Abstract
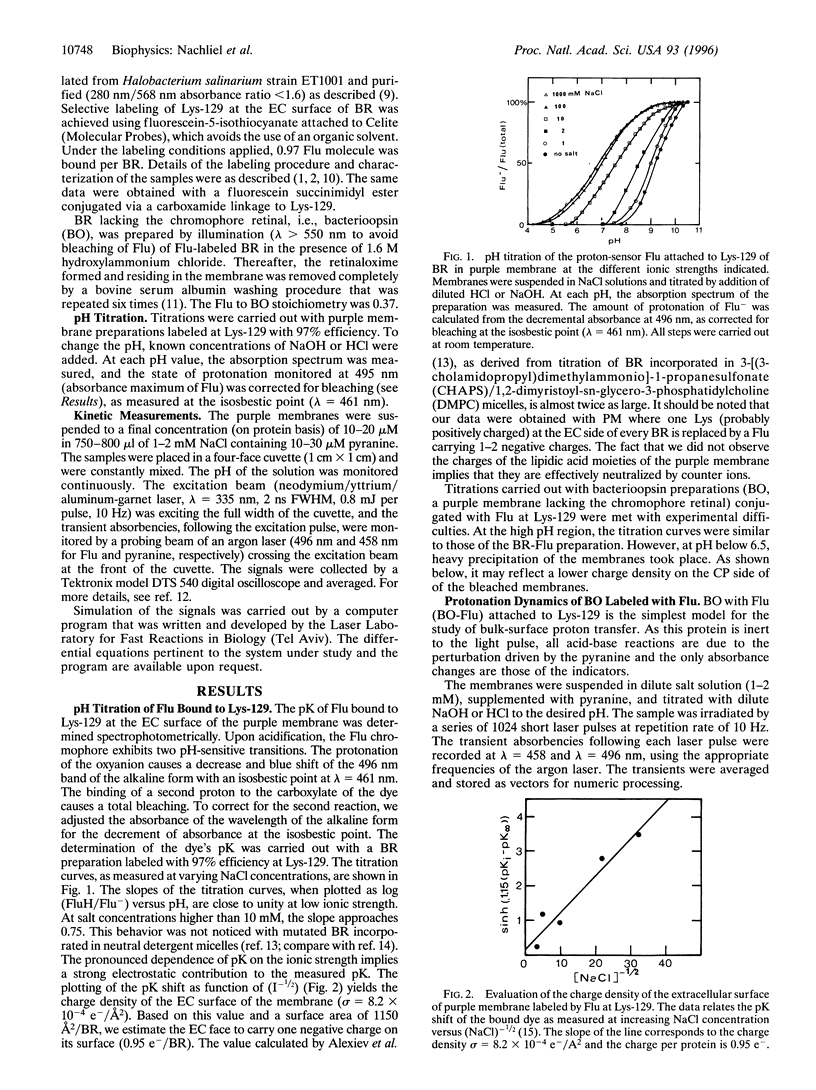
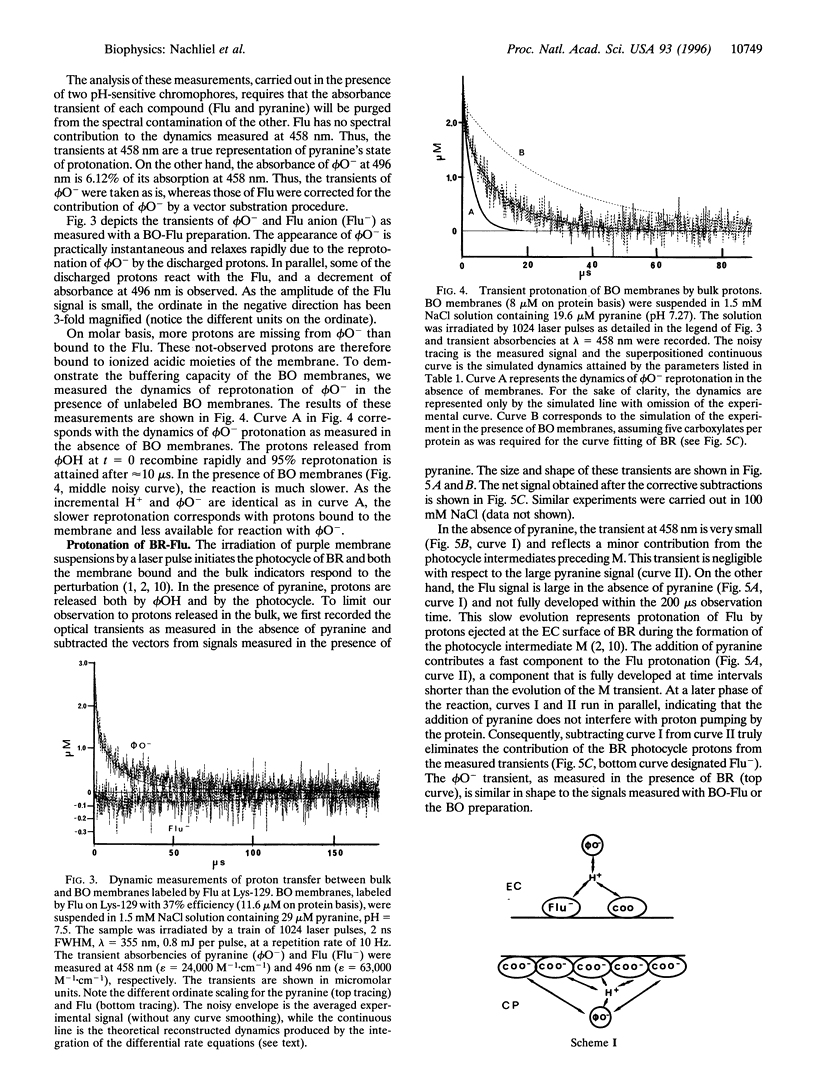
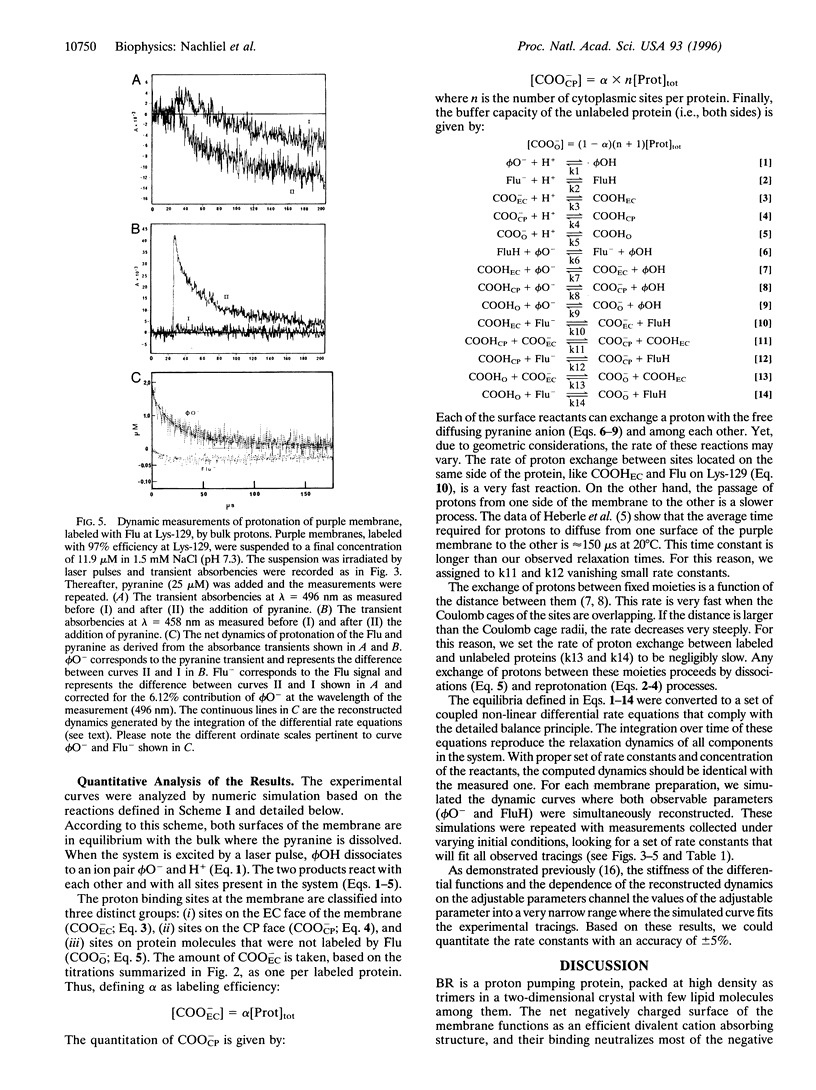
The dynamics of proton binding to the extracellular and the cytoplasmic surfaces of the purple membrane were measured by laser-induced proton pulses. Purple membranes, selectively labeled by fluorescein at Lys-129 of bacteriorhodopsin, were pulsed by protons released in the aqueous bulk from excited pyranine (8-hydroxy-1,3,6-pyrenetrisulfonate) and the reaction of protons with the indicators was measured. Kinetic analysis of the data imply that the two faces of the membrane differ in their buffer capacities and in their rates of interaction with bulk protons. The extracellular surface of the purple membrane contains one anionic proton binding site per protein molecule with pK = 5.1. This site is within a Coulomb cage radius (approximately 15 A) from Lys-129. The cytoplasmic surface of the purple membrane bears 4-5 protonable moieties (pK = 5.1) that, due to close proximity, function as a common proton binding site. The reaction of the proton with this cluster is at a very fast rate (3.10(10) M-1.s-1). The proximity between the elements is sufficiently high that even in 100 mM NaCl they still function as a cluster. Extraction of the chromophore retinal from the protein has a marked effect on the carboxylates of the cytoplasmic surface, and two to three of them assume positions that almost bar their reaction with bulk protons. The protonation dynamics determined at the surface of the purple membrane is of relevance both for the vectorial proton transport mechanism of bacteriorhodopsin and for energy coupling, not only in halobacteria, but also in complex chemiosmotic systems such as mitochondrial and thylakoid membranes.