Abstract
Detecting threats and escaping before serious confrontations are important for animals to avoid danger and death. Transient receptor potential ankyrin 1 (TRPA1), a member of the TRP superfamily, is expressed in a subset of sensory neurons and mediates nociception evoked by pungent chemicals. Using behavioral testing, we found that TRPA1 knockout mice failed to avoid entering a chamber filled with vapor of formalin, allyl isothiocyanate, and acrolein. The avoidance behavior was blocked by nasal but not subcutaneous administration of a blocker to TRPA1. We also found that TRPA1 knockout mice did not wake when exposed to formalin during sleep. Additionally, the spinal trigeminal nucleus, the first relay neurons of the trigeminal system, showed massive expression of c-Fos after a brief (3 min) exposure to formalin vapor. TRPA1 seems to be a sentinel for environmental chemicals and induces avoidance behaviors and waking by way of the trigeminal system.
Detecting threats and escaping before serious confrontations are important for animals to avoid danger and death. Transient receptor potential ankyrin 1 (TRPA1), a member of the TRP superfamily, is implicated in nociception of diverse environmental stimuli, including pungent chemicals1,2,3. Irritants, including tobacco smoke4,5 and hyperoxic gas6, activate TRPA1 in the airways and reduce respiratory frequency to protect against further inhalation. TRPA1 is also implicated in the nociception of formalin-induced pain, a model used for evaluating analgesic compounds in laboratory animals7. Despite an increase in TRPA1-related studies, the behavioral consequences of TRPA1 activation, especially in the upper airway, are still unclear.
We hypothesized that activation of TRPA1 would induce not only passive avoidance by reducing respiration but also active avoidance (escape behavior) from an airborne threat stimulus. Our hypothesis is plausible because TRPA1 is expressed in the trigeminal sensory nerves2 and olfactory receptor neurons8, which innervate the nasal cavity. Environmental gas first contacts sensory receptors in the nasal cavities before reaching the laryngo-tracheal and lung regions where TRPA1 induces respiratory arrest3.
Results
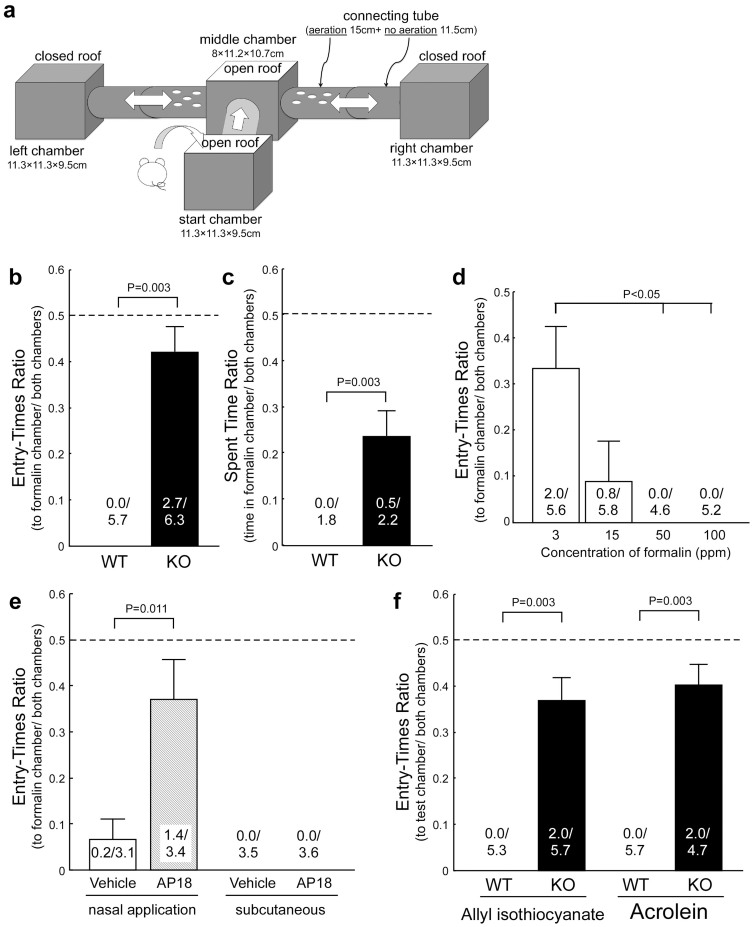
To test our hypothesis, we performed an avoidance/preference test using a homemade apparatus (Fig. 1a) on TRPA1 knockout (KO) mice9 and age-matched wild-type (WT) littermates. During a 5-min test period, WT mice would not enter the apparatus chamber containing 100 ppm formalin (Fig. 1b and c), although they approached the entrance. Avoidance behavior depended on the formalin vapor concentration (Fig. 1d). KO mice entered the chamber without hesitation (Fig. 1b) and even stayed there, but the ratio of time spent was lesser than the chance level of 0.5 (Fig. 1c). The same result was obtained when the test was repeated or prolonged (data not shown).
