Abstract
Background
Dual dependence on alcohol and nicotine is common, with many reports suggesting that more than 80% of alcoholics also smoke cigarettes. Even after cessation of alcohol consumption, many recovering alcoholics continue to smoke. In this exploratory study, we examined how current smoking and a history of alcoholism interacted in relation to brain volumes and neuropsychological performance.
Methods
Participants were 14 abstinent long-term alcoholics (seven current smokers and seven nonsmokers), and 13 nonalcoholics (six current smokers and seven nonsmokers). The groups were equivalent in age, gender, education, and intelligence quotient. Two multiecho magnetization-prepared rapid acquisition with gradient echo (MP-RAGE) scans were collected for all participants using a 3T magnetic resonance imaging scanner with a 32 channel head coil. Brain volumes for each gray and white matter region of interest were derived using FreeSurfer. Participants completed a battery of neuropsychological tests measuring intelligence quotient, memory, executive functions, personality variables, and affect.
Results
Compared to nonsmoking nonalcoholics, alcoholics who smoke (the comorbid group) had volumetric abnormalities in: pre- and para-central frontal cortical areas and rostral middle frontal white matter; parahippocampal and temporal pole regions; the amygdala; the pallidum; the ventral diencephalic region; and the lateral ventricle. The comorbid group performed worse than nonsmoking nonalcoholics on tests of executive functioning and on visually-based memory tests. History of alcoholism was associated with higher neuroticism scores among smokers, and current smoking was associated with higher sensation seeking scores and lower extraversion scores among nonalcoholics.
Conclusion
Results from this exploratory study support and extend prior reports showing that alcoholism and smoking, alone and in combination, are associated with structural brain abnormalities and poorer performance on neuropsychological tests. Therefore, it is important to consider smoking status in alcoholism studies and vice versa.
Keywords: MRI, morphometry, neuropsychology, tobacco, alcohol
Introduction
Cigarette smoking among alcoholics occurs at a considerably higher rate than in the general population. Historically, cigarette smoking rates among heavy drinkers were consistently as high as 90%, although this number has decreased since the early 1990s.1 In 2011, 19% of Americans in the general population were regular smokers.2 Among currently drinking alcoholics, the rate is over twice as high at 45%, and it is as high as 80% in treatment-seeking populations.3 While rates of tobacco use and nicotine dependence vary among alcohol abusers and those who are alcohol dependent,4 it is generally the case that individuals who are the heaviest drinkers also are the heaviest smokers.5 Similarly, smoking alcoholics consume alcohol more frequently than nonsmoking and former-smoking alcoholics,6 and the correlation between smoking and drinking exists for both use and dependence.7 Addressing cigarette smoking in treatment for alcoholism provides an opportunity for improved treatment outcomes, as reductions in smoking are associated with greater success in cessation of alcohol abuse.8
Alcohol and smoking have differential and synergistically harmful effects on many systems in the body, but alcoholics are more likely to die from smoking-related health problems than from those associated with alcohol abuse.9 Chronic smoking and chronic drinking are known to be associated with neurocognitive deficits and brain injury.10–15 As such, it has become clear that studies investigating the effects of either alcohol abuse or cigarette smoking cannot be properly interpreted without information about the use of both substances by the participants studied.
While gray and white matter tissue loss and enlarged ventricles have been well-established in association with long-term alcoholism,16–18 cigarette smoking has been identified as a highly relevant confound in the alcoholism neuroimaging literature.14,15 Both alcoholism and smoking have been shown to be associated with abnormal volumes in numerous brain regions, but many alcoholism studies have failed to report or control for the contribution of cigarette smoking. We have identified ten brain areas that have been shown to be impacted both by alcoholism and by smoking (see Table 1 for references to studies reporting abnormalities for each area). Therefore, we examined the differential and interacting contributions of each condition within these regions. Of the ten areas, six of these areas are cortical gray matter, including prefrontal cortex,14,17,19–24 precentral (motor) cortex,25–27 anterior cingulate cortex,21,25,28,29 the insula,17,21–25 inferior temporal/lingual cortex,21,23,25,26 and superior temporal cortex.21,23,26 Subcortical gray matter areas include the nucleus accumbens5,17,30 and the thalamus.21–23,27,29,31 The primary white matter structure is the corpus callosum.16,21,32–35 Finally, abnormalities of the cerebellum also have been reported in association with alcoholism21 and smoking.23,26,36 Generally, volumetric reductions in these regions were reported in alcoholic and smoking populations,11 but some studies would suggest that nicotine has a protective effect,37 or that larger regions might represent a risk factor for nicotine addiction and/or an effect of chronic nicotine exposure.30
Table 1.
References to studies reporting brain volumetric abnormalities associated with alcoholism and smoking
| Regions | Alcoholism references | Smoking references |
|---|---|---|
| Cortical | ||
| Prefrontal cortex | Pfefferbaum et al19 | Durazzo et al14,15 |
| Cardenas et al20 | Froeliger et al22 | |
| Chanraud et al21 | Gallinat et al23 | |
| Makris et al17 | Zhang et al24 | |
| Motor (precentral) cortex | Fein et al25 | Yu et al26 |
| Almeida et al27 | ||
| Anterior cingulate cortex | Chanraud et al21 | Pan et al28 |
| Fein et al25 | Liao et al29 | |
| Insula | Chanraud et al21 | Froeliger et al22 |
| Makris et al17 | Gallinat et al23 | |
| Fein et al25 | Zhang et al24 | |
| Inferior temporal/lingual cortex | Chanraud et al21 | Gallinat et al23 |
| Fein et al25 | Yu et al26 | |
| Superior temporal cortex | Chanraud et al21 | Gallinat et al23 |
| Yu et al26 | ||
| Subcortical | ||
| Nucleus accumbens | Makris et al17 | Das et al30 |
| Narahashi et al5 | ||
| Thalamus | Gazdzinski et al31 | Liao et al29 |
| Chanraud et al21 | Froeliger et al22 | |
| Gallinat et al23 | ||
| Almeida et al27 | ||
| White matter | ||
| Corpus callosum | Agartz et al32 | Choi et al35 |
| Pfefferbaum et al33,34 | ||
| Chanraud et al21 | ||
| Ruiz et al16 | ||
| Cerebellum | Chanraud et al21 | Yu et al26 |
| Kühn et al36 | ||
| Gallinat et al23 | ||
Note: These six cortical areas, two subcortical structures, the corpus callosum, and the cerebellum, formed the basis of our independently justified research questions, utilizing a planned comparisons statistical approach.
Neuropsychological tests have shown that alcoholics and smokers suffer from deficits in several overlapping domains, including various aspects of memory. For example, alcoholics are impaired on auditory verbal memory tasks such as the Logical Memory subtest of the Wechsler Memory Scale (WMS), which requires participants to remember and recall a short story.38,39 Likewise, smokers perform poorly on other auditory verbal tasks such as a free recall task40 and the Rey Auditory Verbal Learning Task.41 Visual memory, which requires recollection of information seen, also has been found to be impaired in alcoholics (for the Rey-Osterrieth42–44 and WMS Visual Recall45) and in smokers (for the WMS Visual Memory Index46). Finally, working memory, which for the purposes of this paper is defined as the mental function involved in storing and manipulating transitory information (as a component of executive functioning), is impaired in alcoholics and in smokers. For example, poor performance on the working memory tasks of Digit Symbol-Coding has been reported in alcoholics17,39,45,47–51 and smokers.52–54
Processing speed, which may be a component of executive function, can be measured by cancellation tests and has been found to be impaired in conjunction with alcoholism42,48 and with smoking.41,55 Another measure of executive function, the Wisconsin Card Sorting Test (WCST), with its measures of categories completed, perseverative responses, and conceptual responses, has been associated with alcoholism-related deficits21,43,44,48,51,56,57 and smoking-related deficits.58 Visuospatial cognition requires the ability to identify stimuli; locate objects in space; navigate; and conceptualize distances, areas, and volumes. Alcoholics and smokers perform poorly on the Block Design subtest of the Wechsler Adult Intelligence Scale (WAIS), a test that measures visuospatial capacities.40,42,44,51
Finally, scores on the extraversion scale of the Eysenck Personality Questionnaire (EPQ) have been shown to be positively associated with drinking,59 and scores on the extraversion, psychoticism, and neuroticism scales have been positively associated with smoking.60,61
In the present exploratory study, we compared those alcoholics who were smoking while abstaining from alcohol to those who either had quit smoking or never smoked. We sought to answer these questions: What are the independent and combined effects of alcoholism and smoking? Does the presence of a comorbid smoking addiction mask the effects of the alcohol history under investigation? Many research studies have not considered smoking as a variable in investigations of morphological and neuropsychological sequelae of alcoholism (perhaps due to recruitment challenges, project complexity, or statistical power). Thus, if alcoholic participants are observed to be impaired, researchers might misattribute those impairments to the alcohol history when in fact the deficits could be tied to current or past smoking history. To examine this confounding effect, we examined long-term chronic alcoholic participants who had been sober for months or years.
Among alcoholics, cigarettes have been identified as the most widely used substance at 88% prevalence.62 However, many abstinent alcoholics have had experience with illicit drugs. As with nicotine dependence, there also is high comorbidity between alcoholism and illicit drugs. Dawson and colleagues identified rates of past-year illicit drug use exceeding 20% among an emergency department screening group characterized by individuals with at least monthly drinking sessions of four drinks or more.63 Therefore, in order to isolate the effects of cigarette smoking, we excluded participants with any drug history of more than once per week, and those taking any psychiatric medications.
We sought to confirm the morphometric and neuropsychological abnormalities described above, and extend them by examining the effects of alcoholism and smoking together to distinguish the contributions of each. Thus, we examined several independently justified research questions. We expected that the effects of alcoholism and smoking would vary by the absence or presence of the other. That is, we predicted that alcoholism might have differential effects on smokers and nonsmokers, and as such, we planned to explore alcoholism as a factor in smokers and nonsmokers considered separately (regardless of the interaction effect). Likewise, we predicted that smoking would have differential effects on alcoholics and nonalcoholics, and thus, we explored smoking effects in each of those groups separately. Moreover, the specific brain regions and neuropsychological functions we selected to examine were planned based upon regions of interest identified in the literature on alcoholism and smoking (see Table 1). However, we also sought to discover new relationships with additional morphometric and neuropsychological measures as exploratory analyses.
Materials and methods
Participants
This study included 14 abstinent long-term alcoholics (seven current smokers [sAL] and seven nonsmokers [nsAL]), and 13 nonalcoholic controls (six current smokers [sNA] and seven nonsmokers [nsNA]) (see Table 2). Participation was solicited from newspaper and web-based advertisements and from flyers placed in and around the Boston University Medical Campus, the Veterans Affairs Boston Healthcare System, and the Massachusetts General Hospital. This study was approved by the Institutional Review Boards of all participating institutions, and informed consent was obtained from each subject prior to neuropsychological testing and scanning. Participants were reimbursed for time and travel expenses. Neurobehavioral and psychiatric evaluations typically required 6 to 9 hours over 3 or more days. Participants had frequent breaks, and sessions were discontinued and rescheduled if a participant indicated fatigue.
Table 2.
Participant characteristics
| sAL (n=7) | nsAL (n=7) | sNA (n=6) | nsNA (n=7) | Significant outcomes where P<0.05 | |
|---|---|---|---|---|---|
| Age (years) | 51.1 ± 11.2 | 52.7 ± 11.2 | 47.0 ± 7.8 | 50.4 ± 9.8 | |
| Gender (women:men) | 3:4 | 3:4 | 2:4 | 3:4 | |
| Education (years) | 13.2 ± 2.6 | 14.6 ± 1.5 | 14.7 ± 3.3 | 14.3 ± 2.1 | |
| WAIS full scale IQ | 99.0 ± 19.8 | 106.4 ± 16.1 | 105.5 ± 14.8 | 112.3 ± 15.4 | |
| Duration of heavy drinking (years) | 14.7 ± 7.0 | 23.4 ± 11.6 | 0 ± 0 | 0 ± 0 |

|
| Average drinks per day | 10.7 ± 5.5 | 14.5 ± 8.6 | 0.4 ± 0.4 | 0.3 ± 0.4 |

|
| Length of sobriety (years) | 3.9 ± 5.0 | 4.2 ± 7.6 | N/A | N/A | |
| Cigarettes per day | 14.4 ± 4.8 | 0 ± 0 | 6.0 ± 5.5 | 0 ± 0 |

|
| Duration of smoking (years)† | 23.4 ± 15.3 | N/A | 14.2 ± 11.8 | N/A | |
| Total brain volume (cm3) | 1,048.6 ± 147.7 | 1,048.8 ± 114.4 | 1,123.9 ± 101.8 | 1,091.6 ± 142.1 | |
| Race (black:white) | 2:5 | 0:7 | 1:6 | 2:4 | |
| Body mass index | 25.7 ± 4.5 | 30.4 ± 8.4 | 24.6 ± 3.7 | 26.7 ± 4.5 |
Notes: All values are listed as mean ± SD.
