Abstract
A 73-year-old man presented with a 5-month history of intermittent nausea, vomiting, central abdominal discomfort and a 17-pound weight loss over the past year. Laboratory testing, including a complete blood count with differential, liver function testing, amylase and lipase studies were normal. A CT scan showed a bilobed cystic lesion inferior to the body of the pancreas. An endoscopic ultrasound revealed a 5.3×3.9 cm, anechoic, bilobed cystic lesion, extrinsic to the body of the pancreas with a 1–2 mm septation and a normal pancreas. Fine-needle aspiration revealed a milky-white aspirate with negative cytology. Laboratory assessment of the cystic aspirant revealed carcinoembryonic antigen 1.7 ng/mL, amylase 148 units/L, cholesterol 300 mg/dL, and carbohydrate antigen 19-9 3 units/mL. He underwent resection of the mass, with the histopathology confirming a diagnosis of peripancreatic lymphangioma. He did well after the surgery with interval resolution of his symptoms.
Background
Lymphangiomas are rare benign tumours composed of malformed lymphatic vessels that may involve almost any organ,1 but are most commonly found in the pancreas or in the peripancreatic area.2 3 Pancreatic lymphangiomas are uncommon mesenchymal tumours that account for less than 1% of all lymphangiomas.4 5 Lymphangiomas are classified as pancreatic lymphangiomas if they originate from the pancreatic parenchyma, lie in proximity to the pancreas (in our case, at laparotomy the mass was adjacent to the inferior border of the body of pancreas), or are attached to the pancreas by a pedicle.6 They have characteristic endothelial immunoreactivity for endothelial markers CD31, CD34, D2-40, and von Willebrand factor antigen (Factor VIII:R antigen).2 3 7 8 The first reported case of pancreatic lymphangioma was published by Koch in 1913,9 and a recent review reveals only 70 cases of pancreatic lymphangioma in the literature.10 Because pancreatic and peripancreatic lymphangiomas are very rare and cystic in nature, preoperative diagnosis is difficult. These tumours can be easily confused with non-neoplastic cystic lesions of the pancreas, calcified echinococcus cysts, and mucinous cystic neoplasms of the pancreas.11 Although pancreatic lymphangiomas are more common in females2 and children,12 we reported an interesting case of a 73-year-old man diagnosed with peripancreatic lymphangioma. We also described the role of endoscopic ultrasound with a fine-needle aspiration in early diagnosis. Ours is only the ninth case reported in the literature using endoscopic ultrasound-guided fine-needle aspiration in confirming the diagnosis.
Case presentation
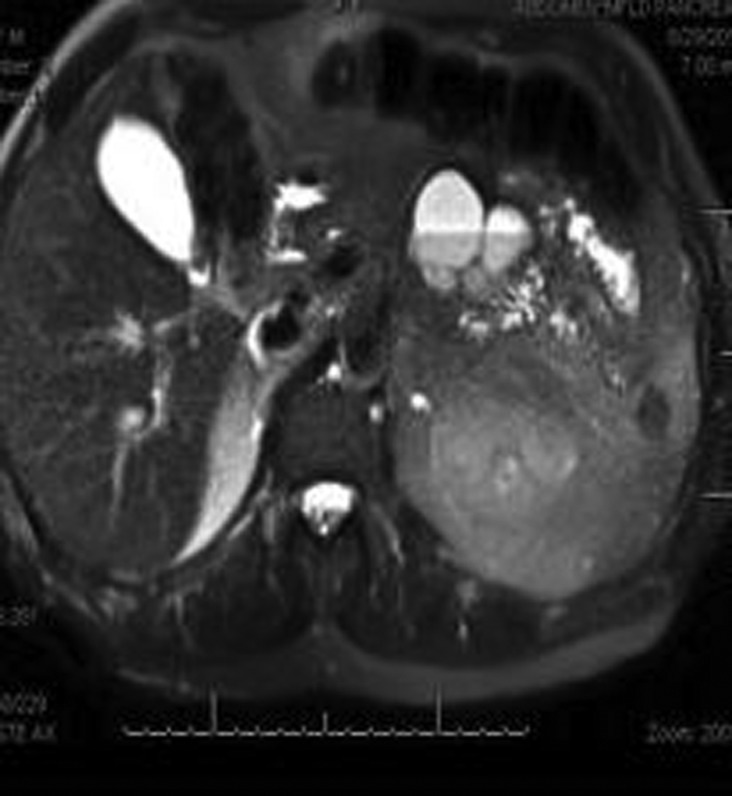
A 73-year-old man presented with a 5-month history of intermittent nausea, vomiting, central abdominal discomfort and a 17-pound weight loss over the past year. Laboratory studies including a complete blood count with differential, liver function testing, amylase and lipase studies were normal. A plain abdominal radiography showed no abnormalities. An abdominal ultrasound revealed two cystic lesions in the region of the tail of the pancreas measuring 3.9×2.9 cm and 1.0×1.0 cm, with a normal gallbladder and a common bile duct. A CT scan showed a 6.6×4.8 cm sharply defined, bilobed cystic lesion in the mid-abdomen, inferior to the body of the pancreas (figure 1). An extrinsic lobulated cystic lesion adjacent to the body of the pancreas was found on MRI (figure 2).
