Abstract
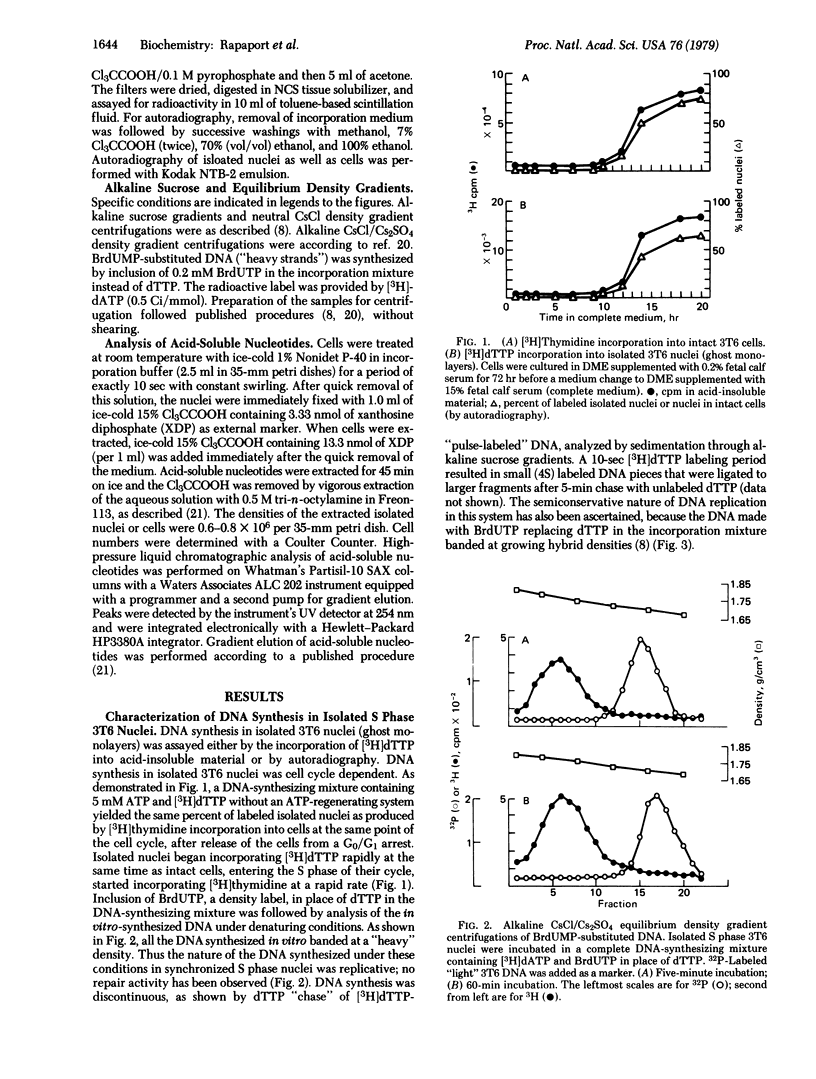
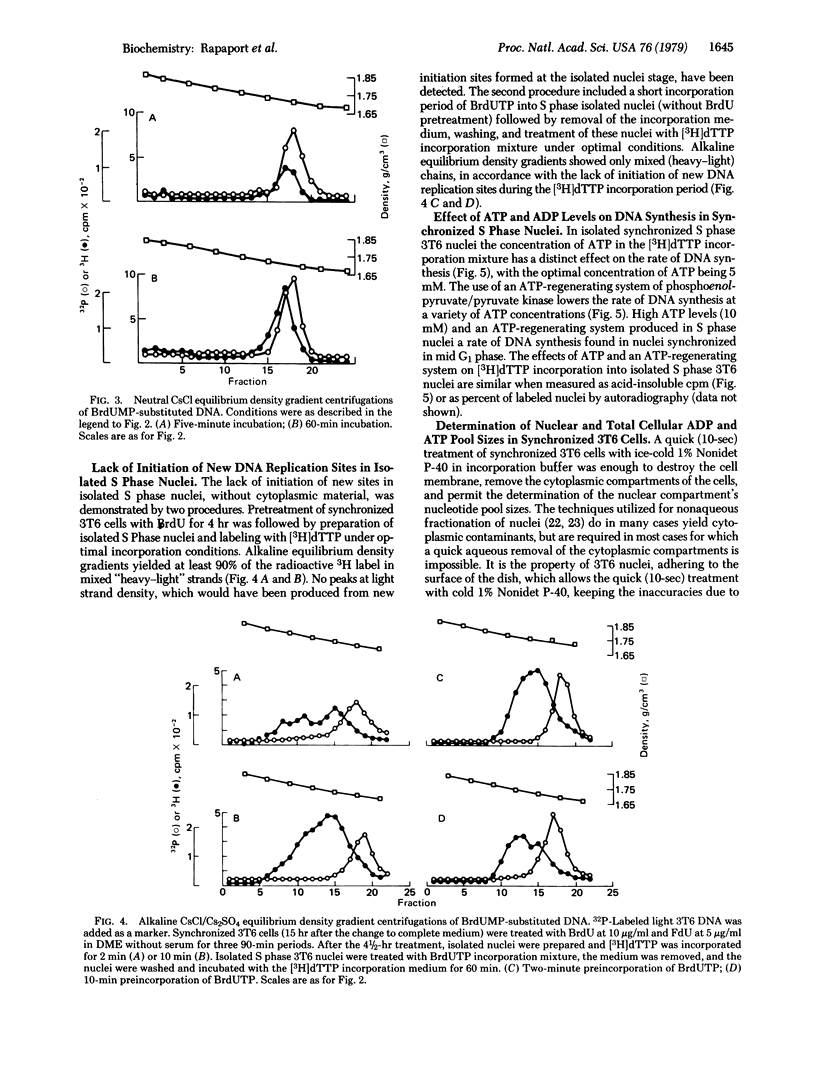
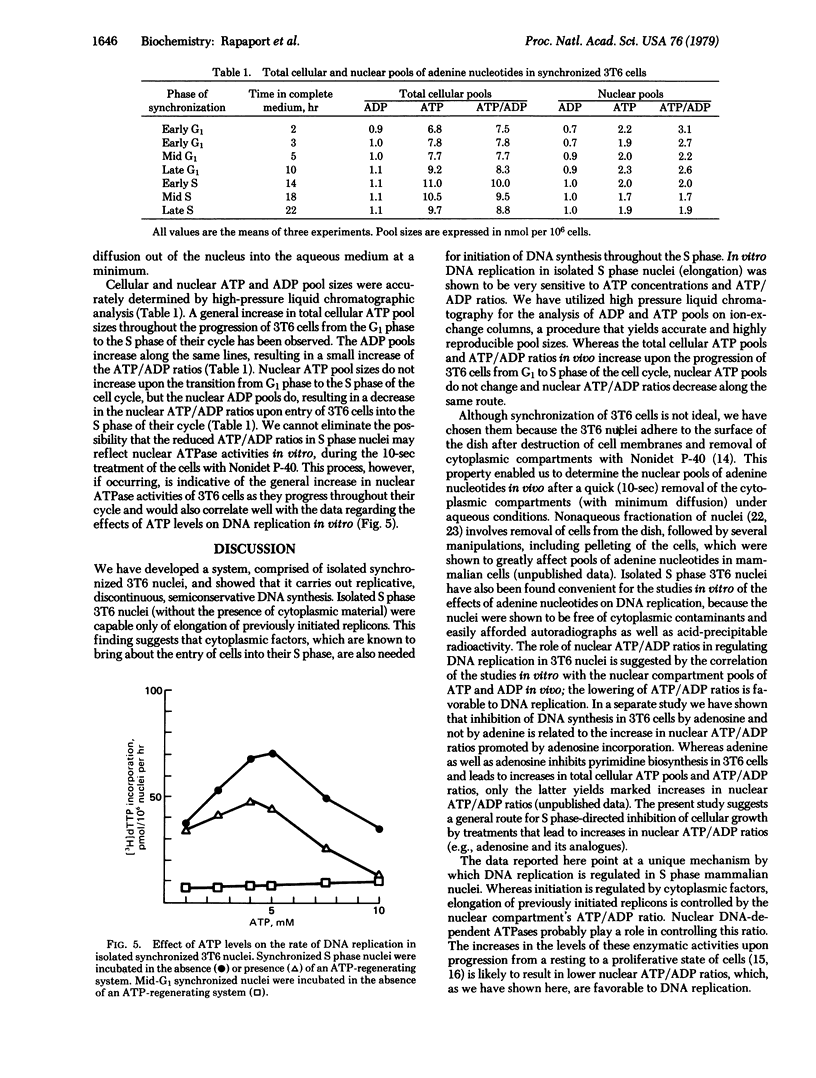
Synchronized 3T6 (mouse fibroblast) ghost monolayers (isolated nuclei) were utilized to study the effects of ATP and ADP levels on DNA replication in vitro. A system yielding discontinuous semiconservative DNA replication (without any detectable repair) in synchronized S phase nuclei has been developed. Lack of initiation of new sites has been observed in isolated S phase 3T6 nuclei without the presence of cytoplasmic material; DNA synthesis is comprised only of elongation at sites where initiation had previously taken place. DNA synthesis in S phase nuclei proceeded optimally at an ATP concentration of 4-5 mM. High ATP levels as well as high ATP/ADP ratios (produced by an ATP-regenerating system at a variety of ATP concentrations) yielded marked inhibition of [3H]dTTP incorporation. The cellular and nuclear pools of ATP and ADP in intact synchronized 3T6 cells were accurately determined by high-pressure liquid chromatography. A good correlation with the studies on isolated nuclei has been observed. Whereas total cellular ATP pools increase during the progression of 3T6 cells from G1 to S phase of the cell cycle, nuclear ATP pools do not increase, and the nuclear ATP/ADP ratios decrease once the cells enter the S phase of their cycle. These experiments suggest that nuclear ATP pools and ATP/ADP ratios act as S phase controls, regulating DNA elongation at sites where its synthesis has previously been initiated by cytoplasmic factors.
Keywords: control of growth, adenine nucleotides, cell cycle
