Abstract
The immune system in the female reproductive tract (FRT) does not mount an attack against HIV or other sexually transmitted infections (STI) with a single endogenously produced microbicide or with a single arm of the immune system. Instead, the body deploys dozens of innate antimicrobials to the secretions of the female reproductive tract. Working together, these antimicrobials along with mucosal antibodies attack many different viral, bacterial and fungal targets. Within the FRT, the unique challenges of protection against sexually transmitted pathogens coupled with the need to sustain the development of an allogeneic fetus have evolved in such a way that sex hormones precisely regulate immune function to accomplish both tasks. The studies presented in this review demonstrate that estradiol and progesterone secreted during the menstrual cycle act both directly and indirectly on epithelial cells and other immune cells in the reproductive tract to modify immune function in a way that is unique to specific sites throughout the FRT. As presented in this review, studies from our laboratory and others demonstrate that the innate immune response is under hormonal control, varies with the stage of the menstrual cycle, and as such is suppressed at mid-cycle to optimize conditions for successful fertilization and pregnancy. In doing so, a window of STI vulnerability is created during which potential pathogens including HIV enter the reproductive tract to infect host targets.
Keywords: Epithelial cells, estradiol, female reproductive tract, mucosal immunity
Introduction
Sexually transmitted infections have reached epidemic proportions throughout the world with more than 20 pathogens transmitted through sexual intercourse. The World Health Organization estimates more than 300 million new infections of Trichonomas vaginalis, Chlamydia trachomatis or Neisseria gonorrhea occur annually throughout the world.1 Some STI can be transmitted vertically to the fetus, resulting in preterm deliveries and/or life-threatening systemic illness in newborn infants. Generally, adolescents and young adults are the demographic age groups most frequently affected with STI, and women are more likely than men to suffer the consequences of these serious infections.
Human immunodeficiency virus (HIV) is recognized as a life-threatening sexually transmitted disease that is unique in its rapid spread and the depth of its impact. With 25 million deaths worldwide and an additional 33.2 million (of which 50% are women) infected worldwide, HIV/AIDS is one of the world’s worst pandemics.2 Since the 1980s, HIV has shifted from a disease spread predominantly through needles and male–male contact to a sexually transmitted disease in which women worldwide are more likely to be infected than men. Presently, women and girls make up almost 57% of all people infected with HIV in Sub-Saharan Africa, where a striking 76% of young people (aged 15–24) living with HIV are female.2
Within the FRT, the mucosal immune system functions as the first line of defense.3–5 In response to the unique requirements of balancing immune protection with procreation, the immune system in the FRT, which consists of both innate and adaptive immune components, is responsive to and precisely regulated by estradiol and progesterone, both of which are produced in a cyclic fashion by the ovary over the course of the menstrual cycle. In preparing the reproductive tract for fertilization and implantation, estradiol and progesterone simultaneously regulate the immune system in the fallopian tubes, uterus, cervix, and vagina to compliment the reproductive process (see6 for review).
The mucosal immune system in the FRT consists of immune cells that migrate into the uterus, cervix, and vagina as well as resident epithelial cells and supportive stromal cells.6 Sex hormones influence the migration of macrophages and dendritic cells as well as T and B cells by affecting the expression of adhesion molecules and chemotactic factors.6–9 Among those cells pivotal in conferring immune protection, epithelial cells are recognized as pluripotential in their ability to confer immune protection. Epithelial cells, in addition to providing barrier protection, transport immunoglobulins (IgA and IgG) into FRT secretions and produce antimicrobials that are both bactericidal and viricidal.7,10 Through the production of cytokines and chemokines, these cells signal the recruitment and activation of other cells of the innate and adaptive immune. What is unique to the FRT is that epithelial cells are responsive to both the direct and indirect effects of sex hormones. 7,9 In this dynamic balance, epithelial cells throughout the FRT respond directly to estradiol and progesterone, as well as indirectly to the cytokines and growth factors produced by resident (fibroblasts) and migratory cells (immune cells) in the reproductive tract. What is clear is that this responsiveness is part of the bidirectional communication that occurs in which epithelial cells direct both reproductive as well as immune function to maintain an effective level of protection, which distinguishes between pathogens, commensals, allogeneic sperm, and the developing fetus. The pleiotrophic capacity of epithelial cells has led to their recognition as sentinels, the functions of which are only now being recognized. 6,11,12
This review will focus on current knowledge regarding the sentinel role of epithelial cells in the human female reproductive tract with special emphasis on uterine and vaginal epithelial cells, especially as it pertains to protection against genital tract pathogens. Our goal is to highlight some of the unique responses of these cells to estradiol and progesterone and to point out that, in addition to direct hormonal effects on particular cells, there are the equally important indirect actions of estradiol mediated through growth factors, cytokines, and chemokines.
Menstrual cycle effects on epithelial cells and immune cells in the FRT
The immune system in the FRT has evolved to be responsive to and precisely regulated by the female sex hormones, estradiol, and progesterone, which are produced in a cyclic fashion by the ovary over the course of the menstrual cycle. In preparing the reproductive tract for fertilization and implantation, estradiol and progesterone simultaneously regulate the immune system in the fallopian tubes, uterus, cervix, and vagina to compliment the reproductive process (See5 for review). We and others have found that over the course of the menstrual cycle, immune cells are present in substantial numbers and nonuniformly distributed in both the stromal layer and the epithelium of the FRT.13–16 When tissues were dispersed by enzymatic or mechanical means for quantitative flow cytometry or functional analyses, leukocytes were found to be 6–20% of the total number of cells within the FRT.17 When Natural killer (NK) cells were included in this analysis, the population of immune cells doubled.18 These studies indicated that T-lymphocytes (CD4+ and/or CD8+) are a major constituent of reproductive tract leukocytes from all tissues. The fallopian tube contained granulocytes as a second major constituent but were significantly less numerous in the other tissues. All tissues contained B-lymphocytes and monocytes as clearly detectable but minor components. Over the course of the menstrual cycle, subtle changes were observed in the migration of macrophages, B cells and neutrophils into the lower tract,17,19 and in dendritic cells entering the squamous epithelium.20
Immune Cell Migration
As seen in Fig. 1, the distribution of immune cells in the FRT varies with the site examined. Exclusive in the uterus are lymphoid aggregates, which consist of a B-cell core surrounded by T cells and an outer halo of macrophages (Fig. 1a, f). The B-cell core (CD19+) was most often seen in large aggregates present in the late proliferative and secretory stages of the cycle.21 Phenotypic analysis indicated that T cells are almost exclusively CD3+, CD8+, and CD4−). Aggregates containing only cells of the CD3+CD4+ phenotype were occasionally found as were individual CD4+ cells located outside the aggregates in the stroma. Monocytes/macrophages (CD14+ cells) were found as a mantle around the T cells. FRT aggregates are anatomically and functionally distinct from Peyer’s patches in the intestine.21 The size of lymphoid aggregates was found to vary with the stage of the menstrual cycle, in that aggregates were significantly larger during the secretory (3000–4000 cells) than the proliferative stage (300–400 cells). The distribution and frequency of CD8+ T cells in aggregates using expression of Vb2 or Vb8 as markers of clonality and Ki-67 as markers of dividing cells led to the conclusion that lymphoid aggregates form largely by the trafficking of cells to nucleation sites within the endometrium, rather than by division of precursor cells.22 The lower reproductive tract, while lacking lymphoid aggregates, contains a full spectrum of immune cells located both within the submucosa and epithelial lining (Fig. 1g–i).
Fig. 1.
Schematic of the mucosal immune system throughout the human female reproductive tract. As seen in the drawing on the left side, the vagina and ectocervix are lined with squamous epithelial cells. Columnar cells are present throughout the upper FRT including the endocervix, uterine endometrium, and fallopian tubes. Panel a–d are confocal photomicrographs showing the distribution of immune cells throughout out the uterus. Frozen sections were directly stained with three fluorescently tagged monoclonal antibodies: an epithelial cell specific antibody (Cy3-labeled clone BerEP4, red color panels a–d), anti-CCR5 (FITC-labeled clone 2D7, green color panels a–d), and anti-CXCR4 (Cy5 labeled 12G5, blue color panels a–d). Panel a shows epithelial gland extending from the myometrial interface (far left) to the luminal epithelium (right-hand side). Panels b through d show higher magnification fields from the same region. The luminal epithelium, in contrast to the adjacent glands, retains a relatively intense BerEP4 expression (red color panels a and d). CCR5 expression is prominent on the lymphoid aggregates located in the stratum basalis immediately adjacent to the myometrium (‘LA’ in panel b). Epithelial expression of CCR5 is low in the stratum basalis and increases in the proximal third of the stratum functionalis. Panel f: This photograph plate consists of a lymphoid aggregate in the uterine endometrium at the late proliferative stage of the menstrual cycle. The three fluorochromes are T cells (Cy3-anti-CD3, red), B cells (FITC-anti-CD19, green), and macrophages (Cy5-anti CD14, blue). Panel e: Vaginal squamous cells expressing GalCer (green) is expressed on parabasal epithelial cells (p) and in the surface regions of the cornified layer. Panel g: CD8+ T cells present in both the submucosa and the squamous epithelium. Panel h: CD14 expression is found on both stroma and squamous epithelium macrophages. Panel i: CD1a-positive dendritic cells (DC) are present in the squamous epithelium (blue). Panel a–d;28 Panel f;22 Panel e, g, h, i.29
CTL Activity
Coincident with aggregate formation in the uterus, White et al. found that CD8+ cytotoxic T-lymphocyte (CTL) activity, measured in a redirected lyses assay, is suppressed in the uterus and fallopian tubes during the secretory stage of the cycle.23 This suppression occurs without any drop in CD8+T cell numbers. In the ectocervix and vagina, in contrast to the upper FRT, we found that CTL activity was measurable in tissues from women at the proliferative or secretory stages of the menstrual cycle.24
Chemokine Receptor Expression
Recognized as important molecules in implantation, 25,26 we measured the expression of chemokine receptors CXCR4 and CCR5 as well as CD4 on uterine epithelial cells and found that expression varies with the stage of the menstrual cycle (Fig. 1a–d).27 All three were low during the proliferative stage of the cycle, peaked at the time of ovulation and then either plateaued (CXCR4, CD4) or declined (CCR5) during the secretory stage of the cycle.27 Chemokine receptors on cultured endometrial epithelial cells showed an up-regulation and polarization of CXCR1, CXCR4, and CCR5 receptors when a human blastocyst was present.25 The distribution and regulation of these receptors in the endometrial epithelium and the human blastocyst suggest that each is essential in the apposition and adhesion phases of human implantation. In addition to expression in the upper FRT, leukocytes and epithelial cells in the lower tract (Fig. 1g–i) express CCR5 and GalCer on both.28 Yeaman et al. showed that basal and parabasal epithelial cells of the ectocervix express CD4, CCR5, and GalCer, unlike the mid-zone and superficial cells lining the lumen.28 Although changes in protein expression were not as pronounced as those seen in the uterus, histological evidence supported the conclusion that CD4 and CCR5 expression was greater during the proliferative stage than during the secretory stage of the cycle. An unintended consequence of chemokine expression in the upper and lower tract is that HIV-1, as a sexually transmitted pathogen, most likely uses these coreceptors to infect cells in the FRT.
Antibodies
The humoral immune system is hormonally controlled and varies with site analysed and stage of the menstrual cycle (for review, see10). In the uterus, levels of pIgR, the epithelial cell receptor responsible for transporting IgA from tissue to lumen varies with the menstrual cycle.29 When expressed as the percentage of total protein, luminal uterine SC levels were highest during the secretory phase, significantly reduced during the proliferative phase and lowest during menstruation. Total SC was also greatest during the secretory phase, averaging approximately 2-fold higher than SC in proliferative and menstrual samples. In other studies, IgG levels in secretions from the uterine mucosa were highest during the peri-ovulatory phase, whereas levels in the fallopian tube were lowest at that time.30 This study reached the conclusion that each organ (fallopian tubes, uterus, cervix, and vagina) and even different sites within each organ can respond independently from each other to changes in hormone levels, producing different types and amounts of secretory proteins. In the lower FRT, IgA, IgG, and lactoferrin levels in cervical mucus were depressed by 10- to 100-fold at mid-cycle relative to that seen early in the proliferative phase, only to rise toward the end of the menstrual cycle.31 When women were placed on oral contraceptives, immunoglobulins and lactoferrin levels were suppressed for the duration of hormone exposure. In other studies, in which cervical mucus was evaluated from 5 days before to 3 days after ovulation, IgA and IgG had a biphasic pattern with a peak before ovulation followed by a small increase after ovulation.32 More recently, Nardelli-Haefliger et al. demonstrated that titers of antihuman papillomavirus 16 virus-like particle (VLP) IgG in cervical secretions dropped approximately 9-fold at mid-cycle during ovulatory cycles suggesting increased vulnerability to pathogens at mid-cycle.33
Chemokines, Cytokines, and Antimicrobials
Chemokines and cytokines are central to the progressive tissue growth and remodeling that occur during each menstrual cycle, while antimicrobials protect against potential pathogens.6 While relatively little has been done to measure changes in upper FRT secretions during the menstrual cycle, analysis of cervical–vaginal secretions demonstrate that chemokines and cytokines (IL-6 and IL-8) as well as endogenously produced antimicrobials [secretory leukocyte protease inhibitor (SLPI), HBD2, HNP1-3, and lactoferrin] dropped significantly at mid-cycle (day 13) and remained depressed for 7–10 days, returning to proliferative stage levels just before menstruation.31,34 In contrast, total protein and transforming growth factor-beta (TGFβ) levels remained unchanged throughout the menstrual cycle. In other studies, human intestinal defensin-5 was highest in cervico-vaginal lavage during the secretory stage of the menstrual cycle.35
Overall, these studies support the hypothesis that hormonal changes during the menstrual cycle regulate the immune system throughout the FRT in a way that is synchronized with reproductive function which optimizes FRT conditions for successful sperm migration, fertilization, implantation, and pregnancy. The extent to which changes in the immune system are directly or indirectly mediated by estradiol is addressed in the following sections.
