Abstract
Background and objectives
Circulating fibroblast growth factor 23 (FGF23) is associated with adverse cardiovascular outcomes in CKD. Whether FGF23 predicts cardiovascular mortality after kidney transplantation, independent of measures of mineral metabolism and cardiovascular risk factors, is unknown.
Design, setting, participants, & measurements
The association between plasma C-terminal FGF23 and cardiovascular mortality was analyzed in a single-center prospective cohort of 593 stable kidney transplant recipients (mean age ± SD, 52±12 years; 54% male; estimated GFR, 47±16 ml/min per 1.73 m2), at a median of 6.1 (interquartile range, 2.7–11.7) years after transplantation. Multivariate Cox regression models were built, adjusting for measures of renal function and mineral metabolism; Framingham risk factors; the left ventricular wall strain markers midregional fragment of pro–A-type natriuretic peptide (MR-proANP) and N-terminal-pro brain natriuretic peptide (NT-proBNP); and copeptin, the stable C-terminal portion of the precursor of vasopressin.
Results
In multivariate linear regression analysis, MR-proANP (β=0.20, P<0.001), NT-proBNP (β=0.18, P<0.001), and copeptin (β=0.26, P<0.001) were independently associated with FGF23. During follow-up for 7.0 (interquartile range, 6.2–7.5) years, 128 patients (22%) died, of whom 66 (11%) died due to cardiovascular disease; 54 (9%) had graft failure. FGF23 was associated with an higher risk of cardiovascular mortality in a fully adjusted multivariate Cox regression model (hazard ratio [HR], 1.88 [95% confidence interval (CI), 1.11 to 3.19]; P=0.02). FGF23 was also independently associated with all-cause mortality (full model HR, 1.86 [95% CI, 1.27 to 2.73]; P=0.001). Net reclassification improved for both cardiovascular mortality (HR, 0.07 [95% CI, 0.01 to 0.14]; P<0.05) and all-cause mortality (HR, 0.11 [95% CI, 0.05 to 0.18]; P<0.001).
Conclusions
Plasma FGF23 is independently associated with cardiovascular and all-cause mortality after kidney transplantation. The association remained significant after adjustment for measures of mineral metabolism and cardiovascular risk factors.
Introduction
Kidney transplantation is the treatment of choice for patients with ESRD. As a result of improved surgical techniques, immunosuppressive therapies, and prevention of opportunistic infections, the short-term prognosis of kidney transplant recipients in particular has improved dramatically over the past decades. However, the risk of premature death due to cardiovascular disease remains greatly increased in kidney transplant recipients compared with the general population (1).
Deregulation of calcium/phosphate metabolism is common in CKD and in kidney transplant recipients with impaired renal function (2). In patients with CKD, circulating levels of fibroblast growth factor 23 (FGF23), parathyroid hormone (PTH), and phosphate have been identified as independent risk factors for cardiovascular disease and all-cause mortality (3–9). Experimental studies demonstrated that FGF23 may be directly involved in the development of left ventricular hypertrophy (LVH) (10). Whether FGF23 predicts cardiovascular mortality in renal transplant recipients is unknown.
Our primary hypothesis was that plasma FGF23 is a risk factor for cardiovascular mortality in renal transplant recipients, independent of Framingham risk factors (recipient age and sex, systolic BP, antihypertensive treatment use, smoking status, diabetes mellitus, plasma total cholesterol, and HDL cholesterol), known correlates of FGF23 (including estimated GFR [eGFR] and proteinuria [11]), and factors related to phosphate metabolism. We also addressed whether FGF23 is associated with the left ventricular wall strain markers midregional fragment of pro–A-type natriuretic peptide (ANP) and N-terminal-pro brain natriuretic peptide (NT-proBNP) and with copeptin, the stable C-terminal portion of the precursor of vasopressin (12), and whether these cardiac markers influence the association between FGF23 and cardiovascular mortality.
Materials and Methods
Study Population
In this prospective cohort study, all renal transplant recipients with a graft functioning for at least 1 year who visited our outpatient clinic (University Medical Center Groningen, The Netherlands) between August 2001 and July 2003 were eligible to participate. This study was a post hoc analysis of a previously published study (13); plasma samples from 593 patients were available. All participants provided written informed consent. Patients were enrolled at a median of 6.1 (interquartile range [IQR], 2.7–11.7) years after transplantation. Baseline visits were postponed if signs of infection were present. Patients diagnosed with cancer other than cured skin cancer were not eligible. The institutional review board approved the study protocol (METc 01/039), which was in adherence with the Declaration of Helsinki. The clinical and research activities being reported are consistent with the Principles of the Declaration of Istanbul as outlined in the Declaration of Istanbul on Organ Trafficking and Transplant Tourism.
Endpoints
The major outcome of the study was cardiovascular mortality. All-cause mortality and death-censored graft failure were secondary outcomes. Adequate collection of up-to-date data on events and mortality was ensured by our continuous surveillance system; general practitioners or referring nephrologists were contacted in case the current status of a patient was unknown. Cause of death was obtained by linking the number of the death certificate to the primary cause of death as coded by a physician, according to the International Classification of Diseases, Ninth Revision (ICD-9). Cardiovascular death was defined as ICD-9 codes 410–447 (14). Follow-up was completed until May 19, 2009. There was no loss to follow-up.
Patient Characteristics and Laboratory Measurements
Relevant transplant characteristics were taken from a database containing information on all renal transplantations performed at our center since 1968. Standard immunosuppressive treatment regimens are published previously (13). Current medication, including active vitamin D use (alfacalcidol or calcitriol); presence of diabetes mellitus; and cardiovascular history were extracted from the medical record. Diabetes mellitus was defined according to the American Diabetes Association guidelines: fasting plasma glucose ≥126 mg/dl or use of antidiabetic medication (15). Cardiovascular history was defined as a history of myocardial infarction, percutaneous transluminal angioplasty or stenting of coronary or peripheral arteries, bypass operation of coronary or peripheral arteries, intermittent claudication, amputation for vascular reasons, transient ischemic attack, or ischemic cerebrovascular accident. BP was measured as described previously (13). Smoking status was recorded with a self-report questionnaire.
Upon entry in the cohort (baseline), blood was drawn after an 8- to 12-hour overnight fasting period. EDTA plasma samples were stored at −80°C until assessment of biochemical measures for this study. Plasma C-terminal FGF23 levels were determined by sandwich ELISA (Immutopics, San Clemente, CA), with intra-assay and interassay coefficients of variation of <5% and <16%, respectively (16). Plasma creatinine concentrations were determined using a modified version of the Jaffe method (MEGA AU 510, Merck Diagnostic, Darmstadt, Germany). PTH and 1,25(OH)2 vitamin D were measured in EDTA plasma using radioimmunoassay, and 25(OH)-vitamin D levels were determined by isotope dilution–online solid phase extraction liquid chromatography–tandem mass spectrometry (17). NT-proBNP levels were measured by immunoassay on an ELECSYS2010 instrument (ELECSYS proBNP, Roche Diagnostics, Germany). MR-proANP was measured with a sandwich immunoassay (MR-proANP LIA; B.R.A.H.M.S) (18). Plasma copeptin was measured using a sandwich immunoassay (B.R.A.H.M.S GmbH/Thermo Fisher Scientific, Hennigsdorf/Berlin, Germany) (19). Serum albumin, calcium, cholesterol, C-reactive protein, glucose, hemoglobin, and phosphate and urinary phosphate, sodium, total protein, and urea were determined by routine laboratory measurements. We calculated eGFR using the CKD-Epidemiology Collaboration equation (20).
