Abstract
EMBO J 32: 2905–2919 ; DOI: 10.1038/emboj.2013.199; published online September 03 2013
Some B cells of the adaptive immune system secrete polyreactive immunoglobulin G (IgG) in the absence of immunization or infection. Owing to its limited affinity and specificity, this natural IgG is thought to play a modest protective role. In this issue, a report reveals that natural IgG binds to microbes following their opsonization by ficolin and mannan-binding lectin (MBL), two carbohydrate receptors of the innate immune system. The interaction of natural IgG with ficolins and MBL protects against pathogenic bacteria via a complement-independent mechanism that involves IgG receptor FcγRI expressing macrophages. Thus, natural IgG enhances immunity by adopting a defensive strategy that crossovers the conventional boundaries between innate and adaptive microbial recognition systems.
The adaptive immune system generates protective somatically recombined antibodies through a T cell-dependent (TD) pathway that involves follicular B cells. After recognizing antigen through the B-cell receptor (BCR), follicular B cells establish a cognate interaction with CD4+ T follicular helper (TFH) cells and thereafter either rapidly differentiate into short-lived IgM-secreting plasmablasts or enter the germinal centre (GC) of lymphoid follicles to complete class switch recombination (CSR) and somatic hypermutation (SHM) (Victora and Nussenzweig, 2012). CSR from IgM to IgG, IgA and IgE generates antibodies with novel effector functions, whereas SHM provides the structural correlate for the induction of affinity maturation (Victora and Nussenzweig, 2012). Eventually, this canonical TD pathway generates long-lived bone marrow plasma cells and circulating memory B cells that produce protective class-switched antibodies capable to recognize specific antigens with high affinity (Victora and Nussenzweig, 2012).
In addition to post-immune monoreactive antibodies, B cells produce pre-immune polyreactive antibodies in the absence of conventional antigenic stimulation (Ehrenstein and Notley, 2010). These natural antibodies form a vast and stable repertoire that recognizes both non-protein and protein antigens with low affinity (Ehrenstein and Notley, 2010). Natural antibodies usually emerge from a T cell-independent (TI) pathway that involves innate-like B-1 and marginal zone (MZ) B cells. These are extrafollicular B-cell subsets that rapidly differentiate into short-lived antibody-secreting plasmablasts after detecting highly conserved microbial and autologus antigens through polyreactive BCRs and nonspecific germline-encoded pattern recognition receptors (Pone et al, 2012; Cerutti et al, 2013).
The most studied natural antibody is IgM, a pentameric complement-activating molecule with high avidity but low affinity for antigen (Ehrenstein and Notley, 2010). In addition to promoting the initial clearance of intruding microbes, natural IgM regulates tissue homeostasis, immunological tolerance and tumour surveillance (Ochsenbein et al, 1999; Zhou et al, 2007; Ehrenstein and Notley, 2010). Besides secreting IgM, B-1 and MZ B cells produce IgG and IgA after receiving CSR-inducing signals from dendritic cells (DCs), macrophages and neutrophils of the innate immune system (Cohen and Norins, 1966; Cerutti et al, 2013). In humans, certain natural IgG and IgA are moderately mutated and show some specificity, which may reflect the ability of human MZ B cells to undergo SHM (Cerutti et al, 2013). Yet, natural IgG and IgA are generally perceived as functionally quiescent.