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alexiev U., Marti T., Heyn M. P., Khorana H. G., Scherrer P. Surface charge of bacteriorhodopsin detected with covalently bound pH indicators at selected extracellular and cytoplasmic sites. Biochemistry. 1994 Jan 11;33(1):298–306. doi: 10.1021/bi00167a039. [DOI] [PubMed] [Google Scholar]
- Alexiev U., Mollaaghababa R., Scherrer P., Khorana H. G., Heyn M. P. Rapid long-range proton diffusion along the surface of the purple membrane and delayed proton transfer into the bulk. Proc Natl Acad Sci U S A. 1995 Jan 17;92(2):372–376. doi: 10.1073/pnas.92.2.372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bauer P. J., Dencher N. A., Heyn M. P. Evidence for chromophore-chromophore interactions in the purple membrane from reconstitution experiments of the chromophore-free membrane. Biophys Struct Mech. 1976 Apr 15;2(1):79–92. doi: 10.1007/BF00535654. [DOI] [PubMed] [Google Scholar]
- Cao Y., Brown L. S., Sasaki J., Maeda A., Needleman R., Lanyi J. K. Relationship of proton release at the extracellular surface to deprotonation of the schiff base in the bacteriorhodopsin photocycle. Biophys J. 1995 Apr;68(4):1518–1530. doi: 10.1016/S0006-3495(95)80324-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dencher N. A., Burghaus P. A., Grzesiek S. Determination of the net proton-hydroxide ion permeability across vesicular lipid bilayers and membrane proteins by optical probes. Methods Enzymol. 1986;127:746–760. doi: 10.1016/0076-6879(86)27059-7. [DOI] [PubMed] [Google Scholar]
