Abstract
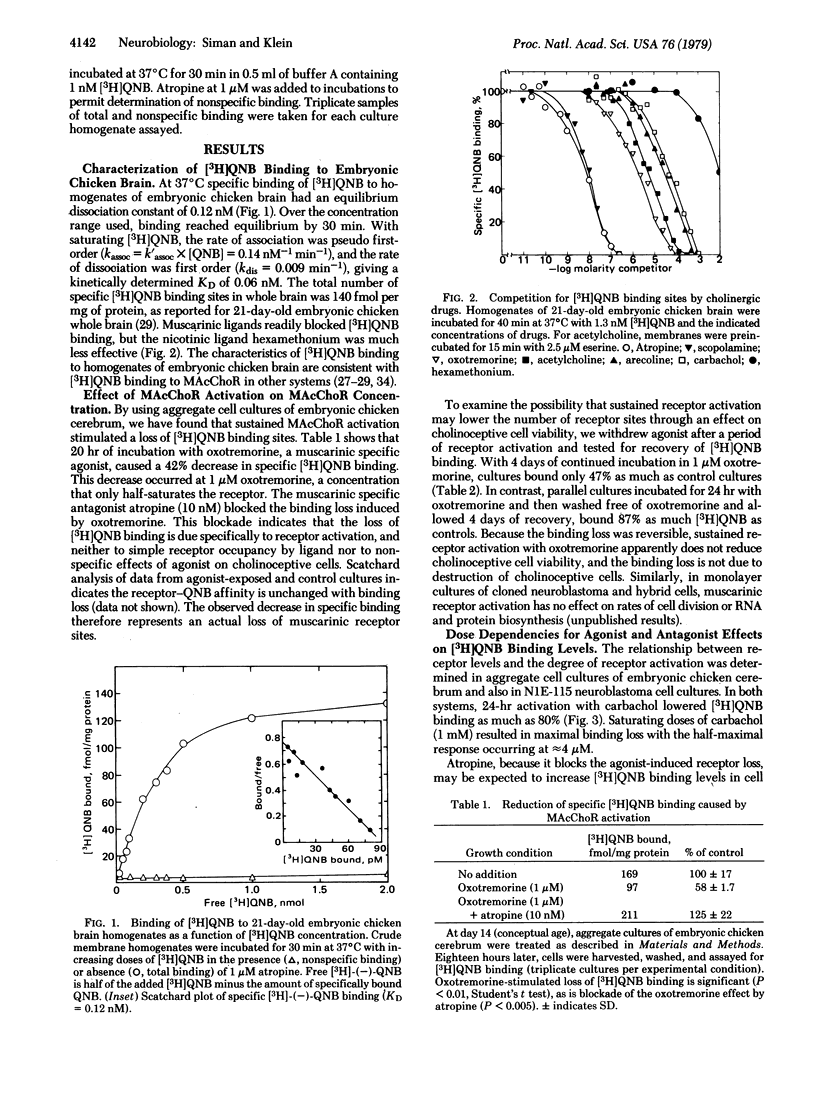
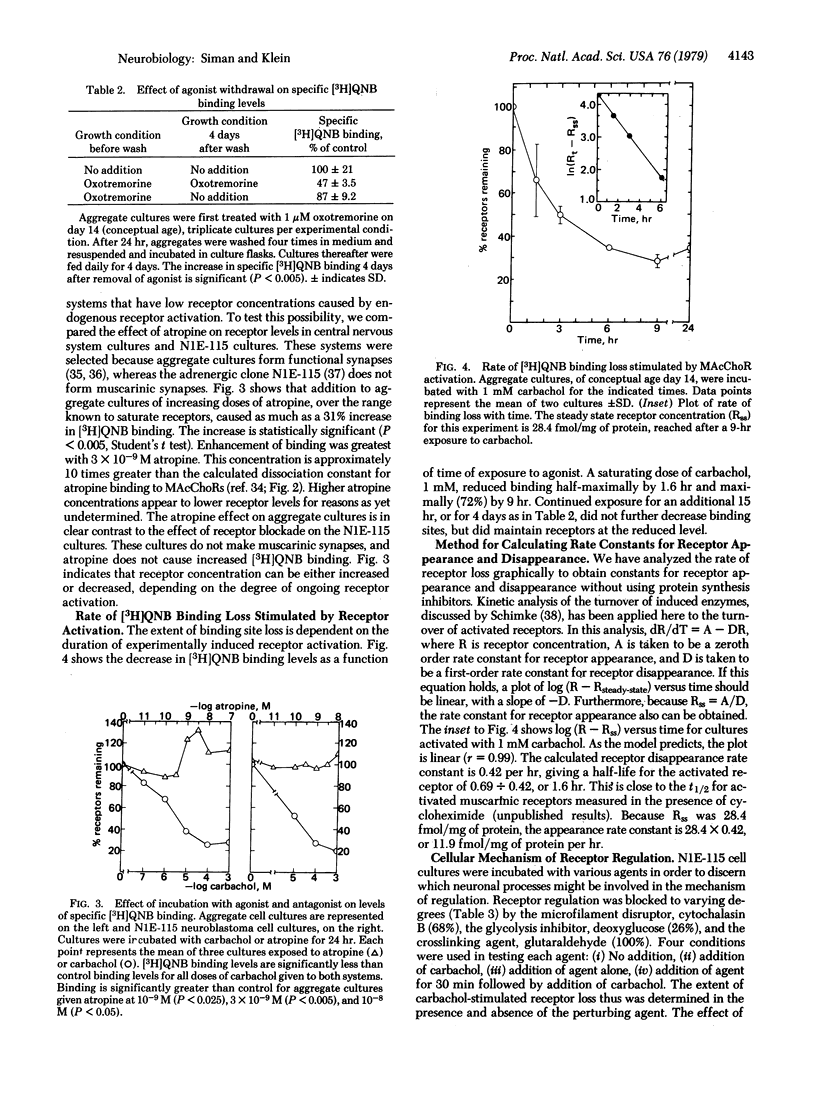
Muscarinic acetylcholine receptor activation induces a loss of muscarinic receptors from cultured neuroblastoma and embryonic chicken cerebrum cells. As measured by specific binding of [3H]quinuclidinyl benzilate, steady-state receptor concentrations decrease 75% in response to receptor-saturating concentrations of cholinomimetic drugs. Both the degree and duration of activation determine the extent of receptor loss. A method for analyzing receptor turnover, which does not rely upon protein synthesis inhibitors, shows that activated receptors have a half-life of 1.6 hr. The regulated rate of receptor disappearance begins as soon as activators are added, and the rate is maintained as long as activators are present. The receptor blocker atropine causes an increase in receptor levels in central nervous system cultures but has no effect on receptors in cultures of adrenergic neuroblastoma cells. Because spontaneous cholinergic activity is expected only in the central nervous system cultures, the increase likely reflects blockade of endogenous regulation. Cytochalasin B blocks receptor regulation, suggesting that regulation may be mediated by a process involving microfilaments.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Amano T., Richelson E., Nirenberg M. Neurotransmitter synthesis by neuroblastoma clones (neuroblast differentiation-cell culture-choline acetyltransferase-acetylcholinesterase-tyrosine hydroxylase-axons-dendrites). Proc Natl Acad Sci U S A. 1972 Jan;69(1):258–263. doi: 10.1073/pnas.69.1.258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anwyl R., Appel S. M., Narahashi T. Myasthenia gravis serum reduces acetylcholine sensitivity in cultured rat myotubes. Nature. 1977 May 19;267(5608):262–263. doi: 10.1038/267262a0. [DOI] [PubMed] [Google Scholar]
- Axline S. G., Reaven E. P. Inhibition of phagocytosis and plasma membrane mobility of the cultivated macrophage by cytochalasin B. Role of subplasmalemmal microfilaments. J Cell Biol. 1974 Sep;62(3):647–659. doi: 10.1083/jcb.62.3.647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bird S. J., Aghajanian G. K. Denervation supersensitivity in the cholinergic septo-hippocampal pathway: a microiontophoretic study. Brain Res. 1975 Dec 19;100(2):355–370. doi: 10.1016/0006-8993(75)90488-6. [DOI] [PubMed] [Google Scholar]
- Burgermeister W., Klein W. L., Nirenberg M., Witkop B. Comparative binding studies with cholinergic ligands and histrionicotoxin at muscarinic receptors of neural cell lines. Mol Pharmacol. 1978 Sep;14(5):751–767. [PubMed] [Google Scholar]
- Burt D. R., Creese I., Snyder S. H. Antischizophrenic drugs: chronic treatment elevates dopamine receptor binding in brain. Science. 1977 Apr 15;196(4287):326–328. doi: 10.1126/science.847477. [DOI] [PubMed] [Google Scholar]
