SUMMARY
Resolved ER stress response is essential for intracellular homeostatic balance, but unsettled ER stress can lead to apoptosis. Here, we show that a pro-apoptotic p53 target, CDIP1, acts as a key signal transducer of ER stress-mediated apoptosis. We identify BAP31, B-cell receptor-associated protein 31, as an interacting partner of CDIP1. Upon ER stress, CDIP1 is induced and enhances an association with BAP31 at the ER membrane. We also show that CDIP1 binding to BAP31 is required for BAP31 cleavage upon ER stress and for BAP31-Bcl-2 association. The recruitment of Bcl-2 to the BAP31-CDIP1 complex, as well as CDIP1-dependent tBid and caspase-8 activation, contributes to BAX oligomerization. Genetic knockout of CDIP1 in mice leads to impaired response to ER stress-mediated apoptosis. Together, our data demonstrate that the CDIP1/BAP31-mediated regulation of mitochondrial apoptosis pathway represents a novel mechanism for establishing an ER-mitochondrial cross-talk for ER stress-mediated apoptosis signaling.
INTRODUCTION
The endoplasmic reticulum (ER) is a specialized intracellular organelle responsible for the proper localization and folding of proteins, which has crucial roles in cellular homeostasis, development and stress responsiveness (Walter and Ron, 2011). In response to cellular stress, a well-established signaling cascade the unfolded protein response (UPR) is activated to maintain and restore proper ER homeostasis. However, as cells are exposed to irremediable ER stress, the apoptotic response is initiated to eliminate these damaged cells (Walter and Ron, 2011). Thus, the dysregulation of ER stress signaling has been implicated in a variety of human diseases including metabolic disorders, neurodegenerative diseases, inflammatory diseases, and cancer (Hotamisligil, 2010; Tabas and Ron, 2011). While the molecular mechanism of ER stress-induced apoptosis is not fully understood, accumulating evidence indicates that chronic or irreversible ER stress results in apoptosis via the regulation of the Bcl-2 family of proteins (Hetz et al., 2006; Rodriguez et al., 2012). Upon ER stress, pro-apoptotic members, such as Bax and Bak, of the Bcl-2 family proteins are recruited to the ER surface and the mitochondria while overexpression of the anti-apoptotic Bcl-2 family members can prevent ER stress-mediated apoptosis (Scorrano et al., 2003). BH3 domain-only family members, PUMA, NOXA, BID and BIM have been implicated in ER stress-induced apoptosis although cells deficient in one or more of these genes are not completely resistant to ER stress-mediated cell death (Chen et al., 2005; Li et al., 2006; Puthalakath et al., 2007). Recent studies have uncovered the critical factors responsible for the cross-talk of apoptosis signals between the mitochondria and ER by identifying the association between ER-localized BAP31 and mitochondrial fission factor Fis1 (Iwasawa et al., 2011). BAP31 is an integral ER membrane protein that functions as an escorting factor in the sorting of newly synthesized membrane proteins within the ER (Wang et al., 2008). In addition to its role in ER protein trafficking, it has been shown that a number of apoptotic pathways involve the cleavage of BAP31 by caspase-8 (Breckenridge et al., 2002; Breckenridge et al., 2003), and that BAP31 functions as a regulator of apoptosis through an interaction with Bcl-2 or Bcl-XL and caspase-8 (Ng et al., 1997; Ng and Shore, 1998).
CDIP1 was identified as a novel p53 target gene that is up-regulated upon DNA damage and a key downstream effector of p53-dependent apoptosis (Brown et al., 2007). Here, we describe that CDIP1 acts as a key signal transducer of ER stress response through its interaction with BAP31 at the ER membrane. We found that the subsequent cleavage of BAP31 occurs in an ER-stress regulated manner, followed by enhanced BAP31-Bcl-2 association, which is dependent on CDIP1 expression. Upon stress the resulting sequestration of Bcl-2 from CDIP1-BAP31 complex and CDIP1-dependent t-Bid and caspase-8 activation result in BAX oligomerization and mitochondrial apoptosis. Thus, the CDIP1/BAP31-mediated regulation of mitochondrial apoptosis pathway represents a novel mechanism for establishing an ER-mitochondrial cross-talk for ER stress-mediated apoptosis.
RESULTS
