Abstract
In this study, a modified infusion procedure and a novel infusion device designed for use in humans (Clinical Device B) were evaluated for delivery of recombinant adeno-associated virus (AAV2) to brain. The device is composed of 1.2 m of fused silica inserted through a 24.6-cm surgical steel cannula designed to fit a standard Leksell® clinical stereotaxic frame and micro-infusion syringe pump. AAV2 encoding the human aromatic L-amino acid decarboxylase gene (AAV-hAADC-2) was infused into the putamen of 4 normal rhesus monkeys as a supportive study for a clinical trial in Parkinson&apos ;s disease (PD) patients. Two infusion protocols were tested: a ramped procedure (slow stepwise increases in rate from 0.2 μL/min to 1μL/min), thought to be essential for convection-enhanced delivery (CED), and a non-ramped infusion at a constant rate of 1 μL/min. The primary endpoints were safety evaluation of the infusion procedures and assessment of transgene expression at 5.5 weeks post-infusion. Clinical observations after vector infusions revealed no behavioral abnormalities during the study period. No differences in gross pathology with either the ramped or non-ramped infusion procedure were observed. Histopathology of the putamen was comparable with both procedures, and revealed only minimal localized inflammatory tissue reaction along the needle track in response to cannula placement and vector infusion. AADC immunohistochemistry demonstrated that vector was distributed throughout the putamen, with no significant difference in volume of immunostaining with either infusion procedure. Serum antibody levels against AAV2 vector exhibited a minor increase after infusion. These results validate the clinical utility of this new infusion device and non-ramped infusion conditions for intraputamenal gene therapy, and have the potential to impact a number of human diseases in which delivery of therapeutics to brain is indicated.
Keywords: Adeno-associated virus, Aromatic L-amino acid decarboxylase, Parkinson's disease, Brain, Infusion device, Putamen, Convection-enhanced delivery, AAV-hAADC-2
Introduction
PD is characterized in part by the progressive loss of dopaminergic neurons in the substantia nigra and a severe decrease of dopamine in the putamen (Hornykiewicz, 1975). Aromatic L-amino acid decarboxylase (AADC) is an enzyme in the dopamine biosynthetic pathway that converts L-dopa to dopamine. We have previously shown that transfer of the cDNA encoding human AADC to rat or non-human primate putamen can reduce effective L-dopa doses in animal models of PD, and thereby restore dopamine to normal levels (Bankiewicz et al., 2000; Sanchez-Pernaute et al., 2001). In human PD patients, this therapy would be expected to lower L-dopa requirements and extend the duration during which clinical benefit of the drug is observed. A challenging feature of this therapy is that vector infusion must be quantitatively and accurately delivered to the putamen and distribution should be as diffuse from the needle delivery site as possible. Focal, concentrated delivery and expression of AADC could result in localized dopamine production in the putamen that may be undesirable.
Parkinsonian rhesus macaques have been used in pharmacological studies with the AADC gene expressed from a recombinant AAV2 vector with a CMV enhancer and promoter (AAV-hAADC-2) (Bankiewicz et al., 2000). The vector was infused by CED with a ramped infusion procedure through a prototype delivery device (‘‘preclini-cal’ ’) composed of a short fused silica catheter connected to a syringe pump by Teflon tubing. Although a standardized preclinical device was proven safe for delivery of vector to the putamen of non-human primates (Bankiewicz et al., 2000), an unacceptable amount of the vector was lost by adsorption to Teflon. The relatively large hold-up volume (or internal volume) of the Teflon tubing also required that the syringe and proximal end of the tubing (end furthest from the cannula) be filled with oil to conserve vector. Thus, a new device (Clinical Device B) was designed for administration of AAV2 vector to humans.
The clinical infusion device was designed to (1) minimize vector loss to the internal surfaces of the catheter and tubing, (2) minimize the hold-up volume of the device such that vector could be dispensed from a syringe placed approximately 1.2 m away from the site of infusion, (3) strengthen and lengthen the cannula material for added stability in the larger human brain, (4) attach to a commonly used stereotaxic surgery frame, and (5) allow manufacturing of a device in compliance with quality system requirements (QSR) for medical devices. The end product of these design requirements was Clinical Device B, a stereotaxic frame-compatible surgical steel cannula and Teflon tubing that are lined with fused silica and fitted to a standard Luer lock at the proximal end for attachment to a programmable syringe pump.
Macromolecules can be administered to large areas of the brain by taking advantage of fluid convection through interstitial spaces in the brain (Bobo et al., 1994; Lieberman et al., 1995). Maximal volume of distribution is achieved by applying a pressure gradient and optimizing dose, infusion time, and volume (Kroll et al., 1996). Typically, the infusion rate is ramped up over about 30 min to avoid an initial pressure buildup at the infusion site that could result in reflux of product along the needle track. However, a non-ramped delivery procedure would allow for a simpler infusion protocol and shorter infusion times that are important considerations for clinical success. The goal of this study was to evaluate the safety and efficacy of the new infusion device (Clinical Device B) and to compare ramped vs. non-ramped infusion procedures in normal monkeys. The primary endpoints of the present study were vector distribution as measured by immunohistochemistry for hAADC expression, and safety as evaluated by clinical observations, histopathology, and antibody formation.
