Abstract
Background
MMP2 has been shown to play an important role in cancer cell invasion and the expression of MMP2 is associated with the poor prognosis of prostate cancer; however, the mechanism of MMP2 expression is largely unknown.
SIRT1 is a nicotinamide adenine dinucleotide (NAD)-dependent histone deacetylase (class III HDAC) that has recently been shown to have implications in regulating cancer cell growth and apoptosis. The purpose of this study is to determine the role of SIRT1 in regulating MMP2 expression and tumor invasion in prostate cancer cells.
Methods
The interfering RNAi was used to knockdown SIRT1 from prostate cancer cells. Immunoblots, RT-PCR, zymographic assays, co-immunoprecipitation analysis and transwell assays were used to examine the effects of SIRT1 silencing on MMP2 expression and activity, on SIRT1 and MMP2 interaction, and on prostate cancer cell invasion. The immuno-histochemical assay was performed to study SIRT1 expression in prostate cancer tissues.
Results
We show that SIRT1 associates and deacetylates MMP2 and SIRT1 regulates MMP2 expression by controlling MMP2 protein stability through the proteosomal pathway. Thus, we demonstrated a novel mechanism in that MMP2 expression can be regulated at the posttranslational level by SIRT1. Furthermore, we determined that SIRT1 inhibition reduced prostate cancer cell invasion and SIRT1 is highly expressed in advanced prostate cancer tissues.
Conclusions
SIRT1 is an important regulator of MMP2 expression, activity, and prostate cancer cell invasion. Overexpressed SIRT1 in advanced prostate cancer may play an important role in prostate cancer progression.
Keywords: SIRT1, MMP2, protein stability, prostate cancer, invasion
Introduction
Prostate cancer is the most frequently diagnosed cancer in men and the second leading cause of cancer deaths in men (1). Prostate cancer develops through a series of stages beginning with prostatic intraepithelial neoplasia (PIN), then continuing through high-grade PIN lesions, invasive cancer, and an androgen-independent state (2–5). The mortality of prostate cancer is mainly due to the progression of localized primary cancer to the advanced stages with metastasis to multiple organs (6, 7).
Matrix metalloproteinases (MMPs) are a family of zinc-dependent endopeptidases that are structurally and functionally related, and are capable of degrading extracellular matrix (ECM) to promote cancer cell invasion (8). In general, MMPs have been found in greater amounts with higher activity in and around malignant cancers than in normal, benign, or premalignant tissues (9) . Among the MMPs, MMP2 in particular has been shown to play an important role in prostate cancer cell migration and invasion, and MMP2 overexpression has been shown to be associated with prostate cancer progression (2, 10). Like many MMPs, MMP2 is secreted as a latent proenzyme and further activated extracellularly (9). MMP2 activity regulation at several levels has been throroughly studied, including by a transcriptional mechanism and by the inhibition of enzymatic activity by endogenous tissue inhibitors of metalloproteinases (11). However, apart from a report showing that phosphorylation modifications can regulate MMP2 activation, the role of posttranslational modification on MMP2 expression, stability, and activity is largely unknown (12). There have been no reports showing that acetylation/deacetylation plays a role in MMP2 expression and activity.
SIRT1 is a nicotinamide adenine dinucleotide (NAD)-dependent histone deacetylase (class III HDAC), which has been reported to play an important role in a variety of physiological processes such as aging, metabolism, neurogenesis, and cell survival, due to its ability to deacetylate both histone and numerous non-histone substrates (13, 14). In this study, we identify SIRT1 as a critical regulator of MMP2 expression, stability, and activity. Our results show that SIRT1 knockdown by RNAi or inhibition by pharmaceutical SIRT1 inhibitors reduces MMP2 protein levels but not mRNA abundance. We also found that SIRT1 silencing destabilizes MMP2 through the proteosomal pathway and reduces zymographic activity. Furthermore, we show that SIRT1 associates with MMP2 and this association can be promoted by hypoxia condition. In addition, we show that SIRT1 deacetylates MMP2 and SIRT1 deacetylase activity is required for MMP2 expression. Finally we demonstrate that SIRT1 silencing decreases prostate cancer cell invasion by targeting MMP2 and SIRT1 is overexpressed in advanced prostate cancer. These results suggest SIRT1 is a positive regulator of MMP2 expression, and overexpressed SIRT1 in advanced prostate cancer may play an important physiological role in prostate cancer progression.
Materials and Methods
Cell lines and antibodies
Prostate cancer cell lines LNCaP and PC3 were obtained from the American Type Culture Collection (Manasses, Virgina). LNCaP cells were maintained in 1× RPMI 1640 plus 10% FBS and PC3 was maintained in 1× DMEM plus 10% FBS. Antibodies to SIRT1 (sc-74504) and MMP2 (H76) (sc-10736) were purchased from Santa Cruz Biotechnology (Santa Cruz, CA). Antibody to acetyl-lysine (06–933) and to SIRT1 (05–707) were purchased from Millipore (Billerica, MA). Antibodies to β –actin were purchased from Sigma-Aldrich (St. Louis, MO).
Establishment of Stable SIRT1-Knockdown Cell Lines
The Phoenix packaging cell line was transfected with pSUPER.retro.puro-SIRT1, or pSUPER.retro.puro vectors (provided by Dr. F. Picard, Laval University, Canada) separately, using Lipofectamine 2000 (Invitrogen, Carlsbad, CA). After 48 hours, the medium containing retrovirus was collected, filtered, treated with polybrene, and transferred to PC3 and LNCaP cell cultures. Infected cells were selected with puromycin and the stably infected colonies were pooled.
Qualitative reverse transcription real-time PCR (qRT-PCR) Analysis
Total RNA was extracted from vector control (pSuper PC3, pSuper LNCaP) and SIRT1 knockdown (siSIRT1 PC3, siLNCaP) cells using TRIzol Reagent (Invitrogen, Carlsbad, CA) according to the manufacturer’s instructions. Reverse transcription was performed to synthesize single stranded cDNA using the SuperScript III First-Strand Synthesis System (Invitrogen, Carlsbad, CA). The qRT-PCR analysis of gene expression was performed using SYBR Green PCR Master Mix (Applied Biosystems, Foster City, CA) on an Applied Biosystems 7500 Real-Time PCR System. Fluorescence data was collected after each cycle. Relative quantification of gene expression was performed by using β-actin as an internal control. The threshold cycle and ∆∆Ct method were used for calculating the relative amount of the target RNA. The experiment was repeated three times.