Sex hormone effects on epithelial cells in culture
Epithelial Cell Tight Junctions
Located throughout the FRT, epithelial cells provide a physical barrier to protect against microbial infection, as well as contribute to an environment for the transport and protection of sperm, ovum, or conceptus.7,36 Stratified squamous epithelial cells line the vagina and ectocervix, while tight junctions between the columnar epithelial cells maintain the integrity of the mucosal monolayer in the endometrium, endocervix, and fallopian tube (Fig. 1). Disruption of the tight junctions in the upper FRT or damage to the squamous epithelial layer in the lower tract can lead to infection, resulting in infertility and potential life-threatening illness. The tight junction barrier permits epithelial cells to functionally polarize to respond to different stimuli and serve as a directional conduit for different factors. For example, the epithelial cell polymeric immunoglobulin receptor (pIgR) traverses the epithelial cell from the basolateral side to the apical side to release IgA into the lumen.37 Uterine epithelial cells secrete cytokines such as TGFβ preferentially at the basolateral surface and TNFα at the apical surface. 38 In culture, columnar uterine epithelial cells generate an electrochemical gradient across a monolayer that reflects the presence of these tight junctions and the integrity of the physical barrier, thus barrier function can be measured as transepithelial resistance (TER).39–45 Physiological concentrations of estradiol significantly reduce the transepithelial resistance of mouse epithelial cells7,46 and porcine vas deferens epithelia,47 as well as disrupt adherens junctions in endothelial cells.48
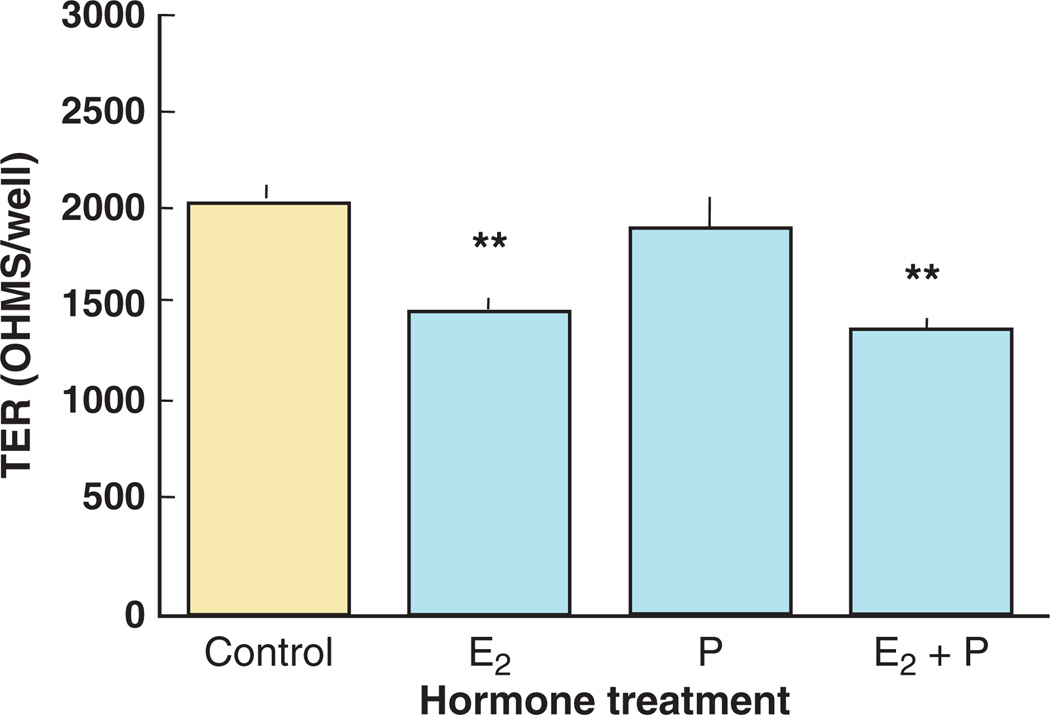
To determine the effect of estradiol and progesterone on human uterine epithelial cell barrier integrity, we grew the highly differentiated cell line ECC-141 in cell inserts until they achieved a consistent maximum TER of about 2000 Ω/insert. As seen in Fig. 2, the addition of 5 × 10−8 m estradiol for 48 hrs reduced TER by more than 25%. Progesterone at 1 × 10−7 m had no effect on TER and also failed to inhibit the reduction in TER caused by estradiol. A lower TER would result in an increase in fluid and molecule transport from the uterine tissue into the lumen, which can contribute to transport of sperm and conceptus.
Fig. 2.
Estradiol inhibits transepithelial resistance (TER) in the human uterine epithelial cell line ECC-1. ECC-1 cells were grown in media supplemented with stripped FBS until maximal TER of about 2000 Ω/well in cell inserts was obtained. Estradiol (5 × 10−8 m) and/or progesterone (1 × 10−7 m) were added to the basolateral compartment of some inserts, and TER was measured after 48 hr. Four wells per group; **significantly different (P < 0.01) from control.
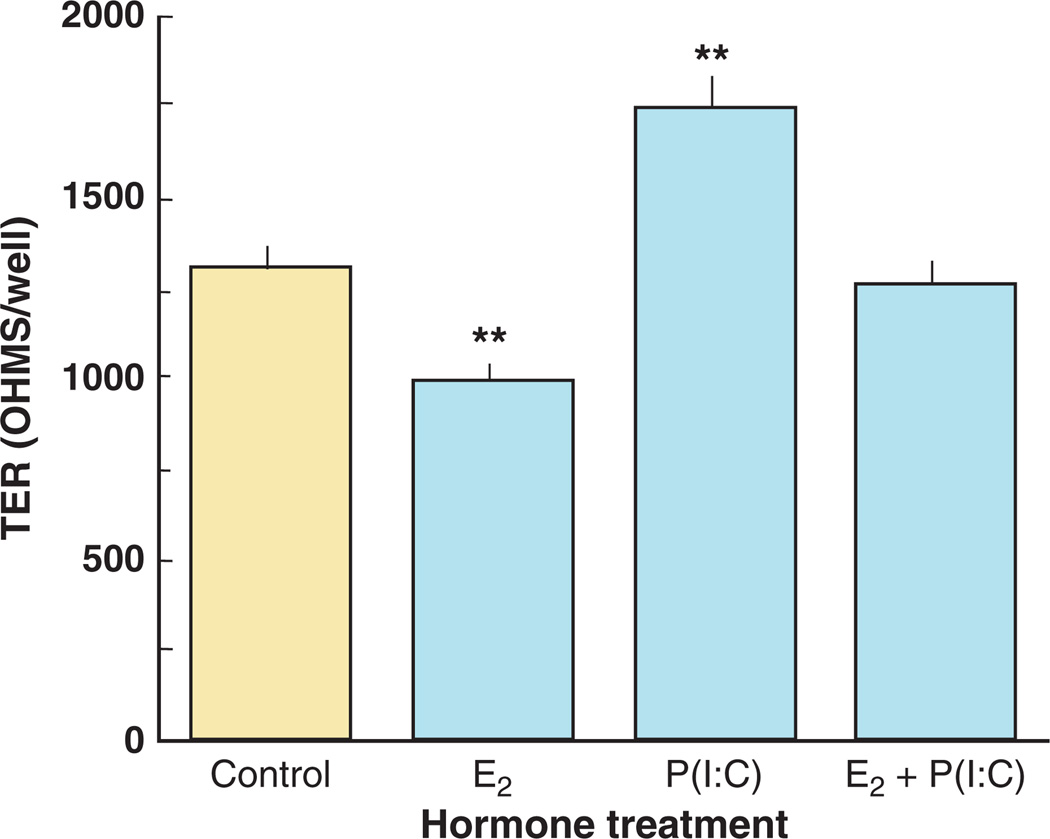
Human uterine epithelial cells express toll-like receptors (TLR) 1 through 9 and are therefore prepared to respond to a range of pathogens that include viruses, bacteria, and fungi.49,50 To determine the effect of a TLR3 agonist on TER in the presence of estradiol, the dsRNA viral mimic poly (I:C) was added to primary polarized human uterine epithelial cells with and without a pretreatment of estradiol for 24 hrs, and TER was measured. As shown in Fig. 3, estradiol treatment, similar to the ECC-1 cells, reduced TER in primary uterine cells. Unexpectedly, we found that in contrast to estradiol, poly (I:C) alone significantly increased TER. This finding suggests that epithelial cells increase barrier protection to viral challenge possibly to limit viral infection. Also shown is the effect of estradiol on poly (I:C) increases in TER. When pre-treated with estradiol, TER stimulation by poly (I:C) is muted and remains unchanged relative to control values. These findings suggest that in the context of viral challenge, which involves TLR3 ligation, estradiol lowers TER without affecting barrier protection. What remains to be determined is whether estradiol reduced TER translates into lower barrier protection in the presence of a viral pathogen.
Fig. 3.
Effect of estradiol and poly (I:C) on TER of primary uterine epithelial cells. Human uterine epithelial cells were cultured in cell inserts in media supplemented with stripped FBS until maximal TER was obtained. The media was changed and some inserts were cultured with 5 × 10−8 m estradiol for 24 hr and then an additional 24 hr with estradiol with or without apical treatment with poly (I:C) and TER was measured. Six wells per group; **significantly different (P < 0.01) from control.
We have previously suggested that the period of ovulation, which is initiated by an increase in estradiol, is a time of risk for infection by HIV because of decreases in a number of innate and adaptive immune mechanisms.5 It is important for reproductive success that the FRT immune system be regulated to prevent attack on sperm and conceptus, and estradiol is likely a major player in modulating specific FRT immune responses. Unfortunately, it is likely that the reduction in uterine epithelial cell TER because of estradiol may also contribute to the potential for infection by microbial pathogens in the upper tract.51
To further understand the modulation of TER in uterine epithelial cells by estradiol, we seeded ECC-1 cells in cell inserts and treated the cells 24 hrs later with estradiol, tamoxifen, or raloxifene. The latter two molecules are selective estrogen receptor modulators (SERMs) and are clinically relevant for the treatment of breast cancer (tamoxifen) and bone resorption (raloxifene). Treatments began 24 hr after seeding (Day 1, when TER was just developing and averaged 350 Ω/well) and cells were retreated at 48-hr intervals. As shown in Table I, TER was affected by all treatments. Consistent with data shown earlier, estradiol inhibited TER by more than 50%. The greater decrease seen here compared to aforementioned experiments is probably because of the effect of estradiol on developing TER whereas prior results were shown after maximum TER had established in untreated wells. This result infers that estradiol, increasing in concentration for the start of ovulation, may reduce barrier integrity and allow certain small pathogens such as HIV to enter underlying tissue and infect target cells. Also of interest was our finding that both SERMs significantly enhanced TER over control values. These results suggest that the use of SERMs, perhaps in a cervical ring that is commercially available for contraceptive purposes, may be beneficial in the protection of infection by microorganisms that cause STI such as HIV-1. Thus, treatment with SERMs might be valuable to close the window of vulnerability to HIV infection afforded by endogenous estradiol.6
Table I.
Effects of estradiol, tamoxifen, and raloxifene on transepithelial resistance of ECC-1 cells. ECC-1 cells were seeded in cell inserts and treatments with estradiol or the SERMs began 24 hr after seeding (Day 1, when TER was just developing and averaged 350 Ω/well) and cells were retreated at 48-hr intervals after TER measurements were assessed. TER was affected by all treatments; estradiol significantly inhibited TER by more than 50% compared to control. SERMs enhanced TER. The data represent the mean and SEM of four wells per group.
| Transepithelial resistance (TER) | |||
|---|---|---|---|
| Days in culture | 3 | 5 | 7 |
| Control | 2193 | 3203 | 3343 |
| 221 | 276 | 123 | |
| Estradiol | 1030b | 1297b | 1430b |
| 175 | 48 | 21 | |
| Tamoxifin | 3067a | 4520a | 5727a |
| 227 | 316 | 334 | |
| Raloxifene | 2960a | 6200a | 9620a |
| 698 | 408 | 811 | |
Significantly (P < 0.01) greater than controls.
Significantly (P < 0.01) lower than controls.
Chemokines, Cytokines, and Antimicrobials
In addition to a physical barrier, uterine epithelial cells provide protection from pathogen infection through the production of antimicrobials, as well as the secretion of chemokines and cytokines that recruit and activate immune cells of both the innate and adaptive immune systems.6 How estradiol regulates the secretion of these soluble factors can determine whether or not infection will occur. The functions of antimicrobial factors in host defense are multifaceted and range from direct killing of invading microbes (bacteria, fungi, and viruses) to linking innate and acquired immunity.7 We and others have previously shown that epithelial cells in the FRT secrete factors that exhibit potent antimicrobial activity and include human α-defensin-5 (HD5), β-defensins 1-4 (HBD1-4), elafin, and SLPI.12,35,52–55 These antimicrobial peptides, some of which are induced in response to TLR ligand exposure, are effective at inhibiting Gram-positive and Gram-negative bacteria, fungi, and enveloped viruses, including Neisseria gonorrhoeae, Chlamydia trachomatis, Candida albicans, herpes simplex virus-2, and HIV-1.56–62 Other uterine epithelial cell factors with antiviral activity, notably against HIV-1, are the chemokines MIP-1α, MIP-1β, RANTES, and SDF-1α, which inhibit HIV-1 infection of target host cells by interfering with the ability of HIV-1 to bind coreceptors CCR5 and CXCR4 found on host cells.63,64 More recently, we demonstrated that human uterine and fallopian tube epithelial cells produce MIP3α and Trappin-2/Elafin and that these recombinant peptides inhibit HIV infection.65,66
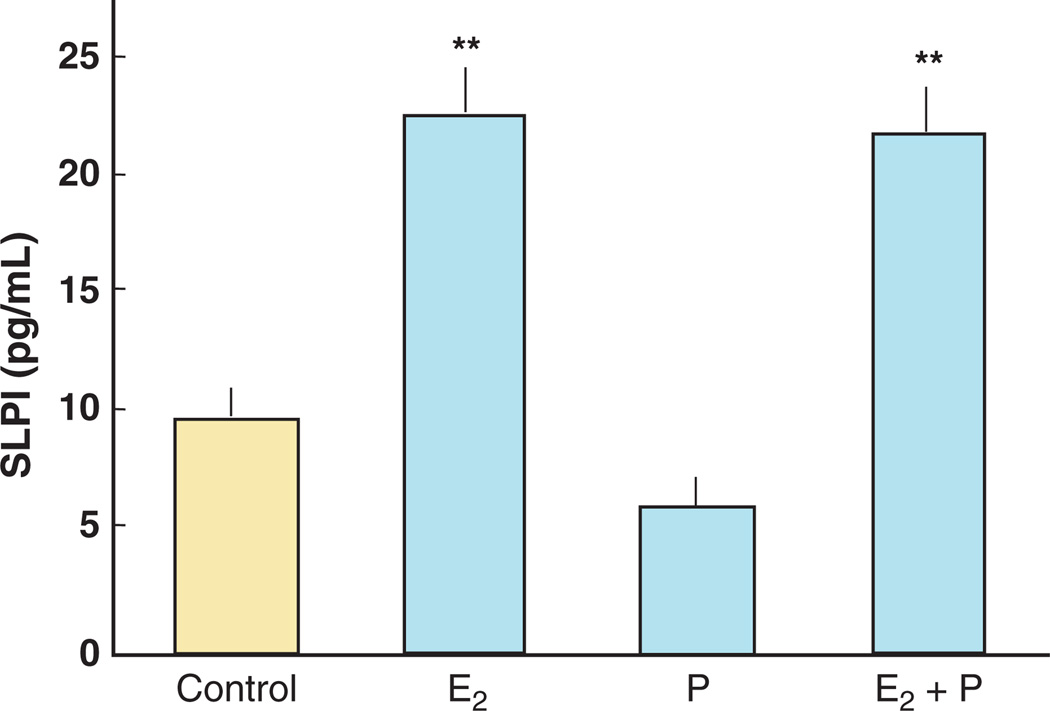
To determine if sex hormones stimulate uterine epithelial cells to secrete the antimicrobial SLPI, we added estradiol and/or progesterone to polarized ECC-1 cells. As shown in Fig. 4, estradiol significantly increased the secretion of SLPI; progesterone neither enhanced SLPI secretion nor negated the estradiol-induced effect. These data indicate that uterine epithelial cells constitutively secrete SLPI and that estradiol enhances that secretion. Further evidence that estradiol induces protection from infection was our study that demonstrated that apical secretions from polarized human uterine epithelial cells from pre-menopausal women, but not from post-menopausal women, had anti-bacterial activity against both Gram-positive and Gram-negative bacteria. 67 The antibacterial activity was partially neutralized with antibody to SLPI. These data support our working hypothesis that endocrine balance during the menstrual cycle modulates the sentinel function of epithelial cells and offers an explanation as why susceptibility to sexually transmitted pathogens varies with stage of the menstrual cycle.68
Fig. 4.
