Abstract
Airborne particulate matter (PM) from urban vehicular aerosols altered glutamate receptor functions and induced glial inflammatory responses in rodent models after chronic exposure. Potential neurotoxic mechanisms were analyzed in vitro. In hippocampal slices, 2 h exposure to aqueous nanosized PM (nPM) selectively altered postsynaptic proteins in CA1 neurons: increased GluA1, GluN2A, and GluN2B, but not GluA2, GluN1 or mGlur5; increased PSD95 and spinophilin, but not synaptophysin, while dentate gyrus (DG) neurons were unresponsive. In hippocampal slices and neurons, MitoSOX red fluorescence was increased by nPM, implying free radical production. Specifically, NO• production by slices was increased within 15 min of exposure to nPM with dose dependence, 1–10 µg/ml. Correspondingly, CA1 neurons exhibited increased nitrosylation of the GluN2A receptor and dephosphorylation of GluN2B (S1303) and of GluA1 (S831 & S845). Again, DG neurons were unresponsive to nPM. The induction of NO• and nitrosylation were inhibited by AP5, an NMDA receptor antagonist, which also protects neurite outgrowth in vitro from inhibition by nPM. Membrane injury (EthidiumD-1 uptake) showed parallel specificity. Finally, nPM decreased evoked excitatory postsynaptic currents (EPSCs) of CA1 neurons. These findings further document the selective impact of nPM on glutamatergic functions and identify novel responses of NMDA receptor-stimulated NO• production and nitrosylation reactions during nPM-mediated neurotoxicity.
Keywords: Air pollution, NMDA, glutamate, nitric oxide, nitrosylation, CA1 neurons
INTRODUCTION
Urban air pollution adversely impacts brain functions in human populations and animal models. Emerging findings show associations of airborne pollutant levels with mild cognitive impairments (Calderon-Garciduenas et al. 2008, Chen & Schwartz 2009, Weuve et al. 2012, Power et al. 2011, Power et al. 2013). Brains from a highly polluted city had premature inflammation and neurodegeneration (Block & Calderon-Garciduenas 2009). Rodents chronically exposed to particulate matter (PM) from diesel engines or urban traffic emissions also developed glial inflammatory responses (Levesque et al. 2011a,b, Levesque et al, 2013, Morgan et al. 2011, Kleinman et al. 2008, Win-Shwe & Fujimaki 2011) and oxidative stress with protein nitrosylation (Levesque et al. 2011b) and lipid peroxidation (Zanchi et al. 2010). Exposure of rats to diesel exhaust also impaired memory functions associated with the hippocampus (Fonken et al. 2011, Win-Shwe et al. 2012).
The basis for hippocampal memory impairments from inhalation of urban air pollutants could include glutamate receptors, which are altered in rodent models by exposure to nPM from diesel exhaust (Win-Shwe et al. 2009) or by nPM of <0.2 µm fractioned from Los Angeles urban freeway air in our prior study (Morgan et al. 2011). The nano-sized PM (ultrafine PM) from combustion engines has consistently shown higher toxicity than larger PM in vivo, e.g. (Li et al. 2013) and in vitro (Li et al. 2003; Gillespie et al. 2013). Inhalation of nPM for 150 h during 10 weeks decreased hippocampal levels of the GluA1 subunit of AMPA receptors (Morgan et al. 2011). The selectivity of responses to nPM is indicated by the absence of changes in GluA2 levels or in the associated synaptic proteins PSD95 or synaptophysin.
In vitro primary hippocampal neuronal cultures also showed inhibition of neurite outgrowth during exposure to nPM at 2 µg/ml for 48 h, an effect rescued by the NMDA receptor antagonist AP5 (Morgan et al. 2011). nPM induced LDH release by hippocampal slice cultures, a measure of cell damage, which was also rescued by AP5. Inhibitory effects of nPM on neurite outgrowth may share mechanisms with the regression of hippocampal CA1 and CA3 dendrites after in vivo exposure to vehicular-derived PM of 2.5 µm size (Fonken et al. 2011).
To further analyze the mechanisms of nPM on glutamatergic functions, we examined the effects of acute nPM on synaptic proteins in hippocampal slices and dissociated neurons. nPM cross cell membranes by non-phagocytic mechanisms (Geiser et al. 2005) consistent with their relatively high hydrophobicity (Xia et al. 2006). Because nPM rapidly induced free radicals in macrophages (Li et al. 2003, Xia et al. 2006), we investigated free radical production in slices and neuronal cultures, with emphasis on nitric oxide (NO•). Glutamatergic subunit nitrosylation was examined because NMDA receptors are vulnerable to oxidative damage (Aizenman et al. 1989, Aizenman et al. 1990, Manzoni et al. 1992, Shi et al. 2013). Neuronal selectivity was assessed by comparing effects of nPM on CA1 pyramidal neurons, which are more vulnerable than DG neurons to nPM toxicity (Fonken et al. 2011), ischemia (Kawasaki et al. 1990) and Alzheimer disease (Morrison & Hof 1997). To evaluate functional outcomes of nPM, synaptic transmission was examined by patch clamp recording.
MATERIAL AND METHODS
Animals
Male C57BL/6J mice (1 month) were purchased from Jackson Laboratories (Sacramento, CA) and pregnant Sprague-Dawley rats from Harlan Labs (Livermore, CA). Animals were maintained and treated using animal procedures conforming to NIH guidelines as approved by the USC Institutional Animal Care & Use Committee (IACUC). Animals were euthanized after isoflurane anesthesia.
nPM collection
Nanoscale particulate matter (nPM <200 nm) was collected continuously for 30 days in a well-studied site in downtown Los Angeles next to the CA-I110 Freeway (Sardar et al. 2005), which has a yearly-average of 10.5±2.9 ug/m3, ranging 6.1 to 15.6 ug/m3 across months (Daher et al. 2013). Using a High-Volume Ultrafine Particle (HVUP) Sampler at 400 L/min (Misra et al. 2002, Ning et al. 2007), nPM were trapped on Teflon filters (20 × 25.4 cm, PTFE, 2 µm pore; Pall Life Sciences, Port Washington, NY). nPM was then transferred to sterile aqueous suspension by vortexing and sonication, with differential elution of components (Morgan et al. 2011)(see Discussion below). Aqueous suspensions of nPM pooled from 30 days of collection as stocks of ca. 200 µg/ml, which were aliquoted and stored at −20 °C. The frozen stocks retain markers of chemical stability for >3 months (Li et al. 2003, Morgan et al. 2011). Endotoxin was not detected in nPM stocks (Limulus amoebocyte assay) (Lonzo Biologics, Hopkinton, MA).
Acute hippocampal slices
Slices were prepared from 1 month old male mice in ice-cold modified artificial cerebrospinal fluid (aCSF) containing 124 mM sucrose, 62 mM NaCl, 3 mM KCl, 3 mM MgCl2, 1.25 mM CaCl2, 26 mM NaHCO3, 1.25 mM NaH2PO4, 10 mM glucose. Transverse hippocampal slices (400 µm) were cut by a Vibratome-1000 (Vibratome Co., St Louis, MO) or a McIlwain tissue chopper (Brinkmann Instruments Inc., Westbury, NY). Slices were transferred to aCSF (124 mM NaCl, 3 mM KCl, 1.5 mM MgCl2, 2.5 mM CaCl2, 26 mM NaHCO3, 1.25 mM NaH2PO4, 10 mM glucose), with pre-incubation for 30 min at 30 °C. The aCSF was continuously bubbled with 95% O2/5% CO2 in the standard electrophysiological paradigm (see below). Following pre-incubation, slices from 3–4 brains were transferred to fresh aCSF (10 slices per 20 ml) at 22 °C, to which nPM suspensions were added. Some experiments included the NMDA antagonist AP5 (D,L-2-amino-5-phosphonopentanoic acid; Sigma Chemical Corp., St. Louis, MO). After 2 h incubation, slices were prepared for patch clamp electrophysiology recordings or Western blot (see below).
