Abstract
BACKGROUND
A new cryopreservation bag for hematopoietic cell transplantation requires validation as a safe alternative to the bag currently being used in the laboratory.
STUDY DESIGN AND METHODS
The new bag was validated using both laboratory and clinical criteria. Laboratory validation proceeded using paired samples of mononuclear cells processed using standard procedures. Cells cryopreserved in the new and old bags were compared for viability, cell counts, CD34 enumeration, colony-forming unit assays, and bag integrity. After completion of laboratory investigations, engraftment with the new bags was followed and compared to historical engraftment using the old bags.
RESULTS
There were no significant differences between the old and new bags detected using laboratory studies. Bag integrity was equivalent. The validation data suggested impaired cell function after cryopreservation in the new bags, but there were no significant differences in engraftment potential using either material. Days to engraftment was longer using the new bags, but statistical analysis revealed an association with CD34 dose and not with cryopreservation bag type.
CONCLUSION
The new bags were noninferior to the old bags. A change in cryopreservation bag type may appear to affect cell function and potentially affect engraftment. Multiple analyses may be needed to understand the effect of cell processing changes.
The Food and Drug Administration (FDA) and Foundation for the Accreditation of Cellular Therapies require blood and marrow stem cell laboratories to process cellular products according to specific regulations that apply current good tissue practice.1,2 These regulations stipulate that a change to a process shall be verified and validated by the laboratory before the implementation of the practice change. Validation and verification serves to ensure that a system change does not create an adverse impact anywhere in the operation.1 Furthermore, as a result of market forces, material and production changes might present with or without notice to the end user. Laboratory processes may need to adapt not only to patient care challenges, but also to economic diversity. One process change that a laboratory may encounter is converting from one cryopreservation storage material to another.
Our blood and marrow transplant (BMT) program faced the discontinuation of the current cryopreservation bags used to store peripheral blood progenitor cells (PBPCs), marrow, and donor lymphocytes. After selection of the new cryopreservation bag, a validation plan was drafted and approved. Validation was performed according to the laboratory quality plan. The complete process of validation and verification spanned several years as data were collected and studied. The transplant program medical director, laboratory director, and laboratory quality supervisor all approved the validation plan and participated in data and quality review.
MATERIALS AND METHODS
The validation plan for the Cryocyte (new) freezing bags (Baxter, Deerfield, IL) consisted of parallel comparisons of mononuclear cell (MNC) apheresis samples that were cryopreserved either in the currently used Cryogenic storage container (old) CPL-99 bags (Charter Medical, Winston-Salem, NC) versus the new bags. The new bags were selected based on near unanimous results of a survey of large BMT program processing laboratories, all of which reported excellent transplant outcomes using the new material. Use of the new bag material was associated with historical transplant success at multiple centers and historical FDA approval of the material. The old bags had demonstrated adequate cryopreservation of cells based on historical clinical outcomes of our BMT program compared to national data. Validation of the new cryopreservation bag proceeded in two phases defined as laboratory and clinical.
Laboratory
Three MNC products collected by standard apheresis methods were obtained from an independent supplier (BRT Laboratories, Baltimore, MD). The MNC donors were not cytokine mobilized. Products were processed within 24 hours of collection. Each product was split evenly in equal volumes. Laboratory data on the equivolume products included complete blood count, total white blood cell (WBC) count (Beckman Coulter, Fullerton, CA), volume, total CD34-positive cell enumeration (Beckman Coulter), viability by trypan blue exclusion (Sigma, St Louis, MO), and colony-forming unit (CFU) assays for CFU–granulocyte-macrophage (CFU-GM), burst and colony-forming unit–erythroid (BFU-E; CFU-E), and CFU–granulocyte-erythroid-macrophage-megakaryocyte (CFU-GEMM; Stem Cell Technologies, Vancouver, British Columbia, Canada). CFU assays were plated based on the viable cell count.
All steps of cryopreservation were performed according to laboratory standard operating procedures in parallel using either the old or the new bags. The controlled rate freezer adjusted for a 1°C/min rate of temperature decrease with compensation. Products were cryopreserved from 2 to 5 days and then thawed according to standard operating procedures in a 37°C water bath. After thaw, the products were washed and sampled for the same laboratory data described previously. Cells sampled from all bags before and after cryopreservation were expected to grow CFUs and remain viable. The bags were to maintain integrity throughout processing. Due to the small sample size, parallel processing was not expected to yield data that established superiority, but rather, parallel processing was expected to show noninferiority as acceptable for implementation.
