Abstract
Data characterizing demographics, treatment patterns, and clinical outcomes in black patients with human epidermal growth factor receptor 2 (HER2)-positive metastatic breast cancer (MBC) are limited. registHER is a large, observational cohort study of patients (n = 1,001) with HER2-positive MBC diagnosed ≤6 months of enrollment and followed until death, disenrollment, or June 2009 (median follow-up of 27 months). Demographics, treatment patterns, and clinical outcomes were described for black (n = 126) and white patients (n = 793). Progression-free survival (PFS) following first-line therapy and overall survival (OS) were examined. Multivariate analyses adjusted for baseline and treatment factors. Black patients were more likely than white patients to be obese (body mass index ≥30), to have diabetes, and to have a history of cardiovascular disease; they were also less likely to have estrogen receptor or progesterone receptor positive disease. In patients treated with trastuzumab, the incidence of cardiac safety events (grade ≥3) was higher in black patients (10.9 %) than in white patients (7.9 %). Unadjusted median OS and PFS (months) were significantly lower in black patients than in white patients (OS: black: 27.1, 95 % confidence interval [CI] 21.3–32.1; white: 37.3, 95 % CI 34.6–41.1; PFS: black: 7.0, 95 % CI 5.7–8.2; white: 10.2, 95 % CI 9.3–11.2). The adjusted OS hazard ratio (HR) for black patients compared with white patients was 1.29 (95 % CI 1.00–1.65); adjusted PFS HR was 1.29 (95 % CI 1.05–1.59). This real-world evaluation of a large cohort of patients with HER2-positive MBC shows poorer prognostic factors and independently worse clinical outcomes in black versus white patients. Further research is needed to identify potential biologic differences that could have predictive impact for black patients or that could explain these differences.
Keywords: Racial disparities, HER2-positive, Metastatic breast cancer, registHER, Observational study, Treatment patterns
Introduction
Previous literature has shown that, while the incidence of breast cancer in black women is lower than in white women, black women are more likely to die as a result of the disease [1–5], are diagnosed at an earlier age, have a higher grade at diagnosis [3, 4, 6], and have a greater risk of recurrence [4]. One factor associated with this racial disparity is being diagnosed at a later stage [1, 3, 7], which may be contributed to by socioeconomic factors and access to care [3, 7]. Reproductive variables associated with breast cancer outcome have been shown to differ between black and white women, including menopausal status, use of contraception, breast feeding behaviors [7, 8], and body mass index (BMI) [4, 7, 8]. However, even after controlling for such factors, disparities in health outcomes are still apparent, with several studies revealing potential underlying biologic differences between black and white breast cancer patients [2, 7]. In particular, a greater incidence of triple-negative breast cancer (TNBC), i.e., estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2) negative or basal-like breast cancer, has been demonstrated in black women [2, 3], even when controlling for age and BMI [4]. Though data are limited, several inherited genetic factors may contribute to this biologic association; Olopade and colleagues have associated specific germline BRCA1 mutations with high-risk African American kindreds [9].
About 20–25 % of breast cancers have overexpression of HER2 [10, 11] which is both prognostic and predictive of treatment benefit. Overexpression is associated with a higher risk of and a shorter time to relapse, as well as poorer prognosis, worse survival, and a higher likelihood of response to both specific chemotherapy regimens and biologic therapy targeted to HER2 [2, 10, 12]. Data characterizing HER2 positive metastatic breast cancer (MBC) in black patients are limited. The objective of this analysis was to examine demographics, treatment patterns, and clinical outcomes in black and white patients with HER2-positive MBC in the registHER observational cohort.
Methods
registHER study design
The registHER study (NCT00105456; clinicaltrials.gov) is a multicenter, prospective, observational, US-based cohort study of patients with newly diagnosed HER2-positive MBC (N = 1,023). Patients were diagnosed within 6 months of enrollment and were recruited from community and academic settings between December 2003 and February 2006. Study design and recruitment details are described elsewhere [13]. The objectives of registHER were to describe the natural history of disease and treatment patterns for patients with HER2-positive MBC and to explore associations between specific therapies and patient outcomes. Patients received care according to their physicians’ standard practice, without study-specified evaluations. Prior or planned treatment with trastuzumab, or any specific HER2-targeted therapy, was not a requirement for enrollment. All patients signed an informed consent and authorized the disclosure of their health information. There were no exclusion criteria; however, patients who did not provide informed consent and did not authorize the disclosure of health information were excluded.
Patient information was recorded at enrollment and updated every 3 months. Information collected included treatment history, sites of progressive disease, tumor response, survival, cardiac safety (grades 3/4/5), and adverse events potentially related to the administration of trastuzumab. First-line treatment patterns are based on treatment received prior to first disease progression and may have been given sequentially or concurrently. History of metabolic disorders, cardiovascular disease (CVD), and data on cardiac safety were collected using predefined checkboxes. Cardiac safety events were defined based on the Common Terminology Criteria for Adverse Events, version 3.0, and selected based on physician subjective opinion [14]. The schedule for tumor assessment and time of tumor progression was determined by treating physicians per their standards of care.
Statistical analysis
Of the 1,023 patients in the registHER cohort, 22 did not receive any treatment during the study and were excluded from the analysis. Due to small numbers, Hispanic patients (n = 56), Asian/Pacific Islander patients (n = 18), and patients classified as “other” races (n = 8) were also excluded. Ultimately, 126 black patients and 793 white patients were included in this study for which demographics, treatment patterns, and clinical outcomes were described (n = 919).