Figure 1.
CT scan of bilobed cystic lesion adjacent to the pancreas.
Figure 2.
MRI revealed an extrinsic bilobed cystic lesion with a normal pancreas.
An endoscopic ultrasound revealed a 5.3×3.9 cm, bilobed, extrinsic cystic lesion in the region of the body of the pancreas with a 1–2 mm thin septation, without hypoechoic component or mural nodules, and a normal pancreas (figure 3). Fine-needle aspiration with a 19-gauge needle was performed with aspiration of 35 mL of milky-white aspirate (figure 4) with negative cytology and mucin staining. Laboratory assessment of the cystic aspirant revealed carcinoembryonic antigen 1.7 ng/mL, amylase 148 U/L, cholesterol 300 mg/dL, and carbohydrate antigen 19-9 3 U/mL. The patient underwent a laparotomy that revealed a thickened mesentery with lymphatic congestion and a cystic mass inferior to the border of the pancreas. The mass was resected en bloc, and on gross examination, revealed a 7.5×7.0 cm soft, tan, multicystic tissue containing haemorrhagic fluid and a wrinkled inner surface with no papillary excrescences. The histological findings were diagnostic as lymphangioma and included numerous lymphoid follicles in the cyst wall (figure 5) and vascular spaces lined by a single layer of cytologically bland endothelial cells (figure 6A) that were immunoreactive for podoplanin (D2-40; figure 6B). The patient recovered well postoperatively with the resolution of symptoms at 6 months of follow-up.
Figure 3.

Endoscopic ultrasound revealed a multicystic lesion in the peripancreatic lesion.
Figure 4.

Fine needle aspiration of the cyst revealed a milky aspirate.
Figure 5.

In apposition to the thin-walled cavernous vascular structures are lymphoid follicles, characteristic of lymphangioma (H&E, low magnification).
Figure 6.

(A) Vascular spaces are lined by a single layer of cytologically bland endothelial cells (H&E, high magnification.) (B) Immunostain for podoplanin (D2-40), immunoreactive for D2-40, a lymphatic endothelial marker (high magnification).
Discussion
Lymphangiomas are benign lesions most commonly found on the skin, although they have been reported to develop anywhere in the body except in the brain and ophthalmic tissues.13 14 Retroperitoneal lymphangiomas rarely project into the abdominal cavity.15–18 While the aetiology of the lymphangiomas is not completely clear, the most popular theory suggests that the lymphangiomas may be the result of congenital malformations of the lymphatic channels19 or of lymphatic obstruction secondary to inflammation.20
Pancreatic lymphangiomas are more common in females and may present at any age.2 Literature review for primary pancreatic lymphangiomas revealed a female predilection for all ages and an equal distribution in all regions of the pancreas.2 The female preponderance can be partially explained by the positive effects of the oral contraceptives, hyperprogesteronemia and pregnancy in promoting the growth of lymphangioma.21 A review by Igarashi et al11 revealed a female-to-male ratio of 29:16 and an average age of presentation of 40 years (range 2–81 years), demonstrating that the lymphangiomas more commonly occur in the younger age group.12 Our patient was a 73-year-old man, in contrast with most reported cases.