Identification of BAP31 as a CDIP1 partner upon ER stress
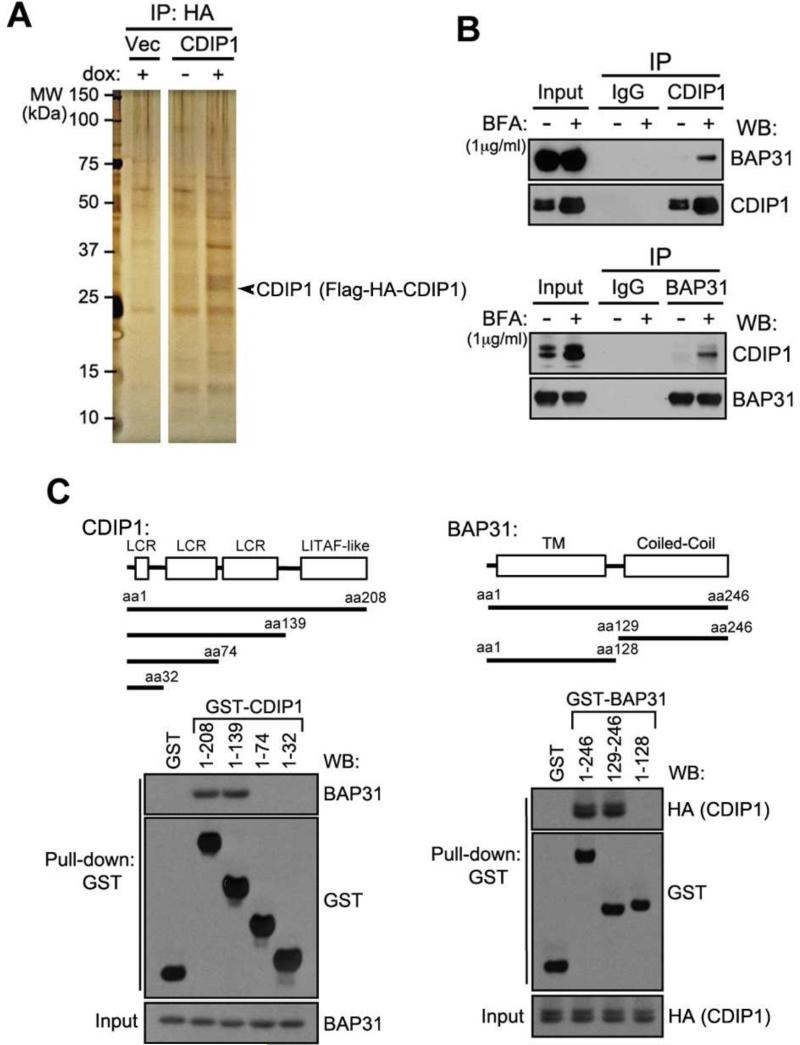
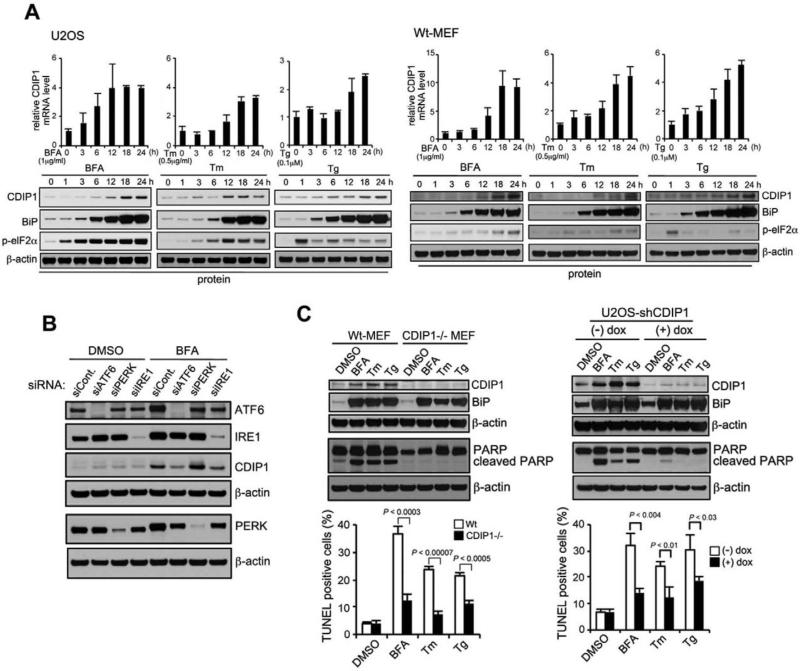
To further understand the role of CDIP1 in the stress response pathway, we performed immunoprecipitation-mass spectroscopy (MS) experiments to identify CDIP1-binding proteins. We utilized the TAP purification approach (Nakatani and Ogryzko, 2003) to isolate CDIP1 interacting proteins by expressing FLAG-HA-double-tagged CDIP1 in doxycycline-regulated manner in U2OS cells. Cell extracts with or without ectopic CDIP1 expression were subjected to purification with anti-FLAG and anti-HA antibody resin, and the resulting precipitates were subjected to LC-MS/MS analysis. Several polypeptides, including BAP31, ATP synthase, HSC70, and reticulon-4, were identified as CDIP1 binding partners (Figure 1A and Table S1). Among these candidate CDIP1 binding proteins, ER protein BAP31 was of interest for the study since there have been a number of reports linking BAP31 to control of ER stress-induced apoptosis. We first confirmed the interaction between CDIP1 and BAP31 in the absence or presence of ER-specific stress (brefeldin A, BFA) by reciprocal immunoprecipitation in U2OS cells. As shown in Figure 1B and Figure S1A, CDIP1 and BAP31 associated endogenously, and their interaction appeared to be enhanced upon BFA treatment and occurred slightly prior to BFA-induced apoptosis. Furthermore, CDIP1 expression was significantly increased in response to BFA treatment (Figure 1B and Figure S1A). We next mapped the region in which CDIP1 and BAP31 interacted by GST pull-down experiments with GST-fusion constructs expressing various sizes of CDIP1 or BAP31 as well as GST alone in HA-CDIP1 transfected U2OS cells. GST pull-down experiments demonstrated that CDIP1 aa75-139 (3rd LCR region) bound to the coiled-coil domain (aa128-246) of BAP31 (Figure 1C). Additionally, confocal image analysis showed that CDIP1 levels increased and it appeared to co-localize with ER-bound BAP31 in response to ER stress (Figure S1B). We further investigated whether CDIP1 is an ER stress-response protein. We challenged U2OS cells and MEF cells with ER stress agents (BFA, tunicamycin (Tm), and thapsigargin (Tg)) and compared expression levels of CDIP1 to other ER stress response markers. Early induction of BiP and p-elF2α was followed by gradual increase of CDIP1 mRNA and protein levels (Figures 2A). It is now well established that three ER resident proteins (IRE1, PERK, and ATF6) function as major sensors of unfolded proteins and initiate series of signaling events to circumvent the ER stress (Walter and Ron, 2011). We next tested whether RNAi-mediated depletion of each of the three ER stress sensors/pathways would affect the ER stress-mediated induction of CDIP1. As shown in Figure 2B, we observed that the depletion of ATF6 or IRE1 inhibited CDIP1 induction upon BFA treatment. We also determined the role of p53 in ER stress-mediated CDIP1 induction. It has been previously reported that p53 expression increases in response to ER stress (Li et al., 2006), but ER stress-mediated CDIP1 induction was only partially affected by the depletion or loss of p53 (Figure S2), suggesting that ER stress-mediated CDIP1 induction is likely regulated by two major ER stress sensor pathways: ATF6 and IRE1, as well as p53. These results indicate that the level of CDIP1 is elevated and associates with BAP31 at the ER region in response to ER stress, implicating CDIP1 function in the ER stress response of the cell.
Figure 1. CDIP1 interacts with BAP31 and ER stress increases this interaction.
(A) Affinity purification of CDIP1 interacting proteins. The CDIP1 interacting proteins were visualized by silver staining. (B) Co-immunoprecipitation of BAP31 by CDIP1. U2OS cells were treated with or without Brefeldin A (BFA) (1 μg ml−1) for 24 h. Proteins were cross-linked with DSP prior to protein extraction. (C)Mapping of CDIP1-BAP31 binding sites by GST pull-down with indicated GST-CDIP1 deletion proteins (aa 1-32, 1-74, 1-139 and 1–208) and GST-BAP31 deletion proteins (aa 1-129, 128-246 and 1–246) were generated for GST pull-down experiments. LCR: low complexity region; TM: transmembrane; and Coiled-Coil: coiled-coil domain. See also Figure S1 and Table S1 & S2.