Dissociated hippocampal neuronal cultures
Primary hippocampal neuronal cultures were derived from embryonic day 18 (E18) rats. Briefly, hippocampi were dissociated in Hank’s balanced salt medium containing trypsin and DNase at 37 °C (Banker & Cowan 1977). Dissociated cells were plated on poly-D-lysine and laminin coated glass coverslips (20,000 cell/cm2), or on 96 well plates (70,000 cell/cm2); media were Dulbecco’s Modified Eagle Medium (DMEM), supplemented with B27 (Invitrogen, Grand Island, NY). Neurons were cultured for 7 or 14 days at 37 °C with 5% CO2 and exposed to nPM and AP5 (50 µM) for 2 h.
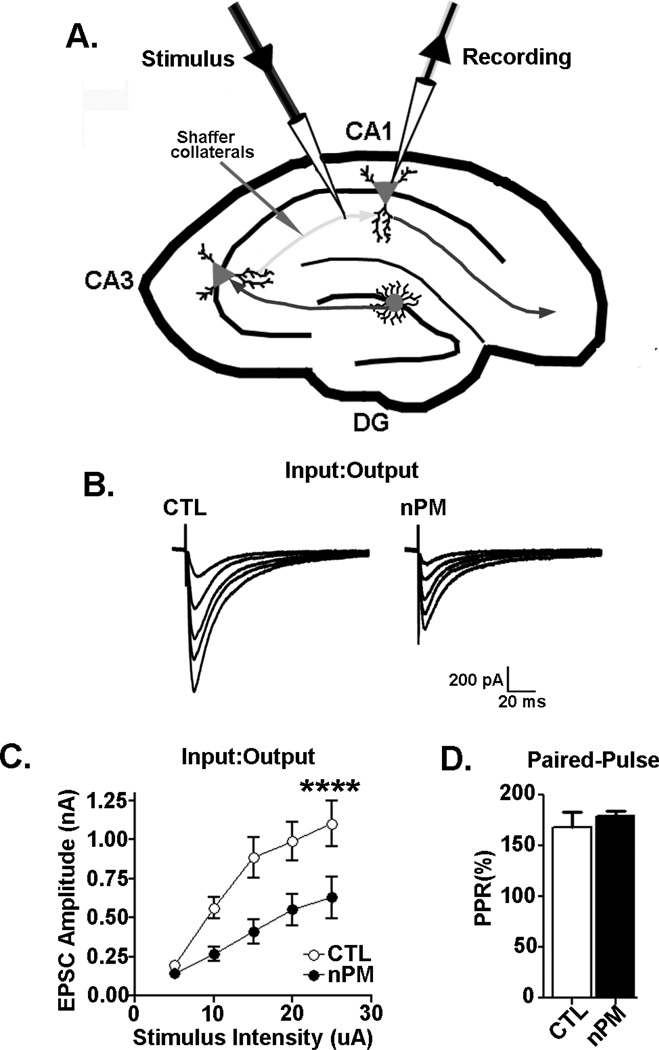
Electrophysiology
After exposure to the nPM suspension or control aCSF (2 h/22 °C), individual hippocampal slices were transferred to a recording chamber at 35 °C (Akopian et al. 2008) and superfused with artificial cerebral spinal fluid (aCSF) at 2 ml/min. Individual CA1 pyramidal neurons were recorded by standard whole-cell voltage clamp methods (Akopian & Walsh 2002) (See Figure 5A). Evoked synaptic currents were recorded using a Multiclamp 700B amplifier and Digidata 1440A (Molecular Devices, Sunnyvale, CA). Patch pipettes were filled with 120 nM CsMeSO4, 10 mM CsCl, 5 mM EGTA, 2 mM MgCl2 10 mM HEPES, 5 mM QX-314 (Sodium channel inhibitor), 2 mM ATP-Mg, 0.25 mM GTP-Na, pH 7.25, 285 mOsm, and spermine (0.1mM). Resistance ranged from 2 to 4 MΩ.
Figure 5. nPM reduces excitatory synaptic function in CA1 neurons.
(A) Schematic of whole cell path clamp recording of CA1 neurons. (B–C) Input/output (I/O) responses of evoked excitatory postsynaptic currents (EPSC) in CA1 perikarya. (B) EPSC traces from CTL and nPM treated CA1 neurons. (C) An ascending sequence of extracellular stimuli at 20 sec intervals was delivered through stimulation of Schaffer collaterals; slices were preincubated with 10 µg/ml nPM for 2 h (C) nPM consistently lowered EPSC amplitude (nPM effect, p<0.0001). (D) The paired-pulse response (PPR) ratio of pre:postsynaptic terminals in the same slices (50 msec intervals at 15 uA); PPR was not altered by nPM.
CA1 pyramidal cells were identified under IR illumination. Passive membrane properties were determined in voltage clamp mode with the “Membrane Test” option of the Clampex 10 software (Molecular Devices, Sunnyvale, CA) by 10 mV depolarizing voltage steps. Series resistance (Rs) was compensated up to 80%. Schaffer collaterals were stimulated by a glass pipette (1 mM NaCl) 100–150 µm from the recorded CA1 neurons. Extracellular stimulation was generated with Master 8 pulse generator, delivered by Iso-Flex stimulus isolation unit (AMPI, Israel).
Synaptic input-output (I/O) curves of synaptic currents were obtained by stimulating stratum radiatum of each slice with ascending stimulus intensities (5–25 µA). I/O curves were plotted as amplitudes of evoked EPSCs vs stimulus intensities of evoked EPSCs. The paired pulse ratio (PPR) was estimated from the average of 5 paired pulse synaptic stimulations with interstimulus intervals of 50 ms every 20 sec. Cells were voltage-clamped at a membrane potential of −60 mV. The PPR (% of the ratio of the second pulse to the first) estimated changes in presynaptic release. The I/O and PPR of CA1 neurons were based on 6–7 neurons measured per condition.
Free radicals
Free radical production was estimated in slices or neurons after 2 h of nPM (10 µg/ml) by MitoSox™ Red (Molecular Probes, Grand Island, NY); MitoSox [2.5 µm] was added to aCSF (slices) or DMEM (neurons) for 15 min (Ma et al. 2011). Fluorescent intensity was quantified in neuronal perikarya by ImageJ software (National Institute of Health) (Figure 3A, S4) and expressed as the mean fluorescent intensity per neuron (N=3–4 cultures per condition) or per hippocampal region (N=7 slices per condition).
Figure 3. MitoSox RED oxidant responses to nPM in hippocampal slices.
Hippocampal slices were incubated with 10 µg/ml nPM for 2 h.
(A) Live slice Mitosox™ RED fluorescence; scale bar, 600 µm. Left panel shows intense fluorescence of neuronal layers against weaker signal from the glial rich neuropil. Center and right panels, CA1 neurons at higher magnification; scale bars, 100 & 40 µm.