Figure 1. Avoidance of a chamber filled with chemical vapor.
(a) Homemade testing apparatus for avoidance/preference behavior. (b, c) WT mice (n = 6) never entered the chamber with formalin (100 ppm) while TRPA1 KO mice (n = 6) entered without hesitation. Numbers at bottom of columns indicate absolute values averaged by group and used in ratio calculations. (d) Dose-response relationship of WT mice (n = 5 per dose) to enter the formalin chamber. (e) Nasal (n = 10 for both vehicle and blocker) but not subcutaneous (n = 6) administration of AP18 blocked avoidance to 50 ppm formalin in WT mice. (f) Similar abnormality in KO mice was observed when formalin was replaced by other TRPA1 agonists allyl isothiocyanate or acrolein (n = 6 per group). Error bars indicate s.e.m.
We next examined whether avoidance could be blocked in WT mice by locally blocking TRPA1 in the nasal cavity. Five minutes before behavioral testing, a specific blocker of TRPA1 (AP18) or vehicle was administered into the nasal cavity or subcutaneous space in the back. Only nasal administration of AP18 significantly reduced avoidance (Fig. 1e). We also tested other irritants that are known TRPA1 agonists (allyl isothiocyanate and acrolein)4. As with formalin, avoidance of these irritants disappeared in KO mice (Fig. 1f).
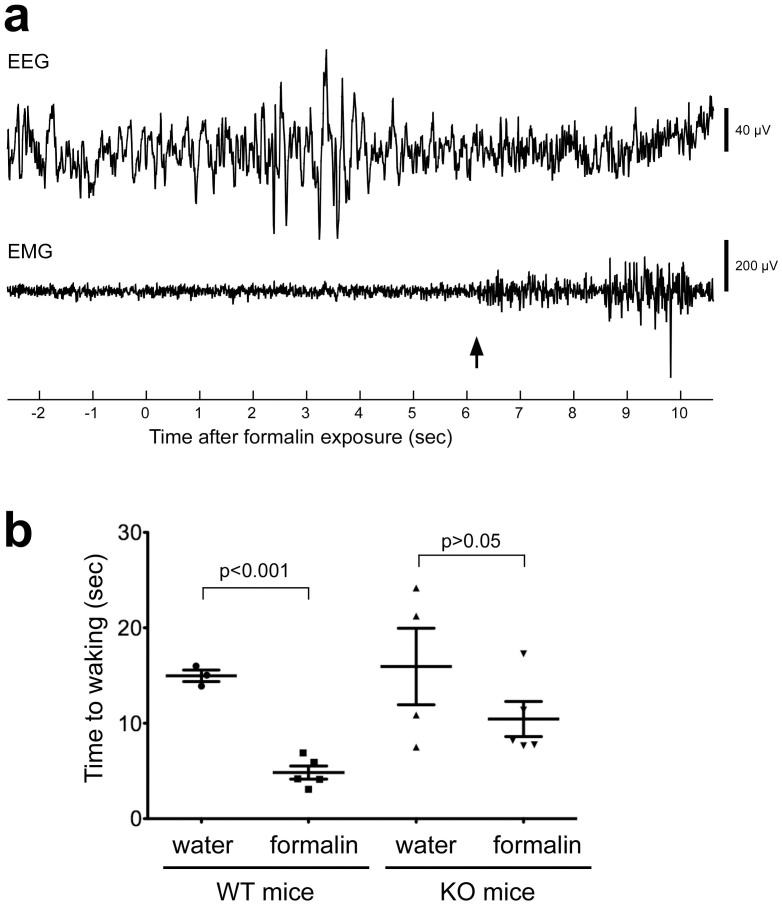
These results show that TRPA1 in the nasal cavity contributes to avoidance behavior in awake, feely-moving mice, indicating that TRPA1 may act as an alarm system. This prompted us to examine another type of alarm: waking from sleep induced by approaching threat. We used WT and KO mice that had EEG and EMG electrodes implanted10 and examined whether exposure to formalin vapor would wake them (Fig. 2a) more rapidly than exposure to water vapor. As expected, formalin-induced arousal was not observed in KO mice, while formalin exposure significantly accelerated wake times in WT mice (from 15.0 ± 0.6 s to 4.8 ± 0.7 s; Fig. 2b).
