Abstract
The present study investigated the anti-inflammatory effects of dichloromethane extract of Auricularia auricula-judae. Dichloromethane extract of Auricularia auricula-judae inhibited Lipopolysaccharide (LPS) -induced nitric oxide (NO) production significantly in a dose-dependent manner in the concentration ≥10 μg/ml (p < 0.05) . Furthermore, RT-PCR results of this study indicated that the extract markedly reduced the expressions of inflammatory cytokines (IL-6, TNF-α and IL-1β) mRNA in LPS-treated murine RAW 264.7 macrophages, which could possibly ameliorate the inflammation. Nevertheless, dichloromethane extract of Auricularia auricula-judae did not show complete inhibition of IL-6 mRNA expression. The inhibition of IL-1β cytokine at protein level was also observed in a dose dependent manner. In conclusion,the current study revealed the previously unknown effect of dichloromethane ethyl extract of Auricularia auricula-judae inhibitions of the production of NO, IL-6, TNF-α and IL-1β in LPS-stimulated macrophages.
Keywords: Auricularia auricula-judae, Anti-inflammatory, Nitric oxide, Inflammatory cytokines
INTRODUCTION
Auricularia auricula-judae is a species of edible Auriculariales mushroom found worldwide, distinguished by its noticeably ear-like shape and brown coloration. A. auricula-judae has been the subject of research for possible medicinal uses and as an important source of new chemical substances with potential therapeutic effects (Misaki et al., 1981). In a study of aqueous extracts of seven species of edible mushrooms tested for their antitumor activity against transplanted Sarcoma 180 in Swiss albino mice, all of the aqueous extracts except that of A. auricula-judae highly inhibited the growth of transplanted tumors. However, another experiments concluded that two glucans isolated from this species had showed potent antitumor properties when used on mice artificially implanted with Sarcoma 180 tumors (Ikekawa, 1969; Misaki et al., 1981). Further, research on hypoglycemic effect of water-soluble polysaccharide (FA) from fruiting bodies of A. auricula-judae was investigated on genetically diabetic mice (KK-Ay) from 10 to 14 weeks of age; mice fed with food including the polysaccharide showed reduced plasma glucose, insulin, urinary glucose and food intake (Yuan, 1998). Other studies on the effects of A. auricula-judae in anticoagulant and platelet aggregation have been investigated (Yoona et al., 2003; Francia, 1999). Nevertheless, there is no report on its therapeutic effect in inflammatory response.
Inflammation is a disorder involving localized increases in the number of leukocytes and a variety of complex mediator molecules. Inflammatory responses play a central role in the pathogenesis of many diseases, initiated by the invasion of pathogens or by tissue injury caused by free radicals, followed with a series of vascular and cellular reactions. Among the leukocytes macrophages are the key players in inflammatory responses and are also the major sources of pro-inflammatory cytokines and enzymes including tumor necrosis factor-α (TNF-α) , interleukins (ILs) , cyclooxygenase (COX) , and inducible nitric oxide synthase (iNOS) (Bonizzi and Karin, 2004; Verma et al., 2010; Duffield, 2003). These genes of pro-inflammatory mediators are strongly induced during inflammation and are responsible for its initiation and persistence. TNF-α and IL-1β are the cytokines that act as signaling molecules for immune cells and coordinate the inflammatory responses (Krakauer, 2004). Nitric oxide (NO) is a free radical that mediates many physiological and pathophysiological processes, including neurotransmission and inflammation (Nathan, 1994). Expression of the inducible isoform of NOS (iNOS) in activated macrophages is mainly responsible for production of pathological concentration of NO during inflammation.
The existing synthetic molecule like nonsteroidal antiinflammatory drugs (NSAIDs) and selective COX-2 inhibitors are known for their side-effect in increasing the incidence of adverse cardiovascular thrombosis (Chowdhury et al., 2009). An alternative therapeutic with minimum side effect from natural plant extract should be a focus in scientific research.
