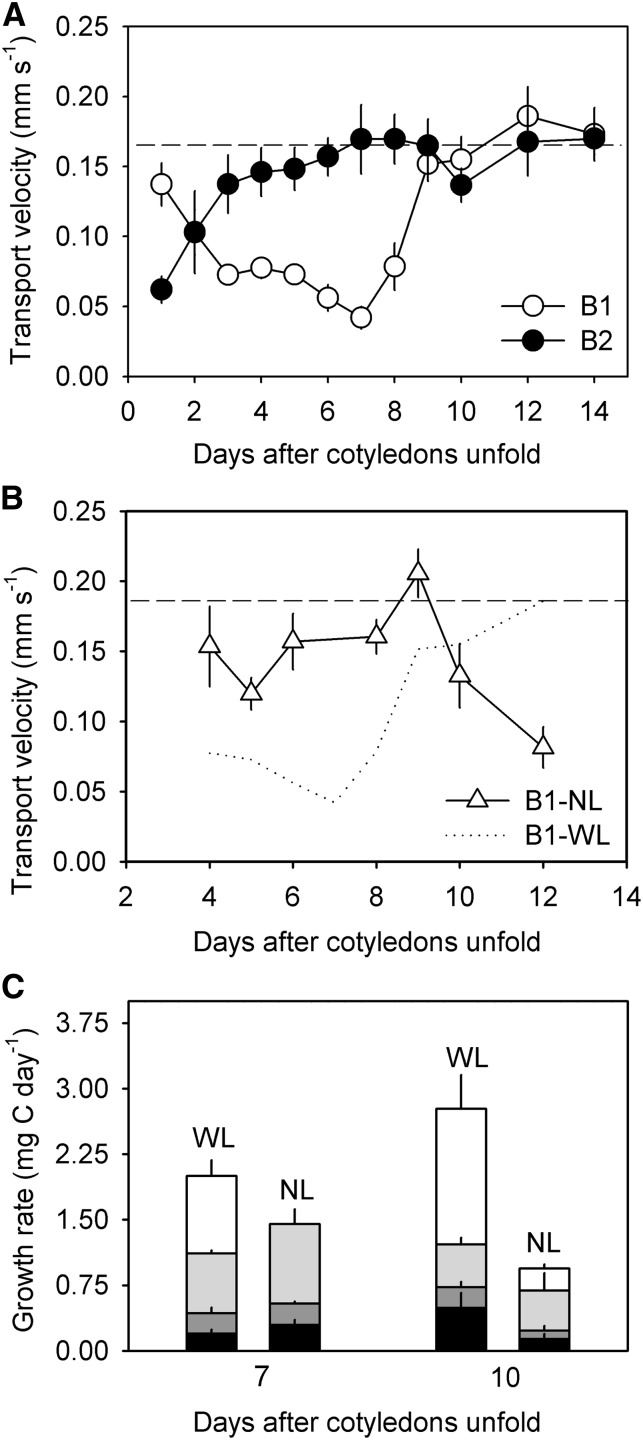
Given that growth in the second true leaf is not initiated until after day 10, vascular bundles attached to this leaf (second bundles) have only one major sink, the roots, during the first week of development (Fig. 1B). Despite this fact, phloem in these bundles demonstrated limited transport toward the roots from day 1 to day 2 (Fig. 3A), a pattern that could result from developmental changes in the shoot apex or cotyledons during this time. Following this period of reduced root investment, the bundles increased their basipetal transport in proportion to their growing cotyledon area (day 3 to day 9; least-squares regression, F5 = 40.5, P = 0.001, R2 = 0.87), quickly achieving velocities similar to those measured in the petioles of adult cucumbers (0.16 ± 0.02 [se] mm s–1; Fig. 3A). To test whether there is a direct correlation between source area and transport velocity in bundles with only one major sink (second bundles), we manipulated cotyledon area on a set of seedlings by either physically cutting one cotyledon in half or by shading it during early development. We then compared phloem transport velocity between the smaller (cut or shaded) and normal-sized cotyledons. We consistently found that transport velocity was slower in bundles attached to the smaller cotyledons (Table I).
Figure 3.
A, Change in seedling phloem transport velocity toward the roots. Two sets of vascular bundles were measured (B1 and B2), which are attached to the first and second developing leaf, respectively. See Figure 2B for schematic of vascular connectivity. B, Transport velocity in the first bundle after leaf removal (NL). The dotted line repeats data from samples with a leaf (WL), which is shown in A. In A and B, the dashed line indicates the average transport velocity measured in the petioles of adult plants. C, Average carbon-specific growth rate (mg C day–1) of seedlings with and without the first developing leaf on day 7 and day 10. Data for the with-leaf treatment are regraphed from Figure 1A. See Figure 1A for legend. In all panels, error bars are ± 1 se.
Table I. Average phloem transport velocity measured on second bundle of both cotyledons after source manipulation.