Clinical
After completion of laboratory validation and implementation of the new bags, a historical cohort of patients was compared prospectively to new patients as they presented through the BMT program. Historical patients transplanted using the old bags were compared to patients transplanted using the new bags. All patients received autologous PBPC transplants. Data compilation spanned 2.3 years and included days to engraftment, cell dose, and a number of additional clinical variables. The critical clinical endpoint was defined as days to engraftment. Engraftment was defined as the first day when the absolute neutrophil count (ANC) was greater than 500 and remained above 500 for 3 consecutive days.
Statistical analysis
The goal of the statistical analyses was to explore the relationship between bag type and days to engraftment, taking into account other possible factors impacting engraftment time. A two-sample t test was initially performed to test if there was a significant difference in the days to engraftment between bags. A general linear model was used to examine the impact of using the new type of blood bag while adjusting for the other covariates. Covariates included in the modeling were patient age at BMT, sex, diagnosis, and CD34+ cell counts. Diagnosis type was collapsed into four main categories: acute myelogenous leukemia, non-Hodgkin’s lymphoma, multiple myeloma, and other. Assumption of normality was questionable for CD34+ counts (several outliers and skewed values; Shapiro-Wilk p < 0.001); therefore, CD34+ counts were used in the analyses after log transformation. Model assumptions were explored using residual diagnostics and plots, which indicated modeling adequacy. Significant was considered to be resulting in a p value of less than 0.05. All analyses were performed with computer software (SAS v9.1.3, SAS Institute, Cary, NC).
RESULTS
Laboratory
The WBC recovery was 83.9% for the old bags (before, 3.1 × 109 total cells [range, 2.0 × 109–4.5 × 109]; after, 2.6 × 109 [range, 1.6 × 109–3.8 × 109]) and 76.7% for the new bags (before, 3.0 × 109 total cells [range, 2.0 × 109–4.0 × 109]; after, 2.3 × 109 [1.3 × 109–3.1 × 109]). The CD34 recovery was 100% for the old bags (before, 5.0 × 109 total cells [range, 1.9 × 109–8.1 × 109]; after, 5.1 × 109 [range, 2.3 × 109–9.8 × 109]) and 80% for the new bags (before, 4.0 × 109 total cells [range, 1.0 × 109–6.5 × 109]; after, 3.2 × 109 [range, 1.1 × 109–6.3 × 109]). The recovered viable cells was 81% for the old bags (before, 98% [range, 98%–98%]; after, 79% [range, 67%–85%]) and 76% for the new bags (before, 98% [range, 97%–99%]; after, 74% [range, 59%–81%]). Recovery was calculated as total cells (WBCs × volume) after cryopreservation/total cells before cryopreservation. Postthaw CFU could be consistently detected. However, the colony assay data were variable and difficult to interpret beyond the detection of growth. Individual results showed that some colony growth using the new material was greater than colony growth after using the old material; the converse was also observed. CFU-GEMM were too few to consider. There were no CFU-GEMM produced before cryopreservation at 2.5 × 104/mL cell concentration and no more than one colony grown at any concentration; therefore, without a concentration curve, postthaw data for both materials were difficult to interpret. CFU-GM results were limited. Most interpretations of the colony data were weighted by BFU-E growth. However, the mean results using the new material were consistently lower than those produced using the old bag material, even though individual data point comparisons showed a slightly more balanced result.
All bags remained intact without breach or rupture during the laboratory validation. The new material was implemented soon thereafter and engraftment results were monitored prospectively.