Statistical Analyses
Variable distribution was tested with histograms and probability plots. Normally distributed variables are presented as mean ± SD, and skewed variables as median (interquartile range). Baseline characteristics upon entry into the cohort were compared between tertiles of FGF23 using one-way ANOVA, Kruskal-Wallis test, and chi-squared tests as appropriate. Skewed data were natural log-transformed for correlation analysis, Cox regression, and backward and forward linear analysis.
The associations between (log-transformed) FGF23 levels and MR-proANP, NT-proBNP, and copeptin were assessed by Pearson correlation analysis. Backward linear regression was used to identify correlates of plasma FGF23 levels. The following covariates were tested: age, sex, donor type (deceased or living), history of one or more acute rejection episodes, cold ischemia time, waist circumference, cardiovascular history, Framingham risk score factors, eGFR, proteinuria, serum phosphate, 24-hour urinary phosphate excretion (representing phosphate intake), 24-hour urinary urea excretion (representing protein intake [21]), plasma 25(OH)-vitamin D, 1,25(OH)2 vitamin D, PTH, C-reactive protein, albumin, hemoglobin, MR-proANP, NT-proBNP, copeptin levels, use of angiotensin-converting enzyme inhibitor or angiotensin receptor blocker (given potential interactions between the renin-angiotensin-aldosterone system and FGF23 [22]), use of active vitamin D, and number of antihypertensive drugs. Variables significantly associated with FGF23 levels using backward linear regression analysis were subsequently tested in a forward linear regression model. Variables that were significant in this model were considered independent correlates of FGF23 levels.
Risk ratios and conditional maximum likelihood estimates of the rate ratio for cardiovascular and all-cause mortality and graft failure were calculated per tertile of FGF23. Confidence intervals were calculated using the Fisher exact test. To further analyze the association between FGF23 and outcomes, tertiles of FGF23 were plotted in Kaplan-Meier curves with a log-rank test. Subsequently, multivariate Cox regression models were built to assess the association between FGF23 and outcomes, adjusted for potential confounders. We tested for effect modifications by invoking multiplicative interaction terms. Sensitivity analyses were performed in a predefined subgroup of patients with an eGFR between 30 and 90 ml/min per 1.73 m2 (n=493).
To determine the goodness of fit of the predictive models, the Akaike information criterion (AIC) was used. AIC is a statistical estimate of the trade-off between the likelihood of a model against its complexity. A lower AIC value indicates a better model fit. Furthermore, discrimination, or the ability of the risk prediction models to distinguish those with an event (i.e., cardiovascular or all-cause mortality or graft failure) from those who do not, was evaluated with the Harrell C-index. The C-index is analogous to the area under the receiver-operating characteristic curve, for which larger values indicate better discrimination.
We assessed net reclassification improvement (NRI) for predefined risk categories of cardiovascular and all-cause mortality (<5%, 5%–10%, and >10%) and calculated integrated discrimination improvement (IDI) (23). The NRI provides reclassification tables constructed separately for study participants with and without events and quantifies the correct movement between categories: upwards for events and downward for nonevents.
Proportionality assumptions were tested using Schoenfeld tests and log-minus-log survival plots. All statistical analyses were performed using SPSS software, version 16.0 for Windows (IBM, Armonk, NY); Stata/SE software, version 11.0 for Windows (College Station, TX); and OpenEpi (openepi.com). P values <0.05 were considered to represent statistically significant differences.
Results
Patient Characteristics
Patient characteristics are summarized in Table 1. Overall, FGF23 levels were 140 (IQR, 95–219) RU/ml, and eGFR was 47±16 ml/min per 1.73 m2. None of the patients used dietary phosphate binders. Patients in the highest FGF23 tertile were older; were more likely to have received a graft from a deceased donor, to have had an acute rejection episode, and to be a smoker; had a longer cold ischemia time; had a higher waist circumference and systolic and diastolic BP; had worse renal function; had lower serum albumin, HDL cholesterol, 1,25(OH)2 vitamin D, and hemoglobin; had higher serum phosphate, NT-proBNP, MR-proANP, copeptin, PTH, and CRP levels; and had more proteinuria.
Table 1.
Clinical characteristics of the cohort
| Variable | All Patients (n=593) | Plasma FGF23 Tertiles | |||
|---|---|---|---|---|---|
| Tertile 1 (n=197) | Tertile 2 (n=198) | Tertile 3 (n=198) | P Value | ||
| Plasma C-terminal FGF23 (RU/ml) | 140 (95–219) | 83 (68–96) | 141 (124–162) | 311 (220–567) | <0.001 |
| Age (yr) | 52±12 | 50±13 | 52±12 | 53±11 | 0.04 |
