Abstract
Fibrosing mediastinitis (FM), also known as granulomatous or sclerosing mediastinitis, is an uncommon but serious cause of chest symptoms. Due to an infectious or inflammatory challenge, production of collagen occurs in the confined space of the mediastinum. Collagen formation leads to compression of vital structures, resulting in cough, chest pain and dyspnea. The majority of cases of FM occur as a result of prior exposure to Histoplasma capsulatum. The following is a case of a previously healthy young woman who presented with a 3-month history of cough, chest pain and trouble breathing, and was subsequently found to have fibrosing mediastinitis. Fibrosing mediastinitis should be considered in the differential diagnosis of cough, chest pain and dyspnea, primarily when findings such as increased venous pressure are present on physical exam and hilar abnormalities are seen on chest radiograph. Clinical presentation, diagnosis and management of fibrosing mediastinitis are discussed.
KEY WORDS: fibrosing mediastinitis, cough, chest pain, dyspnea
CASE
A 38-year-old Brazilian woman presented to the outpatient clinic for evaluation of cough, chest pain and trouble breathing for 3 months. The cough was dry, not associated with wheezing and did not disturb her sleep. The chest pain was sharp and intermittent. It was not worsened with exertion, position change or deep breathing. She denied palpitations or diaphoresis. She described being aware of her breathing, stating she was now short of breath with one flight of stairs. She denied fever, chills, night sweats, sick contacts or weight loss. The patient had emigrated from Brazil 4 years earlier and worked as a house cleaner. She lived with her boyfriend; they had no children or pets. Her only medication was an oral contraceptive. She did not smoke or use illicit drugs and drank alcohol only occasionally. She had no significant family history.
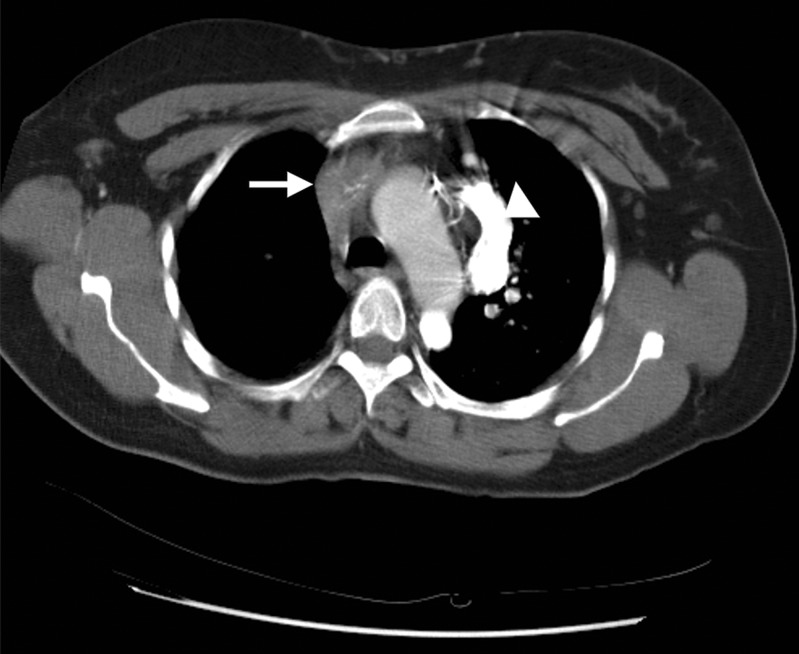
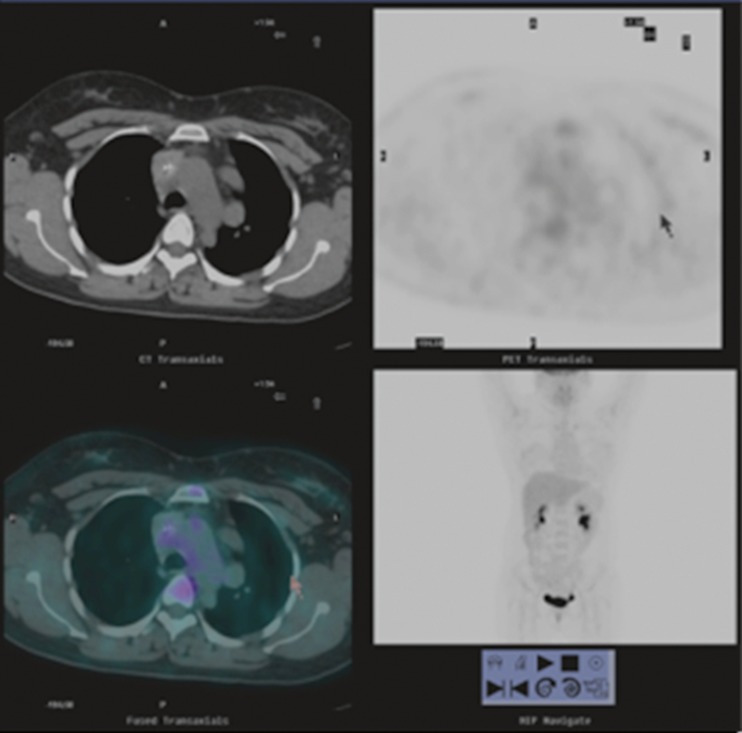
Physical examination was normal, except for numerous superficial veins that were visible across the anterior chest wall. Laboratory examination included complete blood count, comprehensive metabolic panel, HIV antibody screen and urinalysis. Results were normal with the exception of a hemoglobin of 9.6 g. Chest radiograph demonstrated an increased opacity in the right paratracheal region and aortopulmonary window with an area of bulging in the retrocardiac area to the mediastinal