Abstract
Functionally inverted plasma membrane vesicles isolated from the eukaryotic microorganism Neurospora crassa catalyze Mg2+/ATP-dependent Ca2+ uptake. Inhibitors induced efflux studies and isotope-exchange experiments indicate that the Ca2+ is accumulated inside the vesicles against a concentration gradient of about 40-fold, and that the majority of the transported Ca2+ is present essentially in free solution. Comparisons of Mg2+/ATP-driven 45Ca2+ uptake and [14C]SCN-uptake with respect to the Mg2+/ATP concentration dependence, the effects of inhibitors, and the nucleotide and divalent cation specificities indicate that the energy for Ca2+ accumulation is derived from ATP hydrolysis catalyzed by the electrogenic plasma membrane ATPase. Energized Ca2+ uptake is stimulated by the permeant anion SCN- to a degree that varies reciprocally with the ability of this anion to dissipate the membrane potential, and is inhibited by K+ in the presence of nigericin. All of these data point to the conclusion that the active transport of Ca2+ across the Neurospora plasma membrane takes place via a Ca2+/H+ antiporter, which functions to pump Ca2+ out of the intact cell.
Full text
PDF

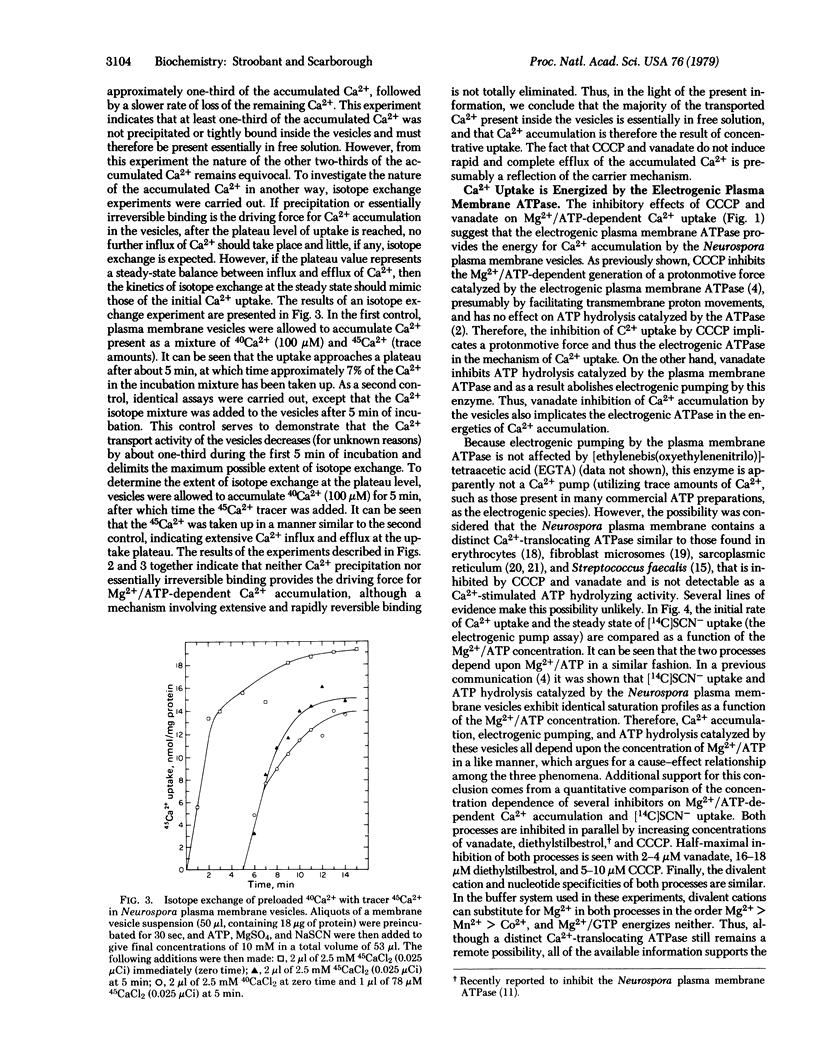
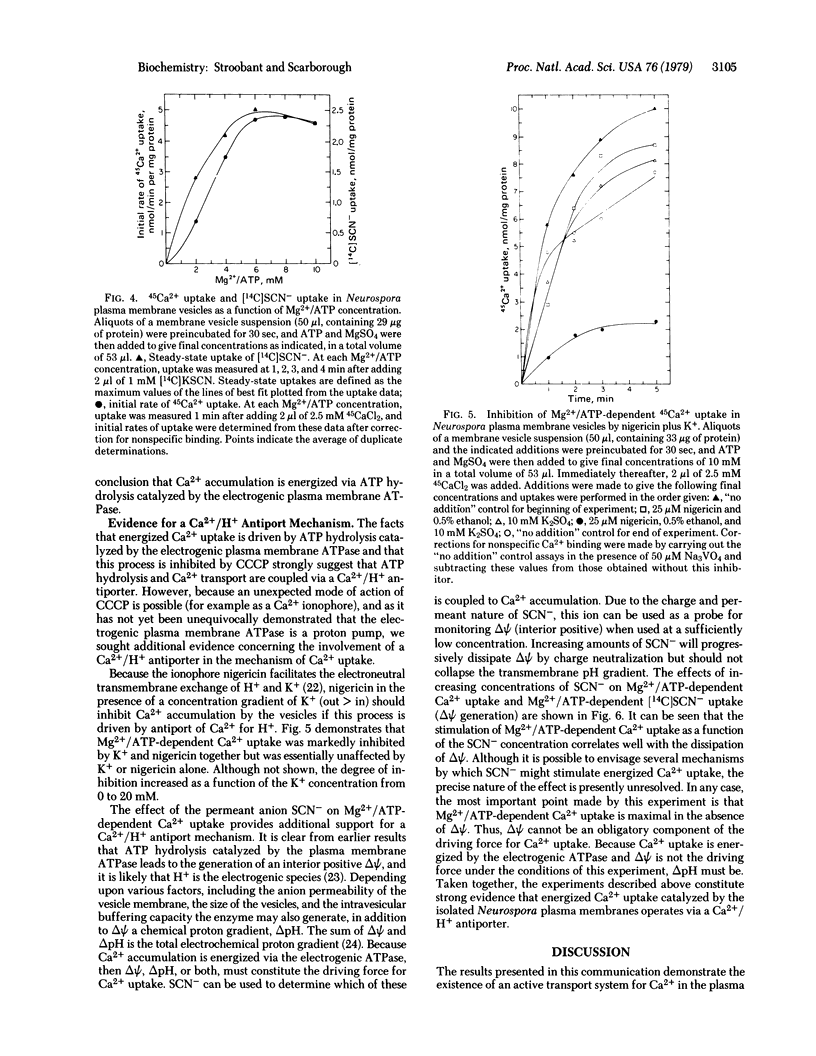

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Belliveau J. W., Lanyi J. K. Calcium transport in Halobacterium halobium envelope vesicles. Arch Biochem Biophys. 1978 Feb;186(1):98–105. doi: 10.1016/0003-9861(78)90468-x. [DOI] [PubMed] [Google Scholar]
- Bowman B. J., Mainzer S. E., Allen K. E., Slayman C. W. Effects of inhibitors on the plasma membrane and mitochondrial adenosine triphosphatases of Neurospora crassa. Biochim Biophys Acta. 1978 Sep 11;512(1):13–28. doi: 10.1016/0005-2736(78)90214-6. [DOI] [PubMed] [Google Scholar]
- Bowman B. J., Slayman C. W. Characterization of plasma membrane adenosine triphosphatase of Neurospora crassa. J Biol Chem. 1977 May 25;252(10):3357–3363. [PubMed] [Google Scholar]