Immunoprecipitation Assay
LNCaP cells or PC3 cells (5×106) were lysed in 100 µl 1% NP-40 IP lysis buffer. Protein A/G (50 µl) was incubated with each sample for 1 hour at 4°C for pre-clearing. Primary antibodies (SIRT1 antibody (B-10) or MMP2 (H-76) or IgG ) were mixed with pre-cleared lysates for 1 hour at 4°C before the addition of 70 µl protein agarose A/G; reactions were continued overnight at 4°C. The agarose beads were washed three times at RT with 1× IP lysis buffer. The samples were separated on 8% PAGE gels and further analyzed with anti-MMP2, anti-SIRT1, and anti-acetyl-lysine antibodies.
Immunoblot
Cells were lysed with 1% Nonidet P-40 lysis buffer (1% NP40, 150mm NaCl, 50mM Tris-HCl) with protease inhibitor (Roche Diagnostics). Protein samples were subsequently separated on an 8% SDS gel and analyzed with anti-SIRT1, anti-MMP2, anti-acetyl-lysine, and β-actin antibodies.
Zymography
We examined culture supernatants for gelatinase activity using the zymography technique described previously (15). MMP2 activity was detected in the culture media of prostate cancer cells by substrate lysis using 7.5% SDS-polyacrylamide gels containing 2 mg/ml gelatin. Aliquots of culture medium were diluted 1:1 with non-reducing Zymogram Sample Buffer (BioRad, Hercules, CA) and underwent electrophoresis at 4° C. After electrophoresis, SDS was removed by incubating the gel twice with 1X Zymogram Renaturation Buffer (BioRad). Gelatinase activity was revealed by overnight incubation at 37° C with Zymogram Development Buffer (BioRad), followed by staining with 0.1% Coomassie Brilliant Blue.
Cell invasion assay
Cell invasion activity of prostate cancer cells was assessed by BD BioCoat Matrigel Invasion Chambers (BD #354480). Briefly, the cells was trypsined, washed with PBS and resuspended in media containing 1% FBS at a density of 5×10^4 cells/ml. 500 ul of the cell suspension was put onto the upper chamber coated matrigel and 500 ul of culture media were added to the lower chamber of the transwell. After 24 hr culturing at 37°C , the transwells were removed from 24-well plates and stained with Diff-Quick solution. The invaded cells were counted under a light microscope.
Immuno-histochemical assay of prostate tissue
The tissue arrays from Cybrdi (CC19-11-007; CC-19-005 and CC19-01) were examined via standard IHC methods (DAB kit, DAB150, Upstate). Briefly, the prostate cancer tissue arrays were deparaffinized and hydrated through Xylene and graded concentrations of alcohol. Antigen retrieval was performed in 10mM sodium citrate buffer, pH 6.0, for 20 min within a microwave oven, followed by treatment with protein blocker for 10 min. Endogenous peroxidase activity was blocked using 3% H2O2. TMAs were further incubated 1hr at RT with antibodies against SIRT1 (1:2000, Millipore), then incubated with a biotin-conjugated secondary antibody followed by streptavidin-horseradish peroxidase detection with 3’-3’-diaminebenzine (DAB) as the substrate. Counter-staining is performed with Hematoxylin.
Results
SIRT1 inhibition reduces MMP2 expression without changing MMP2 mRNA level
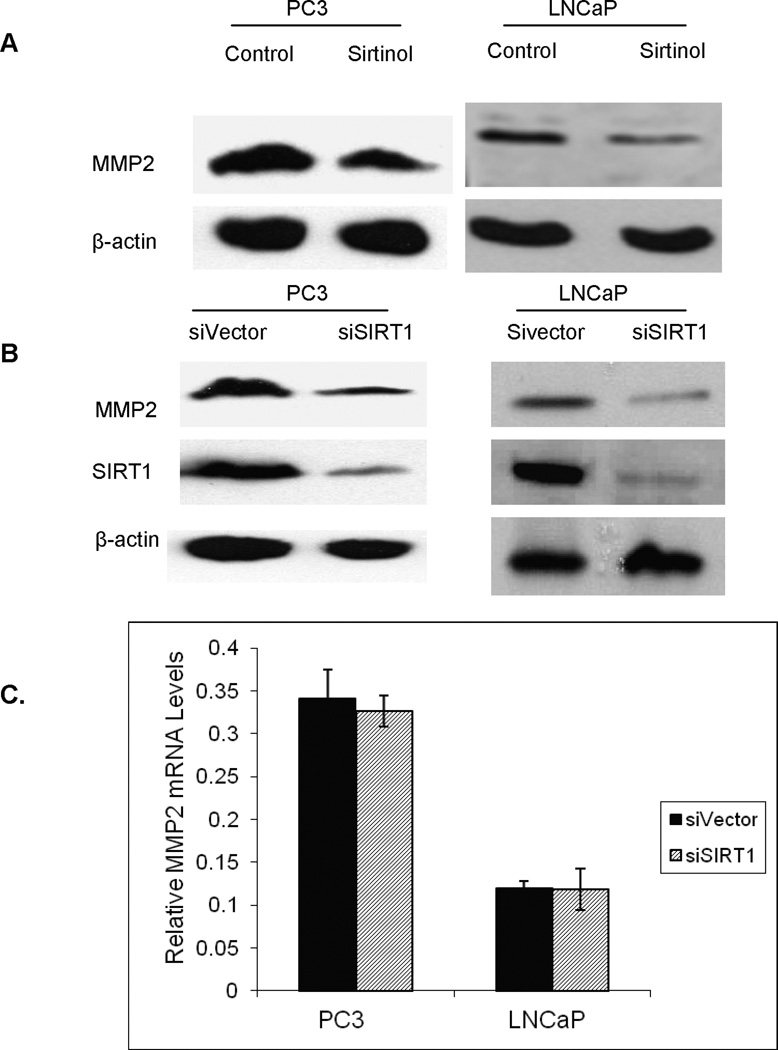
MMP2 is one of the most important MMP proteins involved in the regulation of prostate cancer migration and invasion. In order to study the regulation of SIRT1 on MMP2 expression, we first treated prostate cancer cells with sirtinol, a SIRT1 inhibitor, and found that SIRT1 inhibition suppresses MMP2 expression in both LNCaP and PC3 prostate cancer cells (Figure 1A). Furthermore, we established SIRT1 knockdown in prostate cancer cell lines by infection with the SIRT1 RNAi (siSIRT1) or RNAi vector control (pSuper) to confirm the regulation of SIRT1 on MMP2 expression. Consistent with the SIRT1 inhibition data, SIRT1 knockdown decreases MMP2 expression in both LNCaP and PC3 cells (Figure 1B), while knockdown of other sirtuins that we studied, e.g. SIRT2 and SIRT7, had no effect on MMP2 expression (data not shown). These results suggest that there is some specificity to the regulation of MMP2 expression by SIRT1.
