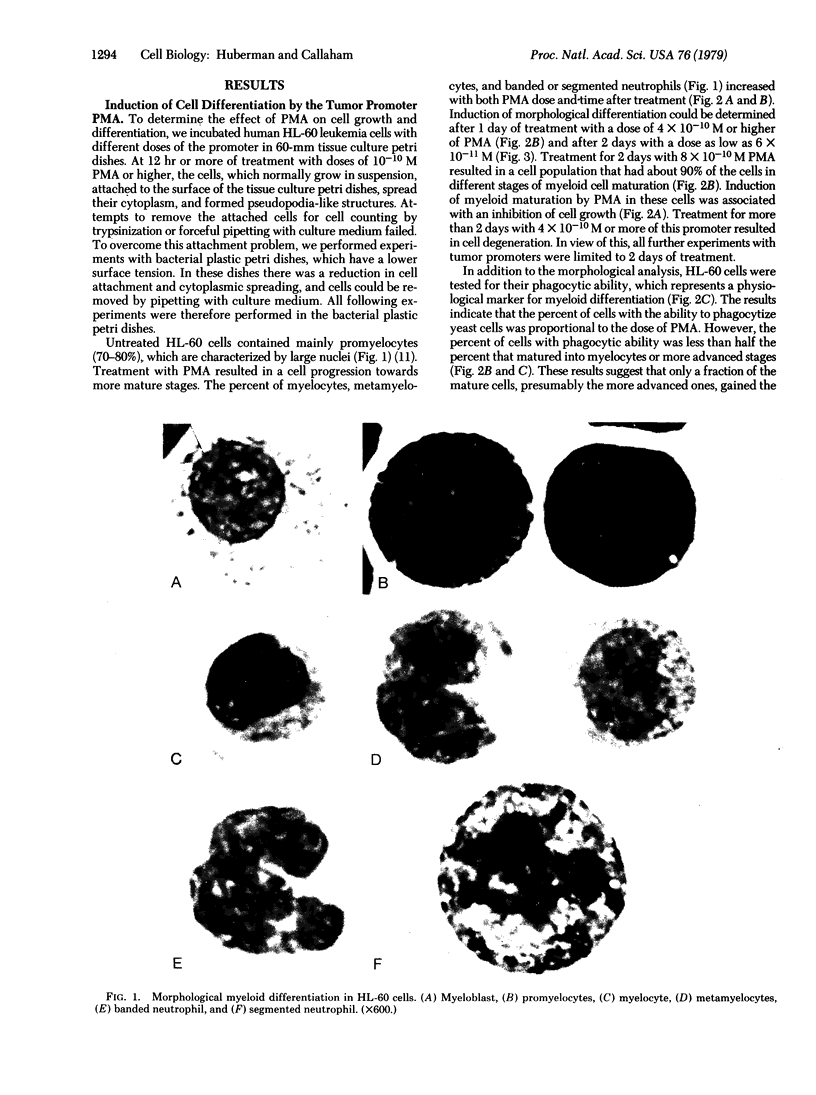
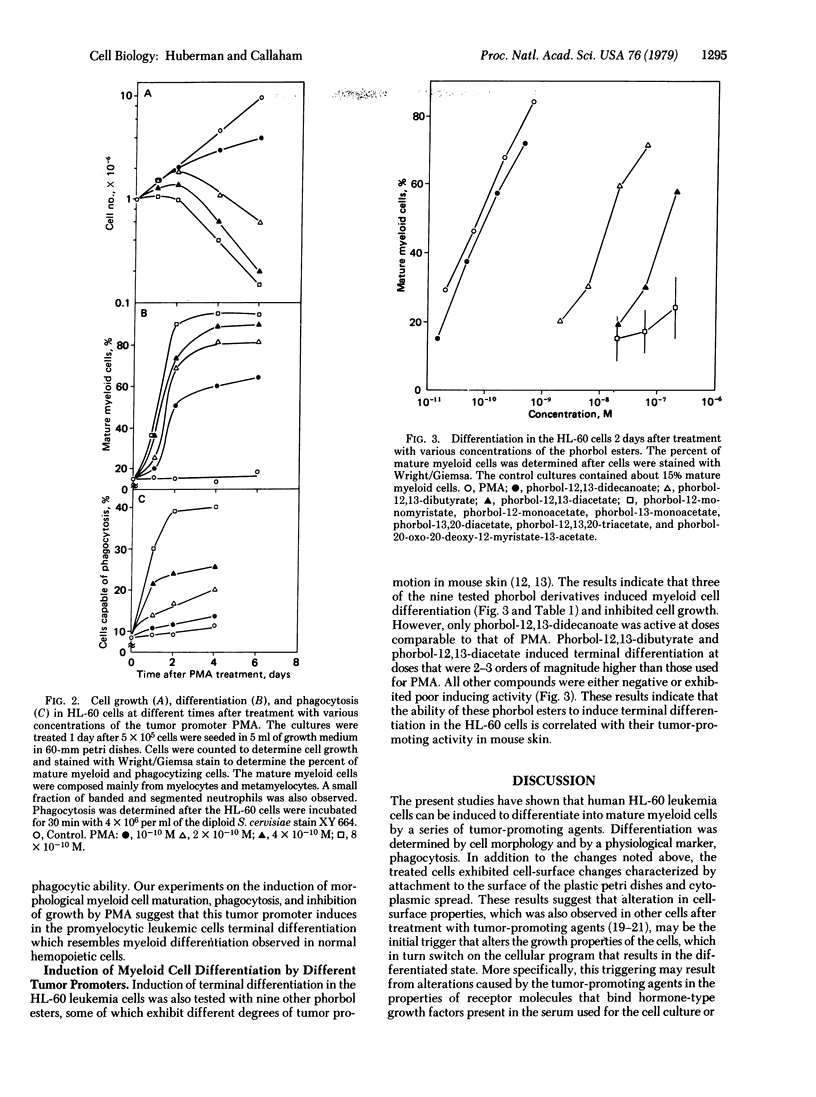
Abstract
Human promyelocytic leukemia cells (HL-60) were induced to differentiate into mature cells by the tumor-promoting agent phorbol-12-myristate-13-acetate and other related phorbol diesters. Differentiation was determined by an increase in the percent of myelocytes, metamyelocytes, and other mature myeloid cells as well as by an increase in the percent of phagocytizing cells. Induction of differentiation could be determined after 2 days of treatment with phorbol-12-myristate-13-acetate at a dose as low as 6 X 10(11) M. A correlation was found between reported tumor-promoting activity of a series of phorbol esters and their ability to induce myeloid differentiation and to inhibit cell growth. It is suggested that tumor-promoting agents like chemicals that induce terminal differentiation in these cells, at extremely low concentrations, may be used as a tool in the study of the control of cell growth, cell differentiation, and malignancy in human leukemic cells.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Augusti-Tocco G., Sato G. Establishment of functional clonal lines of neurons from mouse neuroblastoma. Proc Natl Acad Sci U S A. 1969 Sep;64(1):311–315. doi: 10.1073/pnas.64.1.311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins S. J., Gallo R. C., Gallagher R. E. Continuous growth and differentiation of human myeloid leukaemic cells in suspension culture. Nature. 1977 Nov 24;270(5635):347–349. doi: 10.1038/270347a0. [DOI] [PubMed] [Google Scholar]
- Collins S. J., Ruscetti F. W., Gallagher R. E., Gallo R. C. Terminal differentiation of human promyelocytic leukemia cells induced by dimethyl sulfoxide and other polar compounds. Proc Natl Acad Sci U S A. 1978 May;75(5):2458–2462. doi: 10.1073/pnas.75.5.2458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diamond L., O'Brien S., Donaldson C., Shimizu Y. Growth stimulation of human diploid fibro-blasts by the tumor promoter, 12-0-tetradecanoly-phorbol-13-acetate. Int J Cancer. 1974 May 15;13(5):721–730. doi: 10.1002/ijc.2910130516. [DOI] [PubMed] [Google Scholar]
- Diamond L., O'Brien T. G., Rovera G. Inhibition of adipose conversion of 3T3 fibroblasts by tumour promoters. Nature. 1977 Sep 15;269(5625):247–249. doi: 10.1038/269247a0. [DOI] [PubMed] [Google Scholar]