Clinical
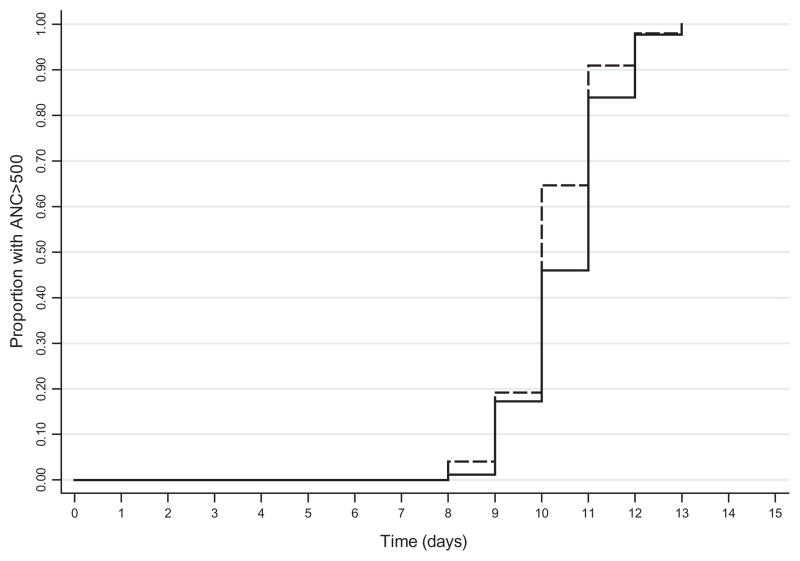
The viability after cryopreservation was 72.3% (n = 99) for the old bag and 72.5% (n = 87) for the new bag. The days to engraftment was plotted for patients receiving PBPC cryopreserved in old and new bags (Fig. 1). From bivariate analyses (Table 1), the products stored in the old bags required significantly fewer days to reach ANC of more than 500 (old bag median number of days, 10 vs. new bag median number of days, 11; Wilcoxon p = 0.0313). There was a significant difference between the proportions of diagnosis type groups due to a higher proportion of multiple myeloma cases and lower proportion of acute myelogenous leukemia cases in the new bag group. The other two diagnosis groups had approximately the same proportion for both bag types (df = 3, p = 0.041). The remaining characteristics did not have a significant difference in distribution between the two series. Since patient characteristics such as age, sex, diagnosis, and CD34+ counts may also have an impact on the engraftment time, these were included in a regression model to control their influence in examining the relationship between bag type and days to engraftment. In adjusted analyses, the bag type was no longer a significant factor (p = 0.289) in days to reaching ANC of greater than 500 (Table 2).
Fig. 1.
Days to engraftment for patients receiving autologous PBPCs cryopreserved in old and new bags. (—) New bag; (----) old bag.
TABLE 1.
Patient characteristics: statistics, frequencies, and time to engraftment by blood bag type (n = 186)
| Old bag (n = 99) | New bag (n = 87) | |
|---|---|---|
| Characteristic | ||
| Sex† | ||
| Male | 47 (47) | 53 (61) |
| Female | 52 (53) | 34 (39) |
| Age (years) | 53.1 (58.1) | 54.0 (51.0) |
| Diagnosis | ||
| AML | 18 (18) | 5 (6) |
| NHL | 28 (28) | 26 (30) |
| MM | 30 (31) | 38 (43) |
| Other | 23 (23) | 18 (21) |
| CD34+ (number of cells/kg infused ×106) | 6.23 (38.14) | 6.00 (19.2) |
| Number of collections infused | 1 (9) | 1 (6) |
| Time to engraftment | ||
| Number of days to ANC > 500† | 10 (5) | 11 (5) |
| Number of days to ANC > 1000† | 10 (6) | 11 (4) |
Data are reported as number (%) or median (range).
p < 0.05; chi-square or Fisher’s exact test used for categorical variables; t test or Wilcoxon rank-sum test used for continuous variables.
AML = acute myelogenous leukemia; MM = multiple myeloma; NHL = non-Hodgkin’s lymphoma.
TABLE 2.
Regression analysis for comparison of bags for time to ANC of more than 500 (n = 186)
| Covariate | β | (95% CI) | p value |
|---|---|---|---|
| Sex (female vs. male) | 0.023 | (−0.247 to 0.294) | 0.863 |
| Age | −0.001 | (−0.013 to 0.011) | 0.850 |
| Diagnosis* | |||
| Other | −0.565 | (−0.949 to −0.180) | 3 df, p < 0.0001 |
| AML | −0.660 | (−1.116 to −0.203) | 0.004 |
| NHL | −1.004 | (−1.349 to −0.658) | 0.005 |
| MM | 0 | <0.0001 | |
| log(CD34+) (number of cells/kg infused × 106) | −0.769 | (−1.020 to −0.519) | <0.0001 |
| Bag type (old vs. new) | 0.147 | (−0.126 to 0.420) | 0.289 |
All diagnosis categories are being compared to the “MM” category.
