Abstract
A prevailing problem in neuroscience is the fast and targeted delivery of DNA into selected neurons. The development of an appropriate methodology would enable the transfection of multiple genes into the same cell or different genes into different neighboring cells as well as rapid cell selective functionalization of neurons. Here, we show that optimized femtosecond optical transfection fulfills these requirements. We also demonstrate successful optical transfection of channelrhodopsin-2 in single selected neurons. We extend the functionality of this technique for wider uptake by neuroscientists by using fast three-dimensional laser beam steering enabling an image-guided “point-and-transfect” user-friendly transfection of selected cells. A sub-second transfection timescale per cell makes this method more rapid by at least two orders of magnitude when compared to alternative single-cell transfection techniques. This novel technology provides the ability to carry out large-scale cell selective genetic studies on neuronal ensembles and perform rapid genetic programming of neural circuits.
Recent advances in studies on neural tissue and the complex morphology and function of neural circuits have gone hand-in-hand with the development of physical and molecular techniques, which have gradually enabled the highly controlled interrogation, modification and perturbation of neural circuits at both the cellular and functional level. In particular, the enormous heterogeneity of neural tissues requires highly cell specific and selective methods for sophisticated studies on function, morphology and connectivity of neural networks. The role of small populations or even single neurons has been demonstrated in neural signal processing1, sensory perception2 and behavioral response to stimulation in motor and somatosensory cortex3. At the same time the role of neuronal circuits, that are often dynamically changing and subsequently reconfiguring their related emergent function, is becoming evident.
To date, a number of techniques have been proposed for labeling and gene transfection of neuronal cells. Most commonly, a bulk volume of cells is transfected using viral gene delivery4, bulk electroporation5, biolistic transfection6 or by creation of a complete transgenic animal7. With most of these techniques the delivery method itself does not provide any cell specificity or selectivity; but by using molecular biology techniques expression of the gene of interest can be targeted to a specific type of cells8, cells defined as being synaptically interconnected9 or to a small defined volume of tissue10. However, with all of these approaches no single-cell specificity is achievable.
This issue has been partially solved by development of manual microinjection11, modified patch-clamping12 and single-cell electroporation13,14,15,16. These approaches are capable of transfecting and labeling single neurons both in vitro and in vivo. However, they are all significantly laborious and inherently limited both in speed and throughput. Indeed recently, an automated single-cell electroporation system has been demonstrated17 capable of improving the throughput to 2 cells/min.
Laser mediated poration of cellular membrane has recently emerged as an exciting technique of gene delivery into mammalian cells. A variety of lasers have been used for the delivery of membrane impermeable substances18. The light-tissue interaction mechanism depends on the light wavelength and pulse duration. In turn these may dictate the required optical dose, achievable transfection efficiency and the cell viability. In particular, a tightly focused femtosecond (fs) pulsed near infrared (NIR) laser beam has proven to provide single-cell selectivity, localized operation and low toxicity with a consistent performance19. Typically, a Titanium-Sapphire laser at a wavelength of approximately 800 nm and pulse duration of 12–200 fs is focused for a few tens of milliseconds through a high numerical aperture (NA) objective on the cellular membrane20. This creates a low density plasma that photochemically ruptures the cell membrane and results in formation of sub-micron pores that last a fraction of a second. The unmatched precision and delicacy of the treatment has enabled mRNA transfection of primary neurons to demonstrate that a transcription factor Elk1 mRNA optoinjected into the soma causes a different cellular response to mRNA injected into the dendrites21. In another study the whole transcriptome was extracted from primary astrocyte cells and optically transfected into individual primary rat hippocampal neurons causing their phenotypical change into astrocytes22.
However, to date no experiments involving DNA transfection into primary neuronal cells have been reported. Notably, non-viral plasmid DNA transfection into post-mitotic cells is typically significantly less efficient than into dividing cell types. In contrast to injection of fluorophores and RNA transfection, plasmid DNA has to be ultimately delivered to the cell nucleus for the gene to be expressed. In intact cells even if plasmid DNA is successfully delivered to the cytoplasm, it is unlikely to cross passively the nuclear membrane23 unless the cell undergoes mitosis during which the nuclear membrane is reorganized. To our knowledge the optimized technique detailed below is the first to provide successful optical transfection of plasmid DNA into neural cells. A scientific field that can particularly benefit from this cell selective transfection is neural circuit mapping and dissection or as has been recently coined “brain mapping”. This is especially important at the functional level where it is crucial to follow precisely a localized excitation of a particular neuron to its downstream functional response in a neural circuit. Optogenetics has enabled optical control of neural function with light and when applied with cell selectivity, this would provide a powerful tool in functional studies and mapping of neural connections24. To date, selective optogenetic photostimulation of a sub-population of neurons has been achieved either by genetic targeting of specific cell types using cell-specific promoters25 or by using localized two-photon excitation with a spatially patterned26, temporally sculpted27 or scanned focused beam28. While the former approach leads to expression of photosensitive proteins in a given type of cells it does not guarantee any spatial confinement of the transfected region nor single cell selectivity within a population of cells. At the same time, the spatially patterned excitation is particularly difficult to put into practice within freely moving in vivo animal models due to the required sophisticated optics such as reconfigurable diffractive elements or high numerical aperture objectives. It would appear that for optogenetic studies on behaving animals, the most convenient solution would be to use a relatively simple and compact portable non-selective illumination source and achieve cell selective excitation by selective transfection of opsins into small, arbitrarily defined populations of neurons. This would provide a robust and reproducible photoexcitation with minimized complexity of the behavioral experiment.
Single cell electroporation of channelrhodopsin-2 (ChR2) has been recently demonstrated29. However, due to its mechanically invasive nature and limitations in terms of throughput and number of transfected cells (<20 cells in one session) it may be difficult to use in more advanced large-scale studies. In contrast, the optical transfection system described here provides throughput of almost 100 cells/minute enabling transfection of thousands of cells within one transfection session, whilst maintaining the normal electrical properties of the neuron. It paves the way towards more complex functionalization of neural circuits involving larger populations of neurons, while guaranteeing single cell selectivity.