| Men (%) | 54 | 56 | 51 | 55 | 0.62 |
| Whites (%) | 95 | 94 | 94 | 95 | 0.41 |
| Cause of kidney disease (%) | 0.75 | ||||
| Primary GN | 28 | 26 | 30 | 29 | |
| GN due to vasculitis/autoimmune disease | 6 | 9 | 5 | 6 | |
| Tubulointerstitial nephritis/pyelonephritis | 16 | 20 | 15 | 14 | |
| Polycystic kidney disease | 18 | 16 | 18 | 20 | |
| Hypertension | 6 | 7 | 5 | 5 | |
| Diabetes | 3 | 4 | 3 | 3 | |
| Other/unknown | 23 | 18 | 24 | 23 | |
| Dialysis duration (mo) | 27 (13–48) | 24 (13–43) | 31 (14–48) | 26 (13–52) | 0.40 |
| Deceased donor transplant (%) | 86 | 78 | 92 | 91 | <0.001 |
| Cold ischemia time (h) | 22 (15–27) | 20 (7–27) | 22 (16–26) | 22 (16–29) | 0.04 |
| Total warm ischemia time (min) | 35 (30–45) | 35 (30–45) | 35 (30–45) | 36 (30–45) | 0.92 |
| Total HLA mismatches (n) | 1 (0–2) | 1 (0–2) | 1 (0–2) | 1 (0–2) | 0.41 |
| History of acute rejection (%) | 45 | 39 | 44 | 52 | 0.04 |
| Transplant vintage (yr) | 6.1 (2.7–11.7) | 5.6 (2.4–11.1) | 5.8 (3.0–11.5) | 7.2 (3.5–12.2) | 0.06 |
| Waist circumference (cm) | 97±14 | 94±13 | 97±14 | 100±13 | <0.001 |
| Systolic BP (mmHg) | 153±23 | 148±21 | 153±22 | 157±25 | <0.001 |
| Diastolic BP (mmHg) | 90±10 | 89±10 | 89±9 | 91±10 | <0.001 |
| Current smoker (%) | 22 | 18 | 18 | 30 | <0.001 |
| Current diabetes (%) | 18 | 19 | 17 | 18 | 0.88 |
| Laboratory results | |||||
| Measurements in serum/plasma | |||||
| Creatinine (mg/dl) | 1.5 (1.3–1.9) | 1.4 (1.1–1.6) | 1.5 (1.3–1.7) | 1.9 (1.4–2.5) | <0.001 |
| Albumin (g/dl) | 4.1±0.3 | 4.1±0.3 | 4.1±0.4 | 4.0±0.3 | <0.001 |
| Calcium (mg/dl) | 9.6±0.6 | 9.5±0.6 | 9.6±0.7 | 9.6±0.7 | 0.20 |
| Total cholesterol (mg/dl) | 218±42 | 214±35 | 219±38 | 221±51 | 0.13 |
| HDL cholesterol (mg/dl) | 42±12 | 45±13 | 42±12 | 40±12 | <0.001 |
| LDL cholesterol (mg/dl) | 137±39 | 137±31 | 136±39 | 138±46 | 0.84 |
| NT-proBNP (pg/ml) | 299 (131–672) | 180 (92–419) | 285 (130–567) | 457 (240–1517) | <0.001 |
| MR-proANP (pmol/L) | 164 (103–275) | 126 (87–188) | 153 (100–231) | 257 (158–390) | <0.001 |
| Copeptin (pmol/L) | 9.3 (5.0–19.5) | 6.5 (3.8–10.4) | 8.6 (4.7–17.4) | 16.1 (7.7–30.0) | <0.001 |
| C-reactive protein (mg/L) | 2.1 (0.8–4.9) | 1.7 (0.6–3.7) | 2.1 (0.8–4.5) | 3.0 (1.2–7.8) | <0.001 |
| Glucose (mg/dl) | 87±23 | 86±22 | 87±26 | 88±20 | 0.59 |
| Phosphate (mg/dl) | 3.3±0.7 | 3.1±0.6 | 3.2±0.6 | 3.6±0.7 | <0.001 |
| Parathyroid hormone (pg/ml) | 86 (57–131) | 72 (51–112) | 88 (61–121) | 101 (68–193) | <0.001 |
| 25-OH vitamin D (ng/ml) | 21±9 | 22±10 | 21±8 | 22±10 | 0.49 |
| 1,25-OH2 vitamin D (pg/ml) | 42±18 | 47±17 | 42±19 | 35±15 | <0.001 |
| Hemoglobin (g/dl) | 13.7±1.5 | 14.2±1.3 | 13.8±1.3 | 13.1±1.7 | <0.001 |
| Estimated GFR (CKD-EPI) (ml/min per 1.73 m2) | 47±16 | 55±13 | 48±14 | 37±15 | <0.001 |
| Measurements in 24-hour urine | |||||
| Sodium excretion (mEq/24 hr) | 140±68 | 139±68 | 138±61 | 143±75 | 0.66 |
| Phosphate excretion (g/24 hr) | 2.3±0.8 | 2.3±0.9 | 2.4±0.8 | 2.2±0.8 | 0.18 |
| Proteinuria (g/24 hr) | 0.2 (0.0–0.5) | 0.2 (0.0–0.3) | 0.2 (0.0–0.4) | 0.3 (0.2–0.8) | <0.001 |
| Urea excretion (g/24 hr) | 10.5±3.2 | 10.6±3.1 | 10.9±3.2 | 10.0±3.2 | 0.03 |
| Medication | |||||
| Calcium supplements (%) | 7 | 5 | 6 | 10 | 0.07 |
| Active vitamin D (alfacalcidol or calcitriol) (%) | 9 | 8 | 5 | 14 | 0.02 |
| ACE inhibitor or ARB (%) | 34 | 30 | 34 | 39 | 0.17 |
| Prednisone dose (mg/d) | 10 (7.5–10) | 10 (7.5–10) | 10 (7.5–10) | 10 (7.5–10) | 0.58 |
| Calcineurin inhibitor (%) | 78 | 77 | 83 | 75 | 0.12 |
| Azathioprine (%) | 34 | 29 | 34 | 38 | 0.17 |
| Mycophenolate mofetil (%) | 40 | 49 | 41 | 31 | <0.001 |
| Sirolimus (%) | 2 | 2 | 2 | 2 | 0.88 |
Values expressed with a plus/minus sign are the mean ± SD; values with parentheses are median and interquartile range. All variables were determined at enrollment except sex, race, cause of the original kidney disease, cold ischemia time, total warm ischemia time, and total number of HLA mismatches, which were assessed at time of transplantation. FGF23, fibroblast growth factor 23; NT-proBNP, N-terminal-pro brain natriuretic peptide; MR-proANP, pro–A-type natriuretic peptide; CKD-EPI, CKD-Epidemiology Collaboration; ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker.
FGF23 Is Independently Associated with Cardiac Markers
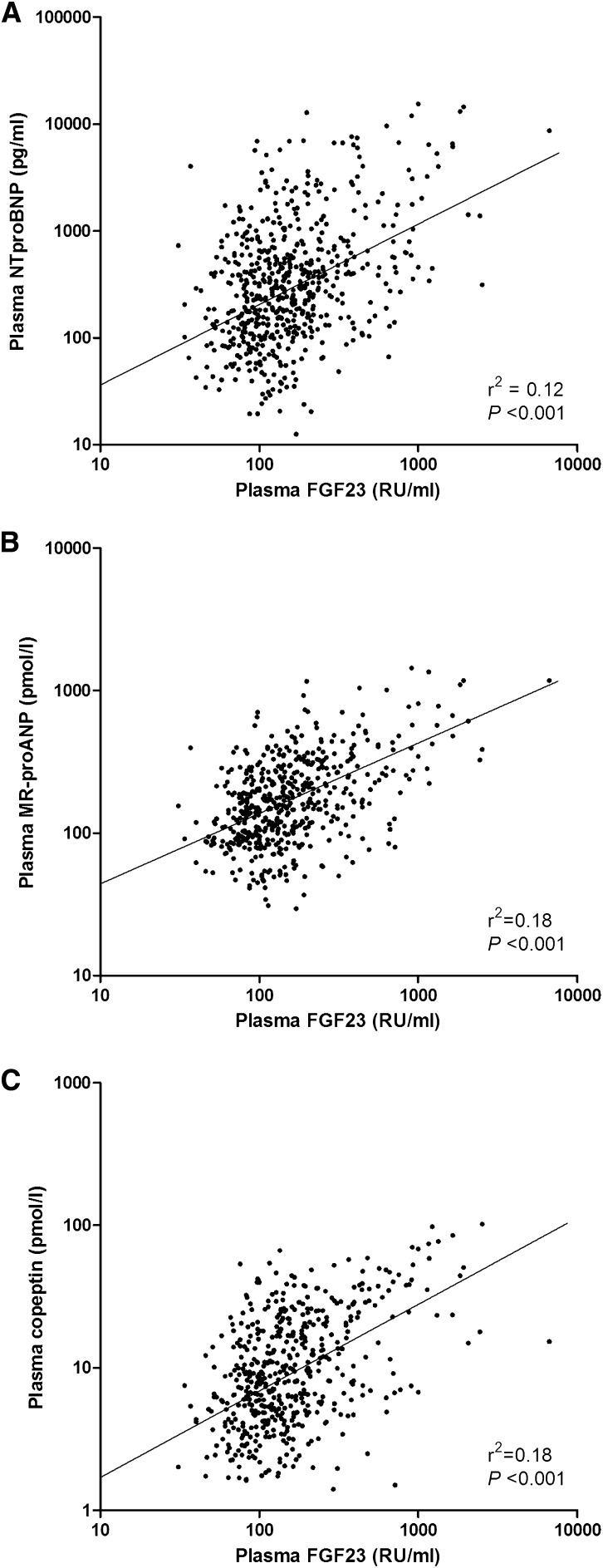
Plasma FGF23 levels were associated with NT-proBNP (r2=0.12; P<0.001), MR-proANP (r2=0.18; P<0.001), and copeptin (r2=0.18; P<0.001) levels in univariate analysis (Figure 1). Using backward and forward multivariate linear regression models, we found that copeptin was strongly and independently associated with plasma FGF23 levels (Table 2). Other independent correlates of FGF23 levels were eGFR, serum phosphate, hemoglobin, and donor type (living or deceased). Results were highly similar when, instead of copeptin, MR-proANP (coefficient, 0.20 [95% CI, 0.12 to 0.28]; P<0.001) or NT-proBNP (coefficient, 0.18 [95% CI, 0.11 to 0.25]; P<0.001) were forced into the final forward regression model.