Figure 2. The effect of ER stress-mediated CDIP1 induction on ER stress-induced apoptosis.
(A) ER stress induction of CDIP1 in U2OS cells (left panel) and MEFs (right panel). U2OS cells or Wt-MEFs were incubated with BFA (1 μg ml−1), Tm (0.5 μg ml−1) or Tg (0.1 μM) for indicated periods. Total RNAs were extracted and subjected to real-time quantitative PCR analysis using a specific primer set for CDIP1 and normalized to β-actin (MEFs) or GAPDH (U2OS). Data shown are mean ± s.d. (in triplicates and measured at the same time). Cell lysates were analyzed by western blotting with indicated antibodies. The blot was cut based on the size of proteins or stripped. (B) ER stress-mediated CDIP1 induction is regulated by ATF6 and IRE1 pathways. U2OS cells with knockdown using siCont., siATF6, siPERK and siIRE1 were treated with BFA (1 μg ml−1) or DMSO for 18 hours. Whole cell lysates were analyzed by western blotting with indicated antibodies. (C) CDIP1 deficiency precludes ER stress-induced apoptosis. Cells were treated with ER stress agents (BFA, Tm, or Tg) as described above for 18 h. Cell lysates were analyzed for the expression of indicated proteins. PARP cleavage was used as an apoptosis marker. Apoptosis was determined by TUNEL assay, followed by enumeration of dUTP-TMR red-positive cells by flow cytometry. Values shown are mean ± s.d. of three different experiments measured at the same time. P-value was calculated using Student's t-test. Left panel: Wt (CDIP1+/+) and CDIP1−/− MEFs. Right panel: U2OS cells with doxycycline-regulatable shCDIP1 were exposed with or without dox for 48 h, prior to ER stress treatment. See also Figure S1 & S2.
Loss of CDIP1 blocks ER stress-induced apoptosis
We next investigated whether ER stress-mediated CDIP1 induction plays any role in ER stress-mediated apoptosis. We first examined the role of CDIP1 in mediating ER stress-induced apoptosis in CDIP1-knockout and Wt MEFs. As expected, CDIP1 level was increased in response to ER stress agents in Wt MEFs and no detectable CDIP1 expression was observed in CDIP1-null MEFs (Figure 2C). However, CDIP1-null cells were still able to induce the expression of an ER stress response pathway marker BiP (Figure 2C), as well as phospho-eIF2α, ATF6 cleavage and XBP1 splicing (data not shown). As shown in Figure 2C, apoptosis induced by the treatment with ER stress agents (BFA, Tm and Tg) was significantly inhibited by ~50% in CDIP1−/− MEFs compared to Wt MEFs. Using Wt- and CDIP1-null MEFs, we also confirmed the effect of CDIP1 loss on ER stress-induced cell death by cell viability assay, demonstrating that CDIP1-null MEFs were more resistant to ER stress agents than Wt MEFs (data not shown). Similarly, stable knockdown of CDIP1 (shCDIP1#1 and shCDIP1#2) in U2OS human cancer cells decreased ER stress-mediated cell death by ~50% compared to that of control cells (shCont) (data not shown). Furthermore, acute suppression of CDIP1 by doxycycline (dox) in U2OS cells stably expressing dox-inducible CDIP1-shRNA (shCDIP1#1) also showed reduced ER stress-induced apoptosis via TUNEL (Figure 2C, right panel). To determine whether BAP31 collaborates with CDIP1 in ER stress-induced apoptosis, we depleted BAP31 expression in U2OS cells. We found that BAP31 depletion inhibited ER stress-induced apoptosis as similar to that seen in CDIP1-depleted or knockout cells (Figure S3A) while BAP31 knockdown did not affect the upregulation of CDIP1 levels in response to ER stress. These results suggest that CDIP1 and BAP31 cooperate to transduce the ER stress-induced apoptotic signal.
CDIP1-BAP31 complex promotes BAP31 binding to Bcl-2 and caspase-8/Bid and Bax activation during ER stress
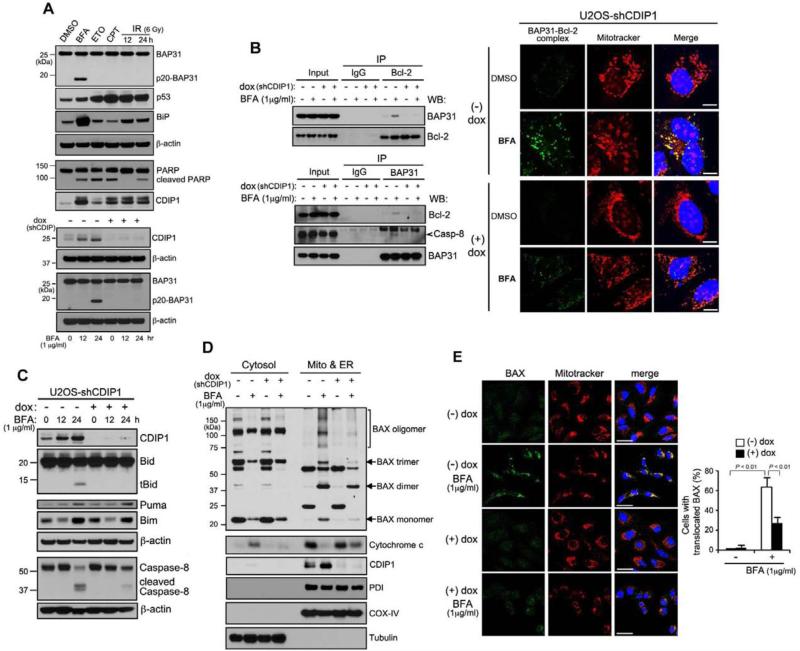
During apoptosis BAP31 is subjected to caspase-8-mediated cleavage into a p20BAP31 fragment that is known to function as a pro-apoptotic factor (Breckenridge et al., 2003; Rosati et al., 2010). We next sought to determine whether other types of stress rather than ER stress could also incite the cleavage of endogenous BAP31. U2OS cells were treated with several commonly used genotoxic stress agents, such as etoposide (ETO), camptothecin (CPT) and ionizing radiation (IR), and the cleavage of endogenous BAP31 was examined. As expected, ER stress-inducing agent BFA treatment revealed the cleavage of BAP31 and the generation of pro-apoptotic p20BAP31 fragment; however, other apoptotic stimuli did not show any cleaved BAP31 products as seen in DMSO-treated cells (Figure 3A, top panel). We next investigated to determine whether CDIP1 induction by ER stress is necessary for BAP31 cleavage and generation of p20BAP31. As shown in Figure 3A (bottom panel), in control U2OS cells upon BFA treatment, BAP31 was cleaved into p20BAP31. However, BAP31 was not cleaved into p20BAP31 upon BFA treatment in CDIP1-depleted cells (Figure 3A, bottom panel). These results suggest that CDIP1 binding to BAP31 is required for BAP31 cleavage and its resulting pro-apoptotic function in response to ER stress. We further examined if other types of apoptosis inducers, such as death ligand TNF-α and p53 overexpression, can provoke the cleavage of the BAP31 and if CDIP1 or BAP31 induction/expression is required for apoptosis mediated by TNF-α, p53 overexpression or IR using CDIP1 or BAP31 knockdowned cells. The treatment of TNF-α, p53 overexpression or IR did not reveal the cleavage of BAP31 (Figure S3B). However, CDIP1 knockdown appeared to inhibit apoptosis-mediated PARP cleavages, more significantly in p53-overexpressed and IR-treated cells, while BAP31 knockdown had no significant effects on PARP cleavages induced by three apoptotic conditions (Figure S3C). Together these data suggest that the cleavage of the endogenous BAP31 to p20BAP31 is ER stress sensitive and is dependent on CDIP1, while CDIP1 likely functions as a general regulator of stress provoked apoptosis.