Estradiol stimulates secretion of SLPI by ECC-1 cells. ECC-1 cells were grown to confluence and high TER in cell inserts. The media was changed and some inserts were cultured with 5 × 10−8 m estradiol and/or progesterone (1 × 10−7 m) for 48 hr. Apical conditioned media were recovered, centrifuged to remove cell debris, and SLPI was measured by ELISA. Four wells per group; **significantly different (P < 0.01) from control or progesterone.
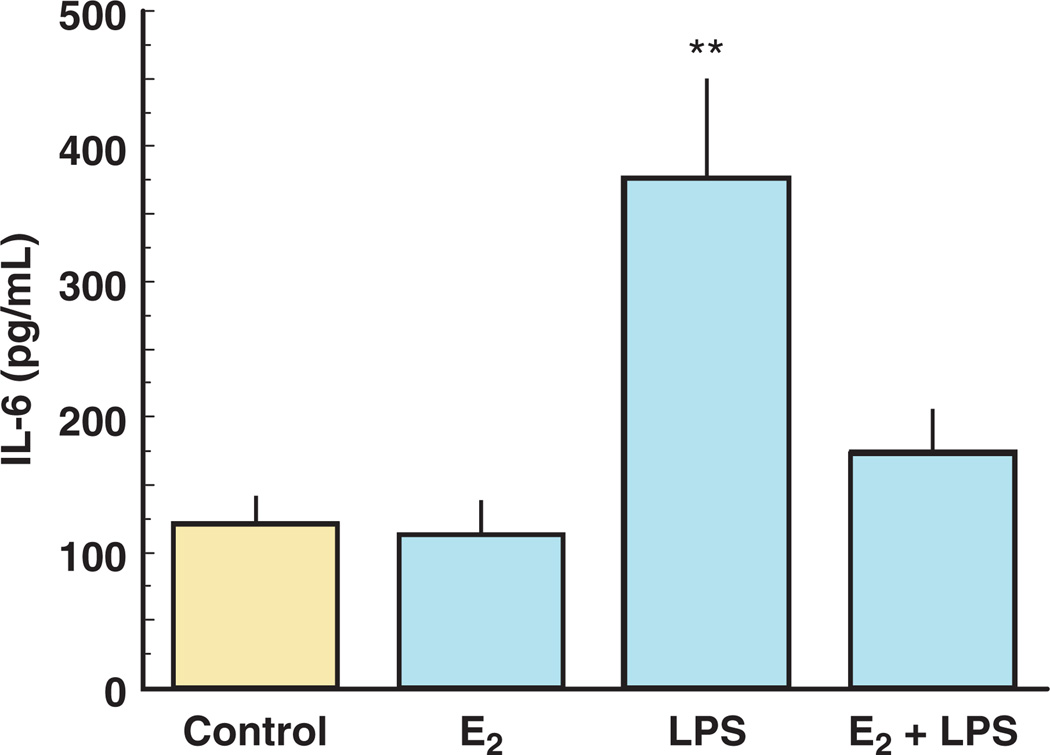
Estradiol also regulates innate immunity by suppressing the secretion and/or expression of pro-inflammatory mediators by epithelial cells. For example, in human keratinocytes, Kande and Watanabe demonstrated the suppression of MCP-1, RANTES, and IP-10 with estradiol treatment.69–71 Previous data in the rodent from our laboratory has also shown that estradiol inhibits the constitutive and TLR-agonist-induced secretion of TNFα and MIP3α by uterine epithelial cells.72 To determine whether estradiol had an effect on the secretion of pro-inflammatory cytokines by human uterine epithelial cells, polarized ECC-1 cells were initially treated with or without estradiol for 24 hrs and then with or without lipopolysaccharide (LPS) for an additional 24 hrs. As shown in Fig. 5, LPS induced an approximate 4-fold increase in IL-6; this increase was almost completely abrogated by pre-treatment with estradiol. These results are consistent with the anti-inflammatory actions of estradiol, which in the FRT, may reduce HIV infection by inhibiting target cell migration and eliminating the ‘immune-activated’ environment often associated with HIV infection. In similar experiments with polarized primary uterine epithelial cells, we found that estradiol enhanced the secretion and mRNA expression of the antimicrobials SLPI and human beta defensin-2 (HBD2).12 Concomitant with these antimicrobial increases was a significant reduction in the LPS- and poly (I:C)-induced secretion of the proinflammatory cytokines MIF, IL-6, and IL-8, as well as a 70% inhibition of mRNA expression of NFkB. Because SLPI is known to inhibit NFkB expression,73–78 these findings suggest that estradiol inhibition of pro-inflammatory cytokines may be mediated through SLPI regulation of NFkB. Overall, these findings indicate that the production of cytokines, chemokines, and antimicrobials by FRT epithelial cells are differentially regulated by estradiol. Further, it suggests that with estradiol regulation epithelial cells that line the uterine cavity have evolved immunologically to be sensitive to viral and bacterial infections as well as the constraints of procreation.
Fig. 5.
Estradiol inhibits LPS-induced IL-6 secretion by uterine epithelial cells. Monolayer cultures of uterine epithelial cells at high TER were treated with 5 × 10−8 m estradiol for 48 hr in the basolateral compartment of cell inserts with some wells receiving an apical treatment of ultra pure lipopolysaccharide (LPS; 100 mg/mL) after 24 hr. Apical conditioned media were recovered, centrifuged to remove cell debris, and IL-6 was measured by ELISA. LPS stimulated a significant increase in IL-6 secretion, which was mostly abrogated by pre-treatment with estradiol. Four wells per group; **significantly different (P < 0.01) from control, estradiol and estradiol + LPS. Adapted from Ref. [12].
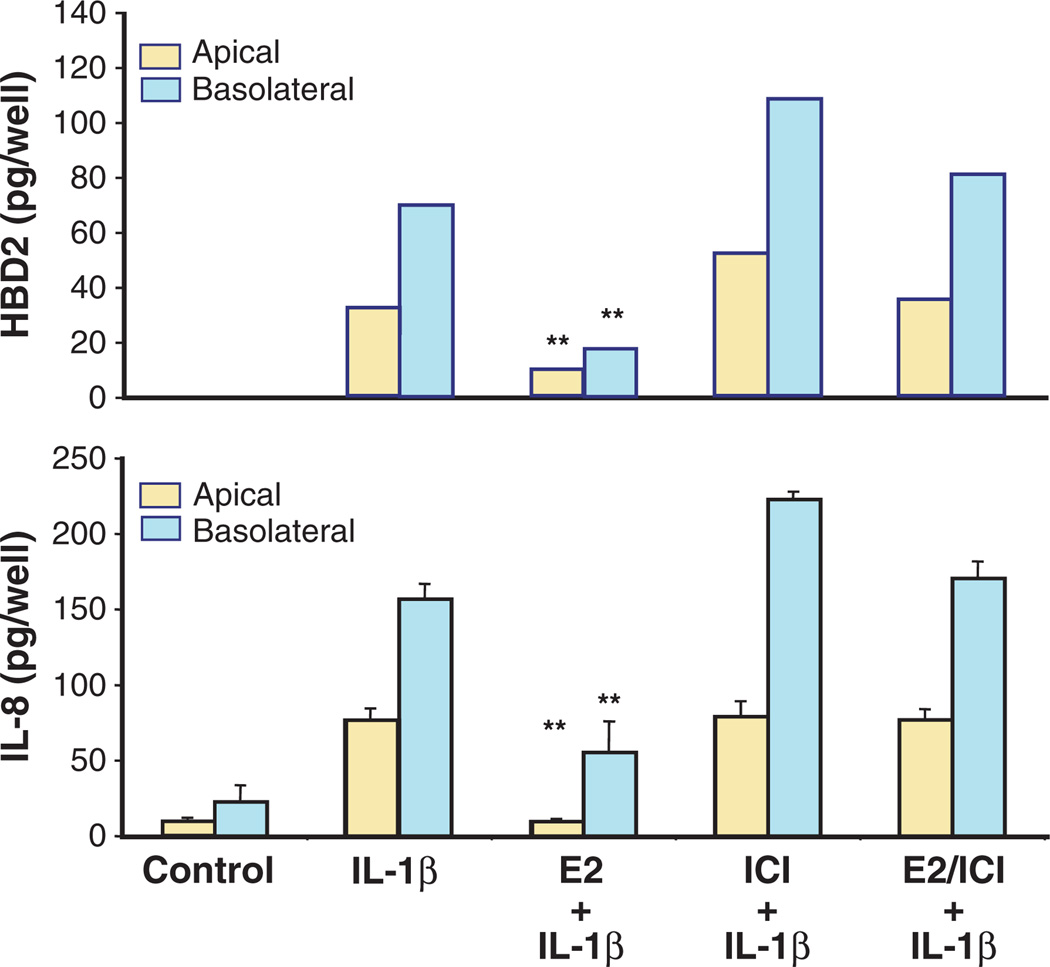
The human uterus is uniquely evolved in its ability to mount immune responses toward pathogens and, at the same time, tolerate an immunologically distinct fetus. Several studies have shown that uterine epithelial cells, when stimulated with pathogens or TLR agonists that mimic pathogenic antigens, can mount a proinflammatory immune response.49,54,79 In vivo, a massive proinflammatory response, although useful against pathogens, might be detrimental to the host and/or fetus. Therefore, immunological regulations are in place that can control and tone down severe inflammatory reactions. One of the key molecules that can regulate inflammatory responses in the upper female reproductive tract is estradiol. Studies from our laboratory and others have demonstrated the down-regulation of immunologically relevant genes by estradiol.12,80 For example, Schaefer et al. have shown that when ECC-1 cells were stimulated by IL-1β, the secretion of both HBD2 as well as IL-8 was enhanced.50 IL-1β is a cytokine known to be involved in the generation of rapid and potent immune responses by upregulating genes involved in the induction of innate immunity (e.g. HBD2 and IL-8). Because prolonged presence of IL-1β is detrimental in the FRT leading to conditions such as preeclampsia and endometriosis,81,82 immunomodulators such as estradiol can dampen this response effectively. As shown in Fig. 6, IL-1β enhanced the secretion of HBD2 and IL-8 by ECC-1 cells. Treatment with estradiol significantly decreased the secretion of both molecules. Treatment with ER antagonist ICI182780 abolished the estradiol-mediated down-modulation of both genes indicating that the observed effect was mediated through the ER receptor. The inhibitory effect of estradiol on IL-1β-mediated inflammatory responses generated by uterine epithelial cells, such as the secretion of HBD2 and IL-8, indicates a link between the endocrine and immune systems and may be crucial for dampening pro-inflammatory responses during the time of ovulation or pregnancy.
Fig. 6.
Estradiol inhibits IL-1β mediated secretion of HBD2 and IL-8 and the inhibition occurs although involvement of the estrogen receptor. ECC-1 cells were preincubated with estradiol, the pure ER antagonist ICI182780, or a combination of steroid and antagonist for 72 hr before stimulation with IL-1β (5 ng/mL). Apical and basolateral conditioned medium were collected following 24-hr stimulation and analysed for HBD2 and IL-8 protein secretion by ELISA. The results are shown as the mean ± SEM. *Significantly different (P < 0.05) from control. **Significantly different (P < 0.01) from control. ***Significantly different (P < 0.001) from control. Gray bars, apical conditioned medium; black bars, basolateral conditioned medium. Adapted from Ref. [50].
Vaginal Cells
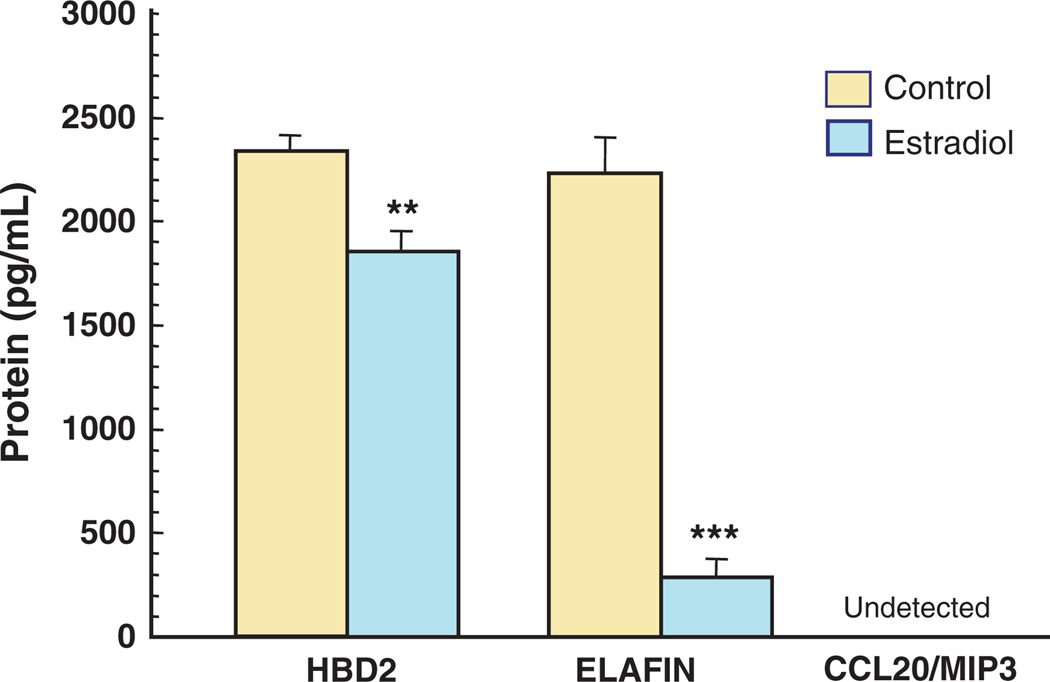
Vaginal surface area ranges from 65 to 108 cm2,83,84 and is lined by multiple layers of squamous nonkeratinized epithelial cells,85 which provide the physical barrier against pathogen entry in the lower reproductive tract. As a mucosal site, the vagina must provide an environment that reconciles its dual and sometimes contradictory roles: reproduction and defense. Additionally, the vagina sustains a commensal bacterial population that supports its major functions. As the sentinels of the innate immune system, vaginal epithelial cells express toll-like receptors (TLRs), 2, 3, 5, and 6.86 TLR stimulation elicits innate immune responses including proinflammatory cytokine production.86 Primary vaginal epithelial cells are sensitive to poly (I:C), TLR3 viral agonist, which induces strong upregulation of IL-1β, IL6, IL8, and MCP-1.86 TNFα is also secreted in response to stimulation by the microbial compounds LPS (TLR4) and peptidoglycan (TLR2).87 Overall, the vagina and its role in innate immune defense remains relatively understudied with the majority of experiments being conducted using cell lines rather than primary isolates. In recent experiments, estradiol significantly decreased the constitutive production of antimicrobials HBD2 and elafin in vitro over 48 hr by freshly isolated squamous vaginal epithelial cells (Fig. 7). In contrast, in vitro results from primary uterine epithelial cells from the upper reproductive tract demonstrate estradiol-dependent upregulation of HBD2 transcription12 and elafin secretion (Fahey et al., unpublished observations). Using vaginal cell lines,88 primary vaginal cell secretion of MIP3α was undetectable in secretions from control and estradiol-treated cells (Fig. 7). The estradiol-induced suppression of HBD2 and elafin secretion by squamous epithelial cells suggests decreased protection by vaginal epithelial cells in vivo during the proliferative and secretory phase of the menstrual cycle when estradiol levels are relatively high.5 These changes in protection could be secondary to physiological changes permitting the survival of sperm deposited in the tract. The opposing responses to estradiol stimulation between the upper and lower tracts may be because of mechanistic changes arising from their differing embryonic origins. A lack of MIP3α production by vaginal epithelial cells could indicate an important role for leukocytes89 and upper reproductive tract epithelial cells in its production, as its presence is detectable in vaginal fluids.
