Sustaining the vast peripheral B cell repertoire is critical for the immunological health of the host. B-cell activating factor of the TNF family (BAFF) is essential for the survival of mature B cells. A recent publication in Immunity by Robert Brink and coworkers has afforded important new insights into how interactions between BAFF and one of its receptors—BAFF receptor (BAFF-R)—function in regulating the development of the mature B cell repertoire.1
The TNF family members BAFF and APRIL (a proliferation-inducing ligand), along with their shared receptors BCMA and TACI were discovered at the turn of last decade. Scientists meandered their way through this morass of factors and shared receptors and came to the realization that signaling through the BAFF-specific receptor BAFF-R was essential for naive B cell development and survival.2 It is now evident that BAFF/BAFF-R interactions regulate the humoral immune system at multiple checkpoints. The first checkpoint occurs during B cell development, where engagement of BAFF-R on a developing B cell by BAFF is required for the continued development and survival of this cell. The second checkpoint occurs by selectively controlling the survival of naive B cells in the periphery. Basal levels of circulating BAFF prevent the persistence of self-reactive B cells due to an enhanced requirement of BAFF-R signaling for this cell type’s survival and activation. However, increased BAFF-R signaling through the introduction of excessive BAFF allows the survival of self-reactive B cells, driving the development of lupus-like symptoms due to a breakdown of peripheral tolerance. Owing to the essential role of BAFF in sustaining peripheral B cell development and survival, blocking interactions between BAFF and BAFF-R leads to the rapid depletion of mature B cells from the circulation and secondary lymphoid organs. This antagonism is the current focus of therapeutic intervention in multiple B cell-mediated pathologies.3–5
Studies examining signaling pathways downstream of BAFF-R have found that the activation of the alternative nuclear factor kappa B (NF-κB2) pathway is essential for the pro-survival effects mediated by this ligand/receptor pair.6,7 Elimination of the NF-κB2 pathway in vivo results in a B-cell deficient phenotype resembling that of both BAFF and BAFF-R knockout mice.8,9 The exact mechanism of how BAFF-R engagement drives a pro-survival signal remains unclear, and there is a gap in our knowledge of the membrane proximal events occurring after BAFF binds to BAFF-R but prior to the activation of the NF-κB2 pathway. In an elegant study, Robert Brink and coworkers have presented data that unifies existing observations to provide some provocative answers to the mysteries surrounding BAFF-R signaling.1
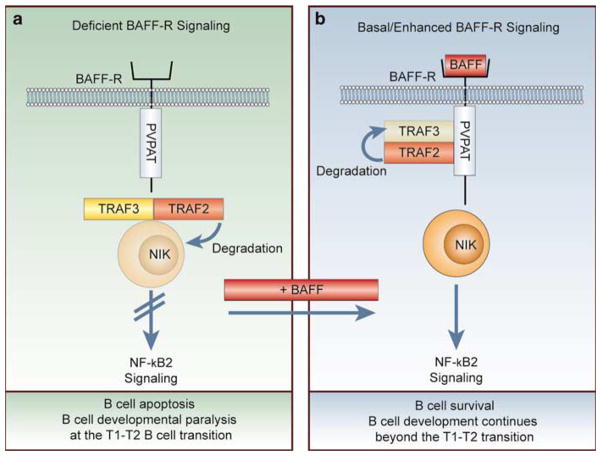
In this report, Gardam et al present compelling evidence of how BAFF imparts survival signals through BAFF-R.1 All the members of the TNF-R family signal through adapter molecules called TNFR-associated factors. On the basis of the observation that the cytoplasmic domain of BAFF-R contains a TRAF3-binding domain and that both TRAF2- and TRAF3-deficient B cells exhibit enhanced in vivo survival and hyperactive NF-κB2 signaling, the authors sought to definitively determine how TRAF2 and TRAF3 negatively regulate BAFF-R signaling in vivo and whether these adapter proteins act redundantly or cooperatively in this role.6,7,10,11 Mice were generated in which TRAF2, TRAF3, or both of these adapter proteins were conditionally deleted only in B cells.1 B cells that lacked either or both TRAF2 and TRAF3 exhibited enhanced survival due to a hyperactive NF-κB2 pathway.1 Remarkably, B cell development and survival in the conditional TRAF2 knockout mouse could now occur independent of BAFF survival signals, as crossing the conditional TRAF2 B cell knockout mice with BAFF−/− mice completely rescued all B cell survival defects that result from BAFF deficiency.1 This result complements a recent observation that TRAF3 knockout B cells also survive independently of BAFF survival signals.11 Interestingly, TRAF2 and TRAF3 single and TRAF2/TRAF3 double knockout B cells exhibited the identical in vivo survival phenotypes and mRNA expression patterns, suggestive of cooperative and non-redundant functions between TRAF2 and TRAF3 in repressing NF-κB2 activation.1 The authors lastly observed that TRAF3 protein is lost in a TRAF2-dependent manner during BAFF-R signaling.1 On the basis of these data, Brink and coworkers propose a model where TRAF2 and TRAF3 cooperate to constitutively suppress the NF-κB2 pathway by driving continual degradation of NIK (NFκB-inducing kinase) in the absence of BAFF-R signaling (Figure 1a). Following binding of BAFF to BAFF-R, TRAF3 is recruited to BAFF-R and is depleted through the activities of TRAF2 (Figure 1b). As TRAF2 and TRAF3 have both been reported to bind NIK, the authors predict that degradation of NIK in the absence of BAFF-R signaling and the degradation of TRAF3 through TRAF2 during BAFF-R signaling occurs through a NIK/TRAF2/TRAF3 complex.12,13 Upon TRAF3 degradation, signaling through the alternative NF-κB2 pathway can now occur, and mature B cells are afforded essential survival and developmental signals (Figure 1b).
