Abstract
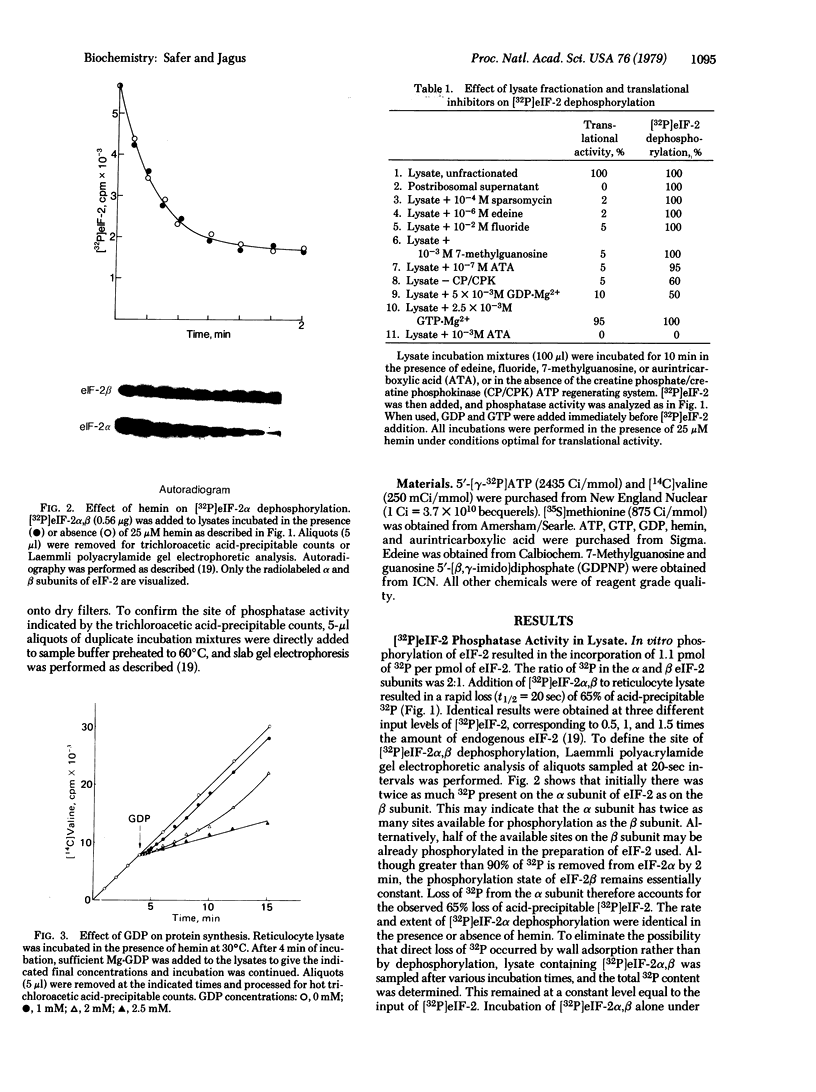
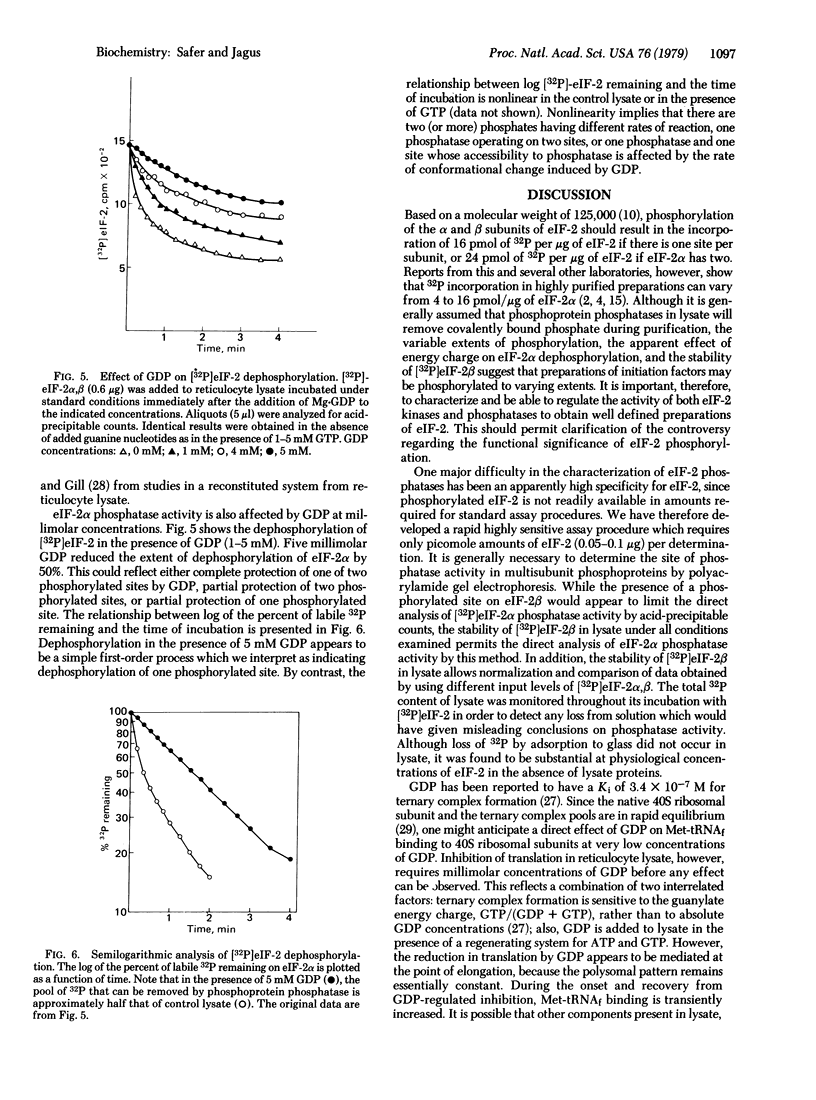
A sensitive assay requiring picomole amounts of [32P]eIF-2 to measure eIF-2 phosphatase activity has been developed. Dephosphorylation of [32P]eIF-2 alpha (38,000-dalton subunit) is extremely rapid (t 1/2 = 20 sec) and occurs at the same rate in both hemin-supplemented and hemin-depleted lysates. In contrast, [32P]eIF-2 beta phosphate is stable under all conditions studied. At concentrations required to produce a transient inhibition of protein synthesis, GDP prevents dephosphorylation of half of the phosphate introduced on eIF-2 alpha by the hemin-controlled repressor. Equimolar GTP is without effect. The concept that the energy charge of the guanylate pool may regulate accessibility of a phosphorylated site on eIF-2 alpha to its phosphatase and the implication of this to the mechanism of hemin-regulated translational control are discussed.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Both G. W., Furuichi Y., Muthukrishnan S., Shatkin A. J. Effect of 5'-terminal structure and base composition on polyribonucleotide binding to ribosomes. J Mol Biol. 1976 Jul 5;104(3):637–658. doi: 10.1016/0022-2836(76)90126-1. [DOI] [PubMed] [Google Scholar]
- Cherbas L., London I. M. On the mechanism of delayed inhibition of protein synthesis in heme-defecient rabbit reticulocyte lysates. Proc Natl Acad Sci U S A. 1976 Oct;73(10):3506–3510. doi: 10.1073/pnas.73.10.3506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farrell P. J., Balkow K., Hunt T., Jackson R. J., Trachsel H. Phosphorylation of initiation factor elF-2 and the control of reticulocyte protein synthesis. Cell. 1977 May;11(1):187–200. doi: 10.1016/0092-8674(77)90330-0. [DOI] [PubMed] [Google Scholar]
- Farrell P. J., Hunt T., Jackson R. J. Analysis of phosphorylation of protein synthesis initiation factor eIF-2 by two-dimensional gel electrophoresis. Eur J Biochem. 1978 Sep 1;89(2):517–521. doi: 10.1111/j.1432-1033.1978.tb12556.x. [DOI] [PubMed] [Google Scholar]
- Goldberg I. H., Mitsugi K. Sparsomycin inhibition of polypeptide synthesis promoted by synthetic and natural polynucleotides. Biochemistry. 1967 Feb;6(2):372–382. doi: 10.1021/bi00854a002. [DOI] [PubMed] [Google Scholar]