(B) Fluorescence in live slices increased above CTL in all neuronal regions: CA1, +50%, p<0.0001; CA2/3, +45%, p<0.01; DG, +35%, p<0.001. Data are shown as the mean fluorescent intensity by % CTL per hippocampal region (N=7 slices per condition).
(C–E) Fixed slices co-stained with Mitosox RED and cell-type markers (green):
(C) neurons (NeuN) have much stronger signal than (D) astrocytes (GFAP) and (E) microglia (IbA1); scale bar, 100 µm.
For nitric oxide (NO•), acute slices were incubated in aCSF (see above) with nPM for 15–60 min. Media were analyzed spectrophotometrically for NO• as nitrite by the Griess reagent [0.1% v/v N-(1-naphthyl) ethylenediamine dihydrochloride, 1% v/v sufanilamide, and 2.5% v/v phosphoric acid; (Sigma)](Xie et al. 2002, Ignarro et al. 1993) using NaNO2 as a standard and normalized to background [nitrite] in aCSF; 4–6 experiments per condition (N=10 slices per experiment).
Cell viability
Cell viability was assessed in hippocampal slices (N=8–12 per condition) by the LIVE/DEAD® assay (Molecular Probes, Grand Island, NY) (Stein et al. 2004). After incubation with nPM, 2 µM Calcein AM and 4 µM EthD-1 were added for 15 min. Wet mounts of slices were directly imaged. EthD-1 positive cells were enumerated in hippocampal regions using Image J software. For primary cultures (N=21–38 per experiment), neurons were co-stained with Calcein AM (Molecular Probes, viable cells) and EthD-1 (injured cells). Viability was expressed as the ratio of injured cells to viable cells.
Immunochemistry
Following nPM treatment, acute hippocampal slices and neuronal cultures were fixed with 4% v/v paraformaldehyde in phosphate buffered saline pH 7.4 (PBS). Subsequently, slices were submerged in 30% sucrose/PBS pH 7.4, then embedded in OTC before sectioning on a cryostat. Tissue sections (30 µm) were permeablized with 1% NP-40/PBS and immunoprobed for 2 h at 22 °C for markers of neurons (NeuN), astrocytes (GFAP), and microglia (Iba1) (Table S1). Immunofluorescence was visualized using Alexa Fluor® antibodies (Molecular Probes, Grand Island, NY). For primary cultures, coverslips were washed and permeablized with 0.2% triton x-100 then immunoprobed with antibodies against glutamate receptors (GluA1, GluN2b) and synaptic proteins (PSD95, spinophillin, synaptophysin) overnight at 4°C (Table S1).
Receptor proteins
After incubation (see above), hippocampal slices were microdissected on a chilled platform into two portions containing the CA1 and DG neuronal regions, with excision of the CA2/3 pyramidal neuron layer and the subiculum. Three microdissected slices per condition were pooled and homogenized in RIPA buffer (EMD Millipore Billerica, MA) by a teflon pestle, followed by centrifugation 10,000 xG/10 min at 4°C. Total supernatant protein concentration was assayed by BCA (Thermo Scientific, Rockford, IL) with bovine serum albumin (BSA) as a standard. For Western blots, 15–20 µg of protein in Laemmli loading buffer (Boston BioProducts, Ashland, MA) was electrophoresed on 8% SDS-polyacrylamide gels, and then by transferred to polyvinylidene fluoride membranes (EMD Millipore, Billerica, MA). Membranes were blocked in 5% BSA for 1 h, and then probed with primary antibodies against AMPA receptor subunits (GluA1, GluA2), metabotropic receptor (mGluR5), NMDA receptor subunits (GluN1, GluN2A, GluN2B) and synaptic proteins (PSD95, spinophillin, synaptophysin) overnight at 4°C, then incubated with secondary antibodies conjugated with an infrared dye (LI-COR Biosciences, Lincoln, NE) (Table S1). Protein levels were analyzed by densitometry using synaptophysin or actin as a loading control as the average of 3–4 experiments, with 3–4 wells per condition (CTL, nPM).
To measure posttranslational modifications on CA1 proteins, membranes were first probed with antibodies to detect-S-nitrosylation (SNO-cysteine) or phosphorylation (GluA1, S831 & S845; GluN2B, S1303). After detection, membranes were stripped and reprobed with antibodies for total proteins. Nitrosylation and phosphorylation were expressed as the ratio of modified to total levels. The specificity of nitrosylation was confirmed by immunoprecipitation of the protein extract (0.1 ml, 1 mg/ml) with 10 µg of antibodies to GluN2A or GluA1, using protein g-Sepharose beads (Pierce, Thermal Scientific) (Ryan et al. 2013); after incubation for 2 h /4 °C, the bead-bound proteins were collected by centrifugation (800g × 10 min), washed in RIPA buffer, and eluted at 99 °C /5 min in Laemmli loading buffer, followed by electrophoresis and Western blotting.
Statistics
Statistical analysis used Prism Version 5 (Graph Pad, La Jolla, CA) and Clampfit 10 software (Molecular Devices, Sunnyvale, CA). Multiple comparisons used ANOVA with Tukey posttest. Single comparison used Student’s t-test. Data are expressed as mean ± SEM; significance level of alpha = 0.05.
RESULTS
Glutamate receptor subunit modification
Following 2 h incubation of mouse hippocampal slices in 10 µg/ml nPM, the CA1 and DG neuronal regions were analyzed for glutamate receptor subunits and synaptic proteins (Figures 1 & 2; Table S2). The Western blot bands corresponded to expected sizes (Fig. S1). In the CA1 region, levels of NMDA receptor subunits were increased selectively: GluN2A (+60%) and GluN2B (+80%) (Figure 1A,B), whereas GluN1 levels did not change (Figure 1C). AMPA receptor subunits also responded selectively with increased GluA1 (+70%) (Figure 1D), but no change in GluA2 (Figure 1E) or in the metabotropic receptor GluR5 (Figure 1F). DG neurons did not show these receptor protein responses (Figure S1; Table S2).
Figure 1. Glutamate receptor protein responses to acute nPM.
Western blots of lysates from the CA1 region of mouse hippocampal slices, microdissected after incubation with nPM (10 µg/ml, 2 h) (A–C) NMDA subunits: (A) GluN2A (+60%); (B) GluN2B (+70%); (C) GluN1, no change; (D–E) AMPA subunits: (D) GluA1 (+70%), (E) GluA2, no change; (F) mGluR5, no change; (G–I) Receptor phosphorylation: (G) GluN2B, pSer1303 (−30%); (H) GluA1, pS831 (−40%); (I) GluA1 pS845 (−40%). Each panel represents the average of 3–4 experiments, with 3–4 wells per condition (CTL, nPM), each containing 3 slices. Values from blots were normalized for synaptophysin, which is unchanged by nPM (Fig. 2). Data is expressed as % CTL per experiment. *p<0.05; ** p<0.01; ***p<0.0001.
Figure 2. CA1 synaptic protein response to acute nPM.
Synaptic and nuclear proteins (Western blot) of the microdissected CA1 region of mouse hippocampal slices incubated with nPM (10 µg/ml, 2 h) (A–B) Postsynaptic: (A) PSD95 (+100%); (B) spinophilin (120 kDa and 96 kDa, total and cleaved) (+40); (C) Presynaptic: synaptophysin (no change); (D) NeuN (neuronal nuclear protein)(no change); Data were obtained and expressed as described in Fig. 1. *p<0.05; ***, p<0.001.