Results
Single-cell optical transfection
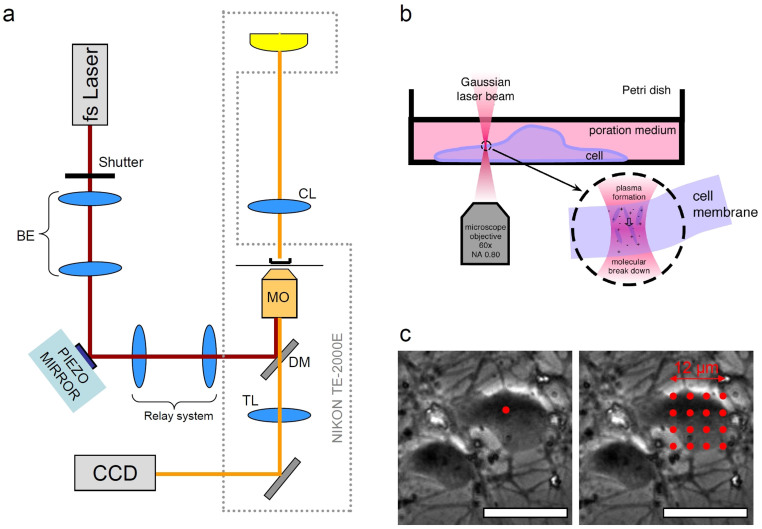
Our optical transfection system is based on a motorized NIKON TE-2000 inverted microscope which provides brightfield, phase contrast (PC), differential interference contrast (DIC) and epi-fluorescence imaging as desired. As shown in Figure 1a, the photoporation beam (800 nm, 200fs @ 80 MHz generated by Coherent MIRA900 pumped by Verdi-V5) was expanded so that it overfilled the back focal plane of the objective in order to guarantee a diffraction limited focal spot in the sample plane (d≈1 μm). A fast piezo-driven steering mirror (SM) was relayed by a 4-f non-magnifying telescope to the back focal plane of the microscope objective to provide fast lateral positioning of the beam without walk-off. This configuration is very similar to that of multiphoton scanning fluorescence microscopy and in principle many existing two-photon microscopy systems can be easily adapted for the purpose of optical transfection, as long as they are capable of stable beam parking for tens of milliseconds. The laser beam was coupled into the microscope objective (NIKON air 60x, NA = 0.8) using a dichroic mirror placed in the upper turret of the microscope. As shown in Figure 1b, the position of the laser focal spot was matched to the imaging plane of the microscope so that the beam was focused on the cell membrane.
Figure 1. Femtosecond optical transfection system.
(a) Schematic of the experimental setup (for details see Methods). BE: Beam Expander; CL: microscope condenser lens; DM: NIR dichroic mirror; TL: tube lens; CCD: digital camera. (b) magnified side-view of a Petri dish with the laser beam tightly focused on the membrane of an adherent neuron. (c) Two irradiation patterns superimposed on phase contrast images of cortical neurons: single spot (left) and square 4 × 4 grid. Scale bars are 20 μm.
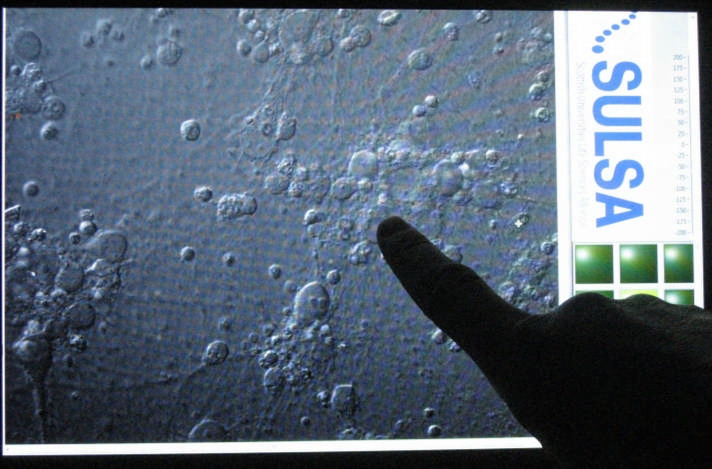
In order to take full advantage of the unique combination of a fully non-mechanical operation and sub-second treatment time per cell provided by femtosecond optical transfection, we developed a user-friendly software (LabView) based on a touch-screen interface implemented upon a tablet computer capable of rapid “point-and-transfect” targeting of selected cells upon a touch of a finger within the whole field of view (Fig. 2, Supplementary Video 1). This approach makes cell targeting and membrane poration easy and robust and enables a truly rapid cell-selective transfection of large populations of cells. Using the integrated user interface the operator can also control all the main features of the motorized microscope, such as stage movement and imaging parameters. The user may also control irradiation parameters such as shutter time and multiple irradiation patterns. In the manual operation mode the speed and throughput of this system are only limited by the human operator.
Figure 2. Touchscreen-based “point-and-transfect” user interface enables rapid positioning of the laser beam and irradiation of selected cells.
In practice, throughput is limited mostly by the reaction speed of the experimentator. (see Supplementary Video 1).