- Goldstein J., Safer B. Use of heparin-Sepharose for the rapid isolation of initiation and elongation factors. Methods Enzymol. 1979;60:165–181. doi: 10.1016/s0076-6879(79)60014-9. [DOI] [PubMed] [Google Scholar]
- Gross M., Mendelewski J. Additional evidence that the hemin-controlled translational repressor from rabbit reticulocytes is a protein kinase. Biochem Biophys Res Commun. 1977 Jan 24;74(2):559–569. doi: 10.1016/0006-291x(77)90340-0. [DOI] [PubMed] [Google Scholar]
- Gross M., Rabinovitz M. Partial purification of a translational repressor mediating hemin control of globin synthesis and implication of results on the site of inhibition. Biochem Biophys Res Commun. 1973 Feb 5;50(3):832–838. doi: 10.1016/0006-291x(73)91320-x. [DOI] [PubMed] [Google Scholar]
- Hoerz W., McCarty K. S. Initiation of protein synthesis in a rabbit reticulocyte lysate system. Biochim Biophys Acta. 1971 Jan 28;228(2):526–535. doi: 10.1016/0005-2787(71)90058-x. [DOI] [PubMed] [Google Scholar]
- Kramer G., Cimadevilla J. M., Hardesty B. Specificity of the protein kinase activity associated with the hemin-controlled repressor of rabbit reticulocyte. Proc Natl Acad Sci U S A. 1976 Sep;73(9):3078–3082. doi: 10.1073/pnas.73.9.3078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kramer G., Henderson A. B., Pinphanichakarn P., Wallis M. H., Hardesty B. Partial reaction of peptide initiation inhibited by phosphorylation of either initiation factor eIF-2 or 40S ribosomal proteins. Proc Natl Acad Sci U S A. 1977 Apr;74(4):1445–1449. doi: 10.1073/pnas.74.4.1445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin D., London I. M. Regulation of protein synthesis: activation by double-stranded RNA of a protein kinase that phosphorylates eukaryotic initiation factor 2. Proc Natl Acad Sci U S A. 1978 Mar;75(3):1121–1125. doi: 10.1073/pnas.75.3.1121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin D., Ranu R. S., Ernst V., London I. M. Regulation of protein synthesis in reticulocyte lysates: phosphorylation of methionyl-tRNAf binding factor by protein kinase activity of translational