The clinical presentation of pancreatic lymphangioma is variable. A number of cases reported asymptomatic lymphangiomas that were diagnosed incidentally during a workup for unrelated diseases.2 22–25 The clinical presentation is typically non-specific and not helpful in the preoperative diagnosis. Lymphangiomas may cause compression of neighbouring organs, resulting in symptoms.22 26
Pancreatic lymphangiomas are cystic in nature and have a broad differential diagnosis including pancreatic pseudocysts, mucinous cystic tumours, solid and papillary cystic epithelial neoplasms, microcystic adenomas, cystic adenocarcinomas, mesenchymal tumours (eg, hemangioma) and duct obstruction.27 Our patient presented with a 5-month history of intermittent nausea, vomiting, abdominal discomfort and a 17-pound weight loss in 1 year. Because cystic lesions of the pancreas and peripancreatic lesions can be easily confused with lymphangiomas of the pancreas, our case could have been easily confused with the mucinous cystic tumours of the pancreas because of the associated weight loss.11
Imaging studies including an ultrasound, CT (shown in figure 1) and MRI (shown in figure 2) are helpful in localising and defining the number of cysts as well as the contents of the cysts.28 29 Lymphangiography that shows a connection of a cystic lesion to the lymphatics, or the presence of fat-like structures in the cystic lesion, may suggest lymphangioma.30 The absence of serological markers for lymphangioma and the non-diagnostic nature of imaging (ultrasound, CT and MRI) make preoperative diagnosis challenging.20
Endoscopic ultrasound with fine-needle aspiration has been reported to have an accuracy of 86% and specificity of 100%.31 32 On imaging studies, pancreatic/peripancreatic cystic lymphangioma can mimic more serious pathological lesions including mucinous cystic neoplasm of the pancreas. An endoscopic ultrasound with fine-needle aspiration has the advantage of aspirating the cystic fluid for analysis, which increases the diagnostic accuracy.33 The chylous appearance of the aspirated fluid with elevated triglycerides confirms the diagnosis of lymphangioma.34
The unique feature of our case includes the use of an endoscopic ultrasound with fine-needle aspiration yielding a milky aspirate from the cyst with elevated cholesterol level of 300 mg/dL to confirm the diagnosis of peripancreatic lymphangioma (figures 3 and 4), which was postoperatively confirmed by histopathology. Ours is only the ninth case reported in the literature using endoscopic ultrasound-guided fine-needle aspiration to confirm the diagnosis.13 35
Our patient was treated with an exploratory laparotomy with complete removal of the cyst with excellent recovery. Reports in the literature support this as the best approach for the treatment of symptomatic lymphangiomas with a low likelihood of recurrence (approximately 7%).4 5 15 25 36 37 Partial resection of pancreatic lymphangioma is associated with a recurrence rate approaching 50% with a mean follow-up period of 2 years.38 Laparoscopic excision should be considered in uncomplicated cystic lymphangiomas.39 There have been no reported cases of malignant transformation of pancreatic/peripancreatic lymphangiomas, so a conservative approach and expectant follow-up remains the best plan for asymptomatic patients.40
Learning points.
Although peripancreatic and pancreatic lymphangiomas are rare tumours, this case illustrates the importance of including lymphangioma in the differential diagnosis when evaluating a cystic lesion of the pancreas.
Endoscopic ultrasound with fine-needle aspiration-based diagnosis of cystic lesions of the pancreatic/peripancreatic lesions can avoid surgeries in patients with asymptomatic, incidental lymphangiomas.
In experienced hands, endoscopic ultrasound-guided fine-needle aspiration is very helpful in elucidating a diagnosis to guide treatment decisions, as surgical removal of this tumour has excellent prognosis.
Endoscopic ultrasound with fine-needle aspiration can provide high resolution imaging of pancreatic and peripancreatic organs and vasculature.
Acknowledgments
The authors thank Ruth Mahmood, MD, of Ministry of Health Care—St. Clare's Hospital for assistance with the patient's care and Marie Fleisner of the Marshfield Clinic Research Foundation's Office of Scientific Writing and Publication for editorial assistance in the preparation of this report.
Footnotes
Contributors: ST, Rafiullah, and AAO provided the patient case; JR conducted the pathology/histology examination and provided slides; all authors helped write the case report, and approved final submission.
Competing interests: None.