Receptor phosphorylation per subunit was decreased by nPM exposure: GluN2B (NMDA subunit) at S1303 (−30%) (Figure 1G) and GluA1 at S831 and S845 (−40%) (Figure 1 H, I). Postsynaptic proteins associated with NMDA receptors were increased: PSD95 (+100%) (Figure 2A), spinophilin (+50%) (Figure 2B); there were no changes in synaptophysin (Figure 2C) or NeuN (Figure 2D). Astrocyte GFAP and microglial/macrophage CD11b were also unchanged by 2 h nPM treatment (not shown).
To further document acute nPM selectivity on pyramidal neurons these findings were extended to dissociated rat hippocampal neurons in culture by immunocytochemistry (Figure S3). nPM increased levels of GluN2B (+20%) (Figure S3A) as in slices; however, GluA1 decreased (−20%) (Figure S3B), which is in the opposite direction from the slice response. Other responses of neurons paralleled those of slices: PSD95 (+20%) (Figure S3C) and spinophilin (+30%) (Figure S3D), but not synaptophysin (Figure S3E). The phosphorylation of GluN2B (S1303) and GluA1 (S831 & S845) was also decreased (not shown). These responses were attenuated by AP5 (NMDA receptor antagonist) in cultured neurons, whereas in slices AP5 did not consistently attenuate nPM effects on total receptor subunit levels.
Free radicals and nitrosylation
Because excessive glutamatergic function increases oxidative stress (Reyes et al. 2012) and because nPM increased free radical production in monocytic cells (Li et al. 2003, Xia et al. 2006), we evaluated oxidant levels with MitoSOX™ red, a mitochondrially targeted ethidium dye that was responsive to increased superoxide and other oxidants induced in lung epithelial cells by urban PM (Zhao et al. 2008). In both hippocampal slices (Figure 3) and dissociated primary neurons (Figure S3), nPM induced MitoSox fluorescence by 40–50%. Hippocampal neuronal layers gave stronger signals than adjacent neuropil (Figure 3). In dissociated neurons, response to nPM was consistently blocked by AP5 (Figure S2), whereas slice responses to nPM + AP5 were inconsistent (not shown).
Nitric oxide (NO•) was a candidate for free radical responses because inhalation of diesel exhaust particles increased brain protein nitrotyrosine (Levesque et al. 2011b). NO• in hippocampal slices exposed to nPM (1, 5, 10 µg/ml) was increased by 20-fold within 15 min (Figure 4A). Again, the induction was blocked by AP5 (Figure 4B). Correspondingly, there were 50% increases of S-nitrosylation of cysteine residues on GluN2A and GluA1 (Figure 4C). The specificity of glutamate receptor nitrosylation was further documented by immunoprecipitation (Figure S3). GAPDH nitrosylation was also increased (+50%)(Figure 4C). Protein s-nitrosylation was greatest in the CA1 region (Table S2). By ethidium uptake (EthD-1), neuronal membrane damage was greater in CA1 than in DG (Figure 4D). In cultured neurons, EthD-1 uptake showed dose-dependent increase in response to nPM, again blocked by AP5 (Figure S4).
Figure 4. NO• and protein nitration responses to nPM.
(A) Induction of NO•, assayed as nitrite in the media of mouse hippocampal slices during incubation with nPM at 1, 5, 10 µg/ml, which induced dose-dependent nitrite production at 15–60 min (Griess reaction). (B) AP5 attenuated nitrite increase by 50% after incubation with nPM 10 µg nPM/ml for 15 or 60 min; (C) In whole hippocampal slices, S-nitrosylation of GluN2A, GluA1, and GAPDH (+50%) was increased 40–50% after incubation with 10 µg nPM/ml for 2 h. The increased S-nitrosylation was blocked by AP5.
(D) LIVE/DEAD® (EthD-1) uptake as a measure of neuronal injury in hippocampal slices after incubation with 10 µg nPM/ml for 2 h (scale bar, 100 µm): CA1 neurons, +50%; no change in CA2/3 or DG neurons; *p<0.05; **p<0.01; ***p<0.001; ****p<0.0001.
Effects of nPM on excitatory postsynaptic currents
Functional consequences of these biochemical changes were explored by analyzing excitatory postsynaptic currents (EPSCs) in hippocampal slices (Figure 5A). Exposure to nPM (10 µg/ml, 2 h) reduced EPSC amplitudes by about 50% in CA1 neurons across a range of stimulus intensities (Input/Output, Figure 5B,C). There were no effects of nPM on CA1 neuron membrane resistance or capacitance (not shown). Moreover, the paired pulse facilitation of CA3 presynaptic Schaeffer collateral terminals was not altered by nPM (Figure 5D), suggesting that nPM did not alter presynaptic transmitter release.
DISCUSSION
We report three new findings on rapid neuronal damage by nPM derived from vehicular traffic using in vitro models of acute hippocampal slices and dissociated neurons. (1) Hippocampal slices responded rapidly to nPM with dose-dependent increases of nitric oxide (NO•) within 15 min. (2) Nitrosylation of several glutamate receptor subunits was increased by 2 h, while phosphorylation of other sites was decreased. In both slice and neuronal cultures, levels of postsynaptic proteins PSD95 and spinophilin were increased. Several changes induced by nPM were blocked by the NMDA receptor antagonist AP5. (3) The amplitude of excitatory postsynaptic currents in CA1 neurons was decreased, while paired-pulse facilitation was unchanged. These findings document that acute exposure to nPM can alter properties of glutamate receptors that are critical to neuronal plasticity and memory processes. These findings suggest mechanisms that contribute to cognitive impairments associated with vehicular-derived pollutants and to the vulnerability of CA1 neurons to excitotoxicity in Alzheimer disease and ischemia (see Introduction).
Dose responses to nPM corresponded to prior studies with these locally derived nPM, in the range of 1–10 µg nPM/ml for in vitro brain cell models (Morgan et al. 2011) and macrophages (Xia et al. 2006). Other PM sources collected from diesel exhaust (Levesque et al. 2011b) or from urban Baltimore (Zhao et al. 2009) were not as active in cytokine induction at levels <10 µg/ml on macrophages or bronchial epithelial cells, respectively.
The components of nPM that induced NO• and altered glutamate receptors could include redox active metals, water soluble organic carbon (WSOC), and long-lived free radicals that persist for 30 days after initial collection (Morgan et al. 2011). Relative to ambient nPM, the present filter-extracted nPM had similar levels of WSOC and redox active metals, but relatively less black carbon, polyaromatic hydrocarbons, steranes, and organic acids because of differential extraction from filters by sonication (Morgan et al. 2011 and its Suppl Table 2).
NO• production in hippocampal slice cultures was increased within 15 min of exposure to nPM, followed by nitrosylation of proteins at 2 h. The rapid increase of NO• within 15 min may be the most rapid free radical response to combustion engine-derived PM in refereed reports. For comparison of the time course, macrophages (RAW 264.7 murine line) exposed to 10 µg nPM/ml had increased H2O2 production at 30 min (Li et al. 2002), followed at 60 min by decreased mitochondrial membrane depolarization (ΔΨm), and then increased MitoSox fluorescence at 2 h of exposure; mitochondrial swelling arose by 4 h, followed at 16 h by loss of cristae and increased mitochondrial [Ca2+](Xia et al. 2006). Note that these prior reports used alternate terminology (ultrafine particles, UFP) which were collected at the same site by co-author Sioutas (Morgan et al. 2011). Although the size range of UFP was similar to the present nPM, they may have differed in chemical composition because they were not extracted from filters (see above). Since this report was submitted, Gillespie et al. (2013) observed increased NO• after 24 h incubation of dopaminergic neurons with 8 µg/ml. Responses to nPM may prove to be even faster than 15 min because nPM appear to cross cell membranes directly without endocytosis (Xia et al. 2006). Thus, the mitochondrial [Ca2+] influx may begin earlier than the 16 h observation time of Xia et al. (2006).