Figure 1.
Scatter diagrams illustrating the associations between fibroblast growth factor 23 (FGF23) and (A) N-terminal-pro brain natriuretic peptide (NT-proBNP), (B) pro–A-type natriuretic peptide (MR-proANP), and (C) copeptin.
Table 2.
Multivariate linear regression analysis for correlates of plasma fibroblast growth factor 23 levels
| Variable | Coefficient for FGF23 (95% CI) | P Value |
|---|---|---|
| eGFR (CKD-EPI) | −0.32 (−0.41 to −0.23) | <0.001 |
| Ln copeptin | 0.26 (0.17 to 0.35) | <0.001 |
| Serum phosphate | 0.15 (0.07 to 0.23) | <0.001 |
| Hemoglobin | −0.14 (−0.22 to −0.06) | <0.001 |
| Donor type (0=living; 1=deceased) | −0.10 (−0.18 to −0.03) | 0.004 |
Excluded from the model: age, sex, history of one or more acute rejection episodes, cold ischemia time, waist circumference, cardiovascular history, systolic and diastolic BP, smoking status, diabetes mellitus, total cholesterol, proteinuria, 24-urinary phosphate and urea excretion, plasma 25(OH)-vitamin D, 1,25(OH)2 vitamin D, C-reactive protein, albumin, parathyroid hormone, use of angiotensin-converting enzyme inhibitor or angiotensin receptor blocker, use of active vitamin D, and total number of antihypertensive drugs. Coefficients are standardized β values. Model fit: R2=0.41. FGF23, fibroblast growth factor 23; CI, confidence interval; eGFR, estimated GFR; CKD-EPI, CKD-Epidemiology Collaboration; Ln; natural log.
Baseline Plasma FGF23 and Outcomes
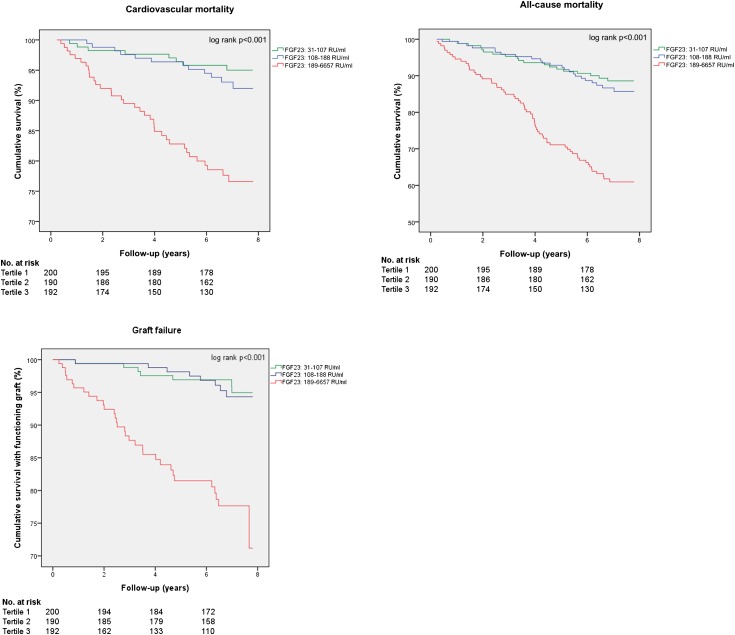
During a median follow-up of 7.0 (IQR, 6.2–7.5) years, 128 patients (22%) died, 66 (11%) of cardiovascular disease; 54 (9%) had graft failure. Kaplan-Meier survival plots demonstrated a higher risk of cardiovascular mortality, all-cause mortality, and graft failure per increasing tertile of plasma FGF23 (Figure 2). Risk ratios for cardiovascular mortality, all-cause mortality, and graft failure were significantly increased in the highest FGF23 tertile (Table 3).
Figure 2.
Kaplan-Meier survival plots of fibroblast growth factor 23 (FGF23) tertiles and cardiovascular mortality. Higher FGF23 levels are associated with an increased incidence of cardiovascular mortality.
Table 3.
Events and risk ratios for cardiovascular and all-cause mortality and graft failure per fibroblast growth factor 23 tertile
| Variable | All Patients | Plasma FGF23 Tertiles | P Valuea | ||
|---|---|---|---|---|---|
| Tertile 1 | Tertile 2 | Tertile 3 | |||
| Cardiovascular mortality | |||||
| Events/patient-years | 66/3722 | 11/1343 | 17/1260 | 38/1119 | |
| Incidence rate/1000 person-years | 17.7 | 8.2 | 13.5 | 34.0 | |
| Risk ratio (95% CI) | Reference | 1.65 (0.73 to 3.89) | 4.15 (2.08 to 8.99) | <0.001 | |
| All-cause mortality | |||||
| Events/patient-years | 128/3722 | 24/1343 | 33/1260 | 71/1119 | |
| Incidence rate/1000 person-years | 34.4 | 17.9 | 26.2 | 63.4 | |
| Risk ratio (95% CI) | Reference | 1.47 (0.84 to 2.59) | 3.55 (2.21 to 5.90) | <0.001 | |
| Graft failure | |||||
| Events/patient-years | 54/3722 | 8/1343 | 8/1260 | 38/1119 | |
| Incidence rate/1000 person-years | 14.5 | 6.0 | 6.3 | 34.0 | |
| Risk ratio (95% CI) | Reference | 1.07 (0.35 to 3.26) | 5.70 (2.62 to 14.15) | <0.001 | |
FGF23, fibroblast growth factor 23; CI, confidence interval.
