Abstract
Background
Trials examining FOLFIRINOX in metastatic pancreatic cancer demonstrate higher response rates compared to gemcitabine-based regimens. There is currently limited experience with neoadjuvant FOLFIRINOX in pancreatic cancer.
Methods
Retrospective review of outcomes of patients with borderline resectable or locally unresectable pancreatic cancer who were recommended to undergo neoadjuvant treatment with FOLFIRINOX.
Results
FOLFIRINOX was recommended for 25 patients with pancreatic cancer, 13 (52%) unresectable and 12 (48%) borderline resectable. Four patients (16%) refused treatment or were lost to follow up. 21 patients (84%) were treated with a median of 4.7 cycles. 6 patients (29%) required dose reductions secondary to toxicity. 2 patients (9%) were unable to tolerate treatment and 3 patients (14%) had disease progression on treatment. 7 patients (33%) underwent surgical resection following treatment with FOLFIRINOX alone, 2 (10%) of which were initially unresectable. 2 patients underwent resection following FOLFIRINOX + stereotactic body radiation therapy (SBRT). The R0 resection rate for patients treated with FOLFIRINOX +/− SBRT was 33% (55% borderline resectable, 10% unresectable). A total of 5 patients (24%) demonstrated a significant pathologic response.
Conclusions
FOLFIRINOX is a biologically active regimen in borderline resectable and locally unresectable pancreatic cancer with encouraging R0 resection and pathologic response rates.
Keywords: Neoadjuvant treatment, borderline resectable, locally unresectable, pancreatic cancer, FOLFIRINOX
Introduction
Pancreatic cancer accounts for only 3% of new cancer diagnoses but is the 4th leading cause of cancer death and at current rates is suspected to become the 2nd leading cause of cancer death by 2015[1]. Surgical resection offers the only hope for long term survival, however nearly 80% of patients present with locally advanced or metastatic disease[2]. A subset of patients present with borderline resectable disease, for which neoadjuvant therapy has an established role in obtaining an R0 surgical resection and improving long term survival[3-6]. Additionally, many patients present with locally advanced unresectable disease that with the proper neoadjuvant treatment could potentially be downstaged to resectable disease [7-19]. In general, neoadjuvant treatment is becoming increasingly explored in hopes of improving the prognosis of pancreatic cancer by treating micrometastases, reducing tumor volume and increasing the likelihood of an R0 resection in locally advanced disease[20-22].
Traditionally, gemcitabine-based regimens have been utilized in the preoperative setting for patients with locally advanced pancreatic cancer[23]. However, neoadjuvant gemcitabine based chemoradiation is associated with a relatively low R0 resection rate and few significant histopathologic responses for borderline resectable and locally unresectable disease[24]. Recently, the use of 5-fluorouracil, irinotecan and oxaliplatin (FOLFIRINOX) has emerged as an alternative in pancreatic cancer. A randomized trial of FOLFIRINOX versus gemcitabine in metastatic pancreatic cancer showed improved median survival from 6.8 to 11.1 months[25]. Further studies of FOLFIRINOX in advanced metastatic disease have demonstrated improved response rates compared with gemcitabine and other historical treatments while maintaining or improving quality of life[26-30]. Because of its efficacy in advanced disease, there is interest in determining whether similar response rates can influence the course of locally advanced disease. However, data is lacking regarding FOLFIRINOX in the neoadjuvant setting for locally advanced pancreatic cancer.
In this series, we retrospectively evaluate our experience with neoadjuvant FOLFIRINOX in patients with borderline resectable and unresectable pancreatic cancer. We specifically evaluate the toxicity of the regimen, the impact of toxicity on delay in surgical resection and postoperative complications and the outcome measures of CA 19-9 response, surgical resection rate, R0 resection rate and histopathologic response to treatment.
Methods
Prior to initiation of the study, Institutional Review Board approval was obtained from the University of Pittsburgh (PRO12060156). A prospectively maintained multidisciplinary pancreatic cancer clinic database was searched to identify patients with locally advanced pancreatic cancer who were recommended to undergo treatment with FOLFIRINOX. Patient demographics, chemotherapy response, operative and postoperative outcomes were retrospectively reviewed. Resectability was defined per American Hepato-Pancreato-Billiary Association/Society of surgical Oncology/ Society for Surgery of the Alimentary Tract (AHPBA/SSO/SSAT) criteria[31]. Toxicity to chemotherapy was evaluated using the National Cancer Institute Common Toxicity Criteria. An R0 resection was defined as at least 1 mm of free margins[32]. Histopathologic response following treatment was graded according to the criteria suggested by the College of American Pathologists[33]. Percentages for outcomes following treatment are calculated based only on patients who initiated therapy (21 overall; 10 unresectable, 11 borderline). Postoperative complications were evaluated using the Clavien-Dindo method[34]. Pancreatic leaks were graded according to the International Study Group on Pancreatic Fistula (ISGPF) definitions[35].