Figure 1.
SIRT1 inhibition down-regulates MMP2 expression without change MMP2 mRNA abundance. A. SIRT1 inhibition decreases MMP2 expression. Prostate cancer cell lines PC3 and LNCaP were treated with SIRT1 inhibitor Sirtinol (50uM) for 12hr and cell lysis were separated on an 8% SDS gel and analyzed with anti-SIRT1, anti-MMP2 and anti-β-actin antibodies. B. SIRT1 knockdown decreases MMP2 expression. Equal amounts of cell lysates from LNCaP and PC3 cells with stably SIRT1 knockdown (siSIRT1) or vector control (siVector) were analyzed by immunoblot analysis with antibodies against SIRT1, MMP2 and β-actin. C. SIRT1 knockdown has no effect on MMP2 mRNA abundance. The mRNA levels of MMP2 were measured by quantitative RT-PCR analysis of 5 µg RNA extracted from SIRT1 siRNA (siSIRT1) or control siRNA (siVector) LNCaP and PC3 cells. The mRNA levels of MMP2 are expressed relative to β-actin transcripts. Each experiment was performed in triplicate and repeated three times. The error bars represent the SEM.
In order to further study the mechanism through which SIRT1 regulates MMP2 expression at the transcriptional or posttranslational modification level, we first performed RT-PCR to determine the regulation of SIRT1 on MMP2 at the mRNA level. The results show that there is no significant change in mRNA levels compared to RNAi vector transfected cells or SIRT1 knockdown cells (Figure 1C). This suggests that SIRT1 most likely regulates MMP2 through post-translational modification.
SIRT1 knockdown or inhibition decreases MMP2 protein stability
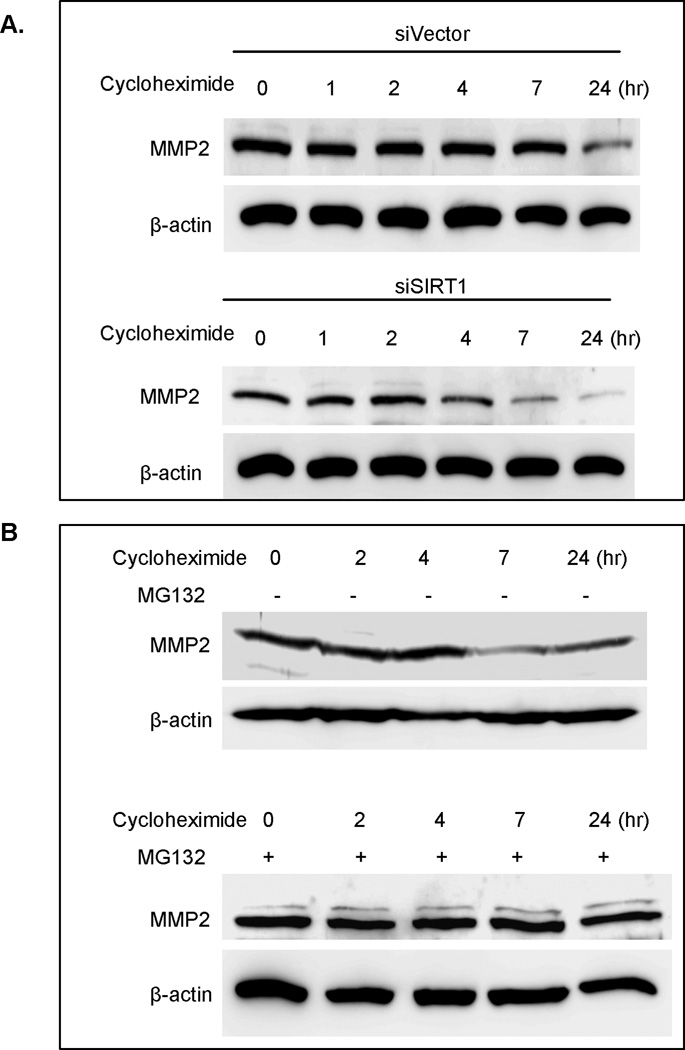
After finding that SIRT1 regulates MMP2 expression but has no effect on MMP2 mRNA expression, we sought to further determine how SIRT1 regulates MMP2 expression at posttranslational modification by studying if SIRT1 inhibition changes MMP2 protein stability. The RNAi vector- or SIRT1 RNAi-infected prostate cancer cell lines were treated with cycloheximide (CHX) to inhibit protein synthesis. The protein level was determined at a series of time points. The results showed that SIRT1 knockdown results in MMP2 degradation starting at four hours and decreasing through seven and twenty-four hours after CHX treatment (Figure 2A), while the siVector infected PC3 cells showed reduced MMP2 levels starting at twenty-four hours after the CHX treatment (Figure 2A). These results indicate that SIRT1 knockdown reduced the MMP2 expression by decreasing MMP2 protein stability.
Figure 2.
SIRT1 regulates MMP2 stability through the ubiquitin-proteasome pathway. A. RNAi vector- (upper panel) or SIRT1 RNAi- (lower panel) transfected LNCaP cells were treated with cycloheximide (10µg/ml). The cell extracts were prepared after treatment with cycloheximide for 0, 1, 2, 4, 7, and 24 hr and equal amounts of cell extracts were separated by SDS gel electrophoresis. The immunoblot was performed with anti-MMP2 and anti-β-actin antibodies. B. The SIRT1 RNAi-transfected LNCaP cells were pretreated with vehicle (upper panel) or MG132 for 1hr (lower panel), then treated with cycloheximide (10µg/ml) for 0, 2, 4, 7, and 24 hr and the immunoblot was performed with anti-MMP2 and anti-β-actin antibodies.