For trend.
In Cox regression models, the highest tertile of FGF23 was consistently associated with a higher risk of cardiovascular mortality after adjustment for Framingham risk factors, history of cardiovascular disease, measures of renal function, phosphate metabolism, and cardiac wall stress (Table 4). To assess whether the association between FGF23 and cardiovascular mortality was influenced by other factors, interaction analyses were performed. We found no evidence for interaction in the association between FGF23 and cardiovascular mortality by eGFR (P for interaction=0.51), proteinuria (P=0.62), 24-hour urinary phosphate excretion (P=0.50), fractional phosphate excretion (P=0.87), or any Framingham risk factor. In addition, no significant interaction by NT-proBNP (P for interaction=0.26), MR-proANP (P=0.32), or copeptin (P=0.19) was observed. NT-proBNP and MR-ANP but not copeptin were independently associated with cardiovascular mortality in the full model (data not shown). The final models provided the best fit (lowest AICs); all models displayed good discrimination as suggested by relatively high Harrell C-indices (Table 4). As a sensitivity analysis, full multivariate Cox regression models were repeated in a subgroup of patients with an eGFR between 30 and 90 ml/min per 1.73 m2 (n=493). In this subgroup, plasma FGF23 remained an independent risk factor for cardiovascular mortality (full model hazard ratio [HR], 2.43 [95% CI, 1.19 to 4.93] per 1 SD of natural log [ln]FGF23; P=0.01) and all-cause mortality (HR, 2.12 [95% CI, 1.29 to 3.49] per 1 SD of lnFGF23; P=0.003).
Table 4.
Cox regression analyses for cardiovascular and all-cause mortality and graft failure per fibroblast growth factor 23 tertile or per 1 SD of natural log fibroblast growth factor 23
| Variable | Plasma FGF23 Tertiles | Natural Log FGF23 | ||||||
|---|---|---|---|---|---|---|---|---|
| Tertile 1 | Tertile 2 | Tertile 3 | P Valuea | Per 1 SD | P Value | AIC | Harrell C-Index | |
| Cardiovascular mortality, HR (95% CI) | ||||||||
| Model 1 | Reference | 1.66 (0.78 to 3.54) | 4.24 (2.17 to 8.29) | <0.001 | 1.97 (1.52 to 2.55) | <0.001 | 645 | 0.706 |
| Model 2 | Reference | 1.62 (0.75 to 3.50) | 4.02 (1.90 to 8.50) | <0.001 | 1.98 (1.44 to 2.74) | <0.001 | 648 | 0.706 |
| Model 3 | Reference | 1.39 (0.55 to 3.50) | 4.20 (1.90 to 9.81) | <0.001 | 1.93 (1.33 to 2.80) | 0.001 | 800 | 0.801 |
| Model 4 | Reference | 1.59 (0.58 to 4.14) | 4.06 (1.55 to 9.98) | 0.003 | 2.27 (1.37 to 3.75) | 0.001 | 479 | 0.810 |
| Model 5 | Reference | 1.86 (0.67 to 5.18) | 2.96 (1.06 to 8.26) | 0.04 | 1.88 (1.11 to 3.19) | 0.02 | 467 | 0.843 |
| All-cause mortality, HR (95% CI) | ||||||||
| Model 1 | Reference | 1.47 (0.87 to 2.48) | 3.64 (2.29 to 5.78) | <0.001 | 1.99 (1.65 to 2.39) | <0.001 | 1245 | 0.694 |
| Model 2 | Reference | 1.36 (0.80 to 2.32) | 2.94 (1.73 to 4.98) | <0.001 | 1.86 (1.46 to 2.36) | <0.001 | 1237 | 0.693 |
| Model 3 | Reference | 1.03 (0.55 to 1.93) | 3.15 (1.69 to 5.85) | <0.001 | 1.93 (1.45 to 2.56) | <0.001 | 1542 | 0.768 |
| Model 4 | Reference | 1.06 (0.53 to 2.11) | 2.63 (1.32 to 5.24) | 0.004 | 2.18 (1.50 to 3.18) | <0.001 | 869 | 0.778 |
| Model 5 | Reference | 1.17 (0.58 to 2.36) | 2.23 (1.11 to 4.49) | 0.02 | 1.86 (1.27 to 2.73) | 0.001 | 858 | 0.799 |
| Graft failure, HR (95% CI) | ||||||||
| Model 1 | Reference | 1.06 (0.40 to 2.83) | 6.29 (2.92 to 13.44) | <0.001 | 3.37 (2.56 to 4.42) | <0.001 | 515 | 0.762 |
| Model 2 | Reference | 0.54 (0.20 to 1.46) | 1.15 (0.47 to 2.84) | 0.41 | 1.49 (0.99 to 2.24) | 0.06 | 463 | 0.861 |
| Model 3 | Reference | 0.53 (0.18 to 1.57) | 0.80 (0.29 to 2.24) | 0.99 | 1.15 (0.73 to 1.82) | 0.55 | 540 | 0.885 |
| Model 4 | Reference | 0.35 (0.10 to 1.22) | 0.39 (0.12 to 1.30) | 0.25 | 1.00 (0.59 to 1.71) | 0.99 | 327 | 0.907 |
| Model 5 | Reference | 0.32 (0.09 to 1.21) | 0.37 (0.11 to 1.32) | 0.25 | 1.03 (0.59 to 1.78) | 0.93 | 331 | 0.909 |
Model 1: crude model; model 2: adjusted for estimated GFR (CKD-Epidemiology Collaboration formula) and proteinuria; model 3: model 2 + adjusted for cardiovascular disease history, Framingham risk factors (recipient age and sex, systolic BP, antihypertensive treatment use, smoking status, diabetes mellitus, plasma total cholesterol, and HDL cholesterol), and transplant vintage; model 4: model 3 + adjusted for serum phosphate, 1,25(OH)2-vitamin D, parathyroid hormone levels, and active vitamin D use; model 5: model 4 + adjusted for N-terminal-pro brain natriuretic peptide, pro–A-type natriuretic peptide, and copeptin. HR, hazard ratio; CI, confidence interval; AIC, Akaike information criterion.
P for linear trend.
Higher FGF23 levels were also associated with a higher risk of all-cause mortality, both when analyzed per tertile and as continuous variable (Table 4). Higher tertiles of FGF23 were also associated with a higher risk of death-censored graft failure in crude analyses, but significance was lost after adjustment for eGFR and proteinuria (Table 4). There was no significant interaction between the risk of graft failure and cardiovascular mortality (P=0.38).