Duration of smoking not available for one sNA participant. In purple: comorbid effects (sAL versus nsNA). In red: smoking effects (sAL versus nsAL or sNA versus nsNA). In blue: alcholism effects (sAL versus sNA or nsAL versus nsNA). In green: cross effects of smoking and alcoholism (sNA versus nsAL).
Abbreviations: IQ, intelligence quotient; nsAL, alcoholic participants – currently not smoking; nsNA, nonalcoholic participants – currently not smoking; sAL, alcoholic participants (the comorbid group); sNA, nonalcoholic participants – current smokers; WAIS, Wechsler Adult Intelligence Scale; SD, standard deviation.
Participants underwent a medical history interview and vision testing, plus a series of questionnaires (eg, handedness, alcohol and drug use) to ensure they met inclusion criteria. The groups also were similar in racial and gender distributions and in body mass index (Table 2). In order to minimize confounding effects from illicit drug use, psychoactive drug use, and psychiatric comorbidity, participants were given an extensive battery of screening tests. They performed a computer-assisted, shortened version of the Diagnostic Interview Schedule Version IV64 that provides lifetime psychiatric diagnoses according to the Diagnostic and Statistical Manual Fourth Edition criteria.65 Individuals were excluded from further participation if any source (Diagnostic Interview Schedule scores, hospital records, referrals, or personal interviews) indicated that English was not their first language, or if they had any of the following: Korsakoff’s syndrome; human immunodeficiency virus; hepatitis; cirrhosis; major head injury with loss of consciousness greater than 20 minutes; stroke; epilepsy or seizures unrelated to alcoholism; Hamilton Rating Scale for Depression66 score over 14; major depressive disorder; bipolar I or II disorder; schizoaffective disorder; schizophreniform disorder; schizophrenia; generalized anxiety disorder; or electroconvulsive therapy. All participants reported that they were not currently taking psychiatric medication and had never used illicit drugs more than once a week.
Participants received a structured interview regarding their drinking patterns, including length of abstinence and duration of heavy drinking, ie, the number of years they consumed more than 21 drinks per week (one drink: 355 mL beer, 148 mL wine, or 44 mL hard liquor). A Quantity Frequency Index,67 which roughly corresponds to number of daily drinks, was calculated for each participant. This measure factors the amount, type, and frequency of alcohol usage over the last 6 months (for the nonalcoholic groups), or over the 6 months preceding cessation of drinking (for the alcoholic groups). For one alcoholic participant with a Quantity Frequency Index value lower than three daily drinks, the last 6 months of heavy drinking was used instead. The alcoholic participants met Diagnostic and Statistical Manual Fourth Edition criteria for lifetime alcohol abuse or dependence for a period of at least 5 years, and had abstained from alcohol for at least 4 weeks prior to testing. Inclusion criteria for the currently-smoking group was based on self-reported cigarettes currently smoked per day, and duration of smoking was the number of years that the participants smoked their current amount of cigarettes.
Clinical evaluation and neuropsychological assessment
In order to assess the neuropsychological measures needed for our planned comparisons described in the Introduction, tests of memory, executive function, visuospatial cognition, affect, social cognition (including facial processing), and personality were administered. These assessments included: the WAIS-IV,68 the WMS-IV,69 the EPQ70 for extraversion, neuroticism, and psychoticism, the Hamilton Rating Scale for Depression,66 and the Multiple Affect Adjective Check List71 for depression, anxiety, and sensation seeking. Additionally, the Advanced Clinical Solutions72 (ACS) for the WAIS-IV was administered to assess social perception, affect recognition from faces and prosody, affect naming, and face recognition. Subjects also were given executive function tests sensitive to frontal brain system disruption, including the WCST,73 two measures from the Delis–Kaplan Executive Function System,74 a modified Trail Making Test versions A and B,75 and the Controlled Oral Word Association Test or FAS test.76,77
Magnetic resonance imaging (MRI) acquisition and processing
Because we were particularly interested in identifying regions with abnormalities that have been implicated in alcoholism and in smoking, we investigated regional volumes while taking into account total brain volumes, so as to highlight regions especially susceptible to the effects of alcoholism and smoking relative to the rest of the brain (or those regions especially relevant as risk factors). This approach has the additional benefit of controlling for the brain size differences associated with gender simply due to differences in head size.
MRI scans were obtained at the Martinos Center for Biomedical Imaging at Massachusetts General Hospital on a 3 Tesla Siemens (Munich, Germany) MAGNETOM Trio Tim scanner with a 32 channel head coil. Image acquisitions included two T1-weighted multiecho magnetization-prepared rapid acquisition with gradient echo (MP-RAGE) scans collected for volumetric analysis and averaged to aid in motion correction (TR = 2530 ms, TE = 1.79 ms, 3.71 ms, 5.63 ms, 7.55 ms [RMS average used], flip angle = 7 degrees, field of view = 256 mm, matrix = 256 × 256, slice thickness = 1 mm with 50% distance factor, 176 interleaved sagittal slices, GRAPPA acceleration factor of 2).
Scans were analyzed using the FreeSurfer processing stream version 5.3.0 (https://surfer.nmr.mgh.harvard.edu) in order to obtain the a priori regional brain volumes described in the Introduction. Volumes of cortical gray matter regions were assessed using FreeSurfer’s automated cortical parcellation algorithm.78,79 Subcortical gray matter regions, corpus callosum regions, ventricular volumes, and other congregate total volumes were derived using a segmentation algorithm.80,81 Volumes of cortically-associated white matter regions were defined according to the overlying gyrus as delineated by the Desikan atlas for FreeSurfer.78,82 The FreeSurfer brain segmentation volume (which excludes the brainstem) was used to define total brain volume.
Statistical analyses
As described in the Introduction, our analyses consisted of independent planned comparisons conducted to confirm and extend literature findings within our conceptual model, followed by exploratory analyses to identify novel associations. The planned comparisons were independently justified (by prior literature) research questions, and the second set of analyses were exploratory. Thus, for both planned and exploratory analyses, multiple comparisons corrections were not applied. As such, results obtained from exploratory analyses should be considered as preliminary findings. All statistical analyses were performed using JMP Pro Version 10.0.1 (SAS Institute Inc, Cary, NC, USA).
Brain volume differences were assessed using a 2 × 2 factorial analysis of covariance (ANCOVA) model, with alcohol history, current smoking status, and their interaction as between-group factors, with age and total brain volume included as covariates. Next, six simple effects comparisons of volumetric differences were performed using ANCOVA: nsNA versus sNA, nsNA versus nsAL, nsNA versus sAL, sNA versus nsAL, sNA versus sAL, and nsAL versus sAL. Results are reported in five sections: 1) interaction effects (with component simple effects), 2) the simple contrast of sAL (the comorbid group) with nsNA, 3) the simple effects associated with alcoholism, 4) the simple effects associated with smoking, and 5) differences between nonalcoholics who smoke and alcoholics who do not smoke.
Neuropsychological scores were scaled by age using normative data, so further correction for age effects was unnecessary. For each subtest score, the interaction of alcohol history and current smoking status was examined using a 2 × 2 factorial analysis of variance (ANOVA) model with all three factors (main effect of smoking, main effect of alcoholism, and the interaction between them). Next, significant simple effects of alcohol history and current smoking on neuropsychological performance were identified using independent samples Student’s t-tests for each of the same six planned comparisons among the groups (as performed for the brain volume analyses). Similarly, results are reported using the same five sections as used for brain volume differences: 1) interactions, 2) comorbid effects, 3) alcoholism, 4) smoking, and 5) differences between smoking nonalcoholics and nonsmoking alcoholics.
Effect size percentages were calculated as the absolute difference between the mean scores of the groups divided by the mean score of all the participants:
Effect sizes for significant findings are presented in Tables 3, 4, S1, S2, and S3. Hypothesis test statistics are presented in Tables S1, S2, and S3.
Table 3.
Least squares mean volumes (in cm3) of regions of interest
| Regions | sAL (n=7) | nsAL (n=7) | sNA (n=6) | nsNA (n=7) | Significant outcomes where P<0.05 (effect size %) |
|---|---|---|---|---|---|
| Cortical | |||||
| L caudal middle frontal | 5.82 ± 1.48 | 5.65 ± 1.49 | 6.18 ± 1.63 | 6.18 ± 1.47 | nsAL < sNA (9%) |
| R caudal anterior cingulate | 1.97 ± 0.75 | 1.74 ± 0.76 | 2.31 ± 0.83 | 1.79 ± 0.75 | nsNA < sNA (26%) |
| R caudal middle frontal | 5.63 ± 1.53 | 4.65 ± 1.53 | 5.68 ± 1.68 | 5.43 ± 1.52 | nsAL < sNA (19%) |
| R pars orbitalis* | 2.49 ± 0.51 | 2.45 ± 0.51 | 2.25 ± 0.56 | 2.64 ± 0.50 | sNA < nsNA (16%) |
| R precentral | 11.50 ± 1.48 | 11.89 ± 1.49 | 12.31 ± 1.63 | 12.39 ± 1.47 |
|
| R rostral middle frontal | 13.81 ± 2.39 | 14.82 ± 2.40 | 13.64 ± 2.64 | 13.37 ± 2.38 | nsNA < nsAL (10%) |
| Subcortical | |||||
| L accumbens area | 0.44 ± 0.18 | 0.52 ± 0.18 | 0.44 ± 0.20 | 0.44 ± 0.18 | nsNA < nsAL (19%) |
| R thalamus proper | 6.85 ± 0.93 | 6.89 ± 0.93 | 6.65 ± 1.02 | 7.09 ± 0.92 | sNA < nsNA (6%) |
|
| |||||
| Cortical | |||||
| L fusiform | 9.07 ± 1.93 | 9.59 ± 1.94 | 8.62 ± 2.12 | 9.30 ± 1.92 | sNA < nsAL (10%) |
| L paracentral | 2.88 ± 0.54 | 2.89 ± 0.54 | 3.06 ± 0.59 | 3.46 ± 0.53 |
|
| L parahippocampal | 1.86 ± 0.48 | 2.05 ± 0.49 | 2.19 ± 0.53 | 2.21 ± 0.48 |
|
| L supramarginal | 9.78 ± 2.02 | 10.42 ± 2.03 | 8.56 ± 2.23 | 10.29 ± 2.01 |
|
| L temporal pole | 2.23 ± 0.38 | 2.36 ± 0.39 | 2.35 ± 0.42 | 2.54 ± 0.38 | sAL < nsNA (13%) |
| R entorhinal | 1.78 ± 0.58 | 1.83 ± 0.58 | 1.51 ± 0.63 | 1.87 ± 0.57 |
|
| R superior parietal | 11.48 ± 3.01 | 10.92 ± 3.02 | 11.28 ± 3.32 | 12.97 ± 2.99 | nsAL < nsNA (19%) |
| R temporal pole | 2.28 ± 0.45 | 1.95 ± 0.45 | 2.22 ± 0.50 | 2.22 ± 0.45 |
|
| L total cortex | 207.85 ± 18.61 | 210.00 ± 18.72 | 205.19 ± 20.53 | 212.67 ± 18.51 | sNA < nsNA (4%) |
| Subcortical | |||||
| L pallidum* | 1.37 ± 0.39 | 1.30 ± 0.39 | 1.34 ± 0.43 | 1.68 ± 0.39 |
|
| L ventral diencephalon | 3.28 ± 0.59 | 3.46 ± 0.59 | 3.6 ± 0.65 | 3.72 ± 0.58 | sAL < nsNA (13%) |
| R amygdala | 1.42 ± 0.22 | 1.46 ± 0.23 | 1.41 ± 0.25 | 1.58 ± 0.22 |
|
| R pallidum | 1.44 ± 0.37 | 1.24 ± 0.37 | 1.37 ± 0.41 | 1.46 ± 0.37 | nsAL < nsNA (16%) |
| White matter | |||||
| L inferior parietal WM* | 9.03 ± 1.71 | 10.04 ± 1.72 | 9.73 ± 1.88 | 9.17 ± 1.7 | |
| L middle temporal WM* | 5.05 ± 1.22 | 5.33 ± 1.23 | 5.64 ± 1.35 | 4.87 ± 1.22 | nsNA < sNA (15%) |
| L superior frontal WM* | 17.14 ± 3.39 | 18.23 ± 3.41 | 18.84 ± 3.74 | 16.99 ± 3.37 | |
| R banks of the superior temporal sulcus WM* | 2.63 ± 0.91 | 3.25 ± 0.91 | 3.16 ± 1.00 | 3.04 ± 0.9 |
|
| R caudal anterior cingulate WM | 2.77 ± 0.54 | 2.86 ± 0.55 | 3.08 ± 0.60 | 2.75 ± 0.54 | nsNA < sNA (12%) |
| R caudal middle frontal WM | 5.85 ± 1.36 | 5.24 ± 1.37 | 6.26 ± 1.50 | 6.1 ± 1.35 |
|
| R fusiform WM | 6.45 ± 1.49 | 6.47 ± 1.50 | 6.85 ± 1.64 | 6.92 ± 1.48 | nsAL < sNA (6%) |
| R inferior temporal WM | 5.93 ± 1.21 | 6.29 ± 1.22 | 6.02 ± 1.34 | 5.57 ± 1.2 | nsNA < nsAL (12%) |
| R rostral middle frontal WM | 12.72 ± 2.61 | 13.44 ± 2.63 | 12.66 ± 2.88 | 11.64 ± 2.6 |
|
| Ventricles | |||||
| Fourth ventricle | 1.71 ± 1.26 | 1.72 ± 1.27 | 2.64 ± 1.39 | 1.84 ± 1.26 | sAL < sNA (47%) |
| R inferior lateral ventricle | 0.44 ± 0.31 | 0.24 ± 0.31 | 0.35 ± 0.34 | 0.22 ± 0.31 |
|
Notes: Group effects were identified with age and total brain volume covariates. All values are listed as mean ± SD. Significant findings for a priori regions are listed above the dashed line, and additional significant findings follow.