Figure 3. CDIP1 promotes Bax oligomerization and mitochondrial translocation during ER stress.
(A) BAP31 cleavage is predominantly ER stress- and CDIP1-dependent. Top panel: U2OS cells were treated with BFA (1 μg ml−1), ETO (25 μM), CPT (500 nM) for 24 hours or γ-irradiation (6 Gy) for 12 and 24 hours. Bottom panel: U2OS cells with dox-regulatable shCDIP1 were treated with or without dox for 48 h prior to BFA treatment. Cell lysates were analyzed for the expression of indicated proteins. Same amounts of proteins were analyzed in two different gels (BAP31, p20BAP31, BiP, p53, β-actin; CDIP1 and PARP). The blot was cut based on the size of proteins. (B) CDIP1 depletion suppresses the BAP31 binding to Bcl-2 and procaspase-8 and BAP31-Bcl-2 interaction on the mitochondria upon ER stress. Left panel: U2OS cells with dox-regulatable shCDIP1 were treated with or without dox (1 μg ml−1) prior to BFA treatment (16 h), followed by Bcl-2 and BAP31 co-immunoprecipitation. Right panel: Representative images of DuoLink using BAP31 and Bcl-2 antibodies in U2OS-shCDIP1 cells upon BFA treatment. The detected endogenous BAP31 and Bcl-2 interaction by proximity ligation assay is shown as green dots. MitoTracker was used as mitochondria-specific marker. Merged images are also shown, and yellow color represents co-localization between BAP31 and Bcl-2 interaction (green) and MitoTracker (red) (scale bar, 10 μm). (C) The effect of CDIP1 depletion on ER stress-induced expression of BH3-only domain proteins. Using the same procedure described in (A), whole cell lysates were analyzed for the expression of indicated proteins. Same amounts of proteins were analyzed in three different gels (CDIP1 and Bid; Puma, Bim and β-actin; caspase-8 and β-actin). (D) ER stress-dependent Bax oligomerization and cytochrome c release requires CDIP1. U2OS cells with dox-regulatable shCDIP1 were grown with or without dox for 24 h and treated with BFA for additional 24 h. Cell lysates were then fractionated into two organellar fractions (cytosol and mitochondria/ER) and blotted with antibodies against BAX, cytochrome c, CDIP1, PDI (ER-specific marker), COX-IV (mitochondria-specific marker) and α-tubulin (cytosol marker). (E) The effect of CDIP1 depletion on Bax translocation to mitochondria upon ER stress. Using the same procedure described in (D), endogenous active form of Bax was visualized by immunostaining with anti-Bax antibody (N-20) by confocal microscopy. The strength of the laser (561nm) used to visualize the MitoTracker staining was determined at a level insufficient to fluoresce the endogenous RFP (data not shown). Merged images are also shown, and yellow color represents co-localization between Bax (green) and MitoTraker (red) (scale bar, 50 μm). The percentage of BAX translocation was determined by counting three different fields (30-50 cells/field). Values shown are mean ± s.d. of three different experiments. P-value was calculated using two-way ANOVA. See also Figures S3, S4 and S5.
It is now clear that BAP31 functions as a regulator of apoptosis through interactions with Bcl-2 or Bcl-XL and procaspase-8 (Ng et al., 1997). To investigate the mechanism by which CDIP1-BAP31 complex participates in ER stress-induced apoptosis, we elucidated whether CDIP1 binding to BAP31 is essential for BAP31-Bcl-2 and BAP31-procaspase-8 associations upon ER stress. We first examined the interaction between Bcl-2 and BAP31 with or without CDIP1 knockdown following BFA treatment. As predicted, when endogenous Bcl-2 was immunoprecipitated from BFA-treated U2OS cells, Bcl-2 binding to BAP31 was enhanced following BFA treatment (Figure 3B, left top panel). However, when CDIP1 was depleted, we detected little Bcl-2 binding to BAP31 in BFA-treated U2OS cells (Figure 3B, left top panel). Using reciprocal co-immunoprecipitation with BAP31 antibody, we also confirmed a decrease of Bcl-2-BAP31 and BAP31-procaspase-8 interactions in CDIP1-depleted cells (Figure 3B, left bottom panel). Bcl-2 is localized to the mitochondria and ER membrane (Krajewski et al., 1993). We next examined the possibility that BAP31 interacts with both Bcl-2 in the mitochondria and Bcl-2 on the ER upon ER stress. We first used the DuoLink technology to detect and visualize BAP31-Bcl-2 association under ER stressed condition. By using antibodies toward BAP31 and Bcl-2, we were able to confirm positive colocalization dots on the mitochondria upon ER stress, indicated by the overlapping signals with MitoTracker; however, BAP31-Bcl-2 interaction signals were significantly decreased by CDIP1 knockdown (Figure 3B, right panel). We next used a set of Bcl-2 expression vectors in which Bcl-2 is targeted specifically to either the ER or the mitochondrial membrane as previously reported (Wang et al., 2001). We confirmed that ER-targeted Bcl-2 and mitochondria-target Bcl-2 were localized to the appropriate organelles, ER and mitochondria, respectively, while we observed that wt-Bcl-2 was ubiquitously distributed in the ER and mitochondria (Figure S4A). Using immunoprecipitation of these cells with targeted Bcl-2 expression, we found that BAP31 was associated with both ER-targeted Bcl-2 and mitochondria-targeted Bcl-2 proteins (Figure S4B).