Progression-free survival (PFS) and overall survival (OS) from time of diagnosis of MBC were analyzed using the Kaplan–Meier method. Patients without a record of disease progression or death were censored at the last followup date or at the data lock date (June 15, 2009), whichever occurred earlier. Multivariate Cox regression models were used to generate hazard ratios (HRs) and 95 % confidence intervals (CIs) to assess the effect of race (black vs. white) on survival outcomes, while adjusting for clinically significant baseline treatment and prognostic factors. Multivariate models were adjusted for baseline factors including age at enrollment, BMI, Eastern Cooperative Oncology Group (ECOG) performance status, serum albumin level, tumor ER/PR status, site of metastatic disease (adjusted using a hierarchical approach, i.e., [1] central nervous system [CNS], [2] bone only or bone plus breast, [3] visceral/other, and [4] node/local), number of metastatic sites, stage of disease at initial diagnosis, history of underlying CVD, and history of other underlying noncardiac comorbid conditions. The final multivariate models further adjusted for first-line treatment variables among patients, including receipt of trastuzumab, taxanes, and hormonal therapy. Differences in PFS and OS for black versus white patients were also investigated within subcategories of baseline and clinical characteristics, as well as treatment factors.
Results
Patient characteristics
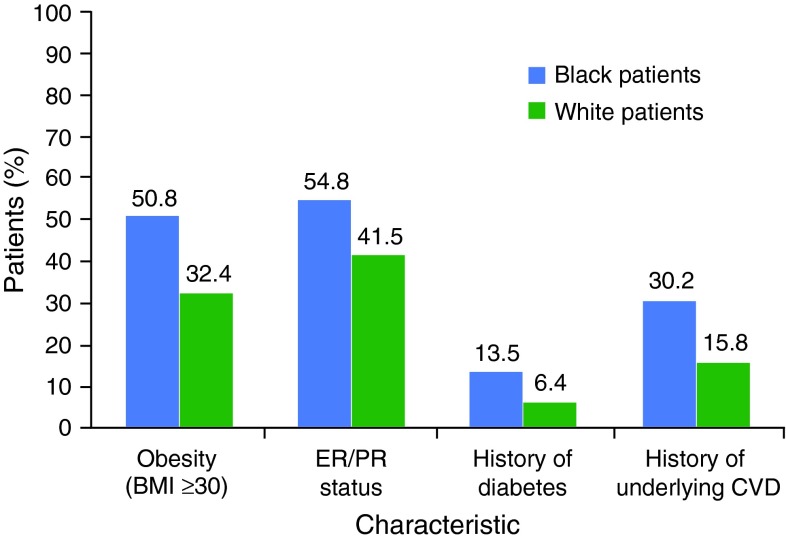
Baseline demographics and clinical characteristics for the black and white patients who were followed until death, disenrollment, or the June 2009 data lock date are shown in Table 1. Black patients were more likely to have discontinued due to death and loss to followup compared with white patients (62.7 vs. 53.1 % and 8.7 vs. 4.9 %, respectively). The median follow-up time was 21.0 months for black patients and 28.4 months for white patients. Black patients were slightly younger, had a higher prevalence of obesity (50.8 vs. 32.4 %), and had more than twice the prevalence of diabetes (13.5 vs. 6.4 %) at baseline than white patients. In addition, twice as many black patients had a history of underlying CVD at baseline than did white patients (30.2 vs. 15.8 %) (see Table 1; Fig. 1). Among black patients, hypertension with complications (11.1 %) and other cardiac diseases (20.6 %) were the most commonly reported CVD conditions.
Table 1.
Baseline demographic and clinical characteristics of black and white patients at diagnosis of MBC
| Characteristic | Black patients (n = 126) | White patients (n = 793) |
|---|---|---|
| Median follow-up, months | 21.0 | 28.4 |
| Median age, years (min–max) | 50 (20–90) | 54 (22–92) |
| Sex, n (%) | ||
| Female | 125 (99.2) | 784 (98.9) |
| BMI, kg/m2 | ||
| <30 | 62 (49.2) | 536 (67.6) |
| ≥30 | 64 (50.8) | 257 (32.4) |
| History of diabetes, n (%) | 17 (13.5) | 51 (6.4) |
| History of underlying CVD (any),a n (%) | 38 (30.2) | 125 (15.8) |
| Arrhythmia | 5 (4.0) | 16 (2.0) |
| Congestive heart failure | 4 (3.2) | 10 (1.3) |
| Hypertension with complications | 14 (11.1) | 33 (4.2) |
| Angina | 1 (0.8) | 2 (0.3) |
| Myocardial infarction | 3 (2.4) | 16 (2.0) |
| Peripheral vascular disease | 2 (1.6) | 10 (1.3) |
| Other underlying cardiac disease | 26 (20.6) | 65 (8.2) |
| Site of metastatic disease at diagnosis, n (%) | ||
| Any CNS | 11 (8.7) | 57 (7.2) |
| Bone only or bone plus breast | 19 (15.1) | 114 (14.4) |
| Visceral | 79 (62.7) | 487 (61.4) |
| Node/local | 17 (13.5) | 133 (16.8) |
| Other sites | 0 (0.0) | 2 (0.3) |
| ECOG performance status at diagnosis, n (%) | ||
| 0–1 | 54 (42.9) | 366 (46.2) |
| 2+ | 12 (9.5) | 43 (5.4) |
| Unknown/missing | 60 (47.6) | 384 (48.4) |
| ER/PR status, n (%) | ||
| ER-positive or PR-positive | 53 (42.1) | 434 (54.7) |
| ER-negative and PR-negative | 69 (54.8) | 329 (41.5) |
| Unknown | 4 (3.2) | 30 (3.8) |
| Clinical stage at initial diagnosis, n (%) | ||
| Stage I–III, MBC ≤12 months after initial diagnosis | 23 (18.3) | 98 (12.4) |
| Stage I–III, MBC >12 months after initial diagnosis | 64 (50.8) | 484 (61.0) |
| Stage IV | 39 (31.0) | 211 (26.6) |
aPatients could be counted in multiple CVD categories
BMI body mass index, CVD cardiovascular disease, CNS central nervous system, ECOG Eastern Cooperative Oncology Group, ER estrogen receptor; max maximum, MBC metastatic breast cancer, min minimum, PR progesterone receptor
Fig. 1.