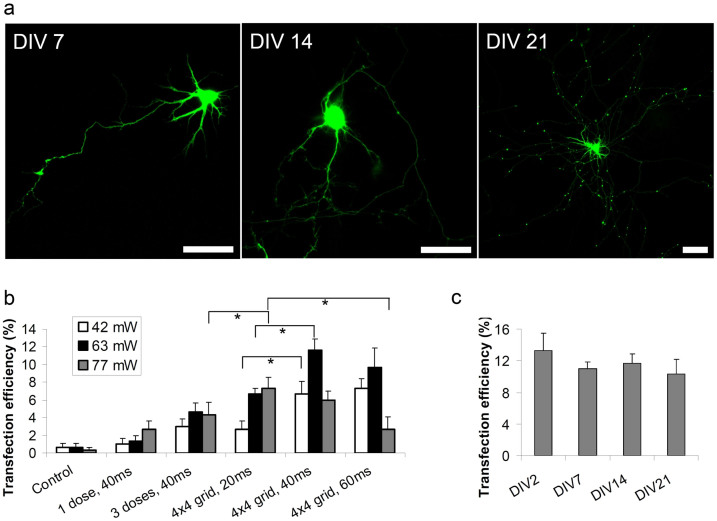
In order to optimize the irradiation parameters to primary neurons, we used a pCAG-YFP plasmid (see Online Methods) and characterized transfection efficiency with a range of powers and irradiation times on DIV14 rat cortical neurons. Typically, at this age cultured neurons exhibit active synaptic connectivity and mature spiking properties30 making them suitable for experimentation. We examined two types of irradiation strategies – a single spot irradiation with one or three consecutive doses (which was a common approach in earlier studies31; and a multiple-site irradiation as shown in Figure 1c. For multiple irradiation, we chose a total of 16 doses placed on a 4 × 4 grid pattern with a 4 μm step between sites. The total dimensions of that pattern are similar to the size of a neural soma and at the same time irradiation spots can be regarded as separate with no accumulation effect between doses. Multi-site irradiation is expected to increase the probability of genetic material crossing the cellular membrane21, especially in the case of large molecules such as plasmid DNA.
In each sample we selectively irradiated 50 cells in a small well defined region of the culture dish and assessed gene expression 48 hrs after transfection. We observed a negligible level of spontaneous transfection (Fig. 3b) with sporadic fluorescent glial cells easily recognizable by their morphology. Figure 3a shows examples of cortical neurons expressing YFP 48 hrs after transfection at various ages of culture. The observed expression levels were significantly above background autofluorescence allowing for easy distinction between expressing and non-expressing cells, indicating significant levels of plasmid DNA was entering the cells. In the majority of cells, expression remained at an easily detectable level for the remaining lifespan of the culture (up to DIV 25).
Figure 3. Optical transfection of plasmid DNA into single cortical neurons.
(a) Representative fluorescence images of neurons transfected with pCAG_YFP at various culture ages. All scale bars 30 μm. (b) Transfection efficiency of pCAG_YFP into DIV14 neurons for various irradiation doses and patterns. (c) Transfection efficiency of pCAG_YFP obtained with the optimum irradiation parameters (P = 63 mW, 4 × 4 grid, T = 40 ms) at various ages of the culture. No statistically significant difference in transfection efficiency was observed. In all experiments cells were assayed for gene expression 48 hrs post transfection. Each data point is the mean (±S.E.M) of n≥6 separate experiments with 50 cells irradiated in each. Stars in (b) denote pairs of data points that are significantly different in unpaired two tailed t-test (P < 0.05).
As presented in Figure 3b, the percentage of cells successfully transfected varied significantly with the irradiation parameters. Clearly, multi-site irradiation patterns provided better transfection efficiency than single and triple irradiation at a given single spot, which confirmed that multiple poration sites increased DNA transition into the cytoplasm. Multi-site irradiation with various power-time combinations resulted in strongly varying transfection efficiency. The nonlinear mechanism of light-tissue interaction makes the poration process sensitive to laser power. At the same time the ionization process that leads to membrane pore formation accumulates over the irradiation time.
Interestingly, the optimum irradiation time is different for each laser power. For the lowest power of P = 42 mW, a longer irradiation time was beneficial while for the highest power of P = 77 mW the opposite was true. This observation is consistent with previous studies32 that demonstrated the intricate interplay between the linear (time) and nonlinear (power) irradiation parameters. In the low power-short time regime not enough pores are formed on the membrane while the combination of a high power and a long irradiation time (e.g. P = 77 mW, T = 60 ms) may result in irreparable membrane damage, cell stress and potentially decreased viability.
We determined that in our system the optimum plasmid DNA transfection is achieved with the 4 × 4 grid pattern, beam power of P = 63 mW measured at the back aperture of the objective and T = 40 ms irradiation time in each spot. These parameters are comparable with those previously used for more simple cell lines31 with single spot triple irradiation. The efficiency achieved with these optimized parameters was 11.7 ± 1.2% (n = 6 separate experiments with 50 irradiated cells in each). Another advantage of this system was that it has been previously reported that in cultured neurons efficiency of other gene transfection techniques decreases with age of a culture33. However, Figure 3c shows that femtosecond optical transfection provides almost constant efficiency throughout the tested range of the culture age, varying between 13.3 ± 2.2% at DIV2 and 10.3 ± 1.8% at DIV21.
Optical transfection of multiple genes
Many experiments require multiple genes to be transfected in parallel. First, using the optimized transfection parameters, we verified successful transfection of plasmid DNA encoding mito-dsRed (Fig. 4a). Then we transfected a population of cells with a 1:1 mixture of dsRed2-Mito and cytoplasmic YFP expressing plasmids (Fig. 4b). We obtained co-expression of both proteins in 68% (17/25 expressing neurons) of successfully transfected cells (Fig. 4b) with 24% (6/25 neurons) expressing only YFP and 8% (2/25 neurons) only showing the mito-dsRed signal.
Figure 4. Representative fluorescence images of optically transfected neurons 48 hrs after transfection expressing (a) mitochondrial DsRed2, (b) simultaneously transfected 1:1 mixture of plasmid DNA encoding for mitochondrial DsRed (Red) and cytoplasmic YFP (Green), (c) cytoplasmic DsRed (Red) and cytoplasmic YFP (Green) sequentially transfected into selected neighboring cells.