Materials and methods
Recombinant vector production
Recombinant AAV2 was generated by a triple transfection protocol (Matsushita et al 1998) Briefly, after expansion of cells from the HEK 293 working cell bank through a series of disposable culture ware in DMEM con taining 10% fetal bovine serum and 2 mM glutamine, cells were co-transfected with 3 plasmids (pAAV-hAADC-2, pHLP19 and pladeno5). After an appropriate transfection time, the medium containing the transfection reagent was replaced with serum-free medium and the cells were incubated further to allow vector production. Cells were harvested, concentrated by centrifugation, and lysed by a freeze/thaw method to release the AAV-hAADC-2 vector. After centrifugation to remove cellular debris, the lysate was treated with Benzonase®, calcium chloride, and precipitated with polyethylene glycol. Vector was purified by 2 cycles of isopycnic gradient ultracentrifugation in cesium chloride. AAV-hAADC-2 was concentrated, and diafiltered with sterile, buffered saline (PBS) containing 5% sorbitol. Poloxamer 188™ (0.001%) was added, the material sterile filtered (0.22 μm), and stored frozen at —70°C. Vector purity was assessed by SDS-PAGE. Purified rAAV2 vector used in this study showed only the 3 structural AAV capsid proteins, VP1, VP2, and VP3, by silver staining of SDS-PAGE gels. Titer was determined by real-time Q-PCR analysis of vector genomes. The rAAV-hAADC-2 vector clone was the same as that described previously (Sanftner et al., 2004).
Animal study
All in vivo portions of the study performed at Charles River Laboratories, Inc., Charles River Sierra Division (Sierra), adhered to the study protocol and the standard operating procedures (SOPs) were established at Sierra. Procedures were in compliance with the USDA Animal Welfare Act <http://www.nal.usda.gov/awic/legislat/usdaleg1.htm> (9 CFR, Parts 1, 2, and 3) and as described in the Guide for the Care and Use of Laboratory Animals <http://oacu.od.nih.gov/regs/guide/guide1.htm> (ILAR publication, 1996, National Academy Press).
Surgical procedures
Magnetic resonance imaging (MRI) was performed on each monkey prior to surgery to identify stereotaxic coordinates (based on the anatomical structure of the putamen). Two sites in each hemisphere were targeted with one site centered in the rostral putamen and a second in the caudal putamen. Adult rhesus monkeys (n = 4) were immobilized with a mixture of ketamine (Ketaset®, 10 mg/kg, intramuscular injection) and Valium® (0.5 mg/kg, intravenous injection), intubated and prepared for surgery. Isotonic fluids were delivered intravenously at 2 mL/kg/h. Anesthesia was induced with isoflurane (Aerane®, Omeda PPD, Inc., Liberty, New Jersey) at 5% v/v, and then maintained at 1%-3% v/v for the duration of the surgery. The animal's head was placed in an MRI-compatible stereotaxic frame. Core temperature was maintained with a circulating water blanket while electrocardiogram, heart rate, oxygen saturation, and body temperature were con tinuously monitored during the procedure. Burr-holes were made in the skull with a dental drill to expose areas of the dura just above the target sites. AAV-hAADC-2 was infused by CED (Bankiewicz et al 2000; Lieberman et al 1995) Each monkey received a total of 3 × 1011 vg in 200 μL spread over 4 sites (50 μL per site with 2 sites per hemisphere). Infusion cannulae were manually guided to the putamen in each brain hemisphere, and the animals received bilateral infusions (i.e., sequential infusions to the rostral and caudal sites within both hemispheres) of AAV-hAADC-2 (1.5 × 1012 vg/mL) at infusion rates of 0.2 μL/min (10 min), 0.5 μL/min (10 min), 0.8 μL/min (10 min), and 1 μL/min (35 min) for the left hemisphere and a constant rate of 1 μL/min (50 min) for the right hemisphere. Actual stereotaxic coordinates for each animal were: MR15102M rostral putamen AP: 18, ML: ±10.5, DV 20, caudal putamen AP: 15, ML: ±13, DV: 20, R211101M rostral putamen AP: 24, ML: ±12.5, DV: 20, caudal putamen AP: 21, ML: ±13.5, DV: 20, MR15109M rostral putamen AP: 12, ML: ±13, DV: 20, caudal putamen AP: 15, ML: ±12, DV: 20, R23700M rostral putamen AP: 21, ML: ±13.5, DV: 21, caudal putamen AP: 24, ML: ±12.5, DV: 20. Approximately 10 min after infusion, the cannulae were removed, the wound sites were closed, and the monkey was monitored for recovery from anesthesia and then returned to its home cage for continuing observations.