It has been shown that the ubiquitin-proteasome pathway plays an important role in MMP2 degradation. After finding that SIRT1 regulates MMP2 stability, we further studied whether the SIRT1-mediated change in MMP2 stability is regulated through the ubiquitin-proteasome pathway. We pretreated the SIRT1 knockdown cells with MG132, a proteosomal inhibitor, then treated the cells with protein synthesis inhibitor cycloheximide to determine whether proteosomal inhibition reduces SIRT1 knockdown-induced MMP2 instability. Our results show that inhibition of the proteosomal pathway by MG132 blocks SIRT1 knockdown-mediated MMP2 degradation (Figure 2B). This result suggests a mechanism in which SIRT1 knockdown-induced MMP2 instability is regulated through the ubiquitin-proteosome pathway.
SIRT1 associates with and deacetylates MMP2, and SIRT1 deacetylase activity is required for regulating MMP2 expression
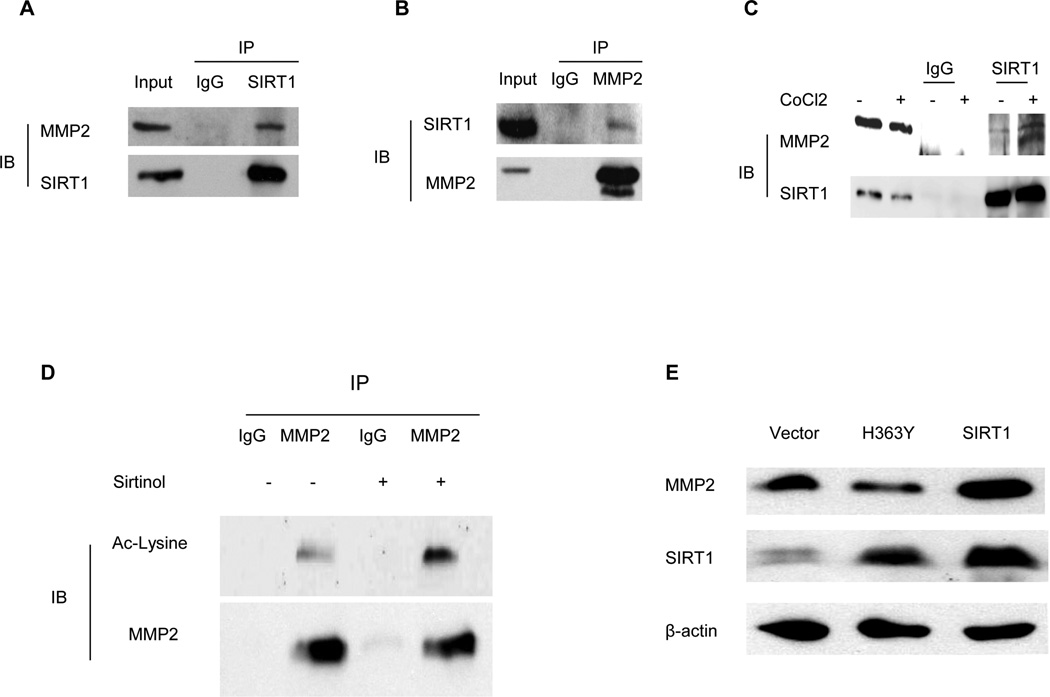
As a protein deacetylase, SIRT1 regulates many protein functions by associating with and deacetylating them (13). In order to study if SIRT1 plays a direct role in MMP2 stability, we studied whether SIRT1 could associate and deacetylate MMP2. First, we performed a co-IP experiment with an anti-SIRT1 antibody in LNCaP and PC3 cells to determine if SIRT1 associates with MMP2. The results showed that endogenous SIRT1 and MMP2 associate with each other in prostate cancer cells (Figure 3A). We also performed a co-IP experiment with anti-MMP2 antibody and a similar result was seen for the association of SIRT1 and MMP2 (Figure 3B). As prostate cancer is often highly hypoxic which are related to poor prognosis, we therefore further studied whether hypoxia condition promotes SIRT1 and MMP2 interaction. We treated the LNCaP cells with CoCl2 to induce the hypoxia condition and performed co-immunoprecipitation with a SIRT1 antibody. We found that CoCl2 treatment increases the interaction of SIRT1 with MMP2 (Figure 3C). Upon finding this association, we sought to further determine if MMP2 could be deacetylated by SIRT1. MMP2 was pulled down by an anti-MMP2 antibody from control or SIRT1 inhibitor treated prostate cancer cells and the acetylation level of MMP2 was determined by performing immunoblot with an anti-acetylated-lysine antibody. The results showed that acetylated MMP2 levels were increased after SIRT1 deacetylase activity inhibition (Figure 3D), suggesting that MMP2 can be deacetylated by SIRT1. To further determine if SIRT1 regulates MMP2 expression through its deacetylase activity, we employed a dominant-negative mutant that abolishes the deacetylase activity of SIRT1 (SIRT1H363Y - mutation of His363 to Tyr). Transfection of SIRT1 H363Y plasmid in prostate cancer cells failed to upregulate MMP2 expression, whereas transfection of wt-SIRT1 in parallel upregulated MMP2 expression (Figure 3E). These results establish that the upregulation of MMP2 expression by SIRT1 requires its deacetylase activity.
Figure 3.