Source area of each plant was reduced by shading or cutting one cotyledon. Measurements were taken 5 d after the cotyledons unfolded. Values reported are cotyledon area (mm2) and average velocity toward the roots (mm s–1). se is in parentheses. Unique letters indicate signficantly different results (α = 0.01). For small and large cotyledon treatments, N is 4 and 5, respectively.
| Measurement | Cotyledon | Shading | Cutting |
|---|---|---|---|
| Cotyl. area | Small | 219 (25)A | 308 (34)A |
| Cotyl. area | Large | 610 (41)B | 596 (50)B |
| Velocity | Small | 0.05 (0.01)C | 0.07 (0.01)C |
| Velocity | Large | 0.12 (0.01)D | 0.14 (0.01)D |
Phloem Transport during Sink Addition
Bundles attached to the first true leaf (first bundles) exhibited a decline in basipetal transport in the phloem during the first week of development, reaching a minimum velocity on day 7 (Fig. 3A), the date when the first leaf blade became visible (Fig. 1B). This point marks the beginning of the transition from a simple one-sink system to a more complex two-sink system (roots and leaf). To examine whether the addition of this new sink could explain the observed drop in transport velocity, we monitored growth and phloem transport in a set of seedlings that had their first leaf removed 1 d prior to measurement. This manipulation resulted in faster basipetal transport from day 4 to day 9, such that phloem in all the bundles transported at a similar velocity (Fig. 3, A and B). Although this manipulation did not significantly change the seedlings’ carbon-based growth rate (mg C day–1; two-sided Student’s t test, t6 = 1.55, P = 0.17), it did result in increased allocation to the roots (two-sided Student’s t test, t6 = 3.68, P = 0.01) and cotyledons (two-sided Student’s t test, t6 = 4.55, P = 0.004) on day 7 (Fig. 3C). It also led to a smaller cotyledon size (two-sided Student’s t test, t6 = –3.55, P < 0.0001) and lower specific leaf area (two-sided Student’s t test, t6 = –5.25, P < 0.0001, data not shown) in manipulated seedlings. Root elongation rate remained the same (8.16 ± 3.3 [se] mm day–1) regardless of whether the first leaf was removed. Taken together, these data suggest that the addition of a new sink (the first leaf) causes a reduction in carbon allocation to the roots and cotyledons that was alleviated by removing the developing tissue. The cost associated with leaf development appears to accrue before leaf expansion, during a time when there is higher respiration and likely greater carbon investment in young tissue (Figs. 1 and 3A).
Phloem Transport after Source Addition
In the second week of development, basipetal phloem transport increased in the bundles attached to the first true leaf (first bundles), suggesting that this leaf quickly begins fixing carbon and thus alleviates its drain on the cotyledons (Fig. 3A). When the first true leaf was removed after day 9, there was a decline in phloem transport toward the roots in the attached bundles (Fig. 3B) and a significant reduction in seedling carbon-based growth rate (two-sided Student’s t test, t6 = –3.40, P = 0.01) from 2.8 ± 0.4 (se) mg C day–1 to 0.95 ± 0.3 (se) mg C day–1 (Fig. 3C). As expected, the slower growth rate of the manipulated seedlings was primarily caused by a reduction in shoot apical growth (two-sided Student’s t test, t6 = –3.33, P = 0.02) resulting from the loss of the first true leaf. In the nonmanipulated seedlings, the first true leaf accounted for 80% of shoot apical growth.
The gradual transition of new leaves from sink to source tissue can be visualized by tracking the movement of CF from cotyledons into developing leaves (Fig. 4). Once a region of the leaf begins exporting carbon, dye from the nearby cotyledons will no longer be transported into the tissue. In this experiment, the sink-source transition occurred between day 9, when the tip of the leaf is no longer importing dye, and day 11, when the leaf is only acting as a carbon source. The timing of this transition is consistent with the sink manipulation experiments (Fig. 3, B and C). At this time, CF applied to the first true leaf travels into the hypocotyl, indicating the leaf is exporting carbon. Together, these data provide support that young leaves begin to act as sources and supplement seedling growth during the second week of development.
Figure 4.
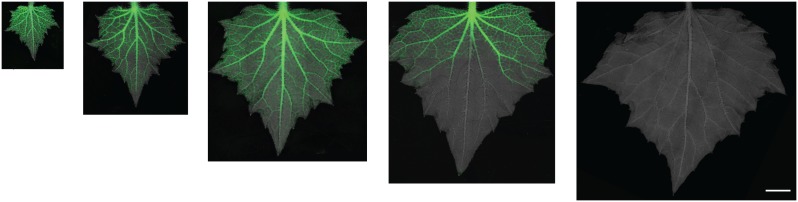
Transition of immature leaves from sink to source tissue. The perfusion of the phloem-mobile tracer (CF; green, online) declines with transport into the leaf, indicating reduced dependence on external carbon. Cucumber leaves were imaged with a fluorescent scope during a 5-d period to visualize their sink-source transition. Bar = 5 mm. [See online article for color version of this figure.]
Heterogeneity in Phloem Transport
Measurements made on individual vascular bundles clearly demonstrated the heterogeneous nature of phloem transport in cucumber seedlings (Fig. 3A). To confirm that this heterogeneity was present within a single plant and not a result of measuring each bundle on a separate individual, we slightly modified our tracking method. Instead of focusing on one bundle, we placed the sensor in the center of the hypocotyl directly under a cotyledon and moved the laser between the two bundles positioned at either corner of the hypocotyl. This method allowed us to measure multiple bundles in one plant, but it had a lower success rate because high fluorescence in one bundle could easily mask the other if they were close together (e.g. seedlings were young). Although we were only able to attain a few measurements using this method (Table II), these measurements were consistent with measurements made on individual bundles (Fig. 3A).
Table II. Phloem transport velocity toward the roots measured in different vascular bundles on the same plant.
Measurements were taken 7 d after the cotyledons unfolded. Values reported are average velocity (mm s–1) with se in parentheses (n = 3).
| Seedlings | First Bundle | Second Bundle |
|---|---|---|
| 1 | 0.08 (0.01) | 0.16 (0.02) |
| 2 | 0.05 (0.00) | 0.17 (0.00) |
| 3 | 0.07 (0.01) | 0.12 (0.01) |
Because both of these methods rely on inferring the arrangement of vascular bundles based on external morphology, we also examined phloem transport using fluorescent microscopy. This involved removing a thin layer of cortical cells from the hypocotyl and using time lapse imaging to record the movement of the phloem-mobile tracer (Fig. 5; Supplemental Video S1). Note that in these images, the number of cells above the sieve tube in the two bundles is not identical, causing some optical distortion. Using this procedure, we clearly document a divergence in the basipetal transport velocity of two vascular bundles. Velocities estimated using this technique were similar to those measured with photodiodes, but it is important to note that because we were not tracking the movement of a bleach front, the measurements are not independent of dye loading rate.