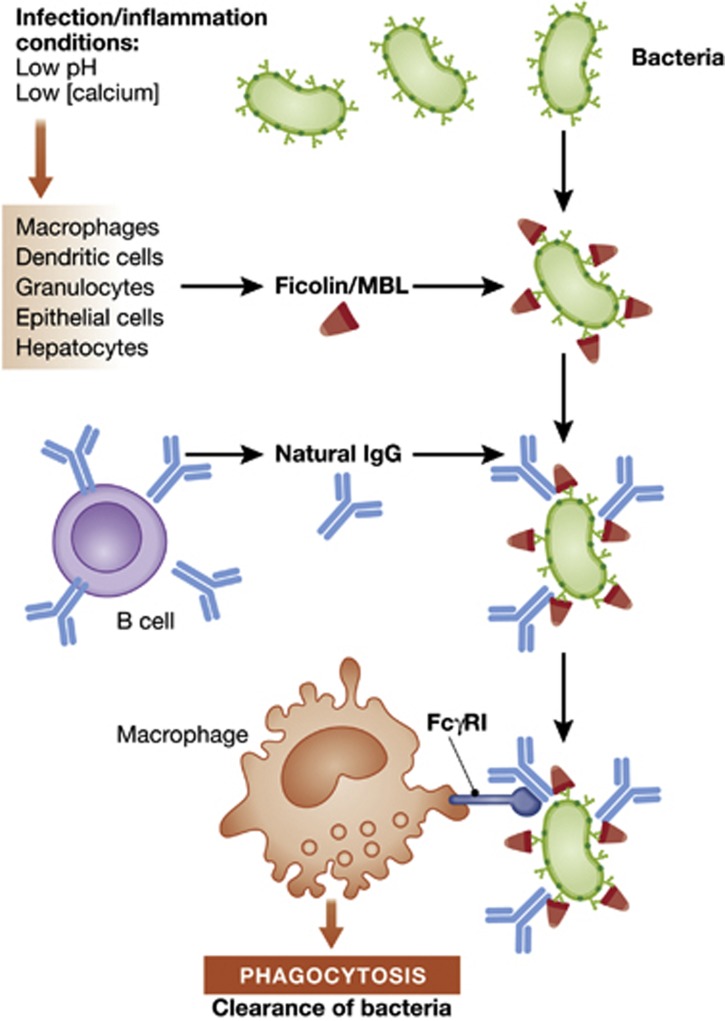
In this issue, Panda et al show that natural IgG bound to a broad spectrum of bacteria with high affinity by cooperating with ficolin and MBL (Panda et al, 2013), two ancestral soluble lectins of the innate immune system (Holmskov et al, 2003). This binding involved some degree of specificity, because it required the presence of ficolin or MBL on the microbial surface as well as lower pH and decreased calcium concentration in the extracellular environment as a result of infection or inflammation (see Figure 1).
Figure 1.
Ficolins and MBL are produced by hepatocytes and various cells of the innate immune system and opsonize bacteria after recognizing conserved carbohydrates. Low pH and calcium concentrations present under infection-inflammation conditions promote the interaction of ficolin or MBL with natural IgG on the surface of bacteria. The resulting immunocomplex is efficiently phagocytosed by macrophages through FcγR1 independently of the complement protein C3, leading to the clearance of bacteria.
Ficolins and MBL are soluble pattern recognition receptors that opsonize microbes after binding to glycoconjugates through distinct carbohydrate recognition domain (CRD) structures (Holmskov et al, 2003). While ficolins use a fibrinogen domain, MBL and other members of the collectin family use a C-type lectin domain attached to a collagen-like region (Holmskov et al, 2003). Similar to pentraxins, ficolins and MBL are released by innate effector cells and hepatocytes, and thus may have served as ancestral antibody-like molecules prior to the inception of the adaptive immune system (Holmskov et al, 2003; Bottazzi et al, 2010). Of note, MBL and the MBL-like complement protein C1q are recruited by natural IgM to mediate complement-dependent clearance of autologous apoptotic cells and microbes (Holmskov et al, 2003; Ehrenstein and Notley, 2010). Panda et al found that a similar lectin-dependent co-optation strategy enhances the protective properties of natural IgG (Panda et al, 2013).
By using bacteria and the bacterial glycan N-acetylglicosamine, Panda et al show that natural IgG isolated from human serum or T cell-deficient mice interacted with the fibrinogen domain of microbe-associated ficolins (Panda et al, 2013). The resulting immunocomplex was phagocytosed by macrophages via the IgG receptor FcγRI in a complement-independent manner (Panda et al, 2013). The additional involvement of MBL was demonstrated by experiments showing that natural IgG retained some bacteria-binding activity in the absence of ficolins (Panda et al, 2013).
Surface plasmon resonance provided some clues regarding the molecular requirements of the ficolin–IgG interaction (Panda et al, 2013), but the conformational changes required by ficolin to interact with natural IgG remain to be addressed. In particular, it is unclear what segment of the effector Fc domain of natural IgG binds to ficolins and whether Fc-associated glycans are involved in this binding. Specific glycans have been recently shown to mitigate the inflammatory properties of IgG emerging from TI responses (Hess et al, 2013) and this process could implicate ficolins and MBL. Moreover, it would be important to elucidate whether and how the antigen-binding Fab portion of natural IgG regulates its interaction with ficolins and MBL.