Fig. 7.
Estradiol decreases the production of HBD2 and elafin in primary vaginal squamous epithelial cells. Vaginal secretions and cells were recovered from volunteers using an Instead™ Menstrual Cup (Instead, La Jolla, CA) following insertion for 1 hr. Cells were cultured overnight in a 96-well plate before treatment with estradiol (5 × 10−8 m) for 48 hrs. Supernatants were recovered, centrifuged, and analysed for HBD2, elafin, and MIP3α production by ELISA (**P < 0.01; ***P < 0.001).
Overall, as seen in Fig. 8, estradiol exerts both stimulatory and inhibitory effects on cytokine, chemokines, and antimicrobials secreted by uterine and vaginal epithelial cells. As shown in this figure, estradiol increases antimicrobial secretion by uterine epithelial cells while inhibiting their secretion by vaginal squamous cells in culture. In contrast, estradiol dampens uterine and vaginal epithelial cytokine/chemokine responses to TLR agonists and IL-1β. Given the mid-cycle suppression of some of these cytokines, chemokines, and antimicrobial during the menstrual cycle,5,34 our findings suggest that these changes are, in part, because of the direct effects of estradiol on squamous cells rather than a dilution by cervical mucus. Whether comparable changes occur in response to estradiol in secretions from the uterine lumen remain to be determined.
Fig. 8.
Schematic of estradiol regulation of innate immune function by human epithelial cells in the upper and lower female reproductive tract. In the uterus, estradiol enhances the secretion of antimicrobial factors and reduces the secretion of induced pro-inflammatory mediators. In contrast to the uterus, estradiol inhibits both the constitutive and induced secretion of antimicrobials in the vagina.
Hormone Effects on FRT Immune Responses in Mice
Our research with human and mouse cells/tissues strives to elucidate the role of estradiol on innate immune responses, including antimicrobial chemokine production. When comparing the production of antimicrobials in human and mouse FRT, some similarities have been observed. Briefly, estradiol up-regulates HBD2 secretion by primary human uterine epithelial cells54 as well as enhances murine HBD2 mRNA in uterine tissues in vivo (Hickey et al. manuscript in preparation). In contrast to the uterus, estradiol down-regulates HBD2 secretion from human vaginal squamous epithelial cells in vitro (Patel et al. manuscript in preparation) and inhibits HBD2 mRNA expression in mouse vaginal tissue (Hickey et al. manuscript in preparation).
Our mouse studies demonstrate the presence of known antimicrobial molecules, of which α defensins, β defensins and SLPI are constitutively expressed in murine vaginal and uterine tissue at the mRNA level.90 In other studies, we have found that estradiol has an inhibitory effect on the secretion of MIP3α from freshly isolated human and murine uterine epithelial cells in vitro.66,91,92 Further supporting this observation is our finding that MIP3α levels in uterine secretions is highest at the diestrus phase of the mouse estrous cycle when estradiol is low (Hickey et al. in preparation). Recent studies in our laboratory indicate that MIP3α is present in vaginal secretions from both human and mouse. However, isolated primary vaginal cells from humans and mice show no MIP3α protein production either constitutively or with estradiol treatment (unpublished observation). This suggests that squamous epithelial cells in the vagina are not the source of MIP3α. Scapini et al. reported that both human epithelial cells and neutrophils secrete MIP3α.89 In the mouse, vaginal tissue neutrophils migrate into vaginal mucosa following estrous,93 which corresponds with a significant increase in MIP3α in vaginal secretions at diestrus (Hickey et al. manuscript in preparation).
As observed in humans and mice, the production of antimicrobial defensins, protease inhibitors, or chemokines in the vaginal and uterine tissue are both site and molecule specific. Differences in vaginal and uterine tissue responses may result from changes in cell numbers and distribution of immune cells during the estrous cycle.94–96 Not only does estradiol directly mediate antimicrobial production by FRT epithelial cells, but estradiol also indirectly regulates chemokine secretion from uterine epithelial cells by stimulating the production of human growth factors from stromal cells97 (Haddad et al. manuscript in preparation). Overall, these studies indicate that animal models are a crucial component for determining the effects of sex hormones in innate immunity and the susceptibility to infection of the FRT.
Direct and indirect effects of sex hormones on immune cells in the FRT
Fibroblasts
Epithelial cell interactions with underlying stromal fibroblasts are essential in facilitating steroid hormone-induced growth and development in the endometrium. Cooke et al. used FRT tissues from estrogen receptor (ER) knockout mice to demonstrate that underlying estrogen receptor–positive stromal cells regulate the differentiation of adjacent epithelial cells.98 These studies indicated that estradiol acts on stromal cells to release one or more paracrine factors that then modulate estradiol effects on FRT epithelial cell for growth and differentiation. Hepatocyte growth factor (HGF), a pleiotrophic agent initially shown to stimulate proliferation of hepatocytes in vivo,99 is produced primarily by stromal fibroblasts.100,101 HGF has been well characterized in terms of its normal physiological roles of increasing epithelial cell motility and proliferation, wound healing, 102 angiogenesis,103,104 epithelial cell scattering, and embryogenesis105 (see review106). Using primary human uterine epithelial cells, Sugawara et al. found that HGF stimulates proliferation, migration, and morphological changes in human uterine epithelial cells and concluded that HGF may modulate the cyclic regeneration and development of the endometrial lining of the uterus.107
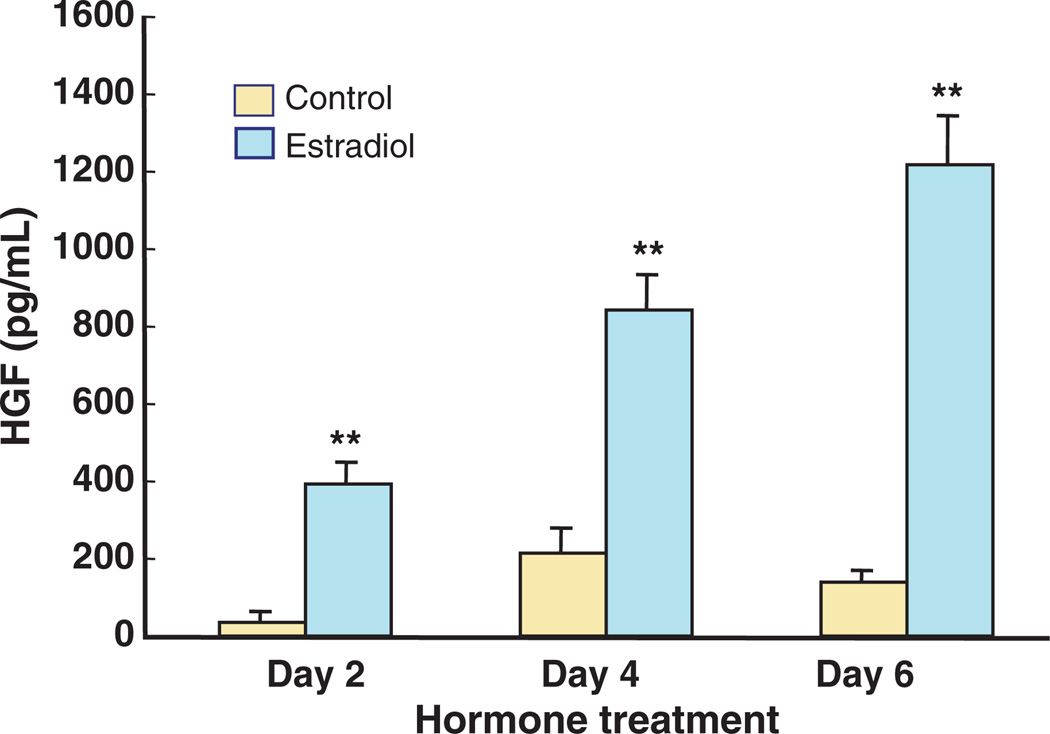
We hypothesized that estradiol might have a regulatory effect on HGF secretion by stromal fibroblasts in the human uterus. Hysterectomy tissues from the uterus were dispersed into epithelial and stromal cell fractions by enzymatic digestion and differential filtering as described previously.36 Stromal fibroblasts were cultured in 24-well culture plates with and without estradiol at a concentration of 10−8 m. Conditioned media from each well were collected at 48-hr intervals and analysed for HGF by ELISA. As shown in Fig. 9, treating uterine stromal fibroblasts with estradiol significantly increased HGF secretion; estradiol-induced secretion of HGF increased with the duration of exposure to estradiol.97 Cells treated with estradiol for 6 days secreted three times more HGF than cells exposed for 2 days. The long-lasting effect of estradiol on HGF secretion by uterine stromal fibroblasts could have implications for cancer or endometriosis where continued HGF secretion might lead to further proliferation of uterine epithelial cells.
Fig. 9.
Estradiol stimulates secretion of HGF by human uterine stromal fibroblasts. Uterine fibroblasts were cultured to confluence in 24-well plates and 1 × 10−8 m estradiol or control media was added on Day 0 and every 2 days thereafter with media change. The conditioned media was recovered every 2 days, centrifuged, and supernatants analysed for HGF by ELISA. HGF secretion increased with estradiol treatment and with ongoing culture. Four wells per group; **, significantly different from control on the day shown (P < 0.01)98.
In related studies using a co-culture system, mouse uterine stromal fibroblasts cultured below uterine polarized epithelial cells in cell inserts produced HGF that significantly increased TER.108 When epithelial cells and/or stromal cells were incubated with anti-HGF or anti-HGF receptor (HGFR) antibody prior to the addition of HGF, the effect of HGF was blocked. Addition of recombinant HGF to the basolateral compartment of polarized epithelial cells increased TER in a dose-dependent manner. These findings indicate that epithelial cells express the HGFR at their basolateral surfaces and that HGFR mediates the effects of HGF on TER.109 In contrast, when cells were incubated with TGFβ, TER was markedly but reversibly suppressed. Based on these findings, we conclude that TGFβ and HGF may play regulatory roles in modulating epithelial cell tight junctions. Studies are underway to determine the relative contributions of hormone balance (Fig. 2 and Table I), pathogen exposure (Fig. 3), cytokine, and HGF secretion (Fig. 9) to innate and adaptive immune protection against potential pathogens in the FRT.
Monocytes
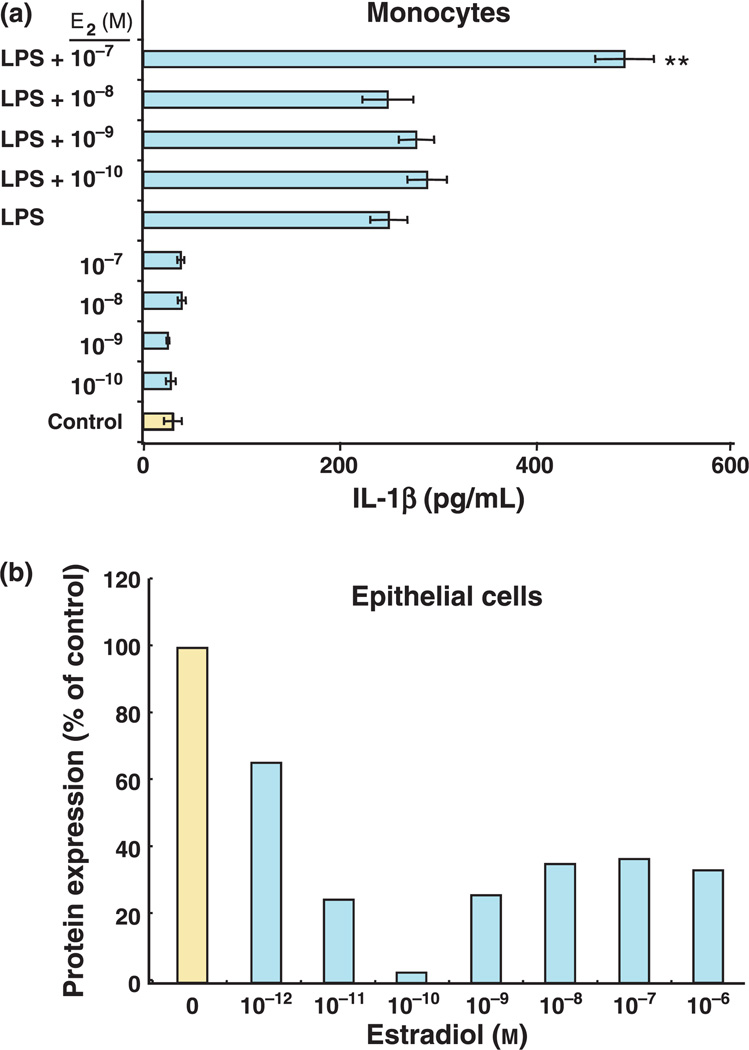
Monocytes circulate through the periphery, mediating immune recognition and pathogen clearance by phagocytosis and indirect stimulation of the immune system through production of key cytokines and chemokines. As shown in Fig. 10 (top), the TLR4 agonist LPS, the key antigenic component of many pathogenic bacteria, stimulates peripheral blood monocyte secretion of IL-1β, thereby causing a proinflammatory response. In the presence of estradiol, this response is further enhanced in a dose-dependent manner (Fig. 10, bottom)110 and may be detrimental to the host especially in the FRT.81,82 Our studies found that increasing estradiol levels in the FRT can down-modulate immune associated genes such as HBD2 and IL-8 (See Fig. 6). We have further shown that this response is mediated through a down-regulation of interleukin-1 receptor type I (IL-1RtI) protein expression (Fig. 10). Overall, estradiol has the ability to enhance pathogen-induced IL-1β expression by monocytes but at the same time decrease IL-1β receptor expression by epithelial cells. These studies demonstrate a close link between the endocrine and immune systems in the FRT that may be crucial for dampening proinflammatory responses during the time of ovulation or pregnancy.
Fig. 10.
Estradiol augments LPS-induced IL-1β levels in monocytes (a). Peripheral blood monocytes were incubated with indicated concentrations of estradiol for 24 hr and then stimulated with 10 ng/mL LPS for an additional 12 hr. Supernatants were collected from these cultures. IL-1β production was quantified by ELISA. (b) Estradiol inhibits interleukin-1 receptor type I (IL-1RtI) protein expression. Whole cell lysates were generated from ECC-1 cells incubated with various concentrations of estradiol for 72 hr. Proteins were resolved by 10% SDS-PAGE and detected with an anti-IL-1RtI antibody. Individual bands were scanned, and the intensity was quantified by computer analysis. The amount of IL-1RtI was normalized to GAPDH levels and plotted as a percentage of the control (representative of two experiments).51,111
Dendritic Cells
Expressed by dendritic cells (DC), DC-SIGN is a calcium-dependent carbohydrate-binding protein.111,112 As an adhesion receptor DC-SIGN interacts with ICAM-2 on endothelial cells to both induce migration of DCs from blood to tissue113 and mediate clustering of DCs with naive T cells through binding of ICAM-3.111 Of equal importance in the FRT, DC-SIGN functions as a pattern recognition receptor that induces specific immune responses upon interaction with a number of pathogens.114 This carbohydrate recognition pattern is the basis of its broad specificity for different pathogens and might also be responsible for its distinct signaling properties. While initial target cells for mucosal transmission of HIV-1 in the reproductive tract have not been clearly determined, studies conducted in nonhuman primate models indicate the potential role of DC as initial targets for mucosal infection and systemic dissemination of HIV-1.115–117 DC can capture HIV-1 using surface expressed C-type lectin receptors (CLRs),118–120 among which DC-SIGN is the best characterized.113 HIV-1 captured by DC can be transferred to other target cells including CD4+ T-lymphocytes via virological synapses, in a process referred to as trans infection.20,121 Understanding the mechanisms by which DC-SIGN and other HIV-1 receptors are regulated on mucosal target cells remains critical to designing strategies to prevent sexual transmission.