Figure 5.
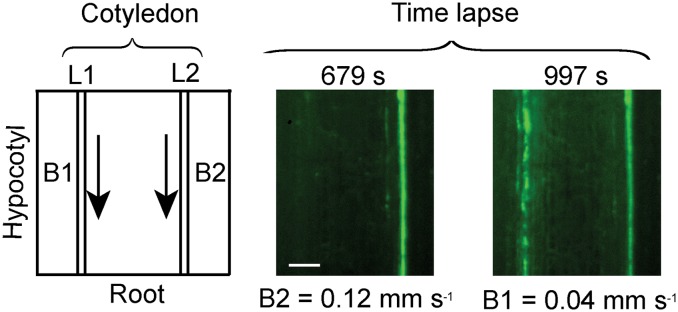
Time-lapse images of dye (CF; green, online) traveling in the phloem of two vascular bundles attached to the same cotyledon on day 7. The left section shows the orientation of the samples. Vascular bundles are labeled B1 and B2 based on their attachment to the first (L1) and second leaf (L2), respectively. The full video is in Supplemental Video S1. The transport velocity for each bundle is noted under each image. Bar = 200 µm. [See online article for color version of this figure.]
Independence of Vascular Bundles
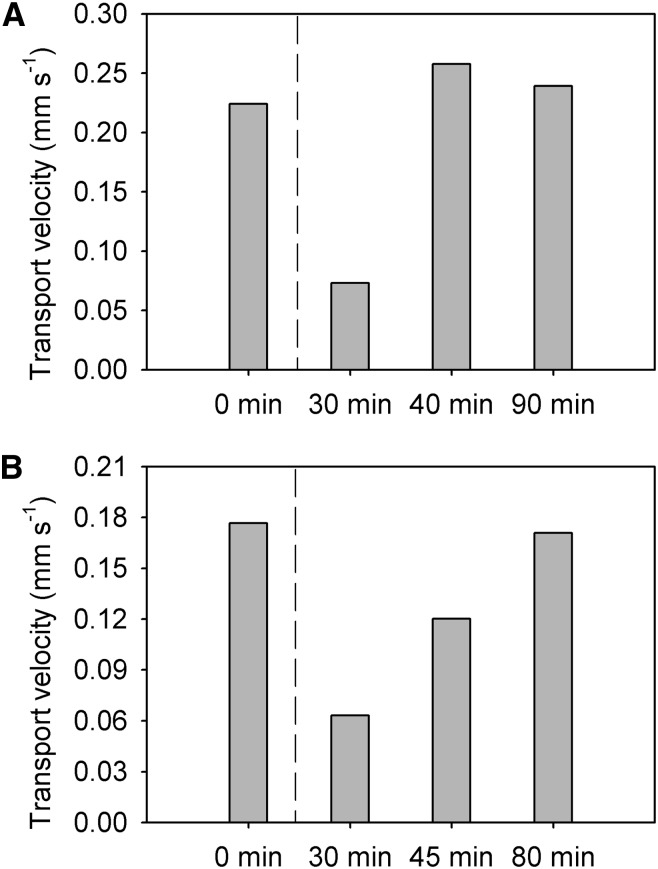
To maintain heterogeneous phloem transport within seedlings, vascular bundles need to operate relatively autonomously over short periods of time. This autonomy is apparent when examining the response of seedlings to damage and/or disturbance. When a cotyledon was cutoff while we were measuring an attached vascular bundle, phloem transport stopped. However, when a cotyledon was removed while measuring a vascular bundle attached to the other cotyledon, transport temporarily slowed but resumed premanipulation velocities within 40 min (Fig. 6A). This lag time is similar to the amount of time it takes a seedling to recover from a physical disturbance such as touching (Fig. 6B). Taken together, these data suggest that vascular bundles from different cotyledons operate independently over short periods of time and therefore provide further support that developmental changes could easily cause heterogeneity in phloem transport in seedlings depending on phloem connectivity.
Figure 6.
Reduction in phloem transport in response to physical disturbance. A, The cotyledon opposite to the measured vascular bundle was cutoff after the first measurement (dashed line). B, The seedling was agitated by shaking after the first measurement (dashed line). Note that seedlings recovered from both disturbances within 1 h.
DISCUSSION
Phloem transport velocity underwent a series of dynamic shifts that coincided with phenological changes during early cucumber seedling development (Figs. 1 and 3). Transport velocity also varied among vascular bundles, leading to significant heterogeneity in phloem transport within individuals, a signature of sectoriality (Table II). When the first true leaf began to export carbon, transport stabilized, reaching velocities similar to those measured in adult plants. These results demonstrate that, in cucumber seedlings, phloem transport is not regulated in a manner that maintains a constant velocity. Instead, we propose that changes in transport velocity during early seedling development occur because cucumber cotyledons cannot fix enough photosynthates to sustain growth in multiple sinks.