Comparative Analyses
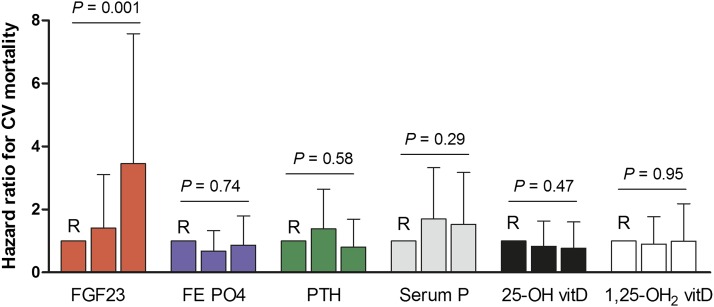
To compare the performance of FGF23 with other measures of mineral metabolism (fractional phosphate excretion, PTH, phosphate, 25-OH vitamin D or 1,25[OH]2 vitamin D) as individual predictors of cardiovascular mortality, separate Cox regression analyses were performed for each of the six exposures, adjusted for age, sex, cardiovascular history, eGFR, proteinuria, and Framingham risk factors. As indicated in Figure 3, FGF23 (highest tertile HR, 3.45 [95% CI, 1.57 to 7.58]; P=0.001) but not the other exposures were strongly associated with cardiovascular mortality.
Figure 3.
Comparative analysis of fibroblast growth factor 23 (FGF23), fractional phosphate excretion (FE PO4), parathyroid hormone, phosphate (P), 25(OH)-vitamin D, or 1,25(OH)2 vitamin D as independent risk factors for cardiovascular (CV) mortality. The model was adjusted for age, sex, cardiovascular history, estimated GFR, proteinuria, and Framingham risk factors. For each exposure, the lowest tertile served as the reference group (R).
NRI and IDI
The NRI was calculated to assess the putative clinical utility of FGF23 as a marker of (cardiovascular) mortality in addition to Framingham risk factors. Reclassification improved mainly in the patients who survived, where adding FGF23 to the model resulted in reclassification into a lower-risk category (Table 5). Overall, net reclassification improved for both cardiovascular mortality (0.07 [95% CI, 0.01 to 0.14]; P<0.05]) and all-cause mortality (0.11 [95% CI, 0.05 to 0.18]; P<0.001). Accordingly, the IDI, which does not rely on prespecified risk categories but represents a continuous measure, was 0.03 (95% CI, 0.01 to 0.05; P=0.004) for cardiovascular mortality and 0.05 (95% CI, 0.03 to 0.07; P<0.001) for all-cause mortality.
Table 5.
Reclassification based on fibroblast growth factor 23 levels
| Without FGF23 | With FGF23 (n) | Total (n) | ||
|---|---|---|---|---|
| <5% | 5%–10% | >10% | ||
| Estimated risk of cardiovascular mortalitya | ||||
| Patients with cardiovascular mortality | ||||
| <5% | 2 | 1b | 3 | |
| 5%–10% | 6 | 6 | ||
| >10% | 2c | 55 | 57 | |
| Total | 2 | 9 | 55 | 66 |
| Patients without cardiovascular mortality | ||||
| <5% | 186 | 12c | 1c | 199 |
| 5%–10% | 46b | 75 | 15c | 136 |
| >10% | 27b | 15 | 178 | |
| Total | 232 | 114 | 167 | 513 |
| Estimated risk of all-cause mortalityd | ||||
| Patients with all-cause mortality | ||||
| <5% | 1 | 1b | 1b | 3 |
| 5%–10% | 3c | 3 | 2b | 8 |
| >10% | 3c | 114 | 117 | |
| Total | 4 | 7 | 117 | 128 |
| Patients without all-cause mortality | ||||
| <5% | 92 | 6c | 98 | |
| 5%–10% | 24b | 41 | 9c | 74 |
| >10% | 5b | 44b | 230 | 279 |
| Total | 121 | 91 | 239 | 451 |
Multivariate model adjusted for Framingham risk factors (recipient age and sex, systolic BP, antihypertensive treatment use, smoking status, diabetes mellitus, plasma total cholesterol and HDL cholesterol) with or without plasma C-terminal fibroblast growth factor 23 level. FGF23, fibroblast growth factor 23.
Net reclassification improvement was 0.07 (95% confidence interval [CI], 0.01 to 0.14; P<0.05).
Numbers of patients who were correctly reclassified by the model with FGF23.
Numbers of patients who were incorrectly reclassified by the model with FGF23.
Net reclassification improvement was 0.11 (95% CI, 0.05 to 0.18; P<0.001).
Discussion
This study identified high plasma FGF23 levels as an independent risk factor for cardiovascular mortality in kidney transplant recipients. The association was consistent in regression models adjusted for renal function, measures of mineral metabolism, and cardiovascular risk factors. The significantly improved NRI suggests that FGF23 levels may have an additional value to Framingham risk factors to predict the risk of cardiovascular mortality in renal transplant recipients.
Our findings are in line with previous data linking high FGF23 levels with incident cardiovascular disease and mortality in the CKD population (3,24,25), and with a higher risk of all-cause mortality after kidney transplantation (26). Furthermore, FGF23 levels have been associated with established cardiovascular risk factors and with a higher risk of cardiovascular events in the general population (27,28). Although patients in the highest FGF23 tertile were characterized by an overall high cardiovascular risk profile, the associations between FGF23 and (cardiovascular and all-cause) mortality remained significant after adjustment for several major cardiovascular risk markers, including Framingham risk factors, suggesting that FGF23 is a strong and independent risk marker. The potential clinical relevance of FGF23 in addition to established risk markers is further supported by improved reclassification (NRI and IDI). Furthermore, FGF23, but not serum phosphate, PTH, or vitamin D, was independently associated with cardiovascular mortality (Figure 3), suggesting a specific role for FGF23. The observed association between elevated FGF23 levels and a higher cardiovascular risk initially positioned FGF23 as a biomarker of phosphate toxicity. More recently, animal studies demonstrated that FGF23 has "off-target" effects, directly contributing to the development of LVH (10). We found that FGF23 was independently associated with MR-proANP and NT-proBNP, both markers of left ventricular wall strain, and with copeptin, a stable peptide derived from the vasopressin precursor (29). All three cardiac markers are used as markers of heart failure (30). Although these associations are in line with a potential role for FGF23 in LVH, we could not demonstrate modulation of the association between FGF23 and cardiovascular mortality by these cardiac markers. Furthermore, only a small amount of FGF23 was explained by copeptin, MR-proANP, and NT-proBNP (Figure 1), suggesting that FGF23 is independently related to cardiovascular and all-cause mortality.
In a previous study, the association between FGF23 and cardiovascular events was modified by sex (31). We could not confirm this effect modulation, which could be explained by differences in renal function between the Heart and Soul study (eGFR, 71±23 ml/min per 1.73 m2) and our cohort (eGFR, 47±16 ml/min per 1.73 m2). Data from 24-hour urine collections allowed us to include phosphate intake (assessed by 24-hour urinary phosphate excretion) (32) and excretion (fractional phosphate excretion) in our analyses. Neither measure interacted with the association between FGF23 levels and cardiovascular mortality.
FGF23 was also associated with a higher risk of graft failure in univariate analysis, in line with a previous study (26). However, the association was no longer significant after adjustment for eGFR and proteinuria in addition to known risk factors for graft failure (Table 3). We hypothesize that renal function after transplantation has a stronger effect on the risk of graft failure than on the risk of mortality (33,34).