Histology and immunohistochemistry
For histological studies, animals were perfused via intracardiac saline infusion followed by 10% neutral buffered formalin (NBF). The brains were then removed and sliced in a brain mold into coronal blocks (8-10 mm). Harvested brain blocks were fixed by immersion in 10% NBF fixative. The tissue blocks were transferred 2-3 days after fixation into ascending concentrations of PBS/sucrose solution (10, 20, and 30%) over a 3- to 5-day period. Brains were frozen in a bath of isopentane, cooled on dry ice and cut serially into 40-μm-thick coronal sections on a cryostat. Every tenth section was stained with Hematoxylin and Eosin (H&E) solutions (Richard Allen Scientific, Kalama zoo, MI) for histopathological analysis. Immunohistochemistry was carried out on free-floating sections with a primary antibody specific for AADC (Chemicon, Temecula, CA, 1:1500). Sections were incubated in 3% hydrogen peroxide for 30 min to quench endogenous peroxidases. After blocking for non-specific binding with 10% normal goat serum, sections were incubated in primary antibody over night at room temperature, then with a biotinylated anti-rabbit IgG antibody (Vector Laboratories, Burlingame, CA, 1:300) with streptavidin-conjugated horseradish peroxidase (Vector Laboratories, 1:300) at room temperature, both for 1 h. The complex was visualized with 3-3'-diaminobenzidine (DAB, Vector Laboratories) and hydrogen peroxide. Sections were mounted on Superfrost Plus® slides (Brain Research Laboratories, Newton, MA), dried, dehydrated in ascending ethanol series, cleared in xylene, and mounted with Cytoseal-XYL (Richard Allen Scientific, Kalamazoo, MI). Anterior-to-posterior distribution of hAADC immu-nostaining was determined by the formula (n × 10 × 40 μm) where n is the number of sections with hAADC-positive cells, 40 μm is the thickness of the section, and every tenth section is examined. We estimated the volume of distribution in serial sections (every tenth), stained for AADC with the Optical Fractionator-Optical Disector design-based stereology method under 63× magnification on a Zeiss microscope equipped with a video camera and Stereoinvestigator™ stereology software (Microbrightfield, Williston, VT). CEE was <5% for each group. Results are reported as mean ± SD. Student's t test was used to measure statistical significance.
Clinical device
The clinical device was produced under contract by a licensed medical device manufacturer. The device was manufactured and assembled in compliance with QSR, the most up-to-date manufacturing practices to provide the best quality assurance for medical devices according to the latest guidelines recommended by the US Food and Drug Administration.
Real-time quantitative PCR
The vector AAV-hAADC-2 used in this study contains the human AADC target cDNA. The real-time Q-PCR primers and probe anneal to exons 2 and 3 of the AADC gene, spanning an intron not present in the vector sequence, thereby minimizing amplification of genomic DNA. Real time Q-PCR was standardized with linearized plasmid DNA containing the vector insert and vector genomes were quantified as described previously (Sommer et al., 2003).
Neutralizing AAV antibody titering
The neutralizing antibody (NAb) titer of serum or plasma was determined in vitro in a cell-based assay. A defined number of AAV2 vector particles encoding a β-galactosi-dase reporter gene (AAV2-LacZ) were incubated with test serum for 1 h at 37°C before addition of the mixture to HEK-293 cells near confluence in 96-well plates. Control (100%) AAV2 transduction was defined as the amount of β-galactosidase activity measured in culture 24 h after transduction with AAV2-LacZ in the presence of naïve mouse serum (NMS). A half-log serial dilution of the test serum in NMS was made to determine the highest dilution of test serum that resulted in 50% or greater inhibition of β-galactosidase expression. Each dilution series was tested in triplicate. A reference plasma with a well-defined AAV2 neutralizing titer was run in each assay and a negative control (NMS only) was used to determine the assay background. The titer of NAb was defined as the two dilutions that bracketed the 50% inhibition level, e.g., 1:100 to 1:316.
Bridging ELISA
Titer plates (96 well) were coated with AAV2 particles and then incubated with test sample (serum or plasma). Plates were rinsed and then incubated with biotinylated AAV2 particles that were then detected with HRP-conjugated streptavidin. The biotinylated AAV2 particles can only be captured by multivalent antibodies forming a bridge between two AAV2 particles. A very low non-specific background signal in this assay permitted testing of undiluted or low dilutions of test articles, and the assay has higher sensitivity than a classical ELISA, in which primary antibody in the test sample is detected by an enzyme-conjugated secondary antibody. The bridging assay allows direct titer comparisons between different species and classes of antibodies. The assay was standardized with known amounts of purified mouse monoclonal antibody ‘A20′ that recognizes AAV2 (Grimm et al., 1998). The quantification limit of this assay is approximately 15 ng/mL anti-AAV2 antibody. Human samples with an NAb titer of 1:100 contained between 1 and 10 μg/mL of antibody equivalent to A20. The average inter-assay variability for 65 human samples that underwent replicate testing by this assay was 23%.
Results
Experimental design
Recombinant AAV2 vectors transduce brain tissue efficiently, but transduction levels decline significantly in the presence of high neutralizing antibody (NAb) titers (>1:1200) (Sanftner et al., 2004). Therefore, 4 male rhesus monkeys with NAb titers of ≤1:100 were selected for AAV2 infusions (Table 1). MRI scans were performed prior to AAV2 delivery to anatomically determine stereotaxic coordinates for vector administration to the putamen. Animals were bilaterally infused with 1.5 × 1011 vg of AAV-hAADC-2 in two 50-μL infusions (7.5 × 1010 vg/site) ineach hemisphere (3.0 × 1011 vg/brain). Ascending infusion rates (ramp) of 0.2 μL/min (10 min), 0.5 μL/min (10 min), 0.8 μL/min (10 min), and 1 μL/min (35 min) were used for the left hemispheres, whereas a constant rate of 1 μL/min for 50 min (non-ramp) was used for the right hemispheres. Animals were monitored for 5.5 weeks, a time span that we have demonstrated is satisfactory for hAADC expression to become relatively stable (unpublished results). Primary endpoints included AADC expression as deter mined by immunohistochemistry and safety assessments as determined by clinical observations and histopathology In addition, serum samples, collected at baseline and at the end of the study, were tested for the presence of both neutralizing and total antibodies against AAV.
