Abstract
The key enzyme of chlorophyll biosynthesis in higher plants, NADPH:protochlorophyllide (Pchlide) oxidoreductase (POR, EC 1.3.1.33), accumulates in its precursor form (pPORA) in barley. pPORA is bound to the chloroplasts and is able to interact with the enzyme's substrate, Pchlide, at both the cytosolic as well as the stromal side of the plastid envelope. The interaction with intraplastidic Pchlide, formed in ATP-containing chloroplasts upon feeding with -aminolevulinic acid, drives vectorial translocation of pPORA across the plastid envelope membranes. In contrast, exogenously applied Pchlide causes the release of the envelope-bound precursor protein to the cytosol. Both processes compete with each other if intra- and extraplastidic Pchlide are applied simultaneously. A cytosolic heat shock cognate protein of Mr 70,000 present in wheat germ and barley leaf protein extracts appears to prevent the release of the pPORA to the cytosol in vivo, however.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Apel K., Santel H. J., Redlinger T. E., Falk H. The protochlorophyllide holochrome of barley (Hordeum vulgare L.). Isolation and characterization of the NADPH:protochlorophyllide oxidoreductase. Eur J Biochem. 1980 Oct;111(1):251–258. doi: 10.1111/j.1432-1033.1980.tb06100.x. [DOI] [PubMed] [Google Scholar]
- Archer E. K., Keegstra K. Current views on chloroplast protein import and hypotheses on the origin of the transport mechanism. J Bioenerg Biomembr. 1990 Dec;22(6):789–810. doi: 10.1007/BF00786931. [DOI] [PubMed] [Google Scholar]
- Arnon D. I. COPPER ENZYMES IN ISOLATED CHLOROPLASTS. POLYPHENOLOXIDASE IN BETA VULGARIS. Plant Physiol. 1949 Jan;24(1):1–15. doi: 10.1104/pp.24.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyle S. A., Hemmingsen S. M., Dennis D. T. Uptake and processing of the precursor to the small subunit of ribulose 1,5-bisphosphate carboxylase by leucoplasts from the endosperm of developing castor oil seeds. Plant Physiol. 1986 Jul;81(3):817–822. doi: 10.1104/pp.81.3.817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chitnis P. R., Harel E., Kohorn B. D., Tobin E. M., Thornber J. P. Assembly of the precursor and processed light-harvesting chlorophyll a/b protein of Lemna into the light-harvesting complex II of barley etiochloroplasts. J Cell Biol. 1986 Mar;102(3):982–988. doi: 10.1083/jcb.102.3.982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cline K., Werner-Washburne M., Andrews J., Keegstra K. Thermolysin is a suitable protease for probing the surface of intact pea chloroplasts. Plant Physiol. 1984 Jul;75(3):675–678. doi: 10.1104/pp.75.3.675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dahlin C., Cline K. Developmental Regulation of the Plastid Protein Import Apparatus. Plant Cell. 1991 Oct;3(10):1131–1140. doi: 10.1105/tpc.3.10.1131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Della-Cioppa G., Bauer S. C., Klein B. K., Shah D. M., Fraley R. T., Kishore G. M. Translocation of the precursor of 5-enolpyruvylshikimate-3-phosphate synthase into chloroplasts of higher plants in vitro. Proc Natl Acad Sci U S A. 1986 Sep;83(18):6873–6877. doi: 10.1073/pnas.83.18.6873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Douwe de Boer A., Weisbeek P. J. Chloroplast protein topogenesis: import, sorting and assembly. Biochim Biophys Acta. 1991 Nov 13;1071(3):221–253. doi: 10.1016/0304-4157(91)90015-o. [DOI] [PubMed] [Google Scholar]
