Abstract
Background
In the aging population cardiovascular disease (CVD) is highly prevalent. Identification of very old persons at high risk of recurrent CVD is difficult, since traditional risk markers loose predictive value with age.
Methods
In a population-based sample of 282 85-year old participants with established CVD from the Leiden 85-plus Study, we studied predictive values of traditional cardiovascular risk markers, a history of major CVD (myocardial infarction, stroke or arterial surgery), and new cardiovascular biomarkers (estimated glomerular filtration rate (MDRD), C-reactive protein (CRP), homocysteine and N-terminal pro B-type natriuretic peptide (NT-proBNP)) regarding 5-year risk of recurrent cardiovascular events and mortality (composite endpoint).
Results
During complete 5-year follow-up 157 (56%) participants died. 109 (39%) had a cardiovascular event or died from cardiovascular causes. Individually related to the composite endpoint were: a history of major CVD (HR 1.5 (95%CI 1.03-2.3)), CRP (HR 1.3 (95%CI 1.03-1.5)), homocysteine (HR 1.4 (95%CI 1.2-2.6)) and NT-proBNP (HR 1.7 (95%CI 1.4-2.1)). A prediction model including all traditional risk markers yielded a C-statistic of 0.59 (95%CI 0.52-0.66). Of all five new markers only addition of NT-proBNP improved the C-statistic (0.67 (95%CI 0.61-0.74, p=0.023)). The categoryless net reclassification improvement for NT-proBNP was 39% (p=0.001), for a history of major CVD 27.2% (p=0.03) and for homocysteine 24.7% (p=0.04).
Conclusions
Among very old subjects with established CVD, NT-proBNP was the strongest risk marker for cardiovascular events and cardiovascular mortality. When estimating risk in secondary prevention in very old age, use of NT-proBNP should be considered.
Introduction
In the aging population, cardiovascular disease (CVD) is highly prevalent and remains a leading cause of death [1,2]. Persons with previous CVD are known to be at high risk of recurrent CVD [3–7]. However, even though secondary preventive treatment is effective in very old age [8–11], treatment at this age is often far from optimal [12–15] and drug adherence is poor [16]. Identifying patients at highest risk of recurrent events can help clinicians to select those very old patients that might benefit most from intensified preventive lifestyle measures and drug treatment [17].
In secondary prevention, traditional risk markers seem to have less predictive value [18]. However, data on their actual value in secondary prevention in very old age are scarce. In search of improvement of risk stratification, some studies found additional predictive value by including information on various degrees of previous CVD [4,19,20]. Many studies have evaluated the additional predictive value of markers of renal dysfunction (MDRD, albuminuria or cystatin C), inflammation (C-reactive protein (CRP)), oxidative stress (homocysteine) or myocardial wall stress (N-terminal pro B-type natriuretic peptide (NT-proBNP)), but nearly all in a primary preventive setting [21–25] and especially aimed at improving prediction in those with intermediate risk. Although some studies have shown incremental predictive value of new biomarkers in populations with established CVD [20,23,26–31], their predictive value in a secondary preventive setting in very old age remains unknown.
We hypothesized that the predictive value of traditional risk markers in secondary prevention in very old age is limited and that addition of information on the history of CVD or new biomarkers (MDRD, CRP, homocysteine and NT-proBNP) might have incremental value for predicting cardiovascular events and cardiovascular mortality.
Methods
Study design and participants
The Leiden 85-plus Study is a prospective population-based study in 85-year-old inhabitants of the city of Leiden in the Netherlands [32]. In brief, between September 1997 and September 1999, 705 people from the 1912-14 birth cohort living in the city of Leiden reached the age of 85 years and were eligible to participate. No exclusion criteria were used. From the 705 people who were eligible at age 85, 92 refused participation and 14 died before enrolment. A total of 599 (87%) people gave informed consent and were enrolled.
At baseline and yearly up to age 90 years participants were visited at their place of residence to obtain extensive data on health and functioning; blood samples and an ECG were taken. Medical history and CVD status were obtained from the medical records of the participant’s physician. Pharmacists provided information on all medication used by the participants.
The Medical Ethics Committee of the Leiden University Medical Center approved the study.
Written informed consent was obtained from all participants. The protocol adhered to the principles of the Declaration of Helsinki.
Baseline Assessment of Risk Markers
Traditional risk markers
Blood pressure was measured on two occasions with a mean interval of two weeks. Systolic blood pressure was recorded at the onset of Korotkoff phase I. The mean of the measured systolic values was used for analyses.
Serum concentrations of total cholesterol and high-density lipoprotein were analyzed on fully automated computerised analyzers (Hitachi 747 and 911; Hitachi, Tokyo, Japan).
Diabetes mellitus was considered present when listed in the medical records of the participant’s physician, when non-fasting glucose concentrations were ≥11.0 mmol/l, or when a participant was taking antidiabetic medication according to the pharmacist’s records.
All participants were interviewed about present smoking habits and were considered as smokers if they were a current smoker of cigarettes, cigars or a pipe.
Nature of the history of cardiovascular disease
For each participant, the primary care physician was interviewed about the CVD history using a standardized questionnaire including questions on present and past cardiovascular pathologies, including myocardial infarction (MI), stroke, surgery for arterial disease, angina, transient ischemic attack (TIA), intermittent claudication and heart failure. The diagnosis of heart failure was based on information as obtained from the participant’s general practitioner or nursing home physician regarding previous events and prevalent disease at entry of the study at age 85 years. An ECG was recorded at baseline and transmitted to the ECG Core laboratory in the Glasgow Royal Infirmary (Scotland, UK) for automated Minnesota Coding [33]. Presence of a MI on the ECG was defined as the presence of Minnesota Code 1-1 or 1-2 (excluding 1-2-8).
Major CVD was considered present if there was a history of MI, stroke, or arterial surgery or if there was a MI on the ECG at baseline [19]. Minor CVD was considered present if there was a history of angina, TIA, intermittent claudication or heart failure.