The in vivo protective role of natural IgG was elegantly demonstrated by showing that reconstitution of IgG-deficient mice lacking the CSR-enzyme activation-induced cytidine deaminase with natural IgG from T cell-insufficient animals enhanced resistance to pathogenic Pseudomonas aeruginosa (Panda et al, 2013). This protective effect was associated with reduced production of proinflammatory cytokines, occurred independently of the complement protein C3 and was impaired by peptides capable to inhibit the binding of natural IgG to ficolin (Panda et al, 2013). Additional in vivo studies will be needed to determine whether natural IgG exerts protective activity in mice lacking ficolin, MBL or FcγRI, and to ascertain whether these molecules also enhance the protective properties of canonical or natural IgG and IgA released by bone marrow plasma cells and mucosal plasma cells, respectively.
In conclusion, the findings by Panda et al show that natural IgG adopts ‘crossover’ defensive strategies that blur the conventional boundaries between the innate and adaptive immune systems. The sophisticated integration of somatically recombined and germline-encoded antigen recognition systems described in this new study shall stimulate immunologists to further explore the often underestimated protective virtues of our vast natural antibody repertoire. This effort may lead to the development of novel therapies against infections.
Footnotes
The authors declare that they have no conflict of interest.
References
- Bottazzi B, Doni A, Garlanda C, Mantovani A (2010) An integrated view of humoral innate immunity: pentraxins as a paradigm. Annu Rev Immunol 28: 157–183 [DOI] [PubMed] [Google Scholar]
- Cerutti A, Cols M, Puga I (2013) Marginal zone B cells: virtues of innate-like antibody-producing lymphocytes. Nat Rev Immunol 13: 118–132 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen IR, Norins LC (1966) Natural human antibodies to gram-negative bacteria: immunoglobulins G, A, and M. Science 152: 1257–1259 [DOI] [PubMed] [Google Scholar]
- Ehrenstein MR, Notley CA (2010) The importance of natural IgM: scavenger, protector and regulator. Nat Rev Immunol 10: 778–786 [DOI] [PubMed] [Google Scholar]
- Hess C, Winkler A, Lorenz AK, Holecska V, Blanchard V, Eiglmeier S, Schoen AL, Bitterling J, Stoehr AD, Petzold D, Schommartz T, Mertes MM, Schoen CT, Tiburzy B, Herrmann A, Köhl J, Manz RA, Madaio MP, Berger M, Wardemann H et al. (2013) T cell-independent B cell activation induces immunosuppressive sialylated IgG antibodies. J Clin Invest 123: 3788–3796 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holmskov U, Thiel S, Jensenius JC (2003) Collectins and ficolins: humoral lectins of the innate immune defense. Annu Rev Immunol 21: 547–578 [DOI] [PubMed] [Google Scholar]
- Ochsenbein AF, Fehr T, Lutz C, Suter M, Brombacher F, Hengartner H, Zinkernagel RM (1999) Control of early viral and bacterial distribution and disease by natural antibodies. Science 286: 2156–2159 [DOI] [PubMed] [Google Scholar]
- Panda S, Zhang J, Tan NS, Ho B, Ding JL (2013) Natural IgG antibodies provide innate protection against ficolin-opsonized bacteria. EMBO J 32: 2905–2919 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pone EJ, Zhang J, Mai T, White CA, Li G, Sakakura JK, Patel PJ, Al-Qahtani A, Zan H, Xu Z, Casali P (2012) BCR-signalling synergizes with TLR-signalling for induction of AID and immunoglobulin class-switching through the non-canonical NF-kappaB pathway. Nat Commun 3: 767–779 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Victora GD, Nussenzweig MC (2012) Germinal centers. Annu Rev Immunol 30: 429–457 [DOI] [PubMed] [Google Scholar]
- Zhou ZH, Zhang Y, Hu YF, Wahl LM, Cisar JO, Notkins AL (2007) The broad antibacterial activity of the natural antibody repertoire is due to polyreactive antibodies. Cell Host Microbe 1: 51–61 [DOI] [PMC free article] [PubMed] [Google Scholar]