Abstract
Double-stranded cDNA synthesized from delta-crystallin mRNA isolated from lens fiber cells of 15-day-old embryonic chicken was cloned in Escherichia coli chi 1776 in the Pst I site of the plasmid pBR322 by using the oligo(dC) . oligo(dG) joining procedure. Twelve Amps Tetr transformants contained sequences complementary to purified delta-crystallin [32P]cDNA. One of the recombinant clones (p delta Cr-2) had an insert of 1241 +/- 240 base pairs, as judged by R-looping analysis with purified delta-crystallin mRNA. The inserted cDNA represents at least 69% of the delta-crystallin coding sequences. p delta Cr-2 was further characterized by restriction analysis, protection of delta-crystallin [3H]cDNA from digestion by S1 nuclease, and hybrid-mediated arrest of delta-crystallin mRNA translation in vitro. p delta Cr-2 provides an invaluable probe for additional analysis of the primary structure, gene organization, and regulated synthesis of delta-crystallin, the principal protein synthesized during lens differentiation in the chicken embryo.
Full text
PDF
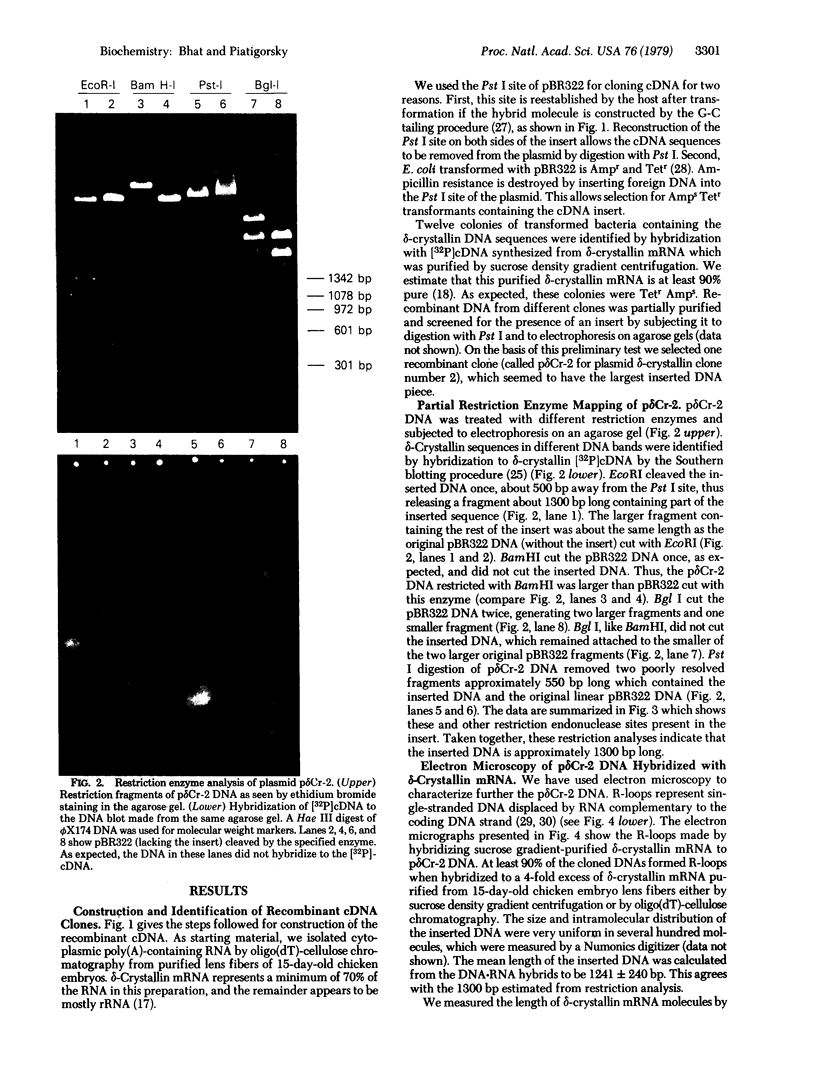


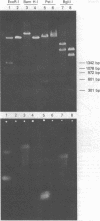
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bolivar F., Rodriguez R. L., Greene P. J., Betlach M. C., Heyneker H. L., Boyer H. W., Crosa J. H., Falkow S. Construction and characterization of new cloning vehicles. II. A multipurpose cloning system. Gene. 1977;2(2):95–113. [PubMed] [Google Scholar]
- Breathnach R., Mandel J. L., Chambon P. Ovalbumin gene is split in chicken DNA. Nature. 1977 Nov 24;270(5635):314–319. doi: 10.1038/270314a0. [DOI] [PubMed] [Google Scholar]
- Clayton R. M., Truman D. E. Molecular structure and antigenicity of lens proteins. Nature. 1967 Jun 17;214(5094):1201–1204. doi: 10.1038/2141201a0. [DOI] [PubMed] [Google Scholar]
- Denhardt D. T. A membrane-filter technique for the detection of complementary DNA. Biochem Biophys Res Commun. 1966 Jun 13;23(5):641–646. doi: 10.1016/0006-291x(66)90447-5. [DOI] [PubMed] [Google Scholar]
- Eguchi G., Okada T. S. Differentiation of lens tissue from the progeny of chick retinal pigment cells cultured in vitro: a demonstration of a switch of cell types in clonal cell culture. Proc Natl Acad Sci U S A. 1973 May;70(5):1495–1499. doi: 10.1073/pnas.70.5.1495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enea V., Vovis G. F., Zinder N. D. Genetic studies with heteroduplex DNA of bacteriophage fl. Asymmetric segregation, base correction and implications for the mechanism of genetic recombination. J Mol Biol. 1975 Aug 15;96(3):495–509. doi: 10.1016/0022-2836(75)90175-8. [DOI] [PubMed] [Google Scholar]
- Genis-Galves J. M., Maisel H., Castro J. Changes in chick lens proteins with aging. Exp Eye Res. 1968 Oct;7(4):593–602. doi: 10.1016/s0014-4835(68)80014-4. [DOI] [PubMed] [Google Scholar]
- Glover D. M., Hogness D. S. A novel arrangement of the 18S and 28S sequences in a repeating unit of Drosophila melanogaster rDNA. Cell. 1977 Feb;10(2):167–176. doi: 10.1016/0092-8674(77)90212-4. [DOI] [PubMed] [Google Scholar]
