Abstract
OBJECTIVES
Primary sarcomas of the mediastinum are relatively rare. This article reviews the surgical outcomes of 21 cases diagnosed with localized mediastinal sarcomas receiving multidisciplinary treatment modalities in Sichuan province, China, from January 1996 to January 2011.
METHODS
Twenty-one cases of histologically diagnosed primary mediastinal sarcoma undergoing surgical treatment were reviewed retrospectively. Disease-free survival (DFS) and overall survival (OS) were statistically analysed. All the patients presented with localized tumours consisting of 5 females and 16 males with a median age of 41.0 years (range: 9.0–68.0 years). Among all cases, 17 (81.0%) had an Eastern Cooperative Oncology Group performance status score of ≤1 at diagnosis. Eight (38.1%) underwent macroscopically complete resection (R0–R1) and 13 (61.9%) had incomplete resection (R2). Ten (47.6%) received postoperative radiotherapy and 7 (33.3%) postoperative chemotherapy.
RESULTS
The median DFS was 17 months (range: 0.4–79.8 months) and the median OS was 27.2 months (range: 0.4–79.8 months). Patients receiving complete resection showed significantly improved DFS (P = 0.031) and OS (P = 0.035) compared with those with incomplete resection. Neither postoperative radiotherapy nor chemotherapy significantly improved DFS (P = 0.770, P = 0.756) or OS (P = 0.905, P = 0.738). However, 7 patients (R2) and 2 (R0–R1 and grade 3) had improved local control with a local recurrence-free survival of 28.9 months (range: 7.6–73.2 months).
CONCLUSIONS
Complete resection should be preferentially attempted compared with incomplete resection and postoperative radiotherapy might yield good local control.
Keywords: Mediastinum, Sarcoma, Surgery, Radiotherapy, Chemotherapy
INTRODUCTION
Soft-tissue sarcoma is a type of malignant neoplasm derived from mesenchymal origin that accounts for ∼1% of all cancers [1]. It arises anywhere in the body but commonly occurs in the extremities or within the abdomen [2]. Mediastinum is a rare primary site for the occurrence of a soft-tissue sarcoma, which comprises <10% of primary mediastinal tumours and <1% of all soft-tissue sarcomas [3, 4].
Due to the rare incidence and large heterogeneity of natural history, primary mediastinal sarcoma has been described rarely in in previous publications, except for few individual case reports and series [5–11]. In addition, clinical experience of treatment strategies was mostly obtained from sarcomas involving other anatomic locations [11, 12]. Surgery is the only curative treatment for soft-tissue sarcomas of localized disease at diagnosis [5], but anatomic constraints and involvement of surrounding vital structures and visceral organs of the deep-seated sarcomas make complete resection of all gross diseases difficult. Moreover, deep location is found to be an independent prognostic factor for the advent of metastasis [13]. Both local recurrence and distant metastasis are commonly seen clinically [14]. The role of the combination of radiotherapy and chemotherapy as part of multidisciplinary treatments is still inconclusive [13, 15–19].
In an attempt to gain more insights into primary mediastinal sarcoma, especially from those cases undergoing an operation at presentation, we have reviewed previous studies of primary mediastinal sarcoma and analysed the clinical and pathological features and multidisciplinary treatment outcomes of 21 consecutive cases treated in Sichuan province over a 15-year time span.
MATERIALS AND METHODS
The authors retrospectively searched the information databases in West China Hospital, Sichuan Province Cancer Hospital and Sichuan Academy of Medical Sciences & Sichuan Provincial People's Hospital and identified 26 consecutive cases of mediastinal sarcoma from January 1996 to January 2011 after obtaining approvals from the Institutional Review Board of these four hospitals. The inclusion criteria: none of the patients had a history of previous soft-tissue sarcomas affecting other sites or any other malignancies documented before the diagnosis of mediastinal sarcomas. Two patients with a final diagnosis of sarcomatoid carcinoma and 3 who failed to undergo surgery had been excluded from further analysis. Medical records of the remaining 21 patients including demographic characteristics, pathological and clinical features, multidisciplinary treatments and survival outcomes were reviewed and recorded. All patients alive were followed up until August 2012.