AML = acute myelogenous leukemia; MM = multiple myeloma; NHL = non-Hodgkin’s lymphoma.
Although not significant, the results suggested that the old bags resulted in a slightly shorter engraftment time when compared to the new bags (difference of old vs. new, 1 day). The slight difference in days to engraftment could be explained in part due to the relationship between CD34+ cell counts and engraftment time. In bivariate analysis there was a significant negative relationship between CD34+ counts and days to ANC of greater than 500 (Spearman ρ = −0.28; p < 0.001). Once CD34+ counts were included in the regression model with bag type for days to ANC of greater than 500, the effect for bag type became nonsignificant (while the overall F test statistic went from F = 4.27 to F = 10.78). There was no evidence of a bag type by CD34+ count interaction (p > 0.10 in models). In adjusted regression analyses using all of the covariates: age, sex, diagnosis, bag type, and CD34+ counts, only the CD34+ counts remained significant for days to ANC of greater than 500 (β for ln[CD34+ counts] = −0.77; p < 0.001). Regression analyses repeated using days to reach an ANC of greater than 1000 showed similar results (data not shown).
Bag breaks were compared between old (six breaks per 295 bags) and new (two breaks per 310 bags). There was no statistical difference in breakage using a chi-square test. However, there was a trend toward improvement in breakage with the new bags. No breaks have occurred with the new bags in the past 6 years.
DISCUSSION
Validation is defined as establishing documented evidence that provides a high degree of assurance that a specific process will consistently produce a product meeting its predetermined specifications and quality attributes.3 For a stem cell cryopreservation bag, the most important specifications are: 1) engraftment capability of the stored cells, which is presumably dependent on stem cell viability and bag integrity. Our validation considered all three properties when developing the validation plan, but the most significant specification, engraftment, was not able to be prospectively assessed. Therefore, the validation plan initially measured MNC viability, colony-forming ability, and bag integrity and then proceeded to a clinical phase to assure adequate engraftment capability through a long-term analysis.
The new bags performed similar to the old bags when compared side by side in the laboratory using live cells of similar composition to stem cells. Nonmobilized donor cells were not an identical cell source to mobilized progenitor cells, but a commercial supplier of mobilized stem cells was not identified. Nonmobilized MNCs were chosen because they were the only cells available that most closely resembled the cells used in practice. Nevertheless, there was a trend toward decreased viability and CFU growth that suggested a slight decrease in postthaw cell function with the new bags. Despite this trend, benchmarking with other laboratories supported the historical success of the new bag in practice though other products were available. Overall, the postthaw viability for both bags may have been depressed because the cells were centrifuged and washed of cryopreservative dimethyl sulfoxide for the study, a process that is not performed during routine practice. Furthermore, some postcryopreservation counts exceed precryopreservation values due to the concentration of cells and the subsequent sample dilution necessary to assess postcryopreservation WBC count. Under practice conditions, the cells are immediately infused after thaw. Nevertheless, the validation was performed in parallel to mitigate misinterpretation of any detrimental effects that the validation method may have introduced.
This conclusion bore out clinically by engraftment times not being related to the cryopreservation bag, but rather the CD34 dose. Although there was a difference in days to engraftment noted on initial review, when controlled for influential variables such as CD34 dose, the difference between the bags became insignificant. It is very likely that changes in transplantation CD34 dose would account for most of the observed engraftment time difference. Engraftment and CD34 dose are clearly related based on previous studies.4 Therefore, one might conclude that there is no significant difference in days to engraftment due to the different bag types. There is, however, a decrease in CD34+ counts, which helps explain the observed increase in engraftment times.