During ER stress-induced apoptosis Bax proteins locate to the ER membrane and the mitochondria and undergo conformational changes resulting in oligomerization (Zong et al., 2003). It is now clear that activation of Bax by BH3-only domain molecules is essential for mitochondrial apoptosis (Tait and Green, 2010). To determine whether ER stress-mediated apoptosis requires the induction of BH3-only molecule(s) and whether the depletion of CDIP1 induction upon ER stress has any effect on the response of BH3-only proteins, U2OS cells were treated with ER stress agent BFA, and the expression of BH3-only molecules, including Bid, Puma, and Bim, was measured with and without CDIP1 knockdown. As shown in Figure 3C, ER stress induced the expression of Bim, Puma and truncated Bid (tBid). Interestingly, CDIP1 knockdown abolished ER stress-mediated truncation of Bid but had no effect on Bim and Puma induction (Figure 3C). Moreover, ER stress-induced caspase-8 activation was abolished by CDIP1 depletion (Figure 3C). Consistently, inhibition of caspase-8 activation with caspase-8-specific inhibitor Z-IETD-FMK blocked ER stress-mediated apoptotic PARP cleavage (data not shown). Together, these findings demonstrate that in addition to the Bcl-2-regulated apoptotic pathway, CDIP1 mediates ER stress-induced apoptosis through caspase-8/Bid pathway.
To further examine the downstream proapoptotic effector Bax upon CDIP1-mediated activation of the BAP31-Bcl-2 and caspase-8/Bid pathways, we next investigated whether CDIP1 is required for ER stress-induced Bax activation/oligomerization. CDIP1 depleted (dox +) or control (dox −) U2OS cells were treated with ER-stress agent BFA for 24 hours. After BFA treatment, bismaleimidohexane (BMH) was used to cross-link the oligomerized Bax proteins. Then, cells were fractionated into cytosolic and organellar (mitochondria and ER) fractions and examined for Bax subcellular localization and oligomerization. As shown in Figure 3D, Bax was mainly found in the cytosol in the absence of stress. However, upon BFA treatment Bax levels were significantly decreased in the cytosol, regardless of CDIP1 status, with concomitant increase in mitochondrial and ER fractions (Figure 3D). More importantly, the extent of oligomerized Bax was also increased in the mitochondrial and ER fractions although CDIP1 depletion significantly inhibited ER stress-induced oligomerization of Bax in the mitochondria and ER (Figure 3D). Consistent with this result, upon BFA treatment cytochrome c release to the cytosol from the mitochondria was accompanied with the accumulation of oligomerized Bax, which was blocked by CDIP1 knockdown. To further define the subcellular localization of Bax, cells were sub-fractionated into heavy membrane (Mitochondria + ER) and light membrane (predominantly ER) fractions. As shown in Figure S5A, there is an increase of oligomerized Bax predominantly in the mitochondrial fraction in response to ER stress but diminished in the mitochondrial fraction of CDIP1-depleted cells. Similar results were obtained using CDIP1−/− and Wt MEFs (Figure S5B, left panel). To further confirm that CDIP1 is required for BAX translocation from cytosol to mitochondria, we examined Bax activation in BFA-treated cells by immunofluorescence via confocal microscopy using an antibody specific for active form of Bax detection. Following BFA treatment, U2OS cells or Wt MEFs appeared to show punctate mitochondrial staining of active Bax (Figure 3E and Figure S5B, right panel). However, CDIP1 knockdown in U2OS cells or CDIP1−/− MEFs efficiently suppressed BFA-induced Bax accumulation in mitochondria (Figures 3D, 3E and S5B). Together, these results indicate that CDIP1 is essential for Bax activation/oligomerization via both BH3-only domain protein Bid activation and the BAP31-Bcl-2 complex formation during ER stress-induced apoptosis and subsequent cytochrome c release.