inhibitor isolated from hemedeficient lysates. Proc Natl Acad Sci U S A. 1976 Sep;73(9):3112–3116. doi: 10.1073/pnas.73.9.3112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lodish H. G., Housman D., Jacobsen M. Initiation of hemoglobin synthesis. Specific inhibition by antibiotics and bacteriophage ribonucleic acid. Biochemistry. 1971 Jun 8;10(12):2348–2356. doi: 10.1021/bi00788a027. [DOI] [PubMed] [Google Scholar]
- Obrig T., Irvin J., Culp W., Hardesty B. Inhibition of peptide initiation on reticulocyte ribosomes by edeine. Eur J Biochem. 1971 Jul 15;21(1):31–41. doi: 10.1111/j.1432-1033.1971.tb01436.x. [DOI] [PubMed] [Google Scholar]
- Ranu R. S., London I. M., Das A., Dasgupta A., Majumdar A., Ralston R., Roy R., Gupta N. K. Regulation of protein synthesis in rabbit reticulocyte lysates by the heme-regulated protein kinase: inhibition of interaction of Met-tRNAfMet binding factor with another initiation factor in formation of Met-tRNAfMet.40S ribosomal subunit complexes. Proc Natl Acad Sci U S A. 1978 Feb;75(2):745–749. doi: 10.1073/pnas.75.2.745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ranu R. S., London I. M. Regulation of protein synthesis in rabbit reticulocyte lysates: purification and initial characterization of the cyclic 3':5'-AMP independent protein kinase of the heme-regulated translational inhibitor. Proc Natl Acad Sci U S A. 1976 Dec;73(12):4349–4353. doi: 10.1073/pnas.73.12.4349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Revel M., Groner Y. Post-transcriptional and translational controls of gene expression in eukaryotes. Annu Rev Biochem. 1978;47:1079–1126. doi: 10.1146/annurev.bi.47.070178.005243. [DOI] [PubMed] [Google Scholar]
- Safer B., Adams S. L., Anderson W. F., Merrick W. C. Binding of MET-TRNAf and GTP to homogeneous initiation factor MP. J Biol Chem. 1975 Dec 10;250(23):9076–9082. [PubMed] [Google Scholar]
- Safer B., Anderson W. F., Merrick W. C. Purification and physical properties of homogeneous initiation factor MP from rabbit reticulocytes. J Biol Chem. 1975 Dec 10;250(23):9067–9075. [PubMed] [Google Scholar]