Therefore, the present study was designed to investigate the anti-inflammatory effects of dichloromethane extract of A.auricula-judae in RAW 264.7 cells with LPS induced NO and cytokine IL-6, TNF-α and IL-1β pro-inflammatory markers.
MATERIALS AND METHODS
Preparations of plant extract.
Dried fruit-bodies of A.auricula-judae were ground in an electric mill, and sieved through 40-mesh. The Dried powder (33.16 g) was suspended in 40 times volume of 80% ethanol and heated two times at 100℃ for 3 hrs. The soluble substances were filtered through filter paper and collected. This process was conducted 3 times. And then collected soluble substances were concentrated in a rotary evaporator. Thereafter, residues was re-suspended with water in a Soxhlet extractor and fractionated with the same volume of dichloromethane (CHCl2) for 24 hrs. The dichloromethane extracts of A.auricular-judae were dissolved in sterile phosphate buffered saline (PBS) and sonificated prior to use.
Cell cultures.
Murine macrophage RAW 264.7 cell (KCLB 40071, Korean Cell Line Bank, Seoul, South Korea) were cultured in phenol red free-Dulbecco’s Modified Eagles Medium (DMEM) supplemented with 10% FBS, 1% Lglutamine, and 100 IU/ml penicillin-100 μg/ml streptomycin at 37℃ in a 5% CO2 humidified air.
Determination of nitric oxide production (NO assay) .
RAW 264.7 cells (2 × 105 cells/ml) were treated with LPS (1 μg/ml) in the presence and absence of dichloromethane extract of A. auricula-judae (10, 30 and 100 μg/ml) for 24 h. To measure nitrite, 100 μl of the cell-free culture medium was removed and placed into a new 96 well flat bottom plate. One hundred microliter of Griess reagent was added into each well and kept for 10 min at room temperature (light protected) . Optical density (OD) was measured at 540 nm using VERSA max microplate reader (Molecular Devices, Sunnyvale, CA. USA) . The nitrite amount in each sample was calculated using a standard curve generated with sodium nitrite (from 0 to 100 μM in cell culture medium) .
Determination of cytokine production with enzymelinked immune-sorbent assay (ELISA) .
RAW 264.7 cells were treated with LPS (1 μg/ml) in the presence or absence of A. auricula-judae extract (10, 30 and 100 μg/ml) for 24 h at 37℃ and 5% CO2 in a humidified air incubator. Culture supernatants were then collected and the levels of cytokines measured. IL-1β concentration in the culture medium was quantified using A solid phase sandwich ELISA commercial kits (Invitrogen, Camerillo, CA. USA) according to the manufacturer’s instructions.
Determination of mRNA expressions of cytokines (RTPCR) .
For examining of mRNA expressions from RAW 264.7 cells treated with LPS (1 μg/ml) in the presence or absence of dichloromethane extract of A. auricula-judae (10, 30 and 100 μg/ml) , total RNA were harvested using Trizol reagent (Invirogen, CA, USA) . Isolated total RNA was incubated with oligo (dT) 15 primer and then reverse transcription was performed using thermal cycler (Mycycler,BIO-RAD) . Then a PCR amplification was undergone with the PCR primers, which include Sense (S) CAGGATGAGGACATGAG-CACC, Antisense (AS) CTCTGCAGACTCAAA-CTCCAC for IL-1β, (S) GTACTCCA-GAAGACCAGAGG, (AS) TGCTGGTGACAAC-CACGGCC for IL-6, (S) TTAACCTA-CGCGCTGAGTTG, (AS) CCTGTAGCCCACGR-CGRAGC for TNF-α, and (S) ATGCTCCTGCTTGAGTATGT, (AS) GGAGGAAGAG-GATGCGGCAGT forβ-actin (as a housekeeping gene) . PCR reactions for IL-1β,IL-6, TNF-α and β-actin were performed using the following conditions: 30 cycles of denaturation at 95℃ for 1 min, annealing at 55℃ for 45 sec and extension at 72℃ for 5 sec; using a thermal cycler (Mycycler, BIO-RAD, USA) and an AccuPower PCR Premix (BioNEER, Dageon, Korea) by following the manufacture’s instruction. RT-PCR products were separated on a 1% TAE agarose gel, stained with ethidium bromide, and visualized using EAGLE-EYE TM (Stratagene, La. Jolla, CA, USA) .