Significant interactions between smoking and alcoholism, where P<0.05. In purple: comorbid effects (sAL versus nsNA). In red: smoking effects (sAL versus nsAL or sNA versus nsNA). In blue: alcholism effects (sAL versus sNA or nsAL versus nsNA). In green: cross effects of smoking and alcoholism (sNA versus nsAL).
Abbreviations: L, left hemisphere; R, right hemisphere; nsAL, alcoholic participants – currently not smoking; nsNA, nonalcoholic participants – currently not smoking; sAL, alcoholic participants (the comorbid group); sNA, nonalcoholic participants – current smokers; WM, white matter; SD, standard deviation.
Table 4.
Scores of neuropsychological tests
| Neuropsychological test | sAL (n=7) | nsAL (n=7) | sNA (n=6) | nsNA (n=7) | Significant outcomes where P<0.05 (effect size %) |
|---|---|---|---|---|---|
| WMS: designs 1 | 8.4 ± 1.6 | 10.6 ± 1.5 | 10.5 ± 3.0 | 12.4 ± 2.5 |
|
| WMS: designs 1 content | 9.7 ± 2.2 | 9.4 ± 1.3 | 10.2 ± 2.0 | 13.4 ± 3.6 | sAL < nsNA (35%) |
| WMS: designs 1 spatial* | 7.7 ± 2.4 | 12.1 ± 1.5 | 10.3 ± 2.3 | 11.0 ± 1.9 |
|
| WMS: designs II spatial* | 7.9 ± 1.3 | 10.9 ± 1.3 | 11.2 ± 2.6 | 9.4 ± 2.7 | sAL < nsAL (31%) |
| WAIS: cancellation | 9.9 ± 3.6 | 8.3 ± 2.6 | 11.3 ± 1.2 | 9.9 ± 2.6 | nsAL < sNA (31%) |
| WCST: categories completed | 3.7 ± 2.0 | 4.7 ± 2.2 | 4.7 ± 2.4 | 6.0 ± 0.0 | sAL < nsNA (48%) |
| WCST: percent conceptual level responses percentile | 17.0 ± 19.9 | 41.4 ± 28.5 | 33.2 ± 31.2 | 55.4 ± 14.5 | sAL < nsNA (105%) |
| EPQ: extraversion | 7.1 ± 2.3 | 7.4 ± 3.7 | 5.8 ± 2.1 | 9.4 ± 2.4 | sNA < nsNA (48%) |
| EPQ: neuroticism | 6.1 ± 5.2 | 5.7 ± 2.9 | 2.8 ± 2.6 | 1.9 ± 2.3 | nsNA < nsAL (93%) |
|
| |||||
| ACS: faces I* | 6.4 ± 3.0 | 10.3 ± 3.9 | 10.8 ± 2.8 | 9.1 ± 3.6 |
|
| ACS: faces II* | 5.6 ± 3.8 | 10.6 ± 3.0 | 10.5 ± 3.1 | 9.7 ± 3.3 |
|
| ACS: faces spatial | 6.4 ± 2.6 | 9.6 ± 3.1 | 9.7 ± 2.2 | 10.0 ± 3.2 |
|
| MAACL: sensation seeking | 49.9 ± 5.5 | 50.6 ± 9.2 | 53.7 ± 5.2 | 46.3 ± 4.9 | nsNA < sNA (15%) |
Notes: All values are listed as mean ± SD. Significant a priori findings are listed above the dashed line, and additional significant findings are below. All scores on neuropsychological tests are age scaled scores except WCST: categories completed.
Significant interactions between smoking and alcoholism, where P<0.05. In purple: comorbid effects (sAL versus nsNA). In red: smoking effects (sAL versus nsAL or sNA versus nsNA). In blue: alcholism effects (sAL versus sNA or nsAL versus nsNA). In green: cross effects of smoking and alcoholism (sNA versus nsAL).
Abbreviations: ACS, advanced clinical solutions; EPQ, Eysenck Personality Questionnaire; MAACL, Multiple Affective Adjective Check List; nsAL, alcoholic participants – currently not smoking; nsNA, nonalcoholic participants – currently not smoking; sAL, alcoholic participants (the comorbid group); sNA, nonalcoholic participants – current smokers; WAIS, Wechsler Adult Intelligence Scale; WCST, Wisconsin Card Sorting Test; WMS, Wechsler Memory Scale; SD, standard deviation.
Normality assumptions for all analyses were assessed using normal probability plots, and it was determined that only one score, WCST Categories Completed, was not normally distributed. Those scores were rank transformed prior to ANOVA examination of the interaction of smoking and alcoholism, and the Wilcoxon rank sums test was used in place of the t-test. All models reported did not violate homoscedasticity assumptions as determined by Levene’s test.84 For the volume measures, the interactions of covariates (age and brain volume) and group effects were examined to determine if they satisfied the homogeneity-of-regression assumption. Only one model indicated such an interaction: the right white matter underlying the fusiform. Thus, this finding is not included in our results. Outliers were assessed first in regard to the distributions of the outcome measures (volumes and neuropsychological performance), and then leverage points were identified within significant models. There were no outliers within outcome measures greater than three standard deviations from the mean in each group. Leverage points were defined as any individual observation with a Cook’s distance above 1.0, for each analysis separately.85 There were five leverage points identified above 1.0. When the participant exerting leverage for right ventral diencephalon was removed, the group difference (sAL versus sNA) in volumes no longer was significant, and thus, this contrast was not included in our results. Similarly, when a leverage point for the subcortical gray matter volume was removed, the group difference (nsAL versus nsNA) in volume no longer was significant, and thus, this contrast was not included in our results. For all other results, when the participant exerting the leverage point was removed, all models remained significant. Thus, all statistics reported included all observations.
Results
Participant characteristics
The extent to which the smoking and alcoholism groups differed on demographics and other characteristics is summarized in Tables 2 and S1. None of the groups varied significantly by age, gender, race, education, WAIS Full Scale IQ, nor total brain volume. By definition, both of the smoking groups (sAL and sNA) smoked more than both of the nonsmoking groups (nsNA and nsAL). Likewise, both of the alcoholic groups (nsAL and sAL) drank more heavily and for a longer duration than nonalcoholics (sNA and nsNA). The alcoholic smoking group smoked an average of 8.4 more cigarettes per day compared to the nonalcoholic smoking group.
Brain volume regions of interest
We evaluated several regional volumes of interest: cortical regions, subcortical gray matter, cortically associated white matter regions, the corpus callosum, and the cerebellum, as specified in the Introduction and Methods sections. Within the cortical areas, we observed significant results for the following regions: four prefrontal areas (left and right caudal middle frontal, right pars orbitalis, right rostral middle frontal), the right precentral cortex, and the anterior cingulate cortex (caudal portion). Within the subcortical structures, we observed significant results for the left nucleus accumbens and the right thalamus. Results are summarized in Tables 3 and S2, and Figure 1, and are described below.
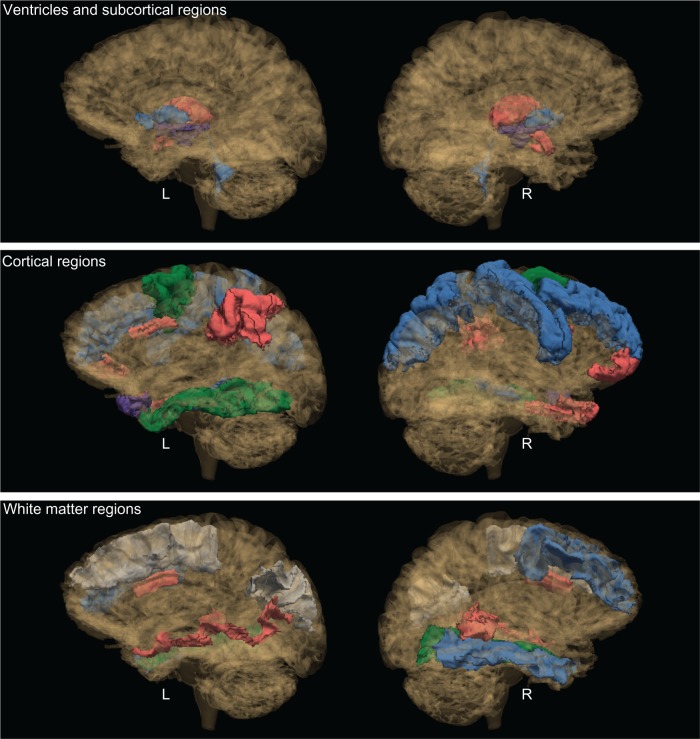
Figure 1.
Significant group contrasts for brain regions.
Notes: See video link for 3D images: http://www.youtube.com/watch?v=ttsTmoqnbig. Purple: comorbid effects (sAL versus nsNA). Red: smoking effects (sAL versus nsAL or sNA versus nsNA). Blue: alcoholism effects (sAL versus sNA or nsAL versus nsNA). Green: cross effects of smoking and alcoholism (sNA versus nsAL). The above brain was chosen from a random nonsmoking nonalcoholic participant. The color scheme of the regions was chosen as follows: A region is colored according to the above color scheme. If multiple group contrasts were significant for a particular region, the color corresponding to the largest effect (smoking or alcoholism) is displayed in the figure. A region is colored purple only if the red or blue contrasts are not present. The cross effects (sNA versus nsAL) are displayed if no other contrasts were significant. Interactions of group effects were significant for two regions: the left pallidum and the white matter underlying the right banks of the superior temporal sulcus. For these regions, the group contrast with the larger effect size is displayed in the figure.
Abbreviations: L, left hemisphere; R, right hemisphere; nsAL, alcoholic participants – currently not smoking; nsNA, nonalcoholic participants – currently not smoking; sAL, alcoholic participants (the comorbid group); sNA, nonalcoholic participants – current smokers.
Volume differences associated with the interaction of alcoholism and smoking
A significant interaction between current smoking status and alcoholism history was indicated for the right pars orbitalis (a prefrontal region). The volumes for sNA were found to be 16% smaller than nsNA, whereas no difference was detected among alcoholics between smokers and nonsmokers in this region.
Volume differences between the comorbid group and nonsmoking nonalcoholic controls
The comorbid group also had 7% smaller right precentral cortex volumes than the nsNA group.
Volume differences associated with alcoholism
Among nonsmokers, the left nucleus accumbens was found to be 20% larger for alcoholics (nsAL versus nsNA). Also among nonsmokers, the rostral middle frontal cortex was 10% larger in association with alcoholism. Likewise, the volume of the right precentral cortex was 7% smaller among sAL than sNA.
Volume differences associated with smoking
Among nonalcoholics, the right thalamus was found to be 6% smaller for smokers (sNA versus nsNA). Conversely, the right caudal anterior cingulate was 26% larger (sNA versus nsNA).
Volume differences between smoking nonalcoholics and nonsmoking alcoholics
For both the left and right hemispheres, the caudal middle frontal cortex was found to be smaller in association with nsAL as compared to sNA (9% on the left and 19% on the right).
Volume differences not confirmed
We did not identify significant interactions or simple effects among the groups on the other brain areas shown to be impacted by alcoholism and/or smoking: the insula, inferior temporal/lingual cortex, superior temporal cortex, the corpus callosum, and the cerebellum.
Exploratory regional brain volume analyses
Volume differences associated with the interaction of alcoholism and smoking
Interactions between current smoking status and alcoholism history were observed for several regions: the left pallidum, along with the white matter associated with the left inferior parietal gyrus, the left middle temporal gyrus, the left superior frontal gyrus, and the right banks of the superior temporal sulcus. The left pallidum was found to be about 25% larger in nsNA than in sAL, nsAL, and sNA. The significant interaction indicated that for the left pallidum, the effects of smoking and alcoholism did not appear to be additive. For the white matter of the left inferior parietal gyrus, the simple effects were not significant, but a significant interaction was observed wherein the nsAL and sNA had the largest volumes. The left middle temporal white matter volume was 15% larger in sNA than nsNA, with the interaction indicating that the effect was smaller for sAL versus nsAL. For the left superior frontal white matter, there were no significant pairwise simple effects, but the significant interaction indicated unexpected opposite effects of smoking and alcoholism: nsAL and sNA had the largest volumes, while sAL and nsNA had smaller volumes. The white matter underlying the right banks of the superior temporal sulcus was about 20% smaller in sAL than both nsAL and sNA, indicating a significant additive effect for this region.
No other interactions were observed. Simple effects for further exploratory results are presented in Table 3.