Possible limitations of our study include its post hoc nature and the use of ICD-9 codes rather than an independent adjudication committee to define the endpoint cardiovascular mortality. Although we adjusted for several confounders, including measures of renal function, which appeared to be the most important determinant of FGF23 levels, the possibility of residual confounding cannot be fully excluded because not all determinants of FGF23 are currently known. The sensitivity analyses in patients with an eGFR of 30–90 ml/min per 1.73 m2 reduces the risk of confounding by renal function. The fact that diabetic and hypertensive nephropathy were under-represented in our cohort, and that our cohort consisted almost entirely of white patients, limits the generalizability of our study. Because FGF23 was measured only at a single time point in this cohort, we could not perform a repeated-measures analysis. Prevalent cohorts such as this may suffer from survivorship biases; these generally lead to an underestimation of the hazard ratio (35). Using a relatively large number of covariates in the final Cox regression model might result in overfitting the model; however, the stepwise Cox models and the low AIC in the final model suggest that overfitting was no issue in our analyses. Finally, we did not have extensive structural or functional cardiac data (e.g., from echocardiography) available to further explore the mechanisms behind the observed association between FGF23 and cardiovascular mortality. Strong points of this study, on the other hand, are the use of 24-hour urine collections, which enabled us to adjust for proteinuria and phosphate excretion, the availability of multiple markers of cardiovascular risk, and the complete follow-up.
In conclusion, to our knowledge this is the first study to identify FGF23 as an independent risk factor for cardiovascular mortality after kidney transplantation. Although it may be relevant to consider therapies reducing FGF23 levels (36) to improve cardiovascular outcomes, it should be kept in mind that high FGF23 levels may serve an important physiologic goal, namely to keep phosphate balance. A recent study demonstrated that specific FGF23 blockade with a neutralizing antibody did reduce secondary hyperparathyroidism but increased serum phosphate, aortic calcification, and mortality (37). Whether specific reduction of FGF23 or rather reducing phosphate load may improve cardiovascular prognosis after kidney transplantation should be addressed in future prospective studies.
Disclosures
None.
Acknowledgments
The authors would like to thank Wendy Dam for excellent technical assistance.
M.H.D.B. is supported by personal development grants from the Dutch Kidney foundation (KJPB.08.07) and the University Medical Center Groningen (Mandema stipend). This work is supported by a consortium grant from the Dutch Kidney Foundation (NIGRAM consortium, grant no. CP10.11). The NIGRAM consortium consists of the following principal investigators: Piet ter Wee and Marc Vervloet (VU University Medical Center, Amsterdam, The Netherlands); René Bindels and Joost Hoenderop (Radboud University Medical Center Nijmegen, The Netherlands); and Gerjan Navis, Jan-Luuk Hillebrands, and Martin de Borst (University Medical Center Groningen, The Netherlands).
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
References
- 1.Jardine AG, Gaston RS, Fellstrom BC, Holdaas H: Prevention of cardiovascular disease in adult recipients of kidney transplants. Lancet 378: 1419–1427, 2011 [DOI] [PubMed] [Google Scholar]
- 2.Larsson T, Nisbeth U, Ljunggren O, Jüppner H, Jonsson KB: Circulating concentration of FGF-23 increases as renal function declines in patients with chronic kidney disease, but does not change in response to variation in phosphate intake in healthy volunteers. Kidney Int 64: 2272–2279, 2003 [DOI] [PubMed] [Google Scholar]
- 3.Ärnlöv J, Carlsson AC, Sundström J, Ingelsson E, Larsson A, Lind L, Larsson TE: Higher fibroblast growth factor-23 increases the risk of all-cause and cardiovascular mortality in the community. Kidney Int 83: 160–166, 2013 [DOI] [PubMed] [Google Scholar]
- 4.Kestenbaum B, Sampson JN, Rudser KD, Patterson DJ, Seliger SL, Young B, Sherrard DJ, Andress DL: Serum phosphate levels and mortality risk among people with chronic kidney disease. J Am Soc Nephrol 16: 520–528, 2005 [DOI] [PubMed] [Google Scholar]
- 5.Fliser D, Kollerits B, Neyer U, Ankerst DP, Lhotta K, Lingenhel A, Ritz E, Kronenberg F, Kuen E, König P, Kraatz G, Mann JF, Müller GA, Köhler H, Riegler P, MMKD Study Group : Fibroblast growth factor 23 (FGF23) predicts progression of chronic kidney disease: The Mild to Moderate Kidney Disease (MMKD) Study. J Am Soc Nephrol 18: 2600–2608, 2007 [DOI] [PubMed] [Google Scholar]
- 6.Gutiérrez OM, Januzzi JL, Isakova T, Laliberte K, Smith K, Collerone G, Sarwar A, Hoffmann U, Coglianese E, Christenson R, Wang TJ, deFilippi C, Wolf M: Fibroblast growth factor 23 and left ventricular hypertrophy in chronic kidney disease. Circulation 119: 2545–2552, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gutiérrez OM, Mannstadt M, Isakova T, Rauh-Hain JA, Tamez H, Shah A, Smith K, Lee H, Thadhani R, Jüppner H, Wolf M: Fibroblast growth factor 23 and mortality among patients undergoing hemodialysis. N Engl J Med 359: 584–592, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kovesdy CP, Ahmadzadeh S, Anderson JE, Kalantar-Zadeh K: Secondary hyperparathyroidism is associated with higher mortality in men with moderate to severe chronic kidney disease. Kidney Int 73: 1296–1302, 2008 [DOI] [PubMed] [Google Scholar]
- 9.Tonelli M, Curhan G, Pfeffer M, Sacks F, Thadhani R, Melamed ML, Wiebe N, Muntner P: Relation between alkaline phosphatase, serum phosphate, and all-cause or cardiovascular mortality. Circulation 120: 1784–1792, 2009 [DOI] [PubMed] [Google Scholar]
- 10.Faul C, Amaral AP, Oskouei B, Hu MC, Sloan A, Isakova T, Gutiérrez OM, Aguillon-Prada R, Lincoln J, Hare JM, Mundel P, Morales A, Scialla J, Fischer M, Soliman EZ, Chen J, Go AS, Rosas SE, Nessel L, Townsend RR, Feldman HI, St John Sutton M, Ojo A, Gadegbeku C, Di Marco GS, Reuter S, Kentrup D, Tiemann K, Brand M, Hill JA, Moe OW, Kuro-O M, Kusek JW, Keane MG, Wolf M: FGF23 induces left ventricular hypertrophy. J Clin Invest 121: 4393–4408, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vervloet MG, van Zuilen AD, Heijboer AC, ter Wee PM, Bots ML, Blankestijn PJ, Wetzels JF, MASTERPLAN group study : Fibroblast growth factor 23 is associated with proteinuria and smoking in chronic kidney disease: An analysis of the MASTERPLAN cohort. BMC Nephrol 13: 20, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Struck J, Morgenthaler NG, Bergmann A: Copeptin, a stable peptide derived from the vasopressin precursor, is elevated in serum of sepsis patients. Peptides 26: 2500–2504, 2005 [DOI] [PubMed] [Google Scholar]