contour. Heart size was normal and no consolidations were identified. Chest computed tomography (CT) with contrast showed a 2.8 × 2.8 cm partially calcified, mediastinal mass with obstruction of the superior vena cava (SVC) (Fig. 1). Large left-sided collateral veins were noted. No lung mass, endobronchial lesion, mediastinal or hilar adenopathy were present. Because neoplasm was a concern, positron emission tomography (PET)-CT to evaluate for disease outside the mediastinum was performed. Results showed that the mediastinal mass had mild fluorodeoxyglucose (FDG) uptake, suggesting a benign etiology, based on the intensity of the uptake and lack of intraabdominal or retroperitoneal pathology (Fig. 2). Mediastinoscopy and biopsy of the mass revealed benign vascular adipose tissue and a benign lymph node. Two weeks post-procedure, the patient returned complaining of right sided neck pain, as well as right sided facial and neck swelling. CT of the neck showed thrombosis of the right internal jugular vein with multiple venous collaterals and complete obstruction of the superior vena cava.
Figure 1.
Chest CT with contrast showing mediastinal mass with calcification (arrow). Dilated superior intercostal and accessory hemiazygous veins due to superior vena cava obstruction (arrow head).
Figure 2.
PET-CT Mild FDG uptake, consistent with a benign etiology for the mass.
Additional studies to help identify the etiology of the mass were performed. A urine histoplasma antigen was negative. A serum angiotensin converting enzyme level (ACE) was sent to evaluate for sarcoid and was normal. Bronchoalveolar lavage (BAL) showed benign bronchial cells, abundant macrophages, few multinucleated giant cells, polymorphonuclear leukocytes and lymphocytes. Cultures for bacteria, acid-fast bacilli, and fungi were all negative. Gomori methenamine silver stain of the BAL fluid was negative for fungi. The patient underwent right anterior thoracotomy with mediastinal biopsy. Histology demonstrated dense fibrous and collagenized tissue with minimal chronic inflammation. Trichrome staining highlighted collagen bundles (Fig. 3). Congo red stain was negative for amyloid. Special stains for microorganisms, including fungi, were negative. Histopathology was consistent with sclerosing or fibrosing mediastinitis (FM).
Figure 3.
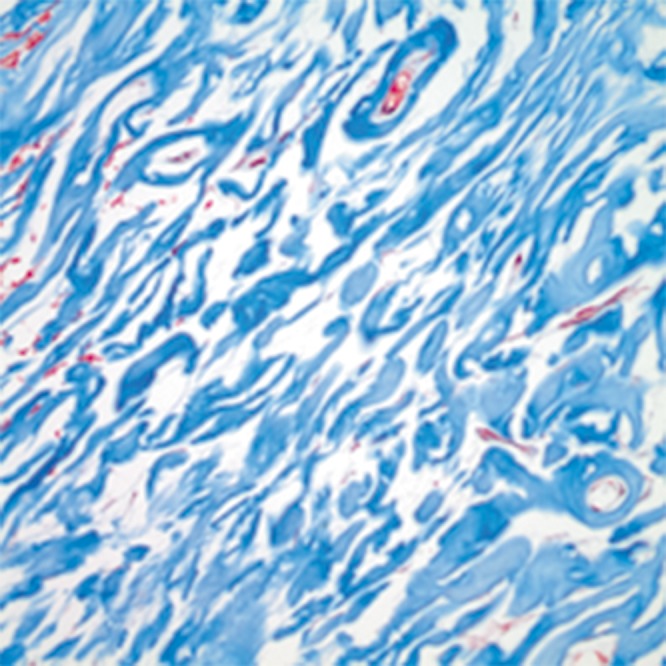
Fibrosing mediastinitis trichrome stain, 10X blue stain highlights the collagen bundles and red stain indicates blood vessels.