Figure 1.
TRAF2 and TRAF3 cooperatively and constitutively repress the alternative NF-κB pathway, with BAFF-R signaling relieving this suppression. In all reports describing deficient BAFF-R signaling, blunted naive mature B cell survival and development is the result (a). When BAFF-R signaling is deficient, TRAF3 and TRAF2 cooperate to constitutively degrade NIK and thus block activation of the alternative NK-κB pathway (a). It is thought that both TRAF2 and TRAF3 bind to NIK and cooperate in its degradation, with the heterocomplex of TRAF2/TRAF3/NIK depicted in (a) purely hypothetical. Upon ligation of BAFF-R by BAFF (b), TRAF3 is recruited to an atypical TRAF3-binding site located in the cytoplasmic tail of BAFF-R (depicted by the amino acid sequence PVPAT).6 TRAF3 is then targeted for degradation in a TRAF2-dependent manner. Owing to depleted TRAF3 levels, NIK is no longer the subject of continual degradation and can activate the alternative NF-κB pathway. Through NF-κB2 signaling, B cell survival and the transitional B cell developmental checkpoints occur.
This report provides convincing evidence that both TRAF2 and TRAF3 are required for the repression of the alternative NF-κB pathway, with these adapter proteins together provoking a requirement for BAFF in mediating peripheral B cell survival. Indeed, the default state of the B cell appears to be survival if it were not for the activities of TRAF2 and TRAF3, as indicated by their selective deletion from B cells and the resulting enhanced survival capacity of these cells. Their activities in suppressing B cell survival and development and in enforcing the BAFF/BAFF-R checkpoint may have arisen as a preventative mechanism to suppress the breakdown of peripheral B-cell tolerance and prevent B cell hyperplasia. It is interesting to note that TRAF2 and TRAF3 constitutively suppress B cell survival but have no impact on T cell survival as deletion of TRAF2 and TRAF3 from the T cell lineage does not perturb the size or survival of the peripheral T cell population, despite these cells exhibiting a hyperactive NF-κB2 pathway.1 This indicates divergent survival mechanisms utilized by naive T and B cells, and raises the question as to why these two lymphocyte populations rely on differing pathways through which to receive pro-survival signals. Indeed, this compartmentalization may be a fail-safe mechanism towards averting or dampening an autoimmune response in the presence of enhanced pro-survival signals in vivo, such as increases in serum BAFF levels seen in patients with autoimmune manifestations. If both B and T lymphocytes relied on BAFF-R signaling to survive, excess BAFF would enhance survival of both populations rather than only B cells. This would create a possible situation of enhanced autoimmunity by rendering both populations refractory to peripheral tolerance mechanisms. Thus, segregating the survival factors required by each lymphocyte population as well as the adapter proteins through which these receptors signal through would be advantageous for avoiding the development of autoimmune conditions.
Important questions remain as to exactly how TRAF2 antagonizes NF-κB2 signaling in the absence of BAFF-R signaling while playing a positive role in NF-κB2 activation during BAFF-R signaling by mediating TRAF3 degradation. How TRAF2 mediates degradation of TRAF3 in the absence of a TRAF2 binding site in the cytoplasmic domain of BAFF-R is unclear. Further biochemical analyses of how TRAF2, TRAF3, NIK and the cytoplasmic region of BAFF-R interact with each other in the presence and absence of BAFF will be necessary to fully establish how this cytokine provides pro-survival signals to B cells through the NF-κB2 pathway. Further studies such as those presented by Robert Brink and coworkers promise to contribute essential answers to the pivotal question as to how B cells survive.