- 13.de Vries AP, Bakker SJ, van Son WJ, van der Heide JJ, Ploeg RJ, The HT, de Jong PE, Gans RO: Metabolic syndrome is associated with impaired long-term renal allograft function; not all component criteria contribute equally. Am J Transplant 4: 1675–1683, 2004 [DOI] [PubMed] [Google Scholar]
- 14.Zelle DM, Corpeleijn E, van Ree RM, Stolk RP, van der Veer E, Gans RO, Homan van der Heide JJ, Navis G, Bakker SJ: Markers of the hepatic component of the metabolic syndrome as predictors of mortality in renal transplant recipients. Am J Transplant 10: 106–114, 2010 [DOI] [PubMed] [Google Scholar]
- 15.American Diabetes Association: Diagnosis and classification of diabetes mellitus. Diabetes Care 31[Suppl 1]: S55–S60, 2008 [DOI] [PubMed] [Google Scholar]
- 16.Heijboer AC, Levitus M, Vervloet MG, Lips P, ter Wee PM, Dijstelbloem HM, Blankenstein MA: Determination of fibroblast growth factor 23. Ann Clin Biochem 46: 338–340, 2009 [DOI] [PubMed] [Google Scholar]
- 17.Doorenbos CR, de Cuba MM, Vogt L, Kema IP, van den Born J, Gans RO, Navis G, de Borst MH: Antiproteinuric treatment reduces urinary loss of vitamin D-binding protein but does not affect vitamin D status in patients with chronic kidney disease. J Steroid Biochem Mol Biol 128: 56–61, 2012 [DOI] [PubMed] [Google Scholar]
- 18.van Hateren KJ, Alkhalaf A, Kleefstra N, Groenier KH, de Jong PE, de Zeeuw D, Gans RO, Struck J, Bilo HJ, Gansevoort RT, Bakker SJ: Comparison of midregional pro-A-type natriuretic peptide and the N-terminal pro-B-type natriuretic peptide for predicting mortality and cardiovascular events. Clin Chem 58: 293–297, 2012 [DOI] [PubMed] [Google Scholar]
- 19.Morgenthaler NG, Struck J, Alonso C, Bergmann A: Assay for the measurement of copeptin, a stable peptide derived from the precursor of vasopressin. Clin Chem 52: 112–119, 2006 [DOI] [PubMed] [Google Scholar]
- 20.Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF, 3rd, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J, CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration) : A new equation to estimate glomerular filtration rate. Ann Intern Med 150: 604–612, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mitch WE: Beneficial responses to modified diets in treating patients with chronic kidney disease. Kidney Int Suppl (94): S133–S135, 2005 [DOI] [PubMed] [Google Scholar]
- 22.de Borst MH, Vervloet MG, ter Wee PM, Navis G: Cross talk between the renin-angiotensin-aldosterone system and vitamin D-FGF-23-klotho in chronic kidney disease. J Am Soc Nephrol 22: 1603–1609, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pencina MJ, D’Agostino RBS, Sr, D’Agostino RB, Jr, Vasan RS: Evaluating the added predictive ability of a new marker: From area under the ROC curve to reclassification and beyond. Stat Med 27: 157–172, discussion 207–212, 2008 [DOI] [PubMed] [Google Scholar]
- 24.Seiler S, Reichart B, Roth D, Seibert E, Fliser D, Heine GH: FGF-23 and future cardiovascular events in patients with chronic kidney disease before initiation of dialysis treatment. Nephrol Dial Transplant 25: 3983–3989, 2010 [DOI] [PubMed] [Google Scholar]
- 25.Kendrick J, Cheung AK, Kaufman JS, Greene T, Roberts WL, Smits G, Chonchol M, HOST Investigators : FGF-23 associates with death, cardiovascular events, and initiation of chronic dialysis. J Am Soc Nephrol 22: 1913–1922, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wolf M, Molnar MZ, Amaral AP, Czira ME, Rudas A, Ujszaszi A, Kiss I, Rosivall L, Kosa J, Lakatos P, Kovesdy CP, Mucsi I: Elevated fibroblast growth factor 23 is a risk factor for kidney transplant loss and mortality. J Am Soc Nephrol 22: 956–966, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gutiérrez OM, Wolf M, Taylor EN: Fibroblast growth factor 23, cardiovascular disease risk factors, and phosphorus intake in the health professionals follow-up study. Clin J Am Soc Nephrol 6: 2871–2878, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ärnlöv J, Carlsson AC, Sundström J, Ingelsson E, Larsson A, Lind L, Larsson TE: Serum FGF23 and risk of cardiovascular events in relation to mineral metabolism and cardiovascular pathology. Clin J Am Soc Nephrol 8: 781–786, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Abbasi A, Corpeleijn E, Meijer E, Postmus D, Gansevoort RT, Gans RO, Struck J, Hillege HL, Stolk RP, Navis G, Bakker SJ: Sex differences in the association between plasma copeptin and incident type 2 diabetes: The Prevention of Renal and Vascular Endstage Disease (PREVEND) study. Diabetologia 55: 1963–1970, 2012 [DOI] [PubMed] [Google Scholar]
- 30.Sabatine MS, Morrow DA, de Lemos JA, Omland T, Sloan S, Jarolim P, Solomon SD, Pfeffer MA, Braunwald E: Evaluation of multiple biomarkers of cardiovascular stress for risk prediction and guiding medical therapy in patients with stable coronary disease. Circulation 125: 233–240, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ix JH, Chonchol M, Laughlin GA, Shlipak MG, Whooley MA: Relation of sex and estrogen therapy to serum fibroblast growth factor 23, serum phosphorus, and urine phosphorus: The Heart and Soul Study. Am J Kidney Dis 58: 737–745, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Vervloet MG, van Ittersum FJ, Büttler RM, Heijboer AC, Blankenstein MA, ter Wee PM: Effects of dietary phosphate and calcium intake on fibroblast growth factor-23. Clin J Am Soc Nephrol 6: 383–389, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Schnitzler MA, Lentine KL, Gheorghian A, Axelrod D, Trivedi D, L’Italien G: Renal function following living, standard criteria deceased and expanded criteria deceased donor kidney transplantation: impact on graft failure and death. Transpl Int 25: 179–191, 2012 [DOI] [PubMed] [Google Scholar]
- 34.Yarlagadda SG, Coca SG, Formica RN, Jr, Poggio ED, Parikh CR: Association between delayed graft function and allograft and patient survival: A systematic review and meta-analysis. Nephrol Dial Transplant 24: 1039–1047, 2009 [DOI] [PubMed] [Google Scholar]
- 35.Arrighi HM, Hertz-Picciotto I: The evolving concept of the healthy worker survivor effect. Epidemiology 5: 189–196, 1994 [DOI] [PubMed] [Google Scholar]
- 36.Wolf M: Update on fibroblast growth factor 23 in chronic kidney disease. Kidney Int 82: 737–747, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shalhoub V, Shatzen EM, Ward SC, Davis J, Stevens J, Bi V, Renshaw L, Hawkins N, Wang W, Chen C, Tsai MM, Cattley RC, Wronski TJ, Xia X, Li X, Henley C, Eschenberg M, Richards WG: FGF23 neutralization improves chronic kidney disease-associated hyperparathyroidism yet increases mortality. J Clin Invest 122: 2543–2553, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]