Neuropsychological performance
In addition to measures of mood and social cognition, we evaluated several a priori domains of neuropsychological function: memory, executive functioning, visuospatial cognition, and personality, as specified in the Introduction and Methods. Within these domains, we observed significant results for the following tests: WMS Designs (four scores), WAIS Cancellation, WCST (two scores), and the EPQ (two scores). Results are summarized in Tables 4 and S3, and Figure 2.
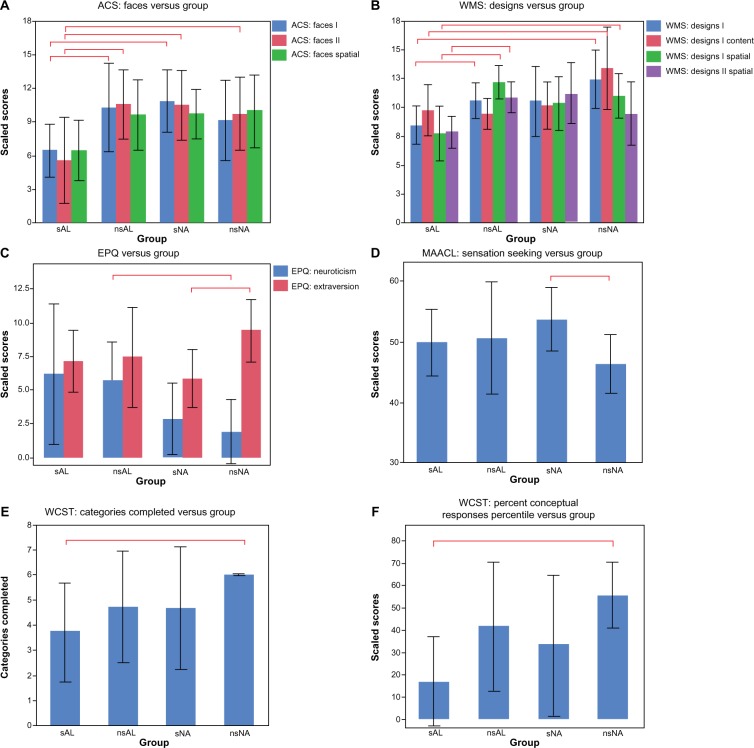
Figure 2.
Shown here are the significant findings of differences in neuropsychological measures.
Notes: The bar graphs show the mean values. Each error bar is constructed using one standard deviation above and below the mean. The red bars comparing the columns indicate significant findings, with P<0.05.
Abbreviations: ACS, advanced clinical solutions; EPQ, Eysenck Personality Questionnaire; MAACL, Multiple Affective Adjective Check List; nsAL, alcoholic participants – currently not smoking; nsNA, nonalcoholic participants – currently not smoking; sAL, alcoholic participants (the comorbid group); sNA, nonalcoholic participants – current smokers; WAIS, Wechsler Adult Intelligence Scale; WCST, Wisconsin Card Sorting Test; WMS, Wechsler Memory Scale.
Differences associated with the interaction of alcoholism and smoking
The interaction effect between alcoholism and smoking was significant for Designs I Spatial and Designs II Spatial (see Table 4 and Figure 2B). For Designs I Spatial (immediate recall), the scores of the sAL group were significantly worse than those of both the nsAL and nsNA groups; sNA scores were in between. The significant interaction indicated a large effect of smoking for alcoholics in comparison to the significantly smaller impairment associated with smoking observed for controls. In other words, there was a large effect of alcoholism for smokers in comparison to the significantly smaller impairment associated with alcoholism observed for nonsmokers. For Designs II Spatial (delayed recall), sAL scored significantly worse than nsAL, and a similar interaction was observed.
Differences between the comorbid group and nonsmoking nonalcoholics
In addition to the effect of smoking described above, Designs I and Designs I Content scaled scores were observed to be about a third lower for the sAL than for nsNA (Table 4). Additionally, several subscores of the WCST showed significant differences between the two groups. All nsNA participants completed all six categories of the test, while the sAL group completed only an average of 3.7 categories (Figure 2E). Conceptual level responses also indicated a strong disadvantage for the sAL group, which scored at the 17th percentile, performing significantly below average, while nsNA scored at the 55th percentile (Figure 2F).
Differences associated with alcoholism
For the EPQ, nsAL were found to be 93% more neurotic than nsNA (Figure 2C).
Differences associated with smoking
For the Designs I subtest of the WMS, nsAL performed 20% better than sAL (Figure 2B). For the EPQ: extraversion, sNA were 48% less extroverted than nsNA (Figure 2C).
Differences between smoking nonalcoholics and nonsmoking alcoholics
For the Cancellation subtest of the WAIS, nsAL performed 31% better than sNA.
Neuropsychological effects not confirmed
The groups did not differ significantly on tests measuring auditory verbal memory (WAIS: Logical Memory I and II), working memory (WAIS: Letter-Number Sequencing and WAIS: Digit Symbol-Coding), or visuospatial abilities (WAIS: Block Design).
Exploratory neuropsychological analyses
Differences associated with the interaction of alcoholism and smoking
The interaction effect between history of alcoholism and current smoking was significant for ACS Faces I and II (see Table 4). For both immediate and delayed recall, the scores for sAL were significantly worse than both nsAL and sNA. For delayed recall, sAL additionally scored significantly worse than nsNA (Figure 2A). In summary, for both scores, the sAL group scored about 50% worse than nsAL and sNA. As was observed for Designs, the interaction indicated a large effect of alcoholism for smokers in comparison to the significantly smaller impairment associated with alcoholism observed for nonsmokers.
No other significant interactions were observed. Simple effects for further exploratory results are presented in Table 4.
Discussion
Both alcoholism and smoking have been shown to be associated with impairments in brain and behavior, as measured by regional brain volumes and neuropsychological performances. However, the literature examining the effects of comorbid addictions, and comparing the effects of alcoholism with those of smoking, is sparse. We examined these effects and found evidence of associations of both alcoholism and smoking with regard to: 1) brain volumes for cortical regions, subcortical regions, gyrally-associated white matter regions, and ventricles; and 2) measures of memory, executive functioning, personality, and social cognition.
A number of our findings confirmed and extended the a priori hypotheses suggested by previous literature.20,31 Exploratory analyses revealed further evidence of a relationship of alcoholism and smoking to brain volume and neuropsychological performance.
Brain morphometry
Comorbidity
In studies by Durazzo et al83 and Gazdzinski et al,31 the alcoholic participants had been abstinent for a brief duration (1 week). By comparison, our alcoholics had been abstinent for a much longer duration (about 4 years). Thus, our findings suggested that the abnormalities continue and are persistent. Nevertheless, findings from all of these studies indicated abnormalities associated with alcoholism and smoking, despite differences in the exact brain regions involved.
Parahippocampal volumes were found to be smaller in the comorbid group (sAL) than in nonalcoholics with or without a smoking history. The parahippocampal gyrus abuts the hippocampus and is a crucial structure for memory processing. The neuropsychological measures involving memory reported here indicated that sAL performed worse on WMS Designs and ACS Faces subtests, which require the ability to remember designs and faces, respectively.
Separate effects of alcoholism or smoking
Although Makris et al reported that the right nucleus accumbens was smaller in alcoholics than in nonalcoholics (smoking was not reported),17 unexpectedly, the present study found that nsAL had larger left nucleus accumbens volumes than nsNA, an effect that was not observed for sAL. The nucleus accumbens is a core component of the mesocorticolimbic reward circuitry, which is important for emotional and motivational functions and for memory processing. Further research could help explain why larger nucleus accumbens volumes would be associated with addiction, eg, predisposition to alcoholism or risk-taking behaviors.86 Our results revealed similar findings in another region: the right rostral middle frontal cortex, a component of the prefrontal cortex. This region was smaller in nonalcoholics compared to alcoholics. Previous research has indicated opposite findings,19 although smoking effects were not examined in the same manner. A study by Fein et al showed that abstinent alcoholics had significantly smaller primary motor cortex compared to normal controls.25 Within our sample, we observed smaller primary motor cortex in association with smoking.
Our results revealed that the right thalamus was smaller in sNA compared to nsNA. This confirmed findings from literature suggesting that smokers had a smaller right thalamus.23,27 It is interesting to note that [3H] nicotine binding studies have shown that the thalamus has the highest density of nicotinic acetylcholine receptors in the human brain.87 Das et al speculated that the highest number of nicotinic receptors are found in the same regions where smokers had smaller volumes, eg, in the cerebral cortex, thalamus, substantia nigra, and striatum.30 Thus, these regions might be more susceptible to damage caused by excessive amounts of nicotine.
Among nonalcoholics, smoking was associated with smaller right pars orbitalis (a component of the prefrontal cortex), a region that has been implicated in language production. Pars orbitalis is part of a well-mapped neural circuitry involved in social cognition.88 In the present study, the results of the social cognition tests that involved identifying faces indicated that among alcoholics, smokers performed worse than nonsmokers. In both cases (volume of the pars orbitalis and scores on ACS Faces I and II), we detected an interaction wherein the combination of smoking and alcoholism was significantly worse than either alone.
Our results indicated that smokers had a larger right caudal anterior cingulate gray matter volume than nonsmokers. This result is in contrast with the finding presented in the meta analysis by Pan et al, which indicated that smokers had a smaller anterior cingulate.28 Only one morphometric study was identified that examined the white matter adjacent to the anterior cingulate,26 and, as in the present study, those researchers reported that smokers had a larger volume of anterior cingulate white matter.
Besides looking at the interaction effects, one way to address whether smoking or alcoholism had a larger effect is to compare smoking nonalcoholics and alcoholics who do not smoke (sNA versus nsAL). For both left and right caudal middle frontal volumes, nsAL had smaller volumes than sNA, implying that alcoholism had more of an effect on these volumes than smoking.
From the exploratory data, we also found that the left and the right pallidum were smaller in nsAL than nsNA. The ventral pallidum is a part of the mesocorticolimbic circuitry, which is involved in motivation and emotion, and activated by environmental stimuli associated with rewards. Further, it has been suggested in the literature that the addictive properties of alcohol are regulated by GABAA1 receptors in the ventral pallidum.89,90 Thus, we may speculate that the binding of ethanol to GABA receptors may result in cytoarchitectonic modifications which cause reduced volumes.
Neuropsychological performance
Comorbidity
The associations of these addictions to abnormalities in brain morphometry occur in conjunction with neuropsychological deficits. Among alcoholics, smoking was associated with a variety of impaired spatial memory functions (as measured by WMS Designs subtests and ACS Faces subtests), and executive functions (as measured by the WCST). The Designs subtests assess the ability to remember correct location (Designs Spatial) and correct detail (Designs Content) of the design immediately after seeing the design (Designs I), and after some delay (Designs II). Our findings indicated that the combination of smoking and alcoholism was associated with worse scores than either alone. Of note, although others also have reported learning and memory deficits associated with comorbidity,20,31 we expanded such findings to include abnormalities in memory for spatial aspects of facial stimuli. Regarding executive functioning, only the comorbid group performed worse on the WCST task for both measures: categories completed and percent conceptual responses. The conceptual response score measures understanding of the sorting principle needed for the task by totaling contiguous responses consisting of three or more correct answers. Thus, these results could be interpreted to mean that abstinent alcoholics who smoke are impaired with regard to the reasoning needed to understand an abstract categorical system.
Separate effects of alcoholism or smoking
The evidence presented regarding personality could provide a partial explanation of the deficits observed. The neuroticism measure derived from the EPQ represents temperament aspects characterized by levels of negative affect, including depression and anxiety. In contrast, the EPQ questionnaire also assesses extraversion, which is characterized by high levels of positive affect, such as talkativeness and outgoing qualities. These two personality traits distinguished smokers from drinkers: alcoholics were found to be more neurotic, while smokers were found to be less extraverted. The exploratory analyses revealed a significant pattern of deficits associated with face discrimination, recognition, and perception, in addition to location information associated with the faces. Taken together, these results suggested that both alcoholism and smoking have an impact in relation to cognitive abilities, mental function, and personality.
Limitations
This is a preliminary retrospective study for which we carefully chose, from our larger sample of alcoholics, those participants who did not differ significantly with respect to demographic characteristics, and who were free from potential confounds (eg, psychiatric medications and history of excessive drug use). Consequently, our sample size was small, and additional participants would be needed to increase the statistical strength of the data and to make strong inferential claims. As such, these results should be considered tentative, interpreted with caution, and confirmed in future studies with larger sample sizes. Some studies have shown that aging effects may be nonlinear,82,91 while our analyses assumed a linear impact of age. Detrimental impacts of smoking and alcoholism can also vary by age,92 but we did not find evidence for this within our sample. Gender effects also exist, and thus, the relationships of smoking and alcoholism should be explored in a sample large enough to explore gender effects.
Despite the specific planned nature of the independent comparisons we made based upon regions of interest identified in the alcoholism and smoking literatures, the exploratory analyses we conducted were not corrected for multiple comparisons. This approach may inflate the possibility of making a Type I error. However, it reveals the effects of alcoholism for smokers separately from the effects of alcoholism for nonsmokers, and thereby provides useful avenues for validation and future research. Additionally, this study is cross-sectional, which limits the causal interpretation of results: the findings may be risk factors, consequences, or caused by other factors (eg, body weight, nutrition, exercise, genetic predispositions, etc).