Because this was a retrospective study, there was no standardized protocol in place during the course of treatment. Restaging typically consisted of a repeat CT scan and assessment of the CA 19-9 level. The decision of when to restage was at the discretion of the treating oncologist and varied based on individual patient factors. Treatment decisions were made by a multi-disciplinary conference group that consisted of oncologists, surgeons, radiation oncologists, radiologists and gastroenterologists. Patients who received radiation therapy underwent fiducial placement by endoscopic ultrasound and were treated with stereotactic body radiosurgery with 3 fractions of 12 Gy for a total of 36 Gy.
Results
FOLFIRINOX was recommended to twenty-five patients who were to undergo treatment for locally advanced pancreatic cancer between February 2011 and October 2012. Median age was 59 and 52% of patients were female (Table I). 13 patients (52%) had unresectable disease and 12 patients (48%) were borderline resectable based on AHPBA/SSO/SSAT criteria. Eighteen patients (72%) had proximal pancreatic tumors, while 7 patients (28%) had distal pancreatic tumors. Four patients (16%) either refused treatment with FOLFIRINOX or were lost to follow up prior to initiating treatment.
Table 1. Demographics of patients recommended neoadjuvant treatment with FOLFIRINOX.
| Patient Demographics | |
|---|---|
| Age | |
| Median (Range) | 59 (42-73) |
| Gender, n(%) | |
| Male | 12 (48%) |
| Female | 13 (52%) |
| Resectability | |
| Borderline | 12 (48%) |
| Unresectable | 13 (52%) |
| Ca 19-9 | |
| Mean ± SD | 1819 ± 4207 |
| Median (Range) | 371 (8-19000) |
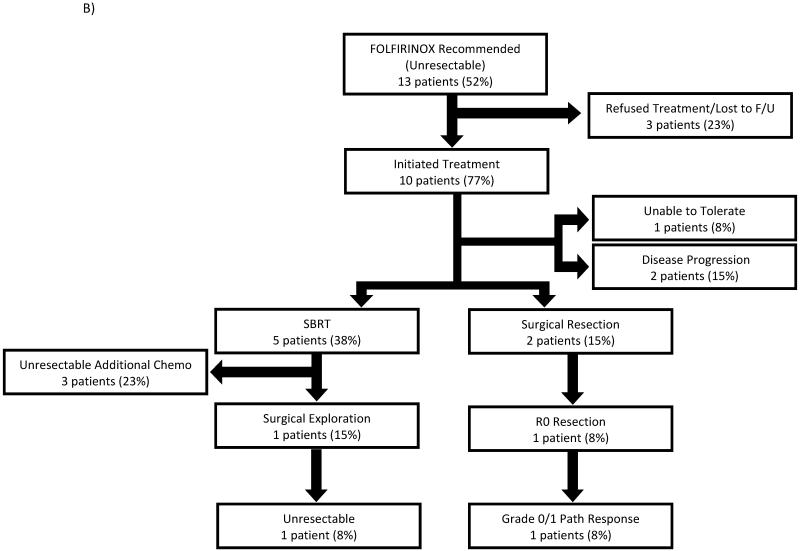
Patients received a mean of 5 cycles of FOLFIRINOX with most patients (57%) receiving between 4-6 cycles (Table II). Patients who proceeded to surgery were treated with a mean of 6 cycles. 6 patients (29%) required dose reductions during their course of treatment and 5 patients (24%) required temporary delay in treatment due to toxicity. The mean delay in treatment was 1.5 weeks, with no delay longer than 2 weeks. Toxicity from treatment is listed in Table III using NCI Common Toxicity Criteria grading. No patients were denied surgery due to toxicity or side effects related to treatment. A flow chart for outcomes of patients undergoing treatment for borderline resectable and unresectable disease is depicted in Figure I. FOLFIRINOX was discontinued prior to surgical resection in 24% of those that initiated treatment (5 patients). Reason for discontinuation of therapy included inability to tolerate side effects (2 patients, 10%) and disease progression (3 patients, 14%). 5 patients (24%) were treated with radiation therapy in addition to FOLFIRINOX. 4 patients (19%) were treated with additional chemotherapy following FOLFIRINOX + SBRT.