- Safer B., Anderson W. F. The molecular mechanism of hemoglobin synthesis and its regulation in the reticulocyte. CRC Crit Rev Biochem. 1978;5(4):261–290. doi: 10.3109/10409237809177144. [DOI] [PubMed] [Google Scholar]
- Safer B., Jagus R., Kemper W. M. Analysis of initiation factor function in highly fractionated and unfractionated reticulocyte lysate systems. Methods Enzymol. 1979;60:61–87. doi: 10.1016/s0076-6879(79)60008-3. [DOI] [PubMed] [Google Scholar]
- Tahara S. M., Traugh J. A., Sharp S. B., Lundak T. S., Safer B., Merrick W. C. Effect of hemin on site-specific phosphorylation of eukaryotic initiation factor 2. Proc Natl Acad Sci U S A. 1978 Feb;75(2):789–793. doi: 10.1073/pnas.75.2.789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trachsel H., Ranu R. S., London I. M. Regulation of protein synthesis in rabbit reticulocyte lysates: purification and characterization of heme-reversible translational inhibitor. Proc Natl Acad Sci U S A. 1978 Aug;75(8):3654–3658. doi: 10.1073/pnas.75.8.3654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trachsel H., Staehelin T. Binding and release of eukaryotic initiation factor eIF-2 and GTP during protein synthesis initiation. Proc Natl Acad Sci U S A. 1978 Jan;75(1):204–208. doi: 10.1073/pnas.75.1.204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Traugh J. A., Tahara S. M., Sharp S. B., Safer B., Merrick W. C. Factors involved in initiation of haemoglobin synthesis can be phosphorylated in vitro. Nature. 1976 Sep 9;263(5573):163–165. doi: 10.1038/263163a0. [DOI] [PubMed] [Google Scholar]
- Walton G. M., Gill G. N. Nucleotide regulation of a eukaryotic protein synthesis initiation complex;. Biochim Biophys Acta. 1975 May 1;390(2):231–245. doi: 10.1016/0005-2787(75)90344-5. [DOI] [PubMed] [Google Scholar]
- Walton G. M., Gill G. N. Preferential regulation of protein synthesis initiation complex formation by purine nucleotides. Biochim Biophys Acta. 1976 Sep 20;447(1):11–19. doi: 10.1016/0005-2787(76)90090-3. [DOI] [PubMed] [Google Scholar]
- de Haro C., Datta A., Ochoa S. Mode of action of the hemin-controlled inhibitor of protein synthesis. Proc Natl Acad Sci U S A. 1978 Jan;75(1):243–247. doi: 10.1073/pnas.75.1.243. [DOI] [PMC free article] [PubMed] [Google Scholar]