It was difficult to differentiate the effects of smoking history in alcoholic individuals, because we examined alcoholics who were current smokers at the time of testing. However, based on the qualitative data we had obtained, we determined that many of the currently nonsmoking recovering alcoholics had a history of smoking, whereas the nonsmoking nonalcoholic participants had never smoked. Moreover, the comorbid group smoked more than the nonalcoholic smokers. Because the alcoholic smoking group smoked more cigarettes per day compared to the nonalcoholic smoking group, the brain volume changes and poorer performances in the comorbid group could be attributed either to alcoholism or to the larger quantity of cigarettes. Lifetime smoking is known to be associated with volumetric abnormalities, including increased volumes, which perhaps represent predispositions for addiction.30,93–97
Conclusion
Compared to nonsmoking nonalcoholics, alcoholics who smoke (the comorbid group) had the greatest number of cortical and subcortical gray matter volume abnormalities and neuropsychological deficits. Therefore, we recommend that researchers who study alcoholism should, at a minimum, aim to equate their groups by smoking status, ie, include a similar number of smokers within alcoholic and nonalcoholic groups. Structuring the sample in such a way would address confounding but not interaction effects; these need to be examined independently of group matching. Further, when considered in future studies, the interaction of smoking and alcoholism may elucidate methods for smoking cessation programs or alcohol treatment.98–101
Supplementary tables
Table S1.
Statistical comparisons for alcoholism and smoking
| Measure | Contrast | df | t | P-value | d |
|---|---|---|---|---|---|
| Duration of heavy drinking (years) | sNA < sAL | 6 | 5.55 | 0.001 | 3.35 |
| Duration of heavy drinking (years) | nsNA < nsAL | 6 | 5.33 | 0.002 | 3.08 |
| Duration of heavy drinking (years) | sNA < nsAL | 6 | 5.33 | 0.002 | 3.21 |
| Duration of heavy drinking (years) | nsNA < sAL | 6 | 5.55 | 0.001 | 3.20 |
| Average drinks per day | sNA < sAL | 6.08 | 5.00 | 0.002 | 3.02 |
| Average drinks per day | nsNA < nsAL | 6.02 | 4.35 | 0.005 | 2.51 |
| Average drinks per day | sNA < nsAL | 6.03 | 4.33 | 0.005 | 2.61 |
| Average drinks per day | nsNA < sAL | 6.08 | 5.00 | 0.002 | 2.89 |
| Cigarettes per day | nsAL < sAL | 6 | 7.97 | 0.0002 | 4.60 |
| Cigarettes per day | nsNA < sNA | 5 | 2.67 | 0.04 | 1.61 |
| Cigarettes per day | sNA < sAL | 10.06 | 2.91 | 0.02 | 1.75 |
| Cigarettes per day | nsAL < sNA | 5 | 2.67 | 0.04 | 1.61 |
| Cigarettes per day | nsNA < sAL | 6 | 7.99 | 0.0002 | 4.61 |
Notes: For each contrast, degrees of freedom (df), t, P, and Cohen’s d values are presented. Cohen’s d was calculated with the following formula: 2t/√ (N − 2). In purple: comorbid effects (sAL versus nsNA). In red: smoking effects (sAL versus nsAL or sNA versus nsNA). In blue: alcholism effects (sAL versus sNA or nsAL versus nsNA). In green: cross effects of smoking and alcoholism (sNA versus nsAL).
Abbreviations: nsAL, alcoholic participants – currently not smoking; nsNA, nonalcoholic participants – currently not smoking; sAL, alcoholic participants (the comorbid group); sNA, nonalcoholic participants – current smokers.
Table S2.
Volumetric variables presented within models containing age and total brain volume as covariates
| Regions | Contrast | df1 | df2 | t | P-value | d |
|---|---|---|---|---|---|---|
| Fourth ventricle | sAL < sNA | 1 | 9 | 2.93 | 0.02 | 1.77 |
| R inferior lateral ventricle | nsAL < sAL | 1 | 10 | 2.51 | 0.03 | 1.45 |
| R inferior lateral ventricle | nsNA < sAL | 1 | 10 | 3.59 | 0.005 | 2.07 |
| L accumbens area | nsNA < nsAL | 1 | 10 | 2.33 | 0.05 | 1.35 |
| L pallidum* | sNA < nsNA | 1 | 9 | 3.47 | 0.007 | 2.09 |
| L pallidum* | nsAL < nsNA | 1 | 10 | 4.76 | 0.0008 | 2.75 |
| L pallidum* | sAL < nsNA | 1 | 10 | 2.95 | 0.01 | 1.70 |
| L ventral diencephalon | sAL < nsNA | 1 | 10 | 2.98 | 0.01 | 1.72 |
| R amygdala | sNA < nsNA | 1 | 9 | 2.42 | 0.04 | 1.46 |
| R amygdala | sAL < nsNA | 1 | 10 | 3.91 | 0.003 | 2.26 |
| R pallidum | nsAL < nsNA | 1 | 10 | 2.67 | 0.02 | 1.54 |
| R thalamus proper | sNA < nsNA | 1 | 9 | 2.45 | 0.04 | 1.48 |
| R ventral diencephalon | sAL < nsNA | 1 | 10 | 3.16 | 0.01 | 1.82 |
| Total subcortical gray matter | sAL < nsNA | 1 | 10 | 2.29 | 0.05 | 1.32 |
| L caudal middle frontal | nsAL < sNA | 1 | 9 | 2.70 | 0.02 | 1.63 |
| L fusiform | sNA < nsAL | 1 | 9 | 3.13 | 0.01 | 1.89 |
| L paracentral | nsAL < nsNA | 1 | 10 | 4.00 | 0.003 | 2.31 |
| L paracentral | sAL < nsNA | 1 | 10 | 3.81 | 0.003 | 2.20 |
| L parahippocampal | sAL < sNA | 1 | 9 | 3.50 | 0.007 | 2.11 |
| L parahippocampal | sAL < nsNA | 1 | 10 | 3.39 | 0.007 | 1.96 |
| L supramarginal | sNA < nsNA | 1 | 9 | 2.72 | 0.02 | 1.64 |
| L supramarginal | sNA < nsAL | 1 | 9 | 2.94 | 0.02 | 1.77 |
| L temporal pole | sAL < nsNA | 1 | 10 | 2.97 | 0.01 | 1.71 |
| R caudal anterior cingulate | nsNA < sNA | 1 | 9 | 2.31 | 0.05 | 1.39 |
| R caudal middle frontal | nsAL < sNA | 1 | 9 | 2.89 | 0.02 | 1.74 |
| R entorhinal | sNA < nsNA | 1 | 9 | 2.70 | 0.02 | 1.63 |
| R entorhinal | sNA < nsAL | 1 | 9 | 2.28 | 0.05 | 1.37 |
| R pars orbitalis* | sNA < nsNA | 1 | 9 | 3.18 | 0.01 | 1.92 |
| R precentral | sAL < sNA | 1 | 9 | 2.46 | 0.04 | 1.48 |
| R precentral | sAL < nsNA | 1 | 10 | 2.63 | 0.02 | 1.52 |
| R rostral middle frontal | nsNA < nsAL | 1 | 10 | 2.91 | 0.02 | 1.68 |
| R superior parietal | nsAL < nsNA | 1 | 10 | 3.09 | 0.01 | 1.78 |
| R temporal pole | nsAL < sAL | 1 | 10 | 2.93 | 0.01 | 1.69 |
| R temporal pole | nsAL < nsNA | 1 | 10 | 2.36 | 0.04 | 1.36 |
| L total cortex | sNA < nsNA | 1 | 9 | 2.31 | 0.05 | 1.39 |
| L middle temporal WM* | nsNA < sNA | 1 | 9 | 2.33 | 0.04 | 1.41 |
| R banks of the superior temporal sulcus WM* | sAL < nsAL | 1 | 10 | 2.77 | 0.02 | 1.60 |
| R banks of the superior temporal sulcus WM* | sAL < sNA | 1 | 9 | 2.46 | 0.04 | 1.48 |
| R caudal anterior cingulate WM | nsNA < sNA | 1 | 9 | 2.41 | 0.04 | 1.45 |
| R caudal middle frontal WM | nsAL < nsNA | 1 | 10 | 2.33 | 0.04 | 1.35 |
| R caudal middle frontal WM | nsAL < sNA | 1 | 9 | 2.89 | 0.02 | 1.74 |
| R fusiform WM | nsAL < sNA | 1 | 9 | 2.38 | 0.04 | 1.44 |
| R inferior temporal WM | nsNA < nsAL | 1 | 10 | 2.42 | 0.04 | 1.40 |
| R rostral middle frontal WM | nsNA < nsAL | 1 | 10 | 4.36 | 0.001 | 2.52 |
| R rostral middle frontal WM | nsNA < sAL | 1 | 10 | 2.27 | 0.05 | 1.31 |
Notes: For each contrast, degrees of freedom (df), t, P, and Cohen’s d are presented. Cohen’s d was calculated with the following formula: 2t/√(N – 2)
Significant interactions between smoking and alcoholism, where P<0.05. In purple: comorbid effects (sAL versus nsNA). In red: smoking effects (sAL versus nsAL or sNA versus nsNA). In blue: alcholism effects (sAL versus sNA or nsAL versus nsNA). In green: cross effects of smoking and alcoholism (sNA versus nsAL).
Abbreviations: L, left hemisphere; R, right hemisphere; nsAL, alcoholic participants – currently not smoking; nsNA, nonalcoholic participants – currently not smoking; sAL, alcoholic participants (the comorbid group); sNA, nonalcoholic participants – current smokers; WM, white matter.
Table S3.
Neuropsychological variables
| Neuropsychological test | Contrast | df | t | P-value | d |
|---|---|---|---|---|---|
| ACS: faces I* | sAL < nsAL | 9.65 | 2.23 | 0.05 | 1.29 |
| ACS: faces I* | sAL < sNA | 9.77 | 3.08 | 0.01 | 1.86 |
| ACS: faces II* | sAL < nsAL | 11.4 | 2.74 | 0.02 | 1.58 |
| ACS: faces II* | sAL < sNA | 10.99 | 2.43 | 0.03 | 1.47 |
| ACS: faces II* | sAL < nsNA | 11.79 | 2.18 | 0.05 | 1.26 |
| ACS: faces spatial | sAL < sNA | 10.99 | 2.43 | 0.03 | 1.47 |
| ACS: faces spatial | sAL < nsNA | 11.56 | 2.27 | 0.04 | 1.31 |
| EPQ: extraversion | sNA < nsNA | 10.95 | 2.87 | 0.02 | 1.73 |
| EPQ: neuroticism | nsNA < nsAL | 11.53 | 2.76 | 0.02 | 1.59 |
| MAACL: sensation seeking | nsNA < sNA | 10.44 | 2.60 | 0.03 | 1.57 |
| WAIS-IV: cancellation | nsAL < sNA | 8.7 | 2.75 | 0.02 | 1.66 |
| WCST: categories completed | sAL < nsNA | NA | 2.53 | 0.01 | 0.68 |
| WCST: percent conceptual level responses percentile | sAL < nsNA | 11 | 4.13 | 0.002 | 2.38 |
| WMS-IV: designs I | sAL < nsAL | 11.94 | 2.56 | 0.03 | 1.48 |
| WMS-IV: designs I | sAL < nsNA | 10.26 | 3.55 | 0.005 | 2.05 |
| WMS-IV: designs I content | sAL < nsNA | 10.05 | 2.34 | 0.04 | 1.35 |
| WMS-IV: designs I spatial* | sAL < nsAL | 10.02 | 4.23 | 0.002 | 2.44 |
| WMS-IV: designs I spatial* | sAL < nsNA | 11.51 | 2.86 | 0.01 | 1.65 |
| WMS-IV: designs II spatial* | sAL < nsAL | 12 | 4.17 | 0.001 |
Notes: For each contrast, degrees of freedom (df), t, P, and Cohen’s d are presented. Cohen’s d was calculated with the following formula: 2t/√ (N – 2). For WCST: categories completed, Wilcoxon rank-sum Z is reported in place of t, and r = Z/√N in place of d.
*Significant interactions between smoking and alcoholism, where P<0.05. In purple: comorbid effects (sAL versus nsNA). In red: smoking effects (sAL versus nsAL or sNA versus nsNA). In blue: alcholism effects (sAL versus sNA or nsAL versus nsNA). In green: cross effects of smoking and alcoholism (sNA versus nsAL).
Abbreviations: ACS, advanced clinical solutions; EPQ, Eysenck Personality Questionnaire; MAACL, Multiple Affective Adjective Check List; nsAL, alcoholic participants – currently not smoking; nsNA, nonalcoholic participants – currently not smoking; sAL, alcoholic participants (the comorbid group); sNA, nonalcoholic participants – current smokers; WAIS, Wechsler Adult Intelligence Scale; WCST, Wsconsin Card Sorting Test; WMS, Wechsler Memory Scale.