New risk markers
MDRD was calculated as follows: MDRD (ml/min/1,73 m2) = 186 * (serum creatinine (umol/l) / 88, 4)-1154, * age (in years) –0,203 * 0,742(for females).
Plasma concentrations of CRP were measured using a fully automated Hitachi 747 analyzer (Hitachi, Tokyo, Japan; detection limit 1 mg/l; coefficient of variation <5%).
Concentrations of homocysteine were measured in plasma samples with a fluorescence polarisation immunoassay after reduction to the free form with an IMx analyzer (Abbott, Abbott Park, IL, USA; coefficient of variation 2.2-2.5%).
NT-proBNP was determined with a chemiluminescent enzyme immunoassay (CLEIA) procedure (Roche, Switzerland) and was carried out on a PATHFAST (Mitsubishi Chemical Medience Corporation, Tokyo.) Detection range was 15 - 30 000 pg/mL and the coefficient of variation was < 5%.
Composite endpoint
The composite endpoint ‘cardiovascular morbidity and cardiovascular mortality’ was defined as incident fatal and non-fatal myocardial infarction, incident fatal and non-fatal stroke or any other cardiovascular mortality, whichever came first.
Up to age 90 years all incident fatal and non-fatal MIs were annually registered using data from the primary care physician, ECGs and death registration forms. Incident MI on the ECG was defined as the appearance of Minnesota Code 1-1 or 1-2, or Minnesota Code 1-3 in combination with the first appearance of Minnesota Code 5-x in the same myocardial area [33]. A fatal incident MI was categorised by cause of death codes I21-I23 (ICD 10). Information on incident stroke was collected annually from the primary care physician up to age 90 years. A fatal incident stroke was categorised by cause of death codes I61-I69 (ICD10). All participants were followed for mortality until age 90 years. Date and cause of death were obtained from civic and national registries. Causes of death were divided into cardiovascular causes (ICD-10 codes I00-I99) and non-cardiovascular causes (all other ICD-10 codes). Assignment of cause of death was done blinded for baseline and follow-up study data.
Statistical analysis
Variables that were unevenly distributed were log transformed.
For all traditional risk markers, a history of major CVD, as well as, the four new biomarkers, hazard ratios (HRs) and corresponding 95% confidence intervals (CIs) for cardiovascular morbidity and mortality were calculated univariate and multivariable, using Cox proportional hazards models, all adjusted for sex. Continuous variables were entered into the model per SD increase.
We constructed prediction models with the traditional risk markers (reference model), and with combinations of the traditional and new risk markers. All biomarkers were entered in the models as continuous variables. For each participant the linear predictor score (X-β) was calculated, using Cox proportional hazard models, all adjusted for sex. Using the continuous predicted risks from each model, C-statistics and receiver operating characteristic (ROC) curves with p-values (level of significance 5%) and 95% CIs were calculated.
We compared the tertiles of predicted risk of the traditional risk marker model and of new models with the observed 5-year incidence of the endpoint, using Kaplan-Meier plots adjusted for competing risks [34] and the log rank test.
Since clinically meaningful risk categories in secondary prevention are not defined, we also calculated the categoryless Net Reclassification Index (NRI), comparing new models to the reference model [35,36].
We estimated the integrated discrimination improvement (IDI) [37], a quantification of the difference in sensitivities and ‘one minus specificities’ between new models and the reference model over all possible cut-offs. In addition, we calculated the relative integrated discrimination improvement (rIDI) by dividing the integrated discrimination improvement by the discrimination of the reference model [38].
As a sensitivity analysis regarding the prognostic value of NT-proBNP we repeated all analyses with exclusion of participants with a history of heart failure.
To investigate the validity of our results, we repeated the calculations of C-statistics using cross validated X-beta values obtained by the ‘jack-knife’ method [39]. This was done for the prediction models with 1/ all traditional risk markers, 2/ all traditional risk markers plus NT-proBNP, and 3/ all traditional risk markers plus all five new markers.
Data analysis was performed using SPSS 20 for Windows (SPSS Inc., Chicago, IL, USA). Differences in C-statistics were analysed using Stata/IC 10.0.
Results
Baseline characteristics
Of the 599 participants, all aged 85 years at baseline, 300 (50%) had established CVD. Of these 300 participants, for 282 (94%; 109 males, 173 females) all clinical and laboratory data were available and they were included in our analyses. Of these 282 participants 55% had a history of major CVD (32% with MI or MI on the baseline ECG, 19% with stroke and 13% with arterial surgery) and 45% had a history of minor CVD (Table 1). Median NT-proBNP levels were 649 pg/ml (IQR 231-1477) in the subgroup with major CVD, and 405 pg/ml (174-1196) in the subgroup with minor CVD (p=0.035).
Table 1. Baseline cardiovascular characteristics of participants with a history of cardiovascular disease at age 85 years (N=282).
|
|
N (%), mean (SD) or median (IQR) a | |
|---|---|---|
| Traditional risk markers |
||
| Men | 109 (39%) | |
| Systolic blood pressure, mm Hg | 155 (19) | |
| Total chol, mmol/L | 5.7 (1.2) | |
| HDL chol, mmol/L | 1.3 (0.40) | |
| Diabetes | 49 (17%) | |
| Current smoking | 47 (17%) | |
| Nature of cardiovascular disease history c | ||
| Myocardial infarction | 90 (32%) | |
| Stroke | 52 (19%) | |
| Arterial surgery | 37 (13%) | |
| Angina | 105 (38%) | |
| Transient ischemic attack | 72 (26%) | |
| Intermittent claudication | 34 (12%) | |
| Heart failure | 68 (24%) | |
| Major CVD | 155 (55%) | |
| Minor CVD | 127 (45%) | |
| Cardiovascular medication | ||
| Aspirin or oral anticoagulant | 127 (45%) | |
| Antihypertensive medication d | 202 (72%) | |
| Lipid lowering drug | 5 (2%) | |
| New risk markers | ||
| MDRD, ml/min | 57 (15) | |
| CRP, mg/L | 5 (2-9) | |
| HCY, umol/L | 14 (11-17) | |
| NT-proBNP, pg/ml | 495 (198-1314) | |
data presented as N (%) for categorical variables, mean (SD) for normally distributed or median (IQR) for skewed continuous variables,
assessed only in participants with MMSE >18,
according to treating primary care physician
β-blockers, ACE inhibitors, diuretics and/or Calcium channel blockers
Incidence of composite endpoint
Of the 282 participants, 157 (56%) died during the 5-year follow-up, of whom 67 (43%) died from cardiovascular causes. In total 109 (39%) participants experienced the endpoint: 43 (39%) participants experienced a fatal or non-fatal MI, 46 (42%) a fatal or non-fatal stroke, and 20 (18%) died of other cardiovascular causes.