Leaf Development Slows Phloem Transport toward Roots Because of Carbon Limitation
After using carbon reserves in the seeds, cucumbers, similar to other epigeal species, depend on photosynthates produced in their cotyledons to support growth during early seedling development (Lovell and Moore, 1970; Penny et al., 1976). Carbon leaving the cotyledons is transported both basipetally toward the roots and acropetally toward the shoot apex. Cucumber cotyledons are attached to two vascular bundles that run the length of the hypocotyl, each of which connects to different developing leaves (Fig. 2B). Prior to the expansion of the first leaf, basipetal transport in the attached bundles declines, suggesting that carbon is diverted from the roots to the shoot apex (Figs. 1B and 3A). This carbon drain is alleviated when the leaf is manually removed, causing an increase in phloem transport toward the roots in the attached bundle (Fig. 3, B and C). These data indicate that developing leaves take priority over the roots, causing a shift in carbon allocation that results in slower basipetal transport.
The amount of carbon transported into a sink, which is typically referred to as sink strength, is not a property of the sink but the entire phloem network and will change depending on carbon availability, the number of sinks, and their respective activity (Farrar, 1996; Minchin and Thorpe, 1996). According to a model developed by Minchin et al. (1993), the addition of a new sink substantially affects transport into other unequivalent sinks only when carbon is limited. Under this scenario, sinks do not operate at saturation and therefore compete for available carbon. There are two main lines of evidence that the cucumber seedlings in this study were carbon limited. First, sink manipulation, both in terms of natural sink addition (growth of a new leaf) and subtraction (artificial leaf removal), had a direct effect on growth in other sinks (Figs. 1 and 3). Second, sink activity scaled with source area, responding directly to source manipulation. For example, a decrease in basipetal transport velocity occurred when cotyledons were shaded or cut (Table I).
Although the low light level used in this experiment likely contributed to the seedlings’ carbon limitation, previous work has found that cucumber seedlings are carbon limited when grown under high light (Penny et al., 1976), suggesting this limitation is not solely determined by growing conditions. A similar carbon limitation has been observed in seedlings of other species including Juglans regia (Escobar-Gutierrez et al., 1998) but this pattern is not ubiquitous (Schulz, 1994). Peas (Pisum sativum), different from cucumbers, can compensate for reduced cotyledon area, such that removal of one cotyledon does not cause a reduction in root growth because the remaining cotyledon increases its carbon export. One possible explanation for the difference between peas and cucumbers is the structure of their cotyledons. Peas exhibit hypogeal germination, meaning that their cotyledons primarily serve as belowground storage structures. Hypogeal species produce true leaves more quickly than epigeal species such as cucumbers (Lovell and Moore, 1971). Considering the low photosynthetic capacity of cotyledons (in cucumbers, it is one-tenth that of a fully mature leaf [Shibuya et al., 2010]), it is plausible that epigeal species are more carbon limited than hypogeal species because of their delayed leaf development. However, more research is needed to explicitly test whether there is a connection between cotyledon structure and early carbon use.
Shifts in carbon allocation in response to both source and sink manipulation are regulated by complex feedback mechanisms (Paul and Foyer, 2001) that allow plants to compensate for the removal of source area by altering photosynthetic activity and carbon storage (Thaine et al., 1959; Peel and Weatherley, 1962). Recent work has highlighted the potential role of sugar in regulating sink activity in Arabidopsis (Arabidopsis thaliana) seedlings by serving as a direct signal for root growth (Kircher and Schopfer, 2012) and by stimulating auxin biosynthesis (Lilley et al., 2012; Sairanen et al., 2012). With future work in this area, it may be possible to determine whether feedback loops can directly influence phloem transport and how sugar and auxin regulate sink activity in a variety of plant species.
New Carbon Source Increases Transport to Roots and Overall Carbon Availability
In the second week of development, the first new leaf begins to transition from sink to source tissue (Fig. 4), with the tip of the leaf fixing carbon before the base (Turgeon and Webb, 1973). This transition allows for a greater proportion of growth to occur in the roots and appears to coincide with higher basipetal phloem transport velocity (Fig. 3A). During this transition, the leaf begins to support its own growth and reduces its dependency on carbon from the cotyledons. As a result, its subsequent removal causes a reduction in both phloem transport toward the roots and seedling growth rate (Fig. 3, B and C). Previous research on Cucurbita pepo shows that young leaves begin fixing carbon when they are only 8% expanded (Turgeon and Webb, 1975) and export carbon when they are only 45% expanded (Turgeon and Webb, 1973). During this time, leaf cells begin producing enzymes involved in raffinose and stachyose synthesis, the sugars critical for phloem loading via polymer trapping (Turgeon and Webb, 1975; Pharr and Sox, 1984). In this study, the first leaf stopped importing carbon by day 11, when it was approximately 38% expanded.
After the first leaf started supporting its own growth, basipetal transport in the phloem plateaued around 0.16 ± 0.01 (se) mm s–1 (day 9; Fig. 3A). Transport after this point remained relatively stable, exhibiting only a minor drop prior to the development of the second true leaf (day 10). This apparent stability is not surprising considering that cucumber seedlings increase carbon fixation as they mature and become sink-limited adults (Mayoral et al., 1985). The average basipetal velocity in the phloem after day 9 was similar to velocities measured in adult cucumbers and other cucurbits, including Cucurbita maxima (0.19 mm s–1; Mullendore et al., 2010), C. pepo (0.18 m s–1; Crafts and Lorenz, 1944), and cucumber (0.15 mm s–1; Jensen et al., 2011). The consistency of these measurements is remarkable considering that they were determined using different methods (magnetic resonance imaging, growth and dye tracking, respectively) on plants grown under various environmental conditions. Considering this fact, it seems likely that there are anatomical and/or physiological constraints on phloem transport or sink growth in Cucurbitaceae that cause adult plants to transport at the same velocity.