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chen S. C., Brown P. R., Rosie D. M. Extraction procedures for use prior to HPLC nucleotide analysis using microparticle chemically bonded packings. J Chromatogr Sci. 1977 Jun;15(6):218–221. doi: 10.1093/chromsci/15.6.218. [DOI] [PubMed] [Google Scholar]
- Grummt F. Diadenosine 5',5'''-P1,P4-tetraphosphate triggers initiation of in vitro DNA replication in baby hamster kidney cells. Proc Natl Acad Sci U S A. 1978 Jan;75(1):371–375. doi: 10.1073/pnas.75.1.371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grummt F., Paul D., Grummt I. Regulation of ATP pools, rRNA and DNA synthesis in 3T3 cells in response to serum or hypoxanthine. Eur J Biochem. 1977 Jun 1;76(1):7–12. doi: 10.1111/j.1432-1033.1977.tb11564.x. [DOI] [PubMed] [Google Scholar]
- Gurney T., Jr, Foster D. N. Nonaqueous isolation of nuclei from cultured cells. Methods Cell Biol. 1977;16:45–68. doi: 10.1016/s0091-679x(08)60091-6. [DOI] [PubMed] [Google Scholar]
- Hachmann H. J., Lezius A. G. An ATPase depending on the presence of single-stranded DNA from mouse myeloma. Eur J Biochem. 1976 Jan 15;61(2):325–330. doi: 10.1111/j.1432-1033.1976.tb10025.x. [DOI] [PubMed] [Google Scholar]
- Hamlin J. L., Pardee A. B. Control of DNA synthesis in tissue culture cells. In Vitro. 1978 Jan;14(1):119–127. doi: 10.1007/BF02618178. [DOI] [PubMed] [Google Scholar]
- Hatanaka M., Del Giudice R., Long C. Adenine formation from adenosine by mycoplasmas: adenosine phosphorylase activity. Proc Natl Acad Sci U S A. 1975 Apr;72(4):1401–1405. doi: 10.1073/pnas.72.4.1401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hershey H. V. Effect of ATP analogs of DNA synthesis in isolated nuclei. Biochim Biophys Acta. 1977 Dec 2;479(3):256–264. doi: 10.1016/0005-2787(77)90108-3. [DOI] [PubMed] [Google Scholar]