Furthermore, it was clear from the outset of the validation plan that the most important criteria to define an effective cryopreservation bag was successful patient engraftment. Unfortunately, successful engraftment was impossible to assess accurately without implementation of the bag in practice. Therefore, to address the need for a replacement cryopreservation bag and continued engraftment success, additional engraftment data were tracked prospectively on the new bags to compare with historical data on the old bags. This continuous validation approach appears as the only viable option to address the important clinical criteria that may be affected by a process change. Our validation results highlight the obstacles in assessing some important criteria before implementation. Clinical-phase evaluation showed no difference in engraftment potential between the old and new bags. However, large numbers of tests needed to be performed to derive these results, and these studies had to be performed retrospectively.
Recent publications on cryopreservation bag validation have highlighted the need for assurance of bag integrity.5 The bag failures reported by Khuu and coworkers6 directed attention towards mechanical properties of the freezing bags used in cellular therapies. While bag integrity was a priority included in our bag validation plan, it was not our principal focus. It was recognized that the industrial design of cryopreservation bags focuses on material resistance to breakage in a cold environment. Therefore, it was possible for our validation to concentrate more on patient engraftment. Patient-centered laboratory and clinical markers of the engraftment potential were emphasized in this plan. Industry validation of mechanical properties provide a foundation from which patient-centered laboratories may expand the validation effort.
The clinical validation phase revealed a trend toward improved bag integrity with the new bags. This trend may have achieved significance because one of the two breaks encountered while using the new bags was caused by handling and not from the bag integrity. This bag was noted to be pinched in the cassette. Nevertheless, this bag break was included in the analysis. In addition, the second break noted with the new bags occurred early on in transition to the new bag size with zero occurring over the past 6 years. There may have been a transition learning process with the new bag’s shape. No bag breakages resulted in adverse patient outcomes.
There are no published validation studies of cryopreservation bags for stem cell processing in the United States, although studies have been performed to investigate the effects of storage environment on cell function.7,8 Process changes occur in the stem cell laboratory and there are few guidances available that have been produced by practitioners in the field. More research and investigation is needed to address the medical process validation to enable rapid adjustment to changing market pressures without significantly affecting patient care. As manufactures’ patents are bought and sold with production rights, the new production lots should have a practical validation plan presented with the change approval.
ABBREVIATIONS
- ANC
absolute neutrophil count
- BMT
Blood and Marrow Transplant
Footnotes
CONFLICT OF INTEREST
There are no conflicts of interest reported by any of the authors.
References
- 1.FACT-JACIE. International standards for cellular therapy product collection, processing and administration. 4. 2008. [Google Scholar]
- 2.Food and Drug Administration. Current good tissue practices. 21CFR1271.150. [Google Scholar]
- 3.Food and Drug Administration. Guideline on general principles of process validation. 1987 [cited 2010 Nov 29]. Available from: http://www.fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/ucm124720.htm.
- 4.Stewart DA, Guo D, Luider J, Auer I, Klassen J, Ching E, Morris D, Chaudhry A, Brown C, Russell JA. Factors predicting engraftment of autologous blood stem cells: CD34+ subsets inferior to the total CD34+ cell dose. Bone Marrow Transplant. 1999;23:1237–43. doi: 10.1038/sj.bmt.1701800. [DOI] [PubMed] [Google Scholar]
- 5.Proctor J, Regen D. Validation of cryobags. ISCT Telegraft. 2006;13:8–10. [Google Scholar]
- 6.Khuu HM, Cowley H, David-Ocampo V, Carter CS, Kasten-Sportes C, Wayne AS, Solomon SR, Bishop MR, Childs RM, Read EJ. Catastrophic failures of freezing bags for cellular therapy products: description, cause, and consequences. Cytotherapy. 2002;4:539–49. doi: 10.1080/146532402761624700. [DOI] [PubMed] [Google Scholar]
- 7.Fleming KK, Hubel A. Cryopreservation of hematopoietic and non-hematopoietic stem cells. Transfus Apher Sci. 2006;34:309–15. doi: 10.1016/j.transci.2005.11.012. [DOI] [PubMed] [Google Scholar]
- 8.Valeri CR, Pivacek LE. Effects of the temperature, the duration of frozen storage, and the freezing container on in vitro measurements in human peripheral blood mono-nuclear cells. Transfusion. 1996;36:303–8. doi: 10.1046/j.1537-2995.1996.36496226141.x. [DOI] [PubMed] [Google Scholar]