Vascular Bundles Can Transport Sugar at Different Velocities
Early in development, variation in basipetal phloem transport was measured in different vascular bundles (Table II) and visually confirmed using fluorescent microscopy (Fig. 5; Supplemental Video S1). Some of this variation could be caused by differences in the timing of sieve tube development. For example, the second bundles exhibited slower basipetal transport on day 1 compared with day 2 (Fig. 3A), when there was no apparent change in carbon sources and sinks. However, anatomical development cannot explain all of the observed heterogeneity because all the bundles were capable of transporting at a similar velocity by day 4, a point that was demonstrated by the sink manipulation experiments (Fig. 3B). Therefore, we propose that bundles operate autonomously, and the observed heterogeneity is caused by differences in the sources and sinks attached to each bundle.
To further explore sectoriality in phloem transport, we examined how cutting off a cotyledon impacted transport in bundles not attached to the removed cotyledon. This disturbance only temporarily impacted transport in these bundles (Fig. 6A). The lag time for recovery was similar to the one observed when the seedling experienced a minor disturbance such as shaking (Fig. 6B). Taken together, these data support the idea that phloem transport within a plant is sectorial (Watson and Casper, 1984) and that future work examining whole plant transport needs to consider phyllotaxy and/or vascular connectivity.
It is important to note that cucumbers have bimorphic phloem (bicollateral fascicular phloem that flanks the xylem and extrafascicular phloem in the cortex). The phloem tracer (CF) does not produce a strong signal in the extrafascicular phloem, and evidence suggests that this type of phloem is not involved in sugar transport (Turgeon and Oparka, 2010; Zhang et al., 2010). The primary source of the signal detected in this study is from the external fascicular phloem because the internal fascicular phloem has a small translocation area and is deeper in the tissue. Moreover, the internal phloem does not appear to extend the length of the hypocotyl in seedlings, terminating prematurely about 1 cm above the soil (a similar observation was made by Fischer [1883]). Because the extrafascicular and the internal fascicular phloem were not monitored in this study, it is possible that more variation exists in transport velocity within the stem than reported here.
CONCLUSION
Seedling physiology is critical to plant establishment and has important implications for species’ distributions in their natural environments (Kitajima, 2002; Savage and Cavender-Bares, 2012) and their success in agriculture (Pujol et al., 2005). In this study, we demonstrate that phloem transport responds dynamically to changes in sink and source development and that transport velocity within a single plant can be heterogeneous. As cucumber seedlings age, their phloem network expands and transport stabilizes at velocities similar to those experienced in adult plants, a change that may coincide with the transition from being source to sink limited. Altogether, these results indicate that in cucumber seedlings, transport is not regulated at the level of velocity, but instead, transport velocity is determined by source-sink relations. More research is needed to understand the extent to which the observed patterns are species specific and to clarify how carbon allocation might differ among young seedlings of different species.
MATERIALS AND METHODS
Plant Material and Growth Conditions
Cucumbers (Cucumis sativus) of the variety Marketmore 76 were grown under laboratory conditions (average temperature, 22.7°C ± 0.3°C [se] and humidity, 31.7% ± 3.7% [se]) in an all-purpose potting soil composed of sphagnum moss, perlite, and vermiculite (Sunshine Mix #1, Sungro). Each seedling was grown in a separate cell of a 28-cell flat and during the first week received 15 to 20 mL of water every other day. Watering was increased to about 20 to 25 mL per plant (every other day) during the second week. Seedlings were illuminated with a fluorescent light at a low light level (approximately 30 μmol m–2 s–1). Under these conditions, the seedlings were slightly etiolated and had hypocotyls that were over 3 cm long, which is the minimum length required for our measurements. Seedling development was monitored for 2 weeks following cotyledon unfolding (day 0). Three of the seedlings were moved from the lab to a greenhouse and allowed to mature for a few months. Lights were kept on in the greenhouse for 12 h, and the temperature was kept between 19°C and 26°C.
Seedling Growth and Carbon Budget
Four sets of 10 seedlings were harvested every day for 2 weeks. The length of the translocation pathway (roots and hypocotyl), the area of source material (cotyledon and true leaves), and the dry weight of each organ (dried at least 48 h at 65°C) were measured for each seedling. A set of 10 dried seedlings was sent to the Earth System Center for Stable Isotopic Studies at Yale University to determine the total carbon content of root, stem, cotyledon, bud, and true leaf material. For bud and root material, more than 10 plants were sampled because of their small mass.
Gas exchange was measured on the cotyledons of 10 seedlings for 1 week (days 2 to 9) using a LI-COR 6400 with a leaf chamber fluorometer. Measurement conditions were as follows: light level, 30 μmol m–2 s–1 (same as growth conditions); reference CO2, 400 μmol mol–1 (ambient level in laboratory); leaf temperature, 20.0°C ± 0.16°C (sd); vapor pressure deficit, 1.06 ± 0.06 kPa (sd). Daily measurements were taken at 2 pm except on day 7, when measurements were taken every 2 h from 8 am to 6 pm. On day 9, measurements were also taken on the first true leaf. These data, along with cotyledon and leaf area measurements, were used to estimate daily carbon fixation. Because rates of carbon assimilation likely change as leaves expand, there is some bias in estimates of carbon fixation and respiration. However, these estimates are of a similar magnitude to those reported in an earlier study on cucumber seedlings (Halevy, 1964).
Measuring Phloem Velocity
Phloem transport velocity was measured by tracking the movement of a phloem-mobile, fluorescent tracer (CF dye) from the cotyledons to the roots in intact seedlings using a high-gain photodiode with a method modified from Jensen et al. (2011). The tracer was introduced into the phloem by applying 50 μmol of 0.01 m CF diacetate in 1:10 mixture of acetone and distilled, deionized water to a cotyledon after gently abrading its adaxial surface with fine-grain sandpaper (500 grit). CF diacetate is a membrane-permeant form of the dye that is readily converted to CF by esterases inside living cells (Rotman and Papermaster, 1966; Oparka and Read, 1994). After the dye was applied, the site of application was covered with a thin piece of plastic to prevent desiccation.