- Carpenter G., Cohen S. 125I-labeled human epidermal growth factor. Binding, internalization, and degradation in human fibroblasts. J Cell Biol. 1976 Oct;71(1):159–171. doi: 10.1083/jcb.71.1.159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooper B., Handin R. I., Young L. H., Alexander R. W. Agonist regulation of the human platelet alpha-adrenergic receptor. Nature. 1978 Aug 17;274(5672):703–706. doi: 10.1038/274703a0. [DOI] [PubMed] [Google Scholar]
- Crain S. M., Bornstein M. B. Organotypic bioelectric activity in cultured reaggregates of dissociated rodent brain cells. Science. 1972 Apr 14;176(4031):182–184. doi: 10.1126/science.176.4031.182. [DOI] [PubMed] [Google Scholar]
- Deutsch J. A. The cholinergic synapse and the site of memory. Science. 1971 Nov 19;174(4011):788–794. doi: 10.1126/science.174.4011.788. [DOI] [PubMed] [Google Scholar]
- Enna S. J., Yamamura H. I., Snyder S. H. Development of muscarinic cholinergic and GABA receptor binding in chick embryo brain. Brain Res. 1976 Jan 9;101(1):177–183. doi: 10.1016/0006-8993(76)91001-5. [DOI] [PubMed] [Google Scholar]
- Fambrough D. M. Acetylcholine receptors. Revised estimates of extrajunctional receptor density in denervated rat diaphragm. J Gen Physiol. 1974 Oct;64(4):468–472. doi: 10.1085/jgp.64.4.468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galper J. B., Klein W., Catterall W. A. Muscarinic acetylcholine receptors in developing chick heart. J Biol Chem. 1977 Dec 10;252(23):8692–8699. [PubMed] [Google Scholar]
- Galper J. B., Smith T. W. Properties of muscarinic acetylcholine receptors in heart cell cultures. Proc Natl Acad Sci U S A. 1978 Dec;75(12):5831–5835. doi: 10.1073/pnas.75.12.5831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garber B. B., Moscona A. A. Reconstruction of brain tissue from cell suspensions. I. Aggregation patterns of cells dissociated from different regions of the developing brain. Dev Biol. 1972 Feb;27(2):217–234. doi: 10.1016/0012-1606(72)90099-1. [DOI] [PubMed] [Google Scholar]
- Gavin J. R., 3rd, Roth J., Neville D. M., Jr, de Meyts P., Buell D. N. Insulin-dependent regulation of insulin receptor concentrations: a direct demonstration in cell culture. Proc Natl Acad Sci U S A. 1974 Jan;71(1):84–88. doi: 10.1073/pnas.71.1.84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldfine I. D., Smith G. J., Wong K. Y., Jones A. L. Cellular uptake and nuclear binding of insulin in human cultured lymphocytes: evidence for potential intracellular sites of insulin action. Proc Natl Acad Sci U S A. 1977 Apr;74(4):1368–1372. doi: 10.1073/pnas.74.4.1368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartzell H. C., Fambrough D. M. Acetylcholine receptors. Distribution and extrajunctional density in rat diaphragm after denervation correlated with acetylcholine sensitivity. J Gen Physiol. 