- Hershey H. V., Stieber J. F., Mueller G. C. Dna synthesis in isolated HeLa nuclei. A system for continuation of replication in vivo. Eur J Biochem. 1973 Apr;34(2):383–394. doi: 10.1111/j.1432-1033.1973.tb02770.x. [DOI] [PubMed] [Google Scholar]
- Kirsch W. M., Leitner J. W., Gainey M., Schulz D., Lasher R., Nakane P. Bulk isolation in nonaqueous media of nuclei from lyophilized cells. Science. 1970 Jun 26;168(3939):1592–1595. doi: 10.1126/science.168.3939.1592. [DOI] [PubMed] [Google Scholar]
- Krokan H., Bjorklid E., Prydz H. DNA synthesis in isolated HeLa cell nuclei. Optimalization of the system and characterization of the product. Biochemistry. 1975 Sep 23;14(19):4227–4232. doi: 10.1021/bi00690a012. [DOI] [PubMed] [Google Scholar]
- Liu L. F., Wang J. C. Micrococcus luteus DNA gyrase: active components and a model for its supercoiling of DNA. Proc Natl Acad Sci U S A. 1978 May;75(5):2098–2102. doi: 10.1073/pnas.75.5.2098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lynch W. E., Brown R. F., Umeda T., Langreth S. G., Lieberman I. Synthesis of deoxyribonucleic acid by isolated liver nuclei. J Biol Chem. 1970 Aug 10;245(15):3911–3916. [PubMed] [Google Scholar]
- Otto B. DNA-dependent ATPases in concanavalin A stimulated lymphocytes. FEBS Lett. 1977 Jul 1;79(1):175–178. doi: 10.1016/0014-5793(77)80377-3. [DOI] [PubMed] [Google Scholar]
- Rao P. N., Johnson R. T. Mammalian cell fusion: studies on the regulation of DNA synthesis and mitosis. Nature. 1970 Jan 10;225(5228):159–164. doi: 10.1038/225159a0. [DOI] [PubMed] [Google Scholar]
- Rapaport E., Zamecnik P. C. Incorporation of adenosine into ATP: formation of compartmentalized ATP. Proc Natl Acad Sci U S A. 1976 Sep;73(9):3122–3125. doi: 10.1073/pnas.73.9.3122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rapaport E., Zamecnik P. C. Presence of diadenosine 