CDIP1 deficiency leads to the protection against ER stress-induced cell death in vivo
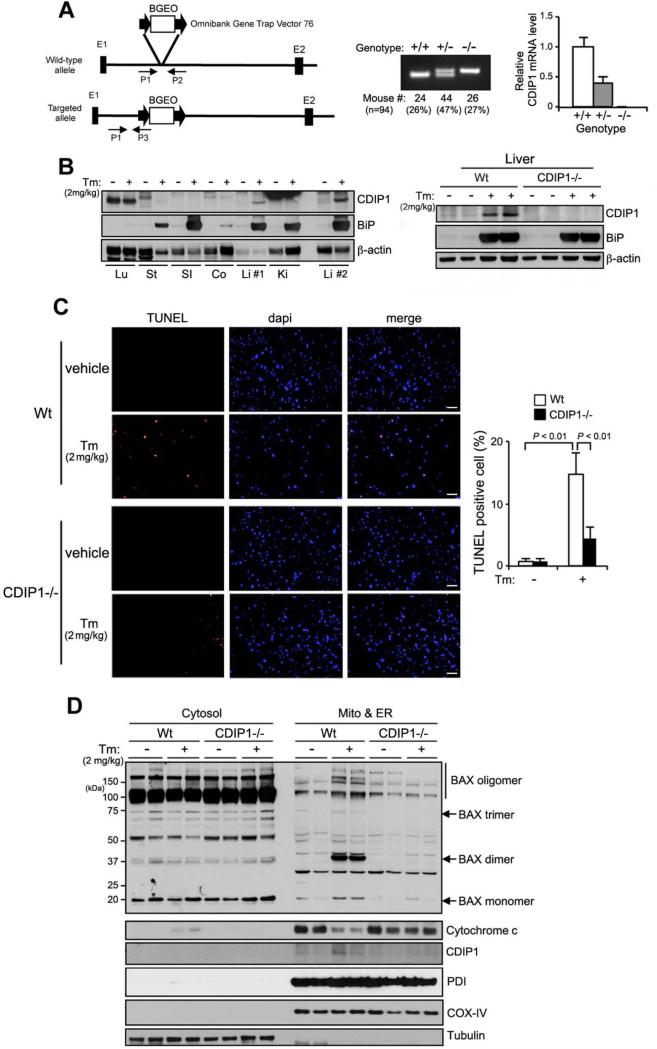
So far, we demonstrated that CDIP1-BAP31 complex acts in a coherent feedback upon ER stress, modulating pro-apoptotic response through mitochondrial death machinery. Since we discovered and explored the role of CDIP1-BAP31 complex in ER stress response pathway in vitro, we investigated whether CDIP1 also plays any role in ER stress response in vivo using a CDIP1-knockout mouse. CDIP1 knockout mice were generated using ES cells carrying a OmniBank gene trap mutation (Zambrowicz et al., 1998) in the first intron of the CDIP1 gene (Figure 4A). The knockout was confirmed by genotyping and measurement of CDIP1 mRNA levels using quantitative real-time PCR assay (Figure 4A). To examine levels of CDIP1 expression in vivo with or without ER stress, Wt mice were treated with Tm (2 mg kg−1) or 150 mM dextrose (vehicle) by intraperitoneal administration. As shown in Figure 4B, CDIP1 was primarily induced in the liver in response to ER stress agent Tm, and as expected, ER stress-mediated CDIP1 induction was absent in CDIP1-null liver tissue. To elucidate the physiological role of CDIP1 in ER stress-induced apoptosis, Wt and CDIP1-deficient littermates (4-6 weeks old) were exposed to Tm for 18 hours. Liver tissues were collected and analyzed for cell death by TUNEL-based assays. There was almost complete abrogation of ER stress-induced apoptosis in the liver of CDIP1-null animals after Tm exposure (Figure 4C), while the population of TUNEL positive cells was markedly higher in the Wt liver after Tm exposure. We have shown that CDIP1 induction contributes to Bax activation/oligomerization and subsequent cytochrome c release in vitro (Figures 3D, 3E). We next examined whether CDIP1 deficiency could affect the ER stress-induced Bax oligomerization in CDIP1-null liver. As shown in Figure 4D, Bax oligomerization was increased in the mitochondrial and ER fractions following Tm treatment along with cytochrome c release to cytosol in the Wt liver but not CDIP1-null liver. All of these data together in the CDIP1 knockout animals confirmed that in response to ER stress signals, CDIP1-BAP31 complex recruits Bcl-2, leading to Bax activation/oligomerization and subsequent stimulation of cytochrome c release and cell death.
Figure 4. CDIP1 deficiency leads to impaired ER stress-induced cell death in mouse.
(A) A gene trap vector was inserted into the first intron of CDIP1 (left panel). PCR assay was shown using tail genomic DNA from three different genotypes: Wt (+/+), CDIP1 heterozygous (+/−) and CDIP1-null (−/−) littermates. Relative CDIP1 mRNA levels were measured by real-time quantitative PCR analysis (right panel). (B) CDIP1 is predominantly induced in liver tissue upon ER stress. Wt or CDIP1-null mice were intraperitoneally administrated with Tm (2 mg kg−1) or vehicle (150 mM dextrose). After 24 hours, tissues were removed and subjected to western blot analysis: Lu: lung, St: stomach, SI: small intestine, Co: colon, Li #1 & Li #2: liver, and Ki: kidney. (C) CDIP1-deficient mouse is resistant to ER stress-induced apoptosis. Wt- or CDIP1-null mice were injected with Tm as described above. Frozen sections of liver from vehicle- or Tm- treated mice were subjected to TUNEL staining to detect apoptotic cells (scale bar, 50 μm). The percentage of TUNEL-positive cells was determined by counting four different sections (approximate 100 cells/section) per liver of four different mice. Values shown are mean ± s.d. (n=4). P-value was calculated using two-way ANOVA. (D) ER stress-dependent Bax oligomerization and cytochrome c release requires CDIP1 in vivo. Liver tissues from vehicle- or Tm-administered Wt and CDIP1-null mice were fractionated and analyzed for the expression of the indicated proteins. See also Figure S5.
DISCUSSION
Here, we identify a novel regulatory mechanism of ER stress-induced cell death pathway. We discovered a pro-apoptotic p53 target CDIP1 as a new player in ER stress-induced apoptosis. Through a proteomics approach, we identified the ER protein BAP31 as an interacting partner of CDIP1. We show that during ER stress CDIP1 is induced with increased binding to BAP31. This CDIP1-BAP31 complex is critical for the ER stress-mediated cell death and for the formation of the BAP31-Bcl-2 complex. As a result of the CDIP1-BAP31 interaction, Bax-dependent mitochondrial apoptotic pathway is triggered through the subsequent Bax activation/oligomerization, ultimately leading to cell death. More importantly, we further demonstrate that CDIP1 deficiency in mice substantially impairs Bax-dependent mitochondrial apoptotic pathway and ER stress-induced apoptosis.
Accumulating evidence clearly indicates that there are physical, biochemical and functional links between the ER and the mitochondria through components of proteins and protein complexes of the ER-mitochondria interface (Friedman et al., 2011; Grimm, 2012). Similarly here, we identify CDIP1-BAP31 complex as a new critical component in transducing the ER stress-mediated apoptotic signal between the ER and the mitochondria. Furthermore, we demonstrate that CDIP1 is essential for BAP31-Bcl-2 interaction on the mitochondria and BAP31 cleavage into p20BAP31, and as a result, the lack of CDIP1 impairs the downstream signaling of BAX activation/oligomerization in the mitochondria for apoptosis. We also demonstrate that CDIP1 is required for BH3-only protein Bid-mediated Bax activation resulting in mitochondrial apoptosis, suggesting that both mitochondrial apoptosis pathways are involved in ER stress-induced apoptosis.