- Friend C., Scher W., Holland J. G., Sato T. Hemoglobin synthesis in murine virus-induced leukemic cells in vitro: stimulation of erythroid differentiation by dimethyl sulfoxide. Proc Natl Acad Sci U S A. 1971 Feb;68(2):378–382. doi: 10.1073/pnas.68.2.378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furmanski P., Silverman D. J., Lubin M. Expression of differentiated functions in mouse neuroblastoma mediated by dibutyryl-cyclic adenosine monophosphate. Nature. 1971 Oct 8;233(5319):413–415. doi: 10.1038/233413a0. [DOI] [PubMed] [Google Scholar]
- Huberman E., Mager R., Sachs L. Mutagenesis and transformation of normal cells by chemical carcinogens. Nature. 1976 Nov 25;264(5584):360–361. doi: 10.1038/264360a0. [DOI] [PubMed] [Google Scholar]
- Huebner R. J., Todaro G. J. Oncogenes of RNA tumor viruses as determinants of cancer. Proc Natl Acad Sci U S A. 1969 Nov;64(3):1087–1094. doi: 10.1073/pnas.64.3.1087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ichikawa Y. Differentiation of a cell line of myeloid leukemia. J Cell Physiol. 1969 Dec;74(3):223–234. doi: 10.1002/jcp.1040740303. [DOI] [PubMed] [Google Scholar]
- Ishii D. N., Fibach E., Yamasaki H., Weinstein I. B. Tumor promoters inhibit morphological differentiation in cultured mouse neuroblastoma cells. Science. 1978 May 5;200(4341):556–559. doi: 10.1126/science.644318. [DOI] [PubMed] [Google Scholar]
- Kluge N., Gaedicke G., Steinheider G., Dube S., Ostertag W. Globin synthesis in Friend-erythroleukemia mouse cells in protein- and lipid-free medium. Effects of dimethyl-sulfoxide, iron and erythropoietin. Exp Cell Res. 1974 Oct;88(2):257–262. doi: 10.1016/0014-4827(74)90239-0. [DOI] [PubMed] [Google Scholar]
- Lee L. S., Weinstein I. B. Tumor-promoting phorbol esters inhibit binding of epidermal growth factor to cellular receptors. Science. 1978 Oct 20;202(4365):313–315. doi: 10.1126/science.308698. [DOI] [PubMed] [Google Scholar]
- Lozzio C. B., Lozzio B. B., Yang W. K., Ichiki A. T., Bamberger E. G. Absence of thymus-derived lymphocyte markers in myelogenous leukemia (Ph1+) cell line K-562. Cancer Res. 1976 Dec;36(12):4657–4662. [PubMed] [Google Scholar]