SIRT1 associates with and deacetylases MMP2 and SIRT1 deacetylase activity is required for MMP2 expression regulation. A. Co- immunoprecipitation was performed using anti-SIRT1 antibodies and IgG as a control with LNCaP cell lysates and immunoblot was performed with anti-MMP2 and anti-SIRT1 antibodies respectively. B. Co-immunoprecipitation was performed using anti-MMP2 antibodies and IgG as a control and immunoblot was performed with anti-SIRT1 and anti-MMP2 antibodies. C. The LNCaP cells were treated with or without CoCl2 at 200 uM for 5 hours, then co- immunoprecipitation was performed using SIRT1 antibody and immunoblot was performed with anti-MMP2 and anti-SIRT1. D. Co-immunoprecipitation was performed with anti-MMP2 antibodies or IgG as a control using equal amounts of LNCaP cell lysates from vehicle treated or SIRT1 inhibitor Sirtinol treated cells. Immunoblot was performed with acetylated lysine antibodies to compare the acetylation level of MMP2 in response to SIRT1 inhibitor treatment. The membrane was erased and re-blotted with MMP2 to determine the co-immunoprecipitation efficiency of MMP2 antibody. E. SIRT1 deacetylase activity is required for SIRT1 effects on MMP2 expression. LNCaP cells was transfected with a SIRT1 expression vector (SIRT1), or a dominant-negative SIRT1 vector (H363Y), or an empty vector (Vector). At 48 hr after the transfection the cells were harvested for immunoblot analysis with anti-MMP2 to determine the level of MMP2 expression, and anti-His tag to determine the expression wt SIRT1 and H363Y SIRT1.
SIRT1 knockdown or inhibition reduces MMP2 zymographic activity and prostate cancer cell invasion
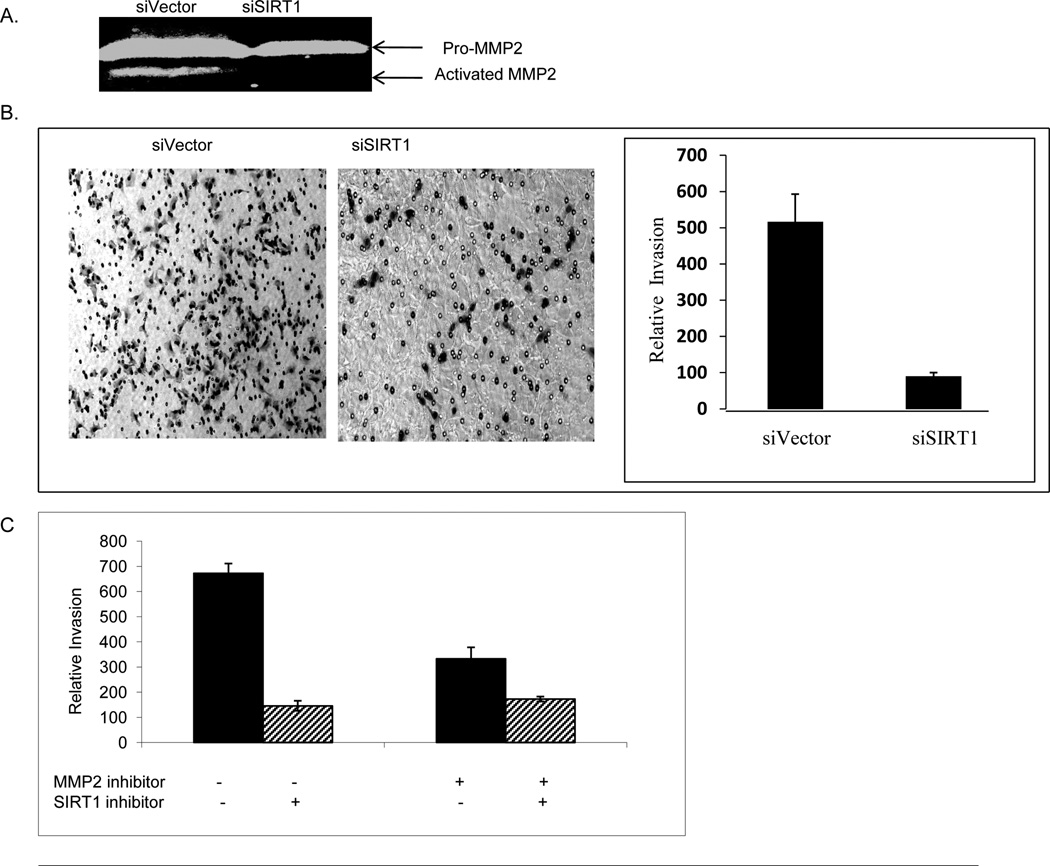
As functional MMP2 needs to be secreted outside of cells in order to degrade the ECM, to determine if the MMP2 protein level or stability has a functional effect on MMP2 activity, we performed a zymography analysis to detect gelatinase activity of MMPs using in cell culture supernatants. We compared the zymographic activity between the supernatants from the vector control and SIRT1 knockdown cells, and results show that supernatants from control cell cultures had higher levels of gelatinase activity than supernatants from knockdown cell cultures (Figure 4A).
Figure 4.
SIRT1 inhibition reduces MMP2 activity and invasion activity. A. SIRT1 knockdown decreases zymographic activity of MMP2. PC3 (2×106) cells were seeded in 10 cm plate with 5 ml medium and incubated for 24 hr. Culture supernatant was harvested and diluted 1:1 with non-reducing Zymogram Sample Buffer. 10 ug of protein from culture supernatant was loaded onto 7.5% SDS-polyacrylamide gels containing 2 mg/ml gelatin. The MMP2 zymographic activity was analyzed. B. SIRT1 knockdown decreases prostate cancer cell invasion. 2.5×104 siVector or siSIRT1 PC3 cells in 500ul were loaded onto matrigel in transwells coated with matrigel. After 24 hr of culture at 37°C, the transwells were removed from 24-well plates and stained with Diff-Quick solution. The invading cells were counted for quantity analysis and the representative imaging was taken under a light microscope. Each experiment was performed in triplicate and repeated three times. The error bars represent the SEM. C. MMP2 inhibition impairs SIRT1 inhibition-induced invasion suppression. PC3 cells were pretreated with vehicle or MMP2 inhibitor (MMP2 inhibitor I, 1.7uM, Millipore) for 1 hr, then the cells were treated with SIRT1 inhibitor Sirtinol (100uM) for 24 hr. 2.5×104 cells in 500 ul were loaded onto transwells coated with matrigel. Cell invasion was analyzed as described in Figure 4B.
Next, we studied the effects of SIRT1 knockdown on prostate cancer invasion by using a trans-well chamber assay system coated matrigel on the top of an ECM-like membrane that prevents the movement of non-invasive cells. The results show that SIRT1 knockdown PC3 cells exhibited a 5-fold decrease in invasion compared to in control cells after a 24-hour incubation period (Figure 4B). These results indicate that inhibiting SIRT1 significantly lowers the ability of cells to invade through a matrigel.