A small clip was attached to the base of the hypocotyl to hold one end of a bifurcating fiber-optic cable close to a vascular bundle (Fig. 2A). The second end of the cable was attached to a light source with a 20-mA narrow-band light-emitting diode (470 nm, B5B-437-IX, Roithner Last Technik) filtered by a 472-nm narrow band-pass filter (NT67-027, Edmund Optics). The unified end of the cable was connected to a high-gain photodiode (SHD033, International Light Instruments) with a 535-nm narrow band-pass filter (NT67-031, Edmund Optics). Data from the photodiode was logged using a program created in LabVIEW 2010 (National Instruments) that was designed to interface between the computer and the radiometer (ILT1700, International Light Instruments).
Velocity was determined by bleaching the dye in the hypocotyl for 6 to 8 s with a 473-nm diode-pumped solid-state laser (BML-473-20FEB, 20 mW, Lasermate) positioned 15 to 30 mm above the fiber-optic cable. Velocity was calculated based on the distance between the laser point and the center of the photodiode and the time it took for the bleach front to travel between the two points (Fig. 2A). Measurements were taken during a 2- to 3-h window when dye levels were high enough to bleach but not high enough to be saturated. This allowed for two to three measurements per plant. Individual vascular bundles were targeted with the tip of the fiber-optic cable, and the exact location of the bundles was noted (Fig. 2C). Initially, the orientation of the sensor was checked by injecting safranin dye into the hypocotyl with a syringe through the clip after each measurement, but this proved unnecessary because of the predictability of the vascular arrangement in the seedlings. Plants were measured under an E.shine LED Grow Light (300 W) at a light level of 100 μmol m–2 s–1 between 8 am and 6 pm (daylight hours). There was no evidence of any diurnal changes in phloem transport during this time period. Velocity measurements were also taken on one petiole of three mature cucumber plants under the same conditions. In general, making successful measurements appeared to depend on the anatomy of the plant (straightness of the bundles and thickness of cortical tissue), the precise angle of the sensor and laser, and the timing of dye movement. All of these factors and the general sensitivity of seedlings to physical disturbance made it difficult to measure more than one to two plants a day. Cucumbers proved to be an ideal species for this study because the fluorescent tracer was easily detected through their semitranslucent stems.
Phloem transport velocity was measured on multiple cohorts of seedlings until a sampling of five to six seedlings per day per pair of vascular bundles was achieved. Vascular bundles were numbered (1 or 2) depending on whether they were connected to the first or second developing leaf (Fig. 2B). The sensitivity of plants to disturbance and the positioning of their vascular tissue made it challenging to measure more than one vascular bundle on each plant. As result, every measurement was made on a separate individual, except on three occasions when we were able to measure two bundles on the same plant. This was achieved by positioning the sensor between two bundles and moving the laser to target each bundle individually. Additionally, on day 7, transport was directly observed in a set of six seedlings on a Zeiss AxioZoom.V16 using a X1 lens with a GFP filter (filter set 38). Images were made with an AxioCam MRm using the Zeiss program Zen Blue 2012. The seedlings were prepared the day before by carefully removing a layer of the outer cortex with a razor blade and placing a cover slip over the exposed tissue. After application of CF, time-lapse imaging was used to document the movement of the dye into the hypocotyl.
Source and Sink Manipulation Experiments
To test the effect of sink manipulation on developing seedlings, phloem velocity was measured on four to five seedlings that had their first leaf primordium removed (cut at the base of the petiole) 1 d prior to measurement. Measurements were taken on days 4, 5, 6, 8, 9, 10, and 12. Based on preliminary tests, leaf removal did not affect bundles that were not directly connected to the first developing leaf (second bundle), and therefore, measurements were only made on bundles attached to this leaf (first bundle). A similar manipulation was used to determine the effect of leaf removal on growth on day 7 and day 10. Sampling of biomass was as described above and was completed on four sets of 10 plants for each day of treatment.
To examine the impact of source area on phloem transport, cotyledon area was decreased either by cutting or shading one cotyledon per plant on a set of eight plants. For the cutting treatment, the cotyledon was cut in half transversely with a razor blade 1 d prior to taking measurements. For the shading treatment, it was covered with a black piece of paper for 3 d prior to taking the measurements. In both treatments, transport velocity was measured in the bundles attached to the second true leaf (second bundle). Measurements were taken before the second leaf began to expand (day 5) when there was only one primary sink (the roots) for the attached vascular bundles.
Phloem Anatomy and Leaf Development
Several aspects of seedling development were examined by tracking the movement of CF within seedlings. First, connections between different carbon sources (cotyledons and leaves) and individual vascular bundles were examined by tracing the movement of the dye through the hypocotyl at different stages of development. Second, six seedlings were harvested every other day of development to examine average sieve tube lumen area in each vascular bundle. Measurements were only made on translocating tubes that were identified by the presence of CF dye. Third, the transition of the first true leaf from sink to source material was monitored by tracking the movement of CF into its veins (Roberts et al., 1997). The dye should only infiltrate into parts of the leaf that serve as strong sinks. One to 1.5 h after CF was applied to both cotyledons, young leaves were imaged on a Zeiss AxioZoom.V16 with a GFP filter (see above for more details). Based on the transport velocity measurements and preliminary tests, this was enough time for the dye to infiltrate the leaves.