Table II. Treatment characteristics of neoadjuvant FOLFIRINOX.
| Treatment Characteristics | |
|---|---|
| Treatment Cycles | |
| Mean | 5 |
| 1-3, n(%) | 6 (29%) |
| 4-6, n(%) | 12 (57%) |
| ≥7, n(%) | 2 (10%) |
| Dose Reduction, n(%) | 6 (29%) |
| Treatment Delays, n(%) | 5 (24%) |
| Delay Duration, weeks (mean) |
1.5 |
| Admission During Tx, n(%) | 4 (19%) |
| Additional Treatment, n(%) | |
| Chemotherapy | 4 (19%) |
| SBRT | 12 (57%) |
Table III. Toxicity following treatment with neoadjuvant FOLFIRINOX.
| Treatment Toxicity | ||
|---|---|---|
|
| ||
| Grade 2 | Grade 3 | Grade 4 |
| Fatigue (5%) | Weight Loss (5%) | Neutropenia (5%) |
| Weight loss (5%) | Hypoalbuminemia (5%) | Leukopenia (5%) |
| Nausea (10%) | Diarrhea (5%) | |
| Vomiting (10%) | Neuropathy (5%) | |
| Dehydration (10%) | Leukopenia (5%) | |
| Blurry vision (5%) | Neutropenia (10%) | |
| Leukopenia (14%) | Thrombocytopenia (10%) | |
| Neutropenia (5%) | Splenic vein thrombosis (5%) | |
| Thrombocytopenia (10%) | ||
Figure I. Flowchart depicted outcomes of patients treated with neoadjuvant FOLFIRINOX for borderline resectable disease (A) and unresectable, locally advanced disease (B).
Seven patients (33%) demonstrated a CA 19-9 response of at least 50% decrease following treatment with FOLFIRINOX alone. Eleven patients (52%) who initiated treatment underwent surgical exploration, 8 (73%) of the borderline resectable patients and 3 (30%) of the unresectable patients. Of these, 1 patient from each group was found to have metastatic disease, resulting in an overall resection rate of 43%, with 7 borderline resectable patients (64%) and 2 unresectable patients (20%) undergoing surgical resection. The two unresectable patients who were successfully resected had encasement of the superior mesenteric and celiac arteries prior to initiation of treatment. The R0 resection rate overall was 33%; with 55% of borderline resectable patients and 10% of unresectable patients achieving R0 resection. Five patients (56% of those resected) required portal vein resection. 7 patients were resected after FOLFIRINOX alone (5 R0) and 2 patients were resected after FOLFIRINOX + SBRT (2 R0). Histopathologic response to therapy was evaluated for all patients undergoing resection. Grade 0, complete pathologic response with absence of viable tumor cells, was seen in 1 patient (5%) after FOLFIRINOX alone. Grade 1, moderate pathologic response, was seen in 4 patients (19%), 3 after FOLFIRINOX alone and 1 after FOLFIRINOX + SBRT.
There was no perioperative mortality. Postoperative morbidity affected 77% of patients with a majority of complications (60%) being Grade I or II. There was one grade IIIa complication, (a peritoneal abscess requiring interventional radiology drainage) and one grade IV complication (a small bowel perforation requiring exploration). Two patients (15%) had pancreatic leaks, one grade A and one grade B.
Discussion
Pancreatic cancer is a highly lethal disease, due in part to the fact that only 15% of patients have resectable disease at presentation. For the significant number of patients with borderline resectable and locally unresectable disease, surgical resection offers the best hope for long-term survival and outcomes for those who are not ultimately resected are poor[36]. These patients have a significant potential to benefit from neoadjuvant therapy as they are either not surgical candidates or at high risk for R1 resection.
Neoadjuvant treatment, including chemotherapy or chemoradiation for borderline resectable and locally unresectable pancreatic cancer offers several advantages including: the potential to downstage unresectable or borderline resectable tumors to enable surgical resection, avoiding surgery in patients whose suffer disease progression while on neoadjuvant treatment and circumventing the delay caused by postoperative recovery and surgical complications in delivering adjuvant therapy[37]. Furthermore, recent evidence suggests that pancreatic cancer appears to be a systemic disease at the time of diagnosis. By the time most pancreatic cancers are diagnosed, the primary tumor already contains clonal populations of cells that are capable of giving rise to distant metastases[38]. Preclinical models have demonstrated that circulating pancreatic cells are present even in animals with premalignant pancreatic lesions, suggesting that pancreatic cancer is systemically disseminated even in its early stages[39]. These data suggest a tremendous potential benefit to neoadjuvant chemotherapy, allowing for treatment of circulating cells prior to surgery and before they have the opportunity to seed distant metastases. The benefits of neoadjuvant chemotherapy are further supported by mathematical modeling, which suggests treatment with chemotherapy prior to surgery will slow the growth rate of pancreatic cells and translate into improved survival[40].