Statistical analysis.
All data are presented as means SEM. Data were analyzed by one-way analysis of variance (ANOVA) and by using Student’s t-test (version 9.1, SAS,NC, USA) . Differences were considered to be significant at p < 0.05 level.
RESULTS AND DISCUSSION
Inflammatory response plays an important role not only in the normal physiology but also in pathology when inflammation is recognized as a major driving force leading to development of malignancy such as cancer. Medicinal plants and their products have been used for many centuries to treat different kinds of acute and chronic inflammatory diseases like wound healing, edema and rheumatoid arthritis (Abrham et al., 2010). However, regardless of the recognized effects of several traditionally used medicinal herbs, their pharmacological activities have not been thoroughly investigated concerning their immunological effects. In the current study, we have investigated the anti-inflammatory effect of dichloromethane extract of A.auricula-judae in RAW 264.7 cells.
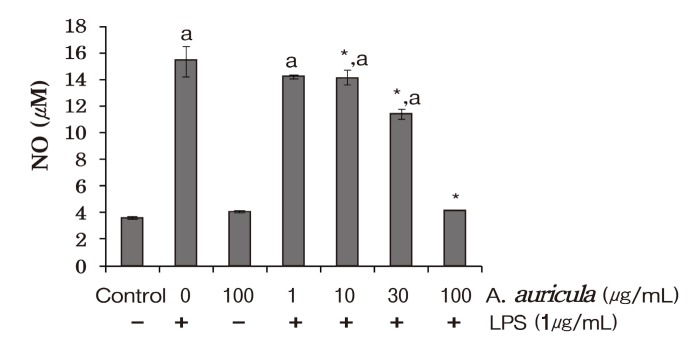
To understand the role of dichloromethane extract of A.auricula-judae as anti-inflammatory agent, we utilized LPS induced inflammatory responses inhibition test in RW264.7 cells. Initially, we investigated the effect of dichloromethane extract of A. auricula-judae on LPS-induced NO production in elicited macrophages. Several stimuli, in addition to LPS, which include TNF-α and IFN-γ can result in the production of massive amount of NO by the activated macrophages, which can participate in the pathological processes in several acute and chronic inflammatory disorders (Murakami et al., 2003; Hyun et al., 2004). Furthermore, many cell types, especially macrophages are known to induce NO production via stimulation with bacterial LPS (Kiemer et al., 2002; Numata et al., 1998). NO is produced in large amounts from the amino acid L-arginine by the inducible nitric oxide synthase (iNOS) . Hence, drugs that inhibit NO production initiated by LPS have appreciable therapeutic effect in the treatment of major inflammatory and infectious diseases (Boucher et al.., 1999; Albina and Reichner, 1998; Zhang and Ghosh, 2000). In this study, we demonstrate that the plant extracts under study significantly inhibited NO production in LPS-stimulated RAW 264.7 cells.
As shown in Fig. 1, we observed that dichloromethane extract of A. auricula-judae inhibited LPS-induced NO production significantly in a dose-dependent manner in the concentration ≥ 10 μg/ml (p < 0.05) . To our knowledge, this result is first report for the effects of dichloromethane extract of A. auricula-judae in murine macrophages. The inhibition of NO observed at highest dose (73.5%) was highly suppressive reducing to the NO production to the basal levels observed in control group (p < 0.01) . The effective dose range used in this experiment is also comparable to plasma levels as reported in pharmacokinetic studies.