Select clinical characteristics in black and white patients at the time of metastatic breast cancer diagnosis. BMI body mass index, CVD cardiovascular disease, ER estrogen receptor, PR progesterone receptor
The distribution of sites of metastatic disease at diagnosis was similar for black and white patients, with visceral sites being the most common and CNS being the least common for patients of both races. More black patients than white patients, however, had an ECOG performance status of ≥2 at diagnosis, had ER/PR-negative disease, and presented with de novo stage IV MBC or recurrent stage IV MBC within 12 months of initial stage I–III diagnosis (see Table 1).
Adjuvant treatment patterns in patients with recurrent disease
Treatment patterns in black and white patients with recurrent disease were similar (Table 2). Among these patients, the majority received adjuvant systemic therapy (66.7 % of black patients, 75.4 % of white patients). Approximately 7 % of both black and white patients received neoadjuvant or adjuvant trastuzumab.
Table 2.
Early-stage treatment patterns in black and white patients with recurrent disease
| Treatment, n (%) | Black patients (n = 87) | White patients (n = 582) |
|---|---|---|
| Neoadjuvant systemic therapy | 22 (25.3) | 118 (20.3) |
| Adjuvant systemic therapy | 58 (66.7) | 439 (75.4) |
| Neoadjuvant/adjuvant trastuzumab | 6 (6.9) | 44 (7.6) |
Patients diagnosed in stages 0–III only or with missing clinical cancer stage
Treatment patterns prior to first disease progression
Initial regimens for MBC usually included trastuzumab, and its percentage of use was comparable between black and white patients (81 and 84 %, respectively). Consistent with the observed difference in ER/PR statuses, more black patients received chemotherapy and fewer received hormonal therapy with or without chemotherapy than white patients (Table 3).
Table 3.
Treatment patterns for MBC prior to first disease progression among black and white patients
| Treatment, n (%) | Black patients | White patients |
|---|---|---|
| Trastuzumab-based first-line regimensa | (n = 102) | (n = 670) |
| With chemotherapy only | 79 (77.5) | 438 (65.4) |
| With hormonal therapy only | 5 (4.9) | 42 (6.3) |
| With chemotherapy and hormonal therapy | 12 (11.8) | 138 (20.6) |
| Trastuzumab alone | 6 (5.9) | 52 (7.8) |
| Non-trastuzumab-based first-line regimens | (n = 24) | (n = 123) |
| Chemotherapy only | 9 (37.5) | 63 (51.2) |
| Hormonal therapy only | 10 (41.7) | 44 (35.8) |
| Chemotherapy and hormonal therapy | 4 (16.7) | 6 (4.9) |
| Untreated | 1 (4.2) | 10 (8.1) |
aThose in the trastuzumab-based first-line regimens are defined as patients receiving ≥21 days of trastuzumab in first-line therapy
MBC metastatic breast cancer
In patients who did not receive a trastuzumab-based regimen as first-line treatment, fewer black patients received chemotherapy only or were untreated, while more black patients received hormonal therapy or combined chemotherapy and hormonal therapy than white patients (see Table 3). The use of anthracycline and cumulative dose were comparable between black and white patients (data not shown).
Cardiac safety outcomes
Reported grade ≥3 cardiac events in black and white patients treated with trastuzumab (in any line) are shown in Table 4. There was a slightly higher incidence of grade ≥3 cardiac events in black patients (13/119 [10.9 %]) than in white patients (59/746 [7.9 %]). The incidence of congestive heart failure was 4.2 % (n = 5) in black patients and 1.6 % (n = 12) in white patients.
Table 4.
Incidence of reported cardiac adverse events (grades ≥3) in black and white patients treated with trastuzumab
| Adverse event, n (%) | Black patients (n = 119) | White patients (n = 746) |
|---|---|---|
| Any | 13 (10.9) | 59 (7.9) |
| Angina pectoris | 0 (0.0) | 2 (0.3) |
| Atrial arrhythmia | 1 (0.8) | 4 (0.5) |
| Cardiac disorder (NOS) | 2 (1.7) | 11 (1.5) |
| Congestive heart failure | 5 (4.2) | 12 (1.6) |
| Left ventricular dysfunction | 3 (2.5) | 22 (2.9) |
| Myocardial infarction | 2 (1.7) | 2 (0.3) |
| Pericardial effusion | 0 (0.0) | 5 (0.7) |
| Ventricular arrhythmia | 0 (0.0) | 1 (0.1) |
NOS not otherwise specified
When stratified by underlying disease history, black patients treated with trastuzumab and with a history of diabetes, hypertension with complications, or CVD were more likely to have cardiac safety events than white patients with the same conditions. Specifically, of the black patients who reported a history of diabetes and who had received ≥1 dose of trastuzumab (n = 15), three (20.0 %) reported any cardiac safety event compared with four white patients (n = 48; 8.3 %). Similarly, of the black patients who reported a history of hypertension with complications and who had received ≥1 dose of trastuzumab (n = 14), two (14.2 %) had any cardiac safety event compared with two white patients (n = 29; 6.9 %). A history of CVD appeared to have less of an impact; of the black patients with any history of CVD and who had received ≥1 dose of trastuzumab (n = 36), five patients (13.9 %) had a cardiac safety event compared with 12 white patients (n = 117; 10.3 %).
Efficacy and clinical outcomes
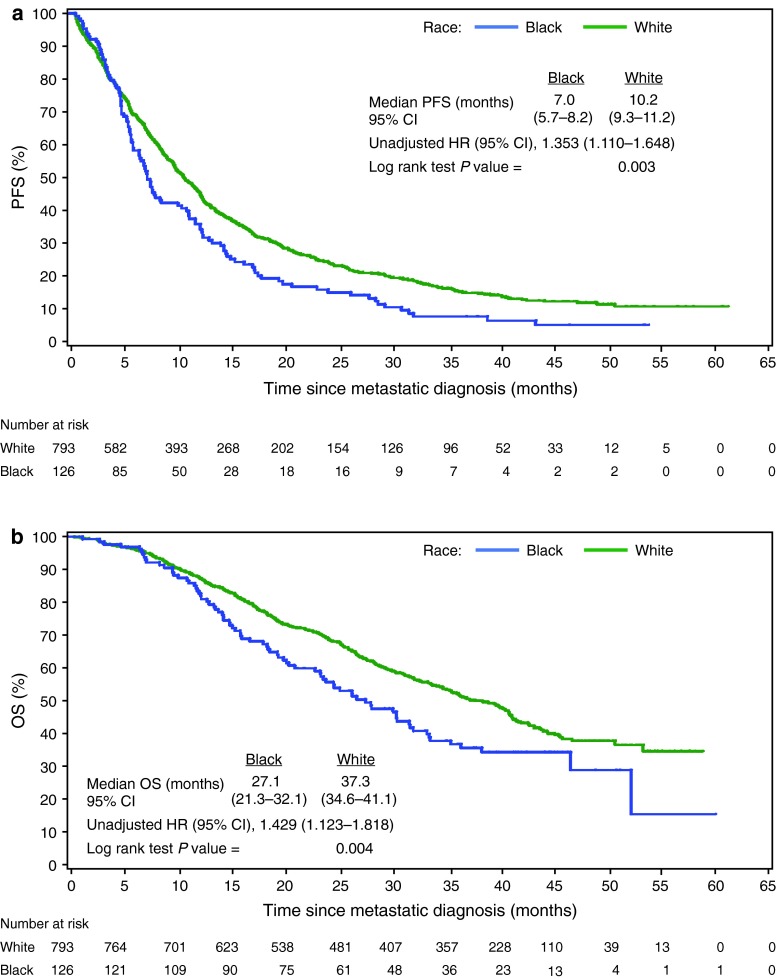
The unadjusted median first-line PFS was substantially lower among black patients than among white patients (7.0 months [95 % CI 5.7–8.2] vs. 10.2 months [95 % CI 9.3–11.2]; Fig. 2a). Unadjusted median OS was also substantially lower among black patients than among white patients (27.1 months [95 % CI 21.3–32.1] vs. 37.3 months [95 % CI 34.6–41.1]; Fig. 2b).
Fig. 2.
Kaplan–Meier plots showing first-line a PFS and b OS since diagnosis of metastatic breast cancer in black and white patients. CI confidence interval, HR hazard ratio, OS overall survival, PFS progression-free survival
To determine whether race had an effect on survival independent of baseline prognostic and treatment factors, we conducted a multivariate analysis controlling for baseline demographics, treatment patterns, and clinical characteristics. After adjusting for these factors, black patients had a significantly greater risk for disease progression compared with white patients (adjusted HR, 1.29; 95 % CI 1.05–1.59; P = 0.015), as well as a significantly greater risk of death (adjusted HR, 1.29; 95 % CI 1.00–1.65; P = 0.049).
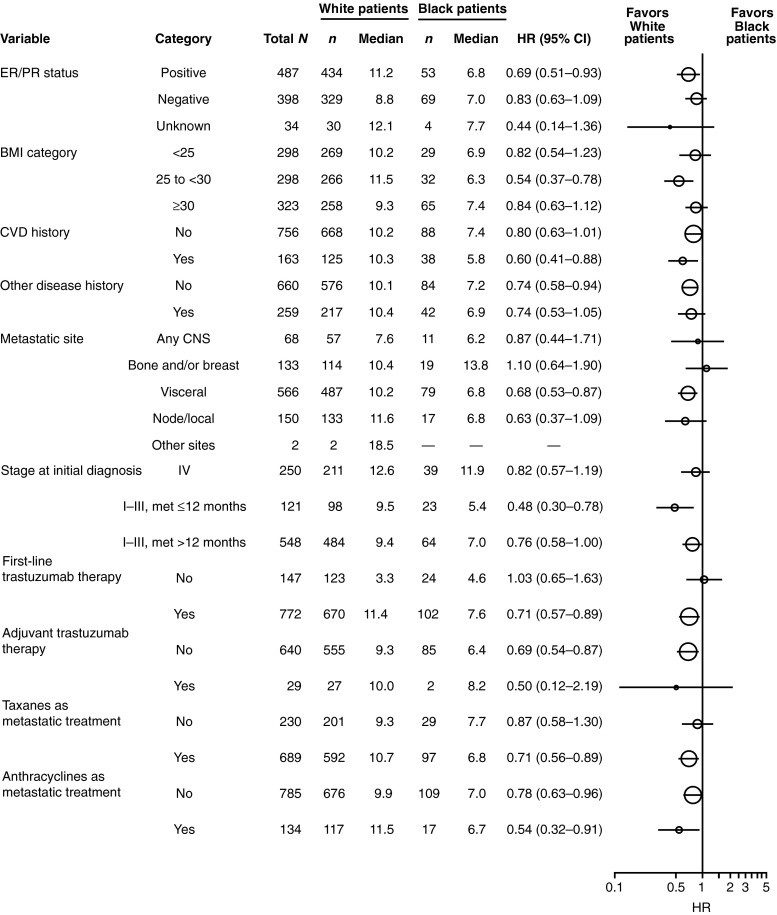
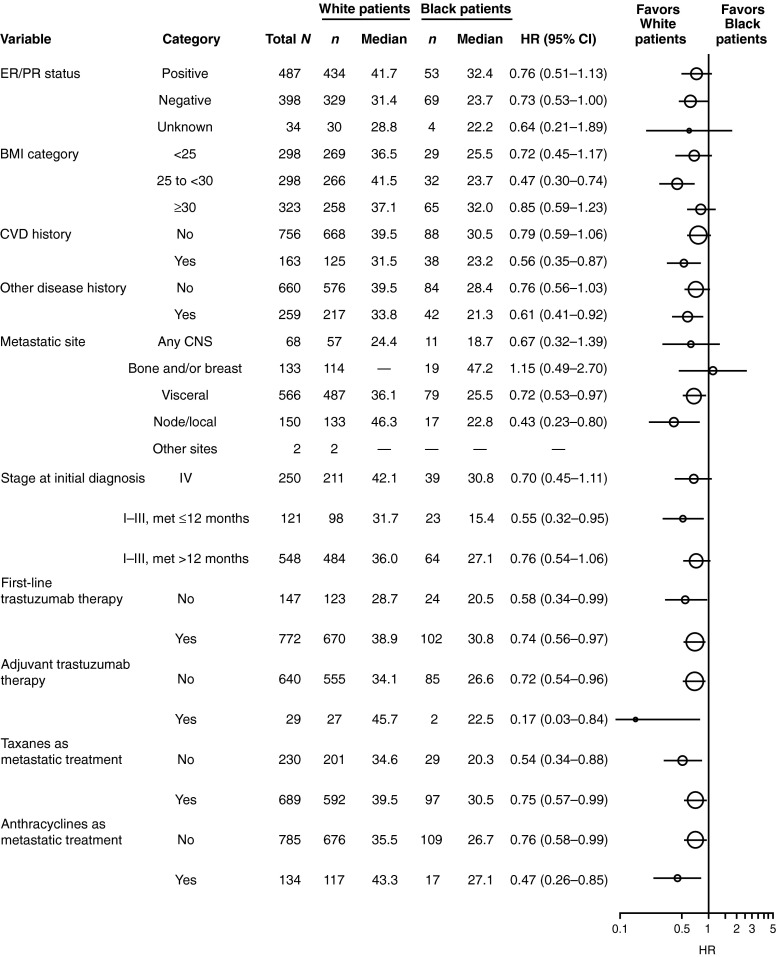
In PFS and OS analyses within subcategories of baseline and clinical characteristics (ER/PR status, BMI category, history of CVD, other disease history, metastatic site, and stage at initial diagnosis), as well as treatment patterns (first-line trastuzumab or adjuvant trastuzumab; taxanes or anthracyclines for metastatic disease), the median PFS and OS consistently favored white patients (Figs. 3, 4). There was a trend toward poorer PFS and OS in black patients compared with white patients for both ER/PR-positive and ER/PR-negative statuses.
Fig. 3.
Analysis of progression-free survival (PFS) by baseline characteristics and treatment patterns in black and white patients. BMI body mass index, CI confidence interval, CNS central nervous system, CVD cardiovascular, ER estrogen receptor, HR hazard ratio, met metastases, PR progesterone receptor
Fig. 4.
Analysis of overall survival (OS) by baseline characteristics and treatment patterns in black and white patients. BMI body mass index, CI confidence interval, CNS central nervous system, CVD cardiovascular, ER estrogen receptor, HR hazard ratio, met metastases, PR progesterone receptor
Among patients with disease progression after first-line therapy for advanced disease, rates of metastasis to node/local sites were nearly twice as high in black patients (22.2 vs. 13.0 %) as in white patients, while rates of metastases to bone only or bone plus breast were approximately twice as high in white patients as in black patients (21.1 vs. 10.2 %, respectively). Black and white patients had similar rates of metastases to CNS sites (18.5 vs. 20.8 %), visceral sites (47.2 vs. 43.9 %), and other sites (1.9 vs. 1.1 %), respectively.
Discussion
In this national registry study conducted in a real-world setting of patients with HER2-positive MBC, black patients were found to have more adverse prognostic factors compared with white patients, including the greater BMI and a higher proportion of ER/PR-negative tumors. These differences in prognostic factors may have contributed to the observed decrease in PFS following first-line therapy (unadjusted median: 7.0 months [95 % CI 5.7–8.2] vs. 10.2 months [95 % CI 9.3–11.2]), and OS (unadjusted median: 27.1 months [95 % CI 21.3–32.1] vs. 37.3 months [95 % CI 34.6–41.1]) in black patients compared with white patients. Multivariate analyses confirmed that, after adjustment for differences in prognostic factors, black patients had a 30 % greater risk of disease progression in the first-line setting compared with white patients, as well as a 30 % increase in risk of death.
Black patients in this study had a higher prevalence of obesity and diabetes; both metabolic disorders have independently been associated with breast cancers that exhibit aggressive biological characteristics and have poor prognoses [15]. Obesity and diabetes share similar biological mechanisms for their associations with breast cancer. In addition, in line with current population-based, epidemiologic data [16, 17], black patients in this study had a higher rate of underlying CVD at baseline compared with white patients and were more likely to present with de novo stage IV MBC or recurrent stage IV MBC within 12 months of initial stage I–III diagnosis. However, black patients consistently demonstrated poorer outcomes, even after adjusting for differences in BMI, the prevalence of underlying CVD between black and white patients, and stage at initial diagnosis.
The unexplained poorer outcomes in black patients with HER2-positive MBC, despite controlling for prognostic factors, suggest that other influences, in particular those underlying tumor and host biology, may play a role in the etiology and health outcomes of patients with HER2-positive breast cancer. One illustration of this can be observed in patients with tumors that are hormone receptor-negative, which are associated with poorer prognosis compared with hormone receptor-positive tumors [18]. Consistent with the findings in registHER, data from the large, population-based Surveillance, Epidemiology, and End Results (SEER) database have shown that African American breast cancer patients are more likely to have ER/PR-negative tumors than European American [19] and non-Hispanic white breast cancer patients [20].
Interestingly, while black patients in the registHER cohort of HER2-positive patients were more likely to have ER/PR-negative disease compared with white patients, there was a trend toward worse outcomes in black patients within both the ER/PR-positive and ER/PR-negative subsets. While the literature on this finding is limited, an early-stage analysis from a recent, large, randomized trial population treated with anthracycline- and taxane-containing chemotherapy found that racial differences in outcome were observed primarily in the ER-positive group but not in the ER-negative or HER2-positive groups [21].
There is evidence demonstrating that there are racial/ethnic differences in breast cancer molecular subtypes in addition to HER2-positive disease, as well as gene alterations, which have been associated with breast cancer outcomes. TNBC, which is associated with poor prognosis, an aggressive and early pattern of metastases, and limited therapeutic options [22], occurs at a higher rate in indigenous African women [3], as well as in African American women [2, 4, 23, 24], compared with white women. In a recently completed sequence analysis of 434 breast cancers in Nigerian patients, high frequencies of BRCA1 and BRCA2 mutations were found (7.1 and 3.9 %, respectively), and 16 different BRCA1 mutations were detected [25]. Alterations in p53 were significantly more common in African American women than in white women (odds ratio, 4.00; 95 % CI 1.77–9.01) in a study of 247 tumor samples, even after adjustment for disease stage at diagnosis and other prognostic factors [26]. In 141 tumor tissue samples analyzed from 72 African American and 69 Latina women with breast cancer, more than 70 % of the patients with HER2-positive disease had elevated Akt levels, a level higher than reported for other ethnic groups in most studies [27].
Given the increased prevalence of poor prognosis molecular subtypes and genetic alterations in black women with breast cancer, and the unexplained unfavorable outcomes in black patients in registHER, the current hypothesis-generating descriptive study suggests that expanded research into the role of the biology of HER2-positive MBC across race/ethnicity may provide an improved understanding of the etiology of these observations and could identify possible new targets for therapy.
To determine if racial disparities were evident for trastuzumab-associated cardiac safety events in registHER, reported cardiac safety events were compared in white and black patients treated with trastuzumab. Because the number of events in the analysis was small, results should be interpreted with caution. The incidence of cardiac safety events (collected via predefined checkboxes and physician subjective opinion) was slightly greater in black patients than white patients (10.9 vs. 7.9 %, respectively), as was the incidence of congestive heart failure (4.2 vs. 1.6 %, respectively). The contributing factors to this modest difference are unknown, but they are likely to include a higher incidence of hypertension that requires treatment in black vs. white patients, as it is known to be a risk factor for trastuzumab-induced cardiac toxicity [28]. Additional research is needed to investigate risk factors for cardiac toxicity by race among patients receiving trastuzumab.
The fact that socioeconomic data were not collected in registHER, and therefore data could not be adjusted for socioeconomic status in this analysis, is a weakness of the study. Socioeconomic factors like account for some proportion of breast cancer outcome disparities, but a majority of pooled and population-based studies show a persistence of survival differences in black patients and white patients after adjustment for socioeconomic factors [7, 29]. Interestingly, despite the observed increase in breast cancer mortality in African American breast cancer patients, these patients are more likely to experience delays in completion of adjuvant chemotherapy, are more likely to receive potentially inappropriate and substandard healthcare, are less likely to undergo preventive cancer screening, and are less likely to participate in clinical trials [7, 30].
In addition to the lack of socioeconomic data in registHER, an inherent limitation of this study is possible “confounding by indication” because of the nonrandomized, observational nature of the registHER study. The analysis may also include possible residual confounding due to the inability to control for prior treatments, treatment compliance, tumor size, menopausal status, and New York Heart Association Functional Classification. CVD and cardiovascular safety data were collected via predefined checkboxes and physicians subjective opinion rather than via validated methods. Also, the overall findings should be interpreted with caution due to the small number of events. Limited information was collected for cause of death (options included only “cancer” and “other”) and was missing for 32/538 deaths (>5 % of deaths) in the registHER cohort (n = 1,001), which precluded the calculation of breast cancer-specific mortality rates in this study.
Conclusions
These real-world data from the US-based registHER study show consistently poorer outcomes in black patients with HER2-positive MBC compared with white patients with the same disease, outcomes which were not explained by the higher prevalence of high-risk prognostic factors among black patients. These findings suggest a need to better understand possible underlying biological differences that could have a predictive or prognostic significance for black patients with HER2-positive MBC. Further prospective research is now ongoing through a second disease-based registry, SystHERs, which includes the collection of archival tumor and germline DNA to better understand not only the biology of resistance but also racial and other host differences, treatment patterns, and outcomes for patients with HER2-positive MBC.
Acknowledgments
The authors thank Shibao Feng for early contributions to the analyses and Lee Bennett and Yun Wu of RTI-Health Solutions for statistical programming. Support for third-party writing assistance for this manuscript was provided by Genentech, Inc. This work was sponsored by Genentech, Inc.
Conflict of interest
PAK and AMB have received consulting fees from Genentech. MUY has served on an advisory board for Roche/Genentech. HSR and PAK have received research funding for their institutions from Genentech. BY and OOA are full-time employees of Genentech and own Roche stock. DT serves in an uncompensated capacity on a Steering Committee for a registry study funded by Genentech/Roche. MM and DAY have no disclosures.
Ethical standards
These experiments comply with the current laws of the country in which they were performed.
References
- 1.Bickell NA, Wang JJ, Oluwole S, Schrag D, Godfrey H, Hiotis K, Mendez J, Guth AA. Missed opportunities: racial disparities in adjuvant breast cancer treatment. J Clin Oncol. 2006;24:1357–1362. doi: 10.1200/JCO.2005.04.5799. [DOI] [PubMed] [Google Scholar]
- 2.Carey LA, Perou CM, Livasy CA, Dressler LG, Cowan D, Conway K, Karaca G, Troester MA, Tse CK, Edmiston S, Deming SL, Geradts J, Cheang MC, Nielsen TO, Moorman PG, Earp HS, Millikan RC. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA. 2006;295:2492–2502. doi: 10.1001/jama.295.21.2492. [DOI] [PubMed] [Google Scholar]
- 3.Huo D, Ikpatt F, Khramtsov A, Dangou JM, Nanda R, Dignam J, Zhang B, Grushko T, Zhang C, Oluwasola O, Malaka D, Malami S, Odetunde A, Adeoye AO, Iyare F, Falusi A, Perou CM, Olopade OI. Population differences in breast cancer: survey in indigenous African women reveals over-representation of triple-negative breast cancer. J Clin Oncol. 2009;27:4515–4521. doi: 10.1200/JCO.2008.19.6873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stead LA, Lash TL, Sobieraj JE, Chi DD, Westrup JL, Charlot M, Blanchard RA, Lee JC, King TC, Rosenberg CL. Triple-negative breast cancers are increased in black women regardless of age or body mass index. Breast Cancer Res. 2009;11:R18. doi: 10.1186/bcr2242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tian N, Goovaerts P, Zhan FB, Wilson JG. Identification of racial disparities in breast cancer mortality: does scale matter? Int J Health Geogr. 2010;9:35. doi: 10.1186/1476-072X-9-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Stark A, Kapke A, Schultz D, Brown R, Linden M, Raju U. Advanced stages and poorly differentiated grade are associated with an increased risk of HER2/neu positive breast carcinoma only in white women: findings from a prospective cohort study of African-American and white-American women. Breast Cancer Res Treat. 2008;107:405–414. doi: 10.1007/s10549-007-9560-5. [DOI] [PubMed] [Google Scholar]
- 7.Newman LA. Breast cancer in African-American women. Oncologist. 2005;10:1–14. doi: 10.1634/theoncologist.10-1-1. [DOI] [PubMed] [Google Scholar]
- 8.Kwan ML, Kushi LH, Weltzien E, Maring B, Kutner SE, Fulton RS, Lee MM, Ambrosone CB, Caan BJ. Epidemiology of breast cancer subtypes in two prospective cohort studies of breast cancer survivors. Breast Cancer Res. 2009;11:R31. doi: 10.1186/bcr2261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Olopade OI, Fackenthal JD, Dunston G, Tainsky MA, Collins F, Whitfield-Broome C. Breast cancer genetics in African Americans. Cancer. 2003;97:236–245. doi: 10.1002/cncr.11019. [DOI] [PubMed] [Google Scholar]
- 10.Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science. 1987;235:177–182. doi: 10.1126/science.3798106. [DOI] [PubMed] [Google Scholar]
- 11.Kallioniemi O-P, Holli K, Visakorpi V, Koivula T, Helin HH, Isola JJ. Association of C-erbB-2 protein over-expression with high rate of cell proliferation, increased risk of visceral metastasis and poor long-term survival in breast cancer. Int J Cancer. 1991;49:650–655. doi: 10.1002/ijc.2910490504. [DOI] [PubMed] [Google Scholar]
- 12.Estevez L, Seidman A. HER2-positive breast cancer: incidence, prognosis, and treatment options. Am J Cancer. 2003;2:169–179. doi: 10.2165/00024669-200302030-00002. [DOI] [Google Scholar]
- 13.Brufsky AM, Mayer M, Rugo HS, Kaufman PA, Tan-Chiu E, Tripathy D, Tudor IC, Wang LI, Brammer MG, Shing M, Yood MU, Yardley DA. Central nervous system metastases in patients with HER2-positive metastatic breast cancer: incidence, treatment, and survival in patients from registHER. Clin Cancer Res. 2011;17:4834–4843. doi: 10.1158/1078-0432.CCR-10-2962. [DOI] [PubMed] [Google Scholar]
- 14.National Cancer Institute (2006) Common terminology criteria for adverse events v3.0 (CTCAE). http://ctep.cancer.gov/protocolDevelopment/electronic_applications/docs/ctcaev3.pdf. Accessed 28 March 2013
- 15.Vona-Davis L, Rose DP. Type 2 diabetes and obesity metabolic interactions: common factors for breast cancer risk and novel approaches to prevention and therapy. Curr Diabetes Rev. 2012;8:116–130. doi: 10.2174/157339912799424519. [DOI] [PubMed] [Google Scholar]
- 16.Jolly S, Vittinghoff E, Chattopadhyay A, Bibbins-Domingo K. Higher cardiovascular disease prevalence and mortality among younger blacks compared to whites. Am J Med. 2010;123:811–818. doi: 10.1016/j.amjmed.2010.04.020. [DOI] [PubMed] [Google Scholar]
- 17.Banegas MP, Li C. Breast cancer characteristics and outcomes among Hispanic black and Hispanic white women. Breast Cancer Res Treat. 2012;134:1297–1304. doi: 10.1007/s10549-012-2142-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Amend K, Hicks D, Ambrosone CB. Breast cancer in African-American women: differences in tumor biology from European-American women. Cancer Res. 2006;66:8327–8330. doi: 10.1158/0008-5472.CAN-06-1927. [DOI] [PubMed] [Google Scholar]
- 19.Joslyn SA. Hormone receptors in breast cancer: racial differences in distribution and survival. Breast Cancer Res Treat. 2002;73:45–59. doi: 10.1023/A:1015220420400. [DOI] [PubMed] [Google Scholar]
- 20.Ooi SL, Martinez ME, Li CI. Disparities in breast cancer characteristics and outcomes by race/ethnicity. Breast Cancer Res Treat. 2011;127:729–738. doi: 10.1007/s10549-010-1191-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sparano JA, Wang M, Zhao F, Stearns V, Martino S, Ligibel JA, Perez EA, Saphner T, Wolff AC, Sledge GW, Jr, Wood WC, Davidson NE. Race and hormone receptor-positive breast cancer outcomes in a randomized chemotherapy trial. J Natl Cancer Inst. 2012;104:406–414. doi: 10.1093/jnci/djr543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Anders CK, Deal AM, Miller CR, Khorram C, Meng H, Burrows E, Livasy C, Fritchie K, Ewend MG, Perou CM, Carey LA. The prognostic contribution of clinical breast cancer subtype, age, and race among patients with breast cancer brain metastases. Cancer. 2011;117:1602–1611. doi: 10.1002/cncr.25746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ihemelandu CU, Leffall LD, Jr, Dewitty RL, Naab TJ, Mezghebe HM, Makambi KH, Adams-Campbell L, Frederick WA. Molecular breast cancer subtypes in premenopausal and postmenopausal African-American women: age-specific prevalence and survival. J Surg Res. 2007;143:109–118. doi: 10.1016/j.jss.2007.03.085. [DOI] [PubMed] [Google Scholar]
- 24.Lund MJ, Trivers KF, Porter PL, Coates RJ, Leyland-Jones B, Brawley OW, Flagg EW, O’Regan RM, Gabram SG, Eley JW. Race and triple negative threats to breast cancer survival: a population-based study in Atlanta, GA. Breast Cancer Res Treat. 2009;113:357–370. doi: 10.1007/s10549-008-9926-3. [DOI] [PubMed] [Google Scholar]
- 25.Fackenthal JD, Zhang J, Zhang B, Zheng Y, Hagos F, Burrill DR, Niu Q, Huo D, Sveen WE, Ogundiran T, Adebamowo C, Odetunde A, Falusi AG, Olopade OI. High prevalence of BRCA1 and BRCA2 mutations in unselected Nigerian breast cancer patients. Int J Cancer. 2012;131:1114–1123. doi: 10.1002/ijc.27326. [DOI] [PubMed] [Google Scholar]
- 26.Jones BA, Kasl SV, Howe CL, Lachman M, Dubrow R, Curnen MM, Soler-Vila H, Beeghly A, Duan F, Owens P. African-American/White differences in breast carcinoma: p53 alterations and other tumor characteristics. Cancer. 2004;101:1293–1301. doi: 10.1002/cncr.20500. [DOI] [PubMed] [Google Scholar]
- 27.Wu Y, Mohamed H, Chillar R, Ali I, Clayton S, Slamon D, Vadgama JV. Clinical significance of Akt and HER2/neu overexpression in African-American and Latina women with breast cancer. Breast Cancer Res. 2008;10:R3. doi: 10.1186/bcr1844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tan-Chiu E, Yothers G, Romond E, Geyer CE, Jr, Ewer M, Keefe D, Shannon RP, Swain SM, Brown A, Fehrenbacher L, Vogel VG, Seay TE, Rastogi P, Mamounas EP, Wolmark N, Bryant J. Assessment of cardiac dysfunction in a randomized trial comparing doxorubicin and cyclophosphamide followed by paclitaxel, with or without trastuzumab as adjuvant therapy in node-positive, human epidermal growth factor receptor 2-overexpressing breast cancer: NSABP B-31. J Clin Oncol. 2005;23:7811–7819. doi: 10.1200/JCO.2005.02.4091. [DOI] [PubMed] [Google Scholar]
- 29.Du XL, Lin CC, Johnson NJ, Altekruse S. Effects of individual-level socioeconomic factors on racial disparities in cancer treatment and survival: findings from the National Longitudinal Mortality Study, 1979–2003. Cancer. 2011;117:3242–3251. doi: 10.1002/cncr.25854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Abidoye O, Ferguson MK, Salgia F. Lung carcinoma in African Americans. Nature. 2007;4:118–129. doi: 10.1038/ncponc0718. [DOI] [PubMed] [Google Scholar]