Univariate and multivariable analyses
Table 2 presents the univariate and multivariable hazard ratios (HRs) for the endpoint for the traditional risk markers, for a history of major CVD, and for the four new biomarkers. In univariate analyses (all adjusted for sex) of the traditional risk markers, current smoking (HR 1.9 (95% CI 1.2-3.0)) was associated with a higher risk. A history of major CVD yielded a HR of 1.5 (95% CI 1.03-2.3). Of the new biomarkers CRP (HR 1.3 (95% CI 1.03-1.5)), homocysteine (HR 1.4 (95% CI 1.1-1.6)) and NT-proBNP (HR 1.7 (95% CI 1.4-2.1)) were associated with a higher risk, whereas MDRD was not (HR 0.83 (95% CI 0.68-1.01)).
Table 2. Univariate (adjusted for sex) and multivariable hazard ratios (HR) for five-year cardiovascular morbidity and cardiovascular mortalitya, depending on traditional and new risk markers, in participants with a history of cardiovascular disease (N=282).
|
|
HR (95% confidence interval) for cardiovascular morbidity and mortalitya
|
||||||||
|---|---|---|---|---|---|---|---|---|---|
|
|
Univariate | Multivariable |
|||||||
|
|
All traditional | All trad. | All trad. | All trad. | All trad. | All trad. | All trad. | ||
|
|
+ major CVD | + MDRD (SD) | + CRP (SD) | + HCYb (SD) | + NT-proBNP (SD) | +all new | |||
| Traditional risk markers | |||||||||
| Men | 1.4 (0.96-2.0) | 1.3 (0.85-2.0) | 1.2 (0.79-1.9) | 1.4 (0.93-2.2) | 1.3 (0.86-2.0) | 1.3 (0.85-2.0) | 1.3 (0.83-1.9) | 1.2 (0.74-1.8) | |
| RR (∆10 mmHg) | 0.95 (0.86-1.1) | 0.93 (0.84-1.0) | 0.93 (0.84-1.0) | 0.93 (0.84-1.0) | 0.92 (0.83-1.0) | 0.95 (0.85-1.1) | 0.96 (0.86-1.1) | 0.96 (0.87-1.1) | |
| Total chol (SD) | 1.1 (0.91-1.4) | 1.1 (0.92-1.4) | 1.1 (0.91-1.4) | 1.1 (0.91-1.4) | 1.2 (0.94-1.4) | 1.1 (0.91-1.4) | 1.2 (0.99-1.5) | 1.2 (0.98-1.5) | |
| HDL chol (SD) | 1.0 (0.83-1.3) | 1.0 (0.82-1.2) | 1.0 (0.3-1.3) | 1.1 (0.87-1.3) | 1.1 (0.87-1.3) | 1.1 (0.88-1.3) | 1.1 (0.86-1.3) | 1.1 (0.92-1.4) | |
| DM | 1.1 (0.66-1.8) | 1.1 (0.70-1.9) | 1.1 (0.68-1.9) | 1.1 (0.67-1.8) | 1.1 (0.67-1.8) | 1.2 (0.70-1.9) | 1.0 (0.62-1.7) | 1.0 (0.62-1.7) | |
| Current smoking | 1.9 (1.2-3.0) | 1.9 (1.2-3.1) | 1.9 (1.2-3.1) | 2.0 (1.2-3.2) | 2.0 (1.2-3.1) | 1.8 (1.1-3.0) | 1.8 (1.1-3.0) | 1.8 (1.1-2.9) | |
| Major CVD c | 1.5 (1.03-2.3) | 1.5 (1.001-2.2) | 1.5 (1.01-2.3) | ||||||
| New risk markers | |||||||||
| MDRD (SD) | 0.83 (0.68-1.01) | 0.81 (0.65-3.7) | 1.1 (0.85-1.3) | ||||||
| CRP (SD) | 1.3 (1.03-1.5) | 1.3 (1.1-1.6) | 1.2 (0.97-1.4) | ||||||
| HCYb (SD) | 1.4 (1.1-1.6) | 1.3 (1.1-1.6) | 1.2 (0.99-1.6) | ||||||
| NT-proBNP (SD) | 1.7 (1.4-2.1) | 1.8 (1.4-2.2) | 1.6 (1.3-2.1) | ||||||
CRP, homocysteine and NT-proBNP are log transformed;a including incident stroke, myocardial infarction or cardiovascular mortality;b HCY: homocysteine;
including a history of myocardial infarction, stroke or arterial surgery
Added individually to a multivariable model with all the traditional risk markers, the estimates showed no major change. In a multivariable analysis with all old and new markers, current smoking (HR 1.8 (95% CI 1.1-2.9)), a history of major CVD (HR 1.5 (95% CI 1.01-2.3)) and NT-proBNP (HR 1.6 (95% CI 1.3-2.1)), were still independently associated with an increased risk of cardiovascular morbidity/mortality.
C-statistics
The combination of traditional risk markers had a C-statistic of 0.59 (95% CI 0.52-0.66) (Table 3). Addition of NT-proBNP alone to all traditional risk markers increased the C-statistic to 0.67 (95% CI 0.61-0.74) with a p-value for ∆ C-statistic of 0.023 (Figure 1).