Despite the numerous proposed benefits to neoadjuvant treatment of borderline resectable and locally unresectable, outcomes for regimens that are currently being utilized leave significant room for improvement. A recently published meta-analysis of 111 studies analyzing various neoadjuvant chemotherapy regimens, a majority combined with radiotherapy (94%), included a subset of patients with borderline resectable/unresectable tumors. In these patients the resection rate following treatment was 33%, with an R0 resection rate of only 26%[41]. Another meta-analysis of 14 phase II clinical trials revealed a resection rate of 32% with 20% R0 resection rate in locally advanced patients treated with neoadjuvant chemotherapy with or without radiation[42]. Gemcitabine-based chemoradiation has been the historical treatment of choice for borderline resectable and locally unresectable disease, however studies looking specifically at such regimens also demonstrate a relatively low resection rate. A meta-analysis of preoperative gemcitabine with or without chemoradiation in patients with locally advanced pancreatic cancer resulted in 27% of patients being surgically resected and a R0 resection rate of 23%[24]. Another recently published study of 215 patients who received neoadjuvant chemoradiation with gemcitabine for locally advanced pancreatic cancer had a 26% resection rate with 10% achieving a R0 resection[7]. Current neoadjuvant treatments have increased the number of R0 resections for locally advanced disease, but many patients remain unresectable after treatment and they may benefit from a more effective neoadjuvant regimen.
This retrospective study follows 25 patients with borderline resectable and locally unresectable pancreatic ductal adenocarcinoma through their course of treatment with FOLFIRINOX in the preoperative setting. We demonstrate that neoadjuvant treatment with FOLFIRINOX is safe, feasible, and has acceptable tolerability outside of a clinical trial. 84% of patients with locally advanced pancreatic cancer who were offered the regimen underwent treatment. No patients were denied surgery due to treatment toxicity and there were no significant delays in surgical resection for patients who had a response to treatment, an important outcome to consider with any neoadjuvant therapy. Additionally, postoperative morbidity and the pancreatic leak rate for patients receiving FOLFIRINOX is historically comparable to patients who were not treated with neoadjuvant therapy[43].
This series suggests that FOLFIRINOX is a highly active neoadjuvant regimen in borderline resectable and locally unresectable pancreatic cancer. CA 19-9 response of >50%, which has been linked to survival in studies evaluating response to treatment[44-46], was seen in 33% of patients treated with FOLFIRINOX. Our overall resection rate of 43% and R0 rate of 33% are encouraging given the locally advanced disease in the patients evaluated. There have been two series published evaluating FOLFIRINOX preoperatively for locally advanced pancreatic cancer that demonstrated similar results to our findings. A study by Hosein et al retrospectively evaluated 18 patients with locally advanced pancreatic cancer, of which 78% were unresectable and 22% were borderline resectable[47]. Despite treating with a median of 8 cycles, compared with 5 in our study, patients in their series had a similar number of dose reductions (17%) and a similar toxicity profile. Their series demonstrated an overall resection rate of 64% with overall R0 rate of 44%. For unresectable patients, the resection rate was 43% with 36% achieving R0 resection. For the borderline resectable patients, the resection rate was 75% with all of these patients achieving R0 resection. A multi-institutional retrospective study of FOLFIRINOX in locally advanced and metastatic pancreatic cancer demonstrated a 35% resection rate in the locally advanced cohort with no mention of R0 resection rate[29]. 100% of borderline resections and 21% of unresectable lesions made it to surgical resection. Our data, in conjunction with the previously published using FOLFIRINOX in the neoadjuvant setting, suggest that the regimen may be at least as, and likely more biologically active than other neoadjuvant regimens evaluated in the literature and that neoadjuvant FOLFIRINOX is a viable alternative to gemcitabine based regimens currently being utilized. Additionally, it offers a platform to build upon that utilizes non-overlapping chemotherapeutic agents to the historical gemcitabine standard. Although FOLFIRINOX may represent an improvement when considered in the context of historical controls, a majority of unresectable patients remained unresectable following treatment leaving significant room for improvement in this devastating disease.