1972 Sep;60(3):248–262. doi: 10.1085/jgp.60.3.248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahn C. R. Membrane receptors for hormones and neurotransmitters. J Cell Biol. 1976 Aug;70(2 Pt 1):261–286. doi: 10.1083/jcb.70.2.261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kandel E. R., Spencer W. A. Cellular neurophysiological approaches in the study of learning. Physiol Rev. 1968 Jan;48(1):65–134. doi: 10.1152/physrev.1968.48.1.65. [DOI] [PubMed] [Google Scholar]
- Kebabian J. W., Zatz M., Romero J. A., Axelrod J. Rapid changes in rat pineal beta-adrenergic receptor: alterations in l-(3H)alprenolol binding and adenylate cyclase. Proc Natl Acad Sci U S A. 1975 Sep;72(9):3735–3739. doi: 10.1073/pnas.72.9.3735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LILEY A. W. The quantal components of the mammalian end-plate potential. J Physiol. 1956 Sep 27;133(3):571–587. doi: 10.1113/jphysiol.1956.sp005610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LIU C. N., CHAMBERS W. W. Intraspinal sprouting of dorsal root axons; development of new collaterals and preterminals following partial denervation of the spinal cord in the cat. AMA Arch Neurol Psychiatry. 1958 Jan;79(1):46–61. [PubMed] [Google Scholar]
- Lefkowitz R. J., Mullikin D., Williams L. T. A desensitized state of the beta adrenergic receptor not associated with high-affinity agonist occupancy. Mol Pharmacol. 1978 Mar;14(2):376–380. [PubMed] [Google Scholar]
- Martres M. P., Costentin J., Baudry M., Marcais H., Protais P., Schwartz J. C. Long-term changes in the sensitivity of pre-and postsynaptic dopamine receptors in mouse striatum evidenced by behavioural and biochemical studies. Brain Res. 1977 Nov 11;136(2):319–337. doi: 10.1016/0006-8993(77)90806-x. [DOI] [PubMed] [Google Scholar]
- Mickey J., Tate R., Lefkowitz R. J. Subsensitivity of adenylate cyclase and decreased beta-adrenergic receptor binding after chronic exposure to (minus)-isoproterenol in vitro. J Biol Chem. 1975 Jul 25;250(14):5727–5729. [PubMed] [Google Scholar]
- Miledi R., Potter L. T. Acetylcholine receptors in muscle fibres. Nature. 1971 Oct 29;233(5322):599–603. doi: 10.1038/233599a0. [DOI] [PubMed] [Google Scholar]
- Mizel S. B., Wilson L. Inhibition of the transport of several hexoses in mammalian cells by cytochalasin B. J Biol Chem. 1972 Jun 25;247(12):4102–4105. [PubMed] [Google Scholar]
- Mukherjee C., Lefkowitz R. J. Desensitization of beta-adrenergic receptors by beta-adrenergic agonists in a cell-free system: resensitization by guanosine 5'-(beta, gamma-imino)triphosphate and other purine nucleotides. Proc Natl Acad Sci U S A. 1976 May;73(5):1494–1498. doi: 10.1073/pnas.73.5.1494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nahorski S. R. Altered responsiveness of cerebral beta adrenoceptors assessed by adenosine cyclic 3',5'-monophosphate formation and (3H)propranolol binding. Mol Pharmacol. 1977 Jul;13(4):679–689. [PubMed] [Google Scholar]