Table 3. C- statistic, categoryless net reclassification improvement (NRI), integrated discrimination improvement (IDI) and relative IDI (rIDI) of the different models for prediction of five-year cardiovascular morbidity and mortality a in participants with a history of cardiovascular disease (N=282), all compared to a model with traditional risk markers b .
| C-statistic | 95% CI | ∆ C-statistic c | p-value∆c | categoryless NRI c (%) | p-valueNRI c | IDI c | p-valueIDI c | rIDI d | |
|---|---|---|---|---|---|---|---|---|---|
| Traditional risk markers | 0.59 | 0.52-0.66 | |||||||
| + major CVD | 0.60 | 0.53-0.67 | 0.01 | 0.59 | 27.2 | 0.03 | 0.012 | 0.30 | 0.46 |
| + MDRD | 0.59 | 0.52 -0.66 | 0.00 | 0.74 | 6.2 | 0.61 | 0.0025 | 0.81 | 0.095 |
| + CRP | 0.60 | 0.54 -0.67 | 0.01 | 0.65 | 16.8 | 0.17 | 0.008 | 0.47 | 0.31 |
| + Homocysteine | 0.62 | 0.55 -0.69 | 0.03 | 0.27 | 24.7 | 0.04 | 0.019 | 0.13 | 0.72 |
| + NT-proBNP | 0.67 | 0.61 -0.74 | 0.08 | 0.023 | 39.0 | 0.001 | 0.067 | <0.001 | 2.55 |
| + all five | 0.69 | 0.62- 0.75 | 0.10 | 0.0067 | 50.8 | <0.001 | 0.089 | <0.001 | 3.39 |
CRP, homocysteine and NT-proBNP are log transformed;
including incident stroke, myocardial infarction or cardiovascular mortality;
including sex, systolic blood pressure, total cholesterol, HDL cholesterol, diabetes, current smoking;
for comparison against traditional risk markers;
for comparison with the discrimination of the model with the traditional risk markers (0.026 in this study)
Figure 1. ROC curves for cardiovascular morbidity and mortality.
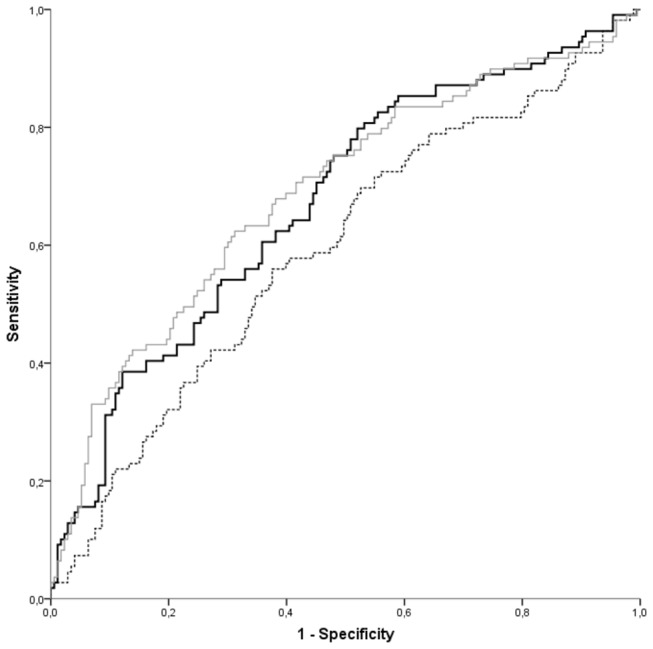
ROC curves for cardiovascular morbidity and mortality of three models: traditional risk markers (dotted line), traditional risk markers plus NT-proBNP (black line, p=0.023), and traditional risk markers plus all five new markers (a history of major CVD, MDRD, CRP, homocysteine and NT-proBNP) (grey line, p=0.0067) (N=282).
Categoryless net reclassification improvement (NRI)
For all different models we calculated the categoryless NRI, the net percentage of participants that is correctly reclassified when the new risk marker is added to the reference model with traditional risk markers (Table 3). Addition of a history of major CVD reclassified 27.2 % (p=0.03) of the participants correctly. For CRP this was 16.8 % (p=0.17), for homocysteine 24.7% (p=0.04) whereas for NT-proBNP it was 39.0 % (p=0.001).
Integrated discrimination improvement (IDI)
The discrimination of the baseline model, based on the predicted probabilities in those with and without events, was 0.026. The IDI after addition of a history of major CVD, or MDRD, CRP or homocysteine was not significant (Table 3). However, addition of NT-proBNP improved the discrimination to 0.067 (p<0.001). Thus for NT-proBNP the relative discrimination improvement, compared to the discrimination of the baseline model with traditional risk markers, was 2.55; this means that the discrimination of the model with addition of NT-proBNP is more than two and a half times as good as the discrimination using the model with the traditional risk markers.
Figure 2 presents the Kaplan-Meier curves, adjusted for competing risks, for 5-year cumulative cardiovascular morbidity or mortality for the model with traditional risk markers, the model with NT-proBNP and the model with all five new markers.
Figure 2. Kaplan-Meier curves, showing cumulative cardiovascular morbidity and mortality.
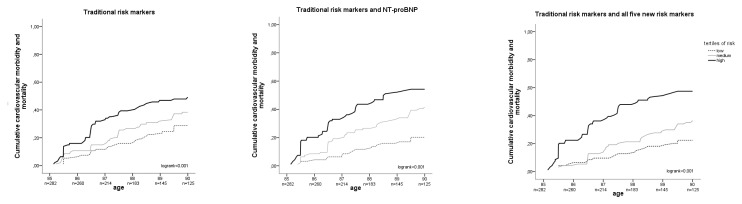
Kaplan-Meier curves, adjusted for competing risks, showing cumulative cardiovascular morbidity and mortality for tertiles of risk for three different models: traditional risk markers only (left graph), traditional risk markers plus NT-proBNP (middle graph), and traditional risk markers plus all five new markers (a history of major cardiovascular disease, MDRD, CRP, homocysteine and NT-proBNP) (right graph) (N=282).
When the analyses were repeated after exclusion of all participants with a history of heart failure according to information as obtained from their general practitioner or nursing home physician at baseline (n=68), results did not materially change (data not shown).