Our most encouraging finding is the rate of pathologic response to treatment (complete 5%, 19% moderate) following FOLFIRINOX alone. This is significantly higher than the complete pathologic response rate with gemcitabine as reported in the literature, which is around 2.5%[48, 49]. Furthermore, complete or near complete pathologic response has been shown to translate into improved prognosis and survival, suggesting a possible survival benefit to patients receiving FOLFIRINOX over gemcitabine-based regimens[48, 49].
In this retrospective study, 24% of patients received radiation therapy alone and 19% went on to receive additional chemoradiation therapy following maximal anticipated effects of FOLFIRINOX. The number of patients receiving radiation is somewhat lower than we would expect in patients with locally advanced disease. Typically, patients were restaged following chemotherapy and then presented at a multi-disciplinary conference where the group made treatment decisions. Patients who had an adequate response to chemotherapy alone proceeded directly to surgery and were not offered radiation. Radiation was typically recommended to those who remained unresectable. While it is difficult to determine retrospectively why patients did not undergo radiation therapy, it may be due to the significant number of patients felt to have become resectable at restaging following FOLFIRINOX treatment. The optimal treatment algorithm for use of neoadjuvant FOLFIRINOX in conjuction with radiation and/or gemcitabine-based regimens has yet to be determined and requires further research. These data also support a paradigm for future trials delineating if particular subgroups of patients may benefit from a FOLFIRINOX compared with a gemcitabine-based regimen.
This study is limited by its retrospective nature and by the small number of patients who have received this therapy. Patients included in our series were young, with a median age of 59 and had good performance status. These patients are most likely to tolerate and benefit from this aggressive regimen. Because of this selection bias, our results may not be typical of the general pancreatic cancer population and at present FOLFIRINOX should be utilized only in carefully selected patients. While we anticipate that the improved resection rate and pathologic response rate demonstrated in our study may translate into improved survival, our data is too immature for a thorough survival analysis, with a median follow up of <9 months. Additionally, the lack of a standardized protocol without a consistent treatment algorithm makes interpretation of these results more challenging [50]. However, these data are not intended to be the definitive evidence to utilize FOLFIRINOX in the neoadjuvant setting, but rather to support the development of more definitive prospective randomized trials of FOLFIRINOX. Despite these limitations, our study suggests that FOLFIRINOX alone, or as part of multimodality approach, is a biologically active regimen in borderline resectable and locally unresectable pancreatic ductal adenocarcinoma with encouraging R0 resection rates when considered in the context of historical treatment results. Further research is needed to determine the utility of additional chemoradiotherapy with FOLFIRINOX and to identify predictors of response in unresectable patients. Additionally, randomized trials with FOLFIRINOX compared with gemcitabine alone or gemcitabine-based regimens are necessary to determine the optimal neoadjuvant therapy in borderline resectable and locally unresectable pancreatic ductal adenocarcinoma.
Synopsis.
This study demonstrates neoadjuvant FOLFIRINOX to be safe and biologically active in borderline resectable and locally unresectable pancreatic cancer with an overall resection rate of 43% and a 44% R0 resection rate. Additionally, there was a significant pathologic response in 24% of patients.
Abbreviation list
- AHPBA/SSO/SSAT
American Hepato-pancreato-billiary Association/Society of Surgical Oncology/Society for Surgery of the Alimentary Tract
- NCI
National Cancer Institute
- f/u
follow up
- SBRT
stereotactic body radiation therapy
- ISGPF
International Study Group on Pancreatic Fistula
Footnotes
Disclosures: None
References