Table 1. Neutralizing anti-AAV serum antibody (NAb) titers and bridging ELISA data in rhesus non-human primates after intraputamenal administration of AAV-hAADC-2.
| Non-human primate ID | Sample | NAb titer | Bridging ELISA (μg/mL anti-AAV Ab) |
|---|---|---|---|
| MR15102M | Pre-treatment | 1:1–1:3.1 1:3.1–1:10 |
0.036 |
| Post-treatment | 1:1–1:3.1 1:3.1–1:10 |
0.24 ± 0.08 | |
| MR15109M | Pre-treatment | 1:3.1–1:10 1:3.1–1:10 |
Below detection (<0.015) |
| Post-treatment | 1:3.1–1:10 1:3.1–1:10 |
0.43 ± 0.35 | |
| R211101M | Pre-treatment | 1:1–1:3.1 1:1–1:3.1 |
0.11 |
| Post-treatment | 1:31–1:100 1:10–1:31 |
0.63 ± 0.07 | |
| R23700M | Pre-treatment | 1:10–1:31 1:31–1:100 |
0.24 |
| Post-treatment | 1:31–1:100 1:100–1:316 |
1.3 ± 0.7 |
The results for two NAb assays are shown. The bridging ELISA was standardized with anti-AAV2 mouse monoclonal antibody. The average of two results is shown for post-treatment samples and a single result is shown for pre-treatment samples.
Infusion device development and vector recovery
A prototype infusion device for human use (“Clinical Device A”) was composed of a 25-cm stainless steel cannula, made to fit a standard Leksell® stereotaxic frame. The cannula was composed of 4 stepped layers of medical grade stainless steel tubing to provide rigidity and minimize internal hold-up volume. The steel cannula was connected to a syringe via 1.2 m of Teflon® tubing. Vector recovery studies at flow rates up to 1 μL/min revealed that 90% of the vector product was adsorbing to the device (Table 2), despite the 0.01% Poloxamer 188 included as a surfactant in the product formulation. A 1-h flush of the device with vector improved subsequent recovery, but vector loss was still approximately 40%. Further testing for vector absorption included different tubing materials at flow rates of <1 μL/min. Excellent vector recovery was observed for fused silica, Tygon®, and silicone tubing. Other materials such as steel, Teflon (PTFE and FEP), and polyimide bound significant amounts of vector. Much of the vector loss was observed only at low flow rates. For example, in Teflon tubing, vector loss was inversely proportional to the linear flow rate. Ninety percent of the vector was lost at 1 μL/min (4 mm/min through 1.2 m of tubing), whereas acceptable vector recovery (>80%) could be attained in the same tubing at flow rates above 100 μL/min. In order to maximize the linear flow rate and to eliminate all contact of vector with Teflon and steel surfaces, we lined the entire core of the clinical device with fused silica of inner diameter 100 Am. In this device (“Clinical Device B”), the steel cannula surrounds the fused silica to provide rigidity, and the fused silica extends 10 mm beyond the tip of the steel cannula (Fig. 1). Two external steps near the needle tip are included to minimize potential reflux along the needle track (Vogelbaum, 2004). An additional 1.2 m of fused silica connects the cannula to a Luer hub and is covered by Teflon tubing only to provide protection. The hold-up volume of this device is approximately 12 μL. The device was manufactured and assembled in accordance with QSR and terminally sterilized by ethylene oxide gas.
Table 2. Vector recovery from preclinical, clinical device A, and clinical device B.
| Preclinical device | Clinical Device A | Clinical Device B | |
|---|---|---|---|
| Product contact surfaces | Fused silica, Teflon®, polypropylene (Luer couplings) | No. 304 stainless steel, Teflon®, polypropylene (Luer lock and syringe) | Fused silica, polypropylene (Luer and syringe) |
| Internal hold-up volume | Variable | 350 μL | 12 μL |
| Vector recovery after <50 μL flush volume (±SD) | 63 ± 16% | 9 ± 4% | 101 ± 6% |
| Vector recovery after 500 μL of flush at 8 μL/min (±SD) | nd | 60 ± 15% | nd |
nd = Not done.
Fig. 1.

AAV vector infusion device designed for human clinical use. The cannula (
 ) is composed of 4 layers of 304 surgical steel fused together by laser welding in a step design, ending in 23-gauge tubing. The steel cannula is lined with fused silica of 100 Am inner diameter (
) is composed of 4 layers of 304 surgical steel fused together by laser welding in a step design, ending in 23-gauge tubing. The steel cannula is lined with fused silica of 100 Am inner diameter (
 , red) which also forms the tip of the delivery device by extending 1 cm beyond the steel. Approximately 1.2 m of additional fused silica covered with Teflon tubing (
, red) which also forms the tip of the delivery device by extending 1 cm beyond the steel. Approximately 1.2 m of additional fused silica covered with Teflon tubing (
 , blue) connect to a Luer hub (
, blue) connect to a Luer hub (
 ). A 1-in., 23-gauge steel spacer (
). A 1-in., 23-gauge steel spacer (
 ) between the fused silica and Teflon tubing is sealed and attached to the Luer hub with medical grade cyanoacrylate glue. Dimensions are in centimeters.