Cross validation of the model with the traditional risk markers led to a C-statistic of 0.53 (95%CI 0.46-0.60) for the traditional risk markers and a C-statistic of 0.64 (95%CI 0.58-0.71) for the model with addition of NT-proBNP. The difference between these two cross validated C-statistics was 0.11 (p=0.001). The cross validated model with addition of all five new markers had a C-statistic of 0.65 (95%CI 0.58-0.71), difference 0.12 (p=0.001).
Discussion
In this study in very old subjects with established CVD, traditional risk markers had little predictive value for recurrent cardiovascular events and cardiovascular mortality. Of all biomarkers NT-proBNP was by far the strongest, adding substantial predictive value beyond the traditional risk markers. Besides NT-proBNP, the nature of the CVD history is also a relevant risk predictor: participants with a history of MI, stroke or arterial surgery had a higher recurrence rate than those with a less severe CVD history. Addition of information about the CVD history improved risk classification in about one in four participants. MDRD was not related to the endpoint, CRP was related but failed to show substantial incremental value, and homocysteine was both related to the endpoint and improved classification. However, NT-proBNP was the only risk marker that showed unambiguous improvement of prediction in all currently advocated methods of evaluation.
In comparison with the literature, in the present study, the traditional risk markers had little predictive value, with a C-statistic of 0.59, compared to 0.67 in secondary prevention in younger age groups [40]; this suggests that, in secondary prevention in very old age, traditional risk markers indeed loose predictive value, as they do in primary prevention [41].
Although we already knew that the nature of the CVD history had prognostic value in very old age [19], we now found it also has incremental value in prediction models in secondary prevention.
New risk markers (renal markers (urinary albumin, eGFR, cystatin C) [31], CRP [30], homocysteine [25,42] and especially NT-proBNP [23,28,43–45] both individually and in various combinations have shown incremental predictive value in secondary prevention [27,46]. However, these studies mainly included younger participants.
We have now demonstrated that in the oldest old with established CVD, NT-proBNP is the most potent predictor for recurrent cardiovascular morbidity and cardiovascular mortality. In line with the literature [26,28,47] CRP and homocysteine, although associated with the outcome, show little additional predictive value when NT-proBNP is also available. As described earlier in very old age [48], CRP was a weaker marker in our study than in younger age groups . In the present study MDRD was not predictive for recurrent cardiovascular morbidity and cardiovascular mortality. Cystatin C may be a superior risk marker of kidney function in very old age [49].
The present study has several strengths. The Leiden 85-plus Study is an observational study of a cohort of very old inhabitants of the city of Leiden with no exclusion criteria and a high participation rate. Therefore, our results can be applied to the general population of the oldest old. Also, the laboratory tests used are easy to obtain in daily practice and are often already available to the physician. A limitation is the relative small sample size of this cohort. We therefore recommend validation of our findings in a larger cohort. Another limitation is that, although addition of NT-proBNP was shown to improve the prediction of cardiovascular morbidity and cardiovascular mortality, we do not know whether incorporating this new risk marker in risk stratification in daily practice indeed improves CVD-free survival. Knowing that participants during the time of our study were not treated according to current guidelines, optimizing such treatment with statins and antihypertensive medication in participants with high NT-proBNP might considerably improve prognosis.
Regarding clinical implications, we think that physicians may already improve their risk estimation by using available knowledge on patients’ CVD history: patients with a history of major CVD are at increased risk. Likewise, CRP and homocysteine are related to bad cardiovascular outcomes and may also be used when available. However, our results call for incorporation of NT-proBNP in risk estimation in secondary prevention in very old age as it improves identification of high-risk patients that will probably benefit most from intensified secondary preventive treatment and follow-up.
In conclusion, the use of NT-proBNP should be considered when estimating risk for recurrent cardiovascular events and cardiovascular mortality in secondary prevention in very old age.
Novelty and Significance
What Is New?
• In secondary prevention in very old patients, measurement of NT-proBNP markedly improves prediction of recurrent cardiovascular events and cardiovascular mortality.
What Is Relevant?
• NT-proBNP might be used to select older people at the highest risk for recurrent cardiovascular events who may benefit most from strict secondary prevention.
Summary
Among very old subjects with established CVD, NT-proBNP was the strongest risk marker for recurrent cardiovascular events and cardiovascular mortality and considerably adds prognostic value on top of traditional risk markers. When estimating risk in secondary prevention in very old age, use of NT-proBNP should be considered.
Funding Statement
The Leiden 85-plus Study was partly funded by an unrestricted grant from the Dutch Ministry of Health, Welfare and Sports. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. No additional external funding was received for this study.