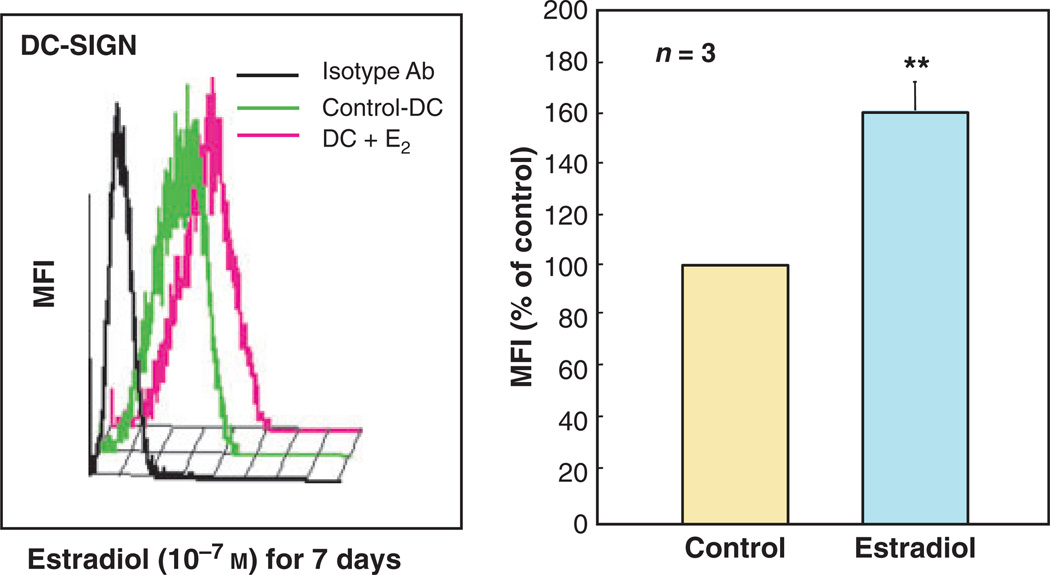
In previous studies aimed at elucidating the role of cell–cell interactions in the endometrium on the expression of HIV-1 receptors, we determined the effects of soluble factors produced by uterine epithelial cells on expression of DC-SIGN by DC (Ochiel et al., submitted). These studies indicated that in addition to the direct effects of estradiol conditioned medium (CM) from polarized primary uterine epithelial cells and ECC-1 cells decreased the expression of DC-SIGN on DC. As a part of these studies, we explored the effects of estradiol on the expression of DC-SIGN by DC. In these experiments, immature DC were differentiated from human monocytes in the presence of IL-4 and GM-CSF as previously described.122 To assess the hormonal effects on DC-SIGN expression, DC were differentiated in the presence or absence of physiological concentrations of estradiol (10−7 m) for 7 days prior to FACS analyses. In contrast to the results obtained with CM from uterine epithelial cells, estradiol significantly enhanced DC-SIGN expression on DC (Fig. 11).
Fig. 11.
Estradiol increases DC-SIGN expression on dendritic cells. Immature DC were derived from human monocytes with IL-4 and GM-CSF for 7 days in the presence or absence of physiological concentrations of estradiol (10−7 m). (a) Overlay histograms for DC-SIGN expression. The histograms depict DC-SIGN expression by control DC (green line) and DC generated with estradiol (red line). The black line represents staining with matched isotype control antibody. (b) Averaged mean fluorescence intensity (MFI) values for DC-SIGN expression (n = 3 donors) are shown as % MFI relative to control DC. *Indicates P < 0.05 compared to control DC.
Our results indicating a stimulatory effect of estradiol on DC-SIGN expression suggests that this sex steroid hormone could increase susceptibility of women to HIV-1 acquisition by enhancing DC-mediated virus uptake and dissemination. The impact of endogenous and exogenous sex steroid hormones on the risk of HIV-1 mucosal transmission in women remains to be fully determined. We recently proposed a model for the existence of a window of vulnerability for HIV-1 infection during a woman’s menstrual cycle.5 During this critical period, 7–10 days of the normal menstrual cycle, critical components of innate and adaptive immune responses are suppressed by estradiol and/or progesterone to facilitate reproductive processes. HIV-1 presumably exploits this time frame, during which antiviral factors are suppressed, to establish and propagate infection in the female genital tract mucosal. 5 The reported findings showing that estradiol increases DC-SIGN expression reveals yet another potential dimension by which sex steroid hormone may modulate susceptibility to HIV-1 infection in women. It is likely that in addition to suppressing antiviral effector mechanisms, the profile of sex steroid hormones present during the window of vulnerability in women may increase HIV-1 acquisition through enhanced expression of HIV-1 capture receptors by DC. More studies are needed to directly test the effects of sex steroid hormones on infection of DC with HIV-1.
NK Cells
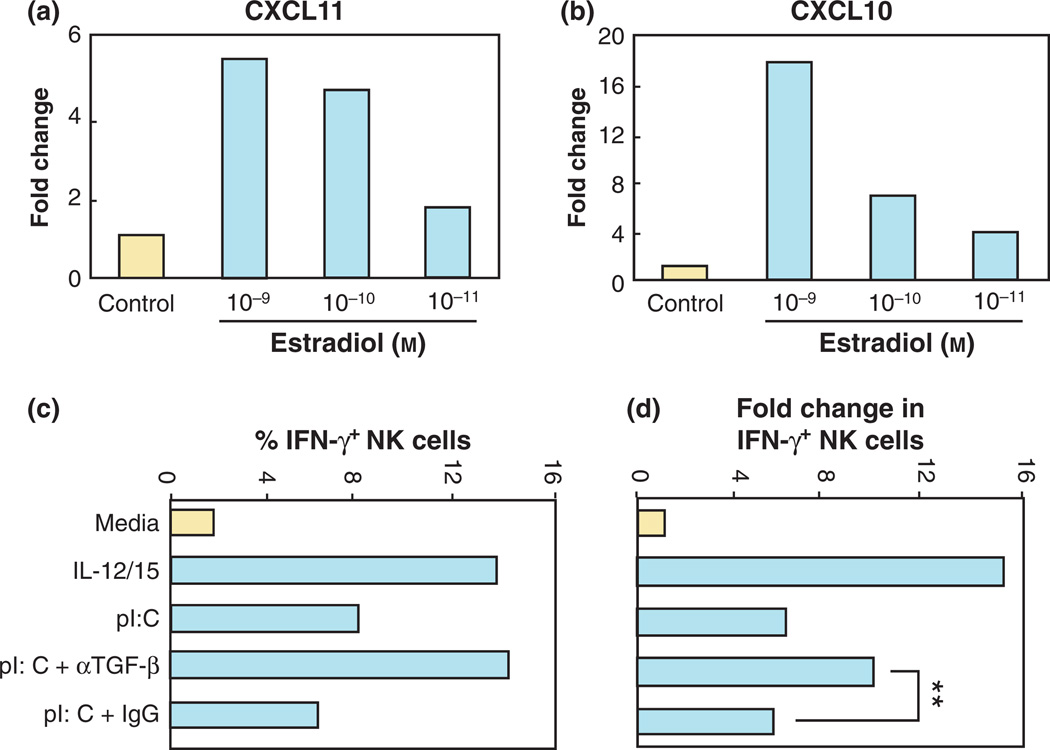
Natural killer cells are important in innate immunity, not only for their ability to kill certain tumor cells and viral-infected cells without prior immunization or MHC restriction, but also for their secretion of immunoregulatory cytokines that contribute to early host responses against viruses, bacteria, and fungi.123 NK cells account for a large percentage of leukocytes in the human endometrium and, because their numbers increase as the menstrual cycle progresses,16–18,124 Sentman et al. hypothesized that recruitment and/or expansion of uterine NK cells are regulated by sex hormones.125 As shown in Fig. 12a,b, they tested the effect of various doses of estradiol on human endometrial slices and found that 10−9 m estradiol significantly enhanced mRNA expression of the chemokines CXCL10 and CXCL11. The expression of five other chemokines was not affected (not shown). Because the estrogen receptor inhibitor ICI182780 blocked the estradiol-induced increase in these two chemokines, the effect was dependent upon estradiol receptor activity (not shown). Progesterone at 10−8 m also increased the mRNA expression of CXCL10 (8.6-fold) and CXCL11 (12.2-fold) in the endometrial organ culture system. 125 Sentman et al. have suggested that the sex hormones do not act directly on NK cells, but rather act on stromal and epithelial cells to produce chemokines for NK cell recruitment.125 Because estradiol and progesterone stimulate production of these cytokines in the endometrium, it is likely that these sex hormones contribute to the migration of NK cells needed for immune surveillance, pathogen response, pregnancy, and normal menstrual cycle function.
Fig. 12.
Effect of estradiol and TGFβ on NK cell immune function. To examine the effect of estradiol on chemokine expression, endometrial tissue sections were incubated with different concentrations of estradiol as indicated for 48 hr, then snap frozen and stored at −80°C. Chemokines CXCL11 (a) and CXCL10 (b) were measured following the isolation of total RNA using TRIzol. Quantitative real-time PCR was used to determine the relative fold expression of each gene compared with medium only. In other studies, endogenous TGFβ suppression of poly (I:C)-induced interferon-γ (IFN-γ) production by uterine NK cells was measured (c and d). Uterine cells were isolated from a hysterectomy patient and cultured with media, IL-12 and IL-15, or poly (I:C) as indicated for 18 hr in the presence of blocking anti-TGFβ monoclonal antibodies (αTGFβ), control IgG (IgG), or media only (media). Uterine NK cells were then analysed for intracellular IFN-γ production by flow cytometry (c). IL-12 and IL-15 in combination and poly (I:C) increase the percent of IFN-γ+ producing NK cells and antibody to TGFβ enhances the number of uterine NK cells induced by poly (I:C) that produce IFN-γ. (d) The data expressed in terms of fold increase in IFN-γ+ NK cells. Adapted from Ref. [125] (bottom) and adapted from Ref. [130].
One innate mechanism that regulates NK activity is TGFβ, which is regulated by sex hormones. TGFβ is typically produced in a pro-form that requires activation; 126 the precise mechanisms that lead to activation of TGFβ in tissues are not well understood. It has previously been shown that uterine NK cell responses to monokines and blood cell NK cell responses to TLR agonists were inhibited by endogenous TGFβ.127,128 Uterine NK cells express TLR and can respond to TLR agonists by producing cytokines. 129 Eriksson et al. explored how endogenous TGFβ modulates the production of interferon-γ (IFN-γ) by human uterine NK cells.130 As shown in Fig. 12c,d, IL-12 and IL-15 in combination and poly (I:C) increase the percent of IFN-γ producing NK cells in the uterus, and antibody to TGFβ enhances the number and fold change of uterine NK cells induced by poly (I:C) that produce IFN-γ. Thus, endogenous TGFβ suppresses poly (I:C)-induced IFN-γ production by uterine NK cells. Because one way that estradiol regulates FRT immunity is by modulating the production and/or activation of TGFβ, it is likely that estradiol regulates NK activity via endogenous TGFβ.
Neutrophils
At mucosal surfaces, neutrophils are responsible for rapid elimination of potential pathogens through phagocytosis of microbes, release of antimicrobial compounds, and production of toxic oxygen and nitrogen species.131 In studies to determine whether ovarian hormones modulate neutrophil phenotype, Smith et al. examined blood neutrophils from women at days 7, 14, 21, and 28 of the menstrual cycle for expression of surface receptors, granule proteins, and intracellular cytokines.19,132 Blood neutrophil phenotype varied during the menstrual cycle with decreased expression of CD89 (IgA Fc receptor) and TNFα during the periovulatory period. In other studies, cytokines were analysed for their ability to enhance the innate immune potential of neutrophils by altering receptor expression and cell function.133 These studies indicated that GM-CSF, known to be produced by FRT epithelial cells, acted synergistically with the chemoattractant IL-8 to promote neutrophil chemotaxis. Use of antibody neutralization and conditioned medium from primary confluent cultures of epithelial cells led to the conclusion that FRT epithelial cells are a potent source of neutrophil chemoattractant activity.
Sex hormone regulation of antimicrobial activity
We have previously shown that polarized human uterine epithelial cells secrete antimicrobial molecules that kill or inhibit pathogenic bacteria, fungi, and viruses.54,67,134 Apical secretion of these molecules is probably one reason why the uterine lumen has a relatively low number of microorganisms compared to the lower FRT despite flow of fluids throughout the entire tract. Secretion of antimicrobials is both constitutive and induced by TLR agonists and microorganisms and is affected by many factors such as chemokines (some chemokines are themselves antimicrobials), cytokines, and steroid hormones.
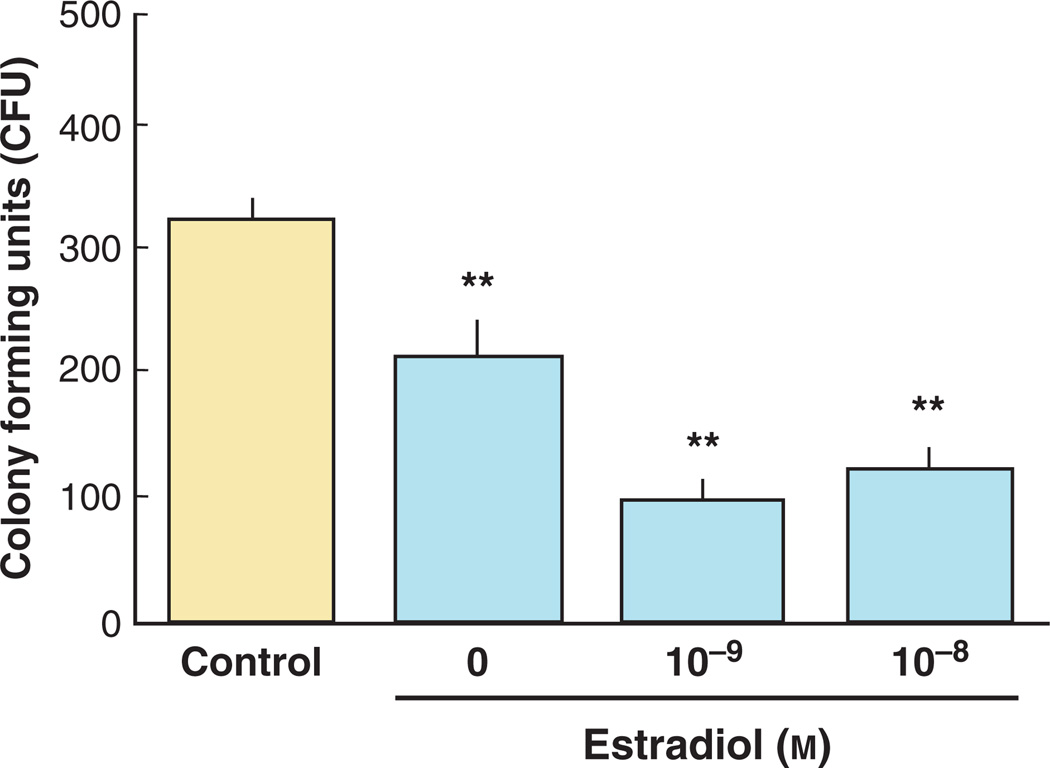
To demonstrate the effect of estradiol treatment on antimicrobial activity, secretions from polarized epithelial cells were tested directly against bacteria (Fig. 13). Human uterine epithelial cells were treated with or without two concentrations of estradiol for 48 hr. An aliquot of apical conditioned media was then incubated with Staphylococcus aureus for 1 hr, and the bacteria were cultured overnight. Control colony-forming units (CFU) refers to the colonies grown in media in the absence of uterine epithelial cells. The inhibition seen with no estradiol represents bacterial growth in the presence of antimicrobials constitutively produced by epithelial cells. The lower CFU seen with secretions obtained from estradiol treatment indicates that estradiol is inducing the secretion of antimicrobial(s) in addition to that seen constitutively. These data reinforce the premise that estradiol can have a significant effect on antimicrobial secretion by human uterine polarized epithelial cells.