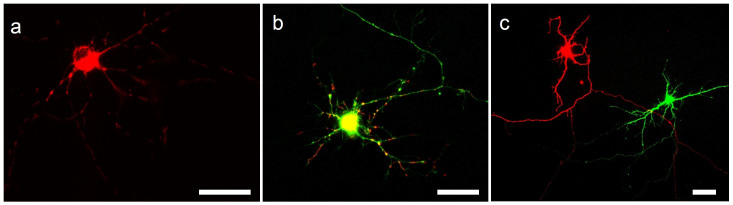
All scale bars represent 30 μm.
Significantly, the single cell selectivity of optical transfection also uniquely enables the easy transfection of different plasmids into neighboring cells. To demonstrate this capability, we first optically transfected a selected population of neurons with YFP, then replaced the DNA containing solution with fresh culture medium, incubated cells for 2 hours to recover and then repeated transfection with a plasmid encoding for cytoplasmic dsRed irradiating a separate but neighboring population of neurons. Figure 4c shows an example of two neighboring neurons expressing the two different fluorescent proteins. We observed no cells expressing both plasmids, which shows that a repetitive transfection procedure can be successfully used for cell-selective gene multiplexing within the same neural circuit, especially if combined with automated image-guided targeting and beam positioning within the sample.
Cell-selective optical transfection with channelrhodopsin-2
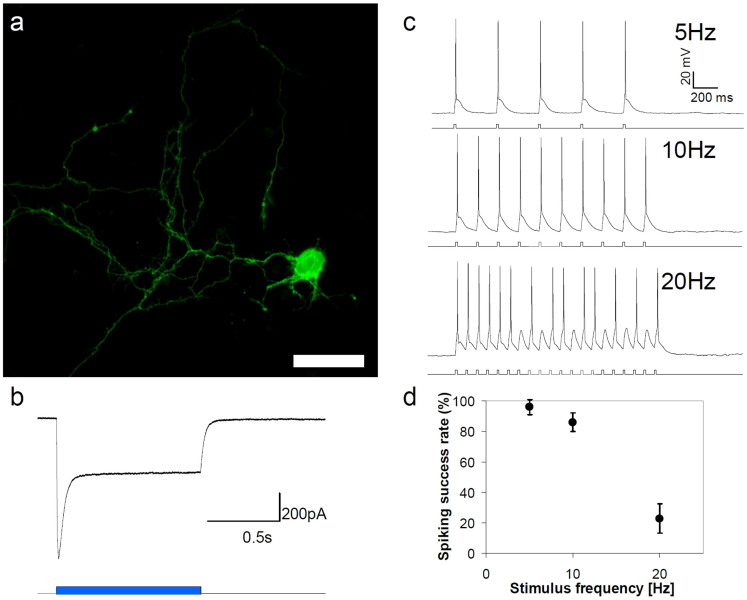
The high throughput and ease of use of femtosecond optical transfection may readily be extended to cell selective functionalization of tens or hundreds of cells in neural circuitry. This is of particular interest in the context of genetic techniques such as optogenetics. Using our optimized system, we selectively transfected plasmid DNA encoding for the YFP-tagged H134R variant of channelrhodopsin-2 Chr2(H134R)34 into DIV 14 rat cortical neurons. All expressing neurons exhibited strong membrane localized YFP signal 48 hrs after transfection with good morphology and no signs of decreased viability (Fig. 5a). In the majority of cells expression remained at a stable level for the remaining lifespan of the culture (up to DIV 25).
Figure 5. Optical transfection of pcDNA3.1/hChR2(H134R)-EYFP into selected single cortical neurons.
(a) Fluorescence (YFP) image of a cultured cortical neuron expressing YFP-tagged CHR2(H134R) 48 hrs post-transfection. Scale bar represents 30 μm. (b) Typical photocurrent evoked in optically transfected neurons by blue light (λ = 470 nm, P = 0.45 mW/mm2). (c) Representative trains of spikes evoked by pulsed (T = 10 ms) blue light excitation (λ = 470 nm, P = 0.45 mW/mm2). (d) Spiking success rate at various stimulation frequencies (train duration was 1 s for all frequencies, λ = 470 nm, P = 0.45 mW/mm2, n = 12 neurons for each data point).
Whole-cell patch-clamp recordings were utilised to determine whether channelrhodopsin was successfully transfected into cultured neurons and to assess whether transfected cells remained functionally viable. Recordings from transfected (YFP positive) and non-transfected (YFP negative) neurons revealed no significant difference in intrinsic membrane properties (transfected: Input resistance Rn = 493.2 ± 55.0 MΩ; Whole cell capacitance Cm = 19.25 ± 2.0 pF; Resting membrane potential Vm = −59.6 ± 1.2 mV; non-transfected: Rn = 395.6 ± 98.7 MΩ; Cm = 24.2 ± 1.8 pF; Vm = −57.8 ± 6.2 mV) or the ability to fire action potentials (data not shown). Thus, of particular importance to electrophysiologists, laser transfection had no adverse effect on neuronal function. Voltage clamp recordings of transfected cells revealed that blue light (λ = 470 nm, P = 0.45 mW/mm2) induced a rapid depolarizing current, tightly locked to exposure to light (Fig. 5b). The light-induced current included an initial transient component with a peak current of 928.8 ± 244.3 pA (n = 12 cells), and a steady state current of 332.5 ± 109.5 pA (n = 12 cells; Fig. 5c). Current clamp recordings demonstrated that light-induced currents were able to depolarize transfected cells above the threshold for action potential firing. Action potentials could be reliably stimulated by 10 ms light pulses delivered at a frequency of 5 Hz (94% spike success rate) or 10 Hz (86% success rate), but less reliably during 20 Hz stimulation (20% success rate) (Fig. 5d). The observed lower percentage of successful spikes at 20 Hz is consistent with the earlier studies on primary neurons in vitro virally transfected with the first generation ChR224 and most likely results from the high photocurrent and slow kinetics of ChR2(H134R)35. Blue light also induced repetitive firing in all cells which had the capacity to fire multiple times in response to a single current stimulus. Negative controls from non-fluorescent cells in transfected cultures (n = 6 cells) and cells from negative control cultures in which transfection was not performed (n = 6) showed no response to light. These measurements confirm that laser mediated optical transfection is capable of cell selective delivery of an opsin-encoding plasmid and the expressing cells have normal electrophysiological properties and can be used for subsequent optogenetic studies.