Acknowledgments
This study was supported by funds from the National Institute on Alcohol Abuse and Alcoholism (NIAAA) grants R01-AA007112 and K05-AA000219 to Dr Marlene Oscar Berman, and the Department of Veterans Affairs Medical Research Service, as well as the Center for Functional Neuroimaging Technologies, P41RR14075. The authors thank all research participants as well as Pooja Parikh, Diane Merritt, Mary M Valmas, and Steven Lehar, who assisted with recruitment, neuropsychological assessment, and MRI data collection. We also thank our collaborator Dr Gordon Harris who provided us mentorship at the Massachusetts General Hospital Athinoula A Martinos Center for Biomedical Imaging.
Footnotes
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Bobo JK, Husten C. Sociocultural influences on smoking and drinking. Alcohol Res Health. 2000;24(4):225–232. [PMC free article] [PubMed] [Google Scholar]
- 2.Adult Cigarette Smoking in the United States: Current Estimate [webpage on the Internet] Atlanta: Center for Disease Control and Prevention; 2011Available from: http://www.cdc.gov/tobacco/data_statistics/fact_sheets/adult_data/cig_smoking/index.htmAccessed July 29, 2013 [Google Scholar]
- 3.Kalman D, Kim S, DiGirolamo G, Smelson D, Ziedonis D. Addressing tobacco use disorder in smokers in early remission from alcohol dependence: the case for integrating smoking cessation services in substance use disorder treatment programs. Clin Psychol Rev. 2010;30(1):12–24. doi: 10.1016/j.cpr.2009.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Falk DE, Yi HY, Hiller-Sturmhofel S. An epidemiologic analysis of co-occurring alcohol and tobacco use and disorders: findings from the National Epidemiologic Survey on Alcohol and Related Conditions. Alcohol Res Health. 2006;29(3):162–171. [PMC free article] [PubMed] [Google Scholar]
- 5.Narahashi T, Soderpalm B, Ericson M, et al. Mechanisms of alcohol-nicotine interactions: alcoholics versus smokers. Alcohol Clin Exp Res. 2001;25(5 Suppl ISBRA):152S–156S. doi: 10.1097/00000374-200105051-00026. [DOI] [PubMed] [Google Scholar]
- 6.John U, Meyer C, Rumpf HJ, Schumann A, Thyrian JR, Hapke U. Strength of the relationship between tobacco smoking, nicotine dependence and the severity of alcohol dependence syndrome criteria in a population-based sample. Alcohol Alcohol. 2003;38(6):606–612. doi: 10.1093/alcalc/agg122. [DOI] [PubMed] [Google Scholar]
- 7.National Institute on Alcohol Abuse and Alcoholism (NIAAA) Alcohol and tobacco. Alcohol Alert. 2007;(No 71):1–6. [Google Scholar]
- 8.Friend KB, Pagano ME. Changes in cigarette consumption and drinking outcomes: findings from Project MATCH. J Subst Abuse Treat. 2005;29(3):221–229. doi: 10.1016/j.jsat.2005.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Littleton J, Barron S, Prendergast M, Nixon SJ. Smoking kills (alcoholics)! shouldn’t we do something about it? Alcohol Alcohol. 2007;42(3):167–173. doi: 10.1093/alcalc/agm019. [DOI] [PubMed] [Google Scholar]
- 10.Durazzo TC, Rothlind JC, Gazdzinski S, Banys P, Meyerhoff DJ. Chronic smoking is associated with differential neurocognitive recovery in abstinent alcoholic patients: a preliminary investigation. Alcohol Clin Exp Res. 2007;31(7):1114–1127. doi: 10.1111/j.1530-0277.2007.00398.x. [DOI] [PubMed] [Google Scholar]
- 11.Glass JM, Adams KM, Nigg JT, et al. Smoking is associated with neurocognitive deficits in alcoholism. Drug Alcohol Depend. 2006;82(2):119–126. doi: 10.1016/j.drugalcdep.2005.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Meyerhoff DJ, Tizabi Y, Staley JK, Durazzo TC, Glass JM, Nixon SJ. Smoking comorbidity in alcoholism: neurobiological and neurocognitive consequences. Alcohol Clin Exp Res. 2006;30(2):253–264. doi: 10.1111/j.1530-0277.2006.00034.x. [DOI] [PubMed] [Google Scholar]
- 13.Oscar-Berman M, Valmas M, Sawyer K, Ruiz SM, Luhar R, Gravitz Z. Profiles of impaired, spared, and recovered processes in alcoholism. In: Pfefferbaum A, Sullivan EV, editors. Alcohol and the Nervous System. New York: Elsevier; 2013. In Press. [Google Scholar]
- 14.Durazzo TC, Meyerhoff DJ, Nixon SJ. Chronic cigarette smoking: implications for neurocognition and brain neurobiology. Int J Environ Res Public Health. 2010;7(10):3760–3791. doi: 10.3390/ijerph7103760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Durazzo TC, Fryer SL, Rothlind JC, et al. Measures of learning, memory and processing speed accurately predict smoking status in short-term abstinent treatment-seeking alcohol-dependent individuals. Alcohol Alcohol. 2010;45(6):507–513. doi: 10.1093/alcalc/agq057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ruiz SM, Oscar-Berman M, Sawyer KS, Valmas MM, Urban T, Harris GJ. Drinking history associations with regional white matter volumes in alcoholic men and women. Alcohol Clin Exp Res. 2013;37(1):110–122. doi: 10.1111/j.1530-0277.2012.01862.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Makris N, Oscar-Berman M, Jaffin SK, et al. Decreased volume of the brain reward system in alcoholism. Biol Psychiatry. 2008;64(3):192–202. doi: 10.1016/j.biopsych.2008.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pfefferbaum A, Sullivan EV, Rosenbloom MJ, Mathalon DH, Lim KO. A controlled study of cortical gray matter and ventricular changes in alcoholic men over a 5-year interval. Arch Gen Psychiatry. 1998;55(10):905–912. doi: 10.1001/archpsyc.55.10.905. [DOI] [PubMed] [Google Scholar]
- 19.Pfefferbaum A, Lim KO, Zipursky RB, et al. Brain gray and white matter volume loss accelerates with aging in chronic alcoholics: a quantitative MRI study. Alcohol Clin Exp Res. 1992;16(6):1078–1089. doi: 10.1111/j.1530-0277.1992.tb00702.x. [DOI] [PubMed] [Google Scholar]
- 20.Cardenas VA, Studholme C, Gazdzinski S, Durazzo TC, Meyerhoff DJ. Deformation-based morphometry of brain changes in alcohol dependence and abstinence. Neuroimage. 2007;34(3):879–887. doi: 10.1016/j.neuroimage.2006.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chanraud S, Martelli C, Delain F, et al. Brain morphometry and cognitive performance in detoxified alcohol-dependents with preserved psychosocial functioning. Neuropsychopharmacology. 2007;32(2):429–438. doi: 10.1038/sj.npp.1301219. [DOI] [PubMed] [Google Scholar]
- 22.Froeliger B, Kozink RV, Rose JE, Behm FM, Salley AN, McClernon FJ. Hippocampal and striatal gray matter volume are associated with a smoking cessation treatment outcome: results of an exploratory voxel-based morphometric analysis. Psychopharmacology (Berl) 2010;210(4):577–583. doi: 10.1007/s00213-010-1862-3. [DOI] [PubMed] [Google Scholar]
- 23.Gallinat J, Meisenzahl E, Jacobsen LK, et al. Smoking and structural brain deficits: a volumetric MR investigation. Eur J Neurosci. 2006;24(6):1744–1750. doi: 10.1111/j.1460-9568.2006.05050.x. [DOI] [PubMed] [Google Scholar]
- 24.Zhang X, Salmeron BJ, Ross TJ, Geng X, Yang Y, Stein EA. Factors underlying prefrontal and insula structural alterations in smokers. Neuroimage. 2011;54(1):42–48. doi: 10.1016/j.neuroimage.2010.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fein G, Shimotsu R, Chu R, Barakos J. Parietal gray matter volume loss is related to spatial processing deficits in long-term abstinent alcoholic men. Alcohol Clin Exp Res. 2009;33(10):1806–1814. doi: 10.1111/j.1530-0277.2009.01019.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yu R, Zhao L, Lu L. Regional grey and white matter changes in heavy male smokers. PLoS One. 2011;6(11):e27440. doi: 10.1371/journal.pone.0027440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Almeida OP, Garrido GJ, Lautenschlager NT, Hulse GK, Jamrozik K, Flicker L. Smoking is associated with reduced cortical regional gray matter density in brain regions associated with incipient Alzheimer disease. Am J Geriat Psychiat. 2008;16(1):92–98. doi: 10.1097/JGP.0b013e318157cad2. [DOI] [PubMed] [Google Scholar]
- 28.Pan P, Shi H, Zhong J, et al. Chronic smoking and brain gray matter changes: evidence from meta-analysis of voxel-based morphometry studies. Neurol Sci. 2013;34(6):813–817. doi: 10.1007/s10072-012-1256-x. [DOI] [PubMed] [Google Scholar]
- 29.Liao Y, Tang J, Liu T, Chen X, Hao W. Differences between smokers and non-smokers in regional gray matter volumes: a voxel-based morphometry study. Addict Biol. 2012;17(6):977–980. doi: 10.1111/j.1369-1600.2010.00250.x. [DOI] [PubMed] [Google Scholar]
- 30.Das D, Cherbuin N, Anstey KJ, Sachdev PS, Easteal S. Lifetime cigarette smoking is associated with striatal volume measures. Addict Biol. 2012;17(4):817–825. doi: 10.1111/j.1369-1600.2010.00301.x. [DOI] [PubMed] [Google Scholar]
- 31.Gazdzinski S, Durazzo TC, Studholme C, Song E, Banys P, Meyerhoff DJ. Quantitative brain MRI in alcohol dependence: preliminary evidence for effects of concurrent chronic cigarette smoking on regional brain volumes. Alcohol Clin Exp Res. 2005;29(8):1484–1495. doi: 10.1097/01.alc.0000175018.72488.61. [DOI] [PubMed] [Google Scholar]
- 32.Agartz I, Momenan R, Rawlings RR, Kerich MJ, Hommer DW. Hippocampal volume in patients with alcohol dependence. Arch Gen Psychiatry. 1999;56(4):356–363. doi: 10.1001/archpsyc.56.4.356. [DOI] [PubMed] [Google Scholar]
- 33.Pfefferbaum A, Lim KO, Desmond JE, Sullivan EV. Thinning of the corpus callosum in older alcoholic men: a magnetic resonance imaging study. Alcohol Clin Exp Res. 1996;20(4):752–757. doi: 10.1111/j.1530-0277.1996.tb01682.x. [DOI] [PubMed] [Google Scholar]
- 34.Pfefferbaum A, Adalsteinsson E, Sullivan EV. Dysmorphology and microstructural degradation of the corpus callosum: Interaction of age and alcoholism. Neurobiol Aging. 2006;27(7):994–1009. doi: 10.1016/j.neurobiolaging.2005.05.007. [DOI] [PubMed] [Google Scholar]
- 35.Choi MH, Lee SJ, Yang JW, et al. Difference between smokers and non-smokers in the corpus callosum volume. Neurosci Lett. 2010;485(1):71–73. doi: 10.1016/j.neulet.2010.08.066. [DOI] [PubMed] [Google Scholar]
- 36.Kühn S, Romanowski A, Schilling C, et al. Brain grey matter deficits in smokers: focus on the cerebellum. Brain Struct Funct. 2012;217(2):517–522. doi: 10.1007/s00429-011-0346-5. [DOI] [PubMed] [Google Scholar]
- 37.Ceballos NA. Tobacco use, alcohol dependence, and cognitive performance. J Gen Psychol. 2006;133(4):375–388. doi: 10.3200/GENP.133.4.375-388. [DOI] [PubMed] [Google Scholar]
- 38.Sullivan EV, Rosenbloom MJ, Pfefferbaum A. Pattern of motor and cognitive deficits in detoxified alcoholic men. Alcohol Clin Exp Res. 2000;24(5):611–621. [PubMed] [Google Scholar]
- 39.Davies SJ, Pandit SA, Feeney A, et al. Is there cognitive impairment in clinically ‘healthy’ abstinent alcohol dependence? Alcohol Alcohol. 2005;40(6):498–503. doi: 10.1093/alcalc/agh203. [DOI] [PubMed] [Google Scholar]
- 40.Hill RD, Nilsson LG, Nyberg L, Backman L. Cigarette smoking and cognitive performance in healthy Swedish adults. Age Ageing. 2003;32(5):548–550. doi: 10.1093/ageing/afg067. [DOI] [PubMed] [Google Scholar]
- 41.Starr JM, Deary IJ, Fox HC, Whalley LJ. Smoking and cognitive change from age 11 to 66 years: a confirmatory investigation. Addict Behav. 2007;32(1):63–68. doi: 10.1016/j.addbeh.2006.03.020. [DOI] [PubMed] [Google Scholar]
- 42.Beatty WW, Hames KA, Blanco CR, Nixon SJ, Tivis LJ. Visuospatial perception, construction and memory in alcoholism. J Stud Alcohol. 1996;57(2):136–143. doi: 10.15288/jsa.1996.57.136. [DOI] [PubMed] [Google Scholar]
- 43.Sullivan EV, Rosenbloom MJ, Lim KO, Pfefferbaum A. Longitudinal changes in cognition, gait, and balance in abstinent and relapsed alcoholic men: relationships to changes in brain structure. Neuropsychology. 2000;14(2):178–188. [PubMed] [Google Scholar]
- 44.Sullivan EV, Fama R, Rosenbloom MJ, Pfefferbaum A. A profile of neuropsychological deficits in alcoholic women. Neuropsychology. 2002;16(1):74–83. doi: 10.1037//0894-4105.16.1.74. [DOI] [PubMed] [Google Scholar]