References
- 1. Roger VL, Go AS, Lloyd-Jones DM, Adams RJ, Berry JD et al. (2011) Heart disease and stroke statistics--2011 update: a report from the American Heart Association. Circulation 123: e18-e209. doi: 10.1161/CIR.0b013e3182009701. PubMed: 21160056. CIR.0b013e3182009701 . PII;Available: 10.1161/CIR.0b013e3182009701 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. McDermott MM (2007) The international pandemic of chronic cardiovascular disease. JAMA 297: 1253-1255. doi: 10.1001/jama.297.11.1253. PubMed: 17374819. [DOI] [PubMed] [Google Scholar]
- 3. Kerr AJ, Broad J, Wells S, Riddell T, Jackson R (2009) Should the first priority in cardiovascular risk management be those with prior cardiovascular disease? Heart 95: 125-129. doi: 10.1136/hrt.2007.140905. PubMed: 18381374. hrt.2007.140905 . PII;Available: 10.1136/hrt.2007.140905 [DOI] [PubMed] [Google Scholar]
- 4. Steg PG, Bhatt DL, Wilson PW, D'Agostino R Sr., Ohman EM et al. (2007) One-year cardiovascular event rates in outpatients with atherothrombosis. JAMA 297: 1197-1206. doi: 10.1001/jama.297.11.1197. PubMed: 17374814. 297/11/1197 . PII;Available: 10.1001/jama.297.11.1197 [DOI] [PubMed] [Google Scholar]
- 5. Bhatt DL, Peterson ED, Harrington RA, Ou FS, Cannon CP et al. (2009) Prior polyvascular disease: risk factor for adverse ischaemic outcomes in acute coronary syndromes. Eur Heart J 30: 1195-1202. doi: 10.1093/eurheartj/ehp099. PubMed: 19339264. ehp099 . PII;Available: 10.1093/eurheartj/ehp099 [DOI] [PubMed] [Google Scholar]
- 6. Arima H, Tzourio C, Butcher K, Anderson C, Bousser MG, et al. (2006) Prior events predict cerebrovascular and coronary outcomes in the PROGRESS trial. Stroke 37: 1497-1502. 01.STR 0000221212.36860.c9 [pii];Available online at: 10.1161/01.STR.0000221212.36860.c9 [doi] [DOI] [PubMed] [Google Scholar]
- 7. Touze E, Varenne O, Chatellier G, Peyrard S, Rothwell PM, et al. (2005) Risk of myocardial infarction and vascular death after transient ischemic attack and ischemic stroke: a systematic review and meta-analysis. Stroke 36: 2748-2755. 01.STR 0000190118.02275.33 [pii];Available online at: 10.1161/01.STR.0000190118.02275.33 [doi] [DOI] [PubMed] [Google Scholar]
- 8. Alhusban A, Fagan SC (2011) Secondary prevention of stroke in the elderly: a review of the evidence. Am J Geriatr Pharmacother 9: 143-152. S1543-5946(11)00050-X [pii];Available online at: 10.1016/j.amjopharm.2011.04.002 [doi] [DOI] [PubMed] [Google Scholar]
- 9. Asberg S, Henriksson KM, Farahmand B, Asplund K, Norrving B et al. (2010) Ischemic stroke and secondary prevention in clinical practice: a cohort study of 14,529 patients in the Swedish Stroke Register. Stroke 41: 1338-1342. doi: 10.1161/STROKEAHA.110.580209. PubMed: 20522818. STROKEAHA.110.580209 . PII;Available: 10.1161/STROKEAHA.110.580209 [DOI] [PubMed] [Google Scholar]
- 10. Deedwania P, Stone PH, Bairey Merz CN, Cosin-Aguilar J, Koylan N, et al. (2007) Effects of intensive versus moderate lipid-lowering therapy on myocardial ischemia in older patients with coronary heart disease: results of the Study Assessing Goals in the Elderly (SAGE). Circulation 115: 700-707. CIRCULATIONAHA106.654756 [pii];Available online at: 10.1161/CIRCULATIONAHA.106.654756 [doi] [DOI] [PubMed] [Google Scholar]
- 11. Shepherd J, Blauw GJ, Murphy MB, Bollen EL, Buckley BM et al. (2002) Pravastatin in elderly individuals at risk of vascular disease (PROSPER): a randomised controlled trial. Lancet 360: 1623-1630. doi: 10.1016/S0140-6736(02)11600-X. PubMed: 12457784. S014067360211600X . PII. [DOI] [PubMed] [Google Scholar]
- 12. Macchia A, Romero M, D'Ettorre A, Mariani J, Tognoni G (2012) Temporal trends of the gaps in post-myocardial infarction secondary prevention strategies of co-morbid and elderly populations vs. younger counterparts: an analysis of three successive cohorts between 2003 and 2008. Eur Heart J 33: 515-522. doi: 10.1093/eurheartj/ehr410. PubMed: 22096090. ehr410 . PII;Available: 10.1093/eurheartj/ehr410 [DOI] [PubMed] [Google Scholar]
- 13. de Ruijter W, de Waal MW, Gussekloo J, Assendelft WJ, Blom JW (2010) Time trends in preventive drug treatment after myocardial infarction in older patients. Br J Gen Pract 60: 47-49: 47–9. doi: 10.3399/bjgp10X482103. PubMed: 20040168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Zuckerman IH, Yin X, Rattinger GB, Gottlieb SS, Simoni-Wastila L et al. (2012) Effect of Exposure to Evidence-Based Pharmacotherapy on Outcomes After Acute Myocardial Infarction in Older Adults. J Am Geriatr Soc, 60: 1854–61. doi: 10.1111/j.1532-5415.2012.04165.x. PubMed: 23003000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Subherwal S, Patel MR, Kober L, Peterson ED, Jones WS, et al. (2012) Missed opportunities: despite improvement in use of cardioprotective medications among patients with lower-extremity peripheral artery disease, underuse remains. Circulation 126: 1345-1354. CIRCULATIONAHA112.108787 [pii];Available online at: 10.1161/CIRCULATIONAHA.112.108787 [doi] [DOI] [PubMed] [Google Scholar]
- 16. Naderi SH, Bestwick JP, Wald DS (2012) Adherence to Drugs That Prevent Cardiovascular Disease: Meta-analysis on 376,162 Patients. Am J Med, 125: S0002-S9343. PubMed: 22748400. 12)00018-6 . PII;Available: 10.1016/j.amjmed.2011.12.013 [DOI] [PubMed] [Google Scholar]