Importantly, our in vivo studies reveal that CDIP1 expression is dramatically up-regulated upon ER stress and further stimulating apoptosis in the liver suggesting functional in vivo relevance of the CDIP1 pathway. Given the fact that ER-stress mediated cellular responses are becoming increasingly significant in the pathogenesis of various disorders including cancer, further studies into the function and downstream effectors of CDIP1 are warrant. Importantly, analysis of recently available high-throughput sequencing data (http://www.cbioportal.org/public-portal/index.do) of patient samples with various types of cancers point to the presence of distinguished genomic alterations in both CDIP1 and BAP31 in subsets of patients with lymphoma (11.1% for BAP31) and sarcomas (7.7% for BAP31; and 5.8% for CDIP1), as well as epithelial cancers (such as invasive breast, bladder, lung and ovarian cancer). Together our data represents a novel mechanism of CDIP1 function for establishing an ER-mitochondrial cross-talk for ER stress-mediated apoptosis signaling through two prominent regulatory routes of mitochondrial apoptosis pathways: caspase-8/Bid-mediated Bax activation and CDIP1-BAP31-Bcl-2→BAX activation/oligomerization.
EXPERIMENTAL PROCEDURES
CDIP1 complex purification and mass spectrometry analysis
A detailed TAP purification procedure has been described previously (Nakatani and Ogryzko, 2003). Briefly, FLAG and HA double-tagged CDIP1 were cloned into pTRE2pur vector (Clontech). U2OS cells were used to generate a stable cell line with dox-inducible HA-FLAG-CDIP1 expression. A dox-inducible HA-FLAG stable U2OS cell line was also generated as control. FLAG-HA-CDIP1 and the proteins complex were sequentially immunoprecipitated with anti-FLAG M2 monoclonal antibody (mAb)–conjugated agarose beads followed by anti-HA 12CA5 mAb-conjugated agarose beads. A small portion of FLAG-HA double affinity-purified materials were separated by 4 to 12% gradient SDS-PAGE and were silver stained. Then the affinity-purified proteins were precipitated with ProteoExtract® Protein Precipitation Kit (Calbiochem) and analyzed by the Mass Spectrometry Facility at Harvard Medical School.
Subcellular fractionation
Cells were resuspended in fractionation buffer A (250 mM sucrose, 20 mM Hepes, 10 mM KCl, 1.5 mM MgCl2, 1 mM EDTA, 1 mM EGTA, protease inhibitor mix, pH 7.5) on ice for 30 min. Cells were disrupted by passing through 26-gauge needles 10 times. Cell lysates were centrifuged at 720 g for 5 min to obtain the nuclei fraction. The supernatant was centrifuged at 15,000 g for 5 min. The pellet was saved as the ER and mitochondria fraction. The supernatant was centrifuged at 14000 rpm for 5 min. The supernatants were collected as the cytosol fraction. The ER and mitochondria fraction was washed once again in fractionation buffer A by centrifugation at 720 g for 5 min to remove any contaminating particles and then recovered by centrifugation at 15,000 g for 5 min. The isolated ER and mitochondria were resuspended in fractionation buffer A for immunoblotting analysis. Fractionation of heavy and light membrane fractions was described previously (Zong et al., 2003).
Generation of CDIP1-null mutant mice
The null mutant mice with complete deletion of 5730403B10Rik gene (mouse CDIP1 gene) were generated using a gene trap technique (Zambrowicz et al., 1998). The mutant mice were generated using an embryonic stem cell clone (IST12371A7; Texas A&M Institute for Genomic Medicine) carrying mutant CDIP1 allele disrupted by the insertion of a gene trap vector with a lacZ (β-galactosidase)-neomycin resistance fusion cassette (β-gal, Omnibank Vector 76) in the first intron. The insertion site, as denoted with an asterisk(*), was confirmed by genomic sequence, which is available upon request. The cloned ES cell was used to generate CDIP1 mutant mice with a FVB background (backcrossed to FVB/N for six generations), which were verified by PCR amplification of the mutant gene in tail DNA from progeny.
Supplementary Material
HIGHLIGHTS.
A pro-apoptotic p53 target CDIP1 regulates ER stress-induced apoptosis.
CDIP1 and BAP31 interact upon ER stress to regulate mitochondrial apoptosis pathway.
CDIP1-null mice show impaired response to ER stress-mediated apoptosis.
CDIP1 mediates an apoptotic signaling link between ER and mitochondria upon stress.
ACKNOWLEDGMENTS
LC-MS/MS analysis was performed by Taplin Biological Mass Spectrometry Facility of Harvard Medical School. We thank S. Aaronson and members of the Lee laboratory for helpful advice and suggestions during the project. This work was partially supported by NIH grants CA142805, CA080058, and CA127247. TN was supported by a fellowship for research abroad from Japan Society for the Promotion of Science and is a recipient of AACR-Bristol-Myers Squibb Oncology Scholar-in-Training. SYH was supported by the Korean Government Research Foundation grant (KRF-2008-357-E00023).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- Breckenridge DG, Nguyen M, Kuppig S, Reth M, Shore GC. The procaspase-8 isoform, procaspase-8L, recruited to the BAP31 complex at the endoplasmic reticulum. Proc Natl Acad Sci U S A. 2002;99:4331–4336. doi: 10.1073/pnas.072088099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breckenridge DG, Stojanovic M, Marcellus RC, Shore GC. Caspase cleavage product of BAP31 induces mitochondrial fission through endoplasmic reticulum calcium signals, enhancing cytochrome c release to the cytosol. J Cell Biol. 2003;160:1115–1127. doi: 10.1083/jcb.200212059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown L, Ongusaha PP, Kim HG, Nuti S, Mandinova A, Lee JW, Khosravi-Far R, Aaronson SA, Lee SW. CDIP, a novel proapoptotic gene, regulates TNFalpha-mediated apoptosis in a p53-dependent manner. EMBO J. 2007;26:3410–3422. doi: 10.1038/sj.emboj.7601779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen L, Willis SN, Wei A, Smith BJ, Fletcher JI, Hinds MG, Colman PM, Day CL, Adams JM, Huang DC. Differential targeting of prosurvival Bcl-2 proteins by their BH3-only ligands allows complementary apoptotic function. Mol Cell. 2005;17:393–403. doi: 10.1016/j.molcel.2004.12.030. [DOI] [PubMed] [Google Scholar]