Independence of Vascular Bundles
Small disturbance can cause phloem transport to stop or slow down temporarily, and larger disturbances can cause more severe wounding responses, including the occlusion of sieve tubes (Knoblauch and Peters, 2010). To examine whether disrupting transport in one vascular bundle effects transport in nonconnected bundles, three disturbance tests were conducted: (1) removal of the cotyledon attached to the bundle being measured, (2) removal of the cotyledon opposite to the bundle being measured, and (3) agitation of the seedling by gently shaking it. After each treatment, phloem velocity was measured repeatedly for about 2 h. Because it is generally rare that measurements can be made continuously for 2 h (see above), these experiments were not replicated.
Supplemental Material
The following materials are available in the online version of this article.
Supplemental Video S1. Basipetal transport of a fluorescent dye (CF) in two vascular bundles in the hypocotyl that are attached to the same cotyledon of a cucumber seedling.
Acknowledgments
We thank Kaare Jensen for his feedback on the experiment and Dustin Haines and two anonymous reviewers for comments on the manuscript.
Glossary
- CF
carboxyfluorescein
References
- Crafts AS, Lorenz OA. (1944) Fruit growth and food transport in cucurbits. Plant Physiol 19: 131–138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Escobar-Gutierrez AJ, Daudet FA, Guadillère JP, Maillard P, Frossard JS. (1998) Modelling of allocation and balance of carbon in walnut (Juglans regia L.) seedlings during heterotrophy-autotrophy transition. J Theor Biol 194: 29–47 [DOI] [PubMed] [Google Scholar]
- Farrar JF. (1996) Sinks—integral parts of a whole plant. J Exp Bot 47: 1273–1279 [DOI] [PubMed] [Google Scholar]
- Fischer A. (1883) Das siebröhrensystem von Cucurbita. Ber Dtsch Bot Ges 1: 276–279 [Google Scholar]
- Froelich DR, Mullendore DL, Jensen KH, Ross-Elliott TJ, Anstead JA, Thompson GA, Pélissier HC, Knoblauch M. (2011) Phloem ultrastructure and pressure flow: Sieve-Element-Occlusion-Related agglomerations do not affect translocation. Plant Cell 23: 4428–4445 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halevy AH. (1964) Effects of gibberellin and growth-retarding chemicals on respiration and catalase activity in various organs of cucumbers seedlings. J Exp Bot 15: 546–555 [Google Scholar]
- Haritatos E, Keller F, Turgeon R. (1996) Raffinose oligosaccharide concentrations measured in individual cell and tissue types in Cucumis melo L. leaves: implications for phloem loading. Planta 198: 614–622 [DOI] [PubMed] [Google Scholar]
- Jensen KH, Lee J, Bohr T, Bruus H, Holbrook NM, Zwieniecki MA. (2011) Optimality of the Münch mechanism for translocation of sugars in plants. J R Soc Interface 8: 1155–1165 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kallarackal J, Bauer SN, Nowak H, Hajirezaei MR, Komor E. (2012) Diurnal changes in assimilate concentrations and fluxes in the phloem of castor bean (Ricinus communis L.) and tansy (Tanacetum vulgare L.). Planta 236: 209–223 [DOI] [PubMed] [Google Scholar]
- Kircher S, Schopfer P. (2012) Photosynthetic sucrose acts as cotyledon-derived long-distance signal to control root growth during early seedling development in Arabidopsis. Proc Natl Acad Sci USA 109: 11217–11221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kitajima K. (2002) Do shade-tolerant tropical tree seedlings depend longer on seed reserves? Functional growth analysis of three Bignoniaceae species. Funct Ecol 16: 433–444 [Google Scholar]
- Knoblauch M, Peters WS. (2010) Münch, morphology, microfluidics - our structural problem with the phloem. Plant Cell Environ 33: 1439–1452 [DOI] [PubMed] [Google Scholar]
- Larson P, Dickson R. (1973) Distribution of imported 14C in developing leaves of eastern cottonwood according to phyllotaxy. Planta 111: 95–112 [DOI] [PubMed] [Google Scholar]
- Lilley JLS, Gee CW, Sairanen I, Ljung K, Nemhauser JL. (2012) An endogenous carbon-sensing pathway triggers increased auxin flux and hypocotyl elongation. Plant Physiol 160: 2261–2270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lovell PH, Moore KG. (1970) A comparative study of cotyledons as assimilatory organs. J Exp Bot 21: 1017–1030 [Google Scholar]
- Lovell PH, Moore KG. (1971) A comparative study of the role of the cotyledon in seedling development. J Exp Bot 22: 153–162 [Google Scholar]
- Marcelis LFM. (1996) Sink strength as a determinant of dry matter partitioning in the whole plant. J Exp Bot 47: 1281–1291 [DOI] [PubMed] [Google Scholar]
- Marshall C. (1996) Sectoriality and physiological organisation in herbaceous plants: an overview. Vegetatio 127: 9–16 [Google Scholar]
- Mayoral ML, Plaut Z, Reinhold L. (1985) Effect of translocation-hindering procedures on source leaf photosynthesis in cucumber. Plant Physiol 77: 712–717 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minchin PEH, Thorpe MR. (1996) What determines carbon partitioning between competing sinks? J Exp Bot 47: 1293–1296 [DOI] [PubMed] [Google Scholar]
- Minchin PEH, Thorpe MR. (2003) Using the short-lived isotope 11C in mechanistic studies of photosynthate transport. Funct Plant Biol 30: 831–841 [DOI] [PubMed] [Google Scholar]