The histological typing and subtyping were conducted based on the World Health Organization Classification of Tumors [20]. Histological grade was assessed according to the updated version of the French Federation of Cancer Centers grading system [21]. Residual tumours after surgery were defined according to the International Union Against Cancer classifications: R0, no residual tumour; R1, microscopic residual tumour; R2, macroscopic residual tumour. The assessment of the microscopic margins on a retrospective review was unreliable due to the anatomic location of mediastinal sarcoma. Therefore, surgical resection margins were only classified as macroscopically complete (R0 or R1) or incomplete resection (R2) [12].
Statistical analysis
Overall survival (OS) was defined as the period from the date of initial diagnosis until death or the last follow-up. Disease-free survival (DFS) was measured from the date of operation until the first event of disease progression or death occurred. Survival curves were analysed by the Kaplan–Meier method and comparisons of survival by means of log-rank test. A two-sided P-value of <0.05 indicated statistical significance.
RESULTS
Clinicopathological characteristics
Clinicopathological characteristics of 21 cases diagnosed with mediastinal sarcoma in this study were summarized in Table 1. Of the 21 patients, 16 were male and 5 were female (ratio 3.2 to 1). The median age of all patients was 41.0 years (range 9.0–68.0 years). Ten patients (47.6%) had a habit or a history of smoking. Seventeen patients (81.0%) had an Eastern Cooperative Oncology Group (ECOG) performance status score of ≤1 at diagnosis. The patients had varying pathological subtypes. The tumours of 13 cases (61.9%) were noted to be located in the posterior mediastinal compartment. Lesion sizes ranged from 5.7 to 16.0 cm (median size: 11.38 cm). Pathological grading revealed that 3 patients (14.3%) had grade 1 tumours, 10 (47.6%) grade 2 and 8 (38.1%) grade 3.
Table 1:
Clinical characteristics (n = 21)
| Variables | Number (n) | Percentage (%) |
|---|---|---|
| Gender | ||
| Male | 16 | 76.2 |
| Female | 5 | 23.8 |
| Age (year) | ||
| Median | 41.0 (9–68) | |
| >40 | 12 | 57.1 |
| ≤40 | 9 | 42.9 |
| Smoking history | ||
| Never smokers | 11 | 52.4 |
| Current or former smokers | 10 | 47.6 |
| Eastern Cooperative Oncology Group | ||
| 0–1 | 17 | 81.0 |
| ≥2 | 4 | 19.0 |
| Histopathological type | ||
| Liposarcoma | 8 | 38.1 |
| Ewing's sarcoma | 3 | 14.3 |
| Chondrosarcoma | 3 | 14.3 |
| Leiomyosarcoma | 3 | 14.3 |
| Rhabdomyosarcoma | 2 | 9.5 |
| Angiosarcoma | 1 | 4.8 |
| Malignant fibrous histiocytoma | 1 | 4.8 |
| Primary tumour site | ||
| Superior | 5 | 23.8 |
| Anterior | 2 | 9.5 |
| Middle | 1 | 4.8 |
| Posterior | 13 | 61.9 |
| Tumour diameter (cm) | ||
| Median | 11.4 (5.7–16.0) | |
| T ≤ 10 cm | 10 | 47.6 |
| T ≥ 10 cm | 11 | 52.4 |
| Tumour grade | ||
| 1 | 3 | 14.3 |
| 2 | 10 | 47.6 |
| 3 | 8 | 38.1 |
Sixteen patients had a documented period of symptoms ranging from 0 to 6 months (median; 3.5 months). Presenting complaints included chest pain in 8 patients (33.3%), back pain in 7 (29.1%), dyspnoea in 4 (16.6%), cough, arm swelling and weight loss in 2 (8.3%), respectively (Table 2). A palpable mass was occasionally found on the surface of the sternum and supraclavicular fossa in 2 patients. Three patients were diagnosed by a chest radiograph during a routine check-up.