- Grunstein M., Hogness D. S. Colony hybridization: a method for the isolation of cloned DNAs that contain a specific gene. Proc Natl Acad Sci U S A. 1975 Oct;72(10):3961–3965. doi: 10.1073/pnas.72.10.3961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guerry P., LeBlanc D. J., Falkow S. General method for the isolation of plasmid deoxyribonucleic acid. J Bacteriol. 1973 Nov;116(2):1064–1066. doi: 10.1128/jb.116.2.1064-1066.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harding J. J., Dilley K. J. Structural proteins of the mammalian lens: a review with emphasis on changes in development, aging and cataract. Exp Eye Res. 1976 Jan;22(1):1–73. doi: 10.1016/0014-4835(76)90033-6. [DOI] [PubMed] [Google Scholar]
- Humphries P., Old R., Coggins L. W., McShane T., Watson C., Paul J. Recombinant plasmids containing Xenopus laevis globin structural genes derived from complementary DNA. Nucleic Acids Res. 1978 Mar;5(3):905–924. doi: 10.1093/nar/5.3.905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meagher R. B., Tait R. C., Betlach M., Boyer H. W. Protein expression in E. coli minicells by recombinant plasmids. Cell. 1977 Mar;10(3):521–536. doi: 10.1016/0092-8674(77)90039-3. [DOI] [PubMed] [Google Scholar]
- Meyer J., Neuwald P. D., Lai S. P., Maizel J. V., Jr, Westphal H. Electron microscopy of late adenovirus type 2 mRNA hybridized to double-stranded viral DNA. J Virol. 1977 Mar;21(3):1010–1018. doi: 10.1128/jvi.21.3.1010-1018.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milstone L. M., Zelenka P., Piatigorsky J. Delta-crystallin mRNA in chick lens cells: mRNA accumulates during differential stimulation of delta-crystallin synthesis in cultured cells. Dev Biol. 1976 Feb;48(2):197–204. doi: 10.1016/0012-1606(76)90084-1. [DOI] [PubMed] [Google Scholar]
- Nguyen-Huu M. C., Stratmann M., Groner B., Wurtz T., Land H., Giesecke K., Sippel A. E., Schütz G. Chicken lysozyme gene contains several intervening sequences. Proc Natl Acad Sci U S A. 1979 Jan;76(1):76–80. doi: 10.1073/pnas.76.1.76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okada T. S., Eguchi G., Takeichi M. The retention of differentiated properties by lens epithelial cells in clonal cell culture. Dev Biol. 1973 Oct;34(2):321–333. doi: 10.1016/0012-1606(73)90361-8. [DOI] [PubMed] [Google Scholar]
- PHILPOTT G. W., COULOMBRE A. J. LENS DEVELOPMENT. II. THE DIFFERENTIATION OF EMBRYONIC CHICK LENS EPITHELIAL CELLS IN VITRO AND IN VIVO. Exp Cell Res. 1965 Jun;38:635–644. doi: 10.1016/0014-4827(65)90387-3. [DOI] [PubMed] [Google Scholar]
- Paterson B. M., Roberts B. E., Kuff E. L. Structural gene identification and mapping by DNA-mRNA hybrid-arrested cell-free translation. Proc Natl Acad Sci U S A. 1977 Oct;74(10):4370–4374. doi: 10.1073/pnas.74.10.4370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piatigorsky J. Subunit composition of delta-crystallin from embryonic chick lens. Analysis of methionine-containing tryptic peptides and cyanogen bromide peptides. J Biol Chem. 1976 Jul 25;251(14):4416–4420. [PubMed] [Google Scholar]
- Piatigorsky J., Webster H. D., Craig S. P. Protein synthesis and ultrastructure during the formation of embryonic chick lens fibers in vivo and in vitro. Dev Biol. 1972 Feb;27(2):176–189. doi: 10.1016/0012-1606(72)90096-6. [DOI] [PubMed] [Google Scholar]