- 17. Bhatt DL, Steg PG, Ohman EM, Hirsch AT, Ikeda Y et al. (2006) International prevalence, recognition, and treatment of cardiovascular risk factors in outpatients with atherothrombosis. JAMA 295: 180-189. doi: 10.1001/jama.295.2.180. PubMed: 16403930. 295/2/180 . PII;Available: 10.1001/jama.295.2.180 [DOI] [PubMed] [Google Scholar]
- 18. Uthoff H, Staub D, Socrates T, Meyerhans A, Bundi B et al. (2010) PROCAM-, FRAMINGHAM-, S. VASA 39: 325-333. doi: 10.1024/0301-1526/a000057. PubMed: 21104622. [DOI] [PubMed] [Google Scholar]
- 19. van Peet PG, Drewes YM, de Craen AJ, Westendorp RG, Gussekloo J et al. (2012) Prognostic value of cardiovascular disease status: the Leiden 85-plus study. Age (Dordr ) . 10.1007/s11357-012-9443-5 [doi]. [DOI] [PMC free article] [PubMed]
- 20. Dorresteijn JA, Visseren FL, Wassink AM, Gondrie MJ, Steyerberg EW et al. (2013) Development and validation of a prediction rule for recurrent vascular events based on a cohort study of patients with arterial disease: the SMART risk score. Heart 99: 866-872. doi: 10.1136/heartjnl-2013-303640. PubMed: 23574971. heartjnl-2013-303640 . PII;Available: 10.1136/heartjnl-2013-303640 [DOI] [PubMed] [Google Scholar]
- 21. Melander O, Newton-Cheh C, Almgren P, Hedblad B, Berglund G et al. (2009) Novel and conventional biomarkers for prediction of incident cardiovascular events in the community. JAMA 302: 49-57. doi: 10.1001/jama.2009.943. PubMed: 19567439. 302/1/49 . PII;Available: 10.1001/jama.2009.943 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Kavousi M, Elias-Smale S, Rutten JH, Leening MJ, Vliegenthart R et al. (2012) Evaluation of newer risk markers for coronary heart disease risk classification: a cohort study. Ann Intern Med 156: 438-444. doi: 10.7326/0003-4819-156-6-201203200-00006. PubMed: 22431676. 156/6/438 . PII;Available: [DOI] [PubMed] [Google Scholar]
- 23. Wannamethee SG, Welsh P, Lowe GD, Gudnason V, Di Angelantonio E, et al. (2011) N-terminal pro-brain natriuretic Peptide is a more useful predictor of cardiovascular disease risk than C-reactive protein in older men with and without pre-existing cardiovascular disease. J Am Coll Cardiol 58: 56-64. S0735-1097(11)01343-X [pii];Available online at: 10.1016/j.jacc.2011.02.041 [doi] [DOI] [PubMed] [Google Scholar]
- 24. Blankenberg S, Zeller T, Saarela O, Havulinna AS, Kee F, et al. (2010) Contribution of 30 biomarkers to 10-year cardiovascular risk estimation in 2 population cohorts: the MONICA, risk, genetics, archiving, and monograph (MORGAM) biomarker project. Circulation 121: 2388-2397. CIRCULATIONAHA109.901413 [pii];Available online at: 10.1161/CIRCULATIONAHA.109.901413 [doi] [DOI] [PubMed] [Google Scholar]
- 25. Humphrey LL, Fu R, Rogers K, Freeman M, Helfand M (2008) Homocysteine level and coronary heart disease incidence: a systematic review and meta-analysis. Mayo Clin Proc 83: 1203-1212. S0025-6196(11)60637-X [pii];Available online at: 10.4065/83.11.1203 [doi] [DOI] [PubMed] [Google Scholar]
- 26. Bibbins-Domingo K, Gupta R, Na B, Wu AH, Schiller NB et al. (2007) N-terminal fragment of the prohormone brain-type natriuretic peptide (NT-proBNP), cardiovascular events, and mortality in patients with stable coronary heart disease. JAMA 297: 169-176. doi: 10.1001/jama.297.2.169. PubMed: 17213400. 297/2/169 . PII;Available: 10.1001/jama.297.2.169 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Shlipak MG, Ix JH, Bibbins-Domingo K, Lin F, Whooley MA (2008) Biomarkers to predict recurrent cardiovascular disease: the Heart and Soul Study. Am J Med 121: 50-57. S0002-9343(07)00844-3 [pii];Available online at: 10.1016/j.amjmed.2007.06.030 [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Blankenberg S, McQueen MJ, Smieja M, Pogue J, Balion C, et al. (2006) Comparative impact of multiple biomarkers and N-Terminal pro-brain natriuretic peptide in the context of conventional risk factors for the prediction of recurrent cardiovascular events in the Heart Outcomes Prevention Evaluation (HOPE) Study. Circulation 114: 201-208. CIRCULATIONAHA105.590927 [pii];Available online at: 10.1161/CIRCULATIONAHA.105.590927 [doi] [DOI] [PubMed] [Google Scholar]
- 29. Battistoni A, Rubattu S, Volpe M (2012) Circulating biomarkers with preventive, diagnostic and prognostic implications in cardiovascular diseases. Int J Cardiol, 157: S0167-S5273. PubMed: 21763018. 11)00606-1 . PII;Available: 10.1016/j.ijcard.2011.06.066 [DOI] [PubMed] [Google Scholar]
- 30. Sattar N, Murray HM, McConnachie A, Blauw GJ, Bollen EL, et al. (2007) C-reactive protein and prediction of coronary heart disease and global vascular events in the Prospective Study of Pravastatin in the Elderly at Risk (PROSPER). Circulation 115: 981-989. CIRCULATIONAHA106.643114 [pii];Available online at: 10.1161/CIRCULATIONAHA.106.643114 [doi] [DOI] [PubMed] [Google Scholar]
- 31. Ford I, Bezlyak V, Stott DJ, Sattar N, Packard CJ et al. (2009) Reduced glomerular filtration rate and its association with clinical outcome in older patients at risk of vascular events: secondary analysis. PLoS Med 6: e16. doi: 10.1371/journal.pmed.1000016. PubMed: 19166266. 08-PLME-RA-1471 . PII;Available: 10.1371/journal.pmed.1000016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. van der Wiel AB, van Exel E, de Craen AJ, Gussekloo J, Lagaay AM et al. (2002) A high response is not essential to prevent selection bias: results from the Leiden 85-plus study. J Clin Epidemiol 55: 1119-1125. doi: 10.1016/S0895-4356(02)00505-X. PubMed: 12507676. S089543560200505X . PII. [DOI] [PubMed] [Google Scholar]