- Christiansen R. O., Steensland H., Loyter A., Saltzgaber J., Racker E. Energy-linked ion translocation in submitochondrial particles. II. Properties of submitochondrial particles capable of Ca++ translocation. J Biol Chem. 1969 Aug 25;244(16):4428–4436. [PubMed] [Google Scholar]
- Gross J., Marmé D. ATP-dependent Ca uptake into plant membrane vesicles. Proc Natl Acad Sci U S A. 1978 Mar;75(3):1232–1236. doi: 10.1073/pnas.75.3.1232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harold F. M. Conservation and transformation of energy by bacterial membranes. Bacteriol Rev. 1972 Jun;36(2):172–230. doi: 10.1128/br.36.2.172-230.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris E. J., Wimhurst J. M., Landaeta I. Ionic and non-ionic mitochondrial phosphate in relation to Ca2 plus and arsenate accumulation. Eur J Biochem. 1974 Jun 15;45(2):561–565. doi: 10.1111/j.1432-1033.1974.tb03582.x. [DOI] [PubMed] [Google Scholar]
- Inesi G. Active transport of calcium ion in sarcoplasmic membranes. Annu Rev Biophys Bioeng. 1972;1:191–210. doi: 10.1146/annurev.bb.01.060172.001203. [DOI] [PubMed] [Google Scholar]
- Kobayashi H., Van Brunt J., Harold F. M. ATP-linked calcium transport in cells and membrane vesicles of Streptococcus faecalis. J Biol Chem. 1978 Apr 10;253(7):2085–2092. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Martonosi A. N., Chyn T. L., Schibeci A. The calcium transport of sarcoplasmic reticulum. Ann N Y Acad Sci. 1978 Apr 28;307:148–159. doi: 10.1111/j.1749-6632.1978.tb41940.x. [DOI] [PubMed] [Google Scholar]
- Mitchell P. Chemiosmotic coupling in oxidative and photosynthetic phosphorylation. Biol Rev Camb Philos Soc. 1966 Aug;41(3):445–502. doi: 10.1111/j.1469-185x.1966.tb01501.x. [DOI] [PubMed] [Google Scholar]