- Nathanson N. M., Klein W. L., Nirenberg M. Regulation of adenylate cyclase activity mediated by muscarinic acetylcholine receptors. Proc Natl Acad Sci U S A. 1978 Apr;75(4):1788–1791. doi: 10.1073/pnas.75.4.1788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noble M. D., Brown T. H., Peacock J. H. Regulation of acetylcholine receptor levels by a cholinergic agonist in mouse muscle cell cultures. Proc Natl Acad Sci U S A. 1978 Jul;75(7):3488–3492. doi: 10.1073/pnas.75.7.3488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schimke R. T. Methods for analysis of enzyme synthesis and degradation in animal tissues. Methods Enzymol. 1975;40:241–266. doi: 10.1016/s0076-6879(75)40020-9. [DOI] [PubMed] [Google Scholar]
- Schlessinger J., Shechter Y., Willingham M. C., Pastan I. Direct visualization of binding, aggregation, and internalization of insulin and epidermal growth factor on living fibroblastic cells. Proc Natl Acad Sci U S A. 1978 Jun;75(6):2659–2663. doi: 10.1073/pnas.75.6.2659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seeds N. W., Vatter A. E. Synaptogenesis in reaggregating brain cell culture. Proc Natl Acad Sci U S A. 1971 Dec;68(12):3219–3222. doi: 10.1073/pnas.68.12.3219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma S. K., Klee W. A., Nirenberg M. Dual regulation of adenylate cyclase accounts for narcotic dependence and tolerance. Proc Natl Acad Sci U S A. 1975 Aug;72(8):3092–3096. doi: 10.1073/pnas.72.8.3092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snyder S. H., Banerjee S. P., Yamamura H. I., Greenberg D. Drugs, neurotransmitters, and schizophrenia. Science. 1974 Jun 21;184(4143):1243–1253. doi: 10.1126/science.184.4143.1243. [DOI] [PubMed] [Google Scholar]
- Snyder S. H., Bennett J. P., Jr Neurotransmitter receptors in the brain: biochemical identification. Annu Rev Physiol. 1976;38:153–175. doi: 10.1146/annurev.ph.38.030176.001101. [DOI] [PubMed] [Google Scholar]
- Stent G. S. A physiological mechanism for Hebb's postulate of learning. Proc Natl Acad Sci U S A. 1973 Apr;70(4):997–1001. doi: 10.1073/pnas.70.4.997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wurtman R. J., Fernstrom J. D. Control of brain neurotransmitter synthesis by precursor availability and nutritional state. Biochem Pharmacol. 1976 Aug 1;25(15):1691–1696. doi: 10.1016/0006-2952(76)90400-7. [DOI] [PubMed] [Google Scholar]
- Yamamura H. I., Snyder S. H. Muscarinic cholinergic binding in rat brain. Proc Natl Acad Sci U S A. 1974 May;71(5):1725–1729. doi: 10.1073/pnas.71.5.1725. [DOI] [PMC free article] [PubMed] [Google Scholar]