Table 2:
Initial symptoms in 21 cases of mediastinal sarcomas
| Complaints | Number (n) | Percentage (%) |
|---|---|---|
| Chest pain | 8 | 33.3 |
| Cough | 2 | 8.3 |
| Dyspnoea | 4 | 16.6 |
| Weight loss | 2 | 8.3 |
| Back pain | 7 | 29.1 |
| Arm swelling | 2 | 8.3 |
Treatment approaches
Surgery as a primary treatment approach was performed in all the 21 patients. Of them, 12 patients were simply treated by wide local excision, 1 with lung gross involvement underwent pulmonary segmental resection and 8 underwent gross resection of residual tumours to reduce the tumour burden and compression of adjacent organs. The adjacent organs were resected in the mediastinum: brachiocephalic veins in 3 cases, superior vena cava in 2, oesophagus in 2, pulmonary lobe in 1 and unilateral phrenic nerve in 1. In total, 8 patients received complete resection and 13 underwent incomplete resection. A 9-year old patient received repeated surgical resection after local recurrence-free survival for 8 months.
Postoperative radiotherapy targeting the mediastinum was employed in 10 patients (47.6%): two-dimensional radiotherapy in 3 (14.3%), three-dimensional conformal radiotherapy in 5 (23.8%) and intensity-modulated radiotherapy in the remaining 2 (9.5%), mainly based on the tumour grade and margin status (dosage range: 50–66 Gray in 25–33 fractions). Eight patients were R2 and the remaining 2 were R0–R1 and grade 3. One patient with spine involvement in later period received two-dimensional radiotherapy, with 10 Gray in two fractions.
Postoperative chemotherapy in combination with local treatment was delivered in 7 patients (33.3%) based on the tumour grade and margin status. Among them, 6 were R2 and 1 R0–R1 and grade 3. Two patients were treated with ifosfamide plus VP-16 and 2 with doxorubicin plus ifosfamide, 4–8 cycles in total. One patient with leiomyosarcoma was treated with six cycles of ifosfamide, doxorubicin and dacarbazine. The 9-year old patient with Ewing's sarcoma mentioned above received 10 cycles of VIDE (vincristine, ifosfamide, doxorubicin and etoposide) alternating with IVA (ifosfamide, vincristine and actinomycin) after gross residual resection, and subsequently underwent re-resection rather than radiotherapy due to local progression. One patient with angiosarcoma received one cycle of ifosfamide plus doxorubicin but subsequently discontinued treatment for severe nausea and vomiting.
Survival outcomes
The median follow-up period of the entire group was 20.7 months. During the follow-up, 10 patients had experienced disease progression: 4 with local progression, 4 with distant progression and 2 with both local and distant progression. Nine patients died of mediastinal sarcomas, 2 of surgical complications and 2 of unrelated diseases. One patient with pleomorphic liposarcoma, found to be involved with the right atrium and the superior vena cava during surgery, died of sudden arrhythmia on postoperative Day 11. Another one with unspecified sarcoma, found to have left atrium involvement intraoperatively, had acute cerebral infarction 1 day postoperation and died 1.1 months later.
At the end of follow-up, the mean and median DFS were 32.7 and 17.0 months (range: 0.4–79.8 months), respectively, and the mean and median OS were 35.0 and 27.2 months (range: 0.4–79.8 months), respectively (Fig. 1). Histological subtypes of well-differentiated liposarcoma and angiosarcoma were associated with the longest survival time. ECOG performance status was correlated with significantly improved OS (P = 0.001) and DFS (P = 0.001). Gender, age, smoking history, primary tumour site, tumour grade and size were not associated with significant differences in survival.
Figure 1:
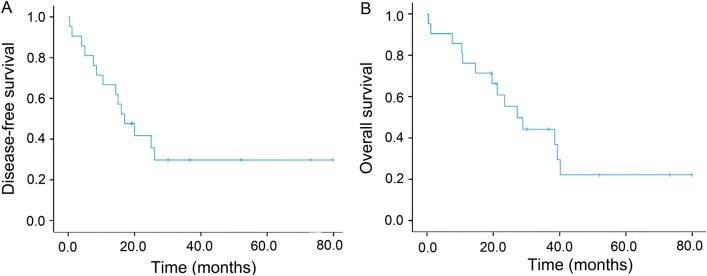
(A) Kaplan–Meier curves of disease-free survival for all patients (n = 21). (B) Kaplan–Meier curves of overall survival for all patients (n = 21).