- Eilers M., Oppliger W., Schatz G. Both ATP and an energized inner membrane are required to import a purified precursor protein into mitochondria. EMBO J. 1987 Apr;6(4):1073–1077. doi: 10.1002/j.1460-2075.1987.tb04860.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erickson A. H., Blobel G. Cell-free translation of messenger RNA in a wheat germ system. Methods Enzymol. 1983;96:38–50. doi: 10.1016/s0076-6879(83)96007-x. [DOI] [PubMed] [Google Scholar]
- Esen A. A simple method for quantitative, semiquantitative, and qualitative assay of protein. Anal Biochem. 1978 Aug 15;89(1):264–273. doi: 10.1016/0003-2697(78)90749-2. [DOI] [PubMed] [Google Scholar]
- Fischer G., Schmid F. X. The mechanism of protein folding. Implications of in vitro refolding models for de novo protein folding and translocation in the cell. Biochemistry. 1990 Mar 6;29(9):2205–2212. doi: 10.1021/bi00461a001. [DOI] [PubMed] [Google Scholar]
- Galat A. Peptidylproline cis-trans-isomerases: immunophilins. Eur J Biochem. 1993 Sep 15;216(3):689–707. doi: 10.1111/j.1432-1033.1993.tb18189.x. [DOI] [PubMed] [Google Scholar]
- Gething M. J., Sambrook J. Protein folding in the cell. Nature. 1992 Jan 2;355(6355):33–45. doi: 10.1038/355033a0. [DOI] [PubMed] [Google Scholar]
- Griffiths W. T. Reconstitution of chlorophyllide formation by isolated etioplast membranes. Biochem J. 1978 Sep 15;174(3):681–692. doi: 10.1042/bj1740681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grossman A. R., Bartlett S. G., Schmidt G. W., Mullet J. E., Chua N. H. Optimal conditions for post-translational uptake of proteins by isolated chloroplasts. In vitro synthesis and transport of plastocyanin, ferredoxin-NADP+ oxidoreductase, and fructose-1,6-bisphosphatase. J Biol Chem. 1982 Feb 10;257(3):1558–1563. [PubMed] [Google Scholar]
- Hallermayer G., Zimmermann R., Neupert W. Kinetic studies on the transport of cytoplasmically synthesized proteins into the mitochondria in intact cells of Neurospora crassa. Eur J Biochem. 1977 Dec;81(3):523–532. doi: 10.1111/j.1432-1033.1977.tb11978.x. [DOI] [PubMed] [Google Scholar]
- Holtorf H., Reinbothe S., Reinbothe C., Bereza B., Apel K. Two routes of chlorophyllide synthesis that are differentially regulated by light in barley (Hordeum vulgare L.). Proc Natl Acad Sci U S A. 1995 Apr 11;92(8):3254–3258. doi: 10.1073/pnas.92.8.3254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joyard J., Block M., Pineau B., Albrieux C., Douce R. Envelope membranes from mature spinach chloroplasts contain a NADPH:protochlorophyllide reductase on the cytosolic side of the outer membrane. J Biol Chem. 1990 Dec 15;265(35):21820–21827. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Madueno F., Napier J. A., Gray J. C. Newly Imported Rieske Iron-Sulfur Protein Associates with Both Cpn60 and Hsp70 in the Chloroplast Stroma. Plant Cell. 1993 Dec;5(12):1865–1876. doi: 10.1105/tpc.5.12.1865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manning-Krieg U. C., Scherer P. E., Schatz G. Sequential action of mitochondrial chaperones in protein import into the matrix. EMBO J. 1991 Nov;10(11):3273–3280. doi: 10.1002/j.1460-2075.1991.tb04891.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müsch A., Wiedmann M., Rapoport T. A. Yeast Sec proteins interact with polypeptides traversing the endoplasmic reticulum membrane. Cell. 1992 Apr 17;69(2):343–352. doi: 10.1016/0092-8674(92)90414-8. [DOI] [PubMed] [Google Scholar]
- Neumann D., Emmermann M., Thierfelder J. M., zur Nieden U., Clericus M., Braun H. P., Nover L., Schmitz U. K. HSP68--a DnaK-like heat-stress protein of plant mitochondria. Planta. 1993;190(1):32–43. doi: 10.1007/BF00195672. [DOI] [PubMed] [Google Scholar]
- Nover L., Scharf K. D. Synthesis, modification and structural binding of heat-shock proteins in tomato cell cultures. Eur J Biochem. 1984 Mar 1;139(2):303–313. doi: 10.1111/j.1432-1033.1984.tb08008.x. [DOI] [PubMed] [Google Scholar]
- Pepperkok R., Scheel J., Horstmann H., Hauri H. P., Griffiths G., Kreis T. E. Beta-COP is essential for biosynthetic membrane transport from the endoplasmic reticulum to the Golgi complex in vivo. Cell. 1993 Jul 16;74(1):71–82. doi: 10.1016/0092-8674(93)90295-2. [DOI] [PubMed] [Google Scholar]
- Reid G. A., Yonetani T., Schatz G. Import of proteins into mitochondria. Import and maturation of the mitochondrial intermembrane space enzymes cytochrome b2 and cytochrome c peroxidase in intact yeast cells. J Biol Chem. 1982 Nov 10;257(21):13068–13074. [PubMed] [Google Scholar]
- Reinbothe C., Apel K., Reinbothe S. A light-induced protease from barley plastids degrades NADPH:protochlorophyllide oxidoreductase complexed with chlorophyllide. Mol Cell Biol. 1995 Nov;15(11):6206–6212. doi: 10.1128/mcb.15.11.6206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reinbothe S., Reinbothe C., Holtorf H., Apel K. Two NADPH:Protochlorophyllide Oxidoreductases in Barley: Evidence for the Selective Disappearance of PORA during the Light-Induced Greening of Etiolated Seedlings. Plant Cell. 1995 Nov;7(11):1933–1940. doi: 10.1105/tpc.7.11.1933. [DOI] [PMC free article] [PubMed] [Google Scholar]
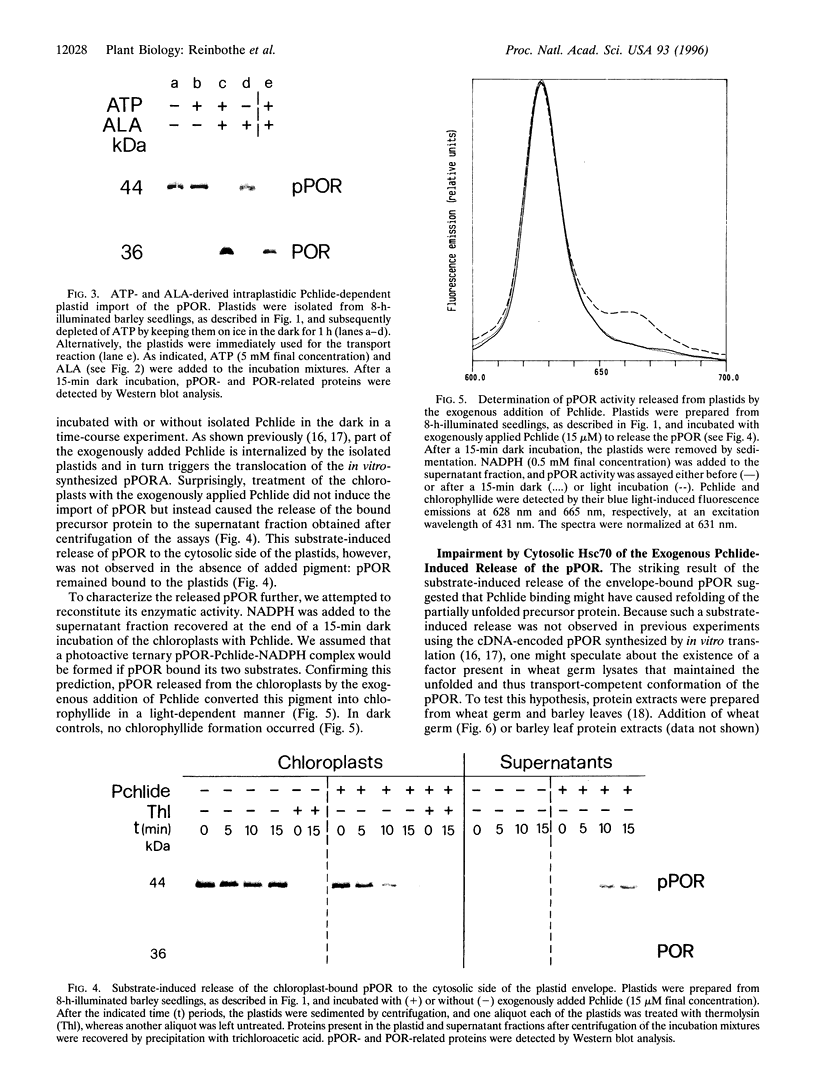