- Minchin PEH, Thorpe MR, Farrar JF. (1993) A simple mechanistic model of phloem transport which explains sink priority. J Exp Bot 44: 947–955 [Google Scholar]
- Mitchell DE, Gadus MV, Madore MA. (1992) Patterns of assimilate production and translocation in muskmelon (Cucumis melo L). 1. Diurnal patterns. Plant Physiol 99: 959–965 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mullendore DL, Windt CW, Van As H, Knoblauch M. (2010) Sieve tube geometry in relation to phloem flow. Plant Cell 22: 579–593 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Münch E (1930) Die Stoffbewegungen in der Pflanze. Gustav Fischer, Jena, Germany [Google Scholar]
- Oparka KJ, Davies HV. (1985) Translocation of assimilates within and between potato stems. Ann Bot (Lond) 56: 45–54 [Google Scholar]
- Oparka KJ, Read ND (1994) The use of fluorescent probes for studies of living plant cells. In N Harris, KJ Oparka, eds, Plant Cell Biology. Oxford University Press, Oxford, pp 27–50 [Google Scholar]
- Paul MJ, Foyer CH. (2001) Sink regulation of photosynthesis. J Exp Bot 52: 1383–1400 [DOI] [PubMed] [Google Scholar]
- Peel AJ, Weatherley PE. (1962) Studies in sieve-tube exudation through aphid mouth-parts: the effects of light and girdling. Ann Bot (Lond) 26: 633–646 [Google Scholar]
- Penny MG, Moore KG, Lovell PH. (1976) Effects of inhibition of cotyledon photosynthesis on seedling development in Cucumis sativus L. Ann Bot (Lond) 40: 815–824 [Google Scholar]
- Pharr DM, Sox HN. (1984) Changes in carbohydrate and enzyme levels during the sink to source transition of leaves of Cucumis sativus L., a stachyose translocator. Plant Sci Lett 35: 187–193 [Google Scholar]
- Pujol B, Mühlen G, Garwood N, Horoszowski Y, Douzery EJP, McKey D. (2005) Evolution under domestication: contrasting functional morphology of seedlings in domesticated cassava and its closest wild relatives. New Phytol 166: 305–318 [DOI] [PubMed] [Google Scholar]
- Rennie EA, Turgeon R. (2009) A comprehensive picture of phloem loading strategies. Proc Natl Acad Sci USA 106: 14162–14167 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts AG, Cruz SS, Roberts IM, Prior D, Turgeon R, Oparka KJ. (1997) Phloem unloading in sink leaves of Nicotiana benthamiana: comparison of a fluorescent solute with a fluorescent virus. Plant Cell 9: 1381–1396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rotman B, Papermaster BW. (1966) Membrane properties of living mammalian cells as studied by enzymatic hydrolysis of fluorogenic esters. Proc Natl Acad Sci USA 55: 134–141 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sairanen I, Novák O, Pěnčík A, Ikeda Y, Jones B, Sandberg G, Ljung K. (2012) Soluble carbohydrates regulate auxin biosynthesis via PIF proteins in Arabidopsis. Plant Cell 24: 4907–4916 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savage JA, Cavender-Bares JM. (2012) Habitat specialization and the role of trait lability in structuring diverse willow (genus Salix) communities. Ecology 93: S138–S150 [Google Scholar]
- Schulz A. (1994) Phloem transport and differential unloading in pea seedlings after source and sink manipulations. Planta 192: 239–248 [Google Scholar]
- Sharkey PJ, Pate JS. (1976) Translocation from leaves to fruits of a legume, studied by a phloem bleeding technique: diurnal changes and effects of continuous darkness. Planta 128: 63–72 [DOI] [PubMed] [Google Scholar]
- Shibuya T, Endo R, Kitamura Y, Kitaya Y. (2010) Potential photosynthetic advantages of cucumber (Cucumis sativus L.) seedlings grown under fluorescent lamps with high red:far-red light. HortScience 45: 553–558 [Google Scholar]
- Sprugel DG, Hinckley TM, Schaap W. (1991) The theory and practice of branch autonomy. Annu Rev Ecol Syst 22: 309–334 [Google Scholar]
- Thaine R, Ovenden SI, Turner JS. (1959) Translocation of labelled assimilates in the soybean. Aust J Biol Sci 12: 349–372 [Google Scholar]
- Turgeon R (1991) Symplastic phloem loading and the sink-source transition in leaves: a model. In JL Bonnemain, S Delrot, WJ Lucas, J Dainty, eds, Recent Advances in Phloem Transport and Assimilate Compartmentation. Ouest Editions/Presses Academiques, Nantes, France, pp 18–22 [Google Scholar]
- Turgeon R, Oparka K. (2010) The secret phloem of pumpkins. Proc Natl Acad Sci USA 107: 13201–13202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turgeon R, Webb JA. (1973) Leaf development and phloem transport in Cucurbita pepo: transition from import to export. Planta 113: 179–191 [DOI] [PubMed] [Google Scholar]
- Turgeon R, Webb JA. (1975) Leaf development and phloem transport in Cucurbita pepo: carbon economy. Planta (Berlin) 123: 53–62 [DOI] [PubMed] [Google Scholar]
- Watson MA, Casper BB. (1984) Morphogenetic constraints on patterns of carbon distribution in plants. Annu Rev Ecol Syst 15: 233–258 [Google Scholar]
- Windt CW, Vergeldt FJ, de Jager PA, van As H. (2006) MRI of long-distance water transport: a comparison of the phloem and xylem flow characteristics and dynamics in poplar, castor bean, tomato and tobacco. Plant Cell Environ 29: 1715–1729 [DOI] [PubMed] [Google Scholar]
- Zhang BC, Tolstikov V, Turnbull C, Hicks LM, Fiehn O. (2010) Divergent metabolome and proteome suggest functional independence of dual phloem transport systems in cucurbits. Proc Natl Acad Sci USA 107: 13532–13537 [DOI] [PMC free article] [PubMed] [Google Scholar]