- Friedman JR, Lackner LL, West M, DiBenedetto JR, Nunnari J, Voeltz GK. ER tubules mark sites of mitochondrial division. Science. 2011;334:358–362. doi: 10.1126/science.1207385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimm S. The ER-mitochondria interface: the social network of cell death. Biochimica et biophysica acta. 2012;1823:327–334. doi: 10.1016/j.bbamcr.2011.11.018. [DOI] [PubMed] [Google Scholar]
- Hetz C, Bernasconi P, Fisher J, Lee AH, Bassik MC, Antonsson B, Brandt GS, Iwakoshi NN, Schinzel A, Glimcher LH, et al. Proapoptotic BAX and BAK modulate the unfolded protein response by a direct interaction with IRE1alpha. Science. 2006;312:572–576. doi: 10.1126/science.1123480. [DOI] [PubMed] [Google Scholar]
- Hotamisligil GS. Endoplasmic reticulum stress and the inflammatory basis of metabolic disease. Cell. 2010;140:900–917. doi: 10.1016/j.cell.2010.02.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwasawa R, Mahul-Mellier AL, Datler C, Pazarentzos E, Grimm S. Fis1 and Bap31 bridge the mitochondria-ER interface to establish a platform for apoptosis induction. EMBO J. 2011;30:556–568. doi: 10.1038/emboj.2010.346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krajewski S, Tanaka S, Takayama S, Schibler MJ, Fenton W, Reed JC. Investigation of the subcellular distribution of the bcl-2 oncoprotein: residence in the nuclear envelope, endoplasmic reticulum, and outer mitochondrial membranes. Cancer research. 1993;53:4701–4714. [PubMed] [Google Scholar]
- Li J, Lee B, Lee AS. Endoplasmic reticulum stress-induced apoptosis: multiple pathways and activation of p53-up-regulated modulator of apoptosis (PUMA) and NOXA by p53. J Biol Chem. 2006;281:7260–7270. doi: 10.1074/jbc.M509868200. [DOI] [PubMed] [Google Scholar]
- Nakatani Y, Ogryzko V. Immunoaffinity purification of mammalian protein complexes. Methods Enzymol. 2003;370:430–444. doi: 10.1016/S0076-6879(03)70037-8. [DOI] [PubMed] [Google Scholar]
- Ng FW, Nguyen M, Kwan T, Branton PE, Nicholson DW, Cromlish JA, Shore GC. p28 Bap31, a Bcl-2/Bcl-XL- and procaspase-8-associated protein in the endoplasmic reticulum. J Cell Biol. 1997;139:327–338. doi: 10.1083/jcb.139.2.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng FW, Shore GC. Bcl-XL cooperatively associates with the Bap31 complex in the endoplasmic reticulum, dependent on procaspase-8 and Ced-4 adaptor. J Biol Chem. 1998;273:3140–3143. doi: 10.1074/jbc.273.6.3140. [DOI] [PubMed] [Google Scholar]
- Puthalakath H, O'Reilly LA, Gunn P, Lee L, Kelly PN, Huntington ND, Hughes PD, Michalak EM, McKimm-Breschkin J, Motoyama N, et al. ER stress triggers apoptosis by activating BH3-only protein Bim. Cell. 2007;129:1337–1349. doi: 10.1016/j.cell.2007.04.027. [DOI] [PubMed] [Google Scholar]
- Rodriguez DA, Zamorano S, Lisbona F, Rojas-Rivera D, Urra H, Cubillos-Ruiz JR, Armisen R, Henriquez DR, Cheng EH, Letek M, et al. BH3-only proteins are part of a regulatory network that control the sustained signalling of the unfolded protein response sensor IRE1alpha. The EMBO journal. 2012;31:2322–2335. doi: 10.1038/emboj.2012.84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosati E, Sabatini R, Rampino G, De Falco F, Di Ianni M, Falzetti F, Fettucciari K, Bartoli A, Screpanti I, Marconi P. Novel targets for endoplasmic reticulum stress-induced apoptosis in B-CLL. Blood. 2010;116:2713–2723. doi: 10.1182/blood-2010-03-275628. [DOI] [PubMed] [Google Scholar]
- Scorrano L, Oakes SA, Opferman JT, Cheng EH, Sorcinelli MD, Pozzan T, Korsmeyer SJ. BAX and BAK regulation of endoplasmic reticulum Ca2+: a control point for apoptosis. Science. 2003;300:135–139. doi: 10.1126/science.1081208. [DOI] [PubMed] [Google Scholar]
- Tabas I, Ron D. Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress. Nat Cell Biol. 2011;13:184–190. doi: 10.1038/ncb0311-184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tait SW, Green DR. Mitochondria and cell death: outer membrane permeabilization and beyond. Nat Rev Mol Cell Biol. 2010;11:621–632. doi: 10.1038/nrm2952. [DOI] [PubMed] [Google Scholar]
- Walter P, Ron D. The unfolded protein response: from stress pathway to homeostatic regulation. Science. 2011;334:1081–1086. doi: 10.1126/science.1209038. [DOI] [PubMed] [Google Scholar]
- Wang B, Heath-Engel H, Zhang D, Nguyen N, Thomas DY, Hanrahan JW, Shore GC. BAP31 interacts with Sec61 translocons and promotes retrotranslocation of CFTRDeltaF508 via the derlin-1 complex. Cell. 2008;133:1080–1092. doi: 10.1016/j.cell.2008.04.042. [DOI] [PubMed] [Google Scholar]
- Wang NS, Unkila MT, Reineks EZ, Distelhorst CW. Transient expression of wild-type or mitochondrially targeted Bcl-2 induces apoptosis, whereas transient expression of endoplasmic reticulum-targeted Bcl-2 is protective against Bax-induced cell death. The Journal of biological chemistry. 2001;276:44117–44128. doi: 10.1074/jbc.M101958200. [DOI] [PubMed] [Google Scholar]
- Zambrowicz BP, Friedrich GA, Buxton EC, Lilleberg SL, Person C, Sands AT. Disruption and sequence identification of 2,000 genes in mouse embryonic stem cells. Nature. 1998;392:608–611. doi: 10.1038/33423. [DOI] [PubMed] [Google Scholar]
- Zong WX, Li C, Hatzivassiliou G, Lindsten T, Yu QC, Yuan J, Thompson CB. Bax and Bak can localize to the endoplasmic reticulum to initiate apoptosis. J Cell Biol. 2003;162:59–69. doi: 10.1083/jcb.200302084. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.