Figure 2. Wake response to formalin vapor.
(a) Typical EEG and neck EMG from WT mouse exposed to formalin. After natural sleep for more than 1 min, inflow to the test chamber was switched to gas containing formalin vapor, denoted as time 0. Waking time is marked by an arrow. (b) Grouped data from WT mice exposed to water vapor (n = 3) and formalin vapor (n = 5), and TRPA1 KO mice exposed to water vapor (n = 4) and formalin vapor (n = 5). Data from 3 trials in a single mouse was averaged and treated as the value for the mouse. Error bars indicate s.e.m.
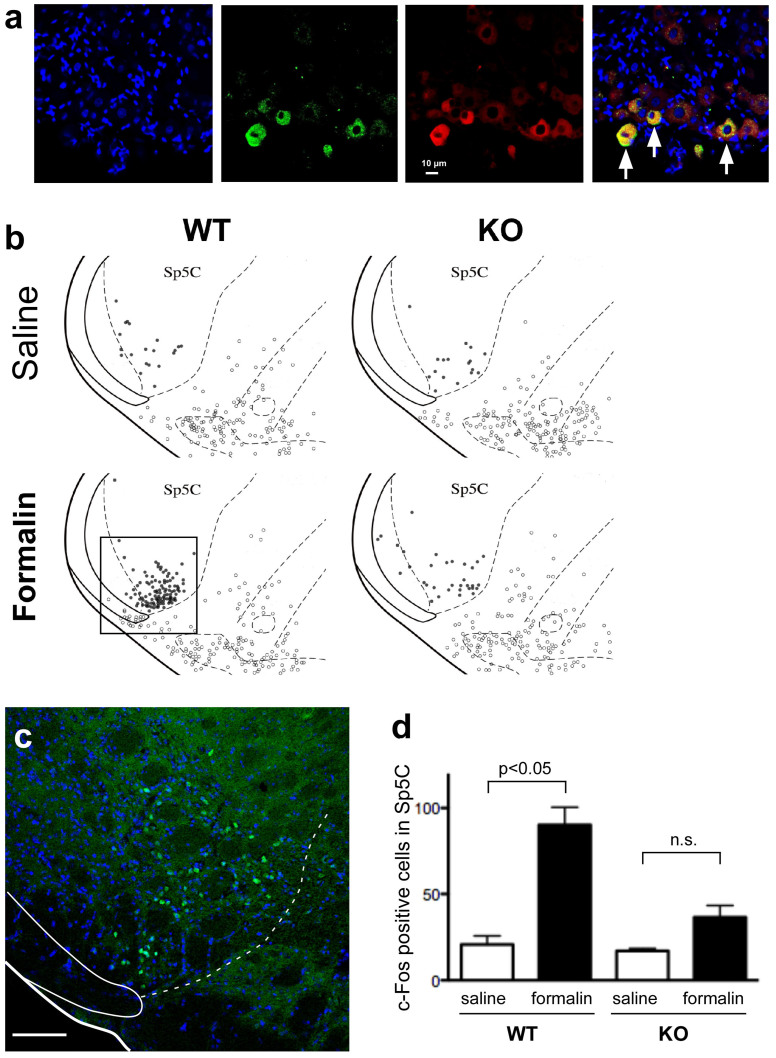
Although TRPA1 is known to exist in trigeminal ganglion neurons2, their projection areas have not been confirmed. Therefore, we examined whether the ethmoidal nerve, a branch of the trigeminal nerve innervating the nasal cavity, expresses TRPA1. As expected, a subset of nasally administered retrograde tracer (DiI)-positive ganglion cells also expressed TRPA1 (Fig. 3a).
Figure 3. Histological evidence showing trigeminal system involvement.