Patient consent: Obtained.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Christie JP, Karlan MS. Lymphangioma of the pancrease with symptoms of ‘acute surgical abdomen’. Calif Med 1969;2013:22–4 [PMC free article] [PubMed] [Google Scholar]
- 2.Paal E, Thompson LD, Heffess CS. A clinicopathological and a immunohistochemical study of ten pancreatic lymphangiomas and a review of the literature. Cancer 1998;2013:2150–8 [DOI] [PubMed] [Google Scholar]
- 3.Colovic RB, Grubor NM, Micev MT, et al. Cystic lymphangioma of the pancreas. World J Gastroenterol 2008;2013:6873–5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Daltrey IR, Jonhson CD. Cystic lymphangioma of the pancreas. Postgrad Medical J 1996;2013:564–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wayne ER, Burrington JD, Bailey WC, et al. Retroperitoneal lymphangioma: an unusual case of acute surgical abdomen. J. Pediatric Surg 1973;2013:831–2 [DOI] [PubMed] [Google Scholar]
- 6.Ng IO, Fan ST, Nicholls J. Lymphangioma of the pancreas. J. Gastroenterol Hepatol 1999;2013:299–302 [DOI] [PubMed] [Google Scholar]
- 7.Gray G, Fried K, Iraci J. Cystic lymphangioma of the pancreas: CT and pathologic findings. Abdom Imaging 1998;2013:78–80 [DOI] [PubMed] [Google Scholar]
- 8.Kalof AN, Cooper K. D2-40 immunohistochemistry—so far! Adv Anat Pathol 2009;2013:62–4 [DOI] [PubMed] [Google Scholar]
- 9.Koch K. Beitrage zur pathologie der Bauchspeicheldruse. Virchows Arch 1913;2013:180–206 [Google Scholar]
- 10.Lyngdoh TS, Konsam R, Th B, et al. Giant cystic lymphangioma of pancreas. ANZ J Surg 2008;2013:673–4 [DOI] [PubMed] [Google Scholar]
- 11.Igarashi A, Maruo Y, Ito T, et al. Huge cystic lymphangioma of the pancreas: report of a case. Surg Today 2001;2013:743–6 [DOI] [PubMed] [Google Scholar]
- 12.Abe H, Kubota K, Noie T, et al. Cystic lymphangioma of the pancreas: a case report with special reference to embryological development. Am J Gastroenterol 1997;2013:1566–7 [PubMed] [Google Scholar]
- 13.Coe AW, Evans J, Conway J. Pancreas cystic lymphangioma diagnosed with EUS-FNA. JOP 2012;2013:282–4 [PubMed] [Google Scholar]
- 14.Saers T, Parusel M, Brockmann M, et al. Lymphangioma of the esophagus. Gastrointest Endosc 2005;2013:181–4 [DOI] [PubMed] [Google Scholar]
- 15.Dubois JD. Lymphangioma of the pancreas masquerading as acute appendicitis and torsion of the left testis. Br J Surg 1981;2013:799–800 [DOI] [PubMed] [Google Scholar]
- 16.Gun I, Schumer W, Veiga S. Cystic lymphangioma of the pancreas: case report. Illinois Med J 1962;2013:651–3 [Google Scholar]
- 17.Blumhagen JD, Wood BJ, Rosenbaum DM. Sonographic evaluation of abdominal lymphangiomas in children. J Ultrasound Med 1987;2013:487–95 [DOI] [PubMed] [Google Scholar]
- 18.Kafka V, Novak Multicystic retroperitoneal lymphangioma in an infant appearing as an inguinal hernia. J Pediatr Surg 1970;2013:573. [DOI] [PubMed] [Google Scholar]
- 19.Allen JG, Riall TS, Cameron JL, et al. Abdominal lymphangiomas in adults. J Gastrointest Surg 2006;2013:746–51 [DOI] [PubMed] [Google Scholar]
- 20.Schneider G, Seidel R, Altmeyer K, et al. Lymphangioma of the pancreas and the duodenal wall: MR imaging findings. Eur Radiol 2001;2013:2232–5 [DOI] [PubMed] [Google Scholar]