All patients underwent surgery: 8 of them had complete resection of their sarcomas while 13 had incomplete resection. Four patients died in the complete resection group compared with 10 in the incomplete resection group at the end of follow-up. Complete resection was significantly associated with improved DFS (P = 0.031) and OS (P = 0.035) (Fig. 2). For the limited follow-up period, it cannot reach the median survival time for the complete resection group. The mean OS and DFS were 49.9 (range: 19.6–79.8 months) and 50.1 months (range: 14.3–79.8 months) in the complete resection group (n = 8) and 24.1 (range: 0.4–73.2 months) and 19.7 months (range: 0.4–73.2 months) in the incomplete resection group (n = 13), respectively.
Figure 2:
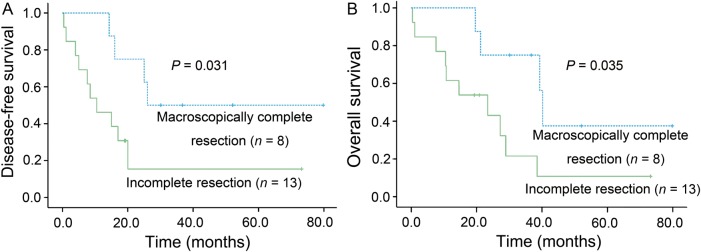
(A) Kaplan–Meier curves of disease-free survival according to response to surgery (P = 0.031). (B) Kaplan–Meier curves of overall survival according to response to surgery (P = 0.035).
Postoperative radiotherapy did not significantly affect DFS (P = 0.770) or OS (P = 0.905). The median DFS and OS in the 10 patients who underwent radiotherapy were 17.0 months (range: 4.0–73.2 months) and 28.9 months (range: 7.6–73.2 months), compared with 15.0 months (range: 0.4–79.8 months) and 27.2 months (range: 0.4–79.8 months) in the 11 counterparts who did not, respectively. However, of the 10 receiving postoperative radiotherapy, good local control was obtained in 7 patients with R2 and 2 with R0–R1 and grade 3, with a local recurrence-free survival of 28.9 months (range: 7.6–73.2 months) (Fig. 3). In the postoperative radiotherapy group, 1 patient died of local recurrence and 5 of distant metastasis. Four patients completed the final follow-up with no evidence of the disease.
Figure 3:
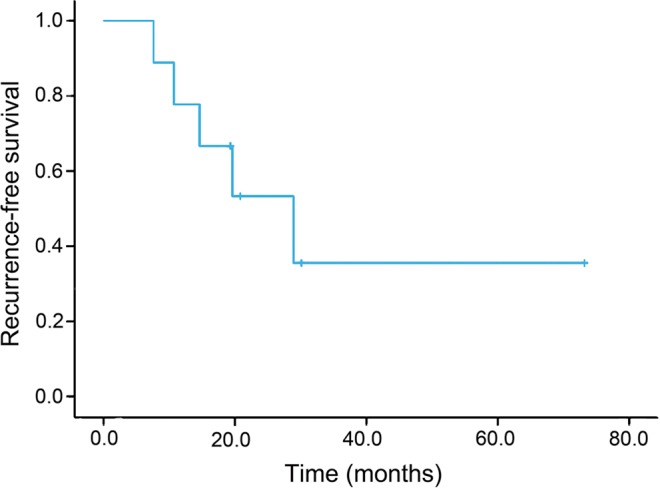
Kaplan–Meier curves of local recurrence-free survival of 9 patients undergoing postoperative radiotherapy.
Postoperative chemotherapy failed to significantly improve DFS (P = 0.756) or OS (P = 0.738). The median DFS and OS in patients who underwent chemotherapy were 20.0 months (range: 4.0–73.2 months) and 38.5 months (range: 7.6–73.2 months) compared with 16.0 months (range: 0.4–79.8 months) and 27.2 months (range: 0.4–79.8 months) in those who did not, respectively.