Fig. 13.
Estradiol induces antibacterial activity in ECC-1 epithelial cells. Human uterine epithelial cells were treated with or without two concentrations of estradiol for 48 hr. The apical conditioned media was recovered after 2 days, centrifuged and an aliquot of supernatants was then incubated with Staphylococcus aureus for 1 hr, and the bacteria were cultured overnight. Control colony-forming units (CFU) refers to the colonies grown in media in the absence of uterine epithelial cells. The inhibition seen with no estradiol represents the bacteria that grew out in the presence of antimicrobials constitutively produced by the cells. Estradiol by itself had no effect on bacterial CFU (not shown). Four wells per group; **significantly different (P < 0.01) from control. Adapted from Ref. [12].
In other studies, we have shown that constitutive conditioned media from human uterine epithelial cells, as well as human cervical and ectocervical epithelial cells inhibit CFU of Candida albicans and Neisseria gonorrhoeae, as well as the infection of TZM-bl cells by HIV-1 (Wira et al. manuscript in preparation). Interestingly, the commensal Lactobacillus crispatus was not affected by any of these conditioned media. This suggests that FRT epithelial cell antimicrobial secretions are selective for potentially pathogenic microorganisms.
Conclusions
In conclusion, analysis of the FRT indicates that the innate and adaptive immune systems are present and functional throughout the menstrual cycle. Not widely appreciated is a growing body of data indicating that the immune system in the fallopian tubes, uterus, cervix, and vagina function independently from each other to changes in hormone levels during the menstrual cycle. The net result is coordinated immune protection that compliments the reproductive demands of each organ. As the first line of this protection, epithelial cells, macrophages, dendritic cells, NK cells, and neutrophils function to meet the challenges of STI, while at the same time supporting an immunologically distinct fetal placental unit. The studies presented demonstrate overwhelmingly that estradiol secreted by the ovary during the menstrual cycle act both directly and indirectly on epithelial cells and other immune cells in the FRT to regulate a spectrum of immune functions specific to each site in the FRT. At the center of immune protection, epithelial cells that line the FRT provide a level of protection not previously recognized. Acting as a physical barrier, these cells are sentinels that function as an integral part of the innate and adaptive immune systems by recognizing and protecting against bacterial and viral challenges as well as signaling the recruitment and activation of underlying immune protection when pathogenic challenge exceeds their protective capacity. Along with other immune cells, epithelial cell responses are uniquely programmed to respond to hormone changes so that immune coverage occurs in a way that confers continuous protection. This review indicates that epithelial cell and immune cell functions are complicated by the recognition that sex hormones alter epithelial permeability, microbicide activity, and cytokine/chemokine secretion in the FRT to ensure maternal and fetal protection. As discussed previously, 5 aspects of the innate, humoral, and cell-mediated immune systems are suppressed by sex hormones to optimize conditions for procreation. Suppression occurs in the upper (fallopian tubes, uterus, endocervix) and lower (ectocervix and vagina) FRT, and coincides with the recruitment of potentially infectable cells and upregulation of coreceptors involved in pathogen uptake. The studies presented suggest that by understanding the ways in which sex hormones regulate epithelial cell and immune cell function in the FRT, new avenues may be identified both to protect against potential pathogens and to enhance the quality of women’s reproductive health.
Acknowledgments
This work was funded by a National Institutes of Health Grants AI-51877, AI-13541, AI-071761 (CRW), and by a grant from the Bill & Melinda Gates Foundation through the Grand Challenges Exploration Initiative (JVF/CRW).
References
- 1. http://www.who.int/mediacentre/factsheets/fs110/en/index.html.
- 2.UNAIDS 2007. AIDS epidemic update. Geneva, Switzerland: JUNPoHAUaWHOW; 2007. [Google Scholar]
- 3.Ogra P, Yamanaka T, Losonsky GA. Local immunologic defenses in the genital tract. In: Fleicher N, editor. Local Immunologic Defenses in the Genital Tract. New York: Alan R. Liss, Inc.; 1981. pp. 381–394. [PubMed] [Google Scholar]
- 4.Wira CR, Fahey JV, White HD, Yeaman GR, Given AL, Howell AL. The mucosal immune system in the human female reproductive tract: influence of stage of the menstrual cycle and menopause on mucosal immunity in the uterus. In: Glasser S, Aplin J, Guidice L, Tabibzadeh S, editors. The Mucosal Immune System in the Human Female Reproductive Tract: influence of Stage of the Menstrual Cycle and Menopause on Mucosal Immunity in the Uterus. New York: Taylor and Francis; 2002. pp. 371–404. [Google Scholar]
- 5.Wira CR, Fahey JV. A new strategy to understand how HIV infects women: identification of a window of vulnerability during the menstrual cycle. AIDS. 2008;22:1909–1917. doi: 10.1097/QAD.0b013e3283060ea4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wira CR, Fahey JV, Sentman CL, Pioli PA, Shen L. Innate and adaptive immunity in female genital tract: cellular responses and interactions. Immunol Rev. 2005;206:306–335. doi: 10.1111/j.0105-2896.2005.00287.x. [DOI] [PubMed] [Google Scholar]
- 7.Wira CR, Grant-Tschudy KS, Crane-Godreau M. Epithelial cells in the female reproductive tract: a central role as sentinels of immune protection. AJRI. 2005;53:1–12. doi: 10.1111/j.1600-0897.2004.00248.x. [DOI] [PubMed] [Google Scholar]
- 8.Fahey JV, Schaefer TM, Channon JY, Wira CR. Secretion of cytokines and chemokines by polarized human epithelial cells from the female reproductive tract. Human Reprod. 2005;20:1439–1446. doi: 10.1093/humrep/deh806. [DOI] [PubMed] [Google Scholar]
- 9.Fahey JV, Schaefer TM, Wira CR. Sex hormone modulation of human uterine epithelial cell immune responses. Intgr Comp Biol. 2006;46:1082–1087. doi: 10.1093/icb/icl036. [DOI] [PubMed] [Google Scholar]
- 10.Kaushic C, Wira CR. IgA and reproductive tract immunity. In: Kaetzel C, editor. IgA and Reproductive Tract Immunity. New York: Kluwer Academic/Plenum Publisher; 2008. pp. 291–320. [Google Scholar]
- 11.Wira CR, Fahey JV. The innate immune system: gatekeeper to the female reproductive tract. Immunology. 2004;111:13–15. doi: 10.1111/j.1365-2567.2003.01796.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fahey JV, Wright JA, Shen L, Smith JM, Ghosh M, Rossoll RM, Wira CR. Estradiol selectively regulates innate immune function by polarized human uterine epithelial cells in culture. Mucosal Immunol. 2008;1:317–325. doi: 10.1038/mi.2008.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dahlenbach-Hellweg G. Histopathology of the Endometrium. 2nd edn. New York: Springer-Verlag; 1975. [Google Scholar]
- 14.Morris H, Edwards J, Tiltman A, Emms M. Endometrial lymphoid tissue: an immunohistological study. J Clin Pathol. 1985;38:644–652. doi: 10.1136/jcp.38.6.644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bulmer JN, Earl U. The expression of class II MHC gene products by fallopian tube epithelium in pregnancy and throughout the menstrual cycle. Immunology. 1987;61:207–213. [PMC free article] [PubMed] [Google Scholar]
- 16.Hunt JS. Immunologically relevant cells in the uterus. Biol Reprod. 1994;50:461–466. doi: 10.1095/biolreprod50.3.461. [DOI] [PubMed] [Google Scholar]
- 17.Givan AL, White HD, Stern JE, Colby E, Gosselin EJ, Guyre PM, Wira CR. Flow cytometric analysis of leukocytes in the human female reproductive tract: comparison of fallopian tube, uterus, cervix, and vagina. Am J Reprod Immunol. 1997;38:350–359. doi: 10.1111/j.1600-0897.1997.tb00311.x. [DOI] [PubMed] [Google Scholar]
- 18.Mselle TF, Meadows SK, Eriksson M, Smith JM, Shen L, Wira CR, Sentman CL. Unique characteristics of NK cells throughout the human female reproductive tract. Clin Immunol. 2007;124:69–76. doi: 10.1016/j.clim.2007.04.008. [DOI] [PubMed] [Google Scholar]
- 19.Smith JM, Wira CR, Fanger MW, Shen L. Human fallopian tube neutrophils: a distinct phenotype from blood neutrophils. AJRI. 2006;56:218–229. doi: 10.1111/j.1600-0897.2006.00410.x. [DOI] [PubMed] [Google Scholar]
- 20.Piguet V, Steinman RM. The interaction of HIV with dendritic cells: outcomes and pathways. Trends Immunol. 2007;28:503–510. doi: 10.1016/j.it.2007.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yeaman GR, Fazleabas AT, Wira CR. Endometrial lymphoid aggregates. Mucosal Immunol Update. 2004;12:6–8. [Google Scholar]
- 22.Yeaman GR, Collins JE, Fanger MW, Wira CR, Lydyard PL. T cell receptor Vb usage in human uterine endometrial lymphoid aggregates shows no clonal restriction: evidence that uterine lymphoid aggregates arise by cell trafficking. Immunology. 2001;102:434–440. doi: 10.1046/j.1365-2567.2001.01199.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.White HD, Crassi KM, Givan AL, Stern JE, Gonzalez JL, Memoli VA, Green WR, Wira CR. CD3+ CD8+ CTL activity within the human female reproductive tract: influence of stage of the menstrual cycle and menopause. J Immunol. 1997;158:3017–3027. [PubMed] [Google Scholar]
- 24.White HD, Crassi K, Wira CR. Cytolytic functional activities of NK cells and cytotoxic T lymphocytes (CTL) are coordinately regulated in the human female reproductive tract. In: Husband AJ, Beagley KW, Clancey RL, Collins AM, Cripps AW, Emery DL, editors. Cytolytic Functional Activities of NK cells and Cytotoxic T Lymphocytes (CTL) are Coordinately Regulated in the Human Female Reproductive Tract. Sydney, Australia: The University of Sydney; 1997. pp. 385–391. [Google Scholar]
- 25.Dominguez F, Galan A, Martin JJL, Remohi J, Pellicer A, Simón C. Hormonal and embryonic regulation of chemokine receptors CXCR1, CXCR4, CCR5 and CCR2B in the human endometrium and the human blastocyst. Mol Human Reprod. 2003;9:189–198. doi: 10.1093/molehr/gag024. [DOI] [PubMed] [Google Scholar]
- 26.Jones RL, Hannan NJ, Kaitúu TJ, Zhang J, Salamonsen LA. Identification of chemokines important for leukocyte recruitment to the human endometrium at the times of embryo implantation and menstruation. J Clin Endocrinol Metab. 2004;89:6155–6167. doi: 10.1210/jc.2004-0507. [DOI] [PubMed] [Google Scholar]
- 27.Yeaman GR, Howell AL, Weldon S, Damien DJ, Collins JE, O’Connell DM, Asin SN, Wira CR, Fanger MW. HIV receptor and coreceptor expression on human uterine epithelial cells during the menstrual cycle. Immunology. 2003;109:137–146. doi: 10.1046/j.1365-2567.2003.01623.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Yeaman GR, Asin S, Weldon S, Demian DJ, Collins JE, Wira CR, Fanger MW, Howell AL. Chemokine Receptor Expression in the Human Cervix: implications for infection by the human Immunodeficiency Virus-type I (HIV-1) Immunology. 2004;113:524–533. doi: 10.1111/j.1365-2567.2004.01990.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sullivan DA, Richardson GS, MacLaughlin DT, Wira CR. Variations in the levels of secretory component in human uterine fluid during the menstrual cycle. J Steroid Biochem. 1984;20:509–513. doi: 10.1016/0022-4731(84)90263-2. [DOI] [PubMed] [Google Scholar]
- 30.Tauber PF, Wettich W, Nohlen M, Zaneveld LJ. Diffusible proteins of the mucosa of the human cervix, uterus, and fallopian tubes: distribution and variations during the menstrual cycle. Am J Obstet Gynecol. 1985;15:1115–1125. doi: 10.1016/0002-9378(85)90394-1. [DOI] [PubMed] [Google Scholar]
- 31.Schumacher GFB. Soluble proteins in cervical mucus. In: Blandau RJ, Moghissi K, editors. Soluble Proteins in Cervical Mucus. Chicago: The University of Chicago Press; 1973. pp. 201–233. [Google Scholar]
- 32.Kutteh KW, Moldoveanu Z, Mestecky J. Mucosal immunity in the female reproductive tract: correlation of immunoglobulins, cytokines, and reproductive hormones in human cervical mucus around the time of ovulation. AIDS Res Hum Retroviruses. 1998;14:S51–S55. [PubMed] [Google Scholar]
- 33.Nardelli-Haefliger DJ, Wirthner D, Schiller J, Lowy D, Hildesheim A, Ponci F, Grandi P. Specific Antibody Levels at the Cervix During the Menstrual Cycle of Women Vaccinated With Human Papillomavirus 16 Virus–Like Particles. J Nat Cancer Inst. 2003;95:1128–1137. doi: 10.1093/jnci/djg018. [DOI] [PubMed] [Google Scholar]
- 34.Keller M, Guzman E, Hazrati E, Kasowitz A, Cheshenko N, Wallenstein S, Cole A, Cole A, Profy A, Wira C, Hogarty K, Herold B. PRO 2000 elicits a decline in genital tract immune mediators without compromising intrinsic antimicrobial activity. AIDS. 2007;21:467–476. doi: 10.1097/QAD.0b013e328013d9b5. [DOI] [PubMed] [Google Scholar]
- 35.Quayle AJ, Martin Porter E, Nussbaum AA, Wang YM, Brabec C, Yip K-P, Mok SC. Gene expression, immunolocalization and secretion of human defensin-5 in human female reproductive tract. Am J Pathol. 1998;152:1247–1258. [PMC free article] [PubMed] [Google Scholar]