) between the fused silica and Teflon tubing is sealed and attached to the Luer hub with medical grade cyanoacrylate glue. Dimensions are in centimeters.
Quantitative recovery of vector was evaluated through mock infusions with preclinical and clinical devices. For the preclinical device, 400 μL of vector solution was drawn from the distal end into a length of Teflon tubing that was then coupled to a 7-cm cannula composed of fused silica surrounded by a 4-cm piece of 27-gauge steel tubing. After filling the cannula at 100 μL/min, an additional 20-AL flush was dispensed before collecting vector for recovery assays. Four samples were collected from two devices at flow rates from 0.2 to 1.0 μL/min (ramped procedure) with a programmable syringe pump. As shown in Table 2, the average vector recovery from the preclinical devices under these conditions was 63 ± 16% (±SD). For the clinical devices, AAV-hAADC-2 vector was diluted to 5 × 1011 vg/mL, loaded into syringes and attached to the devices. After fill, Clinical Device A was flushed with 500 μL of vector solution at 8 μL/min (62.5 min), while Device B was flushed with a total of 50 μL of vector solution at 4 μL/min (12.5 min). Two sequential aliquots of 50 μL were collected from 3 sets of each device at flow rates from 0.2 to 1.0 μL/min. Vector concentration in each sample was determined by real-time quantitative PCR (Q-PCR). Recovery for Clinical Device A was only 60 ± 15% after the extensive 1-h flush, whereas complete recovery of vector (101 ± 6%) was observed for Device B. Potency of vector samples recovered from Clinical Device B was confirmed by determining the infectious titer (Zhen et al , 2004); no significant decrease in specific activity (infectious units/vg) was observed (data not shown).
Immunohistochemistry and quantitation of hAADC expression in vivo
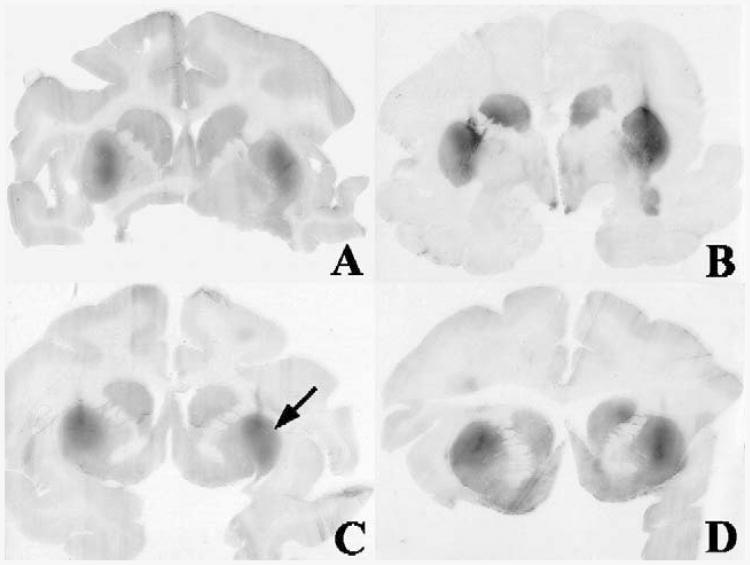
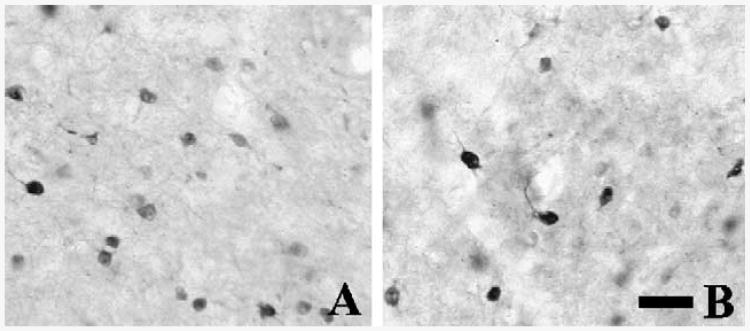
Immunohistochemical analysis of hAADC expression was performed on each brain hemisphere at 5.5 weeks post-AAV-hAADC-2 infusion to determine if the vector distribution was different after ramped (left hemisphere) vs. non-ramped (right hemisphere) infusion with Clinical Device B. All monkeys had evidence of hAADC expression widely distributed within the targeted putamen. Serial sections were examined with brightfield microscopy for hAADC-positive cells. The volume of distribution and Anterior–Posterior (A–P) spread of hAADC transgene-positive cells were determined for all animals. Fig. 2shows immunohistochemical staining for the hAADC transgene in cross-sections through the infusion site. Images are of whole mounts of sections from animals MR15102M (A), MR15109M (B), R23700M (C), and R211101M (D). Sections are oriented from a caudal view with the right hemisphere on the right side of the image and the left hemisphere on the left side of the image. In all animals, transgene expression was primarily localized to the putamen. Occasionally, a small amount of vector was seen to spread from the putamen to the caudate or nucleus accumbens and a few hAADC-positive cells were found in these regions. No hAADC expression was detected in cortical regions except in direct line with the infusion track as illustrated in Fig. 2B. No difference in the number of AADC-positive cells or intensity of hAADC staining was seen in a comparison of the right and left hemispheres. A higher magnification image of the infusion site of the putamen in a representative animal from the left hemisphere that received ramped infusion (Fig. 3A), or the right hemisphere that received non-ramped infusion (Fig. 3B) illustrates the hAADC transgene expression in medium spiny neurons. Immuno-histochemical staining for hAADC expression was seen in all (8/8) of the infused hemispheres. AAV-hAADC-2 ad ministration resulted in good expression and coverage of the putamen with a similar distribution of AAV-hAADC-2 observed with either the ramped or non-ramped infusion procedure.