Discussion
In this study, we have demonstrated that femtosecond optical transfection offers fast and single cell selective DNA gene delivery into primary cortical neurons with >10% efficiency. It is important to note that optical transfection uses naked DNA which not only greatly simplifies the experiment (circumventing the need to create viral, chemical or mechanical vectors) but would also limit potential side-effects, such as an immune response. As such, it is safer and less controversial in the context of its potential use in gene therapy in humans. We also demonstrate that unlike many other transfection technologies this methodology is highly adaptable such that it is possible to use it to delivery either multiple genes, the ability to target single cells within a population of cells, but also easily transfect different cells with different plasmids. Furthermore we have shown that within the tested range of DIV2-DIV25 the age of the neurons is not a barrier to transfection using this technique, and that it does not interfere with their normal physiology. By combining it with optogenetics it is possible to develop a wholly optical methodology to explore neuronal circuits. At the current stage this technique provides a faster and less invasive alternative to the more traditional single cell transfection techniques and is a valuable extension of the neurophotonics toolbox.
As described, the wholly optical basis of this transfection technique makes it a convenient addition to the majority of existing upright and inverted microscopy systems enabling its easy integration with advanced multimodal live cell microscopy and micromanipulation techniques. Importantly, femtosecond optical transfection takes advantage of the popularity and wide availability of two-photon laser scanning imaging systems36 in neurobiology laboratories. Many multiphoton scanning systems are capable of stably positioning their NIR laser beam and when operated at higher power can be used for optical transfection with little modification. In such systems femtosecond optical transfection not only offers a fully optical, non-contact method of genetic modification and labeling of single neural cells but also improves the speed and throughput of single cell transfection by at least two orders of magnitude, matching the ease of cell labeling and manipulation with that of two photon imaging. Moreover, the deep penetration of multiphoton NIR beam opens up the exciting prospect of simultaneous transfection and multiphoton imaging in deep tissue.
Image guided single cell transfection in neural tissue is most commonly performed using two-photon fluorescence imaging of genetically labeled cells37 or ‘shadow imaging' in which the extracellular space is stained with a membrane-impermeable fluorescent dye14. Both these techniques are fully compatible with femtosecond optical transfection and the same NIR laser beam can be used for both imaging and transfection. Moreover, optical transfection can be easily integrated with recently developed spatial light modulator (SLM) based microscopy38 with the advantage of providing highly flexible beam shaping. It is also important to note that image-guided femtosecond optical transfection has been demonstrated to work through an imaging fiber bundle in an endoscopic system39. This paves the way for deep tissue cell selective gene delivery.
Our study shows that femtosecond optical transfection applied to primary neurons is most efficient when multiple poration sites across the cell are created by multiple irradiation. In the data presented, we used a fast motorized steering mirror to translate the laser spot and irradiate in a sequence. This multi-site irradiation can be performed in parallel by splitting the beam into multiple beamlets, thus creating multiple focal spots. We also show that this can be conveniently implemented in a reconfigurable manner by using diffractive optics. Supplementary Figure S1a shows an extended version of the optical transfection system described above that includes an SLM. By displaying a pre-calculated diffractive pattern this optical element can dynamically shape, translate and multiplex the laser beam40 (Supplementary Fig. S1). This capability has been widely used not only in biophotonics for holographic optical trapping but also enabled highly controlled photostimulation and imaging of neurons with subcellular precision26,38. Importantly, a SLM introduces precise three-dimensional beam positioning within the sample, which enables in depth single cell laser poration41 and can be used to enhance membrane poration efficiency42. Provided that the total available power is sufficient, beam multiplexing on an SLM can be also used to irradiate multiple sites in parallel, thus significantly decreasing the total irradiation time per cell. Supplementary Figure S1c shows how the total required power, irradiation time and achievable throughput change with number of multiplexed spots assuming irradiation conditions equivalent to the single beam optimized parameters used in the present study. With 40 ms irradiation time the theoretical achievable throughput reaches 24 cells per second, which in practice means that it is only limited by the speed of imaging, other mechanical parts of the microscope and cell identification algorithms. However, if reconfigurability is not necessary and the optimum irradiation pattern is established a permanent phase mask element can be used in place of the SLM. This significantly decreases the cost and complexity of the system and may improve power efficiency of the optical setup.
As indicated above by combining the use of a fast steering mirror and user-friendly software (LabView) we developed a touch-screen interface where a rapid “point-and-transfect” targeting of selected cells upon a touch of a finger can be achieved (Fig. 2, Supplementary Video 1). We surmise that this powerful approach of cell targeting and membrane poration is the easiest and most robust single cell transfection technology available.
The combination of image guided single cell operation and relatively high throughput makes optical transfection an ideal tool for genetic dissection of neural circuits43. In particular, this technique will be of special relevance to studies that require high-throughput single-cell specific optogenetic functionalization of neurons at the transfection rather than photoexcitation stage, such as in vivo studies on awake animals where implanted optics significantly limit the options for more sophisticated illumination. When combined with imaging of neural activity, femtosecond optical transfection would also enable tracing of post-synaptic activation triggered by an arbitrarily selected population of cells. This can be done in combination with functional magnetic resonance imaging (fMRI) using the opto-fMRI approach44 or with traditional calcium45 or membrane voltage46 imaging, especially in vivo with the recent advances in miniaturized microscopes47. Other potential uses for this novel technology come from combining it with genetically encoded calcium or voltage indicators48 or the “programming” of neural circuits. With regard to the latter, as indicated within a few minutes the NIR laser beam can quickly and precisely genetically modify the function of hundreds or thousands of selected cells, for example making them light-controllable using various types of opsins. When combined with the recent advances in wavelength-multiplexed optogenetic toolbox49 it paves the way for multifunctional light driven programmable neural circuits.
