Abstract
Studies of mouse mammary tumor virus (MMTV) have been impeded by the lack of an in vitro infectivity assay. We have developed a rapid, quantitative in vitro assay for MMTV infectivity based on the detection of positively staining foci by immunoperoxidase. This assay and a 50% end-point titration of MMTV infectivity gave identical virus titers. Infection of a rat hepatoma cell line, a feline kidney cell line, and a normal murine mammary gland cell line by virus from the mouse mammary tumor GR3A cell line was linear with respect to virus concentration. The infectious titers obtained in both homologous and heterologous cell lines were not significantly different, demonstrating a lack of host range specificity. Virus infectivity was inactivated by heating at 55 degrees C and by ultraviolet irradiation. Rabbit anti-MMTV serum neutralized the infectivity with a 50% neutralization end point of 1:5000. Applications of this assay to the study of the immunological, biological, and biochemical characteristics of MMTV are discussed.
Full text
PDF


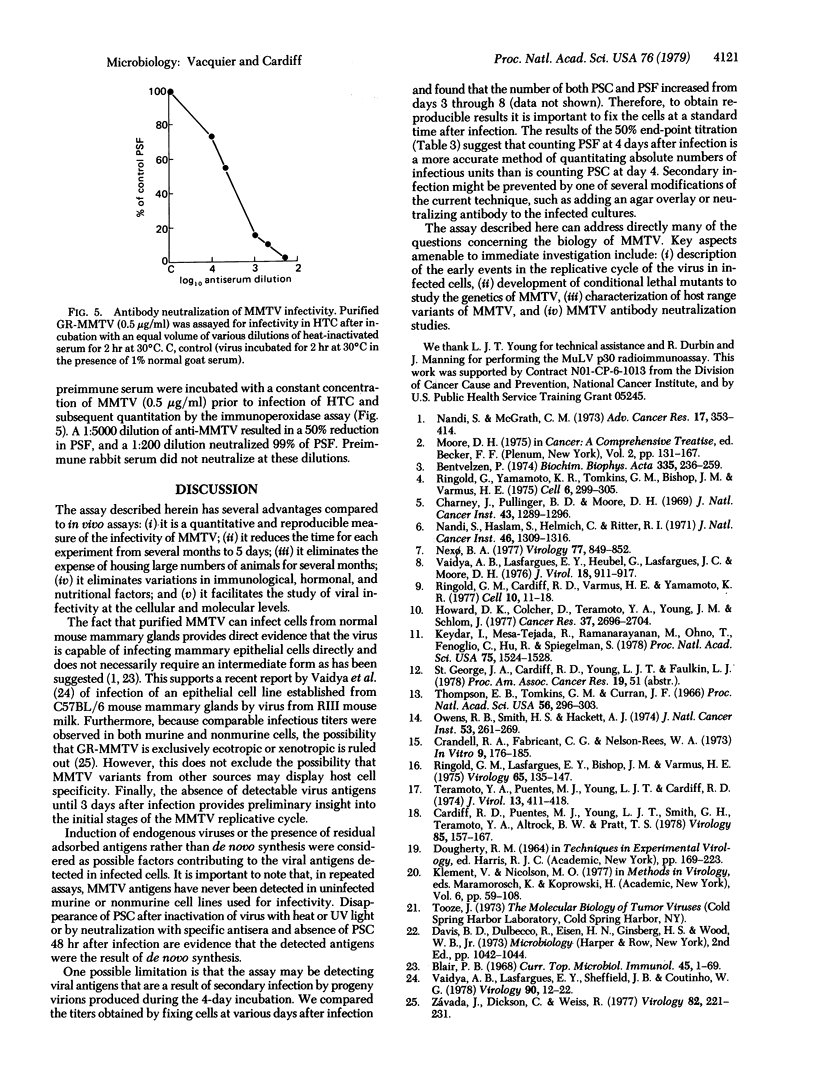
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bentvelzen P. Host-virus interactions in murine mammary carcinogenesis. Biochim Biophys Acta. 1974 Dec 31;355(3-4):236–259. doi: 10.1016/0304-419x(74)90012-2. [DOI] [PubMed] [Google Scholar]
- Blair P. B. The mammary tumor virus (MTV). Curr Top Microbiol Immunol. 1968;45:1–69. doi: 10.1007/978-3-642-50109-8_1. [DOI] [PubMed] [Google Scholar]
- Cardiff R. D., Puentes M. J., Young L. J., Smith G. H., Teramoto Y. A., Altrock B. W., Pratt T. S. Serological and biochemical characterization of the mouse mammary tumor virus with localization of p10. Virology. 1978 Mar;85(1):157–167. doi: 10.1016/0042-6822(78)90420-8. [DOI] [PubMed] [Google Scholar]
- Charney J., Pullinger B. D., Moore D. H. Development of an infectivity assay for mouse mammary-tumor virus. J Natl Cancer Inst. 1969 Dec;43(6):1289–1296. [PubMed] [Google Scholar]
- Crandell R. A., Fabricant C. G., Nelson-Rees W. A. Development, characterization, and viral susceptibility of a feline (Felis catus) renal cell line (CRFK). In Vitro. 1973 Nov-Dec;9(3):176–185. doi: 10.1007/BF02618435. [DOI] [PubMed] [Google Scholar]
- Howard D. K., Colcher D., Teramoto Y. A., Young J. M., Schlom J. Characterization of mouse mammary tumor viruses propagated in heterologous cells. Cancer Res. 1977 Aug;37(8 Pt 1):2696–2704. [PubMed] [Google Scholar]