- 45.Reed RJ, Grant I, Rourke SB. Long-term abstinent alcoholics have normal memory. Alcohol Clin Exp Res. 1992;16(4):677–683. doi: 10.1111/j.1530-0277.1992.tb00660.x. [DOI] [PubMed] [Google Scholar]
- 46.Liu JT, Lee IH, Wang CH, Chen KC, Lee CI, Yang YK. Cigarette smoking might impair memory and sleep quality. J Formos Med Assoc. 2013;112(5):287–290. doi: 10.1016/j.jfma.2011.12.006. [DOI] [PubMed] [Google Scholar]
- 47.Beatty WW, Tivis R, Stott HD, Nixon SJ, Parsons OA. Neuropsychological deficits in sober alcoholics: influences of chronicity and recent alcohol consumption. Alcohol Clin Exp Res. 2000;24(2):149–154. [PubMed] [Google Scholar]
- 48.Ratti MT, Bo P, Giardini A, Soragna D. Chronic alcoholism and the frontal lobe: which executive functions are imparied? Acta Neurol Scand. 2002;105(4):276–281. doi: 10.1034/j.1600-0404.2002.0o315.x. [DOI] [PubMed] [Google Scholar]
- 49.Oscar-Berman M, Kirkley SM, Gansler DA, Couture A. Comparisons of Korsakoff and non-Korsakoff alcoholics on neuropsychological tests of prefrontal brain functioning. Alcohol Clin Exp Res. 2004;28(4):667–675. doi: 10.1097/01.alc.0000122761.09179.b9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Rosenbloom MJ, Pfefferbaum A, Sullivan EV. Recovery of short-term memory and psychomotor speed but not postural stability with long-term sobriety in alcoholic women. Neuropsychology. 2004;18(3):589–597. doi: 10.1037/0894-4105.18.3.589. [DOI] [PubMed] [Google Scholar]
- 51.Oscar-Berman M, Valmas MM, Sawyer KS, et al. Frontal brain dysfunction in alcoholism with and without antisocial personality disorder. Neuropsychiatr Dis Treat. 2009;5:309–326. doi: 10.2147/ndt.s4882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Whalley LJ, Fox HC, Deary IJ, Starr JM. Childhood IQ, smoking, and cognitive change from age 11 to 64 years. Addict Behav. 2005;30(1):77–88. doi: 10.1016/j.addbeh.2004.04.014. [DOI] [PubMed] [Google Scholar]
- 53.Cerhan JR, Folsom AR, Mortimer JA, et al. Correlates of cognitive function in middle-aged adults. Atherosclerosis Risk in Communities (ARIC) Study Investigators. Gerontology. 1998;44(2):95–105. doi: 10.1159/000021991. [DOI] [PubMed] [Google Scholar]
- 54.Stewart MC, Deary IJ, Fowkes FG, Price JF. Relationship between lifetime smoking, smoking status at older age and human cognitive function. Neuroepidemiology. 2006;26(2):83–92. doi: 10.1159/000090253. [DOI] [PubMed] [Google Scholar]
- 55.Spilich GJ, June L, Renner J. Cigarette smoking and cognitive performance. Br J Addict. 1992;87(9):1313–1326. doi: 10.1111/j.1360-0443.1992.tb02740.x. [DOI] [PubMed] [Google Scholar]
- 56.Sullivan EV, Mathalon DH, Zipursky RB, Kersteen-Tucker Z, Knight RT, Pfefferbaum A. Factors of the Wisconsin Card Sorting Test as measures of frontal-lobe function in schizophrenia and in chronic alcoholism. Psychiatry Res. 1993;46(2):175–199. doi: 10.1016/0165-1781(93)90019-d. [DOI] [PubMed] [Google Scholar]
- 57.Fama R, Pfefferbaum A, Sullivan EV. Perceptual learning in detoxified alcoholic men: contributions from explicit memory, executive function, and age. Alcohol Clin Exp Res. 2004;28(11):1657–1665. doi: 10.1097/01.alc.0000145690.48510.da. [DOI] [PubMed] [Google Scholar]
- 58.Razani J, Boone K, Lesser I, Weiss D. Effects of cigarette smoking history on cognitive functioning in healthy older adults. Am J Geriatr Psychiatry. 2004;12(4):404–411. doi: 10.1176/appi.ajgp.12.4.404. [DOI] [PubMed] [Google Scholar]
- 59.Tate DL, Charette L. Personality, alcohol consumption, and menstrual distress in young women. Alcohol Clin Exp Res. 1991;15(4):647–652. doi: 10.1111/j.1530-0277.1991.tb00573.x. [DOI] [PubMed] [Google Scholar]
- 60.Arai Y, Hosokawa T, Fukao A, Izumi Y, Hisamichi S. Smoking behaviour and personality: a population-based study in Japan. Addiction. 1997;92(8):1023–1033. [PubMed] [Google Scholar]
- 61.Spielberger CD, Jacobs GA. Personality and smoking behavior. J Pers Assess. 1982;46(4):396–403. doi: 10.1207/s15327752jpa4604_11. [DOI] [PubMed] [Google Scholar]
- 62.Batel P, Pessione F, Maitre C, Rueff B. Relationship between alcohol and tobacco dependencies among alcoholics who smoke. Addiction. 1995;90(7):977–980. doi: 10.1046/j.1360-0443.1995.90797711.x. [DOI] [PubMed] [Google Scholar]
- 63.Dawson DA, Compton WM, Grant BF. Frequency of 5+/4+ drinks as a screener for drug use and drug-use disorders. J Stud Alcohol Drugs. 2010;71(5):751–760. doi: 10.15288/jsad.2010.71.751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Robins LN, Cottler LB. Making a structured psychiatric diagnostic interview faithful to the nomenclature. Am J Epidemiol. 2004;160(8):808–813. doi: 10.1093/aje/kwh283. [DOI] [PubMed] [Google Scholar]
- 65.American Psychiatric Association . Diagnostic and Statistical Manual of Mental Disorders. 4th ed. Washington, DC: American Psychiatric Association; 2000. [Google Scholar]
- 66.Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23:56–62. doi: 10.1136/jnnp.23.1.56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Cahalan D, Cisin I, Crossley H. American Drinking Practices: A National Study of Drinking Behavior and Attitudes. New Brunswick, NJ: Rutgers Center for Alcohol Studies; 1969. [Google Scholar]
- 68.Wechsler D. Wechsler Adult Intelligence Scale. 4th ed. London: Pearson; 2008. [Google Scholar]
- 69.Wechsler D. Wechsler Memory Scale. 4th ed. S London: Pearson; 2009. [Google Scholar]
- 70.Eysenck HJ, Eysenck MW. Personality and Individual Differences: A Natural Science Approach. New York, NY: Springer Dordrecht; 1985. [Google Scholar]
- 71.Zuckerman M, Lubin B, Robins S. Validation of the multiple affect adjective check list in clinical situations. J Consult Psychol. 1965;29(6):594. doi: 10.1037/h0022750. [DOI] [PubMed] [Google Scholar]
- 72.Pearson N. Advanced Clinical Solutions for WAIS-IV and WMS-IV: Administration and Scoring Manual. London: Pearson; 2009. [Google Scholar]
- 73.Nelson HE. A modified card sorting test sensitive to frontal lobe defects. Cortex. 1976;12(4):313–324. doi: 10.1016/s0010-9452(76)80035-4. [DOI] [PubMed] [Google Scholar]
- 74.Delis DC, Kaplan E, Kramer JH. Delis-Kaplan Executive Function System (D-KEFS) London: Pearson; 2001. [Google Scholar]
- 75.Armitage SG. An analysis of certain psychological tests used for the evaluation of brain injury. Psychol Monogr. 1946;60(1):i–48. [Google Scholar]
- 76.Benton A, Hamsher K, Sivan A. Multilingual Aphasia Examination: Manual of Instructions. Iowa City: AJA Associates; 1994. [Google Scholar]
- 77.Spreen O, Strauss E, editors. A Compendium of Neuropsychological Tests: Administration, Norms, and Commentary. 1st ed. New York: Oxford Univerity Press; 1991. [Google Scholar]
- 78.Desikan RS, Segonne F, Fischl B, et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage. 2006;31(3):968–980. doi: 10.1016/j.neuroimage.2006.01.021. [DOI] [PubMed] [Google Scholar]
- 79.Fischl B, van der Kouwe A, Destrieux C, et al. Automatically parcellating the human cerebral cortex. Cereb Cortex. 2004;14(1):11–22. doi: 10.1093/cercor/bhg087. [DOI] [PubMed] [Google Scholar]
- 80.Fischl B, Salat DH, Busa E, et al. Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron. 2002;33(3):341–355. doi: 10.1016/s0896-6273(02)00569-x. [DOI] [PubMed] [Google Scholar]
- 81.Fischl B, Salat DH, van der Kouwe AJ, et al. Sequence-independent segmentation of magnetic resonance images. Neuroimage. 2004;23(Suppl 1):S69–S84. doi: 10.1016/j.neuroimage.2004.07.016. [DOI] [PubMed] [Google Scholar]
- 82.Salat DH, Greve DN, Pacheco JL, et al. Regional white matter volume differences in nondemented aging and Alzheimer’s disease. Neuroimage. 2009;44(4):1247–1258. doi: 10.1016/j.neuroimage.2008.10.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Durazzo TC, Cardenas VA, Studholme C, Weiner MW, Meyerhoff DJ. Non-treatment-seeking heavy drinkers: effects of chronic cigarette smoking on brain structure. Drug Alcohol Depend. 2007;87(1):76–82. doi: 10.1016/j.drugalcdep.2006.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Howell DC. Statistical Methods for Psychology. 5th ed. Pacific Grove, CA: Duxbury Thomson Learning; 2002. [Google Scholar]
- 85.Cook RD, Weisberg S. Residuals and Influence Regression. New York, NY: Chapman and Hall; 1982. [Google Scholar]
- 86.Thayer RE, Crotwell S, Callahan T, Hutchison K, Bryan A. Nucleus accumbens volume is associated with frequency of alcohol use among juvenile justice-involved adolescents. Brain Sci. 2012;4(2):605–618. doi: 10.3390/brainsci2040605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Court JA, Martin-Ruiz C, Graham A, Perry E. Nicotinic receptors in human brain: topography and pathology. J Chem Neuroanat. 2000;20(3–4):281–298. doi: 10.1016/s0891-0618(00)00110-1. [DOI] [PubMed] [Google Scholar]
- 88.Skuse DH, Gallagher L. Dopaminergic-neuropeptide interactions in the social brain. Trends Cogn Sci (Regul Ed) 2009;13(1):27–35. doi: 10.1016/j.tics.2008.09.007. [DOI] [PubMed] [Google Scholar]
- 89.Harvey SC, Foster KL, McKay PF, et al. The GABA(A) receptor alpha1 subtype in the ventral pallidum regulates alcohol-seeking behaviors. J Neurosci. 2002;22(9):3765–3775. doi: 10.1523/JNEUROSCI.22-09-03765.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.June HL, Foster KL, McKay PF, et al. The reinforcing properties of alcohol are mediated by GABA(A1) receptors in the ventral pallidum. Neuropsychopharmacol. 2003;28(12):2124–2137. doi: 10.1038/sj.npp.1300239. [DOI] [PubMed] [Google Scholar]
- 91.Westlye LT, Walhovd KB, Dale AM, et al. Life-span changes of the human brain white matter: diffusion tensor imaging (DTI) and volumetry. Cereb Cortex. 2010;20(9):2055–2068. doi: 10.1093/cercor/bhp280. [DOI] [PubMed] [Google Scholar]
- 92.Durazzo TC, Pennington DL, Schmidt TP, Mon A, Abe C, Meyerhoff DJ. Neurocognition in 1-month-abstinent treatment-seeking alcohol-dependent individuals: interactive effects of age and chronic cigarette smoking. Alcohol Clin Exp Res. 2013 doi: 10.1111/acer.12140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Madden PA, Bucholz KK, Martin NG, Heath AC. Smoking and the genetic contribution to alcohol-dependence risk. Alcohol Res Health. 2000;24(4):209–214. [PMC free article] [PubMed] [Google Scholar]
- 94.Dick DM, Bierut LJ. The genetics of alcohol dependence. Curr Psychiatry Rep. 2006;8(2):151–157. doi: 10.1007/s11920-006-0015-1. [DOI] [PubMed] [Google Scholar]
- 95.Funk D, Marinelli PW, Le AD. Biological processes underlying co-use of alcohol and nicotine: neuronal mechanisms, cross-tolerance, and genetic factors. Alcohol Res Health. 2006;29(3):186–192. [PMC free article] [PubMed] [Google Scholar]
- 96.Grucza RA, Bierut LJ. Co-occurring risk factors for alcohol dependence and habitual smoking: update on findings from the Collaborative Study on the Genetics of Alcoholism. Alcohol Res Health. 2006;29(3):172–178. [PMC free article] [PubMed] [Google Scholar]
- 97.Hendrickson LM, Guildford MJ, Tapper AR. Neuronal nicotinic acetylcholine receptors: common molecular substrates of nicotine and alcohol dependence. Front Psychiatry. 2013;4:29. doi: 10.3389/fpsyt.2013.00029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Gulliver SB, Kamholz BW, Helstrom AW. Smoking cessation and alcohol abstinence: what do the data tell us? Alcohol Res Health. 2006;29(3):208–212. [PMC free article] [PubMed] [Google Scholar]
- 99.Kodl M, Fu SS, Joseph AM. Tobacco cessation treatment for alcohol-dependent smokers: when is the best time? Alcohol Res Health. 2006;29(3):203–207. [PMC free article] [PubMed] [Google Scholar]
- 100.Ziedonis DM, Guydish J, Williams J, Steinberg M, Foulds J. Barriers and solutions to addressing tobacco dependence in addiction treatment programs. Alcohol Res Health. 2006;29(3):228–235. [PMC free article] [PubMed] [Google Scholar]
- 101.Pennington DL, Durazzo TC, Schmidt TP, Mon A, Abe C, Meyerhoff DJ. The effects of chronic cigarette smoking on cognitive recovery during early abstinence from alcohol. Alcohol Clin Exp Res. 2013;37(7):1220–1227. doi: 10.1111/acer.12089. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1.