- Dencher N. A., Dresselhaus D., Zaccai G., Büldt G. Structural changes in bacteriorhodopsin during proton translocation revealed by neutron diffraction. Proc Natl Acad Sci U S A. 1989 Oct;86(20):7876–7879. doi: 10.1073/pnas.86.20.7876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ehrenberg B. Spectroscopic methods for the determination of membrane surface charge density. Methods Enzymol. 1986;127:678–696. doi: 10.1016/0076-6879(86)27053-6. [DOI] [PubMed] [Google Scholar]
- Fodor S. P., Ames J. B., Gebhard R., van den Berg E. M., Stoeckenius W., Lugtenburg J., Mathies R. A. Chromophore structure in bacteriorhodopsin's N intermediate: implications for the proton-pumping mechanism. Biochemistry. 1988 Sep 6;27(18):7097–7101. doi: 10.1021/bi00418a064. [DOI] [PubMed] [Google Scholar]
- Gutman M. Application of the laser-induced proton pulse for measuring the protonation rate constants of specific sites on proteins and membranes. Methods Enzymol. 1986;127:522–538. doi: 10.1016/0076-6879(86)27042-1. [DOI] [PubMed] [Google Scholar]
- Gutman M., Kotlyar A. B., Borovok N., Nachliel E. Reaction of bulk protons with a mitochondrial inner membrane preparation: time-resolved measurements and their analysis. Biochemistry. 1993 Mar 30;32(12):2942–2946. doi: 10.1021/bi00063a003. [DOI] [PubMed] [Google Scholar]
- Heberle J., Dencher N. A. Bacteriorhodopsin in ice. Accelerated proton transfer from the purple membrane surface. FEBS Lett. 1990 Dec 17;277(1-2):277–280. doi: 10.1016/0014-5793(90)80864-f. [DOI] [PubMed] [Google Scholar]
- Heberle J., Dencher N. A. Surface-bound optical probes monitor protein translocation and surface potential changes during the bacteriorhodopsin photocycle. Proc Natl Acad Sci U S A. 1992 Jul 1;89(13):5996–6000. doi: 10.1073/pnas.89.13.5996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heberle J., Riesle J., Thiedemann G., Oesterhelt D., Dencher N. A. Proton migration along the membrane surface and retarded surface to bulk transfer. Nature. 1994 Aug 4;370(6488):379–382. doi: 10.1038/370379a0. [DOI] [PubMed] [Google Scholar]