In order to further study whether SIRT1 mediated MMP2 expression and activity is responsible for SIRT1 mediated invasion, we pretreated the cells with MMP2 inhibitor followed by SIRT1 inhibitor to determine whether MMP2 inhibition impairs the suppression effects on invasion by SIRT1 inhibition. The result shows that while SIRT1 inhibitor induces 80% inhibition of invasion in vehicle pretreated cells, SIRT1 inhibitor induces only 33% inhibition of invasion in MMP2 inhibitor pretreated cells (Figure 4C). This result indicates that SIRT inhibition-induced suppression in invasion is greatly impaired by MMP2 inhibition, suggesting an important functional role of MMP2 as a downstream target of SIRT1 in invasion regulation.
SIRT1 is highly expressed in advanced prostate cancer tissues
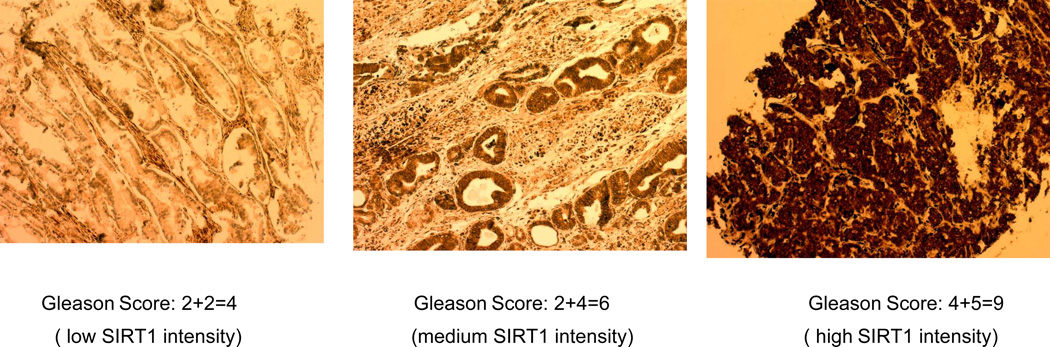
It has been shown that SIRT1 is overexpressed in prostate cancer tissues compared to in normal prostate tissues (16). In order to study whether advanced prostate cancer tissue has a higher SIRT1 expression level, we performed a SIRT1 immunohistochemistry assay using prostate tissues arrays and analyzed 65 cases of prostate cancer with various Gleason scores from 4–9, 6 normal tissues, and 24 benign prostatic hyperplasia (BPH) tissues. Based on the intensity of SIRT1 staining, we divided the tissues into three groups as having low, medium, and high SIRT1 expression, and our results show that SIRT1 expression is lower in normal and BPH tissues, while SIRT1 is highly expressed in advanced prostate cancer tissues (Table 1 & Figure 5). We found that 71.8 % of poorly differentiated tumors (Gleason score 8–9) showed high SIRT1 expression, while 23.5% of moderately differentiated (Gleason score, 6–7) and 0 % of tumors at Gleason score 4–5 showed high SIRT1 expression (Figure 5 and Table1). These results indicate that SIRT1 expression levels are highly up-regulated in advanced prostate cancer and SIRT1 expression level is significantly correlated with tumor Gleason score (Fisher’s test, P = 0.01). All these results suggest an important role of SIRT1 in prostate cancer progression.
Table1.
The correlation of SIRT1 expression levels with Gleason score of prostate cancer tissues. The tissue arrays containing 65 cancer, 6 normal, and 24 BPH prostate tissues from Cybrdi (CC19-11-007; CC-19-005 and CC19-01) were examined via standard IHC methods (DAB kit, DAB150, Upstate) using SIRT1 antibody (1:2000, Upstate). The correlations between SIRT1 expression and tumor Gleason score were evaluated by Fisher’s exact test (p<0.01).
| Tissue Type | Tissue Number | SIRT1 level | ||
|---|---|---|---|---|
| Low N (%) |
Medium N (%) |
High N (%) |
||
| Normal | 6 | 4 (66.6%) | 2 (33.3%) | 0 |
| BPH | 24 | 18 (75.0%) | 6 (25%) | 0 |
| PCA | 65 | 8 (12.3%) | 25 (38.5%) | 32 (49.2%) |
| Gleason Score | ||||
| 4–5 | 9 | 3 (33.3%) | 6 (66.7%) | 0 |
| 6–7 | 17 | 2 (11.8%) | 11 (64.7%) | 4 (23.5%) |
| 8–9 | 39 | 3 (7.7%) | 8 (20.5%) | 28 (71.8%) |
Figure 5.
SIRT1 is highly expressed in advanced prostate cancer tissue. The prostate cancer tissues array from Cybrdi were examined via standard IHC methods (DAB kit, DAB150, Upstate) using SIRT1 antibody (1:2000, Upstate). This figure is the representative of low, medium and high SIRT1 intensity in prostate cancer tissues with Gleason score 4, 6 and 9.
Discussion
MMP2, is also known as gelatinase A or type IV collagenase, is the most widely expressed of all the MMPs and its expression has been shown associated with poor prognosis of prostate cancers (9). Although MMP2 expression has been shown regulated at both transcriptional and posttranscriptional levels, the posttranslational modification especially for deacetylation regulation has not been reported (11, 12). In this study we show that SIRT1 associates with and deacetylates MMP2, and that SIRT1 regulates MMP2 expression through control MMP2 protein stability in the proteosomal pathway. In addition, we show that SIRT1 deacetylation activity is required for MMP2 expression. Thus, we demonstrated a novel mechanism in that MMP2 expression can be regulated at a posttranslational level through deacetylation by SIRT1.