In this study we have demonstrated that femtosecond optical transfection offers fast and single cell selective gene delivery of primary neurons. It can be used for delivery of multiple genes and enables cell selective optogenetics. At the current stage this technique provides a faster and less invasive alternative to the more traditional single cell transfection techniques and is a valuable extension of the neurophotonics toolbox. As it can be easily and cost-effectively combined with the ubiquitous multiphoton microscopy systems it has a great chance of becoming key in the next generation of studies on function and morphology of neural circuits required by the ambitious large-scale brain activity mapping projects50.
Methods
Femtosecond optical transfection setup
The optical transfection system shown in Figure 1a was based on a motorized inverted microscope (Nikon, TE2000-E) which provided brightfield, phase contrast (PC), differential interference contrast (DIC) and epi-fluorescence imaging. The microscope was equipped in two epi-fluorescence carousels using a stage raising expansion kit (Nikon). The infrared optical transfection beam (800 nm, 200fs @ 80 MHz generated by Coherent MIRA900 pumped by Verdi-V5) from the laser output was expanded using a 4x beam expander (BE) and directed to a computer-controlled piezo mirror (PI, S-334.2). The plane of this beam steering mirror was relayed to the back focal plane of the microscope objective (Nikon CFI Achromat, 60x, NA = 0.8, air) using a 2x expanding telescope (comprising two lenses with focal lengths of f1 = 100 mm and f2 = 200 mm). The beam was coupled into the microscope objective using an NIR-reflecting dichroic mirror (Chroma, 675 DCSPXR) placed in the upper carousel of the microscope. The plane of the piezo mirror and the back focal plane of the objective were optically conjugate so that the lateral position of the focal spot in the sample plane could be controlled by tilt of the mirror without any beam aberrations. Beam power at the sample was controlled using a set of polarizing beam splitter (Newport, 10FC16PB.5) and a zero order half-wave plate (Thorlabs RSP1/M) placed at the output of the laser. A fast shutter (NM Laser Products, LST200IR with CX2250 Controller) placed between the beam splitter and beam expander was used to control the irradiation time with 1 ms precision.
Cell culture
Embryonic rat cortical neurons (E18) dissociated from Fisher 344 rat embryos were obtained from Life Technologies (Life Technologies, A10840-01) and cultured following the manufacturer's protocol. In brief, cryopreserved neurons were quickly thawed and plated at a density of 1 × 105 cells on poly-D-lysine (4.5 μg/cm2) - coated glass-bottom 35 mm Petri dishes (11 mm glass well, Fluorodish, WPI) and maintained in complete Neurobasal™ minus Phenol Red (Life Technologies, 12348-017) supplemented with 2% B-27 Supplement (Life Technologies, 17504044) and 0.5 mM GlutaMAX™-I (Life Technologies, 35050-061). The Petri dishes were placed in an incubator at 37°C with a humidified atmosphere of 5% CO2 in air. 50% of culture medium was slowly and gently replaced with fresh medium (same composition) 20 hours after plating and then every 3 days.
Protocol of optical transfection
Neurons were optically transfected around 24 hrs after the routine 50% medium exchange. First, transfection medium was prepared by mixing plasmid DNA with Nupherin-Neuron (Enzo Life Sciences, BML-SE225-0003) at 1 μg:10 μl DNA:Nupherin ratio, following manufacturer's protocol. Final plasmid DNA concentration of 30 μg/ml was obtained by dilution in pre-equilibrated pure Neurobasal medium without Phenol Red. In co-transfection studies concentration of each plasmid was 30 μg/ml.
Culture medium was gently removed from a Petri dish and stored in the incubator (37°C, 5% CO2 for the duration of transfection step). Cells were gently washed once with pre-equlibrated Neurobasal (minus Phenol Red) medium and 150 μl of transfection medium was added to the well. Cells were transported to the optical transfection microscope (with temperature control environmental chamber set at 37°C) and sequentially irradiated. After a desired number of cells were irradiated (in a time not longer than 20 minutes to avoid cytotoxicity) transfection medium was gently removed and cells were washed once with pre-equilibrated complete culture medium. Subsequently, 250 μl of stored pre-conditioned culture medium was added to the culture dish and topped up with 250 μl of fresh pre-equilibrated complete culture medium. Cells were incubated for 24–48 hours before fluorescence imaging. Each data point presented in Figure 3 is the mean (±S.E.M) of n≥6 separate experiments with 50 cells irradiated in each.
Plasmid DNA preparation
All used plasmids: pCAG-YFP (Add-gene, 11180, Principal Investigator: C. Cepko51), pCAG-DsRed (Add-gene, 11151, PI: C. Cepko51), pDsRed2-Mito (Clontech) and pcDNA3.1/hChR2(H134R)-EYFP49 (kindly provided by K. Deisseroth, Stanford University) were prepared using QIAGEN Plasmid Maxi Kit (QIAGEN, 12162) and stored at −20°C. Typically DNA was used at a concentration of 30 μg/ml.
Electrophysiology
Whole cell patch clamp recordings were performed on cultured neurons visualised using infrared differential interference contrast microscopy. Cells were kept at room temperature and perfused with oxygenated (95% oxygen, 5% CO2) artificial cerebral spinal fluid: (in mM) NaCl 127, KCl3, NaH2PO4 1.25, MgCl2 1, CaCl2 2, NaHCO3 26, glucose 10. Neither culture medium nor recording solution were supplemented with all-trans retinal. Although photo-switchability of ChR2 depends on the presence of all-trans retinal it has been previously shown24 that rodent primary neuronal cultures do not need supplementation, especially when culture medium supplement B-27 is used. Patch electrodes with resistance of approximately 4 MΩ were pulled on a horizontal puller (Sutter Instrument, Novato, CA) from borosilicate glass (World Precision Instruments, Sarasota, FL) and filled with an internal solution containing (in mM): KMeSO4 140, NaCl 10, CaCl2 1, HEPES 10, EGTA 1, Mg-ATP 3. Patch-clamp signals were amplified and filtered (4-kHz low-pass Bessel filter) with a MultiClamp 700B amplifier (Molecular Devices) and acquired at ≥10 kHz using a Digidata 1440A A/D board and pClamp software (Molecular Devices). Data were analysed using Clampfit software (Molecular Devices). For each data point n>10 neurons were analyzed.