- Kasianowicz J. J., Bezrukov S. M. Protonation dynamics of the alpha-toxin ion channel from spectral analysis of pH-dependent current fluctuations. Biophys J. 1995 Jul;69(1):94–105. doi: 10.1016/S0006-3495(95)79879-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koch M. H., Dencher N. A., Oesterhelt D., Plöhn H. J., Rapp G., Büldt G. Time-resolved X-ray diffraction study of structural changes associated with the photocycle of bacteriorhodopsin. EMBO J. 1991 Mar;10(3):521–526. doi: 10.1002/j.1460-2075.1991.tb07978.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakasako M., Kataoka M., Amemiya Y., Tokunaga F. Crystallographic characterization by X-ray diffraction of the M-intermediate from the photo-cycle of bacteriorhodopsin at room temperature. FEBS Lett. 1991 Nov 4;292(1-2):73–75. doi: 10.1016/0014-5793(91)80837-s. [DOI] [PubMed] [Google Scholar]
- Subramaniam S., Gerstein M., Oesterhelt D., Henderson R. Electron diffraction analysis of structural changes in the photocycle of bacteriorhodopsin. EMBO J. 1993 Jan;12(1):1–8. doi: 10.1002/j.1460-2075.1993.tb05625.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szundi I., Stoeckenius W. Purple-to-blue transition of bacteriorhodopsin in a neutral lipid environment. Biophys J. 1988 Aug;54(2):227–232. doi: 10.1016/S0006-3495(88)82951-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yam R., Nachliel E., Kiryati S., Gutman M., Huppert D. Proton transfer dynamics in the nonhomogeneous electric field of a protein. Biophys J. 1991 Jan;59(1):4–11. doi: 10.1016/S0006-3495(91)82192-0. [DOI] [PMC free article] [PubMed] [Google Scholar]