Fig. 2.
Immunohistochemistry for AADC in whole mounted brain sections. Brains are shown in coronal section through the infusion site at 5.5 weeks post-infusion. Panels A, B, C, and D represent the four different monkey brains analyzed in this study [MR15102M (A), MR15109M (B), R23700M (C), and R211101M (D)]. All left hemispheres received ramped infusion and all right hemispheres received non-ramped infusion. The black arrow indicates the putamen region.
Fig. 3.
Immunohistochemistry for AADC at high magnification within the putamen. Immunostaining is localized to the medium spiny neurons in the putamen at 5.5 weeks post-infusion. A representative section from the hemisphere that received ramped infusion is shown in Panel A and one from the hemisphere that received non-ramped infusion is shown in Panel B. Scale bar = 100 μm.
Quantitation of the estimated volume of hAADC distribution in serial sections stained with anti-hAADC anti body was performed with Stereoinvestigator™ stereology software (Microbrightfield, Williston, VT). The Anterior– Posterior (A–P) distribution, a one-dimensional measure of distribution from rostral to caudal, and volume of hAADC immunostaining were determined separately for each hemi sphere of the 4 AAV-treated non-human primates (Table 3). The mean A–P distribution for the left hemisphere (ramped delivery) was 9600 μm ± 2422 μm (SD) and the mean volume was 238 mm3 ± 121 mm3. The mean A–P distribution for the right hemisphere (non-ramped delivery) was 9606 Am ± 2037 Am and the mean volume was 284 mm3 ± 55 mm3. There was no significant difference in mean volume or mean A–P distribution between ramped or non-ramped by an unpaired Student's t test (P = 0.9973 for A–P distribution comparison and P = 0.5187 for spread volume comparison). The non-ramped infusion did not result in reflux of vector along the cannula track or a decrease in transgene-derived hAADC distribution. The lack of reflux may also in part be due to the multiple step design of the cannula.
Table 3. Quantitation by immunohistochemistry of AADC anterior-to-posterior (A–P) distance of spread and spread volume in non-human primates infused with AAV-hAADC-2.
| Animal I.D. | A-P distribution | Spread volume |
|---|---|---|
| (μm) | (mm3) | |
| Right (non-ramped infusion) | ||
| MR15109 | 8822 | 272.1 |
| R23700M | 12400 | 346.5 |
| R211101M | 9600 | 301.5 |
| MR15102M | 7600 | 214.4 |
| Mean, right hemisphere | 9606 | 283.6 |
| (non-ramped infusion) | ||
| Standard deviation | 2037 | 55.4 |
| Left (ramped infusion) | ||
| MR15109 | 8400 | 110.2 |
| R23700M | 12800 | 402.3 |
| R211101M | 10000 | 217.3 |
| MR15102M | 7200 | 222.2 |
| Mean, left hemisphere | 9600 | 238.0 |
| (ramped infusion) | ||
| Standard deviation | 2422 | 121.1 |
Histopathology
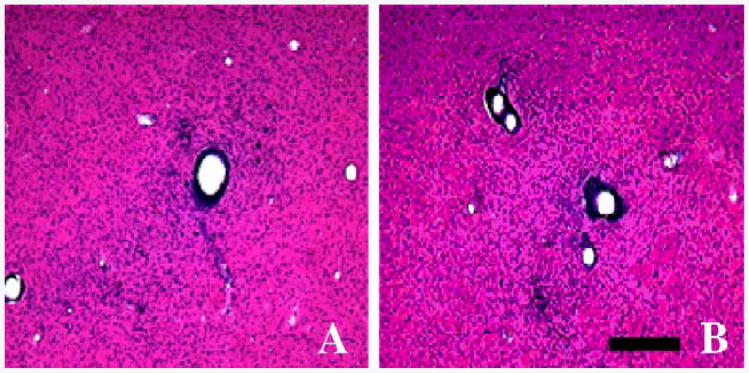
Histopathological analysis of serial sections stained with H&E was performed on all animals to determine the effect of cannula placement and AAV-hAADC-2 infusion with either ramped or non-ramped delivery. Fig. 4 shows H&E-stained sections within the putamen from a representative animal, R211101M, at 5× magnification. Animal R211101M received bilateral CED of AAV-hAADC-2 by the non-ramped infusion procedure in the right hemisphere (Panel A) and the ramped infusion procedure in the left hemisphere (Panel B). Images illustrate the area adjacent to the cannula track at the mid-caudal putamen level. All H&E-stained slides were reviewed by a neuropathologist (Pathology Associates Inc.) blinded to treatment conditions. Some mononuclear cellular infiltration was seen in the putamen with mild perivascular cuffing. Both puta-mina contain a few infiltrated blood vessels and mild parenchymal infiltration. Histopathologic appearance of the right and left hemispheres was similar, with slight inflammatory tissue reaction at the infusion site.