- RABAEY M. Electrophoretic and immunoelectrophoretic studies on the soluble proteins in the developing lens of birds. Exp Eye Res. 1962 Jun;1:310–316. doi: 10.1016/s0014-4835(62)80017-7. [DOI] [PubMed] [Google Scholar]
- Reszelbach R., Shinohara T., Piatigorsky J. Resolution of two distinct embryonic chick delta-crystallin bands by polyacrylamide gel electrophoresis in the presence of sodium dodecyl sulfate and urea. Exp Eye Res. 1977 Dec;25(6):583–593. doi: 10.1016/0014-4835(77)90137-3. [DOI] [PubMed] [Google Scholar]
- Shinohara T., Piatigorsky J. Quantitation of delta-crystallin messenger RNA during lens induction in chick embryos. Proc Natl Acad Sci U S A. 1976 Aug;73(8):2808–2812. doi: 10.1073/pnas.73.8.2808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Sutcliffe J. G. pBR322 restriction map derived from the DNA sequence: accurate DNA size markers up to 4361 nucleotide pairs long. Nucleic Acids Res. 1978 Aug;5(8):2721–2728. doi: 10.1093/nar/5.8.2721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas M., White R. L., Davis R. W. Hybridization of RNA to double-stranded DNA: formation of R-loops. Proc Natl Acad Sci U S A. 1976 Jul;73(7):2294–2298. doi: 10.1073/pnas.73.7.2294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomson I., Wilkinson C. E., Jackson J. F., de Pomerai D. I., Clayton R. M., Truman D. E., Williamson R. Isolation and cell-free translation of chick lens crystallin mRNA during normal development and transdifferentiation of neural retina. Dev Biol. 1978 Aug;65(2):372–382. doi: 10.1016/0012-1606(78)90033-7. [DOI] [PubMed] [Google Scholar]
- Tilghman S. M., Tiemeier D. C., Seidman J. G., Peterlin B. M., Sullivan M., Maizel J. V., Leder P. Intervening sequence of DNA identified in the structural portion of a mouse beta-globin gene. Proc Natl Acad Sci U S A. 1978 Feb;75(2):725–729. doi: 10.1073/pnas.75.2.725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Truman D. E., Brown A. G., Rao K. V. Estimation of the molecular weights of chick -and -crystallins and their subunits by gel filtration. Exp Eye Res. 1971 Nov;12(3):304–310. doi: 10.1016/0014-4835(71)90154-0. [DOI] [PubMed] [Google Scholar]
- Yoshida K., Katoh A. Crystallin synthesis by chick lens. II. Changes in synthetic activities of epithelial and fiber cells during embryonic development. Exp Eye Res. 1971 Mar;11(2):184–194. doi: 10.1016/s0014-4835(71)80022-2. [DOI] [PubMed] [Google Scholar]
- Zelenka P., Piatigorsky J. Isolation and in vitro translation of delta-crystallin mRNA from embryonic chick lens fibers. Proc Natl Acad Sci U S A. 1974 May;71(5):1896–1900. doi: 10.1073/pnas.71.5.1896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zelenka P., Piatigorsky J. Molecular weight and sequence complexity of delta-crystallin mRNA. Exp Eye Res. 1976 Feb;22(2):115–124. doi: 10.1016/0014-4835(76)90038-5. [DOI] [PubMed] [Google Scholar]
- Zwaan J., Ikeda A. Macromolecular events during differentiation of the chicken lens. Exp Eye Res. 1968 Apr;7(2):301–311. doi: 10.1016/s0014-4835(68)80081-8. [DOI] [PubMed] [Google Scholar]