DISCUSSION
Primary sarcomas of the mediastinum are not commonly encountered, most of which are sporadic with no clear reasons [2]. Distinct from extremity and superficial trunk locations, sarcomas originating from the mediastinum are often asymptomatic until they reach a considerable size. The management of mediastinal sarcomas is extremely challenging due to deep location and invasion of adjacent vital structures. Despite multidisciplinary modalities of treatment, the long-term prognosis of mediastinal sarcomas is still poor [5, 6]. Due to the rarity and heterogeneity of mediastinal sarcomas, available data in the literature are relatively limited [5–8]. A US series including 47 patients with mediastinal sarcomas from 1940 to 1991 in New York had the largest number of cases [5], but the participants had a history of radiation exposure and different histological types predominated by malignant peripheral nerve tumour.
Herein, we retrospectively reviewed the clinicopathological features, treatment characteristics and outcomes in 21 patients with primary mediastinal sarcomas who underwent surgery in Sichuan, China, over 15 years, a more contemporary study available. Most of the patients in our cohort had non-specific signs and symptoms. The median age was 41.0 years, which was comparable with 35.0–58.0 years in previous researches [5–8], whereas a much better ECOG performance status was found in our series compared with that reported in another publication [6]. Similar to other publications, there were no significant differences in survival regarding age, gender and smoking history [5]. ECOG performance status was significantly associated with DFS and OS. Tumour grade, size and locations were not associated with significant differences in survival, which were consistent with one previous study [6] but contradictory to others [5, 13].
Soft-tissue sarcomas are preferentially treated by experienced multidisciplinary teams to achieve lower local recurrence rate and higher survival rate. There is general consensus that the overwhelming factor determining survival was the capacity of completely resecting the tumours [6, 7, 11]. In our series, all patients underwent surgery, and 8 had complete resection (38.1%) with a favourable survival outcome, which was comparable with another study [6]. The median OS in two previously published observations were 13 and 18 months [5, 6], but a much longer median OS of 27.2 months was observed in the present series, probably due to the relatively higher ECOG performance scores and better surgical and multidisciplinary treatments. Although the decisive role of surgical resection was relatively clear, it remains extremely challenging to perform wide local excision with negative margins for the distinctive anatomic location similar to retroperitoneum [11], which may require multidisciplinary management. Two case reports suggested that induction chemotherapy followed by surgery might result in complete resection [11, 22], but no patient in our study underwent this strategy, which deserves further investigations.
The role of postoperative radiotherapy in the treatment of mediastinal sarcomas has been seldom studied. However, local recurrence was reported in 64% of cases despite receiving complete resection [5]. In our study, postoperative radiotherapy was mainly performed based on margin status and tumour grade by means of two-dimensional radiotherapy, three-dimensional conformal radiotherapy or intensity-modulated radiotherapy techniques. Although postoperative radiotherapy did not significantly improve survival, it yielded a favourable local control in 7 patients with incomplete resection in our series. Paquette et al. [6] reported similar findings, demonstrating the value of postoperative radiotherapy in patients with incomplete resection. However, patients with complete resection alone had better survival than those undergoing incomplete resection combined with postoperative radiotherapy. Therefore, the effect of postoperative radiotherapy on survival in patients with complete resection remains to be elucidated.
The role of chemotherapy in the treatment of soft-tissue sarcomas, especially postoperative therapy, is not finally settled due to the rarity of this disease. Experiences were mainly obtained based on prospective trials or retrospective studies from sarcoma of the extremities or retroperitoneum [18, 23]. Italiano et al. [17] reported that neither neo-adjuvant nor adjuvant chemotherapy had significant effect on survival. On the contrary, a meta-analysis showed evidence that adjuvant chemotherapy significantly improved the overall recurrence-free survival and noted a trend towards improved OS [24]. Moreover, Eriksson recommended that the combination of doxorubicin and ifosfamide as a standard therapy increased the possibility of surgery with curative intent, and new drugs such as taxanes, gemcitabine and trabectedin also showed promising results [19]. In our study, the patients in the chemotherapy group yielded longer DFS and OS with no statistical significance, probably due to the heterogeneity and limited number of mediastinal cases treated with chemotherapy. After aggressive local treatment, distant metastasis became the major factor responsible for death, potentially suggesting the vital role of chemotherapy in the multidisciplinary treatment modalities.
Because of the rarity of this disease, the number of our series is small. Due to the heterogeneity and small numbers, multivariate analyses cannot be performed and the conclusions of this small subgroup must be interpreted with caution.