- 1.Eheman C, Henley SJ, Ballard-Barbash R, et al. Annual Report to the Nation on the status of cancer, 1975-2008, featuring cancers associated with excess weight and lack of sufficient physical activity. Cancer. 2012;118:2338–2366. doi: 10.1002/cncr.27514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Geer RJ, Brennan MF. Prognostic indicators for survival after resection of pancreatic adenocarcinoma. Am J Surg. 1993;165:68–72. doi: 10.1016/s0002-9610(05)80406-4. discussion 72-63. [DOI] [PubMed] [Google Scholar]
- 3.Landry J, Catalano PJ, Staley C, et al. Randomized phase II study of gemcitabine plus radiotherapy versus gemcitabine, 5-fluorouracil, and cisplatin followed by radiotherapy and 5-fluorouracil for patients with locally advanced, potentially resectable pancreatic adenocarcinoma. J Surg Oncol. 2010;101:587–592. doi: 10.1002/jso.21527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Truty MJ, Thomas RM, Katz MH, et al. Multimodality therapy offers a chance for cure in patients with pancreatic adenocarcinoma deemed unresectable at first operative exploration. J Am Coll Surg. 2012;215:41–51. doi: 10.1016/j.jamcollsurg.2012.03.024. discussion 51-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Katz MH, Pisters PW, Evans DB, et al. Borderline resectable pancreatic cancer: the importance of this emerging stage of disease. J Am Coll Surg. 2008;206:833–846. doi: 10.1016/j.jamcollsurg.2007.12.020. discussion 846-838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Varadhachary GR, Tamm EP, Abbruzzese JL, et al. Borderline resectable pancreatic cancer: definitions, management, and role of preoperative therapy. Ann Surg Oncol. 2006;13:1035–1046. doi: 10.1245/ASO.2006.08.011. [DOI] [PubMed] [Google Scholar]
- 7.Habermehl D, Kessel K, Welzel T, et al. Neoadjuvant chemoradiation with Gemcitabine for locally advanced pancreatic cancer. Radiat Oncol. 2012;7:28. doi: 10.1186/1748-717X-7-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Arvold ND, Ryan DP, Niemierko A, et al. Long-term outcomes of neoadjuvant chemotherapy before chemoradiation for locally advanced pancreatic cancer. Cancer. 2012;118:3026–3035. doi: 10.1002/cncr.26633. [DOI] [PubMed] [Google Scholar]
- 9.Barugola G, Partelli S, Crippa S, et al. Outcomes after resection of locally advanced or borderline resectable pancreatic cancer after neoadjuvant therapy. Am J Surg. 2012;203:132–139. doi: 10.1016/j.amjsurg.2011.03.008. [DOI] [PubMed] [Google Scholar]
- 10.Lee JL, Kim SC, Kim JH, et al. Prospective efficacy and safety study of neoadjuvant gemcitabine with capecitabine combination chemotherapy for borderline-resectable or unresectable locally advanced pancreatic adenocarcinoma. Surgery. 2012;152:851–862. doi: 10.1016/j.surg.2012.03.010. [DOI] [PubMed] [Google Scholar]
- 11.Leone F, Gatti M, Massucco P, et al. Induction gemcitabine and oxaliplatin therapy followed by a twice-weekly infusion of gemcitabine and concurrent external-beam radiation for neoadjuvant treatment of locally advanced pancreatic cancer: A single institutional experience. Cancer. 2012 doi: 10.1002/cncr.27736. [DOI] [PubMed] [Google Scholar]
- 12.Marti JL, Hochster HS, Hiotis SP, et al. Phase I/II trial of induction chemotherapy followed by concurrent chemoradiotherapy and surgery for locoregionally advanced pancreatic cancer. Ann Surg Oncol. 2008;15:3521–3531. doi: 10.1245/s10434-008-0152-3. [DOI] [PubMed] [Google Scholar]
- 13.Pipas JM, Zaki BI, McGowan MM, et al. Neoadjuvant cetuximab, twice-weekly gemcitabine, and intensity-modulated radiotherapy (IMRT) in patients with pancreatic adenocarcinoma. Ann Oncol. 2012;23:2820–2827. doi: 10.1093/annonc/mds109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sahora K, Kuehrer I, Eisenhut A, et al. NeoGemOx: Gemcitabine and oxaliplatin as neoadjuvant treatment for locally advanced, nonmetastasized pancreatic cancer. Surgery. 2011;149:311–320. doi: 10.1016/j.surg.2010.07.048. [DOI] [PubMed] [Google Scholar]
- 15.Sahora K, Kuehrer I, Schindl M, et al. NeoGemTax: gemcitabine and docetaxel as neoadjuvant treatment for locally advanced nonmetastasized pancreatic cancer. World J Surg. 2011;35:1580–1589. doi: 10.1007/s00268-011-1113-8. [DOI] [PubMed] [Google Scholar]