DISCUSSION
The number of ambulatory patients in the United States presenting with respiratory complaints is staggering, with 101 million visits in 2010 alone.1–3 Given the magnitude of this cohort, unusual causes for cough, chest pain and dyspnea may need to be considered in certain clinical settings. Detailed history taking can help stratify patients into groups that require additional evaluation. History of tobacco use or other toxic exposure places patients at greater risk for serious pathology. Episodic shortness of breath may indicate a relapsing/remitting condition such as asthma. The duration cough has been present can be used to help identify diagnostic possibilities. Irwin and Madison describe three categories of cough: acute, lasting less than 3 weeks; subacute, lasting 3–8 weeks; and chronic cough which is greater than 8 weeks in duration.4 They also propose that each category has its own set of etiologies. Our patient had chronic cough, with no history of toxin exposure or reactive airway disease. Chronic cough is most often caused by obstructive lung disease, gastroesophageal reflux disease or postnasal drip syndrome in the majority of ambulatory adult patients.4 After common conditions are eliminated, consideration should be given to a more unusual etiology of respiratory symptoms, as in the clinical vignette.
Fibrosing mediastinitis (FM), also known as granulomatous or sclerosing mediastinitis, is an uncommon but serious cause of chest symptoms. The etiology of FM is thought to be an abnormal host response to an infectious or inflammatory challenge, causing the production of large amounts of collagen and fibrosis.5 The agent most commonly associated with FM is Histoplasma capsulatum. The risk of development of FM is small, with estimates that less than one in 20,000 patients with histoplasmosis infection will develop FM.6 However, an increasing incidence of histoplasmosis has been described in Latin America and is attributed to migration, tourism and population growth.7 Our patient’s urinary Histoplasma antigen test was negative, which came as no surprise, as the urinary Histoplasma antigen is more likely to be positive in the setting of disseminated infection or acute pulmonary infection with a high fungal burden.8 At the time of the test, her potential exposure was remote and her condition localized. Other infectious etiologies include aspergillus,9 blastomycosis,10 tuberculosis11 and mucormycosis.12 These mycobacterial or fungal antigens may lead to fibroblast proliferation in a genetically susceptible host. Peebles and colleagues found that the relative risk of FM was three times higher in individuals with the HLA (human leukocyte antigen)-A2 antigen, suggesting there are host-specific variables in the development of fibrosis.13
Cough, chest pain, dyspnea and hemoptysis are common presenting complaints in patients with FM. The signs and complications of FM are due to the dense collagenous mass causing compression of the components of the mediastinum. Loyd et al. reviewed 71 cases of FM and found the following: cough in 41 %, chest pain in 23 %, dyspnea 31 % and hemoptysis in 31 %.5 Chest pain can be pleuritic and also exertional. Substernal discomfort worsened by swallowing or inspiration is thought to be due to mediastinal lymphadenopathy along with the fibrous mass.14 Hemoptysis may occur, and ranges from mild and intermittent to massive. Dunn et al. describe two cases of FM with massive hemoptysis due to broncholith erosion causing endobronchial arterial bleeding. Both patients required pneumonectomy.14 It is more common for the fibrous mass to cause simple airway obstruction than hemoptysis. Airway obstruction can lead to post-obstructive pneumonia and recurrent infections with fever, night sweats and weight loss as initial complaints. The patient may also be completely asymptomatic and be found to have an abnormal chest radiograph done for an unrelated reason.