Other ROS and oxidants may be induced by nPM besides NO•. As shown for a different urban PM sample by Zhao et al. (2008), we observed that nPM increased the oxidation of mitochondrially-targeted hydroethidine (also called MitoSOX™) by about 50%. Although the increased fluorescence of Mito-SOX™ may indicate increased superoxide, it can also be caused by other mitochondrial derived radicals and oxidants (Zielonka & Kalyanaraman 2010). The subsequent induction of the Nrf2-dependent detoxifying enzymes observed after 10 weeks of in vivo exposure (Zhang et al. 2012) may thus involve multiple oxidants and free radicals.
In the present acute responses, the blockade of NO• production by the NMDA receptor antagonist AP5 has implications for the mechanisms underlying toxic effects of nPM. Activation of either AMPA or NMDA receptors in hippocampal slices rapidly increased NO• production in CA1 neurons within 5 min (Frade et al. 2009). Moreover, NO• production in neurons depended on extra-cellular Ca2+ and recruitment of the PSD95/NOS complex to postsynaptic NMDA receptors (Sattler et al. 1999). In vivo, glutamate and NMDA infusion also rapidly increased NO• levels more in CA1 neurons than DG neurons (Laurenco et al. 2011). Further studies are needed to resolve the role of Ca2+, glutamate, and nNOS in the rapid induction of NO• in relation to possible neuronal depolarization by nPM.
Biochemical modifications of glutamate receptor subunits were also observed. The S-nitrosylation of cysteine residues in GluA1 and GluN2A was increased by 50% after 2 h exposure to nPM in CA1; again, DG neurons were unresponsive. The blockade of S-nitrosylation by AP5 is consistent with its inhibition of NO• induction. Concurrently, nPM decreased phosphorylation of GluA1 (pS831 and pS845) and of GluN2B (pS1303) in slices and neuronal cultures. Because phosphorylation regulates glutamate receptor trafficking to synapses (Santos et al. 2009), the decreased phosphorylation in response to nPM could be a factor in the increased levels of GluA1 and GluN2B. However, the opposite directions of nitrosylation and phosphorylation responses to nPM were not predicted by the induction of both nitrosylation and phosphorylation in GluA1 by glutamate agonists, in which phosphorylation of S831-GluA1 required C875 nitrosylation (Selvakumar et al. 2013). Further studies may address possible oxidative damage by nPM to cysteines or other residues, which may directly modulate phosphorylation; alternatively, indirect actions through phosphatases or kinases may occur.
By Western blots of hippocampal slices, postsynaptic proteins were selectively altered by acute nPM, with increased GluA1 (but not GluA2 or mGluR5), and of GluN2A and GluN2B (but not of GluN1). PSD95 and spinophilin (but not synaptophysin) were also increased. The direction of these changes is consistent with rapid increases of GluN2A and of spine puncta in cultured hippocampal neurons at 70 min after depolarizing pulses of KCl (Baez et al. 2013). Notably, these KCl induced increases of GluN2A were blocked by inhibitors of RNA and protein synthesis. Thus, early responses of synaptic protein levels to nPM may be mediated by gene expression as well as by posttranslational mechanisms of nitrosylation and phosphorylation.
Cytotoxicity from nPM is also indicated by the nitrosylation of GAPDH measured in whole slice proteins. Excitotoxic glutamate levels also increased GAPDH nitrosylation (Hara et al. 2005). By EthD-1 uptake, CA1 neurons showed the greatest membrane damage, which may contribute to their decreased EPSC. This ranking follows the in vivo induction of NO• by depolarizing KCl to a greater extent in CA1 than DG neurons (Baez et al. 2013) and the well-known relative vulnerability of CA1 neurons to ischemia and Alzheimer disease.
Hippocampal neuron cultures responded to nPM in parallel with the hippocampal slices for decreased phosphorylation of GluA1 at S831 and S845. In both slice and neuron cultures, nPM increased PSD95 and spinophilin. In neuron cultures, these responses to nPM were blocked by AP5; because the E18 neurons had limited synapse formation after 7 days of culture, we suggest that AP5 was acting on extrasynaptic NMDA receptors. However, levels of GluA1 protein decreased in cultured neurons, but increased in slices. This divergence may be attributed to the earlier developmental stage of dissociated neuron cultures, whereas the trisynaptic hippocampal circuit was fully formed in slices from 1 month old mice. The neuronal cultures also do not model glial-neuron interactions, as observed in conditioned media from mixed glia exposed to nPM, which altered neurite outgrowth (Morgan et al. 2011). The TNF-alpha secretion induced in mixed glia by nPM is a candidate for modulation of neurite outgrowth (Chang et al. 2012), and for modulation of AMPA receptor subunits (Santello & Volterra 2012)
These electrophysiological studies indicate that nPM can directly alter postsynaptic functions in CA1 pyramidal neurons, which showed the greatest membrane vulnerability by EthD-1 assay. Thus, acute exposure to nPM caused a large reduction (50%) of evoked EPSC amplitudes across a range of stimulus intensity with minor immediate impact on pre-synaptic neurotransmitter release. We suggest a role for the impaired phosphorylation of GluN2B receptor, which in turn phosphorylates GluA1 for trafficking and insertion into the postsynaptic membrane. A postsynaptic location of these changes is indicated by the normal paired pulse facilitation at Schaeffer collateral synapses, implying maintenance of presynaptic transmitter release. The paired-pulse plasticity expressed with synapses activated at short interstimulus intervals is a measure of neurotransmitter release (Fioravante & Regehr 2011). An alternative mechanism, which was not investigated, could involve effects of post-synaptically generated NO• on the excitability of pre-synaptic terminals. Pilot data suggest greater alteration of NMDA receptor currents (Davis et al. 2012). Because of the importance of NO•–dependent GluA1 phosphorylation to memory (Treynelis et al. 2010), we predict that long-term potentiation LTP will be impaired by nPM exposure. For example, LTP is impaired in the mouse GluA1 S845A mutant that cannot be phosphorylated (Lee et al. 2010).
To develop a working model, we need to know if nPM exposure induces a cascade starting with Ca+2 influx from nPM that causes glutamate release, leading to induction of NO•. The role of long-lived free radicals in the nPM suspension (Morgan et al. 2011) to the induced cellular free radicals also remains unknown. Synapse-independent glutamate release can occur at neuritic growth cones (Gelsomino et al. 2013, Soeda et al. 1997). Further downstream may be remodeling of glutamate receptors through nitrosylation and phosphorylation of subunits via pathways that are recognized in LTP. This cascade is applicable to in vivo responses to chronic nPM inhalation initiated by neurons and glial in the olfactory mucosa, which project into the forebrain (Block & Calderon-Garciduenas 2009) and which transport synthetic nPM as far as the hippocampus and cerebellum (Oberdorster et al. 2004).
The present in vitro models do not address the putatively slower adaptive responses to nPM during chronic exposure in vivo, in which GluA1 was decreased (Morgan et al. 2011), in contrast to GluA1 increases from acute in vitro exposure. The present in vitro models of hippocampal slice and neuronal cultures give a basis for dissecting acute effects of nPM on neurons and glia of the nasal mucosa neuroepithelium and the olfactory bulb, which are the initial brain cell contacts of inhaled nPM (Block & Calderon-Garciduenas 2009, Oberdörster et al. 2004).