(a) Trigeminal nerve innervating the nasal cavity expresses TRPA1. Left to right: nuclear staining with Hoechst 33258; TRPA1-like immunoreactivity in the trigeminal ganglion; nasally administered DiI-stained neurons; merged image. Arrows indicate double-stained neurons. Consistent results were obtained in all 3 mice examined. (b) Schematic diagrams showing the distribution of c-Fos-positive cells within (closed circle) and outside (open circle) the spinal trigeminal nucleus caudalis (Sp5C) after brief exposure to formalin vapor. Distribution of c-Fos in 3 sections overlaid in a single diagram. Diagram representative of 1 mouse in each group. (c) Typical photograph taken from WT mouse exposed to formalin vapor. Corresponding area shown as a rectangle in (b). Bar = 100 μm. (d) Numbers of c-Fos-positive cells in Sp5C of WT and TRPA1 KO mice (n = 4 per group). Error bars indicate s.e.m.
Finally, we examined whether exposure to formalin vapor activates secondary sensory neurons in the trigeminal system. A brief (3 min) exposure to formalin increased c-Fos-positive cells in the spinal trigeminal nucleus caudalis of WT but not KO mice (Fig. 3b–d). Therefore, TRPA1 may be involved in early activation of trigeminal neurons in response to formalin exposure.
Discussion
Our results indicate that WT mice detect and recognize formalin, allyl isothiocyanate, and acrolein as dangerous substances via TRPA1. This is the first report showing that stimulation of TRPA1 induces active avoidance behaviors, in addition to respiratory arrest (passive avoidance behavior) and pain sensation. TRPA1 responsible for this active avoidance may be located in the ethmoidal and/or olfactory nerve terminals in the nasal cavity, because only nasal administration of AP18 blocked avoidance in WT mice. We cannot exclude the possible contribution of TRPA1 in other organs (e.g., other parts of the respiratory tract) since we did not check efficiency of subcutaneously administered AP18 in blocking any neurotransmission. However, the dose we used (~200 μmol/kg) was much higher than the reported dose (47.7 μmol/kg, i.p.) that effectively blocked pain-related behavior induced by TRPA1 activation in the plantar region11. Therefore, we can say that nasal TRPA1 is plausible to induce the active avoidance behavior. We cannot exclude the possible involvement of the olfactory system in the observed behavior, but the trigeminal system seems at least partially responsible. Although sensation to pungent chemicals has been reported to mainly depend on the trigeminal nerve12, there is an interaction between trigeminal and olfactory sensations13. Therefore, in combination with the olfactory system, trigeminal TRPA1 likely underlies the waking reaction to allyl isothiocyanate-containing substances14 and may be relevant to sick building syndrome.
Methods
Animals
TRPA1 knockout (KO) mice were purchased from the Jackson Laboratory and genotyped as previously described9. They were maintained as heterozygotes in our facility and crossed to obtain null mutants and wild-type (WT) littermates (male, 20–30 weeks old). All experimental procedures were performed in accordance with the guiding principles for the care and use of animals in the field of physiological sciences published by the Physiological Society of Japan (2003) and approved by the Institutional Animal Use Committees at Kagoshima University.
Homemade avoidance test apparatus
Commercially available avoidance/preference test apparatuses are composed of 2 or 3 chambers that are closely connected, with doors located between the chambers. One problem, however, is that formalin vapor can easily leak between chambers in such an apparatus. To minimize vapor leakage, we made a specially designed apparatus (Fig. 1a) for this study. Three chambers were connected with half mesh tubes so that any leaked vapors from the side chamber would not affect the gas component in the chambers at the opposite side and in the middle. When a side chamber was filled with 50 ppm formalin vapor, the formalin concentration was <1 ppm in the middle chamber and 5–8 ppm at the junction of the closed and open parts of the connection tube (see Fig. 1a). We added an entry chamber located at a right angle to the middle chamber, so that there would be no directional bias when a mouse was first put into the apparatus. Once a mouse moved into the middle chamber from the entry chamber, the door between the 2 chambers was closed. The number of entries and the amount of time spent in each side chamber were recorded. Entry-times ratio was the numbers of entry into one chamber divided by the total numbers of entry into both side chambers. Spent-time ratio was the amount of time spent in one chamber divided by the total amount of time spent in both chambers.