- Reinbothe S., Reinbothe C., Runge S., Apel K. Enzymatic product formation impairs both the chloroplast receptor-binding function as well as translocation competence of the NADPH: protochlorophyllide oxidoreductase, a nuclear-encoded plastid precursor protein. J Cell Biol. 1995 Apr;129(2):299–308. doi: 10.1083/jcb.129.2.299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reinbothe S., Runge S., Reinbothe C., van Cleve B., Apel K. Substrate-dependent transport of the NADPH:protochlorophyllide oxidoreductase into isolated plastids. Plant Cell. 1995 Feb;7(2):161–172. doi: 10.1105/tpc.7.2.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanders S. L., Whitfield K. M., Vogel J. P., Rose M. D., Schekman R. W. Sec61p and BiP directly facilitate polypeptide translocation into the ER. Cell. 1992 Apr 17;69(2):353–365. doi: 10.1016/0092-8674(92)90415-9. [DOI] [PubMed] [Google Scholar]
- Schiebel E., Driessen A. J., Hartl F. U., Wickner W. Delta mu H+ and ATP function at different steps of the catalytic cycle of preprotein translocase. Cell. 1991 Mar 8;64(5):927–939. doi: 10.1016/0092-8674(91)90317-r. [DOI] [PubMed] [Google Scholar]
- Schindler C., Soll J. Protein transport in intact, purified pea etioplasts. Arch Biochem Biophys. 1986 May 15;247(1):211–220. doi: 10.1016/0003-9861(86)90550-3. [DOI] [PubMed] [Google Scholar]
- Schulz R., Steinmüller K., Klaas M., Forreiter C., Rasmussen S., Hiller C., Apel K. Nucleotide sequence of a cDNA coding for the NADPH-protochlorophyllide oxidoreductase (PCR) of barley (Hordeum vulgare L.) and its expression in Escherichia coli. Mol Gen Genet. 1989 Jun;217(2-3):355–361. doi: 10.1007/BF02464904. [DOI] [PubMed] [Google Scholar]
- Simon S. M., Peskin C. S., Oster G. F. What drives the translocation of proteins? Proc Natl Acad Sci U S A. 1992 May 1;89(9):3770–3774. doi: 10.1073/pnas.89.9.3770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Theg S. M., Bauerle C., Olsen L. J., Selman B. R., Keegstra K. Internal ATP is the only energy requirement for the translocation of precursor proteins across chloroplastic membranes. J Biol Chem. 1989 Apr 25;264(12):6730–6736. [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tripathy B. C., Chakraborty N. 5-Aminolevulinic Acid Induced Photodynamic Damage of the Photosynthetic Electron Transport Chain of Cucumber (Cucumis sativus L.) Cotyledons. Plant Physiol. 1991 Jul;96(3):761–767. doi: 10.1104/pp.96.3.761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varagona M. J., Schmidt R. J., Raikhel N. V. Nuclear localization signal(s) required for nuclear targeting of the maize regulatory protein Opaque-2. Plant Cell. 1992 Oct;4(10):1213–1227. doi: 10.1105/tpc.4.10.1213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wienhues U., Becker K., Schleyer M., Guiard B., Tropschug M., Horwich A. L., Pfanner N., Neupert W. Protein folding causes an arrest of preprotein translocation into mitochondria in vivo. J Cell Biol. 1991 Dec;115(6):1601–1609. doi: 10.1083/jcb.115.6.1601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Boer D., Cremers F., Teertstra R., Smits L., Hille J., Smeekens S., Weisbeek P. In vivo import of plastocyanin and a fusion protein into developmentally different plastids of transgenic plants. EMBO J. 1988 Sep;7(9):2631–2635. doi: 10.1002/j.1460-2075.1988.tb03115.x. [DOI] [PMC free article] [PubMed] [Google Scholar]