Optical stimulation
Wide-field stimulation of patch clamped neurons was performed using an LED illumination source (CoolLED, pE-100, central wavelength of 470 nm) attached to the epi-fluorescence illumination port of the microscope. Optical stimulation was synchronized with the electrophysiology measurements by external TTL-triggering of the LED source controlled by the pClamp electrophysiology software (Molecular Devices).
Live cell imaging
Transfected cultures were assayed for expression of fluorescent protein using a wide-field epi-fluorescence imaging system equipped with a multi-color LED-based illumination source (Lumencor, Aura) and EMCCD digital camera (Andor, iXon+). FITC and TRITC fluorescence cubes (both from Nikon) together with 475 nm or 542 nm excitation illumination were used to image GFP/YFP and DsRed expressing cells, respectively. Images were acquired and processed using Micro-Manager and ImageJ imaging suite (NIH).
Author Contributions
M.A., K.D. and F.G.M. conceived the experiments. M.A. designed and constructed the optical transfection setup, developed the software and performed optical transfection experiments. M.A. and M.L.T.-M. prepared cell cultures and plasmids. E.W. and G.M. carried out the electrophysiology measurements. M.A., E.W., G.M., K.D. and F.G.M. performed data analysis. M.A., K.D. and F.G.M. wrote the manuscript with input from all other authors.
Supplementary Material
Supplementary information
Supplementary Video 1
Acknowledgments
This work is supported by the UK Engineering Physical Sciences Research Council (EPSRC). M.A. acknowledges the support of an EPSRC-funded “Rising Star” Fellowship and the Scottish Universities Life Sciences Alliance (SULSA). K.D. is a Royal Society Wolfson-Merit Award holder. F.G.M. acknowledges the support of E. Killick, RS MacDonald and The “BRAINS” 600th anniversary appeal.
References
- Koch C. & Segev I. The role of single neurons in information processing. Nat. Neurosci. 3 Suppl1171–1177 (2000). [DOI] [PubMed] [Google Scholar]
- Barlow H. B. Single units and sensation: a neuron doctrine for perceptual psychology? Perception 1, 371–394 (1972). [DOI] [PubMed] [Google Scholar]
- Brecht M., Schneider M., Sakmann B. & Margrie T. W. Whisker movements evoked by stimulation of single pyramidal cells in rat motor cortex. Nature 427, 704–710 (2004). [DOI] [PubMed] [Google Scholar]
- Naldini L. et al. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science 272, 263–267 (1996). [DOI] [PubMed] [Google Scholar]
- Saito T. & Nakatsuji N. Efficient gene transfer into the embryonic mouse brain using in vivo electroporation. Dev. Biol. 240, 237–246 (2001). [DOI] [PubMed] [Google Scholar]
- O'Brien J. A. & Lummis S. C. Biolistic transfection of neuronal cultures using a hand-held gene gun. Nat. Protoc. 1, 977–981 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Picciotto M. R. & Wickman K. Using knockout and transgenic mice to study neurophysiology and behavior. Physiol. Rev. 78, 1131–1163 (1998). [DOI] [PubMed] [Google Scholar]
- Teschemacher A. G. et al. Targeting specific neuronal populations using adeno- and lentiviral vectors: applications for imaging and studies of cell function. Exp. Physiol. 90, 61–69 (2005). [DOI] [PubMed] [Google Scholar]
- Wickersham I. R. et al. Monosynaptic restriction of transsynaptic tracing from single, genetically targeted neurons. Neuron 53, 639–647 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aronoff R. & Petersen C. C. Layer, column and cell-type specific genetic manipulation in mouse barrel cortex. Front. Neurosci. 2, 64–71 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasri N. N., Govek E. E. & Van Aelst L. Characterization of oligophrenin-1, a RhoGAP lost in patients affected with mental retardation: lentiviral injection in organotypic brain slice cultures. Methods Enzymol. 439, 255–266 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rancz E. A. et al. Transfection via whole-cell recording in vivo: bridging single-cell physiology, genetics and connectomics. Nat. Neurosci. 14, 527–532 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nolkrantz K. et al. Electroporation of single cells and tissues with an electrolyte-filled capillary. Anal. Chem. 73, 4469–4477 (2001). [DOI] [PubMed] [Google Scholar]
- Kitamura K., Judkewitz B., Kano M., Denk W. & Hausser M. Targeted patch-clamp recordings and single-cell electroporation of unlabeled neurons in vivo. Nat. Methods 5, 61–67 (2008). [DOI] [PubMed] [Google Scholar]
- Haas K., Sin W. C., Javaherian A., Li Z. & Cline H. T. Single-cell electroporation for gene transfer in vivo. Neuron 29, 583–591 (2001). [DOI] [PubMed] [Google Scholar]
- Bestman J. E., Ewald R. C., Chiu S. L. & Cline H. T. In vivo single-cell electroporation for transfer of DNA and macromolecules. Nat. Protoc. 1, 1267–1272 (2006). [DOI] [PubMed] [Google Scholar]
- Steinmeyer J. D. & Yanik M. F. High-throughput single-cell manipulation in brain tissue. PLoS ONE 7, e35603 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevenson D. J., Gunn-Moore F. J., Campbell P. & Dholakia K. Single cell optical transfection. J. R. Soc. Interface 7, 863–871 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tirlapur U. K. & Konig K. Targeted transfection by femtosecond laser. Nature 418, 290–291 (2002). [DOI] [PubMed] [Google Scholar]
- Antkowiak M., Torres-Mapa M. L., Stevenson D. J., Dholakia K. & Gunn-Moore F. J. Femtosecond optical transfection of individual mammalian cells. Nat. Protoc. 8, 1216–1233 (2013). [DOI] [PubMed] [Google Scholar]