- 16.Turrini O, Viret F, Moureau-Zabotto L, et al. Neoadjuvant chemoradiation and pancreaticoduodenectomy for initially locally advanced head pancreatic adenocarcinoma. Eur J Surg Oncol. 2009;35:1306–1311. doi: 10.1016/j.ejso.2009.06.005. [DOI] [PubMed] [Google Scholar]
- 17.Milandri C, Polico R, Garcea D, et al. GEMOX plus tomotherapy for unresectable locally advanced pancreatic cancer. Hepatogastroenterology. 2011;58:599–603. [PubMed] [Google Scholar]
- 18.Polistina F, Costantin G, Casamassima F, et al. Unresectable locally advanced pancreatic cancer: a multimodal treatment using neoadjuvant chemoradiotherapy (gemcitabine plus stereotactic radiosurgery) and subsequent surgical exploration. Ann Surg Oncol. 2010;17:2092–2101. doi: 10.1245/s10434-010-1019-y. [DOI] [PubMed] [Google Scholar]
- 19.Strobel O, Berens V, Hinz U, et al. Resection after neoadjuvant therapy for locally advanced, “unresectable” pancreatic cancer. Surgery. 2012;152:S33–42. doi: 10.1016/j.surg.2012.05.029. [DOI] [PubMed] [Google Scholar]
- 20.Brunner TB. Neoadjuvant Therapy for Potentially Resectable Pancreatic Cancer: An Emerging Paradigm? Curr Oncol Rep. 2013 doi: 10.1007/s11912-012-0291-3. [DOI] [PubMed] [Google Scholar]
- 21.Goff SL, Chabot JA. A neoadjuvant strategy for the management of nonmetastatic pancreatic cancer. Cancer J. 2012;18:602–608. doi: 10.1097/PPO.0b013e318279aade. [DOI] [PubMed] [Google Scholar]
- 22.Wolff RA. Neoadjuvant therapy for resectable and borderline resectable adenocarcinoma of the pancreas. Curr Drug Targets. 2012;13:781–788. doi: 10.2174/138945012800564086. [DOI] [PubMed] [Google Scholar]
- 23.Lal A, Christians K, Evans DB. Management of borderline resectable pancreatic cancer. Surg Oncol Clin N Am. 2010;19:359–370. doi: 10.1016/j.soc.2009.11.006. [DOI] [PubMed] [Google Scholar]
- 24.Andriulli A, Festa V, Botteri E, et al. Neoadjuvant/preoperative gemcitabine for patients with localized pancreatic cancer: a meta-analysis of prospective studies. Ann Surg Oncol. 2012;19:1644–1662. doi: 10.1245/s10434-011-2110-8. [DOI] [PubMed] [Google Scholar]
- 25.Conroy T, Desseigne F, Ychou M, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med. 2011;364:1817–1825. doi: 10.1056/NEJMoa1011923. [DOI] [PubMed] [Google Scholar]
- 26.Ychou M, Conroy T, Seitz JF, et al. An open phase I study assessing the feasibility of the triple combination: oxaliplatin plus irinotecan plus leucovorin/ 5-fluorouracil every 2 weeks in patients with advanced solid tumors. Ann Oncol. 2003;14:481–489. doi: 10.1093/annonc/mdg119. [DOI] [PubMed] [Google Scholar]
- 27.Assaf E, Verlinde-Carvalho M, Delbaldo C, et al. 5-fluorouracil/leucovorin combined with irinotecan and oxaliplatin (FOLFIRINOX) as second-line chemotherapy in patients with metastatic pancreatic adenocarcinoma. Oncology. 2011;80:301–306. doi: 10.1159/000329803. [DOI] [PubMed] [Google Scholar]
- 28.Conroy T, Paillot B, Francois E, et al. Irinotecan plus oxaliplatin and leucovorin-modulated fluorouracil in advanced pancreatic cancer--a Groupe Tumeurs Digestives of the Federation Nationale des Centres de Lutte Contre le Cancer study. J Clin Oncol. 2005;23:1228–1236. doi: 10.1200/JCO.2005.06.050. [DOI] [PubMed] [Google Scholar]
- 29.Peddi PF, Lubner S, McWilliams R, et al. Multi-institutional experience with FOLFIRINOX in pancreatic adenocarcinoma. JOP. 2012;13:497–501. doi: 10.6092/1590-8577/913. [DOI] [PubMed] [Google Scholar]
- 30.Gourgou-Bourgade S, Bascoul-Mollevi C, Desseigne F, et al. Impact of FOLFIRINOX Compared With Gemcitabine on Quality of Life in Patients With Metastatic Pancreatic Cancer: Results From the PRODIGE 4/ACCORD 11 Randomized Trial. J Clin Oncol. 2013;31:23–29. doi: 10.1200/JCO.2012.44.4869. [DOI] [PubMed] [Google Scholar]
- 31.Callery MP, Chang KJ, Fishman EK, et al. Pretreatment assessment of resectable and borderline resectable pancreatic cancer: expert consensus statement. Ann Surg Oncol. 2009;16:1727–1733. doi: 10.1245/s10434-009-0408-6. [DOI] [PubMed] [Google Scholar]
- 32.Verbeke CS, Leitch D, Menon KV, et al. Redefining the R1 resection in pancreatic cancer. Br J Surg. 2006;93:1232–1237. doi: 10.1002/bjs.5397. [DOI] [PubMed] [Google Scholar]