Lastly, we note potential links of nPM to brain aging and Alzheimer disease (AD) through glutamatergic functions and the amyloid β-peptide (Aβ). Human brains from a highly polluted city showed premature elevations of Aβ (Calderon-Garciduenas et al. 2012). In a rat model, chronic inhalation of diesel exhaust PM2.5 increased brain amyloid β-peptide (endogenous rat Aβ) (Levesque et al 2011a). Ongoing epidemiological studies address possible associations of air pollution with AD and other dementias. Moreover, glutamate receptors have direct links to Aβ in rodent models. In AD transgenic (ADtg) mice carrying human Aβ, activation of metabotropic glutamate receptors promoted synaptic release of Aβ (Kim et al. 2010), whereas the FDA-approved, NMDA receptor antagonist Memantine decreased the brain Aβ load (Alley et al. 2010). Furthermore, activation of extrasynaptic NMDA receptors (predominantly GluN2B) increased NO• and caused synapse loss (Talantova et al. 2013). Thus, the hippocampal spine loss from inhalation of PM observed in mice exposed to diesel PM2.5 (Fonken et al. 2011) could involve the endogenous mouse Aβ, NO• and glutamate. The newer NMDAR antagonist, NitroMemantine, protected against Aβ-induced synapse loss, as well as induction of NO• and S-nitrosylation (Talantova et al. 2013). Future studies may consider therapeutic interventions with NMDAR antagonists on the synergies between nPM inhalation and Aβ as factors in cognitive impairments associated with aging, as well as onset and progression of AD.
Supplementary Material
ACKNOWLEDGEMENTS
Nahoko Iwata gave excellent assistance in Western blots. We thank Michel Baudry, Enrique Cadenas, and Henry Forman for critical comments. This research was supported by grants to C.E. Finch from the NIH (AG-040683; AG-040753) and the USC Collaborative Research Fund, and to C. Sioutas (South Coast Air Quality Management District (SCAQMD, award #11527). D.A. Davis was supported by NIA training Grant (T32 AG000037).
Abbreviations Defined
- AMPA
α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid
- AP5
D,L-2-amino-5-phosphonopentanoic acid
- CA1
cornu ammonis area 1
- DG
dentate gyrus
- EPSC
excitatory postsynaptic current
- EthD-1
ethidium homodimer-1
- GluA1
glutamate receptor AMPA subunit
- GluN2A/B
glutamate receptor NMDA subunit
- mGluR
metabotropic glutamate receptor
- NO•
nitric oxide
- NMDA
N-methyl-D-aspartic acid
- nPM
nanoscale particulate matter
Footnotes
AUTHORS CONTRIBUTIONS
David A. Davis, acute hippocampal slices, neuronal cultures, Western blots, nitric oxide and superoxide assays, viability assays; Garnik Akopian, electrophysiology; John P. Walsh, electrophysiology; Constantinos Sioutas, nPM collections and administration; Todd E. Morgan and Caleb E. Finch, experimental design and analysis.
REFERENCES
- Aizenman E, Hartnett KA, Reynolds IJ. Oxygen free radicals regulate NMDA receptor function via a redox modulatory site. Neuron. 1990;5:841–846. doi: 10.1016/0896-6273(90)90343-e. [DOI] [PubMed] [Google Scholar]
- Aizenman E, Lipton SA, Loring RH. Selective modulation of NMDA responses by reduction and oxidation. Neuron. 1989;2:1257–1263. doi: 10.1016/0896-6273(89)90310-3. [DOI] [PubMed] [Google Scholar]
- Akopian G, Walsh JP. Corticostriatal paired-pulse potentiation produced by voltage-dependent activation of NMDA receptors and L-type Ca(2+) channels. J Neurophysiol. 2002;87:157–165. doi: 10.1152/jn.00115.2001. [DOI] [PubMed] [Google Scholar]
- Alley GM, Bailey JA, Chen D, Ray B, Puli LK, Tanila H, Banerjee PK, Lahiri DK. Memantine lowers amyloid-beta peptide levels in neuronal cultures and in APP/PS1transgenic mice. J Neurosci Res. 2010;88:143–154. doi: 10.1002/jnr.22172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baez MV, Oberholzer MV, Cercato MC, Snitcofsky M, Aguirre AI, Jerusalinsky DA. NMDA receptor subunits in the adult rat hippocampus undergo similar changes after 5 minutes in an open field and after LTP induction. PLoS One. 2013;8:e55244. doi: 10.1371/journal.pone.0055244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banker GA, Cowan WM. Rat hippocampal neurons in dispersed cell culture. Brain Res. 1977;126:397–342. doi: 10.1016/0006-8993(77)90594-7. [DOI] [PubMed] [Google Scholar]
- Block ML, Calderon-Garciduenas L. Air pollution: mechanisms of neuroinflammation and CNS disease. Trends Neurosci. 2009;32:506–516. doi: 10.1016/j.tins.2009.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calderon-Garciduenas L, Kavanaugh M, Block M, et al. Neuroinflammation, hyperphosphorylated tau, diffuse amyloid plaques, and down-regulation of the cellular prion protein in air pollution exposed children and young adults. J Alzheimers Dis. 2012;28:93–107. doi: 10.3233/JAD-2011-110722. [DOI] [PubMed] [Google Scholar]
- Calderon-Garciduenas L, Solt AC, Henriquez-Roldan C, et al. Long-term air pollution exposure is associated with neuroinflammation, an altered innate immune response, disruption of the blood-brain barrier, ultrafine particulate deposition, and accumulation of amyloid beta-42 and alpha-synuclein in children and young adults. Toxicol Pathol. 2008;36:289–310. doi: 10.1177/0192623307313011. [DOI] [PubMed] [Google Scholar]
- Chen JC, Schwartz J. Neurobehavioral effects of ambient air pollution on cognitive performance in US adults. Neurotoxicology. 2009;30:231–239. doi: 10.1016/j.neuro.2008.12.011. [DOI] [PubMed] [Google Scholar]
- Cheng H, Morgan TE, Finch CE. Nanoscale urban air pollutants inhibit neurite outgrowth via TNF alpha; Soc Neurosci Ann Meeting 2012; 2012. Abstract 189.19. [Google Scholar]
- Daher N, Hasheminassab S, Shafer MM, Schauer JJ, Sioutas C. Seasonal and spatial variability in chemical composition and mass closure of ambient ultrafine particles in the megacity of Los Angeles. Environ. Sci.: Processes & Impacts. 2013;15:283–295. doi: 10.1039/c2em30615h. [DOI] [PubMed] [Google Scholar]
- Davis DA, Akopian G, Sioutas C, Walsh JP, Morgan TE, Finch CE. Nano-particulates from urban vehicular exhaust decrease glutamate receptor function; Soc Neurosci Ann Meet 2012; 2012. Abstract 189.13. [Google Scholar]
- Fonken LK, Xu X, Weil ZM, Chen G, Sun Q, Rajagopalan S, Nelson RJ. Air pollution impairs cognition, provokes depressive-like behaviors and alters hippocampal cytokine expression and morphology. Mol Psychiatry. 2011;16:987–995. doi: 10.1038/mp.2011.76. 973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fioravante D, Regehr WG. Short-term forms of presynaptic plasticity. Curr Opin Neurobiol. 2011;21:269–274. doi: 10.1016/j.conb.2011.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frade JG, Barbosa RM, Laranjinha J. Stimulation of NMDA and AMPA glutamate receptors elicits distinct concentration dynamics of nitric oxide in rat hippocampal slices. Hippocampus. 2009;19:603–611. doi: 10.1002/hipo.20536. [DOI] [PubMed] [Google Scholar]