References
- 1.Gardam S, Sierro F, Basten A, Mackay F, Brink R. TRAF2 and TRAF3 signal adapters act cooperatively to control the maturation and survival signals delivered to B cells by the BAFF receptor. Immunity. 2008;28:391–401. doi: 10.1016/j.immuni.2008.01.009. [DOI] [PubMed] [Google Scholar]
- 2.Mackay F, Schneider P, Rennert P, Browning J. BAFF AND APRIL: a tutorial on B cell survival. Annu Rev Immunol. 2003;21:231–264. doi: 10.1146/annurev.immunol.21.120601.141152. [DOI] [PubMed] [Google Scholar]
- 3.Schiemann B, Gommerman JL, Vora K, Cachero TG, Shulga-Morskaya S, Dobles M, et al. An essential role for BAFF in the normal development of B cells through a BCMA-independent pathway. Science. 2001;293:2111–2114. doi: 10.1126/science.1061964. [DOI] [PubMed] [Google Scholar]
- 4.Gross JA, Dillon SR, Mudri S, Johnston J, Littau A, Roque R, et al. TACI-Ig neutralizes molecules critical for B cell development and autoimmune disease. Impaired B cell maturation in mice lacking BLyS. Immunity. 2001;15:289–302. doi: 10.1016/s1074-7613(01)00183-2. [DOI] [PubMed] [Google Scholar]
- 5.Thompson JS, Bixler SA, Qian F, Vora K, Scott ML, Cachero TG, et al. BAFF-R, a newly identified TNF receptor that specifically interacts with BAFF. Science. 2001;293:2108–2111. doi: 10.1126/science.1061965. [DOI] [PubMed] [Google Scholar]
- 6.Morrison MD, Reiley W, Zhang M, Sun SC. An atypical tumor necrosis factor (TNF) receptor-associated factor-binding motif of B cell-activating factor belonging to the TNF family (BAFF) receptor mediates induction of the noncanonical NF-kappaB signaling pathway. J Biol Chem. 2005;280:10018–10024. doi: 10.1074/jbc.M413634200. [DOI] [PubMed] [Google Scholar]
- 7.Xu LG, Shu HB. TNFR-associated factor-3 is associated with BAFF-R and negatively regulates BAFF-R-mediated NF-kappa B activation and IL-10 production. J Immunol. 2002;169:6883–6889. doi: 10.4049/jimmunol.169.12.6883. [DOI] [PubMed] [Google Scholar]
- 8.Franzoso G, Carlson L, Poljak L, Shores EW, Epstein S, Leonardi A, et al. Mice deficient in nuclear factor (NF)-kappa B/p52 present with defects in humoral responses, germinal center reactions, and splenic microarchitecture. J Exp Med. 1998;187:147–159. doi: 10.1084/jem.187.2.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Caamano JH, Rizzo CA, Durham SK, Barton DS, Raventos-Suarez C, et al. Nuclear factor (NF)-kappa B2 (p100/p52) is required for normal splenic microarchitecture and B cell-mediated immune responses. J Exp Med. 1998;187:185–196. doi: 10.1084/jem.187.2.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Grech AP, Amesbury M, Chan T, Gardam S, Basten A, Brink R. TRAF2 differentially regulates the canonical and noncanonical pathways of NF-kappaB activation in mature B cells. Immunity. 2004;21:629–642. doi: 10.1016/j.immuni.2004.09.011. [DOI] [PubMed] [Google Scholar]
- 11.Xie P, Stunz LL, Larison KD, Yang B, Bishop GA. Tumor necrosis factor receptor-associated factor 3 is a critical regulator of B cell homeostasis in secondary lymphoid organs. Immunity. 2007;27:253–267. doi: 10.1016/j.immuni.2007.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Liao G, Zhang M, Harhaj EW, Sun SC. Regulation of the NF-kappaB-inducing kinase by tumor necrosis factor receptor-associated factor 3-induced degradation. J Biol Chem. 2004;279:26243–26250. doi: 10.1074/jbc.M403286200. [DOI] [PubMed] [Google Scholar]
- 13.Malinin NL, Boldin MP, Kovalenko AV, Wallach D. MAP3K-related kinase involved in NF-kappaB induction by TNF, CD95 and IL-1. Nature. 1997;385:540–544. doi: 10.1038/385540a0. [DOI] [PubMed] [Google Scholar]