- Keydar I., Mesa-Tejada R., Ramanarayanan M., Ohno T., Fenoglio C., Hu R., Spiegelman S. Detection of viral proteins in mouse mammary tumors by immunoperoxidase staining of paraffin sections. Proc Natl Acad Sci U S A. 1978 Mar;75(3):1524–1528. doi: 10.1073/pnas.75.3.1524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nandi S., Haslam S., Helmich C., Ritter R. I. Nodule bioassay of the mouse mammary tumor virus: modifications and quantitative considerations. J Natl Cancer Inst. 1971 Jun;46(6):1309–1315. [PubMed] [Google Scholar]
- Nexo B. A. A plaque assay for murine leukemia virus using enzyme-coupled antibodies. Virology. 1977 Apr;77(2):849–852. doi: 10.1016/0042-6822(77)90504-9. [DOI] [PubMed] [Google Scholar]
- Owens R. B., Smith H. S., Hackett A. J. Epithelial cell cultures from normal glandular tissue of mice. J Natl Cancer Inst. 1974 Jul;53(1):261–269. doi: 10.1093/jnci/53.1.261. [DOI] [PubMed] [Google Scholar]
- Ringold G. M., Cardiff R. D., Varmus H. E., Yamamoto K. R. Infection of cultured rat hepatoma cells by mouse mammary tumor virus. Cell. 1977 Jan;10(1):11–18. doi: 10.1016/0092-8674(77)90134-9. [DOI] [PubMed] [Google Scholar]
- Ringold G. M., Yamamoto K. R., Tomkins G. M., Bishop M., Varmus H. E. Dexamethasone-mediated induction of mouse mammary tumor virus RNA: a system for studying glucocorticoid action. Cell. 1975 Nov;6(3):299–305. doi: 10.1016/0092-8674(75)90181-6. [DOI] [PubMed] [Google Scholar]
- Ringold G., Lasfargues E. Y., Bishop J. M., Varmus H. E. Production of mouse mammary tumor virus by cultured cells in the absence and presence of hormones: assay by molecular hybridization. Virology. 1975 May;65(1):135–147. doi: 10.1016/0042-6822(75)90014-8. [DOI] [PubMed] [Google Scholar]
- Teramoto Y. A., Puentes M. J., Young L. J., Cardiff R. D. Structure of the mouse mammary tumor virus: polypeptides and glycoproteins. J Virol. 1974 Feb;13(2):411–418. doi: 10.1128/jvi.13.2.411-418.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson E. B., Tomkins G. M., Curran J. F. Induction of tyrosine alpha-ketoglutarate transaminase by steroid hormones in a newly established tissue culture cell line. Proc Natl Acad Sci U S A. 1966 Jul;56(1):296–303. doi: 10.1073/pnas.56.1.296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaidya A. B., Lasfargues E. Y., Heubel G., Lasfargues J. C., Moore D. H. Murine mammary tumor virus: characterization of infection of nonmurine cells. J Virol. 1976 Jun;18(3):911–917. doi: 10.1128/jvi.18.3.911-917.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaidya A. B., Lasfargues E. Y., Sheffield J. B., Coutinho W. G. Murine mammary tumor virus (MuMTV) infection of an epithelial cell line established from C57BL/6 mouse mammary glands. Virology. 1978 Oct 1;90(1):12–22. doi: 10.1016/0042-6822(78)90328-8. [DOI] [PubMed] [Google Scholar]