- Miao R. M., Filedsteel A. H., Fodge D. W. Opposing effects of tumor promoters on erythroid differentiation. Nature. 1978 Jul 20;274(5668):271–272. doi: 10.1038/274271a0. [DOI] [PubMed] [Google Scholar]
- Paran M., Sachs L., Barak Y., Resnitzky P. In vitro induction of granulocyte differentiation in hematopoietic cells from leukemic and non-leukemic patients. Proc Natl Acad Sci U S A. 1970 Nov;67(3):1542–1549. doi: 10.1073/pnas.67.3.1542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rohrschneider L. R., Boutwell R. K. The early stimulation of phospholipid metabolism by 12-0-tetradecanoyl-phorbol-13-acetate and its specificity for tumor promotion. Cancer Res. 1973 Aug;33(8):1945–1952. [PubMed] [Google Scholar]
- Rovera G., O'Brien T. G., Diamond L. Tumor promoters inhibit spontaneous differentiation of Friend erythroleukemia cells in culture. Proc Natl Acad Sci U S A. 1977 Jul;74(7):2894–2898. doi: 10.1073/pnas.74.7.2894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sachs L. Regulation of membrane changes, differentiation, and malignancy in carcinogenesis. Harvey Lect. 1974;68:1–35. [PubMed] [Google Scholar]
- Sivak A., Mossman B. T., Van Duuren B. L. Activation of cell membrane enzymes in the stimulation of cell division. Biochem Biophys Res Commun. 1972 Jan 31;46(2):605–609. doi: 10.1016/s0006-291x(72)80182-7. [DOI] [PubMed] [Google Scholar]
- Temin H. M. On the origin of the genes for neoplasia: G.H.A. Clowes memorial lecture. Cancer Res. 1974 Nov;34(11):2835–2841. [PubMed] [Google Scholar]
- Todaro G. J., Huebner R. J. N.A.S. symposium: new evidence as the basis for increased efforts in cancer research. Proc Natl Acad Sci U S A. 1972 Apr;69(4):1009–1015. doi: 10.1073/pnas.69.4.1009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wertz P. W., Mueller G. C. Rapid stimulation of phospholipid metabolism in bovine lymphocytes by tumor-promoting phorbol esters. Cancer Res. 1978 Sep;38(9):2900–2904. [PubMed] [Google Scholar]
- Yamasaki H., Fibach E., Nudel U., Weinstein I. B., Rifkind R. A., Marks P. A. Tumor promoters inhibit spontaneous and induced differentiation of murine erythroleukemia cells in culture. Proc Natl Acad Sci U S A. 1977 Aug;74(8):3451–3455. doi: 10.1073/pnas.74.8.3451. [DOI] [PMC free article] [PubMed] [Google Scholar]