- 33. Macfarlane PW, Latif S (1996) Automated serial ECG comparison based on the Minnesota code. J Electrocardiol 29 Suppl: 29-34. doi: 10.1016/S0022-0736(96)80016-1. PubMed: 9238374. [DOI] [PubMed] [Google Scholar]
- 34. Verduijn M, Grootendorst DC, Dekker FW, Jager KJ, le Cessie S (2011) The analysis of competing events like cause-specific mortality--beware of the Kaplan-Meier method. Nephrol Dial Transplant 26: 56-61. doi: 10.1093/ndt/gfq661. PubMed: 21059831. gfq661 . PII;Available: 10.1093/ndt/gfq661 [DOI] [PubMed] [Google Scholar]
- 35. Pencina MJ, d'Agostino RB, Vasan RS (2010) Statistical methods for assessment of added usefulness of new biomarkers. Clin Chem Lab Med 48: 1703-1711. doi: 10.1515/CCLM.2010.340. PubMed: 20716010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Pencina MJ, D'Agostino RB Sr., Steyerberg EW (2011) Extensions of net reclassification improvement calculations to measure usefulness of new biomarkers. Stat Med 30: 11-21. doi: 10.1002/sim.4085. PubMed: 21204120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Pencina MJ, D'Agostino RB Sr., D'Agostino RB Jr., Vasan RS (2008) Evaluating the added predictive ability of a new marker: from area under the ROC curve to reclassification and beyond. Stat Med 27: 157-172. doi: 10.1002/sim.2929. PubMed: 17569110. [DOI] [PubMed] [Google Scholar]
- 38. Pencina MJ, D'Agostino RB, Pencina KM, Janssens AC, Greenland P (2012) Interpreting incremental value of markers added to risk prediction models. Am J Epidemiol 176: 473-481. doi: 10.1093/aje/kws207. PubMed: 22875755. kws207 . PII;Available: 10.1093/aje/kws207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Van Houwelingen JC, le Cessie S (1990) Predictive value of statistical models. Stat Med 9: 1303-1325. doi: 10.1002/sim.4780091109. PubMed: 2277880. [DOI] [PubMed] [Google Scholar]
- 40. Zhang M, Lu S, Wu X, Chen Y, Song X, et al. (2012) Multimarker approach for the prediction of cardiovascular events in patients with mild to moderate coronary artery lesions. A 3-year follow-up study. Int Heart J 53: 85-90. DN/JST.JSTAGE/ihj/53.85 [pii]. [DOI] [PubMed] [Google Scholar]
- 41. de Ruijter W, Westendorp RG, Assendelft WJ, den Elzen WP, de Craen AJ et al. (2009) Use of Framingham risk score and new biomarkers to predict cardiovascular mortality in older people: population based observational cohort study. BMJ 338: a3083. doi: 10.1136/bmj.a3083. PubMed: 19131384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Anderson JL, Muhlestein JB, Horne BD, Carlquist JF, Bair TL et al. (2000) Plasma homocysteine predicts mortality independently of traditional risk factors and C-reactive protein in patients with angiographically defined coronary artery disease. Circulation 102: 1227-1232. doi: 10.1161/01.CIR.102.11.1227. PubMed: 10982535. [DOI] [PubMed] [Google Scholar]
- 43. Vaes B, de Ruijter W, Degryse J, Westendorp RG, Gussekloo J (2009) Clinical relevance of a raised plasma N-terminal pro-brain natriuretic peptide level in a population-based cohort of nonagenarians. J Am Geriatr Soc 57: 823-829. doi: 10.1111/j.1532-5415.2009.02218.x. PubMed: 19470010. JGS2218 . PII;Available: 10.1111/j.1532-5415.2009.02218.x [DOI] [PubMed] [Google Scholar]
- 44. Di Angelantonio E, Chowdhury R, Sarwar N, Ray KK, Gobin R, et al. (2009) B-type natriuretic peptides and cardiovascular risk: systematic review and meta-analysis of 40 prospective studies. Circulation 120: 2177-2187. CIRCULATIONAHA109.884866 [pii];Available online at: 10.1161/CIRCULATIONAHA.109.884866 [doi] [DOI] [PubMed] [Google Scholar]
- 45. Zethelius B, Berglund L, Sundström J, Ingelsson E, Basu S et al. (2008) Use of multiple biomarkers to improve the prediction of death from cardiovascular causes. N Engl J Med 358: 2107-2116. doi: 10.1056/NEJMoa0707064. PubMed: 18480203. 358/20/2107 . PII;Available: 10.1056/NEJMoa0707064 [DOI] [PubMed] [Google Scholar]
- 46. Pikula A, Beiser AS, Decarli C, Himali JJ, Debette S, et al. (2012) Multiple biomarkers and risk of clinical and subclinical vascular brain injury: the framingham offspring study. Circulation 125: 2100-2107. CIRCULATIONAHA110.989145 [pii];Available online at: 10.1161/CIRCULATIONAHA.110.989145 [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Kistorp C, Raymond I, Pedersen F, Gustafsson F, Faber J et al. (2005) N-terminal pro-brain natriuretic peptide, C-reactive protein, and urinary albumin levels as predictors of mortality and cardiovascular events in older adults. JAMA 293: 1609-1616. doi: 10.1001/jama.293.13.1609. PubMed: 15811980. 293/13/1609 . PII;Available: 10.1001/jama.293.13.1609 [DOI] [PubMed] [Google Scholar]
- 48. Strandberg TE, Tilvis RS (2000) C-Reactive Protein, Cardiovascular Risk Factors, and Mortality in a Prospective Study in the Elderly. Arterioscler Thromb Vasc Biol 20: 1057-1060. doi: 10.1161/01.ATV.20.4.1057. PubMed: 10764673. [DOI] [PubMed] [Google Scholar]
- 49. Manzano-Fernandez S, Boronat-Garcia M, Albaladejo-Oton MD, Pastor P, Garrido IP, et al. (2009) Complementary prognostic value of cystatin C, N-terminal pro-B-type natriuretic Peptide and cardiac troponin T in patients with acute heart failure. Am J Cardiol 103: 1753-1759. S0002-9149(09)00611-0 [pii];Available online at: 10.1016/j.amjcard.2009.02.029 [doi] [DOI] [PubMed] [Google Scholar]