Fig. 1. Inhibition of nitric oxide production measured as nitrite in the cell free culture supernatants of LPS (1 lg/ml) challenged RAW 264.7 murine macrophages. NO concentration in the culture supernatant was determined by Griess assay using sodium nitrite standard curve. Data are presented as means ± SD from three sets of independent experiments. * p< 0.05 represent significant difference compared with cells treated with LPS alone. a p < 0.05 represent significant difference compared with control group.
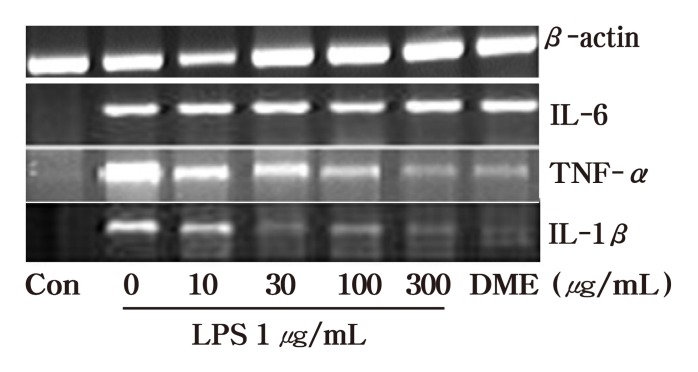
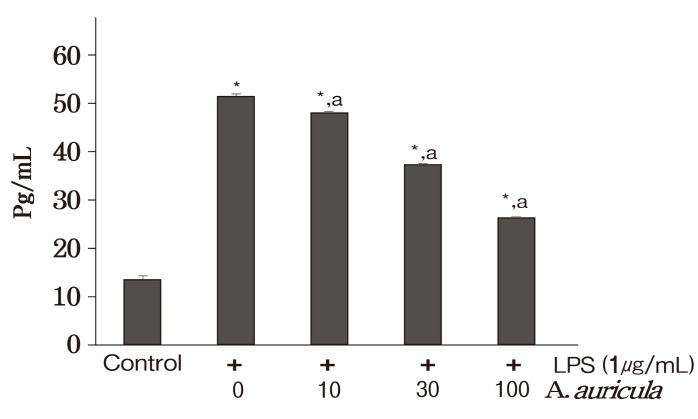
In addition to NO, proinflammatory cytokines are important mediators of inflammatory responses. IL-6, TNF-α, and IL-1β serve as endogenous pyrogens that cause fever by initiating metabolic changes in the hypothalamic thermoregulatory center during inflammation (Wu et al., 2008). These cytokines up-regulate the inflammatory responses and stimulate the production of acute phase reactants. We consequently asses the inhibition of proinflammatory cytokines induced by LPS stimulation in RAW 264.7 macrophages by dichloromethane extract of A. auricula-judae. This study indicates that the extract under investigation markedly reduces the expressions of inflammatory cytokines (IL-6,TNF-α and IL-1β) mRNA in LPS-treated murine RAW 264.7 macrophages (Fig. 2) , which could possibly ameliorate the inflammation. Nevertheless, as observed in the Fig.2 dichloromethane extract of A. auricula-judae did not show complete inhibition of IL-6 mRNA expression. The underlying cause for the incomplete inhibition observed by IL-6 is not known. Besides the incomplete inhibition of mRNA expression of IL-6, the control group treated with dichloromethane extract of A. auricula-judae has shown expression of IL-6 mRNA. This might be the possible reason not to see the inhibition of LPS induced IL-6. The cytokines inhibition at protein level was assed using IL-1β,and the result was similar to mRNA expression showing a dose-dependent inhibition (Fig. 3) .
Fig. 2. Inhibition of LPS induced cytokine mRNA expression in RAW 264.7 murine macrophages. Total RNA was extracted and RT PCR was done to check the expression level of IL-6 TNF-α and IL-1β mRNA. DME (Dichloro Methane Ethyl extract) Con (Control) . Figure is representative of two independent experiments.