- 21.Witte MH, Witte CL. Lymphangiogenesis and lymphologic syndromes. Lymphology 1986;2013:21–8 [PubMed] [Google Scholar]
- 22.St Laurent R. Abdominal Lymphangiomas. Int Surg 1966;2013:596–601 [PubMed] [Google Scholar]
- 23.Khandelwal M, Lichtenstein GR, Morris JB, et al. Abdominal lymphangioma masquerading as a pancreatic cystic neoplasm. J Clinical Gastroenterol 1995;2013:142–4 [DOI] [PubMed] [Google Scholar]
- 24.Rolsman I, Manny J, Fields S, et al. Intra-abdominal lymphangioma. Br J Surg 1989;2013:485–9 [DOI] [PubMed] [Google Scholar]
- 25.Letquart JP, Marcorelles P, Lancien G, et al. [A new case of cystic lymphangioma of the pancreas.] [article in French]. J Chir (Paris) 1989;2013:650–8 [PubMed] [Google Scholar]
- 26.Burkett JS, Pickelman J. The rationale for surgical treatment of mesenteric and retroperitoneal cysts. Am Surg 1994;2013:432–5 [PubMed] [Google Scholar]
- 27.De Jong SA, Pickleman J, Rainsford K. Nonductal tumors of the pancreas. The importance of laparotomy. Arch Surg 1993;2013:734–6 [DOI] [PubMed] [Google Scholar]
- 28.Roisman I, Manny J, Fields S, et al. Intra-abdominal lymphangioma. Br J Surg 1989;2013:485–9 [DOI] [PubMed] [Google Scholar]
- 29.Malnofski MJ, Poulton TB, Nazinitsky KJ, et al. Prenatal ultrasonic diagnosis of retroperitoneal cyctic lymphangioma. J Ultrasound Med 1993;2013:427–9 [DOI] [PubMed] [Google Scholar]
- 30.Salimi Z, Fishbein M, Welvorsen MK, et al. Pancreatic lymphangioma: CT, MRI, and angiographic features. Gastrointest Radiol 1991;2013:248–50 [DOI] [PubMed] [Google Scholar]
- 31.Sedlack R, Affi A, Vazquez-Sequeiros E, et al. Utility of EUS in the evaluation of cystic pancreatic lesions. Gastrointest Endosc 2002;2013:543–7 [DOI] [PubMed] [Google Scholar]
- 32.Raddaoui E. Clinical utility and diagnostic accuracy of endoscopic ultrasound-guided fine needle aspiration of pancreatic lesions: Saudi Arabian experience. Acta Cytol 2011;2013:26–9 [DOI] [PubMed] [Google Scholar]
- 33.Bounds BC, Brugge WR. EUS diagnosis of cystic lesions of the pancreas. Int J Gastrointest Cancer 2001;2013:27–31 [DOI] [PubMed] [Google Scholar]
- 34.Jathal A, Arsenescu R, Crowe G, et al. Diagnosis of pancreatic cystic lymphangioma with EUS-guided FNA: report of a case. Gastrointest Endosc 2005;2013:920–2 [DOI] [PubMed] [Google Scholar]
- 35.Applebaum B, Cunningham JT. Two cases of cystic lymphangioma of the pancreas: a rare finding in endoscopic ultrasonography. Endoscopy 2006;2013:E24–5 [DOI] [PubMed] [Google Scholar]
- 36.Hayashi J, Yamashita Y, Kakegawa T, et al. A case of cystic lymphangioma of the pancreas. J Gastroenterol 1994;2013:372–6 [DOI] [PubMed] [Google Scholar]
- 37.Koshy A, Tandon RK, Kapur BM, et al. Retroperitoneal lymphangioma: a case report with review of the literature. Am J Gastroenterol 1978;2013:485–90 [PubMed] [Google Scholar]
- 38.Hebra A, Brown MF, McGeehin KM, et al. Mesenteric, omental, and retroperitoneal cysts in children: a clinical study of 22 cases. South Medical J 1993;2013:173–6 [DOI] [PubMed] [Google Scholar]
- 39.Gureş N, Gurluler E, Alim A, et al. Cystic pancreatic lymphangioma. Rare Tumors 2012;2013:e27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sanaka MR, Kowalski TE. Cystic lymphangioma of the pancreas. Clin Gastroenterol Hepatol 2007;2013:10–11 [DOI] [PubMed] [Google Scholar]