- Geiser M, Rothen-Rutishauser B, Kapp N, et al. Ultrafine particles cross cellular membranes by nonphagocytic mechanisms in lungs and in cultured cells. Environ Health Perspect. 2005;113:1555–1560. doi: 10.1289/ehp.8006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gelsomino G, Menna E, Antonucci F, et al. Kainate induces mobilization of synaptic vesicles at the growth cone through the activation of protein kinase A. Cereb Cortex. 2013;23:531–541. doi: 10.1093/cercor/bhs026. [DOI] [PubMed] [Google Scholar]
- Gillespie P, Tajuba J, Lippmann M, Chen LC, Veronesi B. Particulate matter neurotoxicity in culture is size-dependent. Neurotoxicology. 2013;36:112–117. doi: 10.1016/j.neuro.2011.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hara MR, Agrawal N, Kim SF, et al. S-nitrosylated GAPDH initiates apoptotic cell death by nuclear translocation following Siah1 binding. Nat Cell Biol. 2005;7:665–674. doi: 10.1038/ncb1268. [DOI] [PubMed] [Google Scholar]
- Ignarro LJ, Fukuto JM, Griscavage JM, Rogers NE, Byrns RE. Oxidation of nitric oxide in aqueous solution to nitrite but not nitrate: comparison with enzymatically formed nitric oxide from L-arginine. Proc Natl Acad Sci U S A. 1993;90:8103–8107. doi: 10.1073/pnas.90.17.8103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawasaki K, Traynelis SF, Dingledine R. Different responses of CA1 and CA3 regions to hypoxia in rat hippocampal slice. J Neurophysiol. 1990;63:385–394. doi: 10.1152/jn.1990.63.3.385. [DOI] [PubMed] [Google Scholar]
- Kim SH, Fraser PE, Westaway D, St. George-Hyslop PH, Ehrlichm ME, Gandy S. Group II metabotropic glutamate receptor stimulation triggers production and release of Alzheimer's amyloid(beta)42 from isolated intact nerve terminals. J Neurosci. 2010;30:3870–3875. doi: 10.1523/JNEUROSCI.4717-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleinman MT, Araujo JA, Nel A, Sioutas C, Campbell A, Cong PQ, Li H, Bondy SC. Inhaled ultrafine particulate matter affects CNS inflammatory processes and may act via MAP kinase signaling pathways. Toxicol Lett. 2008;178:127–130. doi: 10.1016/j.toxlet.2008.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee HK, Takamiya K, He K, Song L, Huganir RL. Specific roles of AMPA receptor subunit GluR1 (GluA1) phosphorylation sites in regulating synaptic plasticity in the CA1 region of hippocampus. J Neurophysiol. 2010;103:479–489. doi: 10.1152/jn.00835.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levesque S, Taetzsch T, Lull ME, et al. Diesel exhaust activates and primes microglia: air pollution, neuroinflammation, and regulation of dopaminergic neurotoxicity. Environ Health Perspect. 2011a;119:1149–1155. doi: 10.1289/ehp.1002986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levesque S, Surace MJ, McDonald J, Block ML. Air pollution & the brain: Subchronic diesel exhaust exposure causes neuroinflammation and elevates early markers of neurodegenerative disease. J Neuroinflammation. 2011b;8:105–115. doi: 10.1186/1742-2094-8-105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levesque S, Taetzsch T, Lull ME, Johnson JA, McGraw C, Block ML. The role of MAC1 in diesel exhaust particle-induced microglial activation and loss of dopaminergic neuron function. J Neurochem. 2013;125:756–765. doi: 10.1111/jnc.12231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li N, Sioutas C, Cho A, et al. Ultrafine particulate pollutants induce oxidative stress and mitochondrial damage. Environ Health Perspect. 2003;111:455–460. doi: 10.1289/ehp.6000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li N, Wang M, Oberley TD, Sempf JM, Nel AE. Comparison of the pro-oxidative and proinflammatory effects of organic diesel exhaust particle chemicals in bronchial epithelial cells and macrophages. J Immunol. 2002;169:4531–4541. doi: 10.4049/jimmunol.169.8.4531. [DOI] [PubMed] [Google Scholar]
- Li R, Navab M, Pakbin P, Ning Z, Navab K, Hough G, Morgan TE, Finch CE, Araujo JA, Fogelman AM, Sioutas C, Hsiai T. Ambient ultrafine particles alter lipid metabolism and HDL anti-oxidant capacity in LDLR-null mice. J Lipid Res. 2013;54(6):1608–1615. doi: 10.1194/jlr.M035014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma T, Hoeffer CA, Wong H, Massaad CA, Zhou P, Iadecola C, Murphy MP, Pautler RG, Klann E. Amyloid beta-induced impairments in hippocampal synaptic plasticity are rescued by decreasing mitochondrial superoxide. J Neurosci. 2011;31:5589–5595. doi: 10.1523/JNEUROSCI.6566-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manzoni O, Prezeau L, Marin P, Deshager S, Bockaert J, Fagni L. Nitric oxide-induced blockade of NMDA receptors. Neuron. 1992;8:653–662. doi: 10.1016/0896-6273(92)90087-t. [DOI] [PubMed] [Google Scholar]
- Misra C, Kim S, Shen S, Sioutas C. A high flow rate, very low pressure drop impactor for inertial separation of ultrafine from accumulation mode particles. J Aerosol Sci. 2002;33:735–752. [Google Scholar]
- Morgan TE, Davis DA, Iwata N, et al. Glutamatergic neurons in rodent models respond to nanoscale particulate urban air pollutants in vivo and in vitro. Environ Health Perspect. 2011;119:1003–1009. doi: 10.1289/ehp.1002973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrison JH, Hof PR. Life and death of neurons in the aging brain. Science. 1997;278:412–419. doi: 10.1126/science.278.5337.412. [DOI] [PubMed] [Google Scholar]
- Ning Z, Geller MD, Moore KF, Sheesley R, Schauer JJ, Sioutas C. Daily variation in chemical characteristics of urban ultrafine aerosols and inference of their sources. Environ Sci Technol. 2007;41:6000–6006. doi: 10.1021/es070653g. [DOI] [PubMed] [Google Scholar]
- Oberdörster G, Sharp Z, Atudorei V, Elder A, Gelein R, Kreyling W, Cox C. Translocation of inhaled ultrafine particles to the brain. Inhal Toxicol. 2004;16:437–445. doi: 10.1080/08958370490439597. [DOI] [PubMed] [Google Scholar]