- Barrett L. E. et al. Region-directed phototransfection reveals the functional significance of a dendritically synthesized transcription factor. Nat. Methods 3, 455–460 (2006). [DOI] [PubMed] [Google Scholar]
- Sul J. Y. et al. Transcriptome transfer produces a predictable cellular phenotype. Proc. Natl. Acad. Sci. USA 106, 7624–7629 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown M. D., Schatzlein A. G. & Uchegbu I. F. Gene delivery with synthetic (non viral) carriers. Int. J. Pharm. 229, 1–21 (2001). [DOI] [PubMed] [Google Scholar]
- Boyden E. S., Zhang F., Bamberg E., Nagel G. & Deisseroth K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 8, 1263–1268 (2005). [DOI] [PubMed] [Google Scholar]
- Zhang F., Wang L. P., Boyden E. S. & Deisseroth K. Channelrhodopsin-2 and optical control of excitable cells. Nat. Methods 3, 785–792 (2006). [DOI] [PubMed] [Google Scholar]
- Papagiakoumou E. et al. Scanless two-photon excitation of channelrhodopsin-2. Nat Meth 7, 848–854 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrasfalvy B. K., Zemelman B. V., Tang J. & Vaziri A. Two-photon single-cell optogenetic control of neuronal activity by sculpted light. Proc. Natl. Acad. Sci. U. S. A. 107, 11981–11986 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prakash R. et al. Two-photon optogenetic toolbox for fast inhibition, excitation and bistable modulation. Nat. Methods, (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Judkewitz B., Rizzi M., Kitamura K. & Hausser M. Targeted single-cell electroporation of mammalian neurons in vivo. Nat. Protoc. 4, 862–869 (2009). [DOI] [PubMed] [Google Scholar]
- Wang J., Hasan M. T. & Seung H. S. Laser-evoked synaptic transmission in cultured hippocampal neurons expressing channelrhodopsin-2 delivered by adeno-associated virus. J. Neurosci. Methods 183, 165–175 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mthunzi P., Dholakia K. & Gunn-Moore F. Photo-transfection of mammalian cells using femtosecond laser pulses: optimisation and applicability to stem cell differentiation. J. Biomed. Opt. 15, 041507 (2010). [DOI] [PubMed] [Google Scholar]
- Rudhall A. P. et al. Exploring the ultrashort pulse laser parameter space for membrane permeabilisation in mammalian cells. Sci Rep 2, 858 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaech S. & Banker G. Culturing hippocampal neurons. Nat. Protoc. 1, 2406–2415 (2006). [DOI] [PubMed] [Google Scholar]
- Nagel G. et al. Light activation of channelrhodopsin-2 in excitable cells of Caenorhabditis elegans triggers rapid behavioral responses. Curr. Biol. 15, 2279–2284 (2005). [DOI] [PubMed] [Google Scholar]
- Lin J. Y. A user's guide to channelrhodopsin variants: features, limitations and future developments. Exp. Physiol. 96, 19–25 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denk W., Strickler J. H. & Webb W. W. Two-photon laser scanning fluorescence microscopy. Science 248, 73–76 (1990). [DOI] [PubMed] [Google Scholar]
- Margrie T. W. et al. Targeted whole-cell recordings in the mammalian brain in vivo. Neuron 39, 911–918 (2003). [DOI] [PubMed] [Google Scholar]
- Nikolenko V., Poskanzer K. E. & Yuste R. Two-photon photostimulation and imaging of neural circuits. Nat. Methods 4, 943–950 (2007). [DOI] [PubMed] [Google Scholar]
- Ma N., Gunn-Moore F. & Dholakia K. Optical transfection using an endoscope-like system. J. Biomed. Opt. 16, 028002 (2011). [DOI] [PubMed] [Google Scholar]
- Čižmár T. et al. Generation of multiple Bessel beams for a biophotonics workstation. Opt. Express 16, 14024–14035 (2008). [DOI] [PubMed] [Google Scholar]
- Torres-Mapa M. L. et al. Integrated holographic system for all-optical manipulation of developing embryos. Biomed. Opt. Express 2, 1564–1575 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Antkowiak M., Torres-Mapa M. L., Gunn-Moore F. & Dholakia K. Application of dynamic diffractive optics for enhanced femtosecond laser based cell transfection. J. Biophotonics 3, 696–705 (2010). [DOI] [PubMed] [Google Scholar]
- Luo L., Callaway E. M. & Svoboda K. Genetic dissection of neural circuits. Neuron 57, 634–660 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desai M. et al. Mapping brain networks in awake mice using combined optical neural control and fMRI. J. Neurophysiol. 105, 1393–1405 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterlin Z. A., Kozloski J., Mao B. Q., Tsiola A. & Yuste R. Optical probing of neuronal circuits with calcium indicators. Proc. Natl. Acad. Sci. U. S. A. 97, 3619–3624 (2000). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterka D. S., Takahashi H. & Yuste R. Imaging voltage in neurons. Neuron 69, 9–21 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh K. K. et al. Miniaturized integration of a fluorescence microscope. Nat. Methods 8, 871–878 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tian L. et al. Imaging neural activity in worms, flies and mice with improved GCaMP calcium indicators. Nat. Methods 6, 875–881 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang F. et al. Multimodal fast optical interrogation of neural circuitry. Nature 446, 633–639 (2007). [DOI] [PubMed] [Google Scholar]
- Alivisatos A. P. et al. Neuroscience. The brain activity map. Science 339, 1284–1285 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuda T. & Cepko C. L. Electroporation and RNA interference in the rodent retina in vivo and in vitro. Proc. Natl. Acad. Sci. U. S. A. 101, 16–22 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary information
Supplementary Video 1