Fig. 3. Inhibition of IL-1β production measured with ELISA in the cell free culture supernatants of LPS (1 lg/ml) challenged RAW 264.7 murine macrophages. IL-1β concentration in the culture supernatant was determined by ELISA using standard curve. Data are presented as means ± SD from three sets of independent experiments. * p< 0.05 represent significant difference compared with cells treated with LPS alone. a p < 0.05 represent significant difference compared with control group.
In conclusion, in the current study, the inhibitory effect of dichloromethane extract of A. auricula-judae in the production of NO, IL-6, TNF-α and IL-1β in LPS-stimulated macrophages has been established for the first time. Further studies on the mechanisms involved in the inhibition of NO and proinflammatory cytokine in vivo is underway.
Acknowledgments
This work was supported by Technology Development Program for Agriculture and Forestry, Ministry for Agriculture,Forestry and Fisheries, Republic of Korea.
References
- 1.Abrham G. Dovrat S. Bessler H. Grossman S. Nir U. Bergman M. Inhibition of inflammatory cytokinesecretion by plant-derived compounds inuviscolide and tomentosin: The role of NF-κB and STAT1. The Open Pharmacology Journal. 2010;4:36–44. doi: 10.2174/1874143601004010036. [DOI] [Google Scholar]
- 2.Albina J.L. Reichner J.S. Role of nitric oxide mediation of macrophage cytotoxicity and apoptosis. Cancer Metastasis Rev. 1998;17:39–53. doi: 10.1023/A:1005904704618. [DOI] [PubMed] [Google Scholar]
- 3.Bonizzi G. Karin M. The two NF-jB activation pathways and their role in innate and adaptive immunity. Trends Immunol. 2004;25:280–288. doi: 10.1016/j.it.2004.03.008. [DOI] [PubMed] [Google Scholar]
- 4.Boucher J.L. Moali C. Tenu J.P. Nitric oxide biosynthesis nitric oxide synthase inhibitors and arginase competition for L-arginine utilization. Cell Mol Life Sci. 1999;55:1015–1028. doi: 10.1007/s000180050352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chowdhury M.A. Abdellatif K.R.A. Don Y. Das D. Suresh M.R. Knaus E.E. Synthesis of celecoxib analogues possessing a N-difluoromethyl-12-dihydropyrid-2-one s-lipoxygenase pharmacophore: Biological evaluation as dual inhibitors of cyclooxygenases and 5-Lipoxygenase with anti-inflammatory activity. J. Med. Chem. 2009;52:1525–1529. doi: 10.1021/jm8015188. [DOI] [PubMed] [Google Scholar]
- 6.Duffield J.S. The inflammatory macrophage: a story of Jekyll and Hyde. Clin Sci. 2003;104:27–38. doi: 10.1042/CS20020240. [DOI] [PubMed] [Google Scholar]
- 7.Francia C. Rapior S. Courtecuisse R. Siroux Y. Current research findings on the effects of selected mushrooms on cardiovascular diseases. International Journal of Medicinal Mushrooms. 1999;1:169–172. [Google Scholar]
- 8.Hyun E. Bolla M. Steinhoff M. John L.W. del Soldato P. Vergnolle N. Anti-inflammatory effects of nitric oxidereleasing hydrocortisone NCX 1022 in a murine model of contact dermatitis. British Journal of Pharmacology. 2004;143:618–625. doi: 10.1038/sj.bjp.0705854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ikekawa T. Uehara N. Maeda Y. Nakanishi M. Fukuoka F. Antitumor activity of aqueous extracts of ediblemushrooms. Cancer Research. 1969;29:734–735. [PubMed] [Google Scholar]