The mice were first allowed to freely explore the apparatus for acclimatization. On the other day, we first tested whether the mice preferred a specific side chamber while both chambers were free of vapor. None of the animals had an initial bias for either chamber. A few days after the initial session using empty chambers, an avoidance test was carried out. The left or right chamber was randomly selected as to whether a piece of cotton paper (1 cm × 1 cm) soaked with a test solution (0.5–20 μL) or distilled water (20 μL) was placed. After 3 min to allow for formalin to fully vaporize, the mouse was put into the entry chamber and behavior was observed. All behavioral tests were performed between 1600 and 2000.
Chemicals and determination of gas concentration
Solutions of formalin (37% formaldehyde), allyl isothiocyanate (AITC, 98%), and acrolein (10 mg/mL) were purchased from Nakarai Chemicals (Tokyo, Japan). A TRPA1 selective blocker, (Z)-4-(4-chlorophenyl)-3-methylbut-3-en-2-oxime (AP18), was synthesized at Kyoto University6. AP18 was dissolved in polyethylene glycol (PEG #400, Nakarai). Animals received an injection of 100 μL of AP18 (60 mM) or vehicle.
Concentration of formalin and acrolein vapor was determined using the detector tube method (Gastec Corp., Ayase, Japan). Concentration of AITC vapor was not measured because a specific detector tube for it was not available. Dose of acrolein (10 μL, <3.3 ppm; below detection threshold) and AITC (0.5 μL) was determined by preliminary testing to be the minimum dose at which WT mice would never enter the test chamber.
Waking experiment
Electrodes for EEG (parietal lobes) and EMG (neck muscles) were implanted under isoflurane (2–3%) anesthesia. After a 1-week recovery period, mice were acclimatized to the measuring chamber. Mice were gently placed in the chamber (a 750-mL container with inflow and outflow gas tubing and water supply) that was continuously flushed with room air (500 mL/min) for 3 h. This procedure was repeated every other day for 1 week (3 times in total). On the experimental day, mice were put into the chamber and EEG and EMG signals were fed into amplifiers through a slip-ring to allow freedom of movement during the experiment. EEG and EMG signals were continuously monitored on a computer screen. When spontaneous non-rapid-eye-movement sleep (low-frequency and high-amplitude EEG and low-amplitude EMG) was observed for more than 1 min, inflow to the chamber was switched to a gas containing either formalin (30 ppm) or water vapor. Waking time was judged by changes in EEG (from low frequency and high amplitude to high frequency and low amplitude) and EMG (from low amplitude to high amplitude). All recordings were obtained over 6 h between 1000 and 1600, which is the resting period for nocturnal mice.
Retrograde tracing experiment
One week after the administration of a tracer, DiI (0.025 mg/mouse; D282, Molecular Probes, Eugene, OR, USA), into the nasal cavity of WT mice (n = 3), the animals were deeply anesthetized by an injection of urethane (1.6 g/kg, i.p.) and transcardially perfused with 0.01 M phosphate-buffered saline (PBS) followed by a fixative solution containing 4% paraformaldehyde in PBS. The trigeminal ganglions were dissected and post-fixed in the same fixative solution for 24 h at 4°C. After cryoprotection with 30% sucrose in PBS, serial transverse frozen sections (40 μm) were cut and immunohistochemically stained for TRPA1. Anti-TRPA1 antibody was a gift from Y. Kubo15. It was visualized with Alexa Fluor 488-conjugated anti-rabbit IgG (1/200, Molecular Probes).
Immunohistochemistry of c-Fos
Mice were exposed to formalin vapor (20 μL on a cotton paper, which makes vapor of 100 ppm) or saline for 3 min in a closed box (1213 mL) that was the same size as the side chamber used in the avoidance test (Fig. 1a). Animals were returned to their home cages for another 2 h. Then, they were anesthetized and their brains were excised as described in the previous section. Serial transverse frozen sections (40 μm) were cut from the brain tissue that included the spinal trigeminal nucleus caudalis (Sp5C) by using area postrema as a landmark. Every second section was collected in PBS and processed as free-floating sections on a shaking table at room temperature. Sections were sequentially incubated with PBS containing 0.3% Triton-X and 1% normal donkey serum for 30 min, rabbit anti-c-Fos antiserum (1/1000, Oncogene Research Products, San Diego, CA, USA) for 60 min, and biotinylated anti-rabbit IgG antibody (1/200, Vector Laboratories, Burlingham, CA, USA) for 90 min. Finally, tissue was incubated with Alexa Fluor 488 streptavidin conjugate (1/200, Molecular Probes) for 90 min in a dark box. The sections were then mounted on a slide glass, coverslipped using mounting medium with DAPI (Vector Laboratories), and examined under a fluorescence microscope. To confirm the specificity of antibodies, incubations without the antibody were conducted as a negative control in each experiment, and no signal was observed. The number of c-Fos-labeled cells was determined for 3 stained sections through the Sp5C; the numbers were counted in a blinded manner, with no knowledge of the treatment.