- 36.Fahey JV, Kaushic C, Wira CR. Human uterine epithelial cells: influence of culture conditions and stromal cells on epithelial cell transepithelial cell resistance. In: Gupta SK, editor. Human Uterine Epithelial Cells: Influence of Culture Conditions and Stromal Cells on Epithelial Cell Transepithelial Cell Resistance. New Delhi: Narosa Publishing House; 1999. pp. 366–378. [Google Scholar]
- 37.Wira CR, Stern J. Endocrine regulation of the mucosal immune system in the female reproductive tract: control of IgA, IgG, and secretory component during the reproductive cycle, at implantation and throughout pregnancy. In: Pasqualini JR, Scholler R, editors. Endocrine Regulation of the Mucosal Immune System in the Female Reproductive Tract: Control of IgA, IgG, and Secretory Component During the Reproductive Cycle, at Implantation and Throughout Pregnancy. New York: Marcel Dekker, Inc.; 1992. pp. 343–368. [Google Scholar]
- 38.Grant KS, Wira CR. Effect of mouse uterine stromal cells on epithelial cell transepithelial resistance (TER) and TNFalpha and TGFbeta release in culture. Biol Reprod. 2003;69:1091–1098. doi: 10.1095/biolreprod.103.015495. [DOI] [PubMed] [Google Scholar]
- 39.Godfrey RWA. Human airway epithelial tight junctions. Microscopy Res Tech. 1997;38:488–499. doi: 10.1002/(SICI)1097-0029(19970901)38:5<488::AID-JEMT5>3.0.CO;2-E. [DOI] [PubMed] [Google Scholar]
- 40.Matter K, Aijaz S, Tsapara A, Balda MS. Mammalian tight junctions in the regulation of epithelial differentiation and proliferation. Curr Opin Cell Biol. 2005;17:453–458. doi: 10.1016/j.ceb.2005.08.003. [DOI] [PubMed] [Google Scholar]
- 41.Mo B, Vendrov AE, Palomino WA, DuPont BR, Apparao KB, Lessey BA. ECC-1 cells: a well-differentiated steroid-responsive endometrial cell line with characteristics of luminal epithelium. Biol Reprod. 2006;75:387–394. doi: 10.1095/biolreprod.106.051870. [DOI] [PubMed] [Google Scholar]
- 42.Planchon S, Fiocchi C, Takafuji V, Roche JK. Transforming growth factor-beta1 preserves epithelial barrier function: identification of receptors, biochemical intermediates and cytokine antagonists. J Cell Physiol. 1999;181:55–66. doi: 10.1002/(SICI)1097-4652(199910)181:1<55::AID-JCP6>3.0.CO;2-M. [DOI] [PubMed] [Google Scholar]
- 43.Shin K, Fogg VC, Margolis B. Tight junctions and cell polarity. Annu Rev Cell Dev Biol. 2006;22:207–235. doi: 10.1146/annurev.cellbio.22.010305.104219. [DOI] [PubMed] [Google Scholar]
- 44.Simons K, Wandinger-Ness A. Polarized sorting in epithelia. Cell. 1990;62:207–210. doi: 10.1016/0092-8674(90)90357-k. [DOI] [PubMed] [Google Scholar]
- 45.Tsukita S, Furuse M, Itoh M. Multifunctional strands in tight junctions. Nat Rev Mol Cell Biol. 2001;2:285–293. doi: 10.1038/35067088. [DOI] [PubMed] [Google Scholar]
- 46.Grant-Tschudy KS, Wira CR. Effect of estradiol on mouse uterine epithelial cell transepithelial resistance (TER) Am J Reprod Immunol. 2004;52:252–262. doi: 10.1111/j.1600-0897.2004.00218.x. [DOI] [PubMed] [Google Scholar]
- 47.Phillips ML, Schultz BD. Steroids modulate transepithelial resistance and Na+ absortion across cultured porcine vas deferens epithelia. Biol Reprod. 2002;66:1016–1023. doi: 10.1095/biolreprod66.4.1016. [DOI] [PubMed] [Google Scholar]
- 48.Groten T, Pierce AA, Huen AC, Schnaper HW. 17 beta-estradiol transiently disrupts adherens junctions in endothelial cells. FASEB J. 2005;19:1368–1370. doi: 10.1096/fj.04-2558fje. [DOI] [PubMed] [Google Scholar]
- 49.Schaefer TM, Desouza K, Fahey JV, Beagley KW, Wira CR. Toll-like receptor (TLR) expression and TLR-mediated cytokine/chemokine production by human uterine epithelial cells. Immunology. 2004;112:428–436. doi: 10.1111/j.1365-2567.2004.01898.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Schaefer TM, Wright JA, Pioli PA, Wira CR. IL-1beta-mediated proinflammatory responses are inhibited by estradiol via down-regulation of IL-1 receptor type 1 in uterine epithelial cells. J Immunol. 2005;175:6509–6516. doi: 10.4049/jimmunol.175.10.6509. [DOI] [PubMed] [Google Scholar]
- 51.Ochiel DO, Fahey JV, Ghosh M, Haddad SN, Wira CR. Innate immunity in the female reproductive tract: role of sex hormones in regulating uterine epithelial cell protection against pathogens. Curr Womens Health Rev. 2008;4:102–117. doi: 10.2174/157340408784246395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.King AE, Critchley HO, Kelly RW. Presence of secretory leukocyte protease inhibitor in human endometrium and first trimester decidua suggests an antibacterial protective role. Mol Hum Reprod. 2000;6:191–196. doi: 10.1093/molehr/6.2.191. [DOI] [PubMed] [Google Scholar]
- 53.King AE, Fleming DC, Critchley HO, Kelly RW. Differential expression of the natural antimicrobials, beta-defensins 3 and 4, in human endometrium. J Reprod Immunol. 2003;59:1–16. doi: 10.1016/s0165-0378(02)00083-9. [DOI] [PubMed] [Google Scholar]
- 54.Schaefer TM, Fahey JV, Wright JA, Wira CR. Innate immunity in the human female reproductive tract: antiviral response of uterine epithelial cells to the TLR3 agonist poly(I:C) J Immunol. 2005;174:992–1002. doi: 10.4049/jimmunol.174.2.992. [DOI] [PubMed] [Google Scholar]
- 55.Valore EV, Park CH, Quayle AJ, Wiles KR, McCray PB, Jr, Ganz T. Human b-defensin-1, an antimicrobial peptide of urogenital tissues. J Clin Invest. 1998;101:1633–1642. doi: 10.1172/JCI1861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Duits LA, Nibbering PH, van Strijen E, Vos JB, Mannesse-Lazeroms SP, van Sterkenburg MA, Hiemstra PS. Rhinovirus increases human betadefensin-2 and -3 mRNA expression in cultured bronchial epithelial cells. FEMS Immunol Med Microbiol. 2003;38:59–64. doi: 10.1016/S0928-8244(03)00106-8. [DOI] [PubMed] [Google Scholar]
- 57.Ganz T. Defensins: antimicrobial peptides of innate immunity. Nat Rev. 2003;3:710–720. doi: 10.1038/nri1180. [DOI] [PubMed] [Google Scholar]
- 58.Hocini H, Becquart P, Bouhlal H, Adle-Biassette H, Kazatchkine MD, Belec L. Secretory leukocyte protease inhibitor inhibits infection of monocytes and lymphocytes with human immunodeficiency virus type 1 but does not interfere with transcytosis of cell-associated virus across tight epithelial barriers. Clin Diagn Lab Immunol. 2000;7:515–518. doi: 10.1128/cdli.7.3.515-518.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Porter E, Yang H, Yavagal S, Preza GC, Murillo O, Lima H, Greene S, Mahoozi L, Klein-Patel M, Diamond G, Gulati S, Ganz T, Rice PA, Quayle AJ. Distinct defensin profiles in Neisseria gonorrhoeae and Chlamydia trachomatis urethritis reveal novel epithelial cell-neutrophil interactions. Infect Immun. 2005;73:4823–4833. doi: 10.1128/IAI.73.8.4823-4833.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Quinones-Mateu ME, Lederman MM, Feng Z, Chakraborty B, Weber J, Rangel HR, Marotta ML, Mirza M, Jiang B, Kiser P, Medvik K, Sieg SF, Weinberg A. Human epithelial beta-defensins 2 and -3 inhibit HIV-1 replication. Aids. 2003;17:F39–F48. doi: 10.1097/00002030-200311070-00001. [DOI] [PubMed] [Google Scholar]
- 61.Tomee JF, Hiemstra PS, Heinzel-Wieland R, Kauffman HF. Antileukoprotease: an endogenous protein in the innate mucosal defense against fungi. J Infect Dis. 1997;176:740–747. doi: 10.1086/514098. [DOI] [PubMed] [Google Scholar]
- 62.Wahl S, McNeely T, Janoff E, Shugars D, Worley P, Tucker C, Orenstein J. Secretory leukocyte protease inhibitor (SLPI) in mucosal fluids inhibits HIV-1. Oral Dis. 1997;3(Suppl 1):S64–S69. doi: 10.1111/j.1601-0825.1997.tb00377.x. [DOI] [PubMed] [Google Scholar]
- 63.Bleul CC, Farzan M, Choe H, Parolin C, Clark-Lewis I, Sodroski J, Springer TA. The lymphocyte chemoattractant SDF-1 is a ligand for LESTR/fusin and blocks HIV-1 entry. Nature. 1996;382:829–833. doi: 10.1038/382829a0. [DOI] [PubMed] [Google Scholar]
- 64.Cocchi F, DeVico AL, Garzino-Demo A, Arya SK, Gallo RC, Lusso P. Identification of RANTES, MIP-1 alpha, and MIP-1 beta as the major HIV-suppressive factors produced by CD8+ T cells. Science. 1995;270:1811–1815. doi: 10.1126/science.270.5243.1811. [DOI] [PubMed] [Google Scholar]
- 65.Ghosh M, Shen Z, Fahey JV, Cu-Uvin S, Mayer K, Wira CR. Trappin-2/Elafin: a novel innate antihuman immunodeficiency virus-1 molecule of the human female reproductive tract. Immunology. 2009;129:207–219. doi: 10.1111/j.1365-2567.2009.03165.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Ghosh M, Shen Z, Schaefer TM, Fahey JV, Gupta P, Wira CR. CCL20/MIP3 alpha is a novel anti-HIV-1 molecule of the human female reproductive tract. Am J Reprod Immunol. 2009;62:60–71. doi: 10.1111/j.1600-0897.2009.00713.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Fahey JV, Wira CR. Effect of menstrual status on antibacterial activity and secretory leukocyte protease inhibitor production by human uterine epithelial cells in culture. J Infect Dis. 2002;185:1606–1613. doi: 10.1086/340512. [DOI] [PubMed] [Google Scholar]
- 68.Beagley KW, Gockel CM. Regulation of innate and adaptive immunity by the female sex hormones oestradiol and progesterone. EFEMS Immunol Med Microbiol. 2003;38:13–22. doi: 10.1016/S0928-8244(03)00202-5. [DOI] [PubMed] [Google Scholar]
- 69.Kanda N, Watanabe S. A17Beta-estradiol inhibits MCP-1 production in human keratinocytes. J Invest Dermatol. 2003;120:1058–1066. doi: 10.1046/j.1523-1747.2003.12255.x. [DOI] [PubMed] [Google Scholar]
- 70.Kanda N, Watanabe S. 17beta-estradiol inhibits the production of RANTES in human keratinocytes. J Invest Dermatol. 2003;120:420–427. doi: 10.1046/j.1523-1747.2003.12067.x. [DOI] [PubMed] [Google Scholar]
- 71.Kanda N, Watanabe S. 17beta-estradiol inhibits the production of interferon-induced protein of 10 kDa by human keratinocytes. J Invest Dermatol. 2003;120:411–419. doi: 10.1046/j.1523-1747.2003.12066.x. [DOI] [PubMed] [Google Scholar]
- 72.Grant-Tschudy KS, Wira CR. Effect of oestradiol on mouse uterine epithelial cell tumour necrosis factoralpha release is mediated through uterine stromal cells. Immunology. 2005;115:99–107. doi: 10.1111/j.1365-2567.2005.02134.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Greene CM, McElvaney NG, O’Neill SJ, Taggart CC. Secretory leucoprotease inhibitor impairs Toll-like receptor 2- and 4-mediated responses in monocytic cells. Infect Immun. 2004;72:3684–3687. doi: 10.1128/IAI.72.6.3684-3687.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Lentsch AB, Jordan JA, Czermak BJ, Diehl KM, Younkin EM, Sarma V, Ward PA. Inhibition of NF-kappaB activation and augmentation of IkappaBbeta by secretory leukocyte protease inhibitor during lung inflammation. Am J Pathol. 1999;154:239–247. doi: 10.1016/s0002-9440(10)65270-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Lentsch AB, Yoshidome H, Warner RL, Ward PA, Edwards MJ. Secretory leukocyte protease inhibitor in mice regulates local and remote organ inflammatory injury induced by hepatic ischemia/reperfusion. Gastroenterology. 1999;117:953–961. doi: 10.1016/s0016-5085(99)70355-0. [DOI] [PubMed] [Google Scholar]
- 76.Nakamura A, Mori Y, Hagiwara K, Suzuki T, Sakakibara T, Kikuchi T, Igarashi T, Ebina M, Abe T, Miyazaki J, Takai T, Nukiwa T. Increased susceptibility to LPS-induced endotoxin shock in secretory leukoprotease inhibitor (SLPI)-deficient mice. J Exp Med. 2003;197:669–674. doi: 10.1084/jem.20021824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Taggart CC, Cryan SA, Weldon S, Gibbons A, Greene CM, Kelly E, Low TB, O’Neill SJ, McElvaney NG. Secretory leucoprotease inhibitor binds to NFkappaB binding sites in monocytes and inhibits p65 binding. J Exp Med. 2005;202:1659–1668. doi: 10.1084/jem.20050768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ward PA, Lentsch AB. Endogenous regulation of the acute inflammatory response. Mol Cell Biochem. 2002;234:225–228. [PubMed] [Google Scholar]
- 79.Jorgenson RL, Young SL, Lesmeister MJ, Lyddon TD, Misfeldt ML. Human endometrial epithelial cells cyclically express Toll-like receptor 3 (TLR3) and exhibit TLR3-dependent responses to dsRNA. Hum Immunol. 2005;66:469–482. doi: 10.1016/j.humimm.2004.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lesmeister MJ, Jorgenson RL, Young SL, Misfeldt ML. 17Beta-estradiol suppresses TLR3-induced cytokine and chemokine production in endometrial epithelial cells. Reprod Biol Endocrinol. 2005;3:74. doi: 10.1186/1477-7827-3-74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Rinehart BK, Terrone DA, Lagoo-Deenadayalan S, Barber WH, Hale EA, Martin JN, Jr, Bennett WA. Expression of the placental cytokines tumor necrosis factor alpha, interleukin 1beta, and interleukin 10 is increased in preeclampsia. Am J Obstet Gynecol. 1999;181:915–920. doi: 10.1016/s0002-9378(99)70325-x. [DOI] [PubMed] [Google Scholar]
- 82.Grosskinsky CM, Halme J. Endometriosis: the host response. Baillieres Clin Obstet Gynaecol. 1993;7:701–713. doi: 10.1016/s0950-3552(05)80459-6. [DOI] [PubMed] [Google Scholar]
- 83.Pendergrass PB, Belovicz MW, Reeves CA. Surface area of the human vagina as measured from vinyl polysiloxane casts. Gynecol Obstet Invest. 2003;55:110–113. doi: 10.1159/000070184. [DOI] [PubMed] [Google Scholar]
- 84.Barnhart KT, Izquierdo A, Pretorius ES, Shera DM, Shabbout M, Shaunik A. Baseline dimensions of the human vagina. Hum Reprod. 2006;21:1618–1622. doi: 10.1093/humrep/del022. [DOI] [PubMed] [Google Scholar]