To sum up, the present study provides several important implications for the treatment of primary mediastinal sarcomas. First and foremost, complete resection should be attempted whenever possible. Furthermore, radiotherapy might contribute to good local control especially those with incomplete resection or high-grade carcinoma. However, the role of chemotherapy is still elusive. Given the rarity and complexity of primary mediastinal sarcomas, treatment options must be optimized by a multidisciplinary forum on a case-by-case basis depending on the clinical and histological characteristics at the time of diagnosis.
Conflict of interest: none declared.
REFERENCES
- 1.Jemal A, Tiwari R, Murray T, Ghafoor A, Samuels A, Ward E, et al. Cancer statistics, 2004. CA Cancer J Clin. 2004;54:8–29. doi: 10.3322/canjclin.54.1.8. [DOI] [PubMed] [Google Scholar]
- 2.Clark MA, Fisher C, Judson I, Thomas JM. Soft-tissue sarcomas in adults. N Engl J Med. 2005;353:701–11. doi: 10.1056/NEJMra041866. [DOI] [PubMed] [Google Scholar]
- 3.Macchiarini P, Ostertag H. Uncommon primary mediastinal tumours. Lancet Oncol. 2004;5:107–18. doi: 10.1016/S1470-2045(04)01385-3. [DOI] [PubMed] [Google Scholar]
- 4.Toro JR, Travis LB, Wu HJ, Zhu K, Fletcher CD, Devesa SS. Incidence patterns of soft tissue sarcomas, regardless of primary site, in the surveillance, epidemiology and end results program, 1978–2001: an analysis of 26,758 cases. Int J Cancer. 2006;119:2922–30. doi: 10.1002/ijc.22239. [DOI] [PubMed] [Google Scholar]
- 5.Burt M, Ihde JK, Hajdu SI, Smith JW, Bains MS, Downey R, et al. Primary sarcomas of the mediastinum: results of therapy. J Thorac Cardiovasc Surg. 1998;115:671–80. doi: 10.1016/S0022-5223(98)70333-2. [DOI] [PubMed] [Google Scholar]
- 6.Paquette M, Truong PT, Hart J, Jones SO, Martens B, Christie JL, et al. Primary sarcoma of the mediastinum: a report of 16 cases referred to the British Columbia Cancer Agency. J Thorac Oncol. 2010;5:898–906. doi: 10.1097/jto.0b013e3181d8fe80. [DOI] [PubMed] [Google Scholar]
- 7.Hahn HP, Fletcher CDM. Primary mediastinal liposarcoma: clinicopathologic analysis of 24 cases. Am J Surg Pathol. 2007;31:1868–74. doi: 10.1097/PAS.0b013e318093f925. [DOI] [PubMed] [Google Scholar]
- 8.Suster S, Moran CA. Primary synovial sarcomas of the mediastinum: a clinicopathologic, immunohistochemical, and ultrastructural study of 15 cases. Am J Surg Pathol. 2005;29:569–78. doi: 10.1097/01.pas.0000157934.50936.3e. [DOI] [PubMed] [Google Scholar]
- 9.Eroglu A, Kürkçüoglu C, Karaoglanoglu N, Gürsan N. Primary leiomyosarcoma of the anterior mediastinum. Eur J Cardiothorac Surg. 2002;21:943–5. doi: 10.1016/s1010-7940(02)00090-8. [DOI] [PubMed] [Google Scholar]
- 10.Marulli G, Rea F, Feltracco P, Calabresse F, Giacometti C, Rizzardi G, et al. Successful resection of a giant primary liposarcoma of the posterior mediastinum. J Thorac Oncol. 2007;2:453–5. doi: 10.1097/01.JTO.0000268681.10367.cf. [DOI] [PubMed] [Google Scholar]
- 11.Coindre JM, Terrier P, Bui NB, Bonichon F, Collin F, Le Doussal V, et al. Prognostic factors in adult patients with locally controlled soft tissue sarcoma. A study of 546 patients from the French Federation of Cancer Centers Sarcoma Group. J Clin Oncol. 1996;14:869–77. doi: 10.1200/JCO.1996.14.3.869. [DOI] [PubMed] [Google Scholar]