5',5''' -P1, P4-tetraphosphate (Ap4A) in mamalian cells in levels varying widely with proliferative activity of the tissue: a possible positive "pleiotypic activator". Proc Natl Acad Sci U S A. 1976 Nov;73(11):3984–3988. doi: 10.1073/pnas.73.11.3984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlaeger E. J. Chromatin replication in isolated nuclei from bovine lymphocytes. Biochem Biophys Res Commun. 1978 Mar 15;81(1):8–18. doi: 10.1016/0006-291x(78)91623-6. [DOI] [PubMed] [Google Scholar]
- Schneider E. L., Stanbridge E. J., Epstein C. J. Incorporation of 3H-uridine and 3H-uracil into RNA: a simple technique for the detection of mycoplasma contamination of cultured cells. Exp Cell Res. 1974 Mar 15;84(1):311–318. doi: 10.1016/0014-4827(74)90411-x. [DOI] [PubMed] [Google Scholar]
- Tsai R. L., Green H. Rate of RNA synthesis in ghost monolayers obtained from fibroblasts preparing for division. Nat New Biol. 1973 Jun 6;243(127):168–170. doi: 10.1038/newbio243168a0. [DOI] [PubMed] [Google Scholar]
- Tseng B. Y., Goulian M. DNA synthesis in human lymphocyts: intermediates in DNA synthesis, in vitro and in vivo. J Mol Biol. 1975 Dec 5;99(2):317–337. doi: 10.1016/s0022-2836(75)80149-5. [DOI] [PubMed] [Google Scholar]
- Winnacker E. L., Magnusson G., Reichard P. Replication of polyoma DNA in isolated nuclei. I. Characterization of the system from mouse fibroblast 3T6 cells. J Mol Biol. 1972 Dec 30;72(3):523–537. doi: 10.1016/0022-2836(72)90172-6. [DOI] [PubMed] [Google Scholar]
- Yanishevsky R. M., Prescott D. M. Late S phase cells (Chinese hamster ovary) induce early S phase DNA labeling patterns in G1 phase nuclei. Proc Natl Acad Sci U S A. 1978 Jul;75(7):3307–3311. doi: 10.1073/pnas.75.7.3307. [DOI] [PMC free article] [PubMed] [Google Scholar]