A more dramatic but less common presentation of FM is superior vena cava syndrome. SVC syndrome is the constellation of findings that result from obstruction of the SVC from thrombosis or external compression. Symptoms may include swelling of the face, neck and arms and visible widening of the veins of the upper body. Loyd reports an incidence of SVC syndrome of 6 % at initial presentation and 11 % at the time of diagnosis.5 Hammoud et al., in a retrospective review of all patients who underwent surgical intervention for FM at their institution, found only one case of SVC obstruction of the 49 cases identified.15 In a retrospective review of 80 patients with FM at the Mayo Clinic, 21 % presented with facial or neck swelling consistent with SVC syndrome.16 The characteristic physical findings of venous distension of the neck and chest wall with facial plethora occurred in our patient 10 days post-mediastinoscopy. However, the numerous chest wall veins present on her initial exam were consistent with elevated venous pressure.
Due to the nonspecific and general nature of presenting complaints, chest radiographs are often obtained as part of the initial workup. They most commonly show hilar abnormalities: hilar mass, mediastinal widening, and mediastinal calcification. It is also possible for chest radiographs to be normal, underestimating the degree of mediastinal involvement with fibrous tissue. Chest CT with contrast is the preferred and most useful imaging modality for FM.17 Chest CT can more clearly assess mediastinal anatomy, as well as the presence or absence of calcification within mediastinal lymph nodes. Intravascular contrast can demonstrate vascular obstruction, specifically SVC obstruction, and collateralization of vessels.
Sherrick et al. describe two distinct radiologic patterns for FM, focal and diffuse.17The patterns appear to have some significance as far as etiology. The focal type is a localized, calcified mass in the paratracheal or subcarinal region. FM is frequently more focal on the right, due to lymphatic drainage patterns. Lymphatics draining both lungs flow to the hilar and subcarinal lymph nodes and then to the right paratracheal chain.18 The focal type is more closely associated with an infectious etiology, is more common than the diffuse form and is not associated with other systemic sclerosing disorders. The diffuse type of FM is noncalcified, affects more than one mediastinal region and can be associated with fibrosis in other anatomic areas. Diffuse FM may be associated with other idiopathic fibrotic disorders, such as retroperitoneal fibrosis, sclerosing cholangitis, orbital pseudotumor, and Reidel’s thyroiditis.19 With the localized, partially calcified paratracheal mass on the right, we believe our patient represents the focal form of FM.
PET scanning and MRI have also been used to evaluate FM. Takalkar et al.20 discuss the value of FDG PET CT imaging in the evaluation of FM. In their case report, the adenopathy demonstrated intense FDG activity, making it suspicious for a neoplastic process. Upon subsequent biopsy, the areas most FDG avid had collections of neutrophils, macrophages and mesothelial cells, more compatible with inflammation. Also present was hyalinized fibrous tissue with lymphoid infiltrate consistent with FM. They hypothesize that this was a more active lesion and that FDG PET CT scanning may be a way to monitor disease activity in FM. Our patient’s PET scan showed only mild FDG uptake, likely indicating a less active lesion and less active disease. Magnetic resonance imaging (MRI) of the chest may also be helpful. Though it poorly depicts calcification, MRI can add information on vascular patency without the need for IV contrast. The adenopathy of FM is usually of low signal intensity on MRI, possibly indicating a benign nature. This low signal intensity may be of use if calcifications are not seen within the mass on CT.21
There is no generally accepted medical treatment for FM. Corticosteroids have been used for their anti-inflammatory effects.22,23 No data on efficacy are available and the isolated cases that have been reported to show improvement have been with the diffuse type of mediastinal involvement.16 Antifungal agents have been tried because of FM’s association with endemic mycoses, though there is usually no evidence of active infection at the time of diagnosis.7 In the 2007 practice guidelines, the Infectious Disease Society of America recommends against the use of antifungal therapy for FM due to histoplasmosis.24 Selective estrogen receptor modulators have been used because of their utility in systemic fibrotic disorders.25 Surgical intervention is technically difficult, as the risk of hemorrhage from dilated collateral vessels is great. Surgical therapies have involved debulking, SVC bypass with spiral vein graft and endovascular interventions such as stenting or balloon angioplasty.26–28 For cases of airway compression or obstruction, removal of the involved airway and associated lung tissue may be required.14 In 70 patients treated with surgical therapies for SVC syndrome of benign etiology, Rizvi et al. reported efficacy for both open surgical repair and endovascular interventions, though graft patency was greater with open surgical procedures.26 Our patient received a brief, tapering course of corticosteroids without change in symptoms. She has had follow-up chest CT documenting stability of the mass.