Statistical procedure
Data were analyzed using the non-parametric tests of Mann-Whitney (for comparison between the two groups) or Kruskal-Wallis (more than two groups). Data were presented as mean ± s.e.m. Differences were considered significant at p < 0.05.
Author Contributions
T.Y. and C.K. carried out experiments and wrote the draft. N.T., Y.M., Y.K., H.K. and Y.O. analyzed data and discussed concepts. T.K. supervised all experiments and wrote the paper. All authors approved the final version of the manuscript.
Acknowledgments
We thank Ms. Miki Sakoda and Hitomi Kasuga for their technical assistance. We also thank the Joint Research Laboratory, Kagoshima University Graduate School of Medical and Dental Sciences, for permitting the use of their facilities. Part of this work was supported by JSPS KAKENHI 25670121.
References
- Talavera K., Nilius B. & Voets T. Neuronal TRP channels: thermometers, pathfinders and life-savers. TiNS 31, 287–295 (2008). [DOI] [PubMed] [Google Scholar]
- Kim Y. S. et al. Expression of transient receptor potential ankyrin 1 (TRPA1) in the rat trigeminal sensory afferents and spinal dorsal horn. J. Comp. Neurol. 518, 687–698 (2010). [DOI] [PubMed] [Google Scholar]
- Nassenstein C. et al. Expression and function of the ion channel TRPA1 in vagal afferent nerves innervating mouse lungs. J. Physiol. 586, 1595–1604 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bessac B. F. & Jordt S.-E. Breathtaking TRP channels: TRPA1 and TRPV1 in airway chemosensation and reflex control. Physiology 23, 360–370 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrè E. et al. Cigarette smoke-induced neurogenic inflammation is mediated by alpha, beta-unsaturated aldehydes and the TRPA1 receptor in rodents. J. Clin. Invest. 118, 2574–2582 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi N. et al. TRPA1 underlies a sensing mechanism for O2. Nat. Chem. Biol. 7, 701–711 (2011). [DOI] [PubMed] [Google Scholar]
- McNamara C. R. et al. TRPA1 mediates formalin-induced pain. Proc. Natl. Acad. Sci. USA. 104, 13525–13530 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakashimo Y. et al. Expression of transient receptor potential channels vanilloid (TRPV) 1-4, melastin (TRPM) 5 and 8, and ankyrin (TRPA1) in the normal and methimazole-treated mouse olfactory epithelium. Acta Oto-Laryngol. 130, 1278–1286 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwan K. Y. et al. TRPA1 contributes to cold, mechanical, and chemical nociception but is not essential for hair-cell transduction. Neuron 50, 277–289 (2006). [DOI] [PubMed] [Google Scholar]
- Nakamura A., Fukuda Y. & Kuwaki T. Sleep apnea and effect of chemostimulation on breathing instability in mice. J. Appl. Physiol. 94, 525–532 (2003). [DOI] [PubMed] [Google Scholar]
- Okubo K. et al. Hydrogen sulfide-induced mechanical hyperalgesia and allodynia require activation of both Cav3.2 and TRPA1 channels in mice. Br. J. Pharmacol. 166, 1738–1743 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doty R. L. et al. Intranasal trigeminal stimulation from odorous volatiles: Psychometric responses from anosmic and normal humans. Physiol. Behav. 20, 175–185 (1978). [DOI] [PubMed] [Google Scholar]
- Cain W. S. & Murphy C. L. Interaction between chemoreceptive modalities of odour and irritation. Nature 284, 255–257 (1980). [DOI] [PubMed] [Google Scholar]
- The 2011 Ig Nobel Chemistry Prize Winners. Ann. Improbable Res. 17(6), 9 (2011). [Google Scholar]
- Nagatomo K. & Kubo Y. Caffeine activates mouse TRPA1 channels but suppresses human TRPA1 channels. Proc. Natl. Acad. Sci. USA. 105, 17373–17378 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]