Fig. 4.
H&E-stained sections within the putamen from a representative animal, R211101M, at 5× magnification. Animal R211101M received bilateral CED of AAV-hAADC-2 using the non-ramped infusion procedure in the right hemisphere (Panel A) and the ramped infusion procedure in the left hemisphere (Panel B). Images illustrate the area adjacent to the cannula track, and were taken at the mid-caudal putamen level. Scale bar = 400 μm.
Development of neutralizing antibodies
Neutralizing antibody (NAb) and total antibody titers to AAV capsid were determined for serum samples collected prior to infusion of vector, and at the time of necropsy. Slight rises in anti-AAV antibody levels were detected by bridging ELISA in all animals after bilateral infusion of AAV-hAADC-2 (Table 1). The animal (R23700M) with the highest serum neutralizing antibody titer (1:10 to 1:100) before treatment had a post-treatment antibody increase to 1:31-1:316 on Day 42. This animal had similar hAADC transgene distribution when compared to the other animals and thus there was no apparent inhibition of vector spread associated with the higher titer.
Clinical observations
Monkeys were evaluated daily for clinical signs, food consumption, and body weight. Post-surgical daily clinical observations indicated that the animals tolerated the CED procedure well and did not display behavioral changes. There were no AAV-hAADC-2 treatment-related clinical signs or changes in body weight. Observations made during the post-treatment period were similar to those commonly observed in laboratory-housed rhesus monkeys that undergo similar surgical procedures.
Discussion
In this study, newly developed infusion devices intended for administration of AAV2 vector in a clinical trial for Parkinson's disease were evaluated along with a comparison of infusion conditions. Mock infusions designed to test vector delivery established that essentially 100% of the intended dose could be delivered with Clinical Device B by avoidance of all contact of vector with Teflon or steel surfaces. Stereotaxic administration of AAV-hAADC-2 into the putamen of 4 non-human primates was performed by comparison of a ramped (graded increase in infusion rate) vs. a non-ramped (constant rate) infusion procedure. The putamen was targeted because it is known to process sensorimotor information, and in PD patients, it is the brain region with the most extensive dopamine depletion. Restoration of dopamine levels specifically in the putamen will likely be beneficial in ameliorating motor complication commonly seen in PD patients. Expression of hAADC was detectable by immunohistochemistry and was distributed broadly in the putamen. Stereological quantitation of the volume of transgene-derived hAADC demonstrated similar distribution in hemispheres receiving either infusion procedure. Furthermore, the constant flow rate did not result in excessive vector deposits along the needle track. Histopa-thologic analysis revealed only slight tissue inflammatory reaction localized to the area of the cannula insertion track suggesting no safety concerns. There was no apparent difference in the degree of cellular infiltration or inflammation between the left and right putamen (i.e., ramped vs. non-ramped infusion). No abnormal clinical observations were seen after surgery and intraputamenal infusion in any animals.
Our observation of comparable transgene expression between ramped vs. non-ramped infusion conditions contra dicts, in part, prior assumptions involving CED (Bankiewicz et al., 2000; Lieberman et al., 1995). A slow ramp-up of delivery rate was thought to be important for establishing the pressure gradient necessary for interstitial fluid con vection (Bobo et al., 1994). However, at least with the devices and conditions described herein for intraputamenal delivery of AAV-hAADC-2, the results indicated that ramp-up of flow rate is not necessary for efficient distribution of vector within the non-human primate putamen.
In addition to device and infusion parameters, another important consideration for effective AAV-mediated gene delivery into any compartment is potential neutralization by anti-AAV antibodies. There is a broad range of pre-existing AAV neutralizing antibody titers in humans (Blacklow et al., 1968) that have the potential of adversely affecting the efficacy of gene therapy techniques. Any AAV-mediated gene therapy approach must anticipate such hurdles. For example, in a model system utilizing SCID mice wherein human AAV2-neutralizing antibody titers could be established at various levels, it was observed that titers <1:10 significantly impacted liver transduction of AAV-Factor IX after intravenous administration (Scallan et al., 2004) (Scallan et al., full manuscript in preparation). Delivery of AAV2 to the putamen is assumed to be less subject to neutralization by circulating antibodies due to the immune-privileged status of the CNS. In fact, studies performed in rats pre-immunized systemically with AAV2 and then infused intrastriatally confirmed significant protection from neutralization with a decrease in transduction observed only when Nab titers exceeded 1:1200 (Sanftner et al., 2004). The current study utilized animals with pre-existing NAb titers ranging from 1:1 to 1:100 in order to exclude neutralizing antibodies as a confounding variable, and these titers had no apparent impact on hAADC expression in putamen. Moreover, post-infusion titers rose only slightly after vector administration, thereby affirming well-targeted and minimally disruptive gene delivery with the current device and infusion conditions. These results also suggest that repeat intrastriatal infusions of AAV2 may be feasible in human patients.