- 10.Kiemer A.K. ü C. Vollmar A.M. Inhibition of LPS-induced nitric oxide and TNF-alpha production by alphalipoic acid in rat Kupffer cells and in RAW 264.7 murine macrophages. Immunol. Cell. Biol. 2002;80:550–557. doi: 10.1046/j.1440-1711.2002.01124.x. [DOI] [PubMed] [Google Scholar]
- 11.Krakauer T. Molecular therapeutic targets in inflammation cyclooxygenase and NF-κB. Curr. Drug. Targets. 2004;3:317–324. doi: 10.2174/1568010043343714. [DOI] [PubMed] [Google Scholar]
- 12.Misaki A. Kakuta M. Sasaki T. Tanaka M. Miyaji H. Studies on interrelation of structure and antitumor effects of polysaccharides: antitumor action of periodate-modifiedbranched (1→3) -β-D-glucan of Auricularia auricula-judaeand other polysaccharides containing (1→3) -glycosidic linkages. Carbohydrate Research. 1981;92:115–129. doi: 10.1016/S0008-6215(00)85986-8. [DOI] [PubMed] [Google Scholar]
- 13.Murakami A. Takahashi D. Hagihara K. Combinatorial effects of nonsteroidal anti-inflammatory drugs and food constituents on production of prostaglandin E2 and tumor necrosis factor alpha in RAW264.7 murine macrophages. Biosci. Biotechnol. Biochem. 2003;67:1056–1062. doi: 10.1271/bbb.67.1056. [DOI] [PubMed] [Google Scholar]
- 14.Nathan C. Xie Q.W. Nitric oxide synthases: roles tolls and controls. Cell. 1994;78:915–918. doi: 10.1016/0092-8674(94)90266-6. [DOI] [PubMed] [Google Scholar]
- 15.Numata M. Suzuki S. Miyazawa N. Miyashita A. Nagashima Y. Inoue S. Kaneko T. Okubo T. Inhibition of inducible nitric oxide synthase prevents LPS-induced acutelung injury in dogs. J. Immunol. 1998;160:3031–3037. [PubMed] [Google Scholar]
- 16.Verma N. Tripathi S.K. Sahu D. Das H.R. Das R.H. Evaluation of inhibitory activities of plant extracts on production of LPS-stimulated pro-inflammatory mediators in J774 murine macrophages. Mol. Cell. Biochem. 2010;336:127–135. doi: 10.1007/s11010-009-0263-6. [DOI] [PubMed] [Google Scholar]
- 17.Wu L.C. Fan N.C. Lin M.H. Chu I.R. Huang S.J. Hu C.Y. Han S.Y. Anti-inflammatory effect of spilanthol from Spilanthes acmella on murine macrophage by down-regulating LPS-induced inflammatory mediators. J. Agric. Food Chem. 2008;56:2341–2349. doi: 10.1021/jf073057e. [DOI] [PubMed] [Google Scholar]
- 18.Yoona S. Yub M. Pyunb Y. Hwangb J. Chuc D. Juneja L. Mourao P.A.S. The nontoxic mushroom Auricularia auricula contains a polysaccharide with anticoagulant activity mediated by antithrombin. Thrombosis Research. 2003;112:151–158. doi: 10.1016/j.thromres.2003.10.022. [DOI] [PubMed] [Google Scholar]
- 19.Yuan Z. He P. Cui J. Takeuchi H. Hypoglycemic effect of water-soluble polysaccharide from Auricularia auricula-judae Quel. on genetically diabetic KK-Ay mice. Bioscience Biotechnology and Biochemistry. 1998;62:1898–1903. doi: 10.1271/bbb.62.1898. [DOI] [PubMed] [Google Scholar]
- 20.Zhang G. Ghosh S. Molecular mechanisms of NF-κB activation induced by bacterial lipopolysaccharide through Toll like receptors. J. Endotoxin. Res. 2000;6:453–457. doi: 10.1179/096805100101532414. [DOI] [PubMed] [Google Scholar]