- Power MC, Weisskopf MG, Alexeeff SE, Coull BA, Spiro A, 3rd, Schwartz J. Traffic-related air pollution and cognitive function in a cohort of older men. Environ Health Perspect. 2011;119:682–687. doi: 10.1289/ehp.1002767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Power MC, Weisskopf MG, Alexeeff SE, Wright RO, Coull BA, Spiro A, 3rd, Schwartz J. Modification by hemochromatosis gene polymorphisms of the association between traffic-related air pollution and cognition in older men: a cohort study. Environ Health. 2013;12:16. doi: 10.1186/1476-069X-12-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reyes RC, Brennan AM, Shen Y, Baldwin Y, Swanson RA. Activation of neuronal NMDA receptors induces superoxide-mediated oxidative stress in neighboring neurons and astrocytes. J Neurosci. 2012;32:12973–12978. doi: 10.1523/JNEUROSCI.1597-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryan TJ, Kopanitsa MV, Indersmitten T, Nithianantharajah J, Afinowi NO, Pettit C, Stanford LE, Sprengel R, Saksida LM, Bussey TJ, O'Dell TJ, Grant SG, Komiyama NH. Evolution of GluN2A/B cytoplasmic domains diversified vertebrate synaptic plasticity and behavior. Nat Neurosci. 2013;16:25–32. doi: 10.1038/nn.3277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santello M, Volterra A. TNFalpha in synaptic function: switching gears. Trends Neurosci. 2012;35:638–647. doi: 10.1016/j.tins.2012.06.001. [DOI] [PubMed] [Google Scholar]
- Santos SD, Carvalho AL, Caldeira MV, Duarte CB. Regulation of AMPA receptors and synaptic plasticity. Neuroscience. 2009;158:105–125. doi: 10.1016/j.neuroscience.2008.02.037. [DOI] [PubMed] [Google Scholar]
- Sardar SB, Philip M, Mayo PR, Sioutas C. Size-fractionated measurements of ambient ultrafine particle chemical composition in Los Angeles using the NanoMOUDI. Envir Sci & Technol. 2005;39:932–944. doi: 10.1021/es049478j. [DOI] [PubMed] [Google Scholar]
- Sattler R, Xiong Z, Lu WY, Hafner M, MacDonald JF, Tymianski M. Specific coupling of NMDA receptor activation to nitric oxide neurotoxicity by PSD-95 protein. Science. 1999;284:1845–1848. doi: 10.1126/science.284.5421.1845. [DOI] [PubMed] [Google Scholar]
- Selvakumar B, Jenkins MA, Hussain NK, Huganir RL, Traynelis SF, Snyder SH. S-nitrosylation of AMPA receptor GluA1 regulates phosphorylation, single-channel conductance, and endocytosis. Proc Natl Acad Sci U S A. 2013;110:1077–1082. doi: 10.1073/pnas.1221295110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi ZQ, Sunico CR, McKercher SR, Cui J, Feng GS, Nakamura T, Lipton SA. S-nitrosylated SHP-2 contributes to NMDA receptor-mediated excitotoxicity in acute ischemic stroke. Proc Natl Acad Sci U S A. 2013;110:3137–3142. doi: 10.1073/pnas.1215501110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soeda H, Tatsumi H, Katayama Y. Neurotransmitter release from growth cones of rat dorsal root ganglion neurons in culture. Neuroscience. 1997;77:1187–1199. doi: 10.1016/s0306-4522(96)00465-4. [DOI] [PubMed] [Google Scholar]
- Stein TD, Anders NJ, DeCarli C, Chan SL, Mattson MP, Johnson JA. Neutralization of transthyretin reverses the neuroprotective effects of secreted amyloid precursor protein (APP) in APPSW mice resulting in tau phosphorylation and loss of hippocampal neurons: support for the amyloid hypothesis. J Neurosci. 2004;24:7707–7717. doi: 10.1523/JNEUROSCI.2211-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Talantova M, Sanz-Blasco S, Zhang X, Xia P, Akhtar MW, Okamoto SI, Dziewczapolski G, Nakamura T, Cao G, Pratt AE, Kang YJ, Tu S, Molokanova E, McKercher SR, Hires SA, Sason H, Stouffer DG, Buczynski MW, Solomon JP, Michael S, Powers ET, Kelly JW, Roberts A, Tong G, Fang-Newmeyer T, Parker J, Holland EA, Zhang D, Nakanishi N, Chen HS, Wolosker H, Wang Y, Parsons LH, Ambasudhan R, Masliah E, Heinemann SF, Piña-Crespo JC, Lipton SA. Aβ induces astrocytic glutamate release, extrasynaptic NMDA receptor activation, and synaptic loss. Proc Natl Acad Sci USA. 2013 doi: 10.1073/pnas.1306832110. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Traynelis SF, Wollmuth LP, McBain CJ, et al. Glutamate receptor ion channels: structure, regulation, and function. Pharmacol Rev. 2010;62:405–496. doi: 10.1124/pr.109.002451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weuve J, Puett RC, Schwartz J, Yanosky JD, Laden F, Grodstein F. Exposure to particulate air pollution and cognitive decline in older women. Arch Intern Med. 2012;172:219–227. doi: 10.1001/archinternmed.2011.683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Win-Shwe TT, Fujimaki H. Nanoparticles and neurotoxicity. Int J Mol Sci. 2011;12:6267–6280. doi: 10.3390/ijms12096267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Win-Shwe TT, Mitsushima D, Yamamoto S, Fujitani Y, Funabashi T, Hirano S, Fujimaki H. Extracellular glutamate level and NMDA receptor subunit expression in mouse olfactory bulb following nanoparticle-rich diesel exhaust exposure. Inhal Toxicol. 2009;21:828–836. doi: 10.1080/08958370802538068. [DOI] [PubMed] [Google Scholar]
- Win-Shwe TT, Yamamoto S, Fujitani Y, Hirano S, Fujimaki H. Nanoparticle-rich diesel exhaust affects hippocampal-dependent spatial learning and NMDA receptor subunit expression in female mice. Nanotoxicology. 2012;6:543–553. doi: 10.3109/17435390.2011.590904. [DOI] [PubMed] [Google Scholar]
- Xia T, Kovochich M, Brant J, et al. Comparison of the abilities of ambient and manufactured nanoparticles to induce cellular toxicity according to an oxidative stress paradigm. Nano Lett. 2006;6:1794–1807. doi: 10.1021/nl061025k. [DOI] [PubMed] [Google Scholar]
- Xie Z, Wei M, Morgan TE, Fabrizio P, Han D, Finch CE, Longo VD. Peroxynitrite mediates neurotoxicity of amyloid beta-peptide1-42- and lipopolysaccharide-activated microglia. J Neurosci. 2002;22:3484–3492. doi: 10.1523/JNEUROSCI.22-09-03484.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang H, Liu H, Davies KJ, Sioutas C, Finch CE, Morgan TE, Forman HJ. Nrf2-regulated phase II enzymes are induced by chronic ambient nanoparticle exposure in young mice with age-related impairments. Free Rad Biol Med. 2012;52:2038–2046. doi: 10.1016/j.freeradbiomed.2012.02.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Y, Usatyuk PV, Gorshkova IA, He D, Wang T, Moreno-Vinasco L, Geyh AS, Breysse PN, Samet JM, Spannhake EW, Garcia JG, Natarajan V. Regulation of COX-2 expression and IL-6 release by particulate matter in airway epithelial cells. Am J Respir Cell Mol Biol. 2009;40:19–30. doi: 10.1165/rcmb.2008-0105OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zielonka J, Kalyanaraman B. Hydroethidine- and Mito-Sox-derived red fluorescence is not a reliable indicator of intracellular superoxide formation: another inconvenient truth. Free Radicl Biol Med. 2010;48:983–1001. doi: 10.1016/j.freeradbiomed.2010.01.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zanchi AC, Fagundes LS, Barbosa F, Jr, Bernardi R, Rhoden CR, Saldiva PH, do Valle AC. Pre and post-natal exposure to ambient level of air pollution impairs memory of rats: the role of oxidative stress. Inhal Toxicol. 2010;22:910–918. doi: 10.3109/08958378.2010.494313. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.