In summary, non-ramped infusion of AAV-hAADC-2 to monkey putamen via a novel infusion device intended for human clinical trials was well tolerated. Transgene (hAADC) expression and distribution in the putamen was comparable to more complicated and time-consuming ramp-up flow conditions. Given that gene therapy of neurodegenerative diseases and other CNS disorders is an expanding field (Tinsley and Eriksson, 2004), the present results suggest that the design of Clinical Device B represents an important transitional step from animal to human studies and this improved AAV-mediated gene delivery system is a key advancement in methodology for this field. While the immediate application is intended for striatal delivery of AAV2 in PD patients, the device and infusion parameters are likely to be applicable for targeting different anatomic sites, delivering a variety of therapeutic drugs, and treating an assortment of CNS clinical indications.
Acknowledgments
The authors thank the members of the Avigen manufacturing team (Jennifer Barr-Davidson, Hoc Nguyen, Joseph Prado, Shawn Chan, Hermia Kwan, James Giang, Liza Africa, Nadia Gowahari, Maria Aguago, Tannie Lee) for production of recombinant vector and Sierra (Sparks, NV) personnel for careful implementation of the in vivo portion of this study. Cheryl Pater is acknowledged for animal resource, contract management, and technical assistance. Peter Little at PAI is acknowledged for expert histopathological analyses. We also thank Dawn McGuire, Alvin Luk, Ruth Lessard, Ken Chahine, and Frasier Wright for their valuable contributions to this project. Lance Sultzbaugh provided skilled editorial and organizational assistance.
References
- Bankiewicz KS, Eberling JL, et al. Convection-enhanced delivery of AAV vector in parkinsonian monkeys; in vivo detection of gene expression and restoration of dopaminergic function using pro-drug approach. Exp Neurol. 2000;164(1):2–14. doi: 10.1006/exnr.2000.7408. [DOI] [PubMed] [Google Scholar]
- Blacklow NR, Hoggan MD, et al. Serologic evidence for human infection with adenovirus-associated viruses. J Natl Cancer Inst. 1968;40(2):319–327. [PubMed] [Google Scholar]
- Bobo RH, Laske DW, et al. Convection-enhanced delivery of macromolecules in the brain. Proc Natl Acad Sci U S A. 1994;91(6):2076–2080. doi: 10.1073/pnas.91.6.2076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimm D, Kern A, et al. Novel tools for production and purification of recombinant adenoassociated virus vectors. Hum Gene Ther. 1998;9(18):2745–2760. doi: 10.1089/hum.1998.9.18-2745. [DOI] [PubMed] [Google Scholar]
- Hornykiewicz O. Brain monoamines and parkinsonism. Natl Inst Drug Abuse Res Monogr Ser. 1975;3:13–21. doi: 10.1037/e472122004-001. [DOI] [PubMed] [Google Scholar]
- Kroll RA, Pagel MA, et al. Increasing volume of distribution to the brain with interstitial infusion: dose, rather than convection, might be the most important factor. Neurosurgery. 1996;38(4):746–752. discussion 752–4. [PubMed] [Google Scholar]
- Lieberman DM, Laske DW, et al. Convection-enhanced distribution of large molecules in gray matter during interstitial drug infusion. J Neurosurg. 1995;82(6):1021–1029. doi: 10.3171/jns.1995.82.6.1021. [DOI] [PubMed] [Google Scholar]
- Matsushita T, Elliger S, et al. Adeno-associated virus vectors can be efficiently produced without helper virus. Gene Ther. 1998;5(7):938–945. doi: 10.1038/sj.gt.3300680. [DOI] [PubMed] [Google Scholar]
- Sanchez-Pernaute R, Harvey-White J, et al. Functional effect of adeno-associated virus mediated gene transfer of aromatic l-amino acid decarboxylase into the striatum of 6-OHDA-lesioned rats. Mol Ther. 2001;4(4):324–330. doi: 10.1006/mthe.2001.0466. [DOI] [PubMed] [Google Scholar]
- Sanftner LM, Suzuki BM, et al. Striatal delivery of rAAV-hAADC to rats with preexisting immunity to AAV. Mol Ther. 2004;9(3):403–409. doi: 10.1016/j.ymthe.2003.12.005. [DOI] [PubMed] [Google Scholar]
- Scallan C, Liu T, et al. Evaluation of AAV Neutralizing Antibodies Using A Passive Immunity Model in SCID Mice. American Society of Gene Therapy; Minneapolis, MN: 2004. Abstract # 753 S286. [Google Scholar]
- Sommer JM, Smith PH, et al. Quantification of adeno-associated virus particles and empty capsids by optical density measurement. Mol Ther. 2003;7(1):122–128. doi: 10.1016/s1525-0016(02)00019-9. [DOI] [PubMed] [Google Scholar]
- Tinsley R, Eriksson P. Use of gene therapy in central nervous system repair. Acta Neurol Scand. 2004;109(1):1–8. doi: 10.1046/j.1600-0404.2003.00240.x. [DOI] [PubMed] [Google Scholar]
- Vogelbaum MA. Convection enhanced delivery for the treatment of malignant gliomas. J Neuro-Oncol. 2004;00:1–17. doi: 10.1007/s11060-004-2243-8. [DOI] [PubMed] [Google Scholar]
- Zhen Z, Espinoza Y, et al. Infectious titer assay for adeno-associated virus vectors with sensitivity sufficient to detect single infectious events. Hum Gene Ther. 2004;15(7):709–715. doi: 10.1089/1043034041361262. [DOI] [PubMed] [Google Scholar]