- 85.Farage M, Maibach H. Lifetime changes in the vulva and vagina. Arch Gynecol Obstet. 2006;273:195–202. doi: 10.1007/s00404-005-0079-x. [DOI] [PubMed] [Google Scholar]
- 86.Herbst-Kralovetz MM, Quayle AJ, Ficarra M, Greene S, Rose WA, Chesson R, Spagnuolo RA, Pyles RB. Quantification and comparison of tolllike receptor expression and responsiveness in primary and immortalized human female lower genital tract epithelia. Am J Reprod Immunol. 2008;59:212–224. doi: 10.1111/j.1600-0897.2007.00566.x. [DOI] [PubMed] [Google Scholar]
- 87.Steele C, Fidel PL. Cytokine and chemokine production by human oral and vaginal epithelial cells in response to Candida albicans. Infect Immun. 2002;70:577–583. doi: 10.1128/IAI.70.2.577-583.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Cremel M, Hamzeh-Cognasse H, Genin C, Delezay O. Female genital tract immunization: evaluation of candidate immunoadjuvants on epithelial cell secretion of CCL20 and dendritic/Langerhans cell maturation. Vaccine. 2006;24:5744–5754. doi: 10.1016/j.vaccine.2006.04.044. [DOI] [PubMed] [Google Scholar]
- 89.Scapini P, Laudanna C, Pinardi C, Allavena P, Mantovani A, Sozzani S, Cassatella M. Neutrophils produce biologically active macrophage inflammatory protein-3a (MIP-3a)/CCL20 and MIP-3b/CCL19. Eur J Immunol. 2001;31:981–988. doi: 10.1002/1521-4141(200107)31:7<1981::aid-immu1981>3.0.co;2-x. [DOI] [PubMed] [Google Scholar]
- 90.Soboll G, Schaefer TM, Wira CR. Effect of toll-like receptor (TLR) agonists on TLR and microbicide expression in uterine and vaginal tissues of the mouse. Am J Reprod Immunol. 2006;55:434–446. doi: 10.1111/j.1600-0897.2006.00381.x. [DOI] [PubMed] [Google Scholar]
- 91.Soboll G, Crane-Godreau M, Lyimo M, Wira CR. Effect of oestradiol on PAMP-mediated CCL20/MIP-3 alpha production by mouse uterine epithelial cells in culture. Immunology. 2006;118:185–194. doi: 10.1111/j.1365-2567.2006.02353.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Crane-Godreau MA, Wira CR. Effects of estradiol on lipopolysaccharide and Pam3Cys stimulation of CCL20/macrophage inflammatory protein 3 alpha and tumor necrosis factor alpha production by uterine epithelial cells in culture. Infect Immun. 2005;73:4231–4237. doi: 10.1128/IAI.73.7.4231-4237.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Jungi TW, Besedovsky HO, Sorkin E, Schardt M. Hormonal control of neutrophil chemotactic activity in the rat vagina. Am J Physiol. 1977;233:R59–R65. doi: 10.1152/ajpregu.1977.233.1.R59. [DOI] [PubMed] [Google Scholar]
- 94.Kaushic C, Frauendorf E, Rossoll RM, Richardson JM, Wira CR. Influence of the estrous cycle on the presence and distribution of immune cells in the rat reproductive tract. Am J Reprod Immunol. 1998;39:209–216. doi: 10.1111/j.1600-0897.1998.tb00355.x. [DOI] [PubMed] [Google Scholar]
- 95.Starkey PM, Clover LM, Rees MC. Variation during the menstrual cycle of immune cell populations in human endometrium. Eur J Obstet Gynecol Reprod Biol. 1991;39:203–207. doi: 10.1016/0028-2243(91)90058-s. [DOI] [PubMed] [Google Scholar]
- 96.Sasaki S, Nagata K, Kobayashi Y. Regulation of the estrous cycle by neutrophil infiltration into the vagina. Biochem Biophys Res Commun. 2009;382:35–40. doi: 10.1016/j.bbrc.2009.02.112. [DOI] [PubMed] [Google Scholar]
- 97.Coleman KD, Wright JA, Ghosh M, Wira CR, Fahey JV. Estradiol modulation of hepatocyte growth factor by stromal fibroblasts in the female reproductive tract. Fertil Steril. 2009;92:1107–1109. doi: 10.1016/j.fertnstert.2008.10.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Cooke PS, Buchanan DL, Young P, Setiawan T, Brody J, Korach KS, Taylor J, Lubahn DB, Cunha GR. Stromal estrogen receptors mediate mitogenic effects of estradiol on uterine epithelium. Proc Natl Acad Sci U S A. 1997;94:6535–6540. doi: 10.1073/pnas.94.12.6535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Lail-Trecker M, Gulati R, Peluso JJ. A role for hepatocyte growth factor/scatter factor in regulating normal and neoplastic cells of reproductive tissues. J Soc Gynecol Investig. 1998;5:114–121. doi: 10.1016/s1071-5576(97)00111-1. [DOI] [PubMed] [Google Scholar]
- 100.Kankuri E, Cholujova D, Comajova M, Vaheri A, Bizik J. Induction of hepatocyte growth factor/scatter factor by fibroblast clustering directly promotes tumor cell invasiveness. Cancer Res. 2005;65:9914–9922. doi: 10.1158/0008-5472.CAN-05-1559. [DOI] [PubMed] [Google Scholar]
- 101.Skibinski G, Elborn JS, Ennis M. Bronchial epithelial cell growth regulation in fibroblast cocultures: the role of hepatocyte growth factor. Am J Physiol Lung Cell Mol Physiol. 2007;293:L69–L76. doi: 10.1152/ajplung.00299.2006. [DOI] [PubMed] [Google Scholar]
- 102.Conway K, Price P, Harding KG, Jiang WG. The molecular and clinical impact of hepatocyte growth factor, its receptor, activators, and inhibitors in wound healing. Wound Repair Regen. 2006;14:2–10. doi: 10.1111/j.1743-6109.2005.00081.x. [DOI] [PubMed] [Google Scholar]
- 103.Grotegut S, von Schweinitz D, Christofori G, Lehembre F. Hepatocyte growth factor induces cell scattering through MAPK/Egr-1-mediated upregulation of Snail. EMBO J. 2006;25:3534–3545. doi: 10.1038/sj.emboj.7601213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Morishita R, Aoki M, Hashiya N, Yamasaki K, Kurinami H, Shimizu S, Makino H, Takesya Y, Azuma J, Ogihara T. Therapeutic angiogenesis using hepatocyte growth factor (HGF) Curr Gene Ther. 2004;4:199–206. doi: 10.2174/1566523043346453. [DOI] [PubMed] [Google Scholar]
- 105.Birchmeier C, Gherardi E. Developmental roles of HGF/SF and its receptor, the c-Met tyrosine kinase. Trends Cell Biol. 1998;8:404–410. doi: 10.1016/s0962-8924(98)01359-2. [DOI] [PubMed] [Google Scholar]
- 106.Zhang YW, Vande Woude GF. HGF/SF-met signaling in the control of branching morphogenesis and invasion. J Cell Biochem. 2003;88:408–417. doi: 10.1002/jcb.10358. [DOI] [PubMed] [Google Scholar]
- 107.Sugawara J, Fukaya T, Murakami T, Yoshida H, Yajima A. Hepatocyte growth factor stimulated proliferation, migration, and lumen formation of human endometrial epithelial cells in vitro. Biol Reprod. 1997;57:936–942. doi: 10.1095/biolreprod57.4.936. [DOI] [PubMed] [Google Scholar]
- 108.Grant-Tschudy KS, Wira CR. Hepatocyte Growth Factor regulation of uterine epithelial cell transepithelial resistance and Tumor Necrosis Factor alpha release in culture. Biol Reprod. 2004;72:814–821. doi: 10.1095/biolreprod.104.035618. [DOI] [PubMed] [Google Scholar]
- 109.Grant-Tschudy KS, Wira CR. Paracrine mediators of mouse uterine epithelial cell transepithelial resistance in culture. J Reprod Immunol. 2005;67:1–12. doi: 10.1016/j.jri.2005.06.001. [DOI] [PubMed] [Google Scholar]
- 110.Pioli PA, Weaver LK, Schaefer TM, Wright JA, Wira CR, Guyre PM. Lipopolysaccharide-induced IL-1 beta production by human uterine macrophages up-regulates uterine epithelial cell expression of human beta-defensin 2. J Immunol. 2006;176:6647–6655. doi: 10.4049/jimmunol.176.11.6647. [DOI] [PubMed] [Google Scholar]
- 111.Geijtenbeek TB, Torensma R, van Vliet SJ, van Duijnhoven GC, Adema GJ, van Kooyk Y, Figdor CG. Identification of DC-SIGN, a novel dendritic cell-specific ICAM-3 receptor that supports primary immune responses. Cell. 2000;100:575–585. doi: 10.1016/s0092-8674(00)80693-5. [DOI] [PubMed] [Google Scholar]
- 112.Granelli-Piperno A, Pritsker A, Pack M, Shimeliovich I, Arrighi JF, Park CG, Trumpfheller C, Piguet V, Moran TM, Steinman RM. Dendritic cell-specific intercellular adhesion molecule 3-grabbing nonintegrin/CD209 is abundant on macrophages in the normal human lymph node and is not required for dendritic cell stimulation of the mixed leukocyte reaction. J Immunol. 2005;175:4265–4273. doi: 10.4049/jimmunol.175.7.4265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Geijtenbeek TB, Kwon DS, Torensma R, van Vliet SJ, van Duijnhoven GC, Middel J, Cornelissen IL, Nottet HS, KewalRamani VN, Littman DR, Figdor CG, van Kooyk Y. DC-SIGN, a dendritic cell-specific HIV-1-binding protein that enhances trans-infection of T cells. Cell. 2000;100:587–597. doi: 10.1016/s0092-8674(00)80694-7. [DOI] [PubMed] [Google Scholar]
- 114.van Kooyk Y, Geijtenbeek TB. DC-SIGN: escape mechanism for pathogens. Nat Rev. 2003;3:697–709. doi: 10.1038/nri1182. [DOI] [PubMed] [Google Scholar]
- 115.Miller CJ, McChesney M, Moore PF. Langerhans cells, macrophages and lymphocyte subsets in the cervix and vagina of rhesus macaques. Lab Invest. 1992;67:628–634. [PubMed] [Google Scholar]
- 116.Miller CJ, Shattock RJ. Target cells in vaginal HIV transmission. Microbes Infect. 2003;5:59–67. doi: 10.1016/s1286-4579(02)00056-4. [DOI] [PubMed] [Google Scholar]
- 117.Miller CJ. Localization of Simian immunodeficiency virus-infected cells in the genital tract of male and female Rhesus macaques. J Reprod Immunol. 1998;41:331–339. doi: 10.1016/s0165-0378(98)00069-2. [DOI] [PubMed] [Google Scholar]
- 118.Jameson B, Baribaud F, Pohlmann S, Ghavimi D, Mortari F, Doms RW, Iwasaki A. Expression of DCSIGN by dendritic cells of intestinal and genital mucosae in humans and rhesus macaques. J Virol. 2002;76:1866–1875. doi: 10.1128/JVI.76.4.1866-1875.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Figdor CG, van Kooyk Y, Adema GJ. C-type lectin receptors on dendritic cells and Langerhans cells. Nat Rev. 2002;2:77–84. doi: 10.1038/nri723. [DOI] [PubMed] [Google Scholar]
- 120.Turville SG, Cameron PU, Handley A, Lin G, Pohlmann S, Doms RW, Cunningham AL. Diversity of receptors binding HIV on dendritic cell subsets. Nat Immunol. 2002;3:975–983. doi: 10.1038/ni841. [DOI] [PubMed] [Google Scholar]
- 121.Wu L, KewalRamani VN. Dendritic-cell interactions with HIV: infection and viral dissemination. Nat Rev. 2006;6:859–868. doi: 10.1038/nri1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Sallusto F, Lanzavecchia A. Efficient presentation of soluble antigen by cultured human dendritic cells is maintained by granulocyte/macrophage colonystimulating factor plus interleukin 4 and downregulated by tumor necrosis factor alpha. J Exp Med. 1994;179:1109–1118. doi: 10.1084/jem.179.4.1109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Trinchieri G. Biology of natural killer cells. Adv Immunol. 1989;47:187–376. doi: 10.1016/S0065-2776(08)60664-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.King A, Wellings V, Gardner L, Loke YW. Immunocytochemical characterization of the unusual large granular lymphocytes in human endometrium throughout the menstrual cycle. Hum Immunol. 1989;24:195–205. doi: 10.1016/0198-8859(89)90060-8. [DOI] [PubMed] [Google Scholar]
- 125.Sentman CL, Meadows SK, Wira CR, Eriksson M. Recruitment of uterine NK cells: induction of CXC chemokine ligands 10 and 11 in human endometrium by estradiol and progesterone. J Immunol. 2004;173:6760–6766. doi: 10.4049/jimmunol.173.11.6760. [DOI] [PubMed] [Google Scholar]
- 126.Khalil N. Post translational activation of latent transforming growth factor beta (L-TGF-beta): clinical implications. Histol Histopathol. 2001;16:541–551. doi: 10.14670/HH-16.541. [DOI] [PubMed] [Google Scholar]
- 127.Eriksson M, Meadows SK, Wira CR, Sentman CL. Unique phenotype of human uterine NK cells and their regulation by endogenous TGF-beta. J Leukoc Biol. 2004;76:667–675. doi: 10.1189/jlb.0204090. [DOI] [PubMed] [Google Scholar]
- 128.Meadows SK, Eriksson M, Barber A, Sentman CL. Human NK cell IFN-gamma production is regulated by endogenous TGF-beta. Int Immunopharmacol. 2006;6:1020–1028. doi: 10.1016/j.intimp.2006.01.013. [DOI] [PubMed] [Google Scholar]
- 129.Eriksson M, Meadows SK, Basu S, Mselle TF, Wira CR, Sentman CL. TLRs mediate IFN-gamma production by human uterine NK cells in endometrium. J Immunol. 2006;176:6219–6224. doi: 10.4049/jimmunol.176.10.6219. [DOI] [PubMed] [Google Scholar]
- 130.Eriksson M, Meadows SK, Wira CR, Sentman CL. Endogenous transforming growth factor-beta inhibits toll-like receptor mediated activation of human uterine natural killer cells. Am J Reprod Immunol. 2006;56:321–328. doi: 10.1111/j.1600-0897.2006.00432.x. [DOI] [PubMed] [Google Scholar]
- 131.Witko-Sarsat V, Rieu P, Descamps-Latscha B, Lesavre P, Halbwachs-Mecarelli L. Neutrophils: molecules, functions and pathophysiological aspects. Lab Invest. 2000;80:617–653. doi: 10.1038/labinvest.3780067. [DOI] [PubMed] [Google Scholar]
- 132.Smith JM, Shen Z, Wira CR, Fanger MW, Shen L. Effects of menstrual cycle status and gender on human neutrophil phenotype. Am J Reprod Immunol. 2007;58:111–119. doi: 10.1111/j.1600-0897.2007.00494.x. [DOI] [PubMed] [Google Scholar]
- 133.Shen L, Fahey JV, Hussey SB, Asin SN, Wira CR, Fanger MW. Synergy between IL-8 and GM-CSF in reproductive tract epithelial cell secretions promotes enhanced neutrophil chemotaxis. Cell Immunology. 2004;230:23–32. doi: 10.1016/j.cellimm.2004.08.004. [DOI] [PubMed] [Google Scholar]
- 134.Schaefer TM, Fahey JV, Wright JA, Wira CR. Migration inhibitory factor secretion by polarized uterine epithelial cells is enhanced in response to the TLR3 agonist poly (I:C) Am J Reprod Immunol. 2005;54:193–202. doi: 10.1111/j.1600-0897.2005.00298.x. [DOI] [PubMed] [Google Scholar]