- 33.Compton CC, Henson DE. Protocol for the examination of specimens removed from patients with carcinoma of the exocrine pancreas: a basis for checklists. Cancer Committee, College of American Pathologists. Arch Pathol Lab Med. 1997;121:1129–1136. [PubMed] [Google Scholar]
- 34.Clavien PA, Barkun J, de Oliveira ML, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250:187–196. doi: 10.1097/SLA.0b013e3181b13ca2. [DOI] [PubMed] [Google Scholar]
- 35.Bassi C, Dervenis C, Butturini G, et al. Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery. 2005;138:8–13. doi: 10.1016/j.surg.2005.05.001. [DOI] [PubMed] [Google Scholar]
- 36.Malik NK, May KS, Chandrasekhar R, et al. Treatment of locally advanced unresectable pancreatic cancer: a 10-year experience. J Gastrointest Oncol. 2012;3:326–334. doi: 10.3978/j.issn.2078-6891.2012.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Evans DB, Rich TA, Byrd DR, et al. Preoperative chemoradiation and pancreaticoduodenectomy for adenocarcinoma of the pancreas. Arch Surg. 1992;127:1335–1339. doi: 10.1001/archsurg.1992.01420110083017. [DOI] [PubMed] [Google Scholar]
- 38.Yachida S, Jones S, Bozic I, et al. Distant metastasis occurs late during the genetic evolution of pancreatic cancer. Nature. 2010;467:1114–1117. doi: 10.1038/nature09515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rhim AD, Mirek ET, Aiello NM, et al. EMT and dissemination precede pancreatic tumor formation. Cell. 2012;148:349–361. doi: 10.1016/j.cell.2011.11.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Haeno H, Gonen M, Davis MB, et al. Computational modeling of pancreatic cancer reveals kinetics of metastasis suggesting optimum treatment strategies. Cell. 2012;148:362–375. doi: 10.1016/j.cell.2011.11.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Gillen S, Schuster T, Meyer Zum Buschenfelde C, et al. Preoperative/neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of response and resection percentages. PLoS Med. 2010;7:e1000267. doi: 10.1371/journal.pmed.1000267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Assifi MM, Lu X, Eibl G, et al. Neoadjuvant therapy in pancreatic adenocarcinoma: a meta-analysis of phase II trials. Surgery. 2011;150:466–473. doi: 10.1016/j.surg.2011.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Cameron JL, Riall TS, Coleman J, Belcher KA. One thousand consecutive pancreaticoduodenectomies. Ann Surg. 2006;244:10–15. doi: 10.1097/01.sla.0000217673.04165.ea. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Katz A, Hanlon A, Lanciano R, et al. Prognostic value of CA 19-9 levels in patients with carcinoma of the pancreas treated with radiotherapy. Int J Radiat Oncol Biol Phys. 1998;41:393–396. doi: 10.1016/s0360-3016(98)00058-3. [DOI] [PubMed] [Google Scholar]
- 45.Nakai Y, Kawabe T, Isayama H, et al. CA 19-9 response as an early indicator of the effectiveness of gemcitabine in patients with advanced pancreatic cancer. Oncology. 2008;75:120–126. doi: 10.1159/000155213. [DOI] [PubMed] [Google Scholar]
- 46.Reni M, Cereda S, Balzano G, et al. Carbohydrate antigen 19-9 change during chemotherapy for advanced pancreatic adenocarcinoma. Cancer. 2009;115:2630–2639. doi: 10.1002/cncr.24302. [DOI] [PubMed] [Google Scholar]
- 47.Hosein PJ, Macintyre J, Kawamura C, et al. A retrospective study of neoadjuvant FOLFIRINOX in unresectable or borderline-resectable locally advanced pancreatic adenocarcinoma. BMC Cancer. 2012;12:199. doi: 10.1186/1471-2407-12-199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chatterjee D, Katz MH, Rashid A, et al. Histologic grading of the extent of residual carcinoma following neoadjuvant chemoradiation in pancreatic ductal adenocarcinoma: a predictor for patient outcome. Cancer. 2012;118:3182–3190. doi: 10.1002/cncr.26651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Zhao Q, Rashid A, Gong Y, et al. Pathologic complete response to neoadjuvant therapy in patients with pancreatic ductal adenocarcinoma is associated with a better prognosis. Ann Diagn Pathol. 2012;16:29–37. doi: 10.1016/j.anndiagpath.2011.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Katz MH, Marsh R, Herman JM, et al. Borderline resectable pancreatic cancer: Need for Standardization and Methods for Optimal Clinical Trial Design. Ann Surg Oncol. 2013 doi: 10.1245/s10434-013-2886-9. Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]