Prognosis in FM depends on the extent and anatomic location of the fibrotic mass, with bilateral and subcarinal involvement suggesting a more severe clinical course and increased mortality.5 In the review by Peikert et al., their group of 80 patients showed an overall survival similar to age matched controls.16 The only two FM related deaths in their cohort occurred in patients that had bilateral mediastinal involvement.1
In conclusion, the vignette highlights FM as an unusual etiology for the common complaints of cough, chest pain and shortness of breath. With over 100 million visits annually for respiratory symptoms, less common etiologies may need to be considered in the appropriate setting. Patients with physical findings of increased venous pressure in the chest and hilar abnormalites on imaging may warrant consideration of FM as a possible explanation.
Acknowledgements
Contributors
None.
Funders
None.
Prior Presentations
None.
Conflicts of Interest
The authors declare that they do not have a conflict of interest.
REFERENCES
- 1.NCHS. National Hospital Ambulatory Medical Care Survey. 2010 outpatient department summary tables. Available from: http://www.cdc.gov/nchs/data/ahcd/nhamcs_outpatient/2010_opd_web_tables.pdf Accessed 24 February 2013.
- 2.NCHS. National Hospital Ambulatory Medical Care Survey: 2010 emergency department summary tables. Available from: http://www.cdc.gov/nchs/data/ahcd/nhamcs_emergency/2010_ed_web_tables.pdf Accessed 24 February 2013.
- 3.NCHS. National Ambulatory Medical Care Survey: 2010 summary tables. Available from: http://www.cdc.gov/nchs/data/ahcd/nhamcs_summary/outpatieny/2010_opd_web_tables.pdf Accessed 24 February 2013.
- 4.Irwin RS, Madison JM. The Diagnosis and Treatment of Cough. N Engl J Med. 2000;343:1715–1721. doi: 10.1056/NEJM200012073432308. [DOI] [PubMed] [Google Scholar]
- 5.Loyd JE, Tillman BF, Atkinson JB, Desprez RM. Mediastinal fibrosis complicating histoplasmosis. Medicine. 1998;67:295–310. doi: 10.1097/00005792-198809000-00002. [DOI] [PubMed] [Google Scholar]
- 6.Hage CA, Wheat LJ, Loyd J, Allen SD, Blue D, Knox KS. Pulmonary Histoplasmosis. Semin Respir Crit Care Med. 2008;29:151–165. doi: 10.1055/s-2008-1063854. [DOI] [PubMed] [Google Scholar]
- 7.Sifuentes-Osnorio J, Corzo-Leon DE, Ponce-de-Leon LA. Epidemiology of Invasive Fungal Infections in Latin America. Curr Fungal Infect Rep. 2012;6:23–34. doi: 10.1007/s12281-011-0081-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kaufmann CA. Histoplasmosis: a Clinical and Laboratory Update. Clin Microbiol Rev. 2007;20:115–32. doi: 10.1128/CMR.00027-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wightman SC, Kim AW, Proia LA, Faber LP, Gattuso P, Warren WH, et al. An unusual case of Aspergillus fibrosing mediastinitis. Ann Thorac Surg. 2009;88:1352–4. doi: 10.1016/j.athoracsur.2009.02.087. [DOI] [PubMed] [Google Scholar]
- 10.Lagerstrom CF, Mitchell HG, Hammon JW. Chronic fibrosing mediastinitis and superior vena cava obstruction from blastomycosis. Ann Thorac Surg. 1992;54(4):764–5. doi: 10.1016/0003-4975(92)91025-5. [DOI] [PubMed] [Google Scholar]
- 11.Weinberger SE Fibrosing Mediastinitis. In: UptoDate, Basow DS(ed) UptoDate, Waltham, MA 2011.