Acetylation and deacetylation are important posttranslational modifications that regulate protein expression and functions. As a histone and protein deacetylase, SIRT1 is involved in many physiological processes due to its ability to deacetylate histone and non-histone substrates (13, 17). SIRT1 has been shown as a nuclear protein that regulates histone deacetylation and transcription factor function within the nucleus (18), but recent evidence has shown that SIRT1 is also localized in the cytoplasm (19, 20). Moreover, SIRT1 is predominantly localized in the cytoplasm in cancer cells while SIRT1 is mainly localized in nucleus in normal cells (21, 22). However, the functional significance of this cytoplasm localization of SIRT1 in cancer cells remains unknown. As MMP2 is a cytoplasmic protein, our findings of the association and deacetylation of MMP2 by SIRT1 suggest an important function of cytoplasmic SIRT1 in regulating MMP2 protein stability and cancer cells invasion. In addition, we found that hypoxia condition promotes SIRT1 and MMP2 interaction. As prostate cancer is often highly hypoxic and related to poor prognosis, our finding suggests that SIRT1 may play an important role in hypoxia-induced prostate cancer progression through deacetylation of MMP2. In a future study, the specific acetylation sites of MMP2 by SIRT1 and their functional role in MMP2 expression, activity and in hypoxia-induced prostate cancer progression will be further elucidated.
MMP2 expression in tissue or serum has been shown positively correlated with poor prognosis and lower survival in prostate cancer (10, 23), and our findings that SIRT1 knockdown or inhibition downregulates MMP2 expression and reduces prostate cancer cell invasion suggest that SIRT1 may play an important role in prostate cancer progression through regulating MMP2. Recently, SIRT1 has been shown to induce epithelial to mesenchymal transition (24), increase angiogenesis (25), and deacetylate cortactin to regulate cell migration (26). In addition, high levels of SIRT1 are found in various cancers, including prostate cancer (16, 27), and SIRT1 expression level is associated with the poor prognosis of gastric carcinomas, B-cell lymphoma, breast cancer, and lung cancer (28–31). In this study our data shows that SIRT1 expression level is positively correlated with the Gleason score of prostate cancer. All of these findings suggest that SIRT1 may be a critical regulator in cancer progression, and inhibition of SIRT1 could be a potential strategy in advanced prostate cancer treatment.
Conclusion
This study indicates that SIRT1 is an important regulator of MMP2 expression and activity, by a mechanism in which SIRT1 deacetylates and stabilizes MMP2 through the proteosomal pathway. Thus we identified a novel mechanism in the regulation of MMP2 expression at the posttranslational level. Our findings that SIRT1 is overexpressed in advanced prostate cancer and SIRT1 knockdown decreases MMP2 and prostate cancer cell invasion, suggest that SIRT1 may play a critical role in prostate cancer progression and that SIRT1 inhibition may be a potential therapeutic target for prostate cancer.
Acknowledgments
We sincerely thank Dr. F. Picard (Laval University, Canada) for reagents. We greatly appreciate the support of Lora Forman (Boston University). This work was supported by grants from the National Cancer Institute (1R21CA141036) (Y.D.), the Clinical and Translational Science Institute award from the NIH (UL1-RR025771) (Y.D.), the National Cancer Institute (CA101992-05A1) (D.V.F.) and Karin Grunebaum Cancer Research Foundation (Y.D.).
References
- 1.Jemal A, Siegel R, Ward E, Hao YP, Xu JQ, Thun MJ. Cancer Statistics, 2009. CA Cancer Journal for Clinicians. 2009;59(4):225–249. doi: 10.3322/caac.20006. [DOI] [PubMed] [Google Scholar]
- 2.Goel HL, Li J, Kogan S, Languino LR. Integrins in prostate cancer progression. Endocrine-related cancer. 2008;15(3):657–664. doi: 10.1677/ERC-08-0019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Scher HI, Heller G. Clinical states in prostate cancer: Toward a dynamic model of disease progression. Urology. 2000;55(3):323–327. doi: 10.1016/s0090-4295(99)00471-9. [DOI] [PubMed] [Google Scholar]
- 4.Dehm SM, Tindall DJ. Molecular regulation of androgen action in prostate cancer. Journal of Cellular Biochemistry. 2006;99(2):333–344. doi: 10.1002/jcb.20794. [DOI] [PubMed] [Google Scholar]
- 5.Busby JE, Kim SJ, Yazici S, Nakamura T, Kim JS, He J, Maya M, Wang X, Do KA, Fan D, Fidler IJ. Therapy of multidrug resistant human prostate tumors in the prostate of nude mice by simultaneous targeting of the epidermal growth factor receptor and vascular endothelial growth factor receptor on tumor-associated endothelial cells. The Prostate. 2006;66(16):1788–1798. doi: 10.1002/pros.20519. [DOI] [PubMed] [Google Scholar]
- 6.Mehlen P, Puisieux A. Metastasis: a question of life or death. Nature Reviews Cancer. 2006;6(6):449–458. doi: 10.1038/nrc1886. [DOI] [PubMed] [Google Scholar]
- 7.Mimeault M, Batra SK. Recent advances on multiple tumorigenic cascades involved in prostatic cancer progression and targeting therapies. Carcinogenesis. 2006;27(1):1–22. doi: 10.1093/carcin/bgi229. [DOI] [PubMed] [Google Scholar]
- 8.Noel A, Jost M, Maquoi E. Matrix metalloproteinases at cancer tumor-host interface. Seminars in Cell & Developmental Biology. 2008;19(1):52–60. doi: 10.1016/j.semcdb.2007.05.011. [DOI] [PubMed] [Google Scholar]