Statistical comparisons for alcoholism and smoking
| Measure | Contrast | df | t | P-value | d |
|---|---|---|---|---|---|
| Duration of heavy drinking (years) | sNA < sAL | 6 | 5.55 | 0.001 | 3.35 |
| Duration of heavy drinking (years) | nsNA < nsAL | 6 | 5.33 | 0.002 | 3.08 |
| Duration of heavy drinking (years) | sNA < nsAL | 6 | 5.33 | 0.002 | 3.21 |
| Duration of heavy drinking (years) | nsNA < sAL | 6 | 5.55 | 0.001 | 3.20 |
| Average drinks per day | sNA < sAL | 6.08 | 5.00 | 0.002 | 3.02 |
| Average drinks per day | nsNA < nsAL | 6.02 | 4.35 | 0.005 | 2.51 |
| Average drinks per day | sNA < nsAL | 6.03 | 4.33 | 0.005 | 2.61 |
| Average drinks per day | nsNA < sAL | 6.08 | 5.00 | 0.002 | 2.89 |
| Cigarettes per day | nsAL < sAL | 6 | 7.97 | 0.0002 | 4.60 |
| Cigarettes per day | nsNA < sNA | 5 | 2.67 | 0.04 | 1.61 |
| Cigarettes per day | sNA < sAL | 10.06 | 2.91 | 0.02 | 1.75 |
| Cigarettes per day | nsAL < sNA | 5 | 2.67 | 0.04 | 1.61 |
| Cigarettes per day | nsNA < sAL | 6 | 7.99 | 0.0002 | 4.61 |
Notes: For each contrast, degrees of freedom (df), t, P, and Cohen’s d values are presented. Cohen’s d was calculated with the following formula: 2t/√ (N − 2). In purple: comorbid effects (sAL versus nsNA). In red: smoking effects (sAL versus nsAL or sNA versus nsNA). In blue: alcholism effects (sAL versus sNA or nsAL versus nsNA). In green: cross effects of smoking and alcoholism (sNA versus nsAL).
Abbreviations: nsAL, alcoholic participants – currently not smoking; nsNA, nonalcoholic participants – currently not smoking; sAL, alcoholic participants (the comorbid group); sNA, nonalcoholic participants – current smokers.
Table S2.
Volumetric variables presented within models containing age and total brain volume as covariates
| Regions | Contrast | df1 | df2 | t | P-value | d |
|---|---|---|---|---|---|---|
| Fourth ventricle | sAL < sNA | 1 | 9 | 2.93 | 0.02 | 1.77 |
| R inferior lateral ventricle | nsAL < sAL | 1 | 10 | 2.51 | 0.03 | 1.45 |
| R inferior lateral ventricle | nsNA < sAL | 1 | 10 | 3.59 | 0.005 | 2.07 |
| L accumbens area | nsNA < nsAL | 1 | 10 | 2.33 | 0.05 | 1.35 |
| L pallidum* | sNA < nsNA | 1 | 9 | 3.47 | 0.007 | 2.09 |
| L pallidum* | nsAL < nsNA | 1 | 10 | 4.76 | 0.0008 | 2.75 |
| L pallidum* | sAL < nsNA | 1 | 10 | 2.95 | 0.01 | 1.70 |
| L ventral diencephalon | sAL < nsNA | 1 | 10 | 2.98 | 0.01 | 1.72 |
| R amygdala | sNA < nsNA | 1 | 9 | 2.42 | 0.04 | 1.46 |
| R amygdala | sAL < nsNA | 1 | 10 | 3.91 | 0.003 | 2.26 |
| R pallidum | nsAL < nsNA | 1 | 10 | 2.67 | 0.02 | 1.54 |
| R thalamus proper | sNA < nsNA | 1 | 9 | 2.45 | 0.04 | 1.48 |
| R ventral diencephalon | sAL < nsNA | 1 | 10 | 3.16 | 0.01 | 1.82 |
| Total subcortical gray matter | sAL < nsNA | 1 | 10 | 2.29 | 0.05 | 1.32 |
| L caudal middle frontal | nsAL < sNA | 1 | 9 | 2.70 | 0.02 | 1.63 |
| L fusiform | sNA < nsAL | 1 | 9 | 3.13 | 0.01 | 1.89 |
| L paracentral | nsAL < nsNA | 1 | 10 | 4.00 | 0.003 | 2.31 |
| L paracentral | sAL < nsNA | 1 | 10 | 3.81 | 0.003 | 2.20 |
| L parahippocampal | sAL < sNA | 1 | 9 | 3.50 | 0.007 | 2.11 |
| L parahippocampal | sAL < nsNA | 1 | 10 | 3.39 | 0.007 | 1.96 |
| L supramarginal | sNA < nsNA | 1 | 9 | 2.72 | 0.02 | 1.64 |
| L supramarginal | sNA < nsAL | 1 | 9 | 2.94 | 0.02 | 1.77 |
| L temporal pole | sAL < nsNA | 1 | 10 | 2.97 | 0.01 | 1.71 |
| R caudal anterior cingulate | nsNA < sNA | 1 | 9 | 2.31 | 0.05 | 1.39 |
| R caudal middle frontal | nsAL < sNA | 1 | 9 | 2.89 | 0.02 | 1.74 |
| R entorhinal | sNA < nsNA | 1 | 9 | 2.70 | 0.02 | 1.63 |
| R entorhinal | sNA < nsAL | 1 | 9 | 2.28 | 0.05 | 1.37 |
| R pars orbitalis* | sNA < nsNA | 1 | 9 | 3.18 | 0.01 | 1.92 |
| R precentral | sAL < sNA | 1 | 9 | 2.46 | 0.04 | 1.48 |
| R precentral | sAL < nsNA | 1 | 10 | 2.63 | 0.02 | 1.52 |
| R rostral middle frontal | nsNA < nsAL | 1 | 10 | 2.91 | 0.02 | 1.68 |
| R superior parietal | nsAL < nsNA | 1 | 10 | 3.09 | 0.01 | 1.78 |
| R temporal pole | nsAL < sAL | 1 | 10 | 2.93 | 0.01 | 1.69 |
| R temporal pole | nsAL < nsNA | 1 | 10 | 2.36 | 0.04 | 1.36 |
| L total cortex | sNA < nsNA | 1 | 9 | 2.31 | 0.05 | 1.39 |
| L middle temporal WM* | nsNA < sNA | 1 | 9 | 2.33 | 0.04 | 1.41 |
| R banks of the superior temporal sulcus WM* | sAL < nsAL | 1 | 10 | 2.77 | 0.02 | 1.60 |
| R banks of the superior temporal sulcus WM* | sAL < sNA | 1 | 9 | 2.46 | 0.04 | 1.48 |
| R caudal anterior cingulate WM | nsNA < sNA | 1 | 9 | 2.41 | 0.04 | 1.45 |
| R caudal middle frontal WM | nsAL < nsNA | 1 | 10 | 2.33 | 0.04 | 1.35 |
| R caudal middle frontal WM | nsAL < sNA | 1 | 9 | 2.89 | 0.02 | 1.74 |
| R fusiform WM | nsAL < sNA | 1 | 9 | 2.38 | 0.04 | 1.44 |
| R inferior temporal WM | nsNA < nsAL | 1 | 10 | 2.42 | 0.04 | 1.40 |
| R rostral middle frontal WM | nsNA < nsAL | 1 | 10 | 4.36 | 0.001 | 2.52 |
| R rostral middle frontal WM | nsNA < sAL | 1 | 10 | 2.27 | 0.05 | 1.31 |
Notes: For each contrast, degrees of freedom (df), t, P, and Cohen’s d are presented. Cohen’s d was calculated with the following formula: 2t/√(N – 2)
Significant interactions between smoking and alcoholism, where P<0.05. In purple: comorbid effects (sAL versus nsNA). In red: smoking effects (sAL versus nsAL or sNA versus nsNA). In blue: alcholism effects (sAL versus sNA or nsAL versus nsNA). In green: cross effects of smoking and alcoholism (sNA versus nsAL).
Abbreviations: L, left hemisphere; R, right hemisphere; nsAL, alcoholic participants – currently not smoking; nsNA, nonalcoholic participants – currently not smoking; sAL, alcoholic participants (the comorbid group); sNA, nonalcoholic participants – current smokers; WM, white matter.
Table S3.
Neuropsychological variables
| Neuropsychological test | Contrast | df | t | P-value | d |
|---|---|---|---|---|---|
| ACS: faces I* | sAL < nsAL | 9.65 | 2.23 | 0.05 | 1.29 |
| ACS: faces I* | sAL < sNA | 9.77 | 3.08 | 0.01 | 1.86 |
| ACS: faces II* | sAL < nsAL | 11.4 | 2.74 | 0.02 | 1.58 |
| ACS: faces II* | sAL < sNA | 10.99 | 2.43 | 0.03 | 1.47 |
| ACS: faces II* | sAL < nsNA | 11.79 | 2.18 | 0.05 | 1.26 |
| ACS: faces spatial | sAL < sNA | 10.99 | 2.43 | 0.03 | 1.47 |
| ACS: faces spatial | sAL < nsNA | 11.56 | 2.27 | 0.04 | 1.31 |
| EPQ: extraversion | sNA < nsNA | 10.95 | 2.87 | 0.02 | 1.73 |
| EPQ: neuroticism | nsNA < nsAL | 11.53 | 2.76 | 0.02 | 1.59 |
| MAACL: sensation seeking | nsNA < sNA | 10.44 | 2.60 | 0.03 | 1.57 |
| WAIS-IV: cancellation | nsAL < sNA | 8.7 | 2.75 | 0.02 | 1.66 |
| WCST: categories completed | sAL < nsNA | NA | 2.53 | 0.01 | 0.68 |
| WCST: percent conceptual level responses percentile | sAL < nsNA | 11 | 4.13 | 0.002 | 2.38 |
| WMS-IV: designs I | sAL < nsAL | 11.94 | 2.56 | 0.03 | 1.48 |
| WMS-IV: designs I | sAL < nsNA | 10.26 | 3.55 | 0.005 | 2.05 |
| WMS-IV: designs I content | sAL < nsNA | 10.05 | 2.34 | 0.04 | 1.35 |
| WMS-IV: designs I spatial* | sAL < nsAL | 10.02 | 4.23 | 0.002 | 2.44 |
| WMS-IV: designs I spatial* | sAL < nsNA | 11.51 | 2.86 | 0.01 | 1.65 |
| WMS-IV: designs II spatial* | sAL < nsAL | 12 | 4.17 | 0.001 |
Notes: For each contrast, degrees of freedom (df), t, P, and Cohen’s d are presented. Cohen’s d was calculated with the following formula: 2t/√ (N – 2). For WCST: categories completed, Wilcoxon rank-sum Z is reported in place of t, and r = Z/√N in place of d.
*Significant interactions between smoking and alcoholism, where P<0.05. In purple: comorbid effects (sAL versus nsNA). In red: smoking effects (sAL versus nsAL or sNA versus nsNA). In blue: alcholism effects (sAL versus sNA or nsAL versus nsNA). In green: cross effects of smoking and alcoholism (sNA versus nsAL).
Abbreviations: ACS, advanced clinical solutions; EPQ, Eysenck Personality Questionnaire; MAACL, Multiple Affective Adjective Check List; nsAL, alcoholic participants – currently not smoking; nsNA, nonalcoholic participants – currently not smoking; sAL, alcoholic participants (the comorbid group); sNA, nonalcoholic participants – current smokers; WAIS, Wechsler Adult Intelligence Scale; WCST, Wsconsin Card Sorting Test; WMS, Wechsler Memory Scale.