- 12.Robertson BD, Bautista MA, Russell TS, Naclerio AL, Porisch ME, Sorrells DL, et al. Fibrosing mediastinitis secondary to zygomycosis in a twenty-two month old child. PIDJ. 2002;21(5):441–2. doi: 10.1097/00006454-200205000-00023. [DOI] [PubMed] [Google Scholar]
- 13.Peebles RS, Carpenter CT, Dupont WD, Loyd JE. Mediastinal fibrosis is associated with human leukocyte antigen-A2. Chest. 2000;117:482–485. doi: 10.1378/chest.117.2.482. [DOI] [PubMed] [Google Scholar]
- 14.Dunn EJ, Ulieny KS, Wright CB, Gottesman L. Surgical implications of fibrosing mediastinitis. Chest. 1990;97:338–46. doi: 10.1378/chest.97.2.338. [DOI] [PubMed] [Google Scholar]
- 15.Hammond ZT, Rose AS, Hage CA, Knox KS, Reiger K, Kesler KA. Surgical management of pulmonary and mediastinal sequelae of histoplasmosis: a challenging spectrum. Ann Thorac Surg. 2009;88:399–403. doi: 10.1016/j.athoracsur.2009.04.041. [DOI] [PubMed] [Google Scholar]
- 16.Peikert T, Colby TV, Midthun DE, Pairolero PC, Edell ES, Schroeder DR, et al. Fibrosing Mediastinitis: clinical presentation, therapeutic outcomes and adaptive immune response. Medicine. 2011;90:412–23. doi: 10.1097/MD.0b013e318237c8e6. [DOI] [PubMed] [Google Scholar]
- 17.Sherrick AD, Brown LR, Harms GF, Meyer JL. The radiographic findings of fibrosing mediastinitis. Chest. 1994;106:484–89. doi: 10.1378/chest.106.2.484. [DOI] [PubMed] [Google Scholar]
- 18.Case records of the Massachusetts General Hospital(Case 6-1989) N Engl J Med. 1989;320:380–9. doi: 10.1056/NEJM198902093200608. [DOI] [PubMed] [Google Scholar]
- 19.Dehner LP, Coffin CM. Idiopathic fibrosclerotic disorders and other inflammatory pseudodtumors. Semin Diagn Pathol. 1998;15(2):161–173. [PubMed] [Google Scholar]
- 20.Takalkar AM, Bruno GL, Makanjoula AJ, El-Haddad G, Lilien DL, Payne KD. A potential role for F-18 FDG PET/CT in evaluation and management of fibrosing mediastinitis. Clin Nucl Med. 2007;32(9):703–9. doi: 10.1097/RLU.0b013e318125035b. [DOI] [PubMed] [Google Scholar]
- 21.Rholl KS, Levitt RG, Glazer HS. Magnetic resonance imaging of fibrosing mediastinitis. AJR. 1985;145(2):255–9. doi: 10.2214/ajr.145.2.255. [DOI] [PubMed] [Google Scholar]
- 22.Ikeda K, Nomari H, Mori T, et al. Successful steroid treatment for fibrosing mediastinitis and sclerosing cervicitis. Ann Thorac Surg. 2007;83(3):1199–201. doi: 10.1016/j.athoracsur.2006.09.034. [DOI] [PubMed] [Google Scholar]
- 23.Kingusa S, Tachibana S, Kawakami M, Orino T, Yamamoto R, Sasaki S. Idiopathic mediastinal fibrosis:report of a case. Surg Today. 1998;28(3):335–8. doi: 10.1007/s005950050135. [DOI] [PubMed] [Google Scholar]
- 24.Wheat LJ, Friefeld AG, Kleiman MB, et al. Clinical Practice Guidelines for the Management of Patients with Histoplasmosis: 2007 Update by the Infectious Diseases Society of America. Clin Inf Dis. 2007;45(7):807–25. doi: 10.1086/521259. [DOI] [PubMed] [Google Scholar]
- 25.Savelli BA, Parshley M, Morganroth M. Successful treatment of sclerosing cervicitis and fibrosing mediastinitis with tamoxifen. Chest. 1997;11(4):1137–40. doi: 10.1378/chest.111.4.1137. [DOI] [PubMed] [Google Scholar]
- 26.Rizvi AZ, Kalra M, Bjarnson H, Bower TC, Schleck C, Gloriczki P. Benign superior vena cava syndrome: stenting is now first line of treatment. J Vasc Surg. 2008;47:372–80. doi: 10.1016/j.jvs.2007.09.071. [DOI] [PubMed] [Google Scholar]
- 27.Doty JR, Flores JH, Doty JB. Superior vena cava obstruction:bypass using spiral vein graft. Ann Thorac Surg. 1999;67(4):1111–6. doi: 10.1016/S0003-4975(99)00145-9. [DOI] [PubMed] [Google Scholar]
- 28.Albers EL, Pugh ME, Hill KD, Wang L, Loyd JE, Doyle TP. Percutaneous vascular stent implantation as treatment of central vascular obstruction due to fibrosing mediastinitis. Circulation. 2011;123:1391–99. doi: 10.1161/CIRCULATIONAHA.110.949180. [DOI] [PMC free article] [PubMed] [Google Scholar]