- Mitchell P. Vectorial chemistry and the molecular mechanics of chemiosmotic coupling: power transmission by proticity. Biochem Soc Trans. 1976;4(3):399–430. doi: 10.1042/bst0040399. [DOI] [PubMed] [Google Scholar]
- Moore L., Pastan I. Energy-dependent calcium uptake by fibroblast microsomes. Ann N Y Acad Sci. 1978 Apr 28;307:177–194. doi: 10.1111/j.1749-6632.1978.tb41942.x. [DOI] [PubMed] [Google Scholar]
- Pressman B. C. Biological applications of ionophores. Annu Rev Biochem. 1976;45:501–530. doi: 10.1146/annurev.bi.45.070176.002441. [DOI] [PubMed] [Google Scholar]
- Rasmussen H., Goodman D. B. Relationships between calcium and cyclic nucleotides in cell activation. Physiol Rev. 1977 Jul;57(3):421–509. doi: 10.1152/physrev.1977.57.3.421. [DOI] [PubMed] [Google Scholar]
- Scarborough G. A. Isolation and characterization of Neurospora crassa plasma membranes. J Biol Chem. 1975 Feb 10;250(3):1106–1111. [PubMed] [Google Scholar]
- Scarborough G. A. Properties of Neurospora crassa plasma membrane ATPase. Arch Biochem Biophys. 1977 Apr 30;180(2):384–393. doi: 10.1016/0003-9861(77)90052-2. [DOI] [PubMed] [Google Scholar]
- Scarborough G. A. The neurospora plasma membrane ATPase is an electrogenic pump. Proc Natl Acad Sci U S A. 1976 May;73(5):1485–1488. doi: 10.1073/pnas.73.5.1485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scarborough G. A. The neurospora plasma membrane: a new experimental system for investigating eukaryote surface membrane structure and function. Methods Cell Biol. 1978;20:117–133. doi: 10.1016/s0091-679x(08)62014-2. [DOI] [PubMed] [Google Scholar]
- Stroobant P., Scarborough G. A. Large scale isolation and storage of Neurospora plasma membranes. Anal Biochem. 1979 Jun;95(2):554–558. doi: 10.1016/0003-2697(79)90771-1. [DOI] [PubMed] [Google Scholar]
- Tsuchiya T., Rosen B. P. Characterization of an active transport system for calcium in inverted membrane vesicles of Escherichia coli. J Biol Chem. 1975 Oct 10;250(19):7687–7692. [PubMed] [Google Scholar]