- 9.Sternlicht MD, Werb Z. How matrix metalloproteinases regulate cell behavior. Annual review of cell and developmental biology. 2001;17:463–516. doi: 10.1146/annurev.cellbio.17.1.463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vandewalle C, Van Roy F, Berx G. The role of the ZEB family of transcription factors in development and disease. Cell Mol Life Sci. 2009;66(5):773–787. doi: 10.1007/s00018-008-8465-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chakraborti S, Mandal M, Das S, Mandal A, Chakraborti T. Regulation of matrix metalloproteinases: an overview. Molecular and cellular biochemistry. 2003;253(1–2):269–285. doi: 10.1023/a:1026028303196. [DOI] [PubMed] [Google Scholar]
- 12.Sariahmetoglu M, Crawford BD, Leon H, Sawicka J, Li L, Ballermann BJ, Holmes C, Berthiaume LG, Holt A, Sawicki G, Schulz R. Regulation of matrix metalloproteinase-2 (MMP-2) activity by phosphorylation. Faseb J. 2007;21(10):2486–2495. doi: 10.1096/fj.06-7938com. [DOI] [PubMed] [Google Scholar]
- 13.Liu T, Liu PY, Marshall GM. The critical role of the class III histone deacetylase SIRT1 in cancer. Cancer research. 2009;69(5):1702–1705. doi: 10.1158/0008-5472.CAN-08-3365. [DOI] [PubMed] [Google Scholar]
- 14.Yamamoto H, Schoonjans K, Auwerx J. Sirtuin functions in health and disease. Molecular Endocrinology. 2007;21(8):1745–1755. doi: 10.1210/me.2007-0079. [DOI] [PubMed] [Google Scholar]
- 15.Oviedo-Orta E, Bermudez-Fajardo A, Karanam S, Benbow U, Newby AC. Comparison of MMP-2 and MMP-9 secretion from T helper 0, 1 and 2 lymphocytes alone and in coculture with macrophages. Immunology. 2008;124(1):42–50. doi: 10.1111/j.1365-2567.2007.02728.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Huffman DM, Grizzle WE, Bamman MM, Kim JS, Eltoum IA, Elgavish A, Nagy TR. SIRT1 is significantly elevated in mouse and human prostate cancer. Cancer research. 2007;67(14):6612–6618. doi: 10.1158/0008-5472.CAN-07-0085. [DOI] [PubMed] [Google Scholar]
- 17.Dai Y, Faller DV. Transcription Regulation by Class III Histone Deacetylases (HDACs)-Sirtuins. Transl Oncogenomics. 2008;3:53–65. doi: 10.4137/tog.s483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Michishita E, Park JY, Burneskis JM, Barrett JC, Horikawa I. Evolutionarily conserved and nonconserved cellular localizations and functions of human SIRT proteins. Molecular biology of the cell. 2005;16(10):4623–4635. doi: 10.1091/mbc.E05-01-0033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhang J. The direct involvement of SirT1 in insulin-induced insulin receptor substrate-2 tyrosine phosphorylation. The Journal of biological chemistry. 2007;282(47):34356–34364. doi: 10.1074/jbc.M706644200. [DOI] [PubMed] [Google Scholar]
- 20.Moynihan KA, Grimm AA, Plueger MM, Bernal-Mizrachi E, Ford E, Cras-Meneur C, Permutt MA, Imai S. Increased dosage of mammalian Sir2 in pancreatic beta cells enhances glucose-stimulated insulin secretion in mice. Cell metabolism. 2005;2(2):105–117. doi: 10.1016/j.cmet.2005.07.001. [DOI] [PubMed] [Google Scholar]
- 21.Byles V, Chmilewski LK, Wang J, Zhu L, Forman LW, Faller DV, Dai Y. Aberrant cytoplasm localization and protein stability of SIRT1 is regulated by PI3K/IGF-1R signaling in human cancer cells. International journal of biological sciences. 6(6):599–612. doi: 10.7150/ijbs.6.599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Stunkel W, Peh BK, Tan YC, Nayagam VM, Wang X, Salto-Tellez M, Ni B, Entzeroth M, Wood J. Function of the SIRT1 protein deacetylase in cancer. Biotechnology journal. 2007;2(11):1360–1368. doi: 10.1002/biot.200700087. [DOI] [PubMed] [Google Scholar]
- 23.Jezierska A, Motyl T. Matrix Metalloproteinase-2 involvement in breast cancer progression: A mini-review. Medical Science Monitor. 2009;15(2):RA32–RA40. [PubMed] [Google Scholar]
- 24.Byles V, Zhu L, Lovaas JD, Chmilewski LK, Wang J, Faller DV, Dai Y. SIRT1 induces EMT by cooperating with EMT transcription factors and enhances prostate cancer cell migration and metastasis. Oncogene. doi: 10.1038/onc.2011.612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Potente M, Ghaeni L, Baldessari D, Mostoslavsky R, Rossig L, Dequiedt F, Haendeler J, Mione M, Dejana E, Alt FW, Zeiher AM, Dimmeler S. SIRT1 controls endothelial angiogenic functions during vascular growth. Genes Dev. 2007;21(20):2644–2658. doi: 10.1101/gad.435107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhang Y, Zhang M, Dong H, Yong S, Li X, Olashaw N, Kruk PA, Cheng JQ, Bai W, Chen J, Nicosia SV, Zhang X. Deacetylation of cortactin by SIRT1 promotes cell migration. Oncogene. 2009;28(3):445–460. doi: 10.1038/onc.2008.388. [DOI] [PubMed] [Google Scholar]
- 27.Kwon HS, Ott M. The ups and downs of SIRT1. Trends Biochem Sci. 2008;33(11):517–525. doi: 10.1016/j.tibs.2008.08.001. [DOI] [PubMed] [Google Scholar]
- 28.Cha EJ, Noh SJ, Kwon KS, Kim CY, Park BH, Park HS, Lee H, Chung MJ, Kang MJ, Lee DG, Moon WS, Jang KY. Expression of DBC1 and SIRT1 is associated with poor prognosis of gastric carcinoma. Clin Cancer Res. 2009;15(13):4453–4459. doi: 10.1158/1078-0432.CCR-08-3329. [DOI] [PubMed] [Google Scholar]
- 29.Jang KY, Hwang SH, Kwon KS, Kim KR, Choi HN, Lee NR, Kwak JY, Park BH, Park HS, Chung MJ, Kang MJ, Lee DG, Kim HS, Shim H, Moon WS. SIRT1 expression is associated with poor prognosis of diffuse large B-cell lymphoma. Am J Surg Pathol. 2008;32(10):1523–1531. doi: 10.1097/PAS.0b013e31816b6478. [DOI] [PubMed] [Google Scholar]
- 30.Lee H, Kim KR, Noh SJ, Park HS, Kwon KS, Park BH, Jung SH, Youn HJ, Lee BK, Chung MJ, Koh DH, Moon WS, Jang KY. Expression of DBC1 and SIRT1 is associated with poor prognosis for breast carcinoma. Hum Pathol. 42(2):204–213. doi: 10.1016/j.humpath.2010.05.023. [DOI] [PubMed] [Google Scholar]
- 31.Tseng RC, Lee CC, Hsu HS, Tzao C, Wang YC. Distinct HIC1-SIRT1-p53 loop deregulation in lung squamous carcinoma and adenocarcinoma patients. Neoplasia. 2009;11(8):763–770. doi: 10.1593/neo.09470. [DOI] [PMC free article] [PubMed] [Google Scholar]