- 12.Gronchi A, Vullo SL, Fiore M, Mussi C, Stacchiotti S, Collini P, et al. Aggressive surgical policies in a retrospectively reviewed single-institution case series of retroperitoneal soft tissue sarcoma patients. J Clin Oncol. 2009;27:24–30. doi: 10.1200/JCO.2008.17.8871. [DOI] [PubMed] [Google Scholar]
- 13.Lewis JJ, Leung D, Heslin M, Woodruff JM, Brennan MF. Association of local recurrence with subsequent survival in extremity soft tissue sarcoma. J Clin Oncol. 1997;15:646–52. doi: 10.1200/JCO.1997.15.2.646. [DOI] [PubMed] [Google Scholar]
- 14.Zhou Z, McDade TP, Simons JP, Ng SC, Lambert LA, Whalen GF, et al. Surgery and radiotherapy for retroperitoneal and abdominal sarcoma: both necessary and sufficient. Arch Surg. 2010;145:426–31. doi: 10.1001/archsurg.2010.70. [DOI] [PubMed] [Google Scholar]
- 15.Gronchi A, Casali P, Mariani L, Miceli R, Fiore M, Lo Vullo S, et al. Status of surgical margins and prognosis in adult soft tissue sarcomas of the extremities: a series of patients treated at a single institution. J Clin Oncol. 2005;23:96–104. doi: 10.1200/JCO.2005.04.160. [DOI] [PubMed] [Google Scholar]
- 16.Blay JY, Le Cesne A. Adjuvant chemotherapy in localized soft tissue sarcomas: still not proven. Oncologist. 2009;14:1013–20. doi: 10.1634/theoncologist.2009-0126. [DOI] [PubMed] [Google Scholar]
- 17.Italiano A, Penel N, Robin YM, Bui B, Le Cesne A, Piperno-Neumann S, et al. Neo/adjuvant chemotherapy does not improve outcome in resected primary synovial sarcoma: a study of the French Sarcoma Group. Ann Oncol. 2009;20:425–30. doi: 10.1093/annonc/mdn678. [DOI] [PubMed] [Google Scholar]
- 18.Eriksson M. Histology-driven chemotherapy of soft-tissue sarcoma. Ann Oncol. 2010;21:vii270–6. doi: 10.1093/annonc/mdq285. [DOI] [PubMed] [Google Scholar]
- 19.Fletcher C, Unni K, Mertens F World Health Organization Classification of Tumors. Pathology and Genetics of Tumours of Soft Tissue and Bone. Lyon: IARC Press; 2002. [Google Scholar]
- 20.Guillou L, Coindre JM, Bonichon F, Nguyen BB, Terrier P, Collin F, et al. Comparative study of the National Cancer Institute and French Federation of Cancer Centers Sarcoma Group grading systems in a population of 410 adult patients with soft tissue sarcoma. J Clin Oncol. 1997;15:350–62. doi: 10.1200/JCO.1997.15.1.350. [DOI] [PubMed] [Google Scholar]
- 21.Berry MF, Sporn TA, Moore JO, D'Amico TA. Giant thoracic liposarcoma treated with induction chemotherapy followed by surgical resection. J Thorac Oncol. 2009;4:768–9. doi: 10.1097/JTO.0b013e31819e77ff. [DOI] [PubMed] [Google Scholar]
- 22.Trovik C, Bauer H, Alvegård T, Anderson H, Blomqvist C, Berlin O, et al. Surgical margins, local recurrence and metastasis in soft tissue sarcomas: 559 surgically-treated patients from the Scandinavian Sarcoma Group Register. Eur J Cancer. 2000;36:710–6. doi: 10.1016/s0959-8049(99)00287-7. [DOI] [PubMed] [Google Scholar]
- 23.Pervaiz N, Colterjohn N, Farrokhyar F, Tozer R, Figueredo A, Ghert M. A systematic meta-analysis of randomized controlled trials of adjuvant chemotherapy for localized resectable soft tissue sarcoma. Cancer. 2008;113:573–81. doi: 10.1002/cncr.23592. [DOI] [PubMed] [Google Scholar]
- 24.Adjuvant chemotherapy for localised resectable soft-tissue sarcoma of adults: meta-analysis of individual data. Lancet. 1